Abstract
The aim of this study was to report on the lesions occurring in the central nervous system (CNS) during experimental classical swine fever (CSF) to clarify the spatial and chronologic distribution of the lesions and virus antigen in the CNS. To learn more about the pathogenetic mechanisms of the lesions during CSF in the CNS and to investigate the role of the virus in these mechanisms, cellular infiltrates and infected cells have been characterized. Twenty-eight pigs were inoculated with the virulent CSF virus isolate Alfort 187 and slaughtered from 2 to 15 postinoculation days; 4 animals of similar background served as a control group. Immunohistochemistry, electron microscopy, and the transferase-mediated deoxyuridine triphosphate nick-end labeling method were used to detect viral antigens and apoptosis. The results showed the presence of nonpurulent meningoencephalitis, occasional microhemorrhages, and apoptosis of the lymphocytes forming the perivascular and interstitital infiltrate in swine with CSF. Macrophages appeared to display little involvement in CNS lesions. The infected cells observed at the early stage of disease were lymphocytes and microglial cells in the rostral portion of the telencephalon, with infection of these cells in other areas in the next stages. The relationship between these lesions and the presence of viral antigen varied according to the type of lesion: hemorrhages were not associated with the presence of antigen in endothelial cells, but infiltrate-cell apoptosis was temporally and spacially associated to viral infection. However, the link between viral infection and the presence of cell infiltrate was far from clear.
Introduction
Classical swine fever (CSF) is caused by an RNA virus belonging to the Flaviviridae family (genus Pestivirus), whose members include bovine diarrhea virus (BDV) and border disease virus, to which CSF is antigenically related. 31 Pestivirus infections give rise to different lesions of the central nervous system depending, mainly, on the timing of infection: transplacental infection may cause demyelinization of the central nervous system (CNS), cerebral cavitation, cerebellar hypoplasia 1, 12, 21, 31 and slight inflammatory lesions when the immune system is first developing, 21, 30 whereas virus persistence is reported when the infection takes place at more advanced stages of gestation. 1 Although most of these diseases do not tend to prompt inflammatory lesions in animals infected after birth, CSF causes an inflammatory reaction leading to nervous signs, mainly paralysis that affects the lower third of the body with a tendency to be generalized. The animals remain reclined on a flank, continuously paddling, 7, 28 owing to a nonpurulent meningoencephalitis accompanied by swelling, proliferation, and degeneration of endothelial cells; moreover, vascular and perivascular cell infiltration next to the presence of perivascular hemorrhages have been described. 11, 20, 28
Even though CNS inflammation is one of the most striking histopathologic alterations, and is considered diagnostic of CSF, 28 very few studies address the nature and origin of the lesions involved; little is known of their chronology or topography, the nature of the cell infiltrate, or the exact relationship between viral presence and CNS lesions, which are observed even when no virus is detected in the CNS. 6 Viral infection has been described in neurons, glial cells, endothelial cells, and CNS cell infiltrate during CSF. 18, 28 A similar viral antigen distribution has recently been reported in a calf with BVD virus type II infection. 2
This paper reports on a study designed to chart the spatial and chronologic distribution of lesions and virus antigen in the CNS, and to characterize cellular infiltrate and infected cells, to learn more about the pathogenesis of CSF-induced lesions of the CNS and determine the exact role of the virus in these lesions.
Material and Methods
Animals, virus, and experimental design
Thirty-two Large White × Landrace pigs of either sex, 4 months old, and serologically negative for CSF, African swine fever, porcine reproductive respiratory syndrome, and Aujeszky's disease were used; live weight at the start of the study was approximately 30 kg. All animals were housed in the Centro de Investigación en Sanidad Animal in Valdeolmos, Madrid, Spain. Four pigs were used as uninfected controls, whereas the other 28 animals each received an intramuscular inoculation of 105 tissue culture infective doses (50%) of CSF virus isolate “Alfort 187,” classified as virulent. 32 Control pigs received only phosphate-buffered saline (PBS), pH 7.2. After virus inoculation, clinical signs and rectal temperature were monitored daily. This experiment was carried out in accordance with the Code of Practise for Housing and Care of Animals used in Scientific Procedures, approved by the European Economic Community Union in 1986 (86/609/EEC).
Processing of specimens for light and transmission electron microscopy
Inoculated pigs were sedated with azaperone (Stresnil, Jannsen Animal Health, Beerese Belgium) and anesthetized and killed with thiopental-sodium (Thiovet, Vet Limited, Leyland, Lancashire, England) in batches of four at 3, 4, 5, 7, 9, 11, and 15 postinoculation days (pid); The remaining four pigs (controls), were killed at the end of the experiment (15 pid). Samples of CNS (rostral and caudal telencephalon, cerebellum, and medulla oblongata) were fixed in 10% buffered formalin solution (pH 7.2) and Bouin's solution for structural and immunohistochemical study.
Samples were dehydrated through a graded series of alcohols, washed with xylol and embedded in paraffin wax by routine techniques for light microscopy. Wax-embedded sections (4 µm) were cut and stained with hematoxylin and eosin (HE). The intensity of histopathologic findings (Table 1) was expressed as: (−) absence, (+) occasional, (++) light, (+++) moderate, (++++) intense.
Histopathologic findings observed on central nervous system of uninoculated (UI) animals and pigs inoculated with CSF virus.∗
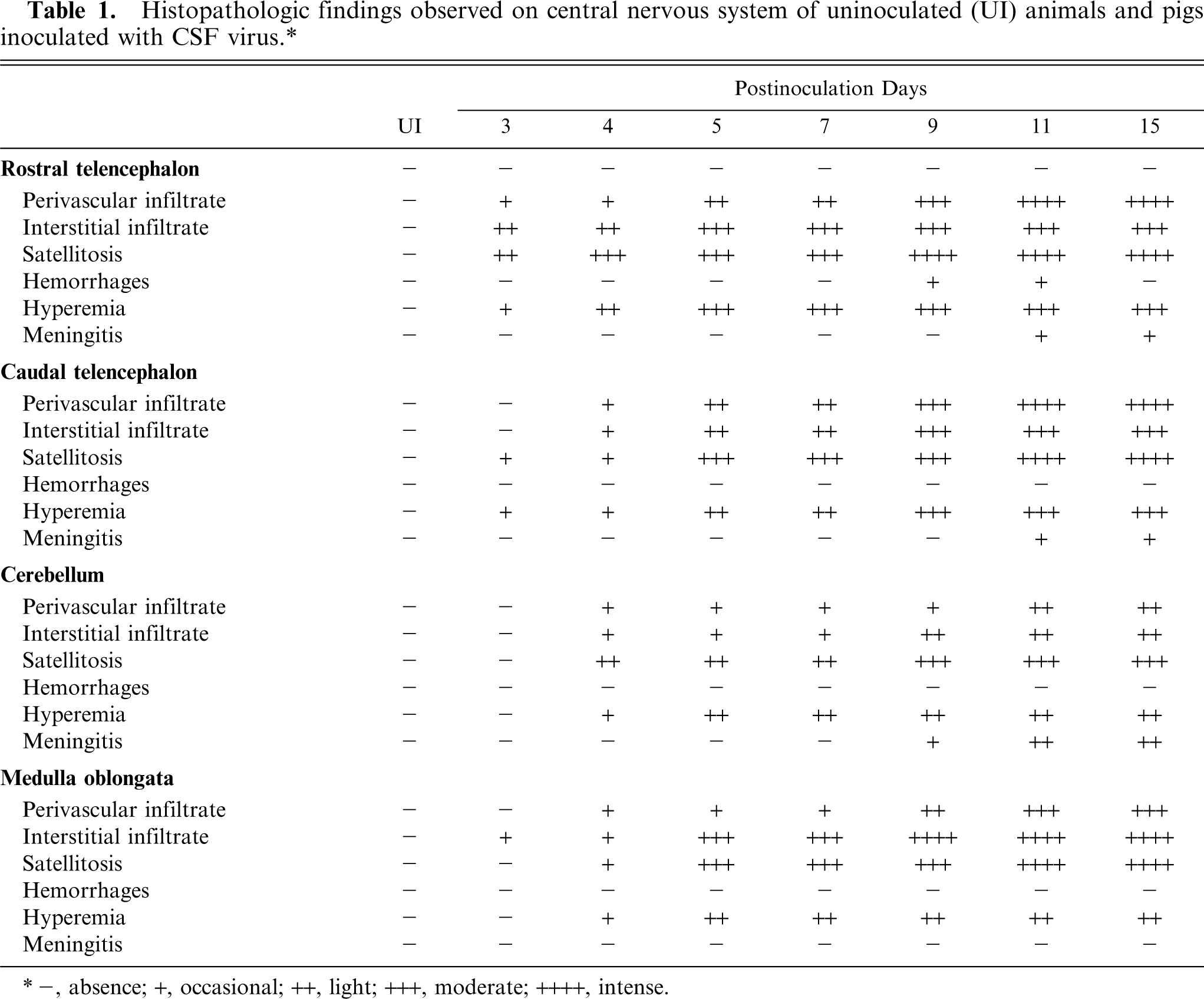
-, absence; +, occasional; ++, light; +++, moderate; ++++, intense.
For transmission electron microscopy, samples were postfixed in 2% osmium tetroxide, dehydrated in acetone, and embedded in Epon 812 (Fluka Chemie AG, Buchs, Switzerland). Epon-812 embedded sections (1 µm) were stained with toluidine blue (1% aqueous solution). Futher sections (50 nm) of CNS areas for ultrastructural examination were counterstained with uranyl acetate and lead citrate, and viewed through a Philips CM-10 transmission electron microscope.
Apoptosis detection and immunohistochemistry
For the detection of DNA fragmentation, the paraffin-wax sections, previously fixed in 10% buffered formalin solution (pH 7.2), were stained by the terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) method, with a kit designated “In Situ Cell Death Detection, POD” (Boehringer Mannheim, Indianapolis, IN, USA), according to manufacturer's instruction.
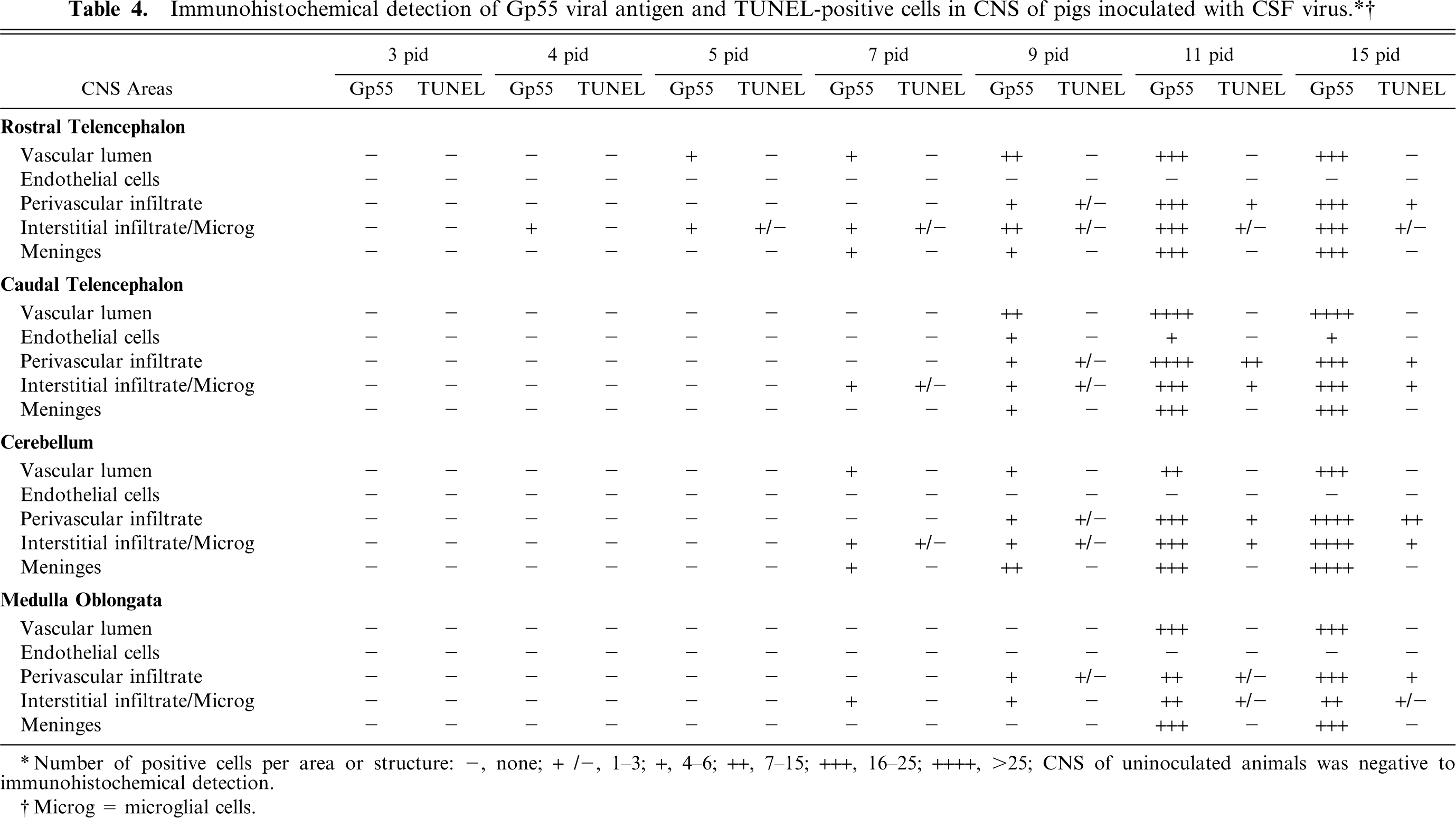
The avidin-biotin-peroxidase complex (ABC) method was employed in the immunohistochemical study. Formalin fixed tissues were used to stain lymphocytes and astrocytes using antibodies anti-CD3, antilight λ-chains, and antiglial fibrillary acidic protein. Bouin's fixed tissues were used to stain for gp55 and macrophages using antibodies WH303 and anti-SWC3, respectively. Details of the primary antibodies used in this study, including dilutions and pretreatments, are summarized in Table 2. PBS and nonimmune serum were used in place of specific primary antibodies as negative controls. Samples from the 4 uninoculated animals were also used as controls. Immunolabeled cells were counted in 25 consecutive areas of 0.20 mm2 or structures that were chosen randomly in 4 diagonally positioned squares. The results for CD3, light-λ-chain, and SWC3 antibodies were given semiquantitatively (Table 3) as number of positive cells per area or structure: (−), none; (+/−), 1–5; (+), 6–10; (++), 11–30; (+++), 31–50; (++++), >50 cells. For WH303 (Gp55) antibody and TUNEL the results were given (Table 4) as number of positive cells per area or structure: (−), none; (+/−), 1–3; (+), 4–6; (++), 7–15; (+++), 16–25; (++++), >25 cells.
Details of the immunolabeling reagents.

1 Veterinary Laboratories Agency, Addlestone, UK; 2BioVet-UCO, Córdoba, Spain; 3Dako, Glostrup, Denmark.
In PBS, pH 7.2, containing normal goat serum 10%. The avidin-biotin complex (ABC) method (Vector Laboratories, Burlingame, CA, USA) was used with 3′3 diaminobenzidine tetrahydrochloride (Sigma-Aldrich Chemie, Steinheim, Germany) as chromogen.
TC = Tri-sodium citrate dihydrate (Merck, Darmstadt, Germany), 0.01 molar (pH 3.2), microwave 10 min.
Protease type XIV (Sigma-Aldrich Chemie, Steinheim, Germany) 0.1%, 10 min.
Immunohistochemical detection of T cells, B cells and macrophages (m⊘) in CNS of pigs infected with CSF virus.∗
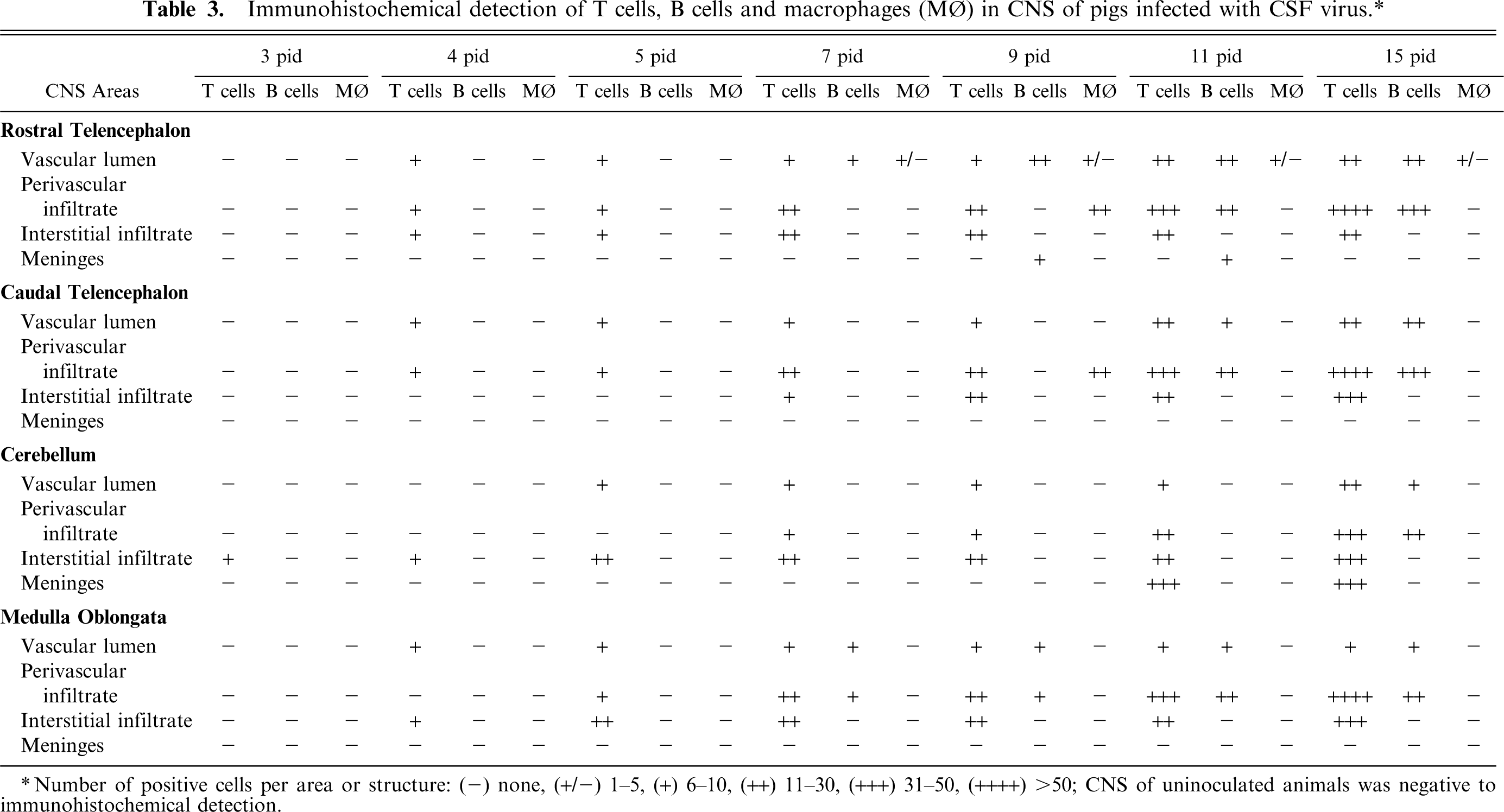
Number of positive cells per area or structure: (-) none, (+/-) 1-5, (+) 6-10, (++) 11-30, (+++) 31-50, (++++) >50; CNS of uninoculated animals was negative to immunohistochemical detection.
Number of positive cells per area or structure: -, none; + /-, 1-3; +, 4-6; ++, 7-15; +++, 16-25; ++++, >25; CNS of uninoculated animals was negative to immunohistochemical detection.
Microg = microglial cells.
To determinate microglial cell distribution, the tomato-lectin technique was used. After dewaxing of tissue sections in xylol, endogenous peroxidase activity was quenched by incubation with 3% hydrogen peroxide in methanol for 30 minutes at room temperature. Sections were rinsed in Tris-buffered saline (TBS) containing 0.05% Triton X-100 (Merck, Darmstadt, Germany). Sections were incubated overnight at 4°C with 20 g/ml biotinylated lectins obtained from Lycopersicon sculentum (Sigma-Aldrich Chemie, Steinheim, Germany). After rinsing in TBS, sections were incubated for 1 hour a room temperature with Vectastain ABC Elite (Vector Laboratories, Burlingame, CA, USA) following the steps described for single immunohistochemistry.
Results
Clinical results
Control animals remained healthy. The clinical signs observed in infected animals were characteristic of CSF. From 2 pid, inoculated animals showed nonspecific symptoms with persistent pyrexia (40.5–41.5°C), varying degrees of anorexia, and constipation. Ocular discharges associated with conjunctivitis were present from 4 to 5 pid. From 6 pid onwards, deterioration was rapid; pigs developed body temperatures of >42°C and showed markedly decreased activity. From 9 pid onwards, all inoculated animals presented semiliquid yellowish-grey diarrhea and tenesmus when rectal temperatures were taken. From 12 pid onwards, the animals displayed nervous signs characterized by incoordination, staggering walk, and paralysis of the posterior third of the body.
Structural and ultrastructural studies
Results are summarized in Table 1. The earliest histopathologic alterations were detected in the rostral portion of the telencephalon at 3 pid: occasional hyperemia was accompanied by a mild (1-cell thick) perivascular and interstitial cell infiltrate comprised of small mononuclear cells identified, by their nucleus/cytoplasm ratio and ultrastructural characteristics, as lymphocytes. The presence of cells with abundant cytoplasm and vesicular nuclei morphologically identified as macrophages was sparse. Histologic and subcellular changes suggesting secretory activation of macrophages—increase in cell size and lysosome numbers, proliferation and dilations of rough endoplasmic reticulum cisternae, and Golgi complexes—were not observed at any stage of the experiment. From 5 pid onwards, cell infiltrate was observed in all areas of the CNS studied, the most striking finding at this stage being a diffuse subependymal infiltrate. By 9 pid, lesions had become more intense: there was a marked increase in the thickness of interstitial and perivascular cell infiltrate (5–7 cells) (Fig. 1), and nonpurulent meningitis was detected at all sites studied; microhemorrhages were apparent in the rostral portion of the telencephalon. At ultrastructural examination, the presence of microhemorrhages was confirmed, though there was no sign of any morphologic changes in endothelial cells that might account for them. There was, however, some evidence of diapedesis of lymphocytes and occasional erythrocytes.
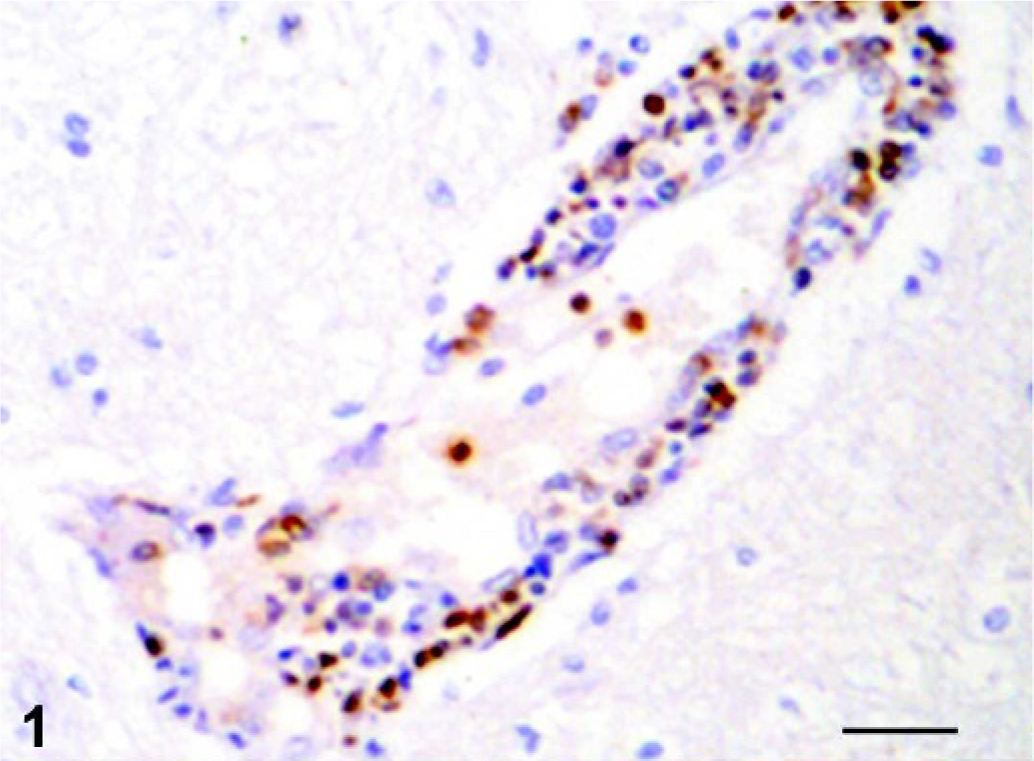
Rostral telencephalon, pig, pid 9. Perivascular mononuclear infiltrate showing immunopositive reaction against gp55 antigen of CSF virus. ABC technique, hematoxylin counterstain. Bar = 100 µm.
Satellitosis was observered from 3 pid onwards in the different studied areas. Neurons displayed no significant morphologic alterations in the course of the experiment.
Immunohistochemical characterization of cell infiltrate
Cell infiltrate identification was confirmed by immunohistochemical examination (Table 3). From their initial appearance onwards, most inflammatory cells displayed a widespread reaction to CD3 (Fig. 2), and were thus identified as T lymphocytes; only a few, scattered cells were positive to SWC3 (macrophages), or to λ-chain (B lymphocytes) (Fig. 3). Occasionally, macrophages were detected in Virchow–Robbins spaces without significant differences between uninoculated and inoculated animals, and between different postinoculation time point. The number of B lymphocytes, observed mainly in interstitial locations, increased slightly towards the end of the experiment.
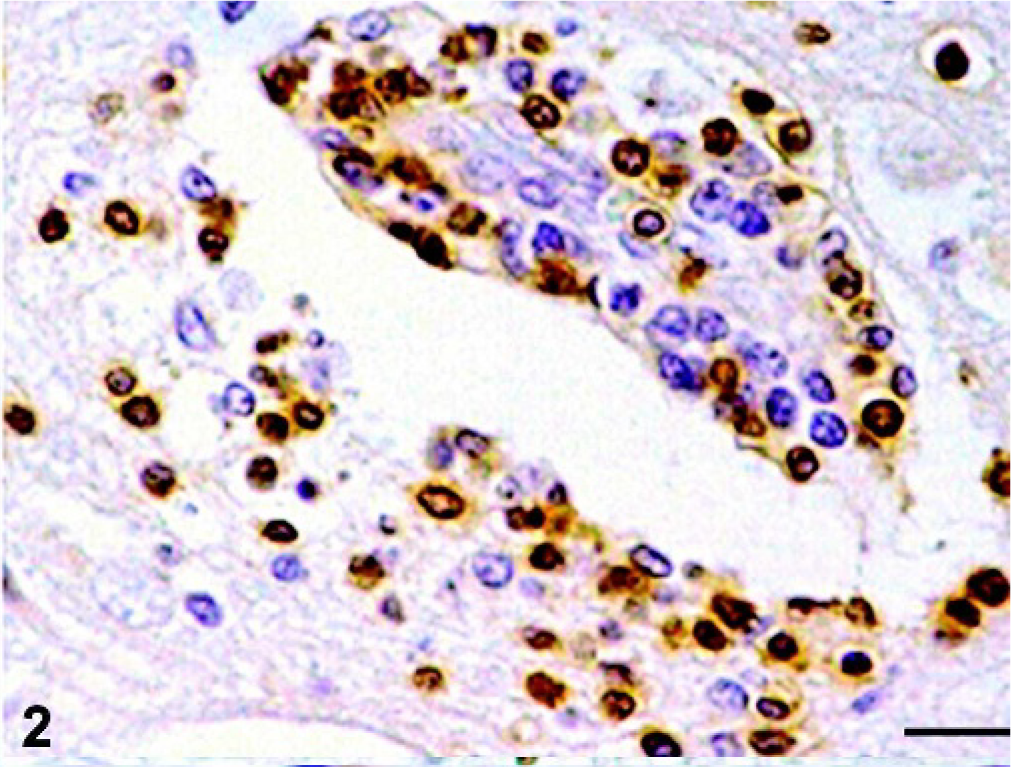
Rostral telencephalon, pig, pid 9. Immunohistochemical detection of CD3 antigen showing immunopositive reaction in lymphocytes of cuff and interstitial cells. ABC technique, hematoxylin counterstain. Bar = 30 µm.
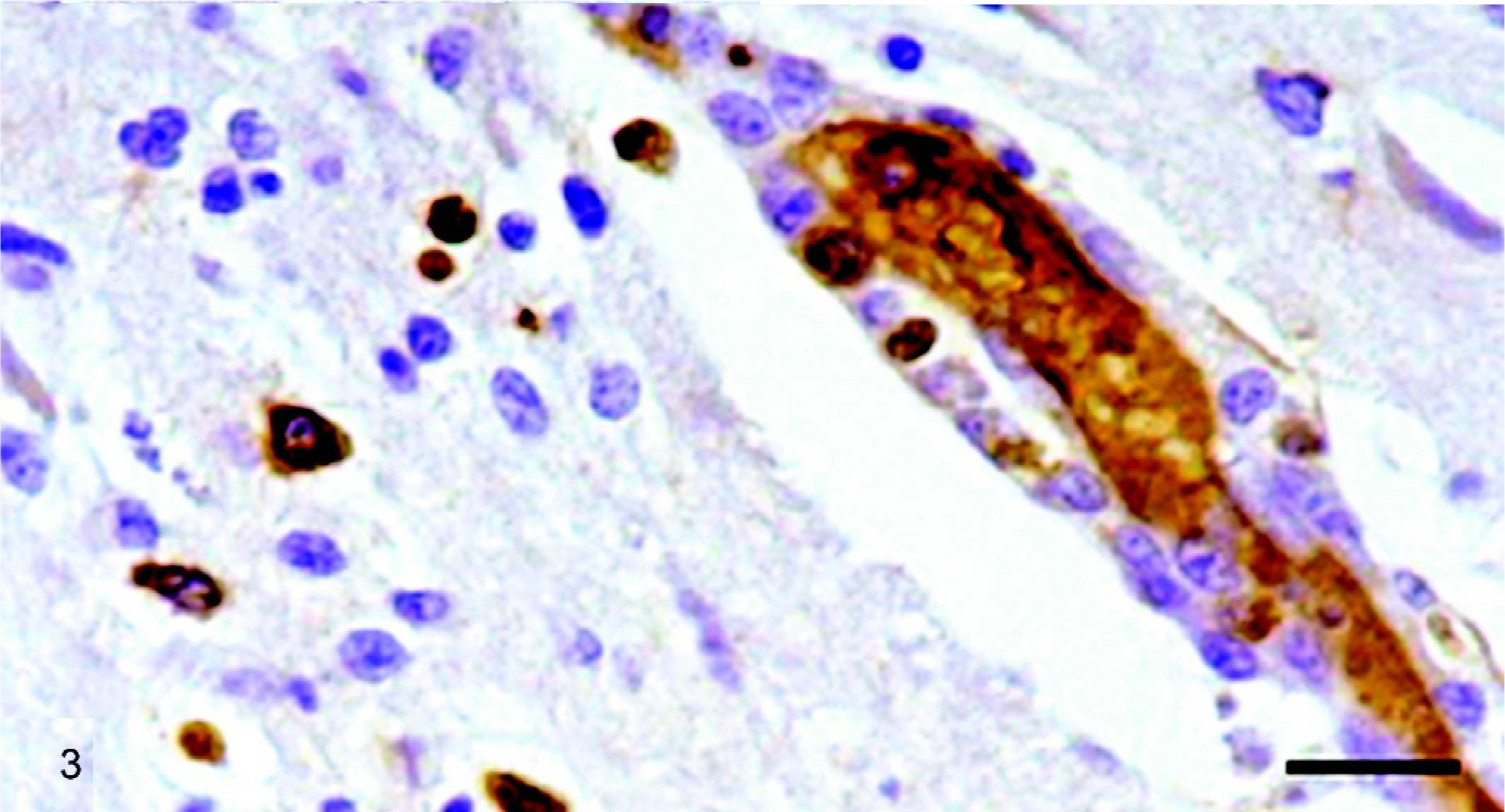
Rostral telencephalon, pig, pid 5. Immunohistochemical detection of light λ chain showing scattered immunopositive reaction in lymphocytes of cuff and interstitial cells. ABC technique, hematoxylin counterstain. Bar = 30 µm.
Tomato-lectin staining disclosed no significant changes in the presence and distribution of microglial cells throughout the experiment, other than their involvement in the satellitosis described earlier.
Apoptosis
From 5 pid, some cell infiltrates displayed greater evidence of pyknosis, karyorrhexis, and nuclear fragmentation, changes associated with cell death by apoptosis. Thereafter, apoptotic changes became increasingly frequent and were observed in numerous cell infiltrates widespread in the different CNS areas studied in our work. TUNEL-positive cells (Table 4) detected from 5 pid mostly involved perivascular infiltrate (Fig. 4) and, to a lesser extent, interstitial infiltrate.
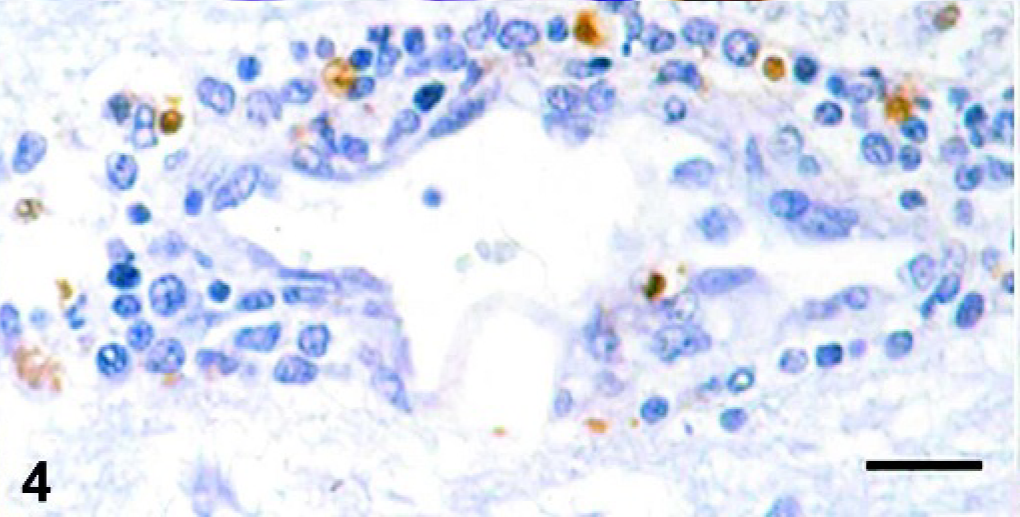
Rostral telencephalon, pig, pid 5. Apoptotic cells in cuff. TUNEL technique. Bar = 30 µm.
Ultrastructural examination exhibited changes consistent with lymphocyte apoptosis observed histologically in interstitial and perivascular infiltrate and confirmed using the TUNEL technique. Apoptotic cells exhibited condensation and margination of chromatin, and fragmentation of lymphocyte nuclei and cytoplasm. Few highly electron-dense membrane-bound cell fragments (apoptotic bodies) were observed.
Astrocytes
Immunohistochemical staining with glial fibrillary acidic protein antibody revealed no significant changes in astrocyte distribution and frequency. However, a slight alteration of the blood–brain barrier was apparent in vessels containing abundant perivascular infiltrate, with a loss of contact between astrocyte cytoplasmic processes and vessel walls. No subcellular alterations were detected.
Viral infection
Results are summarized in Table 4. Occasionally, mononuclear cells of interstitial infiltrate and some microglial cells located in the rostral portion of the telencephalon were immunostained against gp55 at 4 and 5 pid. From 7 pid, virus antigen was detected in interstitial infiltrate and microglial cells in all CNS areas studied, whereas the presence of infected cells in perivascular infiltrate was observed from 9 pid onwards (Fig. 1). From this time point, virus antigen was also detected in a few capillary endothelial cells in the caudal portion of the telencephalon (Fig. 5).
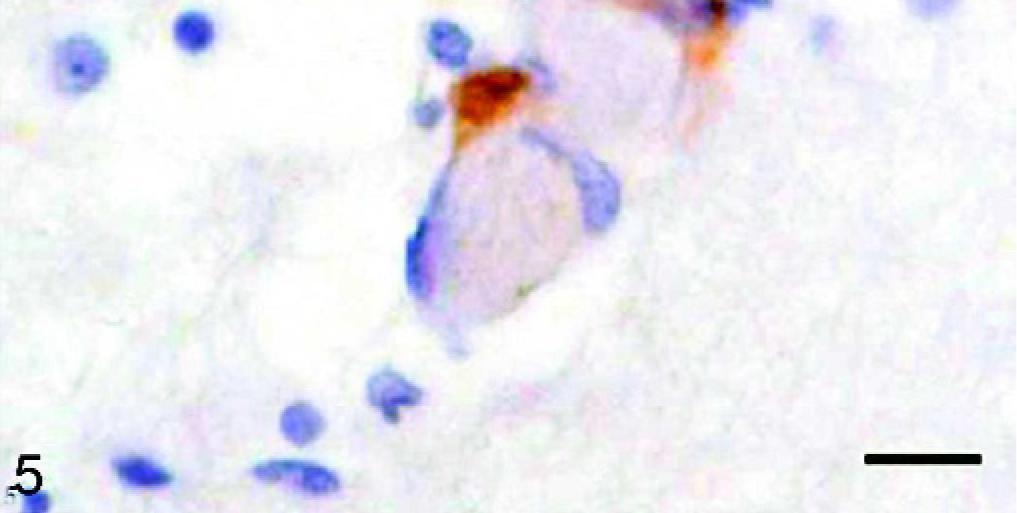
Caudal telencephalon, pig, pid 9. Immunohistochemical detection of gp55 antigen of CSF virus showing immunopositive reaction in endothelial cells of capillaries. ABC technique, hematoxylin counterstain. Bar = 20 µm.
From pid 11 onwards, a significant increase in the presence of cells immunolabeling against CSF virus antigen was observed in all CNS areas studied. Virus antigen was observed mainly in the cellular infiltrate of interstitium, perivascular infiltrate, and meninges. Viral antigen was not detected in neurons or astrocytes in the course of the experiment.
Viral infection of lymphocytes was confirmed by ultrastructural techniques. The major subcellular structures related to infection were cytoplasmic inclusions surrounded by unit membrane, containing fine granular material, and other moderately electron-dense inclusions, which in some areas were condensed and displayed greater electron density. All of these contained spherical particles of 50–55 nm diameter, with an electron-opaque or moderately electron-dense nucleoid, characteristic of CSF virus.
Discussion
This study confirmed the presence of nonpurulent meningoencephalitis in pigs with CSF, and additionally observed both occasional microhemorrhages and apoptosis of the cells forming the perivascular and interstitital infiltrate, a lesion not hitherto reported for this disease. The relationship between these lesions and the presence of viral antigen varied according to the type of lesion. Hemorrhages were not associated with the presence of antigen in endothelial cells, whereas infiltrate-cell apoptosis is associated with the presence of CSF viral antigen. However, the link between viral infection and the presence of cell infiltrate was unclear. Moreover, macrophages—which play a major role in other organs during CSF infection—appeared to display little involvement in CNS lesions.
Studies of CSF have not focused on CNS cells harboring viral antigen and their changing distribution over time. Fluorescence techniques have disclosed viral antigen in endothelial cells, perivascular astrocytes, and a small number of neurons from 4 pid, 28 and viral infection in both glial cells and perivascular infiltrate. 18 The immunoenzyme techniques used here revealed the presence of antigen at this early stage, but only in cell infiltrate and microglial cells in the rostral portion of the telencephalon; thenceforth, wider regional distribution was accompanied by occasional infection of endothelial cells, suggesting a nonuniform distribution of CSF virus through the CNS. Because the virus is distributed in the bloodstream, this time-lag in the detection of viral antigen in CNS studied areas could be attributed to different cellular behavior according with the different microenvironment conditions of CNS areas. These differences might be correlated with the release of chemical mediators that may either favor or inhibit viral invasion of the CNS. 3, 16, 17
Infection of neurons and astrocytes is reported in natural outbreaks of CSF, 18, 28 but was not found either here or in other studies by the authors using the “Quillota” isolate (unpublished data). This may be the result of differences between natural and experimental infection, or possibly to the more prolonged course of the natural infection studied; however, it should also be borne in mind that the immunoenzyme technique used here allows better structure maintenance and morphologic identification than immunofluorescence techniques. In any event, given that nervous-system signs characteristic of the disease were also observed here, possible infection of neurons and astrocytes is unlikely to play a major role in the pathogenesis of CSF. Although neuron infection has been reported in transplacental Pestivirus infections, 13, 29, 31 its role is apparently limited to inducing a weak, focal immune reaction resulting from persistence of the virus in the CNS. In a recently reported case of CNS infection in a cow with meningoencephalitis caused by BDV type II, the authors were unable to determine whether infection had been transplacental or horizontal. 2
Macrophages are the main CSF virus target cell, and play a key role in the pathogenesis of CSF through the synthesis and release of chemical mediators. 8– 10, 23– 25 However, its involvement in CNS lesions would appear to be less significant; few macrophages were found in cell infiltrate, and no major change was observed in microglial cells, which represent the mononuclear phagocyte system in the CNS. It was not possible to account for the perivascular lymphocyte infiltrate observed; its onset did not coincide with the presence of viral antigen, which was not detected until later. Perhaps, inflammatory reaction could be associated with an undetected viral infection of the CNS. The increase in the amount of infiltrate coincided with a rise in the amount of viral antigen in the CNS locations studied, although this may have been due more to the greater presence of cells susceptible to viral infection (lymphocytes) than to any attraction of these cells for the viral antigen present in the CNS. Moreover, the increase in the number of infected lymphocytes in the CNS coincided with a massive invasion of lymphocytes from other organs; 8– 10, 22– 25 many of the cells forming the CNS infiltrate may therefore have been infected on arrival, caused by positive chemotaxis induced by the synthesis and release of chemical mediators in situ or through paracrine action. 16, 17 This would suggest that CSF virus does not have a particular tropism for the CNS; infection is made possible by the virus-induced immune response.
In this study, apoptosis mostly involved perivascular infiltrate and, to a lesser extent, interstitial infiltrate cells from 5 pid onwards; thereafter, pyknosis and karyorrhexis became increasingly frequent. This lesion has not hitherto been reported in studies of the CNS in pigs with CSF, although it has been observed in various lymphoid organs and in peripheral blood, where apoptosis is induced by indirect mechanisms 26, 27 linked to secretory activation of macrophages; 8– 10, 23– 25 subcellular changes characteristic of secretory activation of macrophages were not found in the present study and the release of chemical mediators by these cells in CNS were not detected (unpublished data). However, the rate of infection of infiltrating cells coincided in time, though not in intensity, with apoptosis of these cells, suggesting that infection of infiltrating lymphocytes may be the chief apoptosis-inducing mechanism. This hypothesis is supported by the presence of an apoptosis-inducing Erns glycoprotein in CSF virus, 4 and by observed apoptosis of infected cells. 9 The difference in intensity between the apoptosis detected in the CNS and that observed at the same stage in other organs, 23– 25 where the presence and activation of macrophages has been demonstrated, would suggest that macrophages are mainly responsible for inducing apoptosis in CSF through the release of proinflammatory cytokines. 8– 10, 24, 25 However, the CNS contains a number of mechanisms for inducing apoptosis of cell infiltrate, as a tool for controlling the inflammatory response. 19 Further research is required into the role of the these mechanisms in the pathogenesis of CNS lesions in CSF.
The vascular alterations detected here were very mild, consisting of moderate hyperemia from 3 pid, 28 and occasional microhemorrhages in the rostral portion of the telencephalon from 9 pid. 11 These hemorrhages were not attributed to direct action of the virus on the endothelium, and subcellular endothelial damage was not observed; however, hemorrhages might be related to erythrocyte extravasation possibly prompted by increased vascular permeability. 8 Although the mechanisms responsible for altered permeability require further research, the presence of lymphocyte infiltrate, and the absence of significant changes in macrophages, in CNS and kidney, coupled with the early presence of immune complexes in the kidney, 22 suggest that altered permeability—and possibly the cell infiltrate itself—are immunopathologic in origin, as reported for some human flavivirus infections. 5, 14, 15 Changes observed in hematoencephalic barrier could contribute to increase the vascular permeability. To confirm this hypothesis, it would be necessary to study the cytokine serologic profile and cytokine expression in the CNS of CSF-infected pigs.
Footnotes
Acknowledgements
This work was supported by grants from Ministry of Education and Science (PB98-1033; AGL2003-00252) and Plan Andaluz de Investigación (AGR-0137). We appreciate the technical assistance of “Servicio Central de Apoyo a la Investigación,” University of Córdoba. The authors thank to Ms. M.C. Camacho for assistance in the English translation of the manuscript.