Abstract
To evaluate the diagnostic potential of meat juice for early detection of
Classical swine fever (CSF) is a serious, highly contagious disease in swine. The causative agent is
Skeletal muscle tissue can be easily collected for laboratory examination from abattoir carcasses, wild boars killed in road accidents, and imported meat products. From this material, meat juice can be extracted for further diagnostic analysis. Previous studies have performed pathogen and/or antibody analyses of meat juice for laboratory detection of several infections including salmonellosis, Aujeszky disease, porcine reproductive and respiratory syndrome, trichinellosis,1,6,8,9 and CSF.5,14 However, the existence of numerous CSFV strains of different virulence make the diagnostic approach for CSFV infection less straightforward, as virus distribution may differ with virulence. 2 This fact could influence the suitability of meat juice as a potential diagnostic sample material.
In the present study, the diagnostic potential of meat juice for detection of CSFV RNA was tested by examination of skeletal muscle tissue samples from pigs experimentally infected with 7 CSFV strains of different genotypes and virulence. The performance of meat juice was compared with contemporary analyses of serum collected from the same animal at the same time points. All material originated from CSFV infection experiments in pigs performed at the National Veterinary Institute (Lindholm, Kalvehave, Denmark) in accordance with the requirements of the Danish Animal Experiments Inspectorate.
For evaluation, a real-time reverse transcription polymerase chain reaction (RT-PCR) assay method in an automated RNA extraction system was used on sampled material from a total of 85 pigs. All samples were collected from pigs at time of euthanasia, which varied from day post-inoculation (dpi) 5–28. For pigs infected with CSFV-Bergen, CSFV-Glentorf, CSFV-Romania, CSFV-Israel, and mock-inoculated control pigs, the experimental design included sequential killings in groups of 3–4 on dpi 5, 10, and 21/22, respectively, except for individual pigs, which had to be euthanized at an earlier time point for animal welfare reasons. After collection, the sampled materials were prepared for further examination as follows. Serum was obtained by centrifugation of blood collected in plain tubes with no anticoagulant at 237 g for 10 min and then stored at −40°C until use. Meat juice was obtained from samples of the quadriceps muscle. Thus, muscle tissue was cut into pieces of approximately 2 cm × 2 cm × 2 cm, put into plastic bags, frozen at −20°C, and followed by gentle thawing at 5°C. Through this process, the released meat juice was collected without further preparation into cryotubes and stored at −40°C until use. For each analysis, 100 µl of sample material (serum or meat juice) was used.
The RNA was extracted with a commercial nucleic acid isolation kit, a according to the manufacturer’s instructions using an automated robot. a After RNA extraction, the material was stored at −80°C in the sample cartridge until PCR was carried out. Real time RT-PCR was performed as previously described 12 using the primers CSFV6 and CSFV7 together with a CSFV-specific TaqMan probe (5′-FAM-CCCTGG GTGGTCTAAGTCCTGAGTACAG-TAMRA-3′). The reactions were cycled, b and, to the fluorescence data obtained, a threshold cycle (Ct) value was assigned to each sample as a measure for the virus quantity. The relative quantity of viral RNA (the viral load) in each sample was expressed directly as Ct values without recalculating the genome equivalents.
Data obtained from the serum and meat juice analyses of pigs inoculated with the different CSFV strains were compared. Based on clinical records from the performed experiments, data sets from individual strains were designated into low, moderate, or high virulence (Table 1). To study the possible association between serum and meat juice, a Spearman rank correlation was calculated on the total data set of inoculated animals, and the correlation coefficient (rs) was determined.
Diagnostic value of meat juice in early detection of classical swine fever infection: virulence and genotype information of the 7
dpi = day post-infection; NR = not relevant. Strain ID and genotype designation is obtained from the CSFV database from The European Central Reference Laboratory. RT-PCR-positive and -negative results indicate threshold cycle <40 and ≥40, respectively.
The Ct values of serum and meat juice from individual pigs were depicted with relation to infection day in the experiment and illustrated in Figure 1A–C. The results indicate that CSFV RNA is detected in meat juice, yet in a lower quantity than in serum. Furthermore, throughout the entire experimental period, negative samples of meat juice as well as serum will occur in a group of pigs infected with a CSFV strain of low virulence (Fig. 1A).
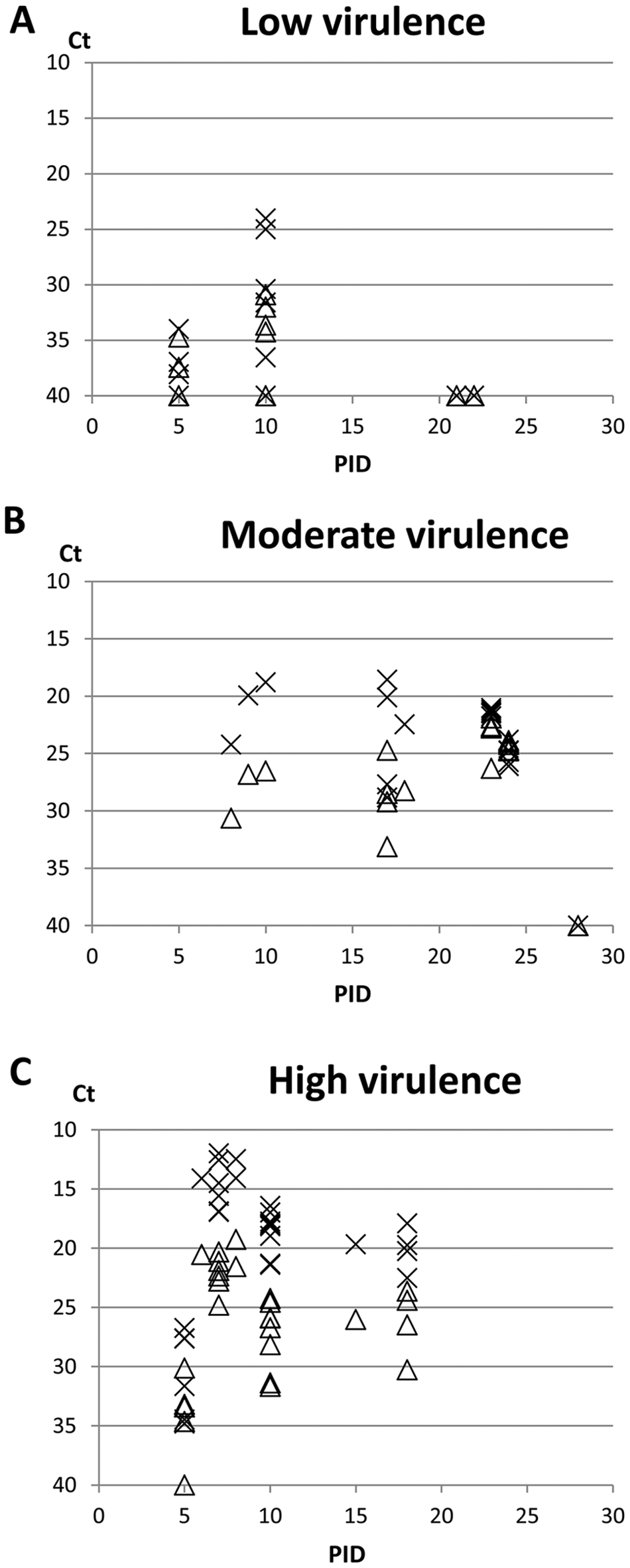
Threshold cycle (Ct) values obtained from examined sample material after real-time reverse transcription polymerase chain reaction procedure. Depicted Ct values are shown for individual pigs and related to day post-infection (PID). Results from the individual groups of pigs inoculated with different
All sample results were assigned to either positive (Ct value < 40) or negative (Ct value ≥ 40) value (Table 1), and by comparison, meat juice sensitivity was calculated to 91% and specificity to 97% of serum. The coefficient of concordance for the 2 tested materials was 93%, indicating that a strong correlation exists. Six pairs of samples were in disagreement; in 5 cases, serum tested positive for CSFV RNA while negative in meat juice, and in 1 case, the relationship was reversed. Four of the disagreements were related to strains in the category of low virulence: 1 sample from a CSFV-Glentorf–infected pig (dpi 5) and 3 samples from CSFV-Bergen–infected pigs (dpi 5/10). Two disagreements were related to the category of high virulence; both samples were collected from CSFV-Romania–infected pigs (dpi 5). All 13 mock-infected control pigs tested negative in serum as well as in meat juice. The calculated correlation coefficient (rs) was = 0.89, with a 95% confidence interval of (0.82; 0.93) and a
In order to address situations where blood and/or target organ material for CSFV diagnosis is not available, the potential of meat juice as diagnostic substrate was evaluated. Previous studies on meat juice and muscular tissue reflect different results and diverging opinions regarding the diagnostic value of this material.5,13,14 Based on the present study with CSFV strains of different virulence, the levels of CSFV RNA in meat juice and corresponding serum samples were compared. By comparison of data from all inoculated pigs, an overall sensitivity of 91% for detection of CSFV in meat juice was calculated. The results obtained in the current study support previous work, 5 which concluded that meat juice is most suitable as substrate for diagnosis of CSFV from pigs infected with strains of high or moderate virulence. In low virulence CSFV infections, virus detection in meat juice as well as in other nonlymphatic organs can be problematic due to low virus load and sparse tissue distribution in the infected pig. Therefore, for low virulent strains, demonstration of serum antibodies several weeks after infection may be of diagnostic value. Furthermore, the results suggest that time point after infection has an important effect in sample collection, as some virus strains, even when classified as highly virulent, will not be disseminated to non–virus-targeted organs within the body of the pig until the end of the first week after infection. Such a problem was encountered in pigs inoculated with CSFV-Romania, where demonstration of viral RNA in meat juice only was possible in 1 out of 3 pigs at dpi 5. Corresponding serum samples were all positive for viral RNA, indicating ongoing viremia. The finding does not correspond with conclusions from a Swiss study, 13 which could not demonstrate any influence of time point of infection for sample collection for detection of CSFV RNA. However, the results from the Swiss and the present study are not directly comparable as the presented data are based on homogenized muscle tissue and meat juice, respectively. In a previous Danish study, 14 a comparison of virus detection in meat juice and corresponding homogenized quadriceps muscle tissue revealed lower Ct values in meat juice than in corresponding muscle tissue samples, indicating that a higher RT-PCR sensitivity for the former sample material exists. The finding is consistent with the Swiss study 13 that could not recommend muscular tissue as diagnostic material for CSFV, regardless of strain virulence, and found that only 58% of the tested samples were CSFV positive by RT-PCR compared with parallel examination of target organs. It is speculated whether the lower sensitivity of muscle tissue samples compared with meat juice might be due to PCR inhibiting factors released by tissue homogenization or dilution of virus content in the muscle tissue preparation process.
In conclusion, the results from the present study indicate that the use of meat juice as diagnostic material for CSFV infection is justified under certain conditions (e.g., when used as material for herd-based screening). However, meat juice cannot be recommended as an isolated diagnostic material for early detection of CSFV infection in individual pigs when blood and/or target organ material is available, as the diagnostic sensitivity for meat juice is lower than that for serum. Potential use of meat juice as a reliable diagnostic sample will depend on the actual strain virulence and on the relative time of sample collection in the specific disease situation.
Footnotes
Acknowledgements
The authors thank MajBritt Eicke for excellent technical assistance.
a.
MagNA Pure LC Total Nucleic Acid Isolation kit, Roche A/S, Hvidovre, Denmark.
b.
MX4000, Stratagene, AH-diagnostics, Aarhus, Denmark.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The current study was financially supported by Directorate for Food, Fisheries and Agri Business in Denmark, grant no. 2007- 776.
