Abstract
This investigation assessed the susceptibility of experimentally infected pigeons to the highly pathogenic avian influenza virus (HPAIV) H5N1 that caused recent outbreaks of avian influenza in birds and humans in several countries of Asia. For this purpose 14 pigeons were infected ocularly and nasally with 108 EID50 and clinical signs were recorded and compared with five chickens infected simultaneously as positive controls. The chickens demonstrated anorexia, depression, and 100% mortality within 2 days postinoculation. Three of the pigeons died after a history of depression and severe neurological signs consisting of paresis to paralysis, mild enteric hemorrhage, resulting in a mortality of 21%. Gross lesions in these pigeons were mild and inconsistent. Occasionally subcutaneous hyperemia and hemorrhage and cerebral malacia were observed. Microscopic lesions and detection of viral antigen were confined to the central nervous system of these pigeons. In the cerebrum and to a minor extent in the brain stem a lymphohistiocytic meningoencephalitis with disseminated neuronal and glial cell necrosis, perivascular cuffing, glial nodules, and in one bird focally extensive liquefactive necrosis could be observed. The remaining nine pigeons showed neither clinical signs nor gross or histological lesions associated with avian influenza, although seroconversion against H5 indicated that they had been infected. These results confirm that pigeons are susceptible to HPAIV A/chicken/Indonesia/2003 (H5N1) and that the disease is associated with the neurotropism of this virus. Although sentinel chickens and most pigeons did not develop disease, further experiments have to elucidate whether or not Columbiformes are involved in transmission and spread of highly pathogenic avian influenza.
Introduction
In the past, a large number of avian influenza viruses (AIV), belonging to 16 hemagglutinin (H1-H16) and nine neuraminidase (N1-N9) subtypes, have been recovered from a variety of domestic and wild avian species worldwide. All subtypes have been found in wild birds from at least nine different orders, but the main reservoir hosts seem to be birds of the orders Anseriformes (geese, ducks, swans) or Chadriiformes (gulls, terns, surfbirds, sandpipers). 25 The wide distribution in these asymptomatically infected wild birds may result in spread to domestic poultry. 2 Highly pathogenic avian influenza (HPAI) is an acute, generalized, fatal disease etiologically restricted only to those AIV strains of the subtypes H5 and H7, which code for a furin-sensitive cleavage site in their hemagglutinin protein and, therefore, are capable of inducing systemic infections. HPAI occurs characteristically in chickens and turkeys. 1 However, other gallinaceous birds such as quails or guinea fowls, as well as ducks, geese, ratites, passerine birds, flamingos, herons, and raptors are also susceptible for the disease. 18, 19 Since 2003, an epidemic emergence of H5N1 in Vietnam, Indonesia, and Thailand has resulted in the culling of millions of birds and led to 173 confirmed cases of human H5N1-influenza, with a total of 93 fatalities reported to the World Health Organization (WHO) until 27 February 2006 (http://www.who.int/csr/disease/avian_influenza). Controversial reports exist about the susceptibility of domestic birds, especially racing pigeons and wild pigeons, and their role as a link between wild and domestic birds and transmission and spread of HPAIV during epizootics over long distances. 10 Whereas several transmission experiments suggest a resistance, one report describes a minimal susceptibility of Columbiformes to infection with HPAIV of the H5 subtype. 16, 18, 20 Here, we focus on the clinical and pathological findings and distribution of viral antigen after experimental inoculation of pigeons with the HPAIV A/chicken/Indonesia/2003 (H5N1).
Materials and Methods
Viruses
The HPAIV A/chicken/Indonesia/2003 (H5N1) was isolated in the national reference laboratory for avian influenza at the Friedrich-Loeffler-Institut, Isle of Riems, Germany, from chicken organs received from Indonesia. The isolate was once propagated in the allantoic cavities of 10-day-old embryonated specific pathogen-free (SPF) chicken eggs. The allantoic fluid was harvested and stored at −70°C. 25
Animals and experimental design
Fourteen 4-month-old, male and female racing pigeons (Columbia livia f. domestica), serologically negative for H5-specific hemagglutination-inhibiting antibodies, were inoculated ocularly and intranasally in each case with 0.5 ml allantoic fluid containing 1 × 108 EID50 (50% embryo infective dose) of the Indonesia/2003 HPAIV. For detection of virus excretion five 12-week-old serologically AIV-negative SPF White Leghorn chickens (VALO SPF, Lohmann, Cuxhaven, Germany) were added to the aviary with the pigeons 48 hours postinoculation (pi). Four pigeons were not inoculated with HPAIV and served as a negative control group. Furthermore five SPF chickens of the same age and breed were simultaneously and identically infected as positive controls and kept separately. Pigeons and chickens were housed in aviaries with free access to food and water. The animals were observed for 19 days pi (dpi), and clinical signs were recorded. All animals used for these investigations were kept under isolated conditions in the biosafety level 3 containment facilities on Isle of Riems. The experimental studies were permitted by the German Animal Welfare Committee, and general care was provided as required.
Sampling
All pigeons and chickens that died or were killed were necropsied for determination of gross lesions and histopathology. Animals were killed when they developed severe clinical signs consisting of severe depression, reluctance to move, neurological signs such as torticollis, paralysis, and loss of consciousness, or after 19 dpi. Tissue samples of all animals were fixed in 4% phosphate-buffered neutral formaldehyde and processed for paraffin-embedding.
Serology
Sera of pigeons and chickens were collected before infection and at the time of euthanasia or at 19 dpi. For detection of H5-specific antibodies the hemagglutination-inhibition test (HI-test) with inactivated H5 antigens was used. 25
Generation of anti-nucleoprotein serum
The influenza virus strain Influenza A/FPV/Rostock/34 was used for the amplification of the viral sequence encoding the nucleoprotein (NP) gene by reverse transcription-polymerase chain reaction (RT-PCR) following standard procedures. The obtained RT-PCR fragment encompassing the complete coding region of the NP gene was cloned into the baculo transfer vector pAcGHLT-A (Pharmingen, Heidelberg, Germany), which resulted in a plasmid pAcGHLTA-NP-Ros encoding a fusion protein of the vector-derived glutathione-S-transferase (GST) and the open reading frame of the NP protein. After generation of a recombinant baculovirus using Baculo-Gold DNA (Pharmingen, Heidelberg, Germany), a recombinant NP expressing baculovirus was generated. The recombinant protein (GST-NP) was purified according to the manufacturer's instructions using GST-binding resin (Novagen, Bad Soden, Germany). Rabbits (SPF, Harlan, and Winkelman; Borchen, Germany) were inoculated intramuscularly four times with 100 µg of the purified GST-NP. The specificity of the anti-NP serum was monitored by Western Blot analysis using lysates of infected and noninfected cell cultures.
Histopathology and immunohistochemistry
Paraffin-wax sections (3 µm) were dewaxed and stained with hematoxylin and eosin (HE). To analyze distribution of HPAIV antigens, sections were mounted on positively charged SuperFrost Plus microscope slides (Menzel, Braunschweig, Germany), dewaxed, and rehydrated. The sections were incubated with the rabbit anti-NP serum. This antibody was applied at a dilution of 1 ° 500 in Tris-buffered-saline (TBS, 0.1 M Tris-base, 0.9% NaCl, pH 7.6). As linker-antibody for the avidin-biotin-complex (ABC) method, a biotinylated goat anti-rabbit (IgG1) (Vector, Burlingame, CA; diluted 1 ° 200 in TBS) was used. As negative control the preimmunization serum of the same rabbit was applied. By means of the ABC method and an immunoperoxidase kit (Vectastain Elite ABC Kit, Vector), a bright red signal was produced from the substrate, 3-amino-9-ethylcarbazole (DAKO AEC substrate-chromogen system; Dako, Carpinteria, CA). The sections were counterstained with Mayer's hematoxylin and sealed with aqueous medium (Aquatex; Merck, Darmstadt, Germany).
Results
Pigeons
Mortality and clinical signs
Ocular and intranasal administration of HPAIV Indonesia/2003 resulted in death of 5 of the 14 pigeons, during a time period of 19 dpi. Survival times varied from 5 dpi (pigeon No. 1) to 7 dpi (pigeons Nos. 2 and 3) to 19 dpi (pigeons Nos. 4 and 5, euthanized both with neurologic symptoms). Clinical signs were observed in all of these five animals (Table 1). Disease progressed over a period of 1 to 3 days from depression to enteric hemorrhage and severe neurological signs consisting of torticollis, nystagmus, and wing paresis to paralysis (Figs. 1 and 2). No clinical signs were observed in the nine pigeons (pigeon Nos. 6–14) euthanized 19 dpi (Table 1).
Clinical signs, time to death, gross and histopathology, and results of immunohistochemistry of pigeons and chickens intranasally and ocularly inoculated with HPAIV A/chicken/Indonesia/2003 (H5N1)
Killed humanely.
Histopathological grading of the lesions: (/) = none, (+) = mild, (++) = moderate, (+++) = severe.
Numbers of IHC-anti-H5N9 positive cells: (/) = none, (∗) = few, (∗∗) = moderate numbers, (∗∗∗) = numerous.

Pigeon No. 4, 19 dpi. Animal with hemiparalysis of the wing.

Pigeon No. 5, 19 dpi. Animal with severe torticollis.
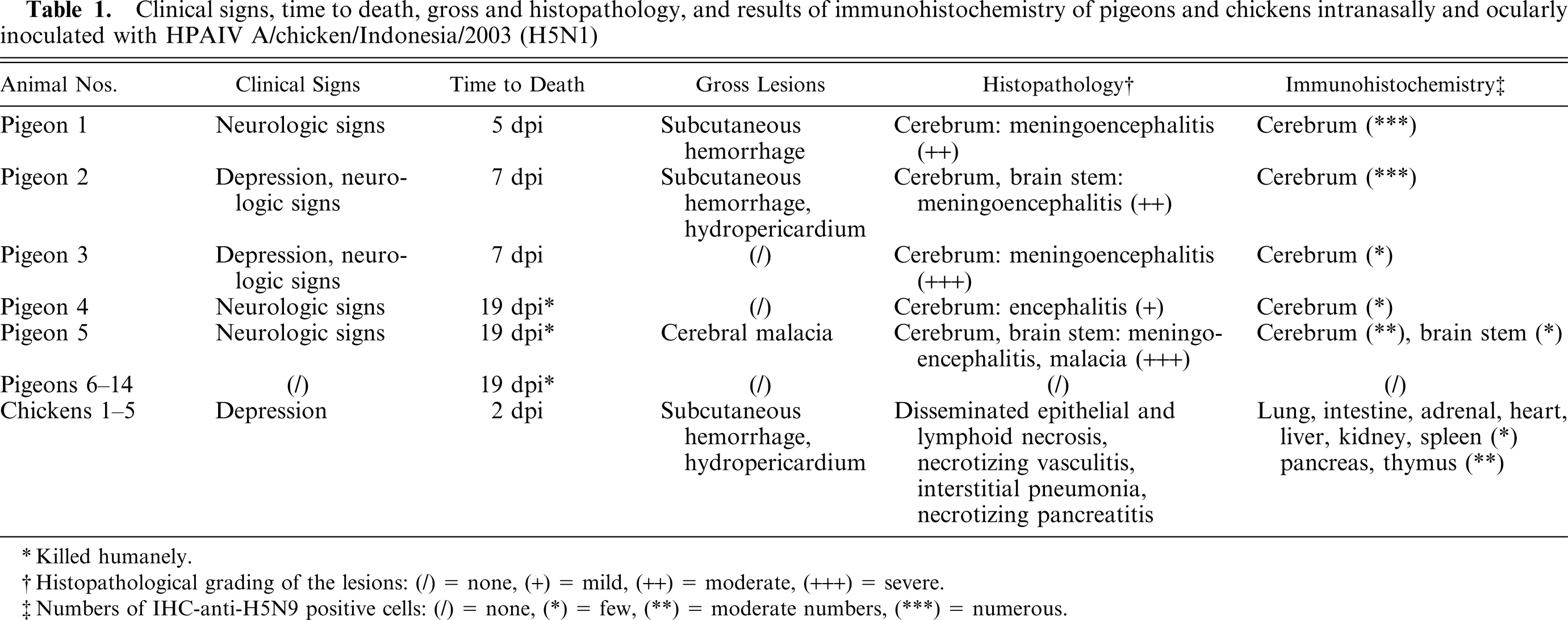
Transversal section of formalin-fixed brain: Pigeon No. 5, 19 dpi. Left hemisphere of the cerebrum with moderate reduction in size and irregularly shaped, fairly demarcated area of liquefactive necrosis of white and gray matter.
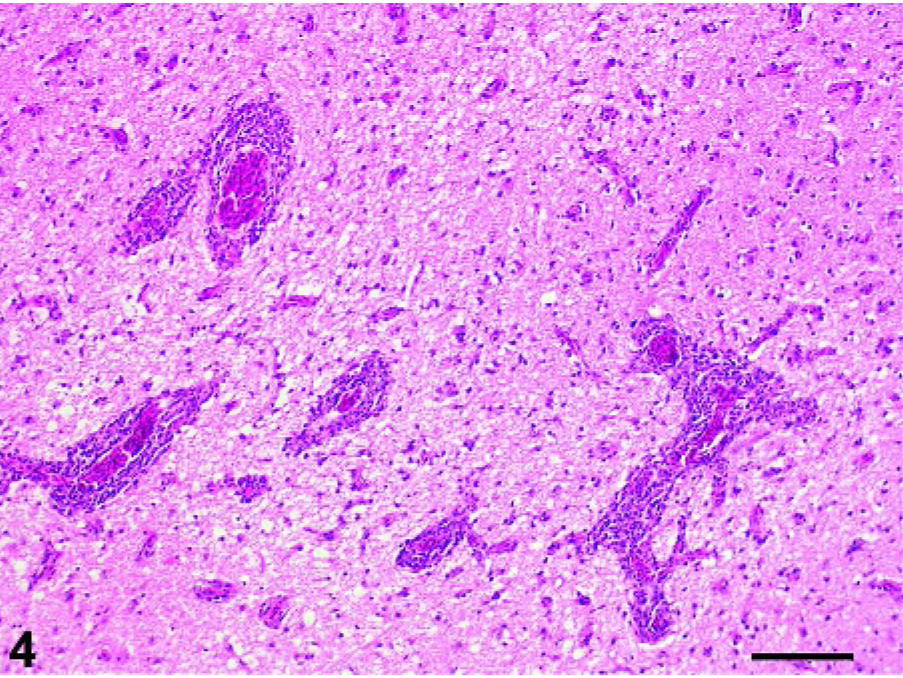
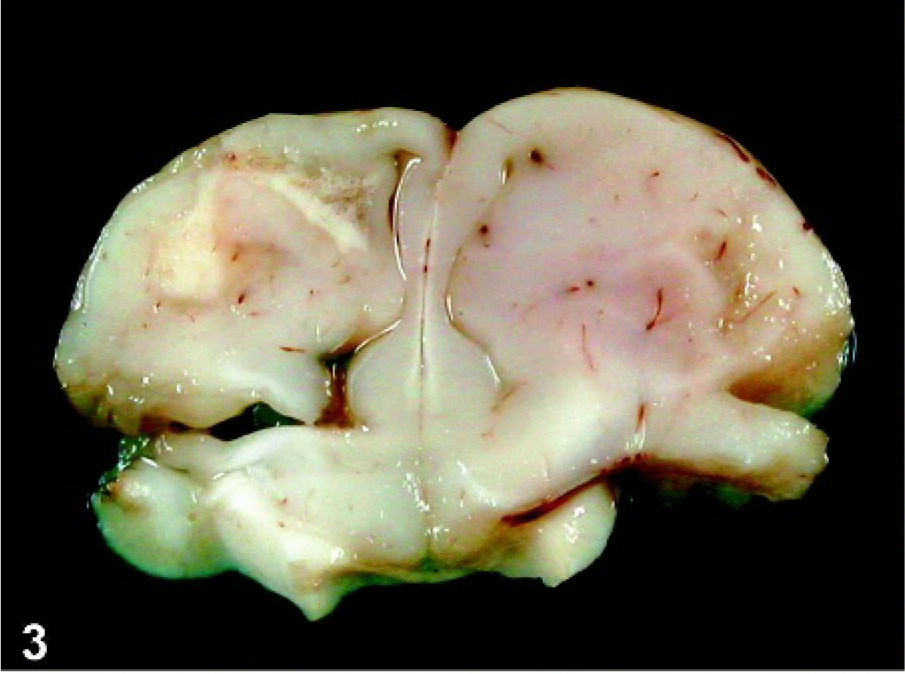
Cerebrum: Pigeon No. 4, 19 dpi. Severe perivascular cuffing of cerebral vessels and mild diffuse gliosis. HE. Bar = 250 µm.
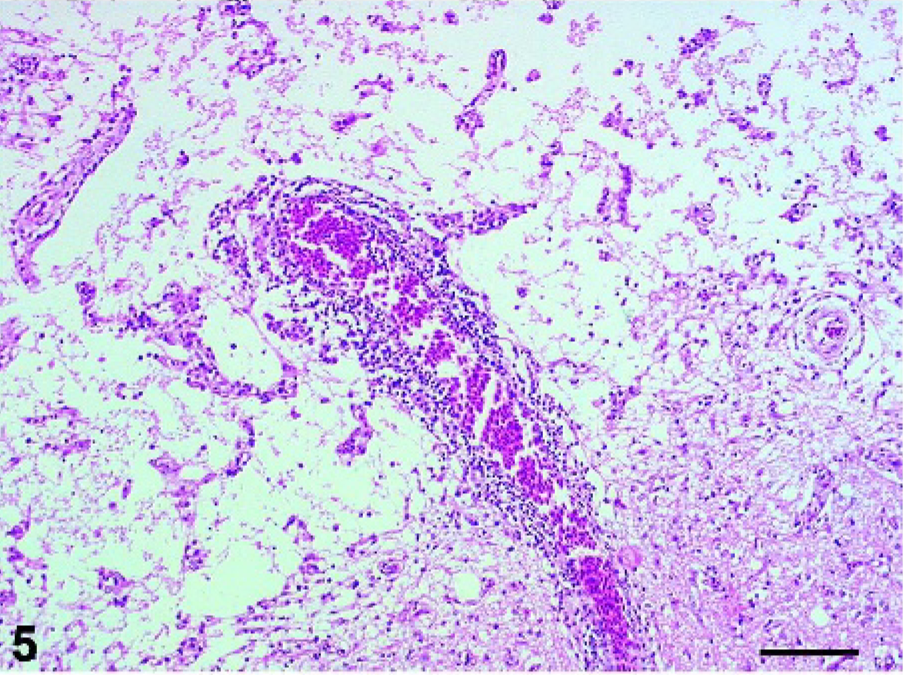
Cerebrum: Pigeon No. 5, 19 dpi. Area of liquefactive necrosis with complete loss of neuropil, remaining blood vessels and few finely vacuolated macrophages (gitter cells). HE. Bar = 250 µm.
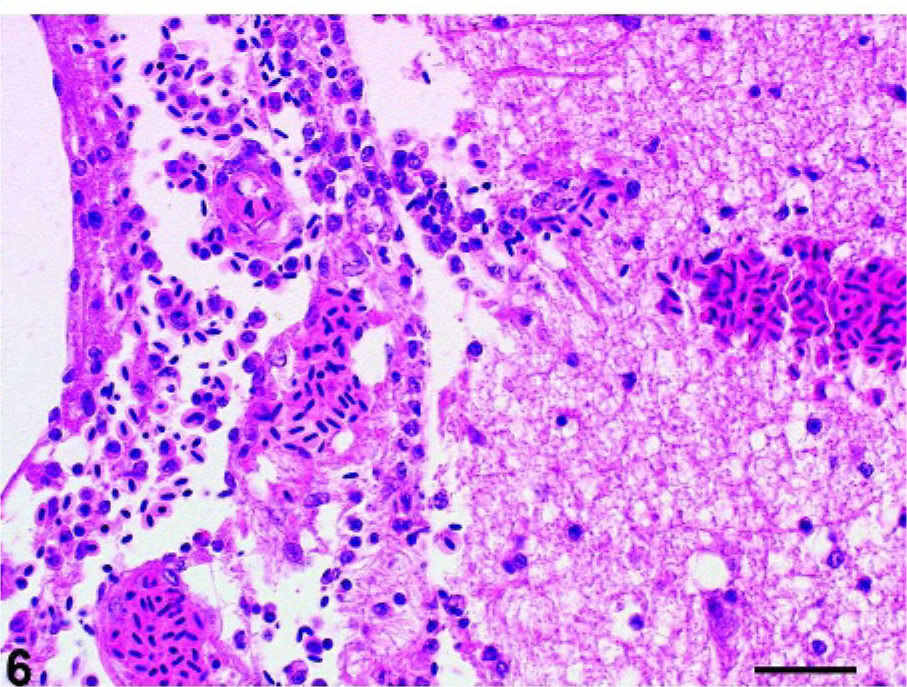
Cerebrum: Pigeon No. 4, 19 dpi. Moderate meningitis with small numbers of mononuclear cells and mild edema. HE. Bar = 175 µm.
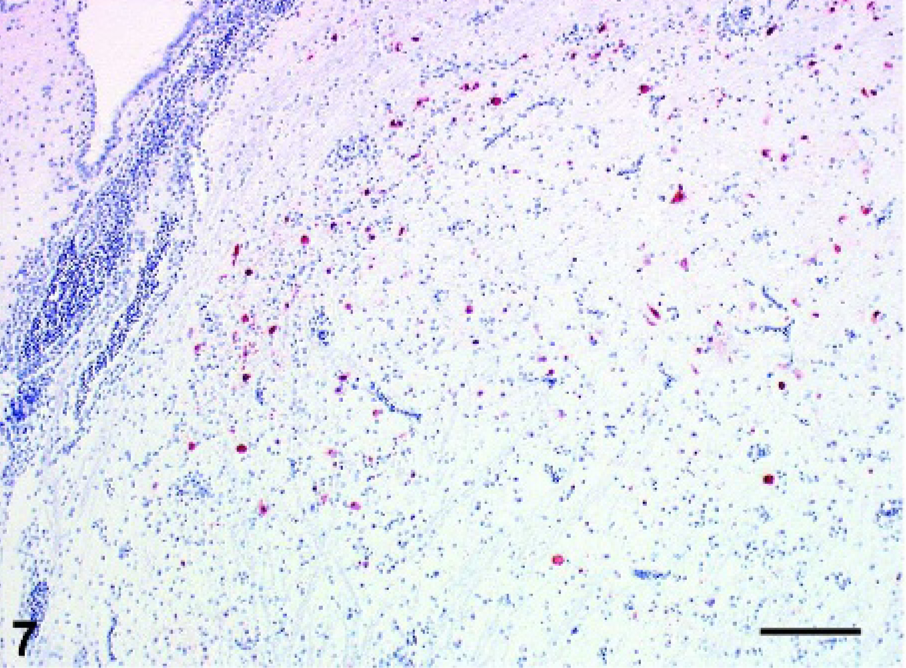
Cerebrum: Pigeon No. 5, 19 dpi. Immunohistochemical staining for AIV nucleoprotein in a moderate number of cerebral neurons and glial cells. ABC method with hematoxylin counterstain. Bar = 500 µm.
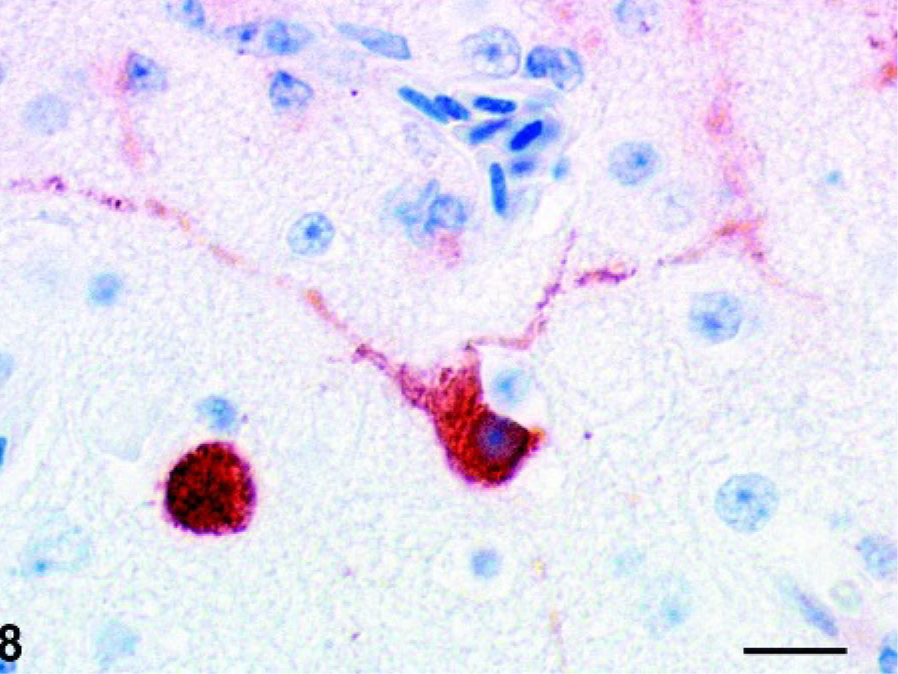
Cerebrum: Pigeon No. 2, 7 dpi. Intranuclear and intracytoplasmic staining for AIV nucleoprotein in a cerebral neuron. ABC method with hematoxylin counterstain. Bar = 25 µm.
Serology
In all of the nonsymptomatic pigeons Nos. 6–14, significant H5-specific titers were detected on day 19 pi (ranging from 1 ° 32 to 1 ° 64).
Gross pathology
At necropsy, only mild and inconsistent lesions were present (Table 1). Whereas pigeons Nos. 1 and 2 showed a focal, well-circumscribed, 2-cm-diameter area of hyperemia and hemorrhage in the subcutaneous venous plexus of the neck, a hydropericardium was found in pigeon No. 2 (Table 1). A focally extensive area of unilateral cerebral malacia was observed in pigeon No. 5. In this case the complete anterior part of the left hemisphere showed a focal, well-delineated indentation with softening and yellow-brown discoloration of the neuroparenchyma as well as hyperemia of adjacent meningeal vessels (Fig. 3). Gross lesions could not be observed in pigeons Nos. 3 and 4 and the animals were killed at 21 dpi.
Histopathology
Histologic lesions were confined to the central nervous system (CNS) of the pigeons that developed clinical signs of HPAI (Table 1). Diffusely, lesions were present mainly in the cerebral gray matter but also in the brain stem. In the cerebrum, multifocal, moderate to severe necrosis of neurons and glial cells and hemorrhages were found in pigeons Nos. 1–5. Corresponding to these lesions, prominent multifocal glial nodules and occasional neuronophagia could be observed in the cerebral gray and white matter. Vessels showed multifocal perivascular cuffing with one to four layers of numerous lymphocytes, fewer histiocytes, and small numbers of heterophils (Fig. 4). In pigeon No. 5, additional extensive, multifocal to coalescent, irregularly shaped, moderately demarcated areas of liquefactive necrosis were found in the cerebrum, characterized by central complete loss of neuropil, leaving empty spaces that contained preexisting arterioles, small amounts of pale eosinophilic material, and few finely vacuolated macrophages (gitter cells; Fig. 5). Milder lesions were found in the brain stem but not in the cerebellum of pigeons Nos. 1 and 5. Rarely, swelling, degeneration, and necrosis of endothelial cells were observed. Four of the five affected pigeons showed moderate diffuse meningitis with small numbers of mainly perivascular mononuclear cells and mild edema (Fig. 6). Histologic examination of the heart, aorta, larynx, lung, spleen, pancreas, kidney, proventriculus, gizzard, and duodenum revealed no lesions. No lesions were observed in the pigeons that did not develop clinical signs.
Immunohistochemistry
Influenza virus antigen strongly colocalized with the histologic lesions in the brain. Intranuclear and intracytoplasmic staining for NP antigen was seen in numerous viable as well as degenerating and necrotic neurons of the cerebrum and brain stem as well as in a few glial cells and capillary and arteriolar endothelial cells of pigeons Nos. 1–3 (Figs. 7 and 8). In contrast, in pigeons Nos. 4 and 5 only a few NP antigen positive neurons and glial cells were detected in the cerebrum and the brain stem but no staining of endothelial cells was observed. In pigeon No. 5 few positive neurons surrounded closely the area of liquefactive necrosis. In pigeons Nos. 1–5 no viral antigen as detected in heart, larynx, lung, spleen, pancreas, kidney, or intestine. Furthermore, viral antigen could not be detected in pigeons Nos. 6–14 (Table 1).
Chickens (positive control group)
Inoculation with HPAIV A/chicken/Indonesia/2003 (H5N1) resulted in 100% mortality in chickens. Clinical signs were mild depression, anorexia, and minimal dyspnea. After occurrence of first clinical signs, the disease progressed rapidly to death within 8–12 hours at 2 dpi. At necropsy, lesions in the chickens consisted of ruptured ovarian follicles with diffuse serofibrinous “egg yolk” peritonitis and subserosal hemorrhages, scattered mucosal hemorrhages in the proventriculus, petechiation in adipose tissue as well as moderate to marked congestion of the lungs, liver, and spleen.
Histologic lesions were observed in a wide variety of organs and consisted of disseminated fibrinoid and necrotizing vasculitis with necrosis of endothelial cells, mild to moderate perivascular, mainly lymphohistiocytic, infiltrates and lymphoid depletion with necrosis in spleen, Peyer's patches, bursa, and perilaryngeal lymph follicles. The lungs showed marked congestion and mild infiltration with few lymphocytes and heterophils within the mildly edematous interstitial connective tissue. Furthermore, discrete foci of single cells or group necrosis of the pancreatic acinar epithelium, proventricular mucosa, and renal tubular epithelium as well as hepatocytes could be observed. Inflammatory response corresponding to these lesions was absent or minimal and consisted of few infiltrating heterophils. Immunohistochemically, intralesional, intranuclear, and intracytoplasmic staining for viral antigen was found within leukocytes of the Peyer's patches, bursa, perilaryngeal lymph follicles as well as cortical and medullary epithelium of the adrenal glands, renal tubules, pancreatic acini, and in few pneumocytes. Endothelial cells of small vessels and capillaries were invariably and diffusely positive for HPAIV antigen.
Sentinel chickens and negative control pigeons
Sentinel chickens and non-infected pigeons were euthanized at day 19 pi. None of the animals showed any clinical signs. Gross pathology and histopathology revealed no lesions in the brain, heart, vessels, larynx, lung, spleen, pancreas, kidney and intestine. Immunohistochemical staining detected no viral antigen in these tissues. Sera of the sentinel chickens revealed no significant H5-specific antibodies on day 19 pi.
Discussion
Ocular and nasal administration of HPAIV A/chicken/Indonesia/2003 (H5N1) resulted in moderate morbidity and mortality in pigeons within 5 to 19 dpi. Clinically, the affected birds showed depression and neurological dysfunction. Histopathology and immunohistochemistry demonstrated a pronounced neurotropism of HPAIV A/chicken/Indonesia/2003 (H5N1) for the cerebrum and brainstem in these pigeons. This is the first complete investigation integrating increased mortality with histopathology and distribution of viral antigen after infection of pigeons with HPAIV of the subtype H5.
Several transmission experiments addressed the susceptibility of pigeons to characterized and quantified HPAIV of the subtypes H5 and H7. 5, 10, 15, 16, 18, 20 However, so far only one report demonstrated death of 1/19 pigeons after intranasal administration of HPAIV A/turkey/Ontario/7732/66 (H5N9). 24 In contrast to these results, our experiments demonstrate that pigeons are susceptible to HPAIV A/chicken/Indonesia/2003 and that this influenza virus exhibits a strong neurotropism in this species. The seroconversion of the nine pigeons that did not develop signs of avian influenza shows that these animals, although primarily infected, were obviously able to clear the virus. Furthermore the lack of clinical symptoms and of seroconversion in the sentinel chickens shows that bird-to-bird transmission of the virus did not occur.
Infections of the CNS with HPAIV of the subtype H5 have been observed in a wide variety of avian and mammalian species. Particularly in mice and rats but also in humans and ferrets, this virus displays a marked neurotropism. 3, 9, 21, 29 Among avian species, the brain was consistently affected in emus, geese, house finches, and budgerigars, but microscopic lesions and viral antigen were also detected in other organs. 17, 19 In chickens, zebra finches, Japanese and Bobwhite quails, turkeys, guinea fowl, pheasants, and partridges, infection was evenly distributed in the body including the brain. 17, 19 Consequently, the pronounced neurotropism in pigeons observed in our experiments seems to be unprecedented at least in avian species. However, the virus may be more widely distributed in pigeons at earlier time points after inoculation.
Multiple basic amino acid residues defining a furin-sensitive cleavage site of the hemagglutinin protein and cellular proteases determine the virulence of avian influenza viruses by rendering them processable in virtually all receptor-positive cells throughout the body. 11, 22 In addition, an influence of the structure of the polymerase PB2 for virulence of Hong Kong H5N1 influenza A virus in mice could be shown. 7 The molecular basis of the neurotropism of certain influenza virus strains is not known but obviously can be influenced by the host and the virus strain. 25 Furthermore, viral adaptation to the host might be important, since enhanced neuropathogenicity of an influenza virus strain (H5N3) could be produced by repeated passages through air sacs and brains of chickens. 23
It has been shown that highly pathogenic influenza virus replicates in endothelial cells throughout the body during early stages of infection in chickens, and virus spread in the body occurs following viremia. 4, 12, 26 After penetrating the blood-brain barrier, infection is propagated to glial cells and neurons. 23 In mice, CNS infections may occur by ascending transsynaptic transmission of H5N1 via vagal and trigeminal nerve fibers. 13, 25 Hence, viral antigen was found mainly in vagal and trigeminal ganglia, and vagal and trigeminal nuclei of the brainstem but not in the cerebral cortex. 14 The exact mechanism of intraneural transport of avian influenza A is unknown but a dependence on integrity of intermediate filaments such as neurofilament has been shown. 14 In contrast to the situation in mice, the more diffuse infection of cortical and midbrain neurons and the sparing of brain stem nuclei of cranial nerves raises the question of whether infection of the CNS in the pigeons follows rather a hematogenic than a neurogenic route. Nevertheless, the exact pathogenesis remains to be further elucidated and is currently under investigation in our laboratory. Whether the observed malacia in pigeon No. 5 was directly caused by viral infection of neuronal structures with necrosis or apoptosis in this area or by ischemia caused by endothelial damage after infection and consequent thrombotic infarction remains unclear. 8 Although adjacent neurons and glial cells stained for AIV NP-antigen and no thrombi could be detected, the focally extensive pattern and the limitation to one animal favors a vascular pathogenesis.
While reported isolations of HPAIV H5N1 were limited to one dead wild pigeon during the outbreak of avian influenza in Hong Kong 2002, the results obtained from this experiment support the view that pigeons are not resistant to infection and might be at least hypothetically involved in transmission of avian influenza. 6 Further experimental studies are ongoing to investigate the shedding of avian influenza virus by infected pigeons.
Footnotes
Acknowledgements
We are grateful for the excellent technical assistance provided by G. Czerwinski and C. Illing.
