Abstract
Disseminated encephalitozoonosis was diagnosed in 2 sibling, juvenile, cotton-top tamarins (Saguinus oedipus) and 3 sibling, neonatal, emperor tamarins (S. imperator) by use of histologic examination, histochemical analysis, electron microscopy, and polymerase chain reaction (PCR) analysis with nucleotide sequencing. All tamarins were captive born at zoos in North America and died with no premonitory signs of disease. The main pathologic findings were myocarditis (4/5), hepatitis (3/5), interstitial pneumonia (3/5), skeletal myositis (3/5), meningoencephalitis (2/5), adrenalitis (2/5), tubulointerstitial nephritis (1/5), myelitis (1/5), sympathetic ganglioneuritis (1/5), and retinitis (1/5). Central nervous system lesions were the most prominent findings in cotton-top tamarins. The inflammation was predominantly lymphocytic and suppurative in cotton-top tamarins, whereas emperor tamarins had granulomatous or lymphoplasmacytic lesions. Intralesional periodic acid-Schiff-, gram-, or acid-fast (or all 3)-positive, oval-to-elliptical shaped organisms were found in 1 cotton-top and the 3 emperor tamarins. By electron microscopy, these organisms were consistent with microsporidia of the genus Encephalitozoon. E. cuniculi genotype III was detected by PCR analysis and sequencing in paraffin-embedded brain, lung, and bone marrow specimens from the cotton-top tamarins. Although PCR results were negative for one of the emperor tamarins, their dam was seropositive for E. cuniculi by ELISA and Western blot immunodetection. These findings and recent reports of encephalitozoonosis in tamarins in Europe suggest that E. cuniculi infection may be an emerging disease in callitrichids, causing high neonatal and juvenile mortality in some colonies. The death of 2 less than 1-day-old emperor tamarins from a seropositive dam supports the likelihood of vertical transmission in some of the cases reported here.
Keywords
Microsporidiosis is a heterogeneous group of diseases in a wide range of invertebrate and vertebrate animal species caused by parasites in the Phylum Microsporidia. Microsporidia are single-celled, spore-forming, obligate, intracellular parasites with a unique organelle, the polar filament. One of the best known microsporidian parasites is Encephalitozoon cuniculi, which infects a variety of rodents, rabbits, carnivores (dogs and blue foxes), nonhuman primates, and humans. 8, 29, 31 In squirrel monkeys (Saimiri sciureus), E. cuniculi infection can be congenital and appears to be most severe in stillbirths and neonates. 1, 2, 27, 32
Microsporidiosis caused by Enterocytozoon bieneusi develops in macaques with and without concurrent simian immunodeficiency virus infection, and principally affects the biliary tree, but it also affects the small intestinal mucosa. 6, 15, 21, 22 In 2003 and 2004, disseminated E. cuniculi infections were documented in emperor (S. imperator) 16 and cotton-top (Saguinus oedipus) 23 tamarins housed at European zoos. Cotton-top tamarins are an endangered species endemic to Colombia, and emperor tamarins are a vulnerable species distributed in Brazil and Perú; 25 both species are subject to captive breeding programs, and cotton-top tamarins also are used as animal models of several human diseases, mostly chronic colitis and colon cancer. 17 We describe disseminated E. cuniculi infection in captive cotton-top and emperor tamarins in North America during outbreaks of disease affecting single family groups at various zoos in 2004 and 1999, respectively.
Materials and Methods
Case histories
All 5 tamarins included in this study were captive born and were housed at 2 zoos in North America; they died spontaneously with no premonitory signs of disease. Table 1 summarizes species, age, sex, and body weight at necropsy. Tamarin Nos. 1 and 2 were siblings born to a breeding pair with a previous high neonatal and juvenile mortality of its offspring. They were housed in an outdoor enclosure with curasows (Crax spp.). The breeding pair and a 1-year-old tamarin from a previous pregnancy never manifested clinical signs of disease, except for a dry, dull coat, with moderate diffuse hypotrichosis of the tail. This dermatosis was present in other cotton-top tamarins at the same facility and had previously responded to zinc sulfate supplementation of 60 ppm in the drinking water.
Species, age, sex, and body weight (BW) at necropsy of 5 tamarins with encephalitozoonosis.

NA = not available (tamarin Nos. 3 and 4 were partly predated).
Tamarin Nos. 3–5 were sibling emperor tamarins. Tamarin Nos. 3 and 4 died a few hours after birth, and tamarin No. 5 died 16 days later. The breeding pair did not manifest clinical signs of disease.
Necropsy and cytologic and histologic examination with special stains
All tamarins that died were necropsied. Touch imprints were obtained from the brain of tamarin No. 2 and were stained by use of the Gram and Ziehl-Neelsen methods. Tissues collected at necropsy were fixed in buffered 10% formalin, embedded in paraffin, sectioned at 5-µm thickness, and stained with hematoxylin and eosin (HE). Special staining of selected tissues from tamarin Nos. 1 and 3–5 included Gram, acid-fast, and periodic acid–Schiff (PAS) methods.
Transmission electron microscopy
Paraffin-embedded brain tissue from tamarin No. 1 and formalin-fixed liver from tamarin No. 5 were processed in routine manner for transmission electron microscopy.18,24,26,30 A Phillips EM 201 (tamarin No. 1) or Zeiss 10 C (tamarin No. 5) transmission electron microscope with a beam set at 60 kV was used.
Extraction of DNA, polymerase chain reaction (PCR) analysis, and nucleotide sequencing
The QIAmp DNA Mini Kit (Qiagen Inc., Valencia, CA) was used to extract DNA from paraffin-embedded lung, brain, and bone marrow tissue sections from tamarin Nos. 1 and 2. The manufacturer's directions were followed, with the modification that, after the dexylenation procedure, lyticase (100 U/200 µl of reaction volume; Sigma Chemical Co., St. Louis, MO) was added to the pellet for incubation at 30°C for 30 minutes prior to digestion with proteinase K. Microsporidia used as controls included tissue culture-harvested E. cuniculi (strains I–III), E. hellem, and E. intestinalis. For microsporidian species identification, the extracted DNA was subjected to a first round of PCR analysis using the rDNA primers MSP-1 (5′-TGAATG(G/T)GTCCCTGT-3′), MSP-2A (5′-TCACTCGCCGCTACT-3′), and MSP-2B (5′-GTTCATTCGCACTACT-3′). A second round of nested PCR analysis was performed using the primers MSP-3 (5′-GGAATTCACACCGCCCGTC(A/G)(C/T)TAT-3′), MSP-4A (5′-CCAAGCTTATGCTTAAGT(C/T)(A/C)AA(A/G)GGGT-3′), and MSP-4B (5′-CCAAGCTTATGCTTAAGTCCAGGGAG-3′) as described. 14, 20, 23 A reagent control without template also was included in each set of the PCR assays. Thermocycling conditions were 95°C for 5 minutes, 35 cycles of 94°C for 2 minutes, 55°C for 2 minutes, and 72°C for 3 minutes, followed by a 10-minute incubation at 72°C. Amplicons were electrophoresed in 1% agarose, viewed after staining with ethidium bromide, excised, and purified from agarose using the QIAquick gel extraction kit (Qiagen Inc.). Nucleotide sequences were determined at the DNA Sequencing Core in the Center for Gene Therapy, Tulane University Health Sciences, Center (New Orleans, LA) using the BigDYE Terminator sequencing and ABI Prism 3700 sequence analysis systems (Applied Biosystems, Foster City, CA).
Serologic testing
Sera from the dam and sire of tamarin Nos. 3–5 were assessed by ELISA and Western blot assay against tissue culture-derived Encephalitozoon species (E. cuniculi, E. hellem, and E. intestinalis) as well as Vittaforma corneae, as described. 9, 12 Briefly, 96-well ELISA plates were coated with solubilized microsporidial proteins, incubated with serial dilutions of test tamarin sera, followed by reactivity with alkaline phosphatase-conjugated, goat antimonkey IgG, and p-nitrophenyl phosphate. Absorbance was measured at 405 nm, and end-point titer was determined as the highest dilution prior to reaching baseline with the mean absorbance value remaining significantly higher than the value for the Tris-buffered saline controls, on the basis of results of Student's t-test analysis. For Western blot immunodetection, microsporidial proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis, blotted onto nylon, incubated with test sera diluted 1 ° 1,000, or Tris-buffered saline as the control, followed by reactivity with alkaline phosphatase-conjugated, goat antimonkey IgG and staining with nitroblue tetrazolium (NBT) and bromochloroindolyl phosphate (BCIP). A second set of blots was incubated with Tris-buffered saline to control for nonspecific staining of the secondary conjugated antibodies.
Results
Gross pathology
At necropsy, tamarin Nos. 1 and 2 had severe atrophy of adipose tissue and mild to moderate splenomegaly. Tamarin No. 1 was moderately dehydrated, with a whitish material adhered to the dorsal surface of the tongue, and locally extensive necrotizing submandibular dermatitis and cellulitis. Tamarin No. 2 had mild fibrinous peritonitis, moderate scoliosis with segmental narrowing of the right hemi-thorax, moderate cardiomegaly attributable to right-sided hypertrophic cardiomyopathy, and mild generalized osteopenia. Touch imprints from the brain of tamarin No. 2 were negative for gram-positive and acid-fast microorganisms. Results of aerobic culture of splenic specimens from tamarin No. 2 were negative.
Grossly, no lesions were detected in tamarin Nos. 3–5. In tamarin No. 3 and 4, the head and right forelimb were partly missing, which was attributed to postmortem predation. The lungs of tamarin Nos. 3 and 4 floated in formalin.
Histologic examination and special staining
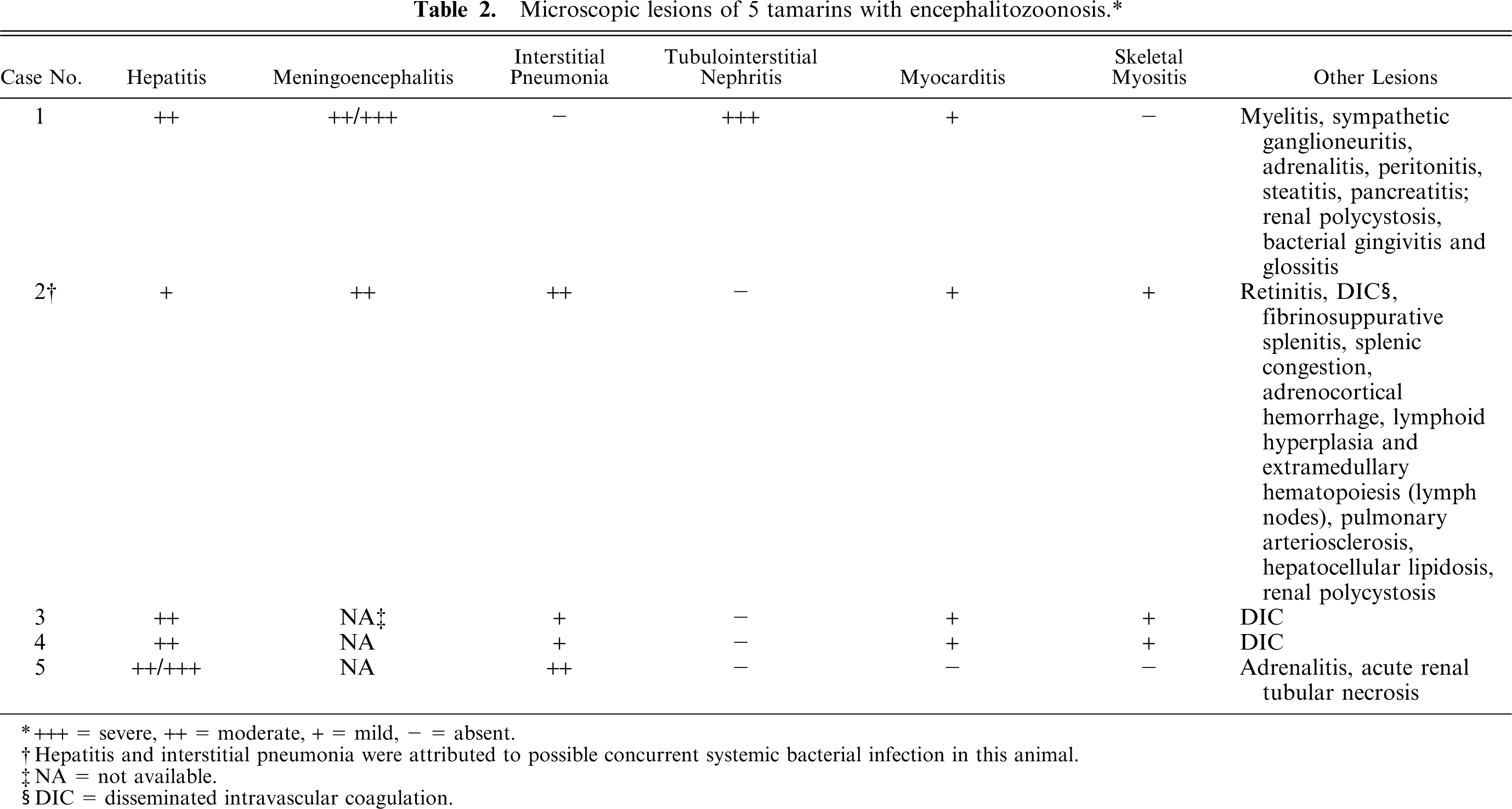
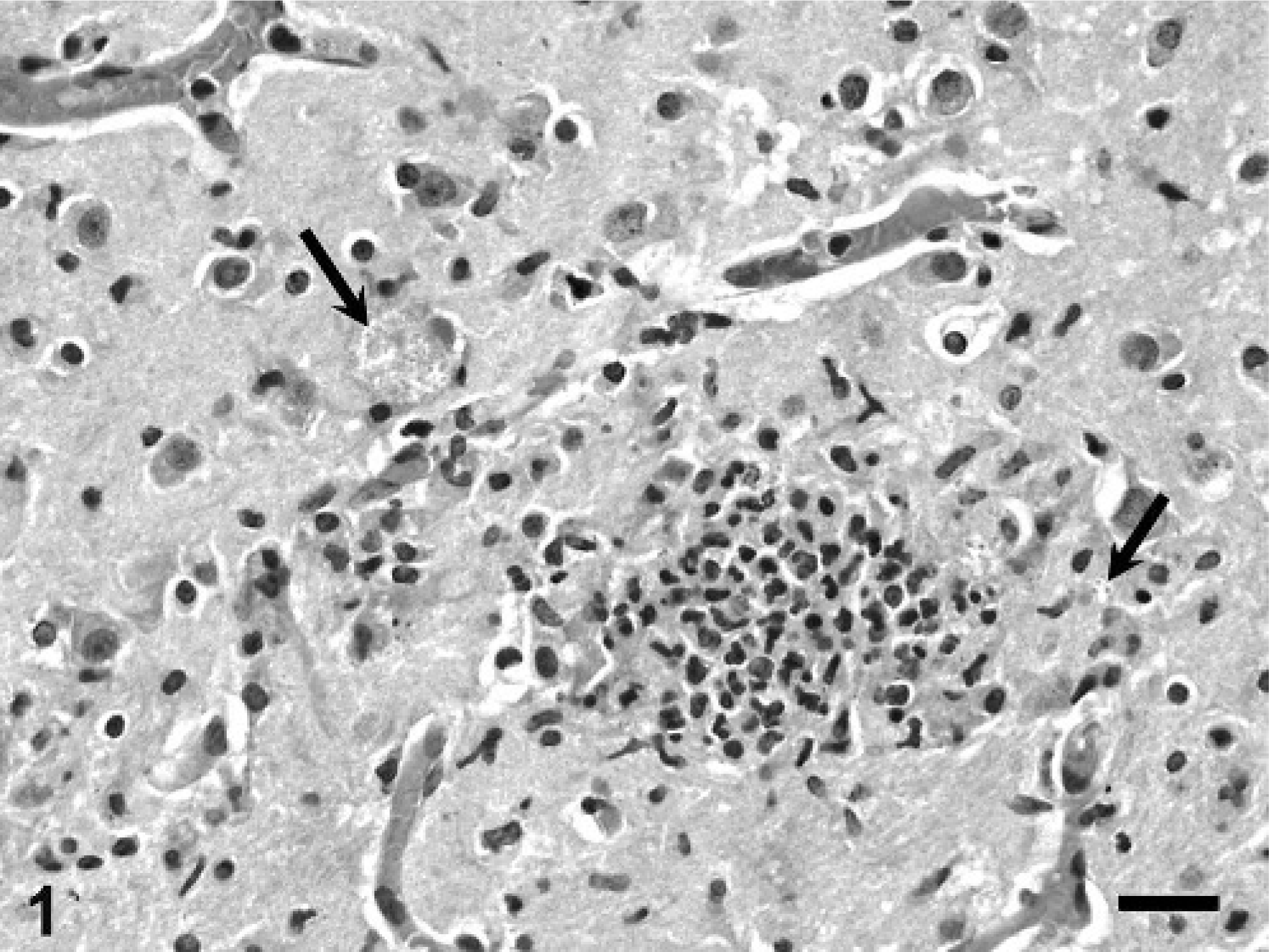
Table 2 summarizes the histologic lesions found in these tamarins. Tamarin No. 1 had numerous aggregates of neutrophils, lymphocytes, and glial cells, with central necrosis distributed randomly throughout the gray and white matter of the cortex, thalamus, hippocampus, cerebellum, and pons, in association with multifocal to diffuse gliosis and lymphocytic perivascular cuffing (Fig. 1). There were numerous intralesional colonies of oval-to-elliptical shaped, slightly basophilic microorganisms, approximately 2 µm in their greatest dimension (Fig. 1). These colonies were also found distant to the sites of inflammation. The pachymeninges had similar, but milder multifocal inflammation. There were small to large areas of mineralization, intralesional and distant to the sites of inflammation, especially in the cerebellum, but also in the cortex and thalamus. The gray matter of the spinal cord and a sympathetic, periadrenal ganglion had inflammation similar to that described in the brain, and similar microorganisms were found in the spinal cord. The kidneys had large foci of tubulointerstitial inflammation composed mostly of lymphocytes and neutrophils, but also containing plasma cells and macrophages. A few intralesional tubules were necrotic and had luminal aggregates of necrotic leukocytes. The liver had numerous foci of lymphocytic and neutrophilic inflammation randomly distributed in the acini and occasionally in the portal areas. The peripancreatic adipose tissue and peritoneum, pancreatic interlobular septae and the zona fasciculata of adrenal glands had similar but milder inflammation. The microorganisms found in the central nervous system reacted positively to Gram and PAS staining (Fig. 2). Organisms were not observed in other organs by use of special stains.
Microscopic lesions of 5 tamarins with encephalitozoonosis.∗
+++ = severe, ++ = moderate, + = mild, - = absent.
Hepatitis and interstitial pneumonia were attributed to possible concurrent systemic bacterial infection in this animal.
NA = not available.
DIC = disseminated intravascular coagulation.
Brain; cotton-top tamarin No. 1. Notice focus of lymphocytic and suppurative encephalitis and gliosis associated with colonies of oval-shaped organisms (arrows). HE. Bar = 32 µm.
Brain; cotton-top tamarin No. 1. Notice large microsporidial colony (arrow). PAS. Bar = 30 µm.
Tamarin No. 2 had similar, but less prominent brain lesions affecting predominantly the gray matter, with no microsporidia detectable. There were large foci of mineralization in the white matter of the cerebellum with extension into the granular layer of the cerebellar folia; the wall of a few small vessels also was mineralized. There was mild multifocal interstitial lymphoplasmacytic and suppurative inflammation in the skeletal muscle and myocardium, with perivascular distribution in the retina. There were numerous fibrin thrombi in glomerular capillaries in the kidney, and in hepatic, splenic, and pulmonary capillaries. Other relevant lesions were moderate, diffuse, acute interstitial pneumonia with acute alveolar hemorrhage; moderate, multifocal, acute fibrinosuppurative splenitis with acute hemorrhage of lymphoid follicles; mild, acute, focal necrosuppurative hepatitis; moderate to severe, acute splenic congestion; and mild, acute, multifocal adrenocortical hemorrhage.
Tamarin Nos. 3 and 4 had microgranulomas distributed randomly throughout the liver that were composed of epithelioid macrophages oriented around small accumulations of oval-to-elliptical shaped microorganisms, approximately 2 µm in their greatest dimension, that were gram positive with acid-fast polar staining. The heart and skeletal muscle had mild perivascular and interstitial infiltrates of lymphocytes associated with necrosis of a few myofibers. The pulmonary interstitium was slightly thickened owing to infiltration of mononuclear inflammatory cells, and some alveoli contained luminal aggregates of neutrophils. The adrenal cortex had a few individual necrotic cells. There were fibrin thrombi in hepatic and myocardial blood vessels.
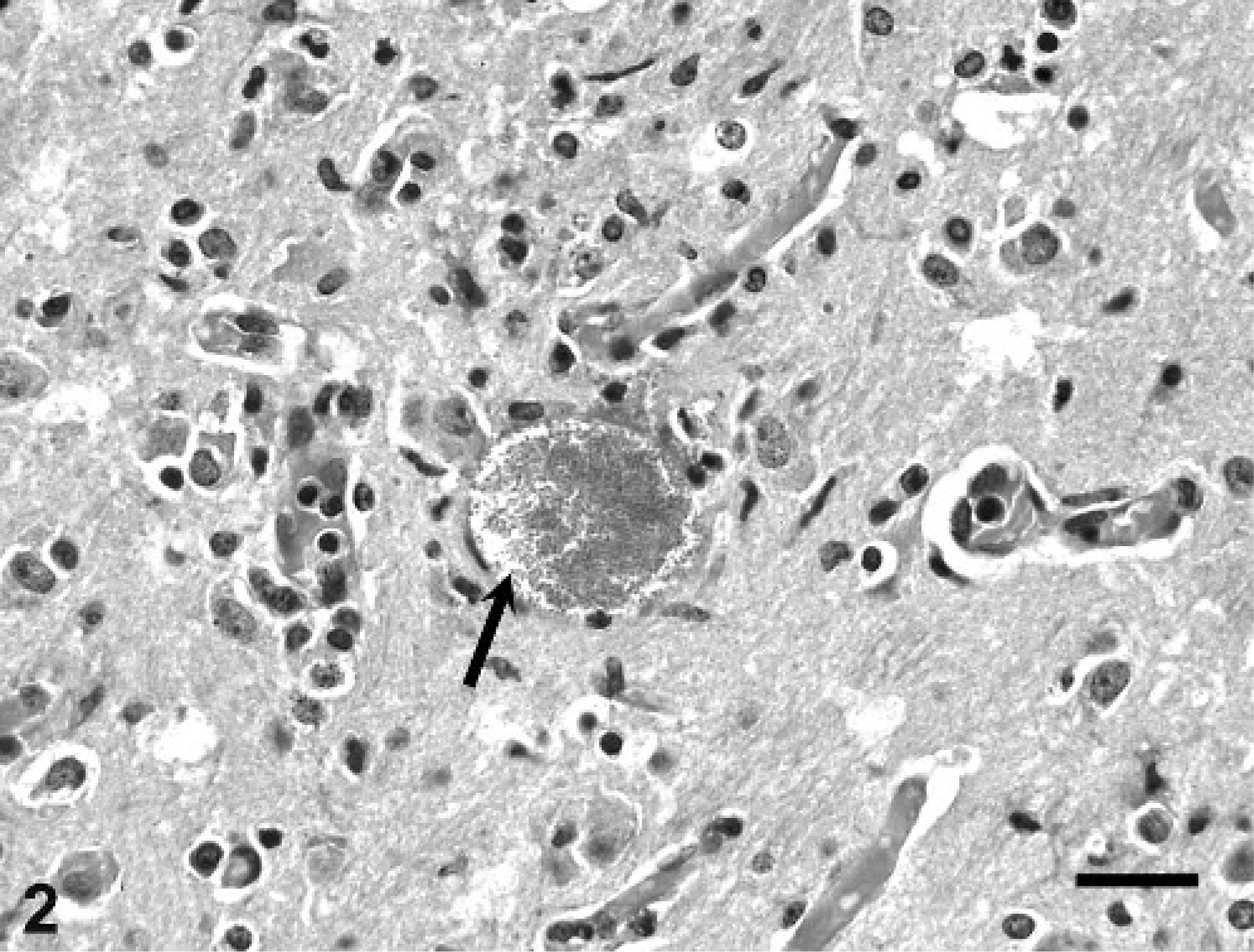
Tamarin No. 5 had aggregates of plasma cells and lymphocytes, and fewer macrophages associated with hepatocellular necrosis distributed randomly throughout the hepatic parenchyma. A few small foci of granulomatous inflammation were detected in the adrenal cortex (Fig. 3). The pulmonary interstitium was diffusely thickened by infiltration of moderate numbers of lymphocytes and macrophages. The kidneys had a few tubules lined by necrotic epithelium or luminal cell debris, or both. The heart had mild, multifocal coagulative necrosis of myofibers, and a large central zone of necrosis was observed in the ventricular septum. The spleen had moderate histiocytosis of the red pulp. There were intralesional organisms similar to those of tamarin Nos. 3 and 4 within the cytoplasm of macrophages in the hepatic, adrenal, pulmonary, and splenic lesions, and in a few renal mesangial cells. These organisms were gram positive (Fig. 4) and partially acid fast, and had a PAS-positive polar cap.
Adrenal gland; emperor tamarin No. 5. Notice well circumscribed focus of granulomatous inflammation in the adrenal cortex (thick arrows) associated with a colony of microsporidia (thin arrow). HE. Bar = 36 µm.
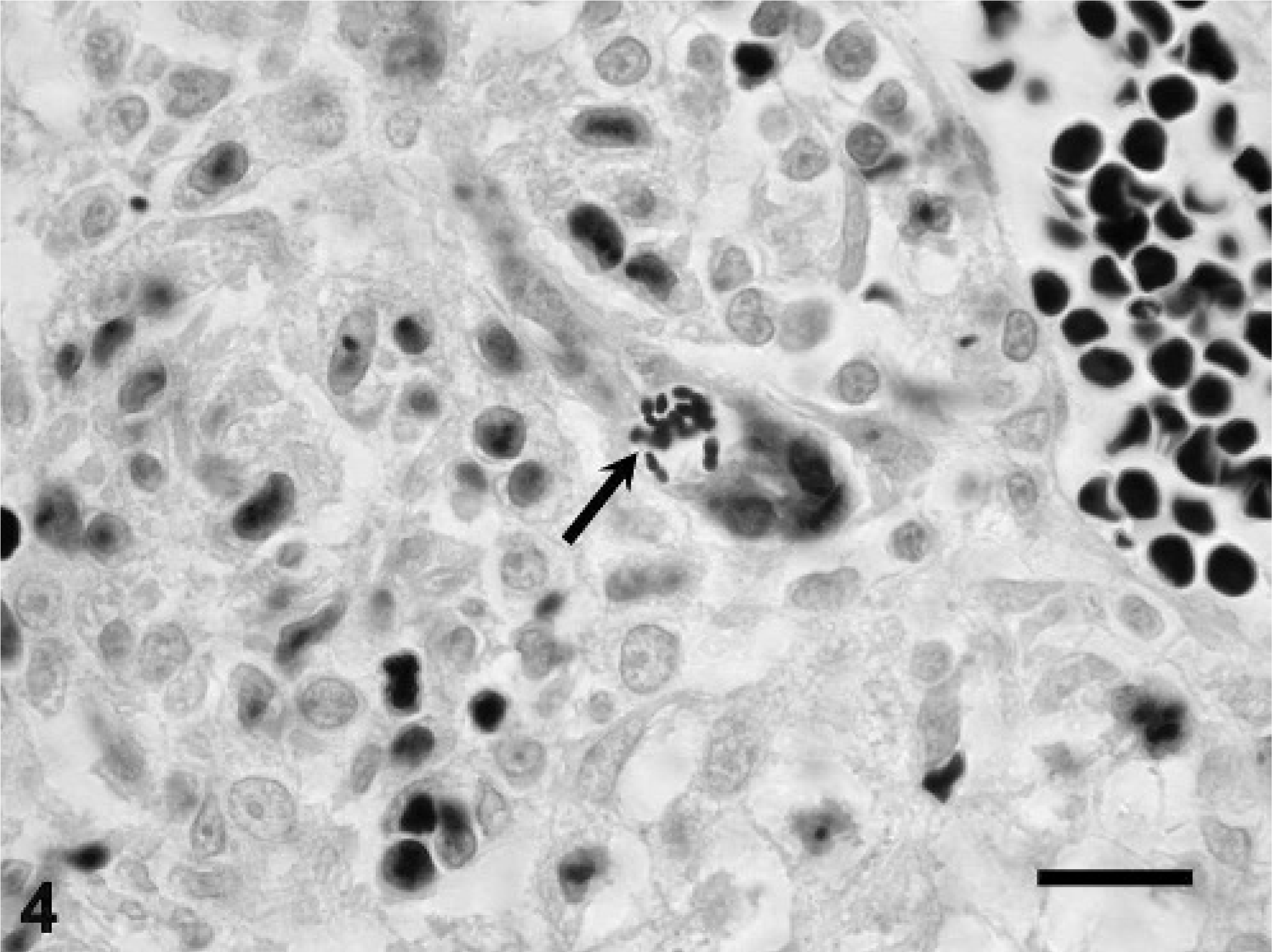
Liver; emperor tamarin No. 5. Notice colony of gram-positive oval-to-elliptical shaped microsporidial organisms (arrow). Gram stain. Bar = 20 µm.
Transmission electron microscopy
The microorganisms observed by histologic examination in tamarin Nos. 1 and 5 consisted of aggregates of mature microsporidial spores within an electron-lucent, parasitophorous vacuole in the host cell that was characteristic of the Encephalitozoon species, E. hellem or E. cuniculi (and different from E. intestinalis, which secretes a substance that causes the parasitophorous vacuole to appear septate). 4, 5 Spores had an electron-lucent endospore covered by an electron-dense exospore, a moderately electron-dense sporoplasm usually with a distinct, more electron-dense, single nucleus (or monokaryon), an anchoring disk, a polaroplast at the anterior pole, and a polar filament with 3–4 (tamarin No. 1) or 4–5 (tamarin No. 5) coils in a single row alignment on each side of the spore (Fig. 5). Spores dimensions averaged 2.5 × 1.5 µm.
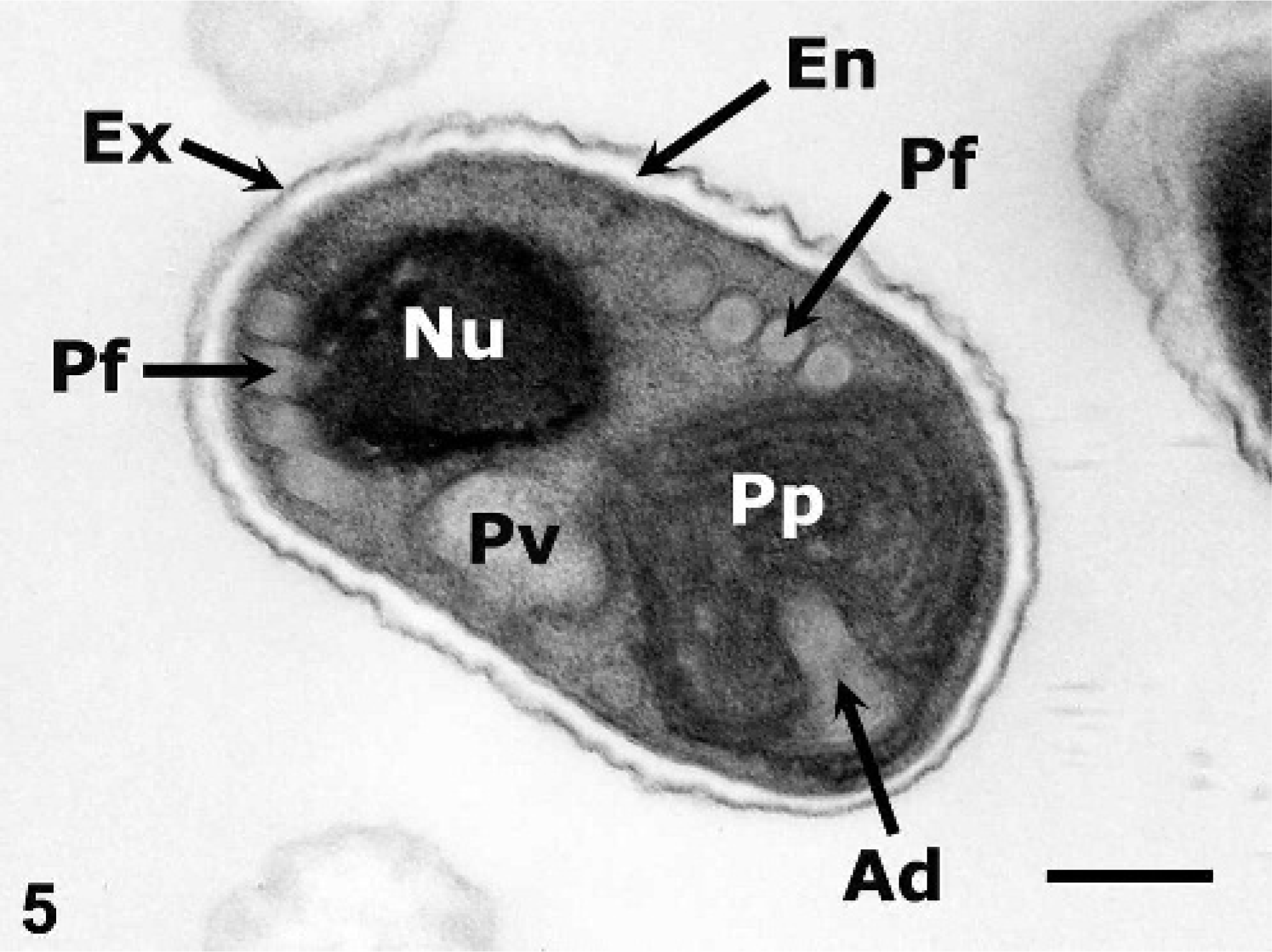
Liver; emperor tamarin No. 5. Notice microsporidial spore with an electron-lucent endospore (En), electron-dense exospore (Ex), moderately electron-dense sporoplasm with an electron-dense nucleus (Nu), polar filament (Pf) with 4 coils in a single row alignment on each side of the spore, anchoring disk (Ad), polaroplast (Pp), and posterior vacuole (Pv). Uranyl acetate and lead citrate. Bar = 0.5 µm.
PCR and DNA sequencing analyses
The DNA was extracted from a pool of lung, brain, and bone marrow from tamarin Nos. 1 and 2, and PCR amplicons of expected size, approximately 300 bp each, 20 were generated. By BLASTn analysis (http://www.ncbi.nlm.nih.gov/BLAST/) and sequence alignment using CLUSTALW (http://clustalw.genome.jp/), the nucleotide sequences of these 2 tamarin microsporidians was identified as E. cuniculi genotype III on the basis of the presence of four 5′-GTTT-3″ repeats. 13 These 2 sequences were identical to each other and were identical to those reported for the E. cuniculi isolate identified in cotton-top tamarins 23 with the assigned GenBank sequence of L29560. 19 Attempts to generate PCR amplicons from paraffin-embedded liver of microsporidia-infected tamarin No. 5 failed.
Serologic testing
The dam of tamarin Nos. 3–5 was seropositive for E. cuniculi, with an ELISA IgG serum titer >1 ° 400,000 against E. cuniculi. By Western blot immunodetection, the serum of the dam expressed the strongest reactivity against E. cuniculi, compared with weak reactivity against E. hellem and E. intestinalis, and virtually no reactivity against V. corneae proteins (Fig. 6). The sire of these tamarins proved to be seronegative for E. cuniculi.
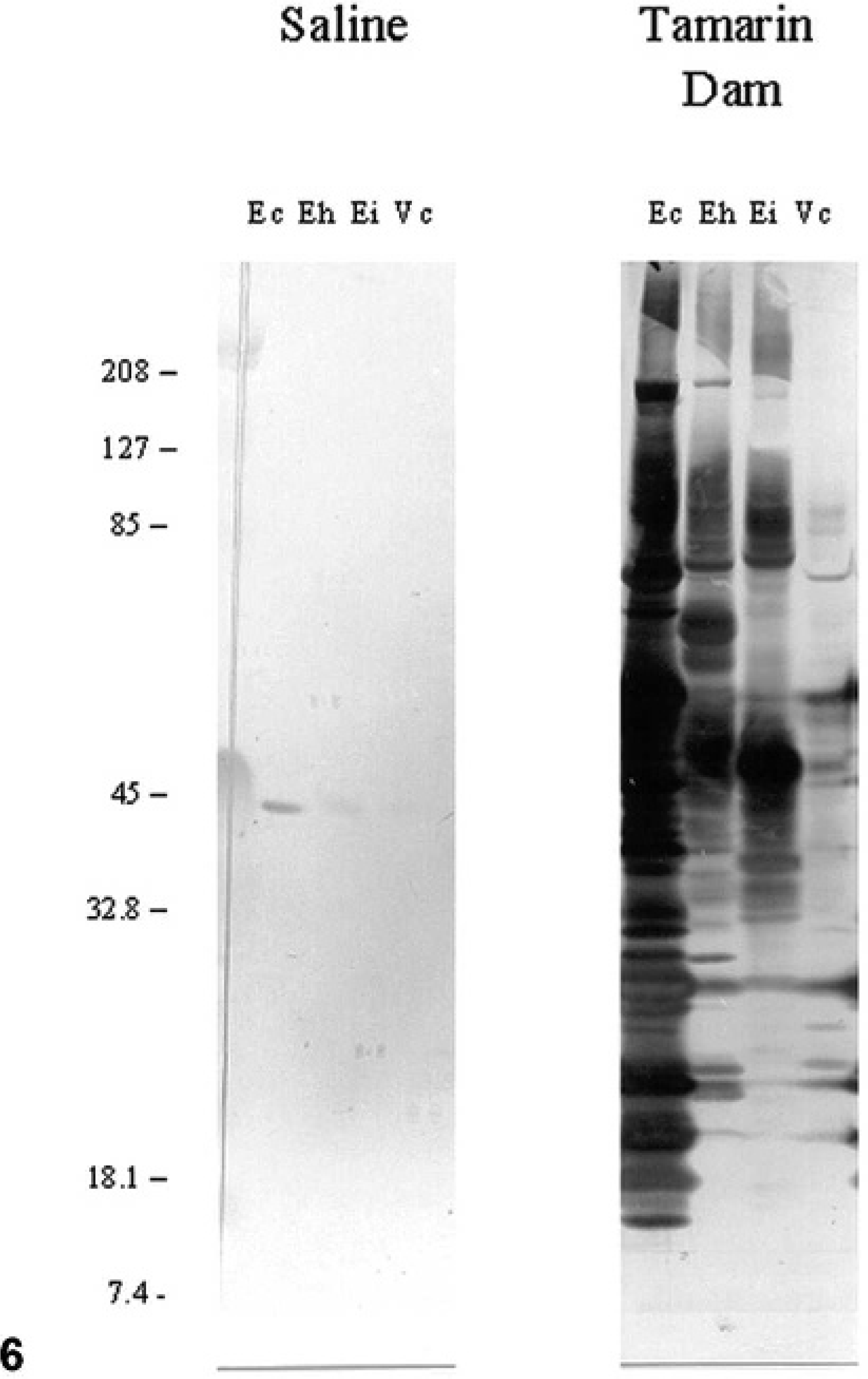
Western blot immunodetection; tamarin dam's serum. Solubilized microsporidial proteins from Encephalitozoon cuniculi (Ec), E. hellem (Eh), E. intestinalis (Ei), and V. corneae (Vc) were exposed to sodium dodecyl sulfate-polyacrylamide gel electrophoresis, blotted onto nylon membranes, and probed with saline or the tamarin dam serum diluted 1 ° 1,000. Blots were then incubated with alkaline phosphatase-conjugated antiserum and stained with nitroblue tetrazolium (NBT) and bromochloroindolyl phosphate (BCIP). Notice the strongest reactivity of tamarin dam serum to E. cuniculi. Protein markers of known molecular mass are noted to the left in kilodaltons.
Discussion
Disseminated encephalitozoonosis was diagnosed in 2 cotton-top and 3 emperor tamarins on the basis of histologic examination, special staining, electron microscopy, serologic testing, or PCR analysis DNA sequencing (or all 5 methods). The ultrastructure of the intralesional microorganisms in tamarin Nos. 1 and 5 was consistent with microsporidia. Spore dimensions and polar filament arrangement were similar to those of microsporidia of the genus Encephalitozoon. The number of polar filament coils ranged from 3 to 5, which is typical of Encephalitozoon species. 31
Similar to previous reports in tamarins, 16, 23 E. cuniculi genotype III (identified originally as a domestic dog isolate) was identified by PCR and DNA sequencing analyses in paraffin-embedded tissues from tamarin Nos. 1 and 2. Results of the PCR assay were negative for tamarin No. 5. However, the dam was seropositive for E. cuniculi on the basis of the ELISA immunoglobulin titer and results of Western blot immunodetection. Therefore, it is likely that tamarin Nos. 3–5 were infected with E. cuniculi as well.
In a previous report of E. cuniculi infection in cotton-top tamarins, 23 pathologic findings consisted mostly of granulomatous hepatitis (both cases) and necrotizing lymphadenitis and splenitis, lesions not seen in the cotton-top tamarins of this report. However, the hepatic lesions were similar to those found in the emperor tamarins included in this study. There was no mention of nervous system lesions in the previous description of encephalitozoonosis in cotton-top tamarins, 23 and this differs from those in the cotton-top tamarins of this report, in which meningoencephalitis was the predominant finding and was accompanied in one of them (tamarin No. 1) by severe tubulointerstitial nephritis. This difference may be attributable to a different course of infection in both colonies because hepatic involvement is usually seen earlier in the course of E. cuniculi infection in other species. 31 The most prominent lesions in the emperor tamarins of this report involved the liver, adrenal glands, and lungs; however, the brain from any of these animals was not available for histologic examination. In the previous report in emperor tamarins, 16 the distribution of lesions was similar to those in the cotton-top tamarins described here (tamarin Nos. 1, 2), except for the vascular involvement, which was not seen in our animals.
The systemic inflammation associated with encephalitozoonosis was considered the main cause of death in tamarin Nos. 1 and 3–5. Although most of the inflammatory lesions in these animals were compatible with encephalitozoonosis and often contained intralesional microsporidia, other diagnostic tests were not carried out to rule out possible concurrent infectious diseases. Although results of aerobic bacterial culture of the spleen were negative for pathogenic bacteria, death was attributed to possible concurrent septicemia in tamarin No. 2 (Table 2) on the basis of the presence of systemic thrombosis (disseminated intravascular coagulation), acute necrosuppurative hepatitis and fibrinosuppurative splenitis, acute interstitial pneumonia, and acute adrenocortical hemorrhage and splenic congestion. However, the possible role of microsporidia in these lesions cannot be ruled out.
In tamarin No. 2, encephalitis was less pronounced than in its sibling (tamarin No. 1); although other tissues had lesions compatible with encephalitozoonosis (especially the skeletal muscle, myocardium, and retina); infection was more widespread in tamarin No. 1. In concordance with this finding, microsporidia were detected by PCR analysis in tamarin No. 2, but not by routine histologic staining and special staining of touch imprints obtained from the brain at necropsy. Unlike tamarin No. 2, tamarin No. 1 had numerous microsporidia visible in HE-stained sections of the brain. Microsporidia were readily visible with HE and special stains in a number of tissues from the emperor tamarins.
Microsporidia are ubiquitous in the environment, and transmission of infection is believed to occur principally through ingestion or inhalation of spores. 3, 7, 11 Transplacental (vertical) transmission in carnivores, lagomorphs, equines, and nonhuman primates also has been reported. 10, 27– 29, 32 As with previous reports in callitrichids and squirrel monkeys, 16, 23, 27, 32 encephalitozoonosis was observed in newborn, neonatal, and juvenile tamarins, whereas adults housed with affected animals did not manifest clinical signs of disease. However, the breeding female which gave birth to tamarin Nos. 3–5 was seropositive for E. cuniculi, which suggested the likelihood of vertical transmission in the emperor tamarins. This was also deemed likely in previous reports of squirrel monkeys and emperor tamarins. 16, 32 The cotton-top tamarin breeding pair for tamarin Nos. 1 and 2 had a history of high neonatal (4 animals) and juvenile (2 animals) mortality of its offspring between 2000 and 2002; a review of the pathologic findings and HE-stained tissue sections from these 6 tamarins failed to reveal lesions or microorganisms consistent with microsporidiosis.
Encephalitozoonosis seems to be an emerging microsporidial disease in captive-bred callitrichids that is associated with appreciable neonatal and juvenile mortality in some colonies in North America and Europe. On the basis of the possibility of vertical transmission, testing of breeding pairs seems to be advisable at this time for captive breeding programs existing for these valuable nonhuman primate species. Testing should include serologic analysis, and calcofluor white staining and PCR analysis of urine.
Footnotes
Acknowledgements
We would like to acknowledge funding through grants RR00164 and AI039968 (ESD, LCB) from the National Institutes of Health, Bethesda, MD.
