Abstract
A great horned owl (Bubo virginianus) was admitted to a rehabilitation clinic with severe neurologic signs that were unresponsive to supportive care. The animal was euthanatized because of a poor prognosis. Marked granulomatous encephalitis with focal brainstem malacia was detected microscopically. The brainstem was the most severely affected brain location and the only place in which schizonts and merozoites, morphologically compatible with Sarcocystis spp., were detected. Immunohistochemistry with the use of polyclonal antisera indicated the presence of Sarcocystis falcatula. The species identification of the protozoa as S. falcatula was confirmed by polymerase chain reaction. To the author's knowledge, this is the first report of spontaneous S. falcatula–associated encephalitis in a great horned owl.
An adult, female great horned owl (Bubo virginianus) was admitted to The Raptor Center (University of Minnesota, St. Paul, MN). 11 Found in southern Minnesota, the owl had been observed for 2 days, during which it was unable to fly. The animal was in a good nutritional state, weighing 1.54 kg. On presentation, the owl was standing, alert, and responsive but had a severe head tilt to the left (Fig. 1). Physical examination revealed a small corneal scar but no signs of trauma or indications of otitis externa. Whole-body radiographs (ventrodorsal and lateral views) demonstrated spleen enlargement and slight displacement of internal organs. Complete blood count revealed an elevated total white cell count (20,000 cells/μl; reference [ref.] range: 6,000–8,000 cells/μl] with heterophilia (14,600 heterophils/μl, 73%; ref. range: 47%) and normal total solids. 14 Biochemistry results were within reference ranges. Initial treatment consisted of parenteral application of nonsteroidal anti-inflammatory drugs (0.2 mg/kg PO [by mouth] SID [once a day] meloxicam a ), systemic application of antibiotics (10 mg/kg enrofloxacin b PO BID [twice a day]), parenteral application of blood ammonia reducer (0.4 mg/kg PO Lactulose c ), and supportive care. The owl was euthanatized 5 days after admission because of poor prognosis.
A complete necropsy was performed at the Minnesota Veterinary Diagnostic Laboratory (University of Minnesota, St. Paul, MN) within 2 hr of death. Various tissues, including brain, spinal cord, skeletal muscle, heart, liver, kidney, lungs, spleen, tibial bone marrow, intestine, proventriculus, adrenal glands, eyes, sciatic nerve, and the skull with inner and middle ears, were fixed in neutral buffered 10% formalin solution for histological examination, paraffin embedded, sectioned at 5 μm, and routinely stained with hematoxylin and eosin. A peroxidase-based polymer system d was used for immunohistochemical detection of protozoal antigens. Polyclonal antisera raised against culture-derived merozoites/tachyzoites of Sarcocystis falcatula, Sarcocystis neurona, Toxoplasma gondii, and Neospora caninum, as well as a monoclonal antibody (mAb) against S. neurona, were used in brain sections as previously described. 10 In addition, a mAb specific for the envelope protein of West Nile virus was used in brain, kidney, and heart as previously described. 23 Blocks of paraffin-embedded brain tissue were used for detection of protozoal nucleic acid as previously described. 10 Briefly, 2 μl of eluted DNA from extracted tissue were used in the first round of a nested 50-μl PCR reaction with apicomplexan panspecific primers targeting the first internal transcribed spacer (ITS-1) region. The PCR reactions were carried out in a commercial thermocycler e under the following reaction conditions: 5.0 μl 10× PCR buffer with MgCl2 (15 mmol), 200 μmol of each deoxyribonucleotide triphosphate (dNTP), 1 μmol of each primer: (ITS1DF, 5′-TACCGATTGAGTGTTCCGGTG-3′; ITS1DR, 5′-GCAATTCACATTGCGTTTCGC-3′), and 1.5 U Taq polymerase. After initial denaturation of templates and primers (94°C, 3 min), 35 cycles of the following conditions were used: 95°C for 40 sec, 58°C for 40 sec, 72°C for 90 sec, followed by a 5-min extension at 72°C. In the second round of the nested reaction, 1 μl of product DNA from the first round was used as the DNA template, and internal primers (ITS1diF, 5′-CGTAACAAGGTTTCCGTAGG-3′; ITS1-diR, 5′-TTCATCGTTGCGCGAGCCAAG-3′) were used. Reaction times and conditions were identical to the first round. Ten microliters of PCR product was electrophoretically separated on a 2% agarose gel stained with ethidium bromide and visualized under ultraviolet light. The PCR products from positive bands were cleaned using the ExoSAP-IT PCR clean-up system f and sequenced at the Division of Biological Sciences DNA sequencing facility (University of California, Davis, CA). A BLAST search (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) was used to compare the resulting sequences with similar sequences available in GenBank.

Great horned owl with Sarcocystis falcatula–associated encephalitis exhibiting severe head tilt to the left.
Brainstem, great horned owl. Focally extensive malacia is present near a necrotic capillary (arrow). The brain parenchyma in the periphery of the malacia was infiltrated by gitter cells and activated astrocytes. Hematoxylin and eosin. Bar = 100 μm.
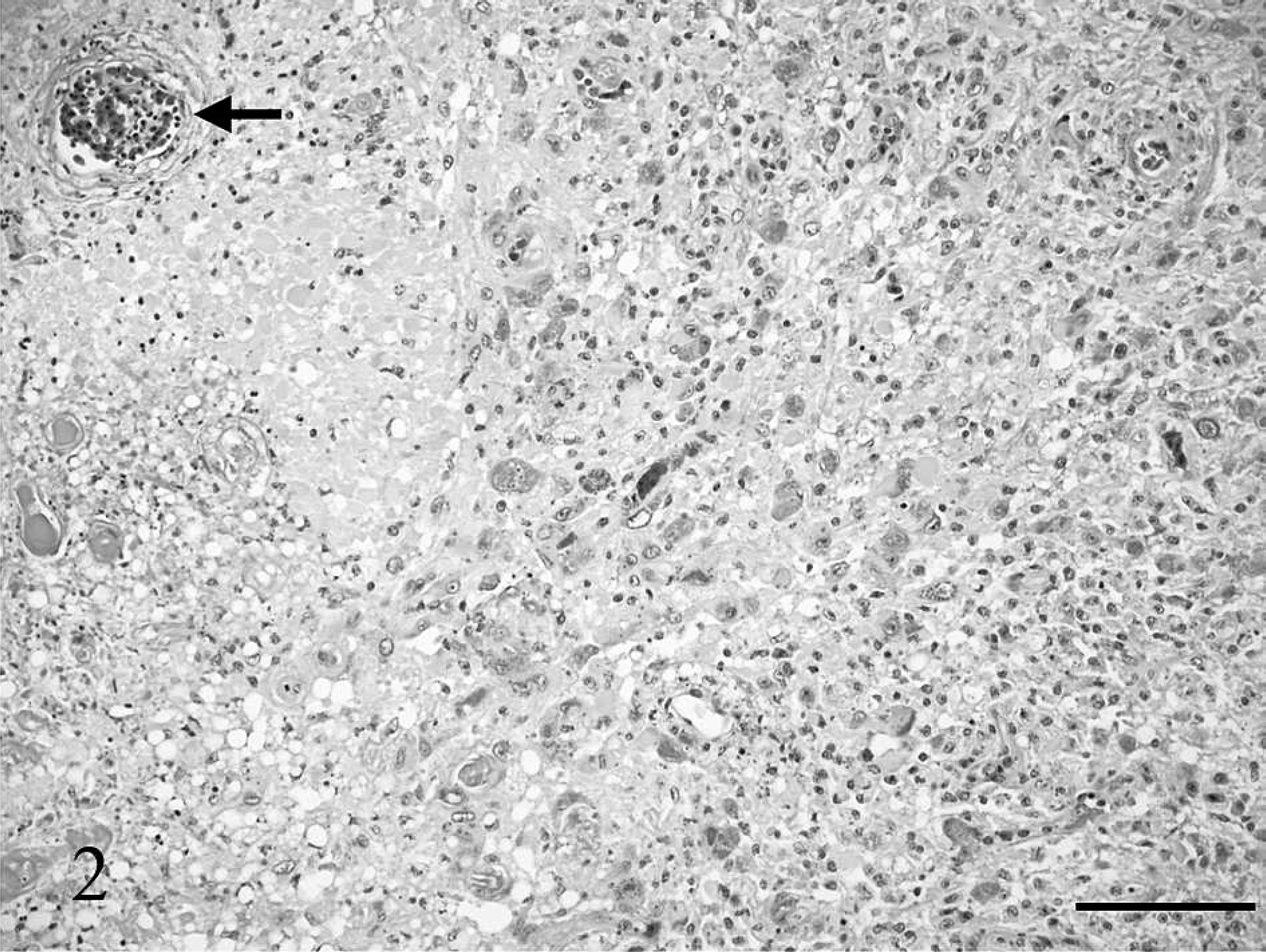
Brainstem, great horned owl. Several protozoal schizonts are present in the neuropil in various stages of development. Large immature schizonts filling the cytoplasm of unidentified cells within the neuropil have large pleomorphic basophilic nuclei (black arrowheads). A single mature schizont consists of a cluster of randomly arranged merozoites (white arrow). An additional intermediate stage schizont consists of a large central nucleus surrounded by peripheral budding merozoites (black arrow). The central nucleus has several small irregular lobular projections, some of which are continuous with the peripheral merozoite nuclei. Occasional free extracellular and intracellular merozoites are present (white arrowheads). Hematoxylin and eosin. Bar = 20 μm.
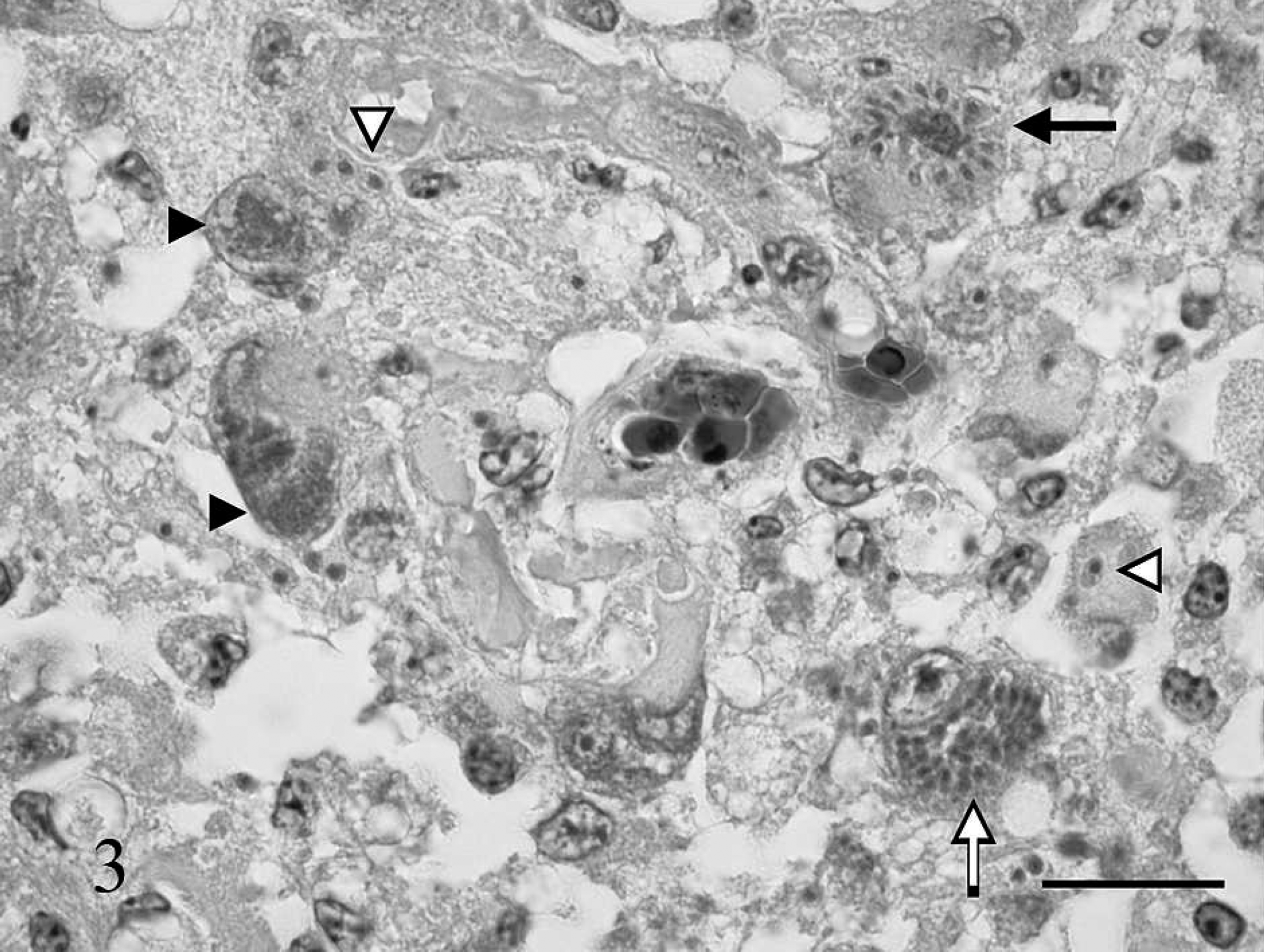
Brainstem, great horned owl. A small vessel with hypertrophied endothelial cells (outlined by arrowheads) has a large well-developed protozoal schizont (arrow) consisting of a randomly arranged cluster of merozoites lying within an endothelial cell. Hematoxylin and eosin. Bar = 25 μm.
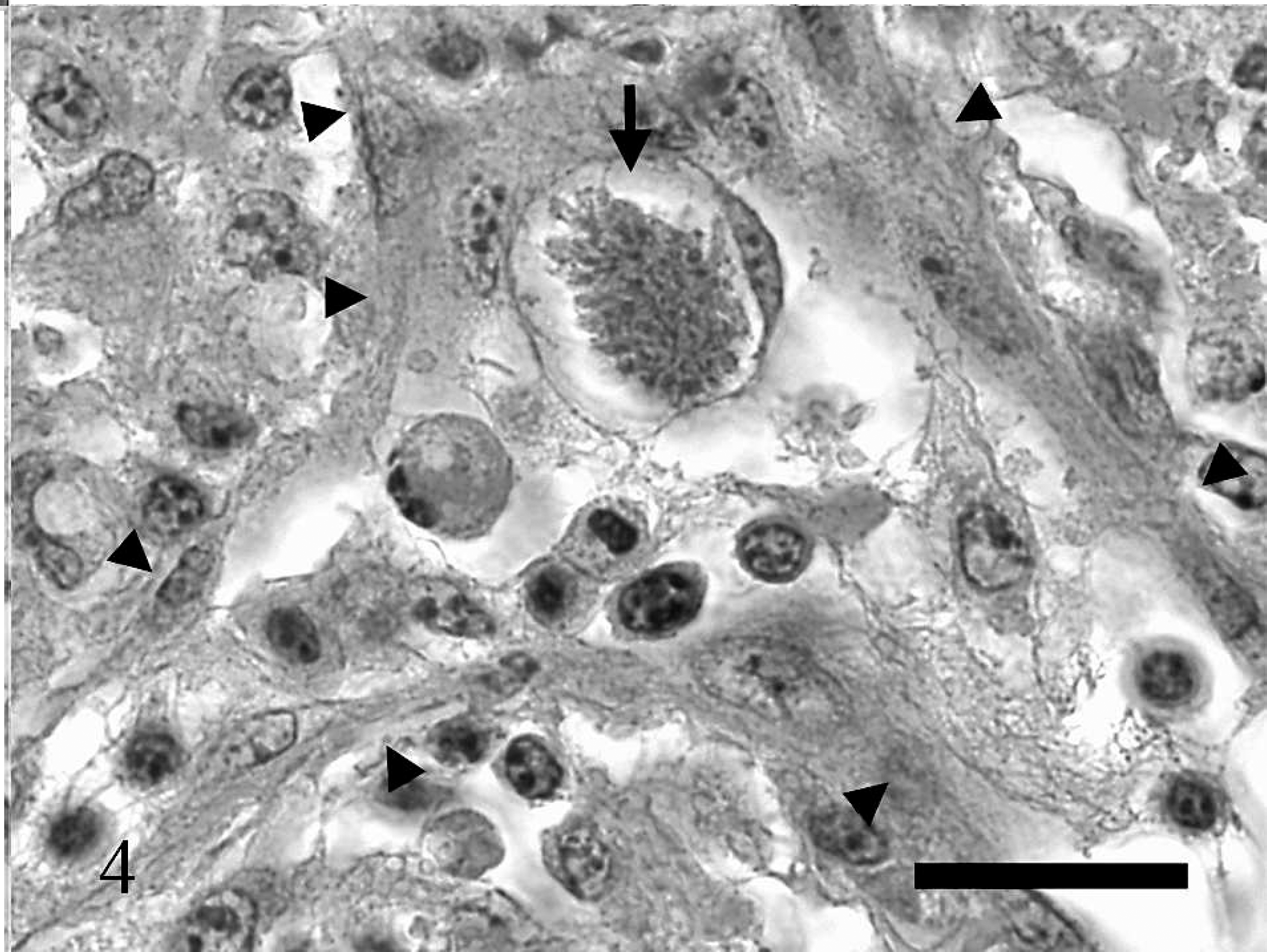
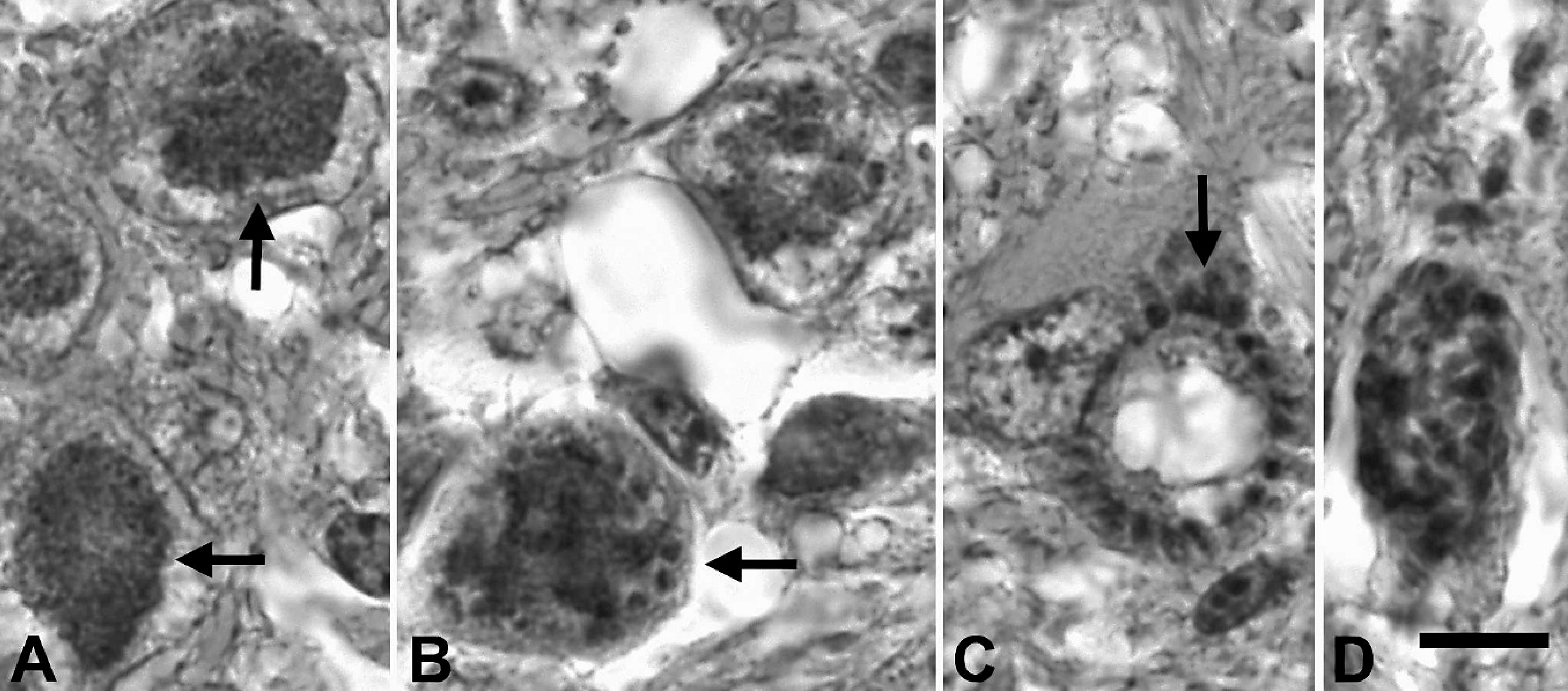
During the postmortem of the owl, significant macroscopic lesions were not detected. The spleen was considered to be of normal size. Histological examination revealed a marked granulomatous meningoencephalitis affecting cerebrum, cerebellum, and brainstem. There was a large focal area of malacia in the ventral midbrainstem with fibrinoid vascular necrosis in the center, infiltration by gitter cells and activated astrocytes in the periphery (Fig. 2). Neurons, cells of the neuropil not further identified, and endothelial cells adjacent to the area of malacia contained numerous protozoal schizonts (Figs. 3, 4). Numerous large immature schizonts with large, fine granular basophilic nuclei were present within neurons, glial cells, or both within the neuropil (Fig. 5A). Some of these large schizont nuclei were irregular in contour with large lobulations, and in others, variable numbers of small budding merozoites extended out peripheral to the large central protozoal schizont nuclei (Fig. 5B). In rare instances, multiple, small, peripherally budding merozoites were present that were continuous with the larger central nuclei compatible with endopolygeny (Figs. 3 [arrow], 5C). Large clusters of well-developed merozoites were also present that consisted either of randomly arranged merozoite clusters or peripherally extending rows of merozoite rosettes organized around central pale-staining residual bodies (Fig. 5C, D). Isolated individual merozoites also were visible within the cytoplasm of large neurons. In addition, individual extracellular merozoites were present within the neuropil (Fig. 5D). By light microscopy, individual merozoites were estimated to be approximately 1.5–2.0 μm × 5–6 μm in size. The combined histologic features were compatible with a Sarcocystis species. 6,8 The leptomeninges had few prominent nodular inflammatory infiltrates that largely consisted of lymphocytes and macrophages. Lymphocytes and plasma cells of the neuropil infiltrated moderately along the ventral commissures of the lateral ventricles and along the meninges covering the dorsal aspect of the thalamus. Protozoal parasites were not detected in this portion of the brain. Numerous capillaries in this region were surrounded by multiple layers of fibrillar astrocytes. Occasional blood vessels had perivascular infiltrates composed of lymphocytes, plasma cells, and macrophages containing brownish pigment interpreted as siderophages. A moderate lymphocytic inflammation of the left vestibulocochlear ganglion and a moderate lymphocytic ganglionitis of spinal ganglia were present. Among several sections of skeletal muscle examined, 2 parasitic structures were found, one of which resembled a large, not further identifiable, protozoal megaloschizont, the second possibly representing a degenerate protozoal tissue cyst approximately 31 μm in diameter that had a thick smooth peripheral wall. Heterophilic, lymphocytic, and histiocytic endocarditis and myocarditis, iridocyclitis, and neuritis (sciatic nerve) were mild, and the femoral bone marrow showed granulomatous inflammation. Protozoal organisms were not detected in these tissues.
Brainstem, great horned owl. Various stages of protozoal schizogony are present.
By immunohistochemistry (IHC), merozoites and schizonts in the brain were limited to the area of malacia and reacted strongly positive to polyclonal antiserum raised against S. falcatula merozoites (Fig. 6). Reaction to polyclonal S. neurona antiserum was variably negative to weakly positive. The parasites did not show any immunoreactivity with polyclonal antisera to T. gondii and N. caninum or to the mAb directed against S. neurona. The presumptive protozoal muscle cyst did not react to any of the anti-protozoal antisera or S. neurona antigen–specific mAb by IHC. West Nile virus antigen was not detected in sections of brainstem, cerebellum, heart, or kidney by IHC.
The PCR analysis revealed an amplification product consistent with the predicted product of a Sarcocystis sp. Subsequent sequencing results were consistent with S. falcatula and differed from S. neurona. The resulting ITS-1 sequence (GenBank EU937517) was 99% similar (681 of 684 bp and 679 of 684 bp) to several published S. falcatula ITS-1 sequences (AF098242 and AF098244). The sequence was only 96% similar (660 of 681 bp) to the most closely related S. neurona ITS-1 sequences in GenBank (AY009113 and AF252407) and only 91% (634 of 692 bp) similar to an Sarcocystis lindsayi ITS-1 sequence (AF387164).
To the authors' knowledge, this is the first report documenting naturally acquired S. falcatula infection of any type in a free-ranging great horned owl. The combined IHC reactions were diagnostic for a S. falcatula–like organism. The pattern of reaction to the various polyclonal antisera and S. neurona mAb were identical to the previously reported reactivity of these reagents to S. falcatula in experimentally infected psittacines, with the exception of a small subset of brain merozoites that also reacted weakly with the S. neurona antigen–specific polyclonal antiserum. 5 However, although IHC reactivity with these polyclonal antisera are useful diagnostic reagents for the routine diagnosis of well-recognized protozoal diseases in known susceptible intermediate hosts, they are not sufficient by themselves to confirm the identity of protozoal species, particularly in tissues of newly identified host species. For instance, a previous study has shown that S. lindsayi schizonts will also react positively with S. falcatula antigen–specific polyclonal antisera. 7 Species identification thus requires additional testing, such as PCR to determine the genetic identity or electron microscopy to possibly identify specific ultrastructural features unique only to the particular organism. The PCR results in this case identified the brain protozoa as S. falcatula.
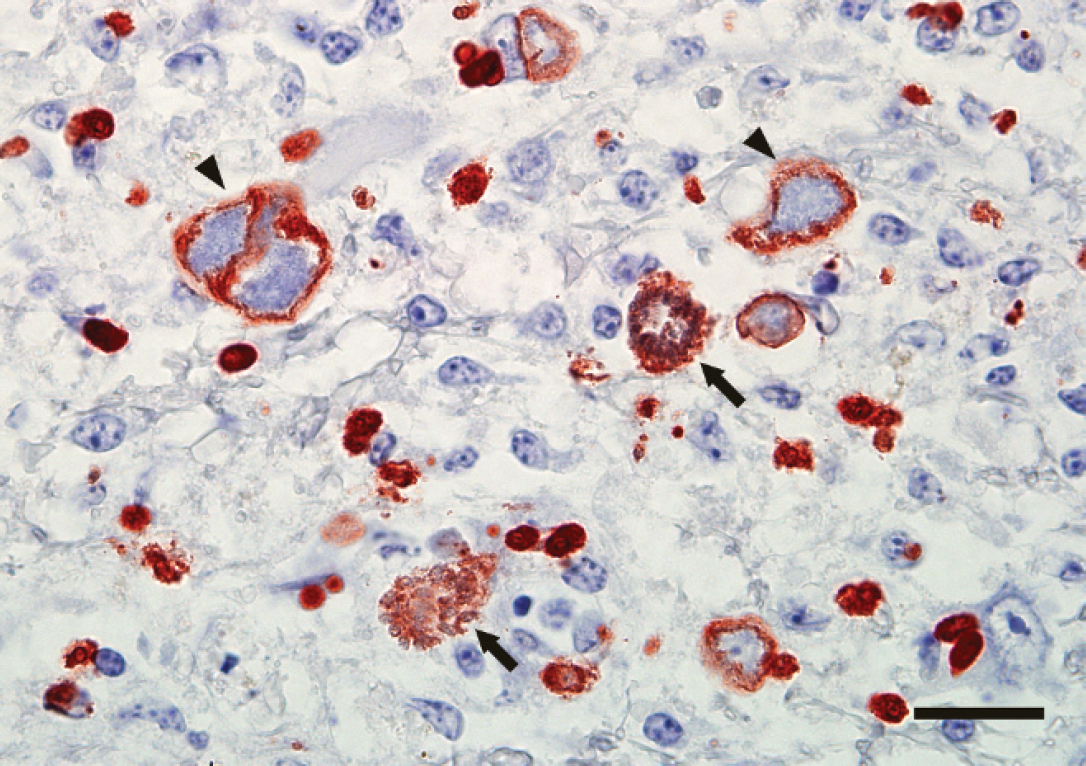
Brainstem, great horned owl. Protozoal schizonts react strongly positive with a Sarcocystis falcatula polyclonal antiserum. Numerous immature schizonts with large, centrally located, negative staining nuclei are surrounded by fine, granular, positively reacting protozoal cytoplasm (arrowheads), as well as additional smaller, densely staining merozoites or very early forming schizonts within unidentified cells in the neuropil. Two mature schizonts composed of clusters of formed merozoites are also present (arrows). Envision™-HRP (horseradish peroxidase), S. falcatula polyclonal antiserum. Bar = 25 μm.
This is the first report of confirmed S. falcatula–associated encephalitis with direct protozoal infection within the central nervous system, including intraneuronal infection, in any susceptible avian host species, although there have been isolated reports of presumptive S. falcatula encephalitis in psittacines based on fluorescent antibody testing, serology, and protozoal histomorphology or S. falcatula–like encephalitis based solely on protozoal histomorphology. 12,17,22 Furthermore, before this report, avian species susceptible to S. falcatula infection have only been reported within the 3 phylogenetic orders Psittaciformes, Passeriformes, and Columbiformes. 1 3,5,13 Although numerous American passerine and columbiforme birds are thought to be susceptible to S. falcatula infection, grackles and cowbirds likely serve as major intermediate hosts for this parasite. 22 Sarcocystis falcatula infection often causes severe disease and death in Old World psittacine and columbiforme birds, whereas the infection usually runs a subclinical course in New World birds. The current findings indicate that at least 1 avian species within the order Strigiformes also might serve as a susceptible intermediate host. It is uncertain whether the great horned owl is a true or aberrant intermediate host because definitive S. falcatula protozoal muscle cysts were not identified in this bird.
The North American opossum (Didelphis virginiana) and South American opossum (Didelphis albiventris) are the definitive hosts for at least 3 Sarcocystis spp. (S. falcatula, S. neurona, and S. lindsayi) known to infect avians, although S. lindsayi has only been documented in South American opossums. 1 4,7,15
Sarcocystis spp. have a life cycle that includes a carnivorous final host and a prey animal that serves as intermediate host. Sexual replication (gametogony) occurs in the intestinal mucosa of the definitive host (opossum) that sheds sporulated sporocysts in the feces. Asexual replication (merogony) occurs within tissues of the intermediate hosts, typically within endothelial cells, circulating leukocytes, and finally striated muscle cells. The definitive host becomes infected by ingesting mature sarcocysts in muscles of latently infected intermediate hosts. Although significant clinical disease is virtually unknown in the definitive host, clinical disease can occur in intermediate hosts and is associated with tissue destruction resulting from rapid asexual multiplication or schizogony after initial oral infection. A fulminant interstitial pneumonia resulting from rupture of protozoa-infected pulmonary endothelial cells is the most common form of clinical disease seen in psittacines after ingestion of S. falcatula sporozoites. 19 21 After endothelial proliferation, the protozoa invade and multiply in muscles, where latent tissue cysts are eventually formed. 5,18 20
Because great horned owls are birds of prey, it is possible that transmission of S. falcatula infection occurred either by ingestion of sporulated sporocysts, similar to other reported intermediate hosts, or by ingestion of the muscle sarcocyst stages. The diet of great horned owls in North America include columbiforme and passeriforme birds that potentially contain S. falcatula sarcocysts in their striated muscles, as well as opossums that might harbor intestinal sporulated sporozoites (Kittredge VC, Wilson PW, Caire W: 2006, An updated checklist of the food items of the great-horned owl [Bubo virginianus: Strigiformes: Strigidae] in Oklahoma. Proceedings of the Oklahoma Academy of Science 86:33 38. Available at http://digital.library.okstate.edu/oas/oas_htm_files/v86/. Accessed on October 1, 2008). Approximately 48% of North American opossums caught in multiple U.S. states contained sporocysts compatible with S. falcatula, with 33% of opossums in middle Michigan harboring S. falcatula oocysts in the feces. 4,9 Direct horizontal transmission via ingestion of muscle cysts would be unique among Sarcocystis species, mimicking the direct asexual horizontal transmission between successive intermediate hosts that occurs with T. gondii. 16
The cause of mild heterophilic, lymphocytic, and histiocytic endocarditis and myocarditis, iridocyclitis, and neuritis (sciatic nerve) and granulomatous inflammation of the femoral bone marrow was not apparent. Mild inflammation of the heart and mild iridocyclitis are a common finding in free-ranging owls in the authors' experience. None of these extraneural and extramuscular lesions might be related to the S. falcatula infection. Sarcocystis falcatula should be considered a differential diagnoses for neurologic disease in great horned owls.
Acknowledgements. The authors thank Dr. Erik Olson for his help in manuscript preparation and the California Animal Health and Food Safety Laboratory System (CAHFS) histology personnel for their excellent assistance with immunohistochemistry.
Footnotes
a.
Metacam®, Boehringer Ingelheim Vetmedica Inc., St. Joseph, MO.
b.
Baytril®, Bayer Healthcare L.L.C. Animal Health Division, Shawnee, KS.
c.
Enulose®, Actavis Mid Atlantic LLC, Columbia, MD.
d.
Envision™-HRP, DAKO North America Inc., Carpinteria, CA.
e.
GeneAmp® 2400, Applied Biosystems, Foster City, CA.
f.
ExoSAP-IT PCR clean-up system, USB Corp., Cleveland, OH.
