Abstract
A 7-year-old female buffalo (Bubalus bubalis) from a local herd in Serres, northern Greece, was presented to a private veterinary clinic with a chronic loss of appetite for 15 days. The clinical examination revealed high fever (41.5°C), lethargy, yellow discoloration of skin and mucous membranes, an abdomen that appeared to be empty, hyperactive rumen motility, and tachypnea. A biochemical profile revealed an elevated total bilirubin concentration and hepatic enzyme activities, whereas globulin, creatinine, and glucose concentrations were within the reference intervals. The animal received a 12-day course of treatment with intramuscular administration of ampicillin and corticosteroids. However, no significant clinical improvement was achieved, and the buffalo was euthanized. Gross necropsy lesions included serous atrophy of adipose tissue and hepatomegaly. Microscopic lesions included necrotizing pyogranulomatous hepatitis with thrombosis, hemorrhages, edema, and fibrosis. Small, nonpigmented, bacterial colonies were harvested in pure culture from the liver and were confirmed as Stenotrophomonas maltophilia by polymerase chain reaction. The bacterium was sensitive to ciprofloxacin, enrofloxacin, colistin, polymyxin, trimethoprim/sulfamethaxazole, and chloramphenicol. In contrast, resistance to ticarcillin, piperacillin, imipenem, ceftazidime, amikacin, gentamicin, tobramycin, and tetracycline was displayed. The bacterial strain carried the L1 metallo-β-lactamase (L1) and tet35 genes, which contribute to high-level resistance to β-lactams and tetracycline, respectively. Although S. maltophilia is widely believed to be a contaminant, the present report suggests that the isolation, identification, and susceptibility testing of this multidrug-resistant bacterium may be of clinical importance in diagnostic samples.
Stenotrophomonas maltophilia (order Xanthomonadales, family Xanthomonadaceae) is a multidrug-resistant, ubiquitous bacterium that is considered a nosocomial pathogen in human medicine. Clinical manifestations of bacterial infection, mainly in immunosuppressed humans, include pneumonia, bacteremia, meningitis, and urinary tract infections. 5 In animals, this bacterium has been isolated as an opportunistic pathogen from squirrel feces, porcine semen, oral flora of snakes, and fleece rot in sheep. 1 Despite doubts about its pathogenicity, the bacterium has also been implicated in chronic respiratory disease among horses, dogs, and cats 1 ; lymphadenitis in goats 8 ; and acute septicemia in a crocodile. 7 The clinical, laboratory, gross, and microscopic findings that are indicative of pyogranulomatous hepatitis caused by S. maltophilia in a buffalo are reported in the current study.
A 7-year-old female buffalo (Bubalus bubalis) from a local herd in Serres, northern Greece, was presented to a private veterinary clinic with a chronic loss of appetite for 15 days. The clinical examination revealed high fever (41.5°C), lethargy, yellow discoloration of the skin and mucous membranes, an empty-appearing abdomen, hyperactive rumen motility, and tachypnea. A dipstick screening for proteinuria, hematuria, and ketone bodies in the urine, which was performed as previously described, 2 was negative. Microscopic examination of Giemsa-stained, thin and thick blood films did not detect any blood parasites. A biochemical indicated that globulin (3.2 g/dl, reference [ref.] interval: 2.4–4.4 g/dl), creatinine (1.1 mg/dl, ref. interval: 0.9–1.8 mg/dl), and glucose (102 mg/dl, ref. interval: 75–117 mg/dl) concentrations were within reference intervals. However, total bilirubin concentration (4.7 mg/dl, ref. interval: 0.8–2.2 mg/dl) and hepatic enzyme activities (sorbitol dehydrogenase 287 IU/l, ref. interval: 2–11 IU/l; gamma-glutamyl transferase 412 IU/l, ref. interval: 8–33 IU/l) were markedly increased.
Serological tests for Leishmania donovani a and Leptospira spp. b were negative, while the Rose Bengal test, c which is used for the diagnosis of infection with smooth Brucella species, did not detect the presence of anti-Brucella antibodies in the serum. The animal was treated intramuscularly with 10 mg/kg of ampicillin d and 0.5 mg/kg of corticosteroids e twice a day for a 12-day period. However, no remarkable clinical improvement was achieved, and the buffalo was euthanized.
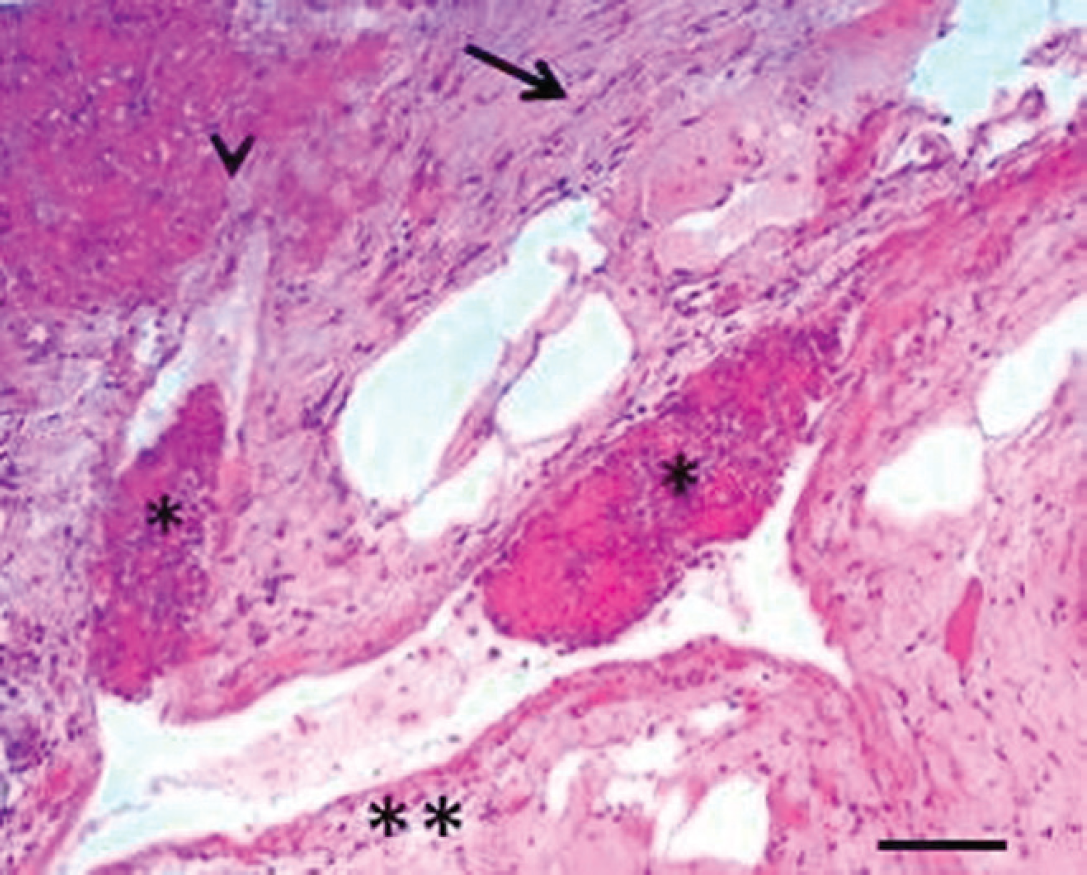
Liver; buffalo (Bubalus bubalis). Lesions of edema (double asterisk), loss of hepatocytes (arrowhead), fibrosis (arrow), and capillary thrombosis (asterisks). Hematoxylin and eosin. Bar = 100 μm.
A necropsy was performed at 1 hr postmortem. Gross lesions included serous atrophy of adipose tissue and hepatomegaly. Parasitological examination of the liver was negative for parasites. Specimens of the liver were cultured on Columbia agar plus 5% sheep blood f aerobically and in an atmosphere that contained 5% CO2 at 37°C. After 48 hr of incubation, small, nonpigmented, bacterial colonies were harvested in pure culture. Bacteria were visible as Gramnegative rods and identified as S. maltophilia with a commercial identification system. f Meanwhile, specimens of the liver cultured on Sabouraud agar g aerobically at 37°C did not yield any fungal colonies. The S. maltophilia isolate was further analyzed by polymerase chain reaction (PCR). Primer sequences were SM1 5′-CAGCCTGC-GAAAAGTA-3′ and SM4 5′-TTAAGCTTGCCACGAACAG-3′, as previously described. 13 The 531-bp amplified product was sequenced on a genetic analyzer h by using dRodamine-labeled terminators. The nucleotide sequences were aligned and revealed 97% homology with S. maltophilia. Minimum inhibitory concentrations were determined by broth microdilution using custom susceptibility plates i according to Clinical and Laboratory Standards Institute guidelines and with breakpoints of Pseudomonas spp. 3 The isolate was sensitive to quinolones (ciprofloxacin and enrofloxacin), colistin, polymyxin, chloramphenicol, and trimethoprim/sulfamethaxazole. Trimethoprim/sulfamethaxazole is the drug of choice for S. maltophilia infections, although resistance mediated by sul genes is emerging worldwide. 12 In contrast, resistance to penicillins (ticarcillin and piperacillin), aminoglycosides (gentamicin, tobramycin, and amikacin), cephalosporins (ceftazidime), β-lactams (imipenem), and tetracycline was displayed. To determine which antibiotic-resistance genes were present in the isolate, DNA samples were analyzed by PCR. The strain was found to carry the L1 metallo-β-lactamase gene (L1) and the tet35 gene, which encode resistance to β-lactams 6 and tetracycline, 11 respectively. The L1 gene was amplified with forward primer 5′-CACACCTGGCAGATCGGCAC-3′ and reverse primer 5′-GCCGCATCCGCGAGGC-3′, while the tet35 gene was amplified with forward primer 5′-GTAATTCTGAGCACTGTCGC-3′ and reverse primer 5′-CTGCCT GGACAAAATTGCTT-3′. Resistance of S. maltophilia to aminoglycosides has been previously described and found to be mediated by enzymes that reduce the uptake of these antibiotics into the cytoplasm. 10 Finally, poor diffusion of penicillins and cephalosporins across the bacterial cell wall has been proposed as the mechanism of S. maltophilia resistance to these compounds. 4
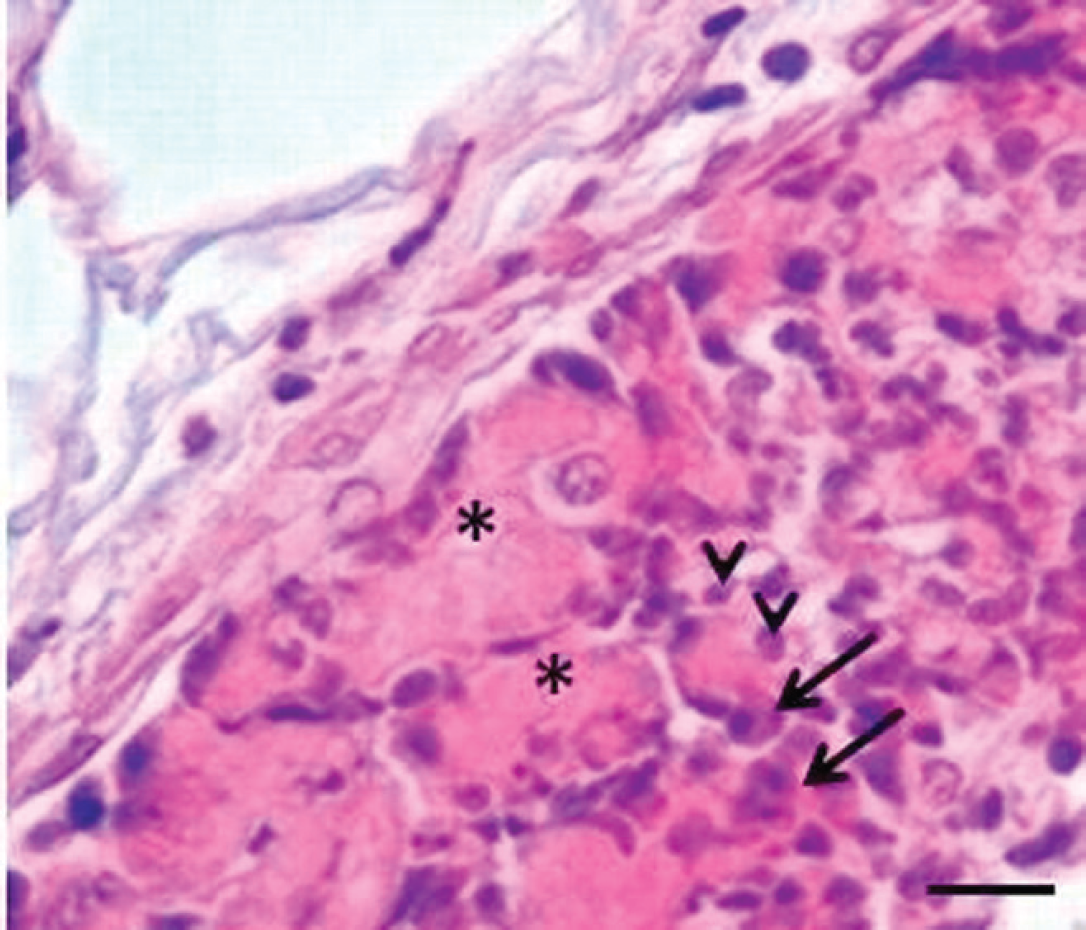
Liver; buffalo (Bubalus bubalis). Thrombi consist of fibrinous material (asterisks), neutrophils (arrowheads), and plasma cells (arrows). Hematoxylin and eosin. Bar = 25 μm.
Specimens of the liver were fixed in 10% neutral buffered formalin. Microscopic examination of hematoxylin and eosin–stained liver sections revealed edema, loss of hepatocytes, fibrosis, and capillary thrombosis (Fig. 1). Thrombi consisted of neutrophils, plasma cells, and fibrinous material (Fig. 2). In addition, hemorrhages and foci of coagulative necrosis with infiltrates of neutrophils, macrophages, and plasma cells were present around the centrilobular vein (Fig. 3).
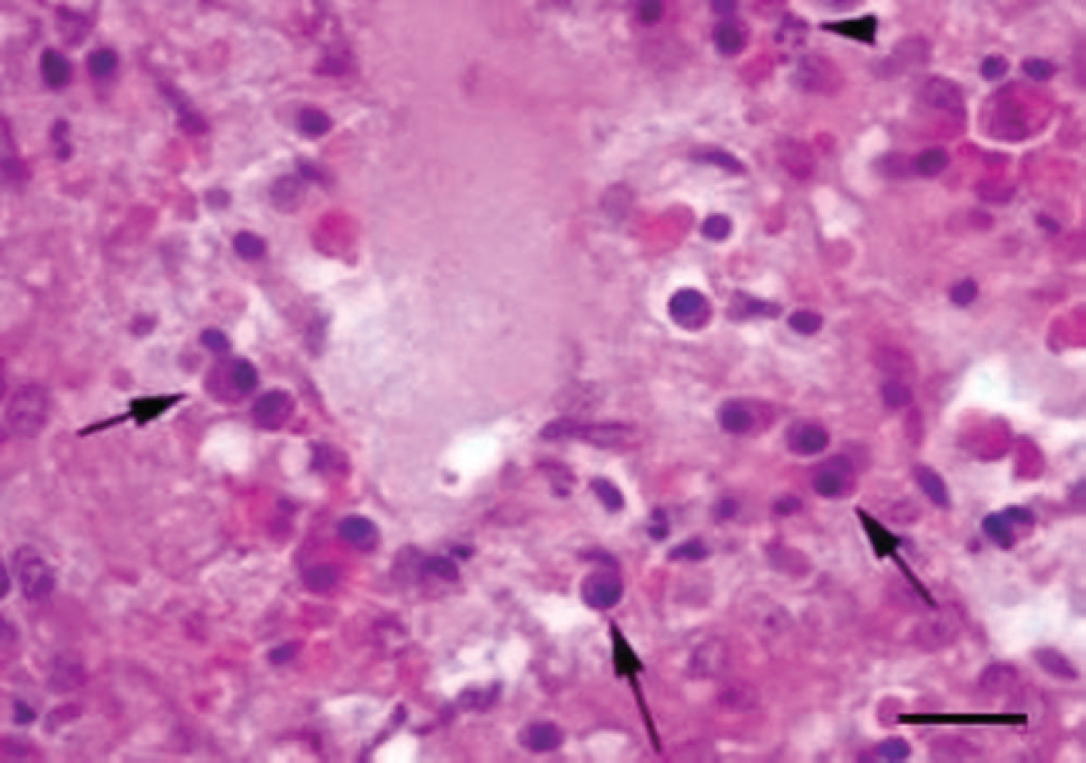
Liver; buffalo (Bubalus bubalis). Hemorrhages and foci of coagulative necrosis with infiltrates of neutrophils (arrowhead) and plasma cells (arrows) around the centrilobular vein. Hematoxylin and eosin. Bar = 25 μm.
The importance of S. maltophilia in veterinary specimens is not easy to pinpoint because it is difficult to distinguish primary infection from contamination. A previous study 5 established that the isolation of S. maltophilia from normally sterile sites is an important finding. The associated infection is mainly based on the elaboration of extracellular enzymes such as DNase, RNase, fibrinolysin, lipases, hyaluronidase, protease, and elastase. In contrast, in a later report, 9 S. maltophilia was isolated from the organs of healthy animals. According to results from the current study, S. maltophilia was the causative agent of hepatic disease in the buffalo and not a contaminant of the liver. This statement is supported in 2 ways. First, it was proved that the animal was suffering from liver disease, as observed by elevations in total bilirubin concentration and hepatic enzyme activities. Second, the results of histopathologic examination and bacteriologic culture indicated that the liver disease was associated with S. maltophilia in pure culture. Thromboembolism was undoubtedly responsible for the lesions observed in the tissue.
To the authors' knowledge, this is the first report of S. maltophilia infection in buffalo. Systemic infections due to this bacterium are generally rare. A previous study 7 reported a case of septicemia that was associated with S. maltophilia in a West African dwarf crocodile. The bacterium was seen histologically in lesions of the valvular endocardium and in foci of necrosis in the myocardium, liver, spleen, pancreas, kidney, and intestinal tract of the dead animal. In the present case, the infection was limited to the liver, probably due to the difference in the virulence of the recovered isolate as well as the diverse immunoresponse of the infected animal. The possibility of a local liver infection due to the loss of rumen mucosal integrity is excluded since gross lesions were not observed within the rumen. Most S. maltophilia infections are associated with high-risk factors such as immune suppression, intravenous drug abuse, contaminated intravascular devices, large skin lesions, or wounds. 5 In the current case, the primary site of infection was not determined. Furthermore, there was no evidence of immune suppression. It could be speculated that the skin lesions, which are very common among buffalo, especially in warm climates, could be the primary site of infection prior to dissemination of the bacterium to the liver.
Infection with S. maltophilia is probably underdiagnosed in the routine veterinary diagnostic laboratory because selective media are not used for bacterial culture. The isolation, identification, and antibiotic susceptibility testing of this multidrug-resistant bacterium may be of clinical importance and should be performed on diagnostic samples.
Acknowledgements
The authors would like to thank Dr. H. Simpson for providing highly technical support.
Footnotes
a.
ID Screen® Leishmania Indirect Hemagglutination, ID VET, Montpellier, France.
b.
Indirect hemagglutination assay (IHA) for detection of leptospirosis, MRL Diagnostics, Cypress, CA.
c.
Atlas Brucella test (Rose Bengal), Atlas Medical, Cambridge, United Kingdom.
d.
Polyflex, Fort Dodge Animal Health, Fort Dodge, IA.
e.
Dexamethasone 5, Vétoquinol North America Inc., Lavaltrie, Quebec, Canada.
f.
API®, bioMérieux (Suisse) SA, Geneva, Switzerland.
g.
Sabouraud Dextrose Agar, Oxoid Ltd., Cambridge, United Kingdom.
h.
ABI PRISM® 3100, Applied Biosystems, Foster City, CA.
i.
Sensititre®, Trek Diagnostics Systems, East Grinstead, England, and MCS Diagnostics BV, Swalmen, The Netherlands.
