Abstract
The health status of a 4-year-old female, dd-haplotype miniature pig deteriorated rapidly, so the animal finally had to be euthanized because of poor clinical condition. Necropsy revealed a massive leukocytic infiltration in the parenchymatous organs of the abdominal cavity. on hematologic cell counting, severe leukocytosis (69.3 X 109 cells/liter) and high-grade basophilia (6.9 X 109 cells/liter) were evident. cytologic examination, as well as analysis of expression of leukocyte differentiation antigens by means of flow cytometry, classified blasts, which accounted for about 22% of leukocytes, as biphenotypic cells co-expressing the myeloid marker SWC3 (CD172a) and the lymphoid markers CD5 and CD25. Hematologic features resembled those seen in humans with chronic myeloid leukemia at blast phase.
Hematologic malignancy is a rare event in pigs. Previous reports include a case of myeloid leukosis in a female Göttingen miniature pig exhibiting massive accumulations of immature myeloid cells, most of them containing eosinophilic granules, in mesenteric lymph nodes, liver, kidneys, and spleen, 6 and a case of myeloid leukosis in a 3-day-old piglet. 1 We describe a case of undifferentiated leukemia in a dd-haplotype miniature pig with special focus on the immunophenotypic characterization of peripheral leukocytes including the tumor cell population.
The 4-year-old sow under investigation belonged to a herd of dd-haplotype miniature pigs 13 —these pigs being inbred for a specific SLA-1 haplotype, in particular the 0401 allele of the SLA-I∗04 group 15 —housed separately from other pigs at the research estate of the University of Veterinary Medicine Vienna, and had never been pregnant. Infertility had been recognized as a herd problem in these inbred pigs. Behavioral depression, subnormal body temperature, and weight loss were noted 1 week before referral to the clinic. Antibiotic and antiphlogistic treatment was started with moderate initial success. As the animal's health status deteriorated, the sow was submitted to the clinic. On admission, the animal was recumbent. Physical examination revealed moderate enophthalmus and moderately anemic conjunctivae and labial mucosa. Body surface was cool, and inner body temperature was 32.3°C. Respiratory rhythm was regular, with a physiologic respiratory rate (18 breaths per minute). Hematologic and serum biochemical analyses were performed, leading to the tentative diagnosis of undifferentiated leukemia. The animal was euthanized because of the poor clinical condition and prognosis.
On pathomorphologic examination, the main finding were severe, generalized lymphadenopathy with systemically enlarged lymph nodes and massive hepatomegaly (liver weight: 4.5 kg, total body mass: 110 kg). Multiple gray nodules of firm consistency and variable size were scattered throughout the liver, kidneys, and ovaries. The spleen had profound hemosiderosis and focal fibrosis. Histologic examination revealed massive infiltration of the affected organs with undifferentiated white blood cells resembling the myeloid series in the liver (Fig. 1) and lymph nodes, but lymphoid morphology in kidneys and ovaries (Fig. 2). Besides these findings, the lungs had moderate alveolar edema and emphysema. Additionally severe cystic hyperplasia of the uterine glands was evident, similar to what had also been found in other sows from this herd.
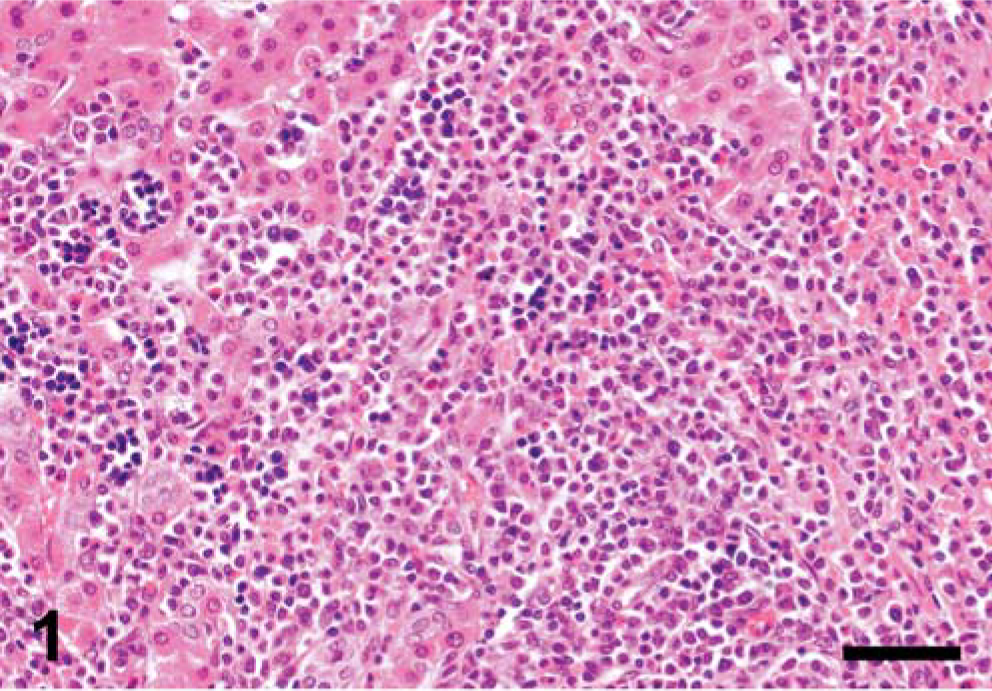
Liver; pig. Liver is infiltrated with neoplastic cells having a myeloid appearance. HE. Bar = 50 µm.
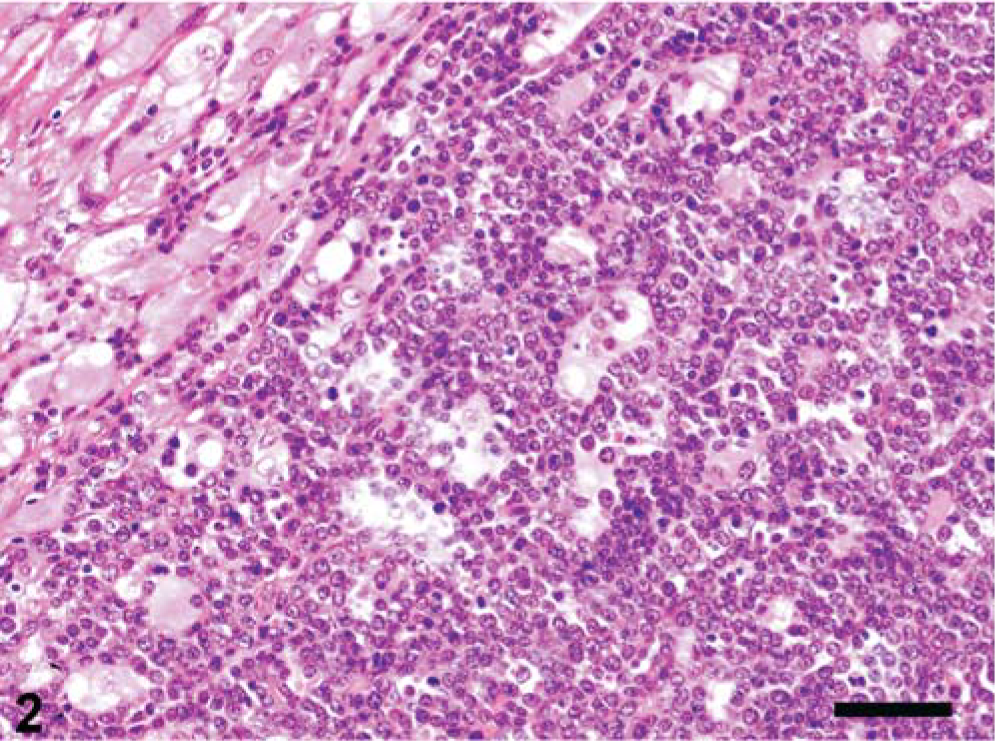
Ovary, pig. Similar to the liver, the ovary exhibits massive infiltration by undifferentiated leukocytes with a more lymphoid appearance. HE. Bar = 50 µm.
Serologic examination for antibodies to porcine reproductive and respiratory syndrome virus, porcine parvovirus, swine influenza virus, Salmonella spp., and Pasteurella multocida toxin were performed by use of commercial ELISA kits (HerdCheck PRRSV-Ab Test Kit, IDEXX, Wörrstadt, Germany; Svanovir PPV-Ab, Svanova Biotech AB, Uppsala, Sweden; HerdCheck SIV Antibody Test Kit H1N1, IDEXX; HerdCheck Swine Salmonella Antibody, IDEXX; PMT ELISA Kit, DakoCytomation, Glostrup, Denmark) and yielded negative results. However, antibody titer to porcine circovirus type 2 as well as Actinobacillus pleuropneumoniae was detected by means of APP-ApxIV Chekit (Bommeli, Liebefeld-Bern, Switzerland) and Ingezim Circovirus IgG/IgM (Ingenasa, Madrid, Spain).
Laboratory investigations were performed by use of the autoanalyzer HITACHI 911 (Roche Diagnostics, Ichiga, Japan) and ADVIA 120 (Bayer Diagnostics Europe Ltd, Dublin, Ireland) with veterinary software adapted for pigs. Serum biochemical results indicated hyperproteinemia of 9.37 g/dl (reference interval [RI]: 5.0–8.0 g/dl), which was attributable to polyclonal hypergammaglobulinemia as revealed by serum protein electrophoresis, and slight increase in γ-glutamyltransferase activity of 57 U/liter (upper reference limit [URL]: 45 U/liter), a cholestasis enzyme in pigs. Low-grade hypokalemia (3.9 mmol/liter; RI: 4.5–6.5 mmol/liter) and hypomagnesemia (0.7 mmol/liter; RI: 0.9–1.7 mmol/liter) also were present, probably attributable to anorexia. Creatine kinase activity was increased fourfold (5,613 U/liter; URL: 1,200 U/liter) because of recumbency.
Hematologic analysis revealed severe nonregenerative anemia (erythrocytes: 3.61 × 1012 cells/liter; RI: 6.0–8.0 × 1012 cells/liter) and profound leukocytosis (69.3 × 109 cells/liter; RI: 10.0–20.0 × 109 cells/liter). Microscopic differential counting revealed moderate neutropenia (4.85 × 109 cells/liter; RI: 5.5–8.0 × 109 cells/liter) and high-grade basophilia (6.93 × 109 cells/liter; URL: 0.1 × 109 cells/liter). Mature lymphocyte numbers were low, but within the physiologic range (3.74 × 109 cells/liter; RI: 3.5–8.0 × 109 cells/liter). Platelets were within the normal range (207 × 109 cells/liter; RI: 180–600 × 109 cells/liter). Besides basophilia, the most prominent finding was the massive number of morphologically undifferentiated cells with low peroxidase index (10.4 × 109 cells/liter) (Figs. 3, 4).
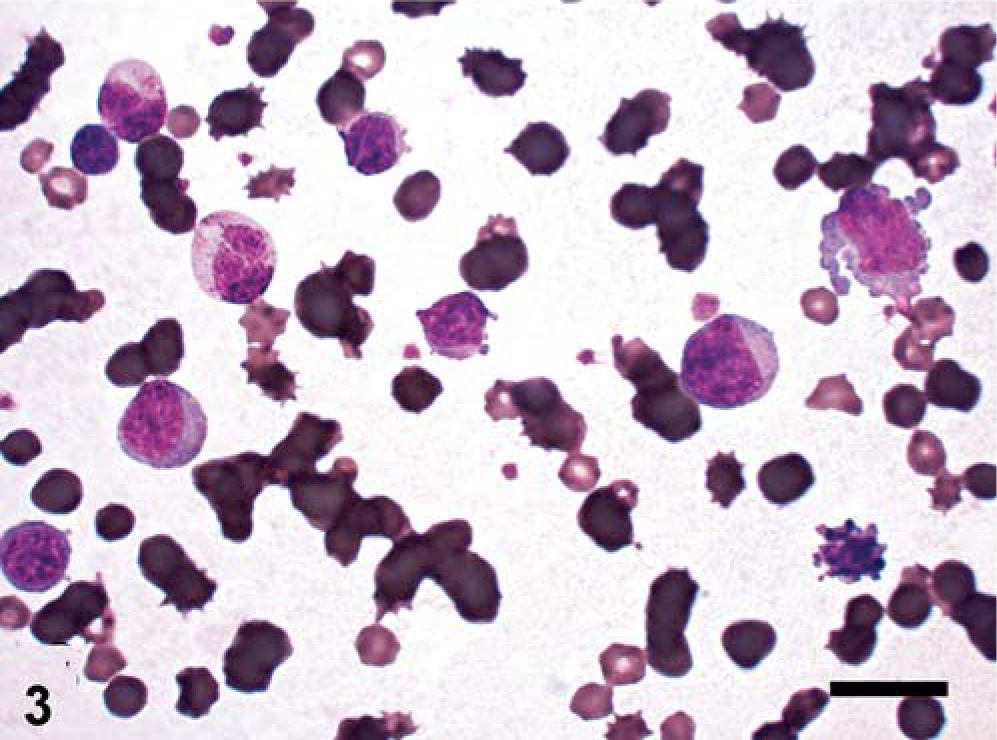
Blood smear, pig. Use of the oil immersion objective reveals atypical blast cells beside phenotypically normal lymphocytes. Two basophils with low to intermediate granularity are situated in the upper left quadrant. Because of hemolysis, there is higher background staining. Wright-Giemsa. Bar = 15 µm.
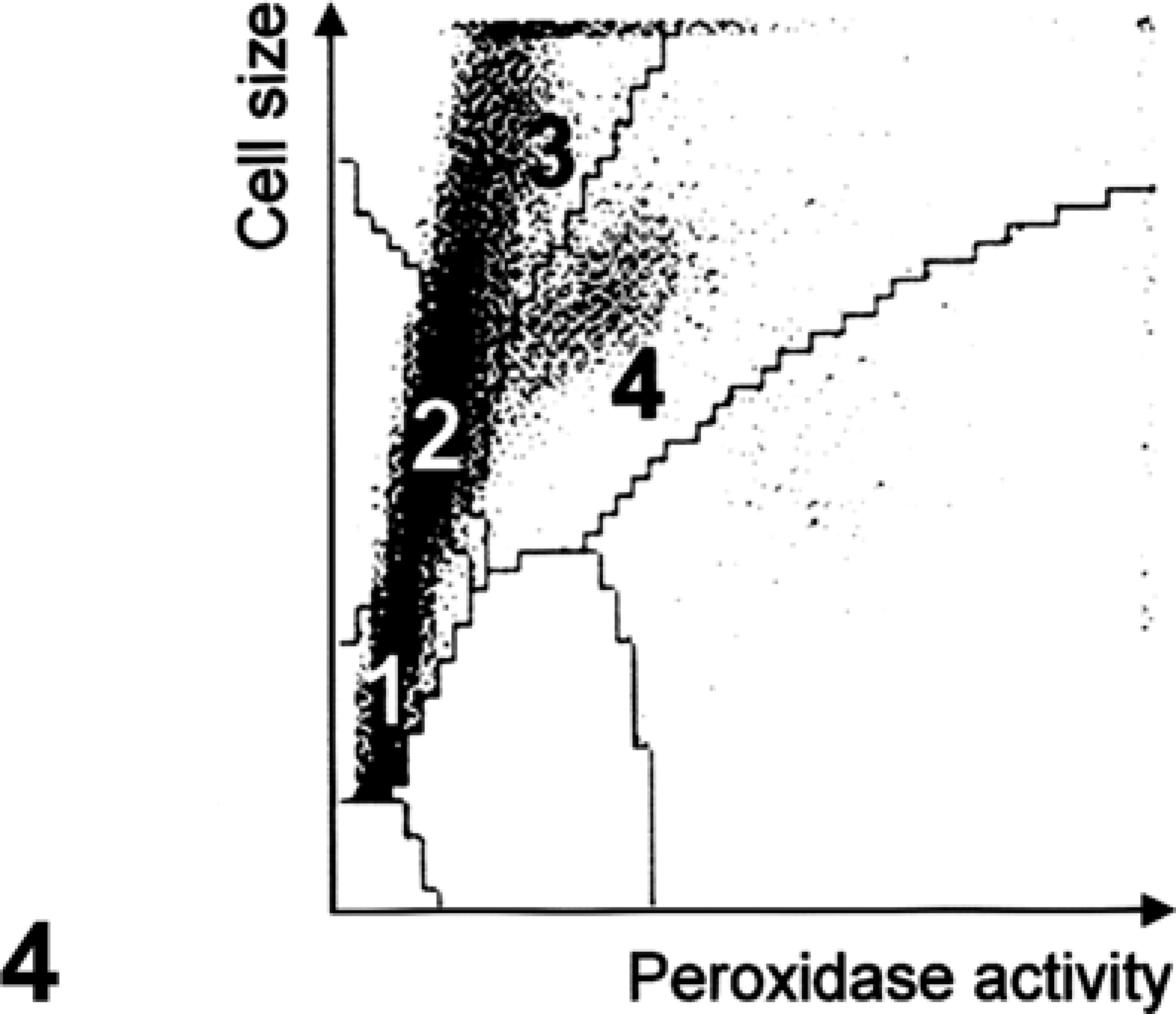
Peroxidase scattergram of leukocytes/blast cells, pig. Printout of the ADVIA 120 peroxidase scattergram. Regions: 1, cells resembling small lymphocytes; 2, monocytes; 3, large unstained cells; and 4, neutrophils.
Hematologic results did not allow classification of these large unstained cells into the lymphoid or myeloid lineage. Therefore, leukocytes were further investigated by flow cytometry with a FACSAria flow cytometer (Becton Dickinson, San Jose, CA) according to a standard protocol. Monoclonal antibodies for the staining of swine leukocyte differentiation antigens as well as their specificity are listed in Table 1. Indirect three-color staining was applied with following primary antibody combinations: 1) CD21, MHC-II, SWC3; 2) CD3, CD8, CD4; 3) CD5, CD8, CD4; 4) CD25, CD8, CD4; 5) CD45RC, CD8, CD4; and 6) TCRγδ, CD8, CD4. Each of these antibody groups forms a combination of the mouse immunoglobulin isotypes IgG1, IgG2a, and IgG2b (see Table 1 for details), and could, therefore, be labeled with the same set of secondary antibodies, consisting of anti–IgG1-PE (SouthernBiotech, Birmingham, AL), anti–IgG2a-Alexa Flour 647 and anti–IgG2b-Alexa Fluor 488 (both Molecular Probes, Eugene, OR). Combination 1 of this antibody panel allows general classification of B cells, myeloid cells, and MHC–II-positive T cells in swine. Combinations 2–6 focus on detailed analysis of porcine T cells, including activation status and subset analysis.
Monoclonal antibodies for immunophenotypic characterization of peripheral blood leukocytes.
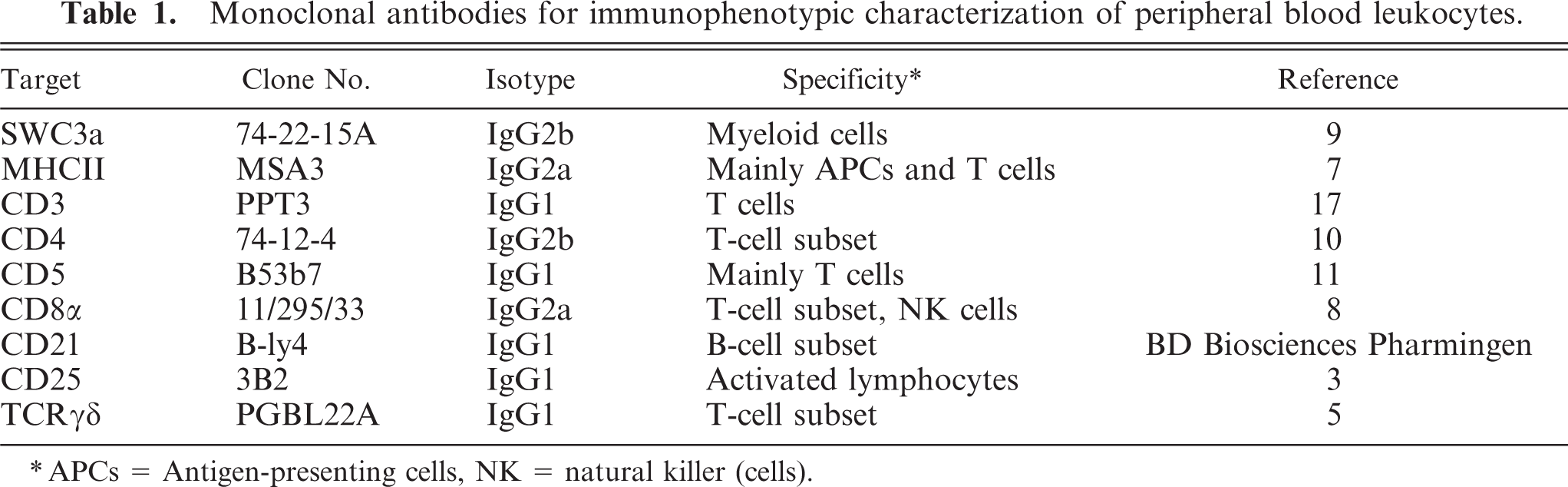
APCs = Antigen-presenting cells, NK = natural killer (cells).
The SWC3 (CD172a) expression pattern 9, 10 was unusual, with two dominant positive populations (SWC3dim and SWC3high, Fig. 5a), which included approximately 78% of the leukocyte population. Leukocyte scatter properties (Fig. 5b) were unusual, too; distinct populations of monocytes and granulocytes could not be identified. Backgating of the three SWC3 surface expression-defined populations (SWC3neg, SWC3dim, and SWC3high) in the scattergram (forward scatter versus side scatter) indicated that all cells belonging to one of these groups defined by similar SWC3 staining intensities were of similar size and granularity (R1–3, Fig. 5b). The SWC3− cells (R1) had the typical morphology of lymphocytes and expressed classical lymphocyte markers CD3 (data not shown), CD4, CD8, and CD5 (Figs. 5e–g), thus confirming the assumption that these cells comprised the majority of the lymphocyte population. However, nearly all analyzed cells were negative for CD21, and only a low frequency of B cells was situated in R1 (Fig. 5d). Most probably, most of the MHC–II-positive cells within R1 were resting T cells, which represent a peculiarity of the porcine immune system. 12 Similar to B cells, γδ-TCR+ cells also formed only a small lymphocyte subpopulation (data not shown). Owing to their morphology as well as the high SWC3 expression, cells in R3 appeared to be monocytes, which have a heterogeneous MHC-II expression pattern in pigs, 12 whereas cells in R2 could not be classified as any normal leukocyte population. They accounted for about 22% of leukocytes corresponding in number to the large unstained cells found when using the ADVIA 120 instrument. Moderate expression of the myeloid-specific surface antigen SWC3 (swine workshop cluster 3), 9 recognized by an IgG2b isoform (74–22-15A) of the monoclonal antibody 74–22-15 10 (IgG1) and previously discussed as a member of the CD172a family, 2 characterized the tumor cells as preferentially belonging to the myeloid lineage. However, the cells in this population also weakly but rather homogeneously expressed the lymphocyte-specific CD5 11 and CD25 3 antigens (Fig. 4g, h), the latter marker being expressed on activated T cells and on regulatory T cells. In contrast, the SWC3high cells in R3 did not express these two markers, supporting the assumption that these cells represented normal monocytes. In conclusion, the tumor cell population in peripheral blood seemed to consist of myeloid cells coexpressing lymphoid markers in low antigen density.
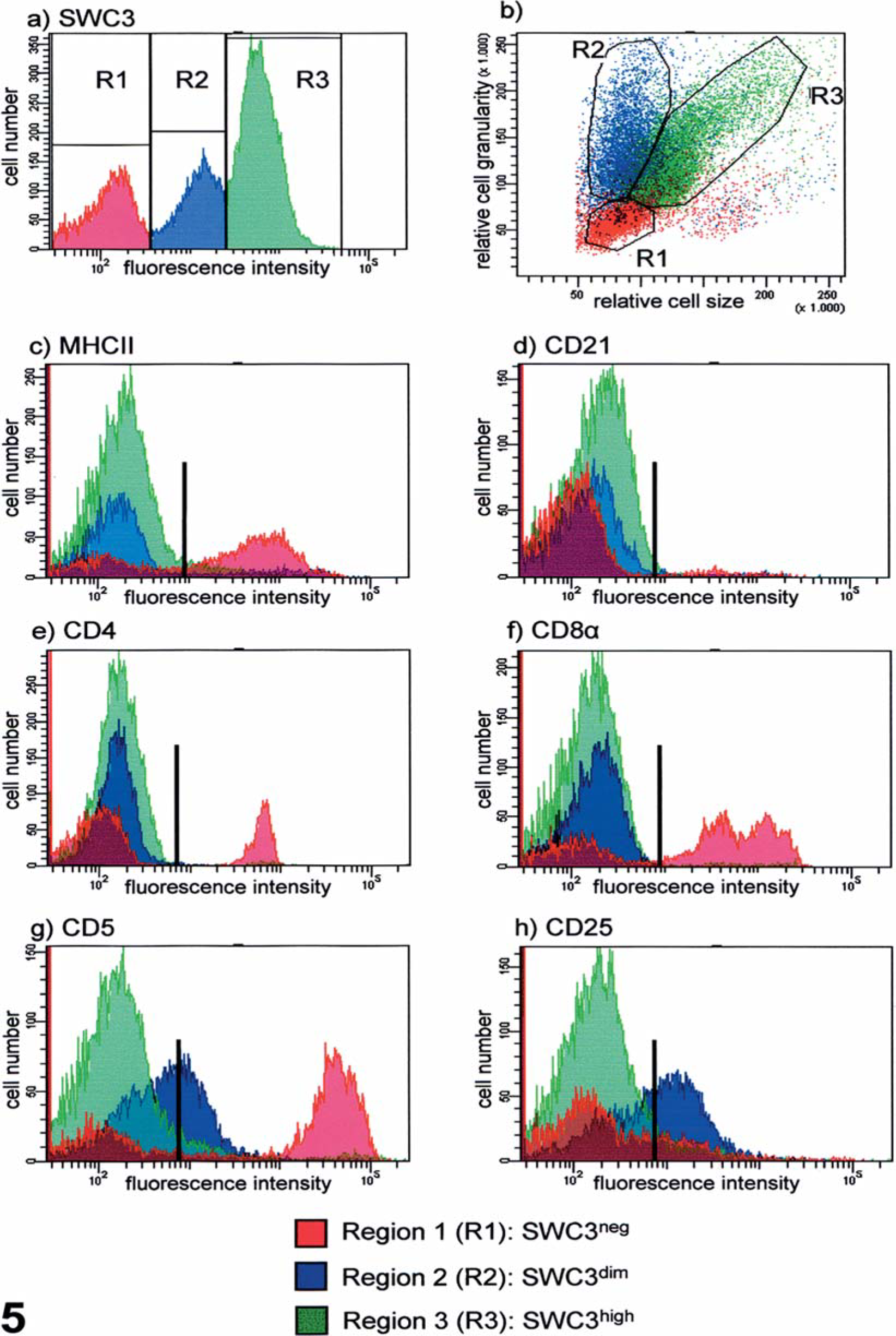
Immunophenotyping of leukocytes/blast cells, pig. Flow cytometric analysis of surface marker expression of peripheral blood leukocytes.
Several hematologic features resembled those seen in human patients with chronic myeloid leukemia (CML), which originates in an abnormal pluripotent bone marrow stem cell, at the transition from the accelerated to blast phase that is characterized by absolute basophilia and a high proportion of blasts. 16 These malignant cells may account for >20% and be of different myeloid or lymphoid lineages, which rarely occur simultaneously. In most instances, the blast lineage is obvious on morphologic examination, but as blasts usually are heterogeneous and rarely differentiated, immunophenotyping is recommended.
Because the clinical history of the sow was incomplete and because the animal was submitted in the final phase of the disease, diagnosis of CML-blast phase is only one of several possible assumptions. Basophilia might support CML because, at least in humans, basophilia is a common finding in the course of this disease. 16 Because leukemia is such a rare event in pigs, to the authors' knowledge, cytogenic investigations with respect to hematologic malignancies have not been performed in pigs at present. Thus, chromosomal aberrations, such as the Philadelphia chromosome, which leads to a BCR/ABL fusion gene with consecutively enhanced activity of a tyrosine kinase in the case of CML in humans, have not been recognized as an etiologic factor in porcine leukemias to date.
In humans, another leukemic condition characterized by detection of blasts coexpressing myeloid and lymphoid markers is termed biphenotypic acute leukemia. 4 This condition is also called acute mixed-lineage leukemia (AMLL) and has been used to propose the hypothesis of lineage infidelity of early progenitor cells. 14 These cells are supposed to transiently express myeloid and lymphoid antigens and possibly to rearrange their immunoglobulin and T-cell receptor (TCR) genes during an early stage of the disease, but down-regulate either myeloid or lymphoid markers in the course of their ontogenesis. Thus, they subsequently enter the myeloid, T-lymphoid, or B-lymphoid differentiation pathway.
To the authors' knowledge, this is the first report of porcine leukemia characterized by the observation of biphenotypic blast cells and is intended to incite awareness of hematologic malignancies in miniature pigs that are kept for research purposes.
