Abstract
Lymphoma diagnosis in dogs and cats is continually evolving as new subtypes and human correlates are being recognized. In humans, T-cell lymphomas with MUM1 expressed and plasma cell neoplasia or B-cell lymphomas with CD3 expressed aberrantly are reported only rarely. We report here a case series of tumors in dogs and cats with CD3 and MUM1 co-expressed as determined by immunocytochemistry or immunohistochemistry. Lineage was assigned for these tumors by 3 board-certified pathologists and a veterinary immunologist based on review of clinical and cellular features and the results of ancillary testing including PCR for antigen receptor rearrangements, flow cytometry, and serum protein electrophoresis with immunofixation. In cats, 7 of 7 tumors, and in dogs, 3 of 6 tumors with CD3 and MUM1 co-expressed had clonal rearrangement of the immunoglobulin gene or serum monoclonal immunoglobulin, consistent with a diagnosis of a plasma cell neoplasia or myeloma-related disorder with CD3 expressed aberrantly. Disease was often disseminated; notably, 3 of 7 feline cases had cutaneous and/or subcutaneous involvement in the tarsal area. In dogs, 3 of 6 cases had a clonal T-cell receptor gamma result and no clonal immunoglobulin gene rearrangement and were diagnosed as a T-cell tumor with MUM1 expressed. The use of multiple testing modalities in our series of tumors with plasma-cell and T-cell antigens in dogs and cats aided in the comprehensive identification of the lymphoproliferative disease subtype.
Keywords
Lymphoproliferative disorders are heterogeneous diseases derived from various B and T lymphocytes that require the use of various tests to arrive at a specific diagnosis. In human medicine, the World Health Organization (WHO) classification of lymphoproliferative disease involves multiple modalities, including evaluation of a large panel of antigen expression by flow cytometry and immunohistochemistry (IHC). This combination of testing continues to identify new subtypes or further characterizes existing subtypes of lymphoproliferative disease. Aberrant expression of proteins used for classification complicates diagnosis, often requiring genetic profiling to correctly subtype disease and provide diagnostic and prognostic information. 35
IHC is used to classify lymphoma subtypes according to WHO classification in animals, and has relied historically upon protein expression of CD3 for T-cell and CD79a for B-cell lineage assignment.26,37,38 Other B-cell antigens have been utilized in more recent studies with evidence of increased specificity, including PAX5.12,40 Protein expression of multiple myeloma oncogene 1 (MUM1), also called interferon regulatory factor 4 (IRF4), has been utilized with other T- and B-cell antigens in dogs and cats in the diagnosis of plasma cell tumors. 27
There are rare reports of lymphoproliferative diseases in which B- and T-cell antigens are co-expressed in domestic animals, and the panel of antibodies used is often variable and limited. One large-scale lymphoma subtype study in dogs found 2 plasma cell tumors in which both CD79a and CD3 were expressed. 38 A CD3- and MUM1-expressing gastric plasma cell tumor was described in a dog. 1 Co-expression of CD20 and CD3 has been described in canine enteropathy–associated T-cell lymphoma, large-cell type 1, 23 in a case report of a dog with mycosis fungoides, 2 and CD20 expression was identified in >50% of dogs with cutaneous epitheliotropic T-cell lymphoma. 11 Additionally, a peripheral T-cell lymphoma with cardiac and nerve involvement in a dog was reported in which both CD3 and CD20 were expressed, with clonal T-cell receptor gamma (TRG) rearrangements. 21 These studies did not investigate expression of MUM1.
We have identified a series of hematopoietic tumors in which CD3 and MUM1 were co-expressed, challenging lineage determination based on immunostaining alone. Our objectives were to describe the clinical presentation and cellular morphology of this series of canine and feline CD3 and MUM1 co-expressing tumors and determine the lineage of these tumors utilizing additional tests.
Materials and methods
Case selection
We identified 13 cases with co-expression of CD3 and MUM1 from submissions to the Colorado State University Clinical Pathology Laboratory (CSU-CP) or Veterinary Diagnostic Laboratory (CSU-VDL; Fort Collins, CO, USA). All cases were identified between August 24, 2016 and November 15, 2018, during routine immunostaining using a panel of antibodies targeting CD3, MUM1, and PAX5; a subset of cases also had immunostaining for CD20, CD18, or CD204. Additional testing, which was limited by sample quantity and the retrospective nature of most of the samples, included PCR for antigen receptor rearrangements (PARR), immunophenotyping by flow cytometry through the CSU Clinical Hematopathology Laboratory (CSU-CH), and serum protein electrophoresis with immunofixation (SPE with IF) by the CSU-CP. Signalment, physical examination findings, and laboratory data were obtained from the sample submission forms and from available case records. Reported laboratory data were confirmed with the CBC and chemistry panel when medical records were available.
Immunocytochemistry and immunohistochemistry
Cytologic samples were stained with modified Wright–Giemsa (Aero-spray). Immunocytochemistry (ICC) for CD3, MUM1, and PAX5 was performed in 8 cases. Cytology slides were reviewed by 2 clinical pathologists (AR Moore, ED Rout). Histologic sections were stained with H&E and IHC by routine processing (Suppl. Table 1) and diagnosed by pathologists through the CSU-VDL diagnostic service. All histology cases were reviewed by one anatomic pathologist (KL Hughes). IHC (n = 5) was performed from 5-µm thick, paraffin-embedded, formalin-fixed (FFPE) tissue sections with labeling for CD3, PAX5, and MUM1. Given the differential of histiocytic sarcoma in some cases, 2 cases were also immunostained with CD18, and 1 case was immunostained with CD204. Additionally, one case was immunostained with CD20 as another B-cell antigen. Deparaffinization, antigen retrieval, and IHC and ICC staining and counterstaining were performed on an automated staining system (Bond-Max, Bond polymer detection system, Leica; Suppl. Table 1).
Serum protein electrophoresis with immunofixation
SPE with IF was performed as described using species-specific reagents, whole serum, and labeled IgG, IgA, and IgM heavy and light chains (Suppl. Table 2). 9
Clonality testing and flow cytometry
Clonality was assessed by the PARR assay as described in dogs and cats.6,28,29 All cases were assessed with Ig and TRG primer sets. Ig primer sets targeted Ig heavy chain V-D-J gene rearrangements and Ig heavy chain incomplete D-J rearrangements. Ig light chain rearrangements were also assessed in cats (Suppl. materials). Flow cytometry was performed as described in dogs and cats28,31 using the antibody panels (Suppl. Table 3) and antibodies and clones (Suppl. Table 4).
Results
We describe 13 cases of CD3 and MUM1 co-expressing tumors in both dogs and cats (Tables 1, 2). Ten of these cases (7 cats, 3 dogs) were shown to be of plasma cell origin based on a combination of morphology, PARR, and SPE with IF. Three cases were shown to be of T-cell origin (3 dogs) based on PARR and flow cytometry. We describe separately the clinical features and diagnostic results for plasma cell tumors and T-cell lymphomas.
Patient characteristics and test results of cases with CD3 and MUM1 co-expressed in tumors in cats and dogs.
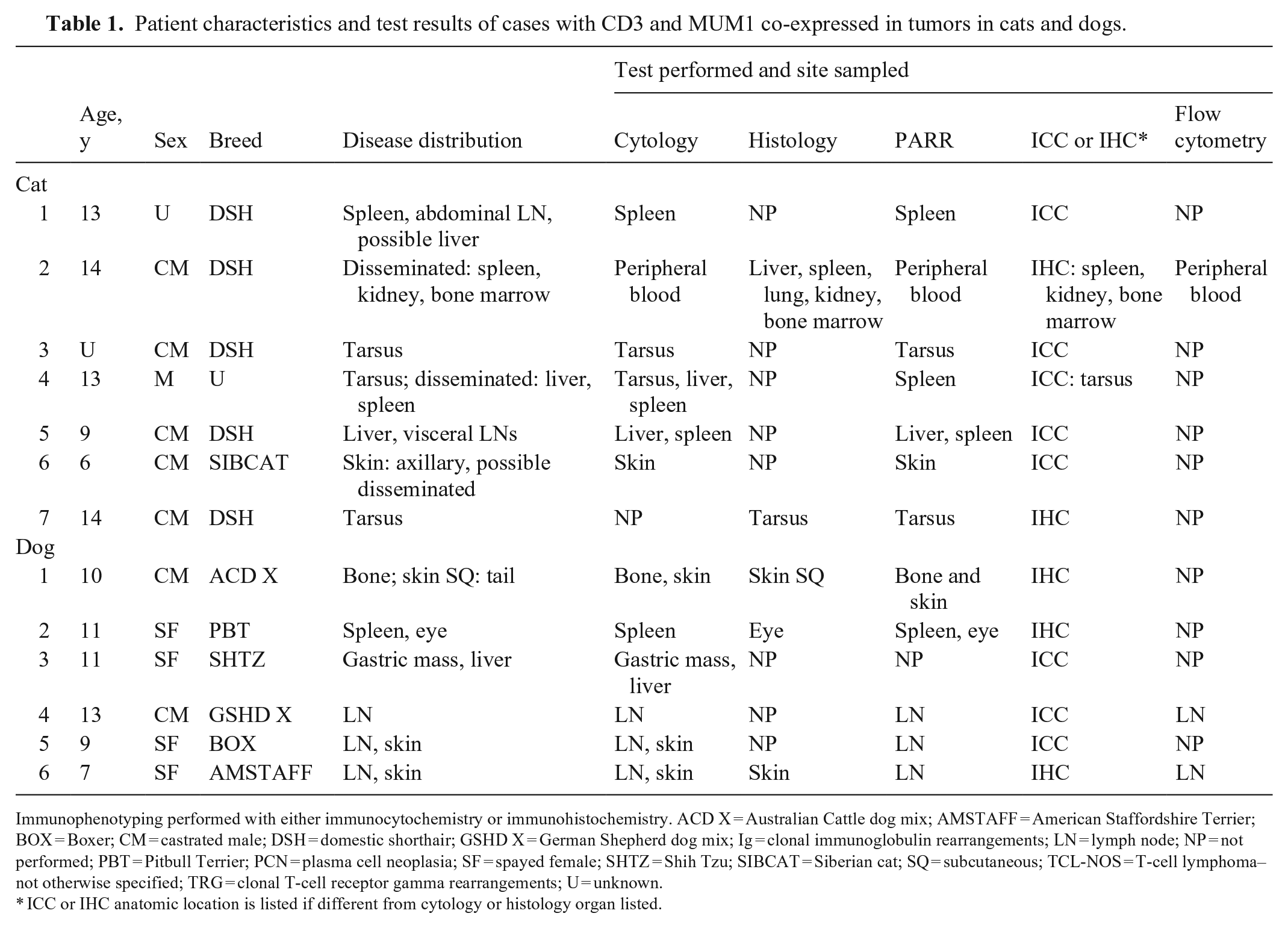
Immunophenotyping performed with either immunocytochemistry or immunohistochemistry.
ACD X = Australian Cattle dog mix; AMSTAFF = American Staffordshire Terrier; BOX = Boxer; CM = castrated male; DSH = domestic shorthair; GSHD X = German Shepherd dog mix; Ig = clonal immunoglobulin rearrangements; LN = lymph node; NP = not performed; PBT = Pitbull Terrier; PCN = plasma cell neoplasia; SF = spayed female; SHTZ = Shih Tzu; SIBCAT = Siberian cat; SQ = subcutaneous; TCL-NOS = T-cell lymphoma–not otherwise specified; TRG = clonal T-cell receptor gamma rearrangements; U = unknown.
* ICC or IHC anatomic location is listed if different from cytology or histology organ listed.
Specific CBC and chemistry tests and subclassification results from dogs and cats with tumors with co-expression of CD3 and MUM1.
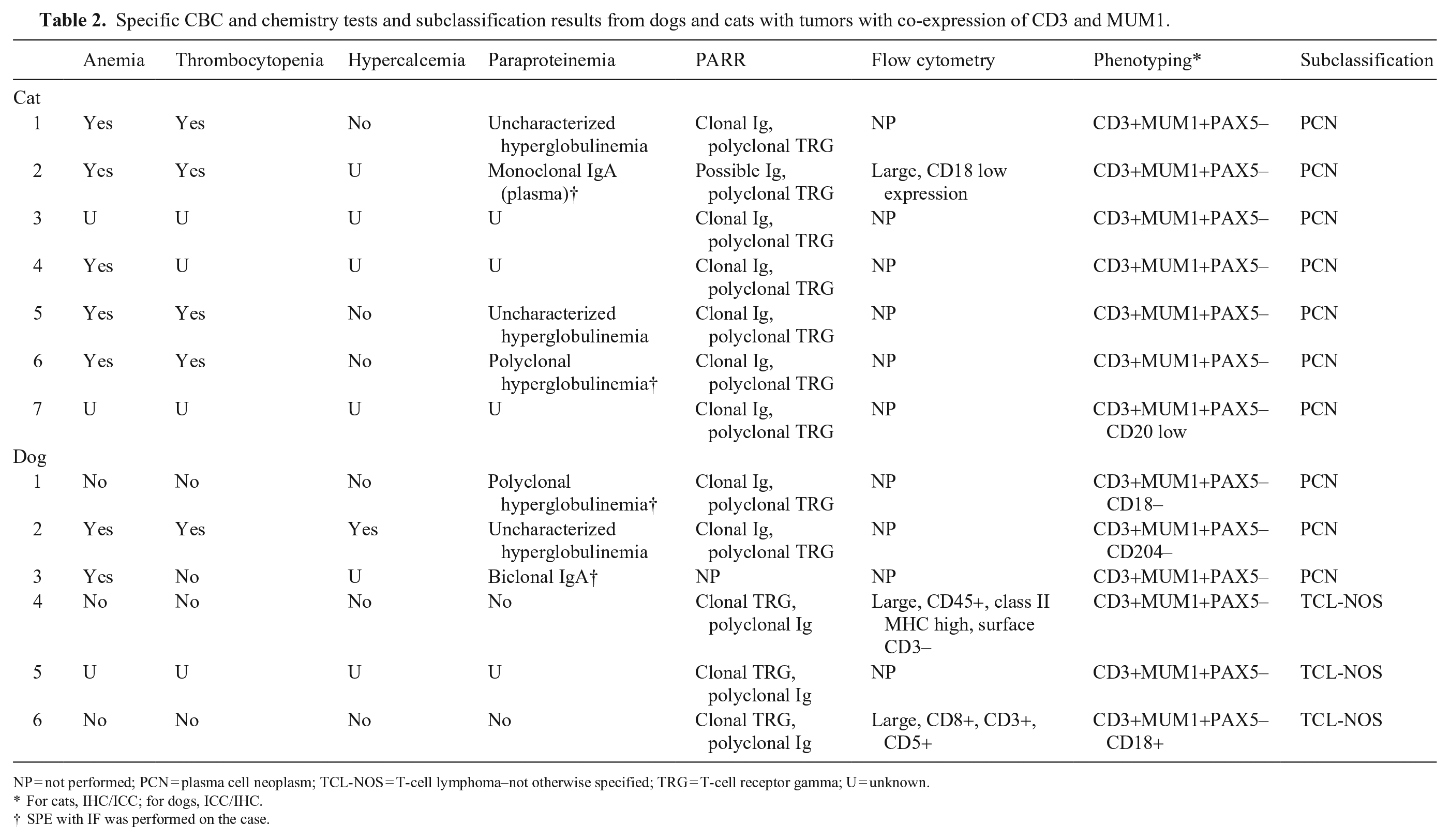
NP = not performed; PCN = plasma cell neoplasm; TCL-NOS = T-cell lymphoma–not otherwise specified; TRG = T-cell receptor gamma; U = unknown.
For cats, IHC/ICC; for dogs, ICC/IHC.
SPE with IF was performed on the case.
Plasma cell tumors
Clinical features (signalment, sites affected, CBC, and chemistry)
All 7 cats and 3 of 6 dogs were diagnosed with plasma cell neoplasia. There was 1 Siberian cat; the remaining were domestic shorthair cats, and 6 of 6 cats in which sex was known were male. There was a mix of dog breeds, with 2 females and 1 male. In cats, the tumor sites varied, and 5 cases had evidence of disseminated disease; 4 cases had cutaneous lesions, with 3 located near the tarsus. In dogs, disease site varied, with bone and skin involvement in 1 case, and abdominal organ involvement in 2 cases (Table 1). Hematologic data (Table 2) was available in 8 plasma cell tumor cases; 7 dogs were anemic and 5 were thrombocytopenic. All 5 cats with available data were anemic and 4 were thrombocytopenic, suggesting possible systemic disease. Three of 4 cats with serum chemistry data were hyperglobulinemic. Similarly, 2 of 3 dogs were hyperglobulinemic. One dog was hypercalcemic, and there was no known lymphocytosis in any patient; however, one cat had circulating Mott cells despite being lymphopenic.
Morphology and immunostaining (cytology, histology, ICC, IHC)
Cytology preparations (n = 8; 5 cats, 3 dogs) and peripheral blood (n = 1; cat) were available. Cytologically, the plasma cell neoplasms were composed of large discrete cells that ranged from well-differentiated plasmacytoid cells to anaplastic cells (Fig. 1) that had moderate-to-abundant amounts of cytoplasm, a round-to-indented or irregularly marginated nucleus, dispersed to finely clumped chromatin, and variably distinct nucleoli. Binucleation or multinucleation (Fig. 1B inset) was frequently present (7 of 8 cases), and fewer cases had mitotic figures (4 of 8). Cytoplasmic inclusions were not uncommon, with 3 cases described as having rare Mott cell morphology (including the peripheral blood sample); 2 cases had a few pink or purple cytoplasmic granules (Fig. 1). Rosettes and clusters were present consistently in one dog (Fig. 2A).
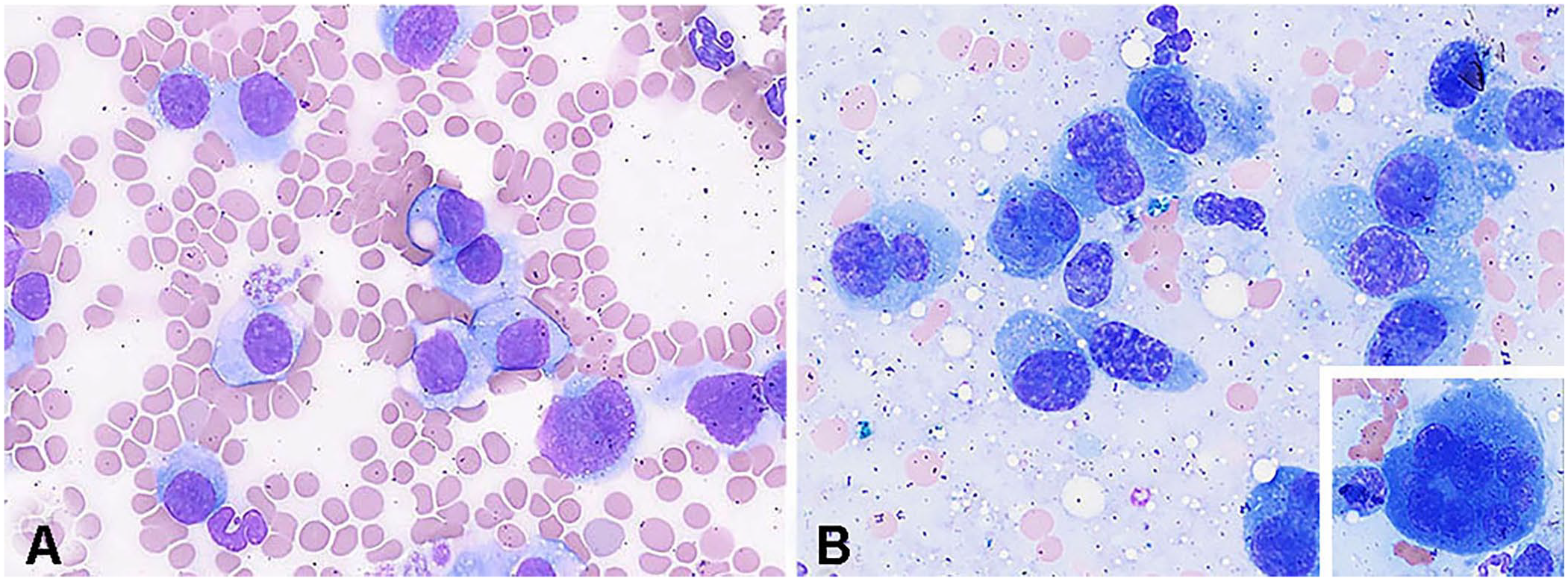
Plasma cell tumor with aberrant expression of CD3 in a cat.
Plasma cell tumor with aberrant expression of CD3 in a gastric mass in dog 3.
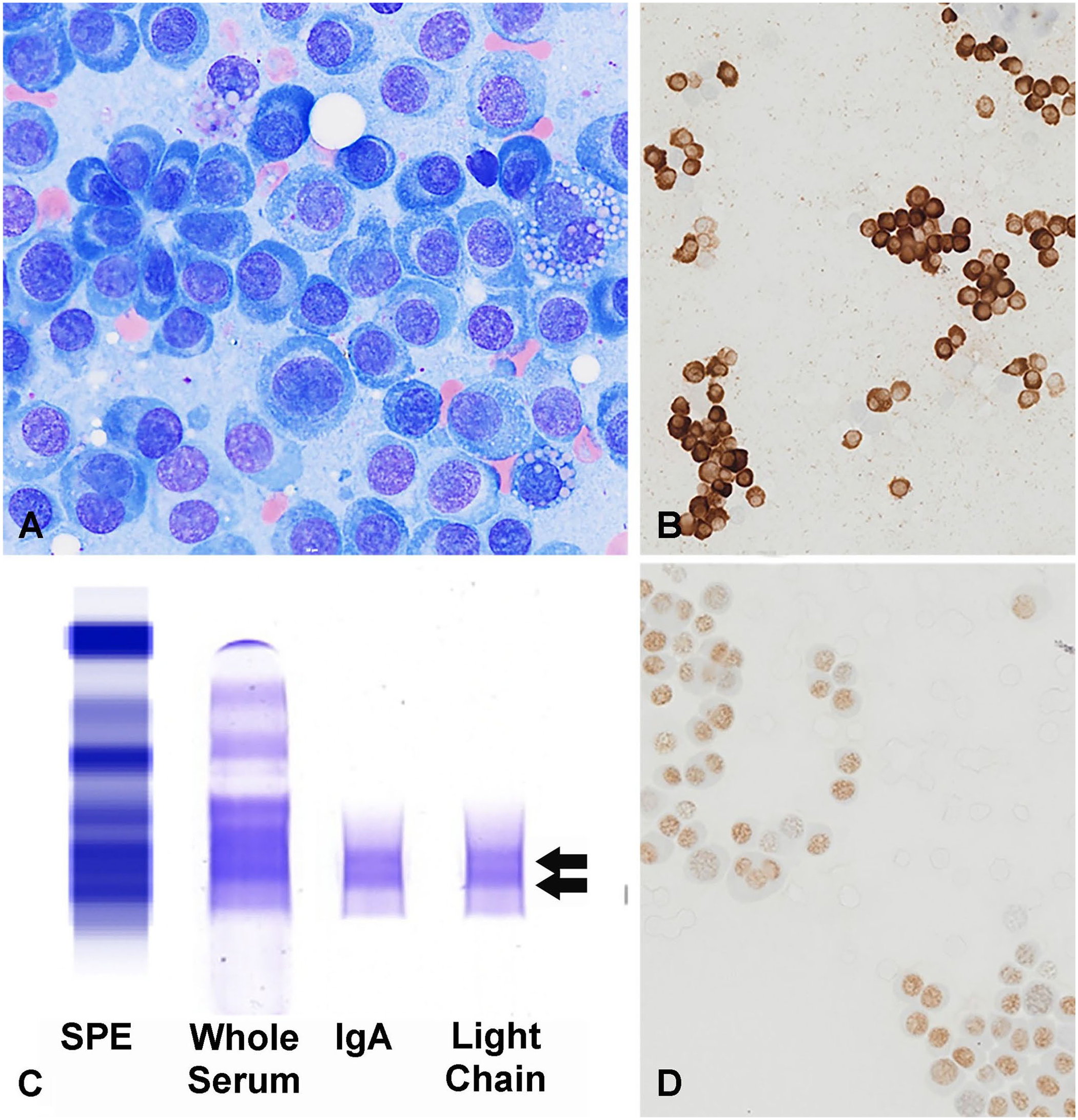
Histologic slides were available for 2 feline cases (Fig. 3) and 2 canine cases; the cellular morphology had some degree of plasmacytoid features in all cases. There was often pleomorphism with binucleate and multinucleate cells (3 of 4), and all cases had variable proportions of cells with perinuclear clearing and eccentrically placed nuclei with moderate-to-abundant eosinophilic cytoplasm and coarse chromatin. One tarsal case in a cat (Fig. 3A) had production of homogeneous extracellular, congophilic material consistent with amyloid.
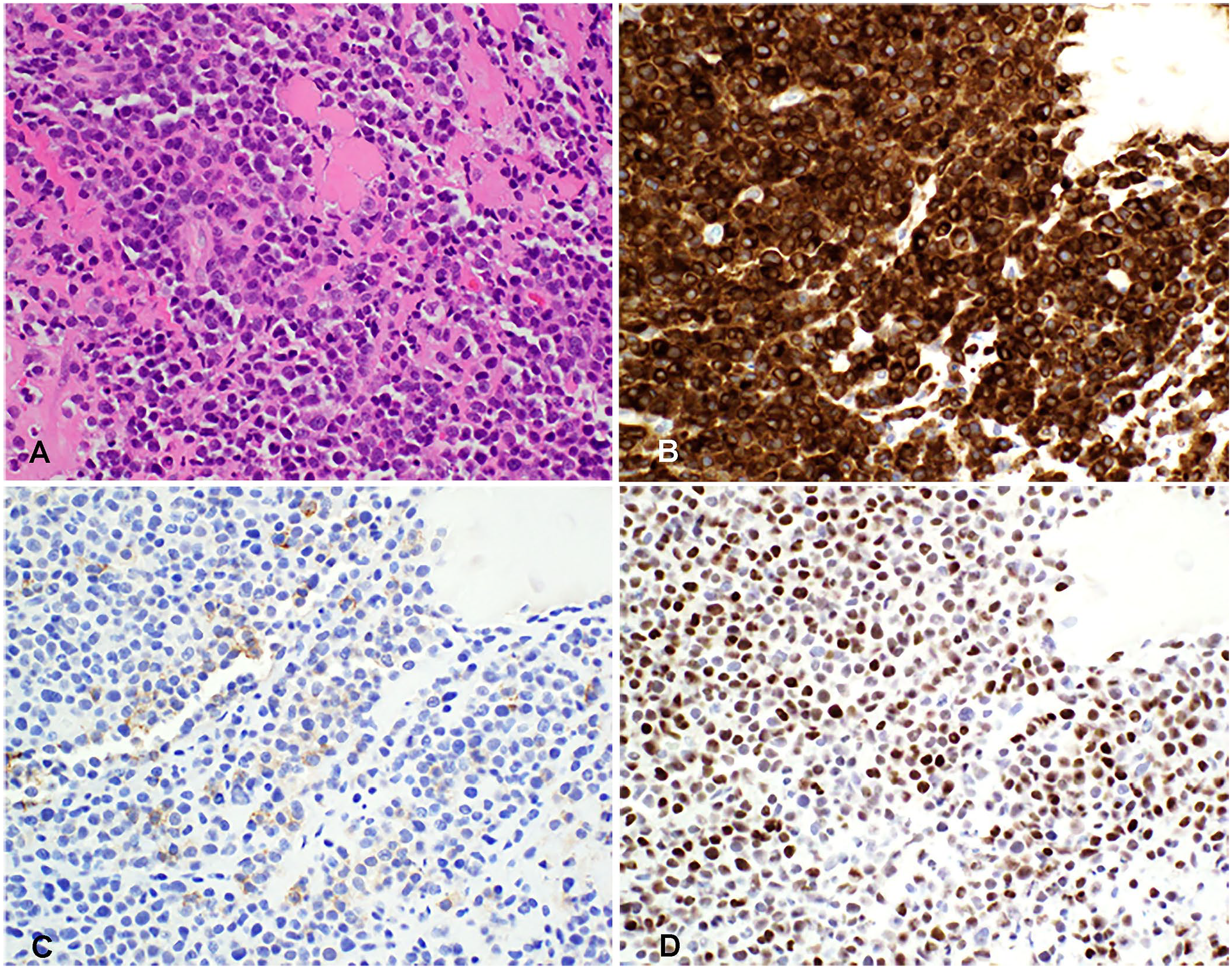
Plasma cell tumor with aberrant expression of CD3 in a subcutaneous tarsal mass in cat 7.
Antigen expression in feline samples was evaluated by cytology and ICC (n = 5), or histopathology and IHC (n = 2). Canine samples were evaluated by cytology and ICC (n = 1), or histopathology and IHC (n = 2). Although, IHC or ICC for antigens was not performed on the same slide, all cases had variable (weak-to-strong) nuclear immunoreactivity for MUM1 and strong cytoplasmic (±membranous) immunoreactivity for CD3 in most of the neoplastic cells (Figs. 2B, 2D, 3B, 3D). PAX5 expression was evaluated in all cases and was negative in all cases. One of the feline cases was also evaluated for CD20 expression and had dim, membranous CD20 expression in <30% of cells (Fig. 3C). CD18 was evaluated in one plasma cell neoplasm in a dog, and was not expressed (Table 2, dog 1; data not shown). CD204 was evaluated in one canine case, and was not expressed by the neoplastic cells (Table 2, dog 2; data not shown).
Additional lineage determination (SPE, PARR, flow cytometry)
SPE with IF were performed in 2 cases in cats and 2 cases in dogs to identify M protein (monoclonal or biclonal gammopathy) for further lineage assessment. One case in a cat was identified as having a monoclonal IgA gammopathy (Table 2, cat 2), and one dog had a biclonal IgA gammopathy by SPE with IF (Table 2, dog 3; Fig. 2C). The other 2 patients had a polyclonal gammopathy (cat 6, dog 1). The dog with the IgA gammopathy did not have sample available for the PARR assay, but the cells were very well differentiated and consistent with a diagnosis of a plasma cell tumor (Fig. 2, dog 3). In the remaining 9 cases, the PARR assay showed a clonal Ig gene rearrangement with polyclonal TRG rearrangements in 8 of 9 cases (Fig. 4). The last case (Table 2, cat 2) had a suspicious clonal Ig gene rearrangement, but the peak amplitude did not reach the objective criterion for clonality. 28 The TRG rearrangements were polyclonal and the cat had a serum M protein, supporting plasma cell lineage. PARR was performed on samples from 2 different anatomic sites in 2 dogs, and the same-sized clonal Ig rearrangement was detected in both samples for each dog (Table 2; dog 1: skin and bone; dog 2: spleen and eye). Flow cytometry of the peripheral blood was performed on cat 2 because of circulating Mott cells despite a normal lymphocyte count. By flow cytometry, the neoplastic cells were large (similar size to neutrophils) and expressed low levels of CD18, the feline pan-leukocyte antigen, similar to levels of expression on normal lymphocytes in cats. Antibodies to cell surface CD3 are not available for cats for flow cytometry.
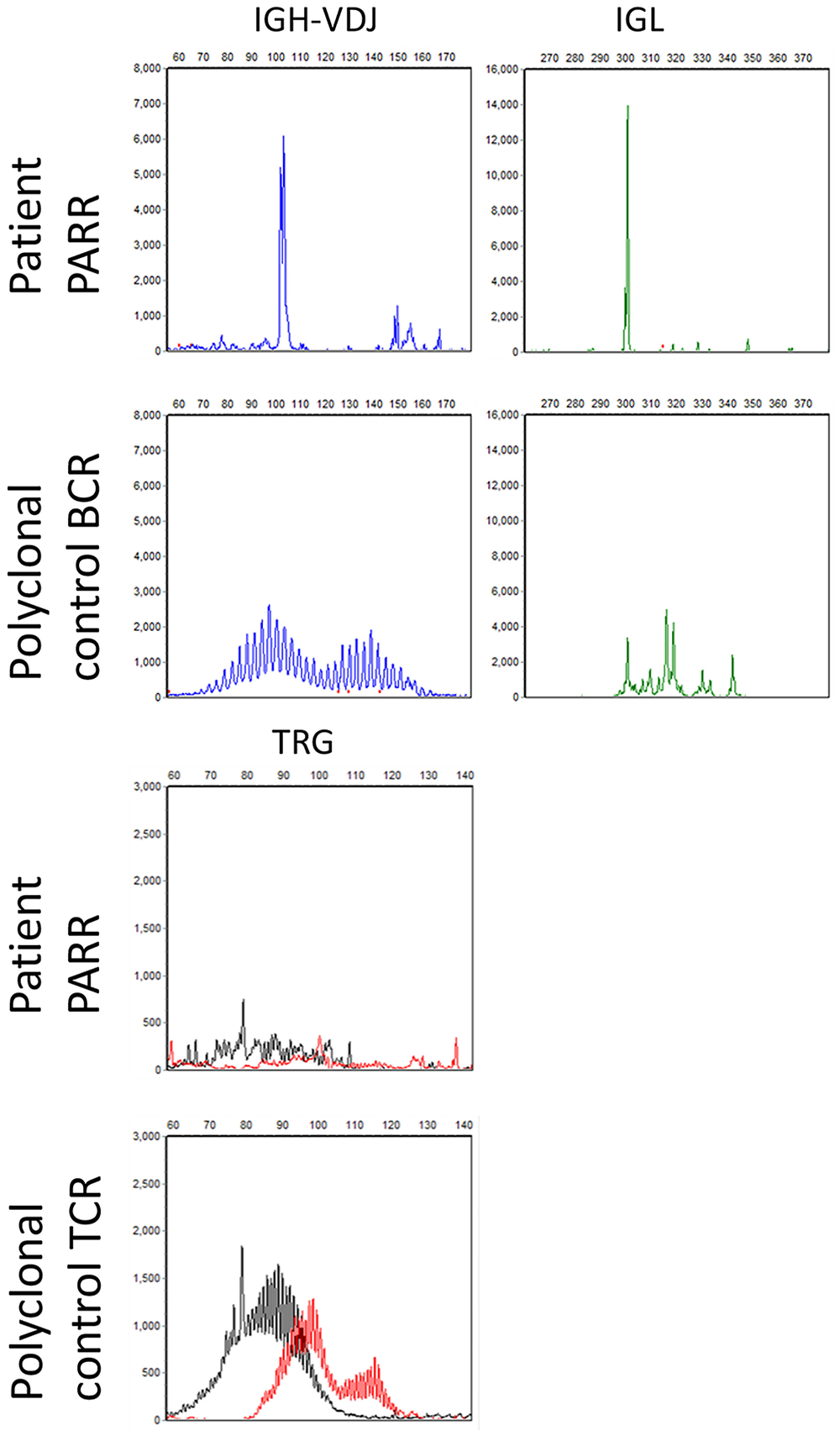
Cat 7 had a clonal rearrangement from the complete Ig heavy (IGH) chain variable (V)-diversity (D)-joining (J) (IGH-VDJ) locus (left, top, blue) and a clonal rearrangement from the Ig light chain (IGL) locus (right, top, green), compared to a polyclonal control. Cat 7 had polyclonal T-cell receptor gamma (TRG) rearrangements similar to the polyclonal control.
T-cell tumors
Clinical features (signalment, sites affected, CBC and chemistry)
All 3 cases of T-cell neoplasms were identified in dogs (Tables 1, 2). Dogs were mixed breeds (2 females, 1 male). All cases had peripheral lymph node involvement and 2 had skin involvement. There was no known lymphocytosis, anemia, thrombocytopenia, hypercalcemia, or hyperglobulinemia in any of these patients.
Morphology and immunostaining (cytology, histology, ICC, IHC)
All T-cell tumors were identified in dogs, and cytology results were available in all cases, with histopathology results available in one case. The cellular morphology was variable and often pleomorphic, with moderate-to-marked anisocytosis and anisokaryosis in all cases. In one case, more anaplastic cells were present cytologically, with cleaved or cerebriform nuclei, dispersed chromatin, and frequent mitotic figures. Binucleate and multinucleate cells were identified cytologically in another case. Histologically, the cells were round, with moderate amounts of cytoplasm and indistinct nucleoli (Fig. 5A).
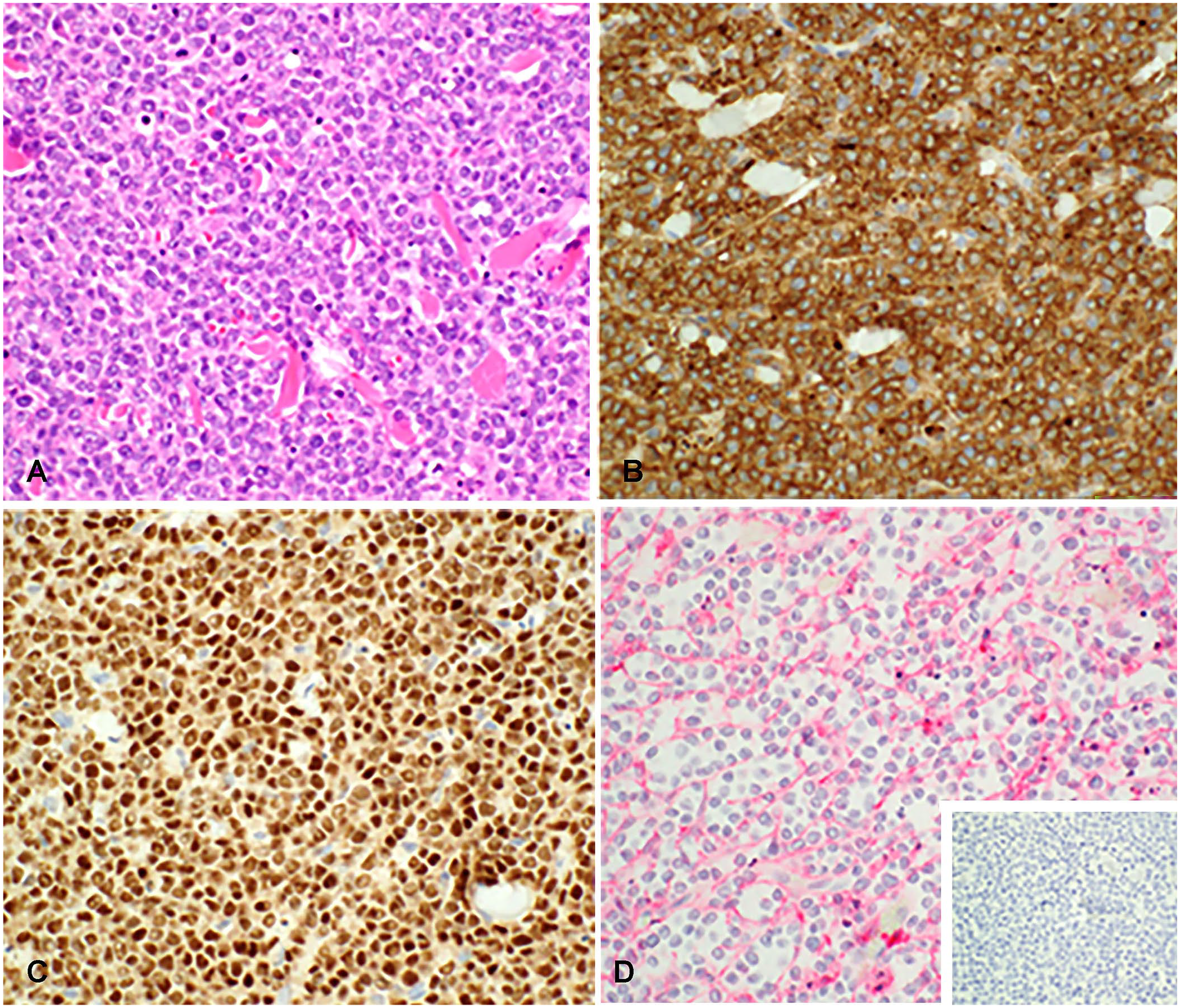
CD8 T-cell neoplasm with MUM1 expression in facial skin, dog 6.
All cases had variable (weak-to-strong) nuclear immunoreactivity for MUM1, and strong cytoplasmic immunoreactivity for CD3, in most of the neoplastic cells. PAX5 expression was evaluated in all cases and was uniformly negative (Fig. 5D inset). CD18 was evaluated in one case and was expressed (Table 2, dog 6; Fig. 5D).
Additional lineage determination (PARR, flow cytometry)
Flow cytometry was performed in 2 dogs, 1 of which had an expansion of large CD8 T cells (Table 1, dog 6; Fig. 6, lymph node). By flow cytometry, the other dog had a homogeneous expansion of large cells that only expressed CD45 (pan-leukocyte antigen) and class II MHC (Table 1, dog 4; data not shown, lymph node). No other mature lineage antigens were expressed by the neoplastic cells by flow cytometry, including surface CD3.
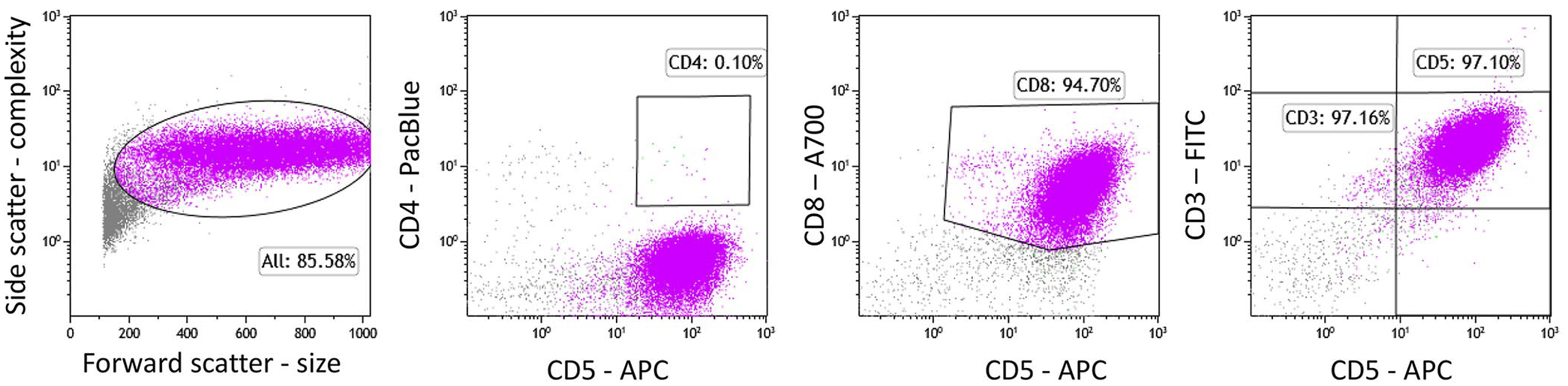
Flow cytometry of the lymph node of dog 6 revealed a homogenous expansion of large T cells that express the T-cell subset antigen CD8 and the pan–T-cell antigens CD3 and CD5 (purple).
The PARR assay was performed in all 3 cases. All cases had polyclonal Ig gene rearrangements and clonal TRG gene rearrangements (Fig. 7). SPE with IF was not performed or recommended in any of these cases.
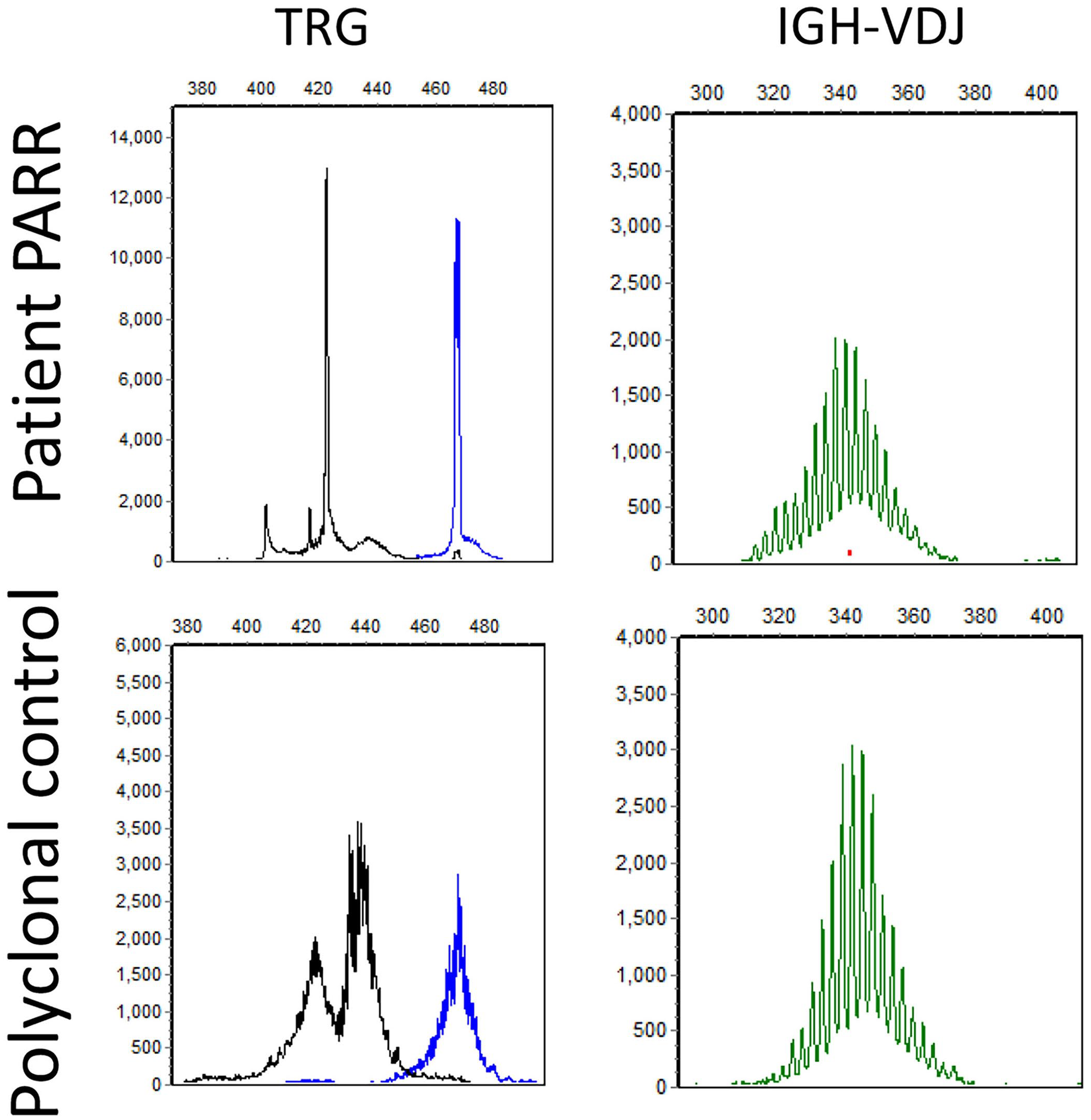
Dog 6 had clonal T-cell receptor gamma (TRG) rearrangements (left, top), compared to the polyclonal control (left, bottom). Dog 6 had polyclonal rearrangements for the complete Ig heavy (IGH) chain variable (V)-diversity (D)-joining (J) (IGH-VDJ) locus (right, top), similar to the polyclonal control (right, bottom).
Discussion
We found that neoplastic cells in plasma cell neoplasms and T-cell lymphomas in dogs can co-express CD3 and MUM1. By contrast, all CD3 and MUM1 co-expressing feline cases were diagnosed as plasma cell tumors. The PARR assay and/or SPE with IF assays were used to help assign lineage.
MUM1/IRF4 was originally identified as a myeloma-associated gene in human myeloma patients, defined as a chromosomal translocation with juxtaposition to the Ig heavy chain gene enhancer, t(6;14)(p25;q32). 17 In the germinal center, IRF4 expression is found in mature centrocytes differentiating toward plasma cells. In humans, MUM1 is expressed to various degrees in diffuse large B-cell lymphoma (DLBCL) subtypes, 14 Reed–Sternberg cells of classic Hodgkin lymphoma, 5 marginal zone lymphoma, small lymphocytic lymphoma, and a number of T-cell tumors. 36 Plasma cell tumors in dogs and cats have historically been identified based on morphology and expression of MUM1. 27 However, MUM1 expression has also been identified in histiocytomas in dogs. 33 In our case series, the reported expression of MUM1 by T-cell lymphomas adds to the complexity of accurate diagnosis of round cell tumors in domestic animals and makes interpretation of MUM1 immunostaining alone a diagnostic dilemma.
PAX5 is a transcription factor necessary for B-cell development and is normally expressed throughout B-cell differentiation, excluding plasma cells. 15 PAX5 downregulation promotes plasma cell differentiaton. 22 In all of the cases in our study, MUM1 expression occurred with a lack of PAX5 expression. Based on the normal loss of expression of PAX5 in plasma cells, a clonal Ig PARR result or M protein, in conjunction with cellular morphology and clinical presentation, these cases are interpreted as plasma cell tumors with aberrant expression of CD3. In cats, myeloma-associated disorders have been reported commonly with extramedullary presentation. 20 In our study, disseminated disease occurred in half of the feline cases, with bone marrow involvement in 1 case; an M protein was documented in 3 of 4 cases with disseminated disease. Furthermore, anemia and thrombocytopenia were present in all feline cases with available data, suggesting more systemic involvement. In dogs with plasma cell tumors, bone involvement (lytic lesion) was reported in 1 dog and an M protein was present in the other 2 dogs, with multiple organs affected.
CD138 is commonly used in human medicine to designate plasma cell origin and is often used in combination with numerous antibodies during the initial diagnostic workflow of high-grade lymphoma. 39 CD138 has been evaluated in canine tissue, and expression was identified in plasma cells and activated B cells. 8 CD138 expression was also identified in 12 of 15 canine DLBCLs, predominantly in cases that were considered nongerminal-center DLBCLs. 8 Development of a more specific marker for plasma cell tumors for veterinary medicine is needed given our results and other reports of MUM1 expression in non–plasma cell tumors in dogs.32,33
The 3 feline cases with involvement of the tarsus provide striking information; 2 had cutaneous lesions without disseminated disease, and 1 had confirmed disease in the liver and spleen. Additionally, during compilation of our case material, an additional CD3 and MUM1 co-expressing feline plasma cell tumor was identified with tarsal location through Antech by one of the authors (A. A. Pavuk), and a clonal Ig was confirmed through the CSU-CH. Tarsal lymphoma in cats has been described previously. 4 All tarsal lymphomas have been described in cats as non-epitheliotropic, and immunophenotyping was performed in 13 of 23 cases using a varied combination of CD3, CD20, CD79a, and CD18. 4 Most cases of tarsal lymphoma were consistent with a B-cell lineage (n = 9), and CD3 was expressed in 4 cases. 4 Within the CD3-expressing cases, 2 were reported to have a small number of neoplastic cells that also expressed CD79a, which is expressed in roughly one-third of feline myeloma–related disease. 20 Additionally, 2 cases had amyloid production, including 1 case that lacked expression of all tested antigens (CD3, CD79a, CD18). 4 We also detected amyloid in one of the feline tarsal cases in our series. Notably, none of the cases were evaluated for MUM1 immunoreactivity, and PARR was not performed to confirm lineage assignment. A 2022 report investigating feline periarticular tumors, primarily in the tarsus, found all cases labeled with MUM1, but CD3 and PAX5 was not investigated; these cases also often had abundant amyloid production. 7 Published data on B-cell antigen expression by plasma cells and our data on CD3 expression in plasma cells in this location call for further evaluation of feline tarsal lymphoproliferative disease to determine what fraction of these cases is a myeloma-related disorder with aberrant expression of CD3.
CD3 is a component of the T-cell receptor and was observed almost 20 y ago to detect T cells in canine FFPE tissues. 13 In humans, a variety of plasmablastic B-cell (plasmablastic lymphoma, plasmablastic plasma cell) tumors have been reported with aberrant expression of CD3. 25 There are also a number of B-cell lineage tumors with reported aberrant expression of CD3, including DLBCL. 24 In our case series, cases with clonal TRG rearrangements had a lack of plasma cell differentiation, no hyperglobulinemia or hypercalcemia, and no evidence of bone involvement. We interpret these tumors as T-cell lymphoma with MUM1 expression. In dogs, cases with a clonally rearranged TRG all had lymph node involvement, and 2 of 3 had skin involvement. Peripheral T-cell lymphoma with skin involvement or cutaneous (non-epitheliotropic) T-cell lymphoma with lymph node involvement could be considered in these cases. Histologically, in one T-cell case, there was no evidence of epitheliotropism, inconsistent with a diagnosis of cutaneous epitheliotropic lymphoma or mycosis fungoides. Involvement of peripheral lymph nodes and abdominal lymph nodes, along with enlarged spleen and liver occurred in the third dog with a clonal TRG. Histopathology was not available in this case, and flow cytometry did not identify surface expression of CD3, CD5, CD4, or CD8. This case is still considered consistent with a T-cell tumor with MUM1 expression but could not be further subtyped by cytology and flow cytometry results alone.
Accurate immunophenotyping is necessary for correct diagnosis of lymphoma subtypes. A preliminary consensus on what antibodies are appropriate for flow cytometry is available 19 ; however, standards for IHC diagnosis of lymphoproliferative disease have not been established in veterinary medicine and continue to evolve as studies identify expression patterns that add to knowledge of antibody specificity. The IHC and ICC antibodies that we used have been used extensively at CSU and have been reported in published works,2,16,18,29,30 but antibody performance can differ between laboratory methods and between clones that purportedly label the same antigen, highlighted in the identification of MUM1 in histiocytomas. 33 Laboratory-specific performance, and the effects of cost and pathologist preference, likely contribute to the observed inconsistencies in basic phenotyping standards for cytology and histopathology. The combination of cellular morphology and a broader panel of antibodies for immunostaining may help avoid inaccurate classification, but is unlikely to diagnose all cases accurately. Use of ancillary tests, including PARR, flow cytometry, and SPE, can aid in the appropriate identification of lymphoma and plasma cell neoplasms.
Flow cytometry is an extremely useful diagnostic modality for identification and classification of lymphoma or leukemia. It should be noted that, to date, there are no plasma cell–specific antibodies for dogs or cats that can be used for cell-surface staining. Therefore, a combination of other tests, as described here, is necessary for identification of plasma cell tumors. However, flow cytometry, with a panel of lymphoid antibodies, was useful for definitive diagnosis of a T-cell tumor in one of our canine cases.
In our case series, the PARR assay was valuable for lineage identification of co-expressing tumors, given that it provided evidence of lineage in 11 of 12 cases in which it was evaluated. PARR has been utilized in other studies with tumors co-expressing typical B- and T-cell antigens. 23 PARR should not be used alone for determining lineage because dual clonal rearrangements can occur in immature neoplasms 34 and when 2 different neoplasms are present. The PARR assay used alone in these cases would not differentiate a plasma cell tumor from a B-cell lymphoma and does not provide prognostic or subtype information. Nevertheless, both flow cytometry and PARR were shown to be valuable ancillary tests and should be used in future cases expressing conflicting or ambiguous IHC or ICC staining.
SPE with IF documented functional Ig production, consistent with plasma cell lineage and was used to determine lineage in 2 of 4 cases. Ig production alone is not specific evidence of plasma cell differentiation; M protein production has been reported in B-chronic lymphocytic leukemia, Burkitt lymphoma, DLBCL, and follicular lymphoma in humans. 10 In the dog, hyperglobulinemia has been reported in 25–68% of B-cell chronic lymphocytic leukemia cases. 3 However, in our case series, M protein production was most supportive of a plasma cell lineage, which was interpreted in conjunction with the cellular morphology and clinical presentation.
In our study, all feline neoplasms with co-expression of CD3 and MUM1 were consistent with a plasma cell lineage by PARR or IF. In dogs with co-expressing tumors, 3 cases were consistent with a plasma cell lineage based on PARR or IF, and 3 were T-cell lineage based on the PARR assay with or without flow cytometry. Our study highlights the need for a more specific plasma cell marker in dogs and cats. In the interim, our study demonstrates the need to use a panel of antibodies and a combination of tests for the appropriate classification of lymphoproliferative disease.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221139799 – Supplemental material for A series of heterogeneous lymphoproliferative diseases with CD3 and MUM1 co-expressed in cats and dogs
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221139799 for A series of heterogeneous lymphoproliferative diseases with CD3 and MUM1 co-expressed in cats and dogs by Kelly L. Hughes, Emily D. Rout, Paul R. Avery, Alana A. Pavuk, Anne C. Avery and A Russell Moore in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Todd Bass and the Histopathology Laboratory (CSU Diagnostic Laboratory, Fort Collins, CO, USA) for assistance with immunohistochemistry and immunocytochemistry.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded in part by CSU Clinical Pathology Research and Development Funds.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
