Abstract
Clinical, hematologic, and immunophenotypic data were studied in 25 dogs with large granular lymphocyte (LGL) lymphocytosis. Primarily large-breed dogs were affected, with an average age at initial diagnosis of 10 years (range 5-14 years). All dogs had persistent (>4 months) LGL lymphocytosis except for three that were euthanized with aggressive disease. Splenomegaly was reported in 12 of 20 dogs in which splenic size was evaluated. The clinical course was heterogeneous and dogs were divided into four groups based on similar clinical and hematologic findings: acute leukemia (3/25), persistent lymphocytosis with anemia (12/25), persistent lymphocytosis without anemia (8/25), and reactive lymphocytosis (2/25). Immunophenotypes varied within groups but were homogeneous among cells from the same patient except in the two dogs classified as reactive LGL lymphocytosis. Analysis of T-cell receptor (TCR) usage identified three main LGL lineages. TCRαβ was expressed in 15/25 (60%) cases. TCRγδ was expressed in 8/25 (32%) cases, and 2/25 (8%) cases were CD3-, compatible with NK cells. β2 integrin expression was distinctive. CD11a was consistently expressed, while CD11b was absent. CD11c was expressed only weakly in 16/25 (64%) cases. The leukointegrin αdβ2 was highly prevalent on all LGL lineages, being expressed in 23/25 (92%) cases. Prominent involvement of the spleen, relative sparing of bone marrow, an unexpectedly large proportion of γδ T-cell LGLs, and the distinctive β2 integrin expression pattern on diverse lineages of LGLs suggest the disease arises from unique populations of lymphocytes that preferentially localize in the splenic red pulp.
Keywords
Interest in large granular lymphocytes (LGLs) has grown due their tumoricidal activity, antiviral effects, and possible immunoregulatory functions. 28 32 44 Clinical disorders associated with abnormally increased numbers of LGLs were first described in people in the middle 1970s. 21 LGL lymphocytosis may be either transient (reactive) or persistent (reactive, preneoplastic, or neoplastic). 18 29 30 32 35 In peripheral blood, these cells have abundant lightly basophilic cytoplasm with azurophilic granules. Despite similar morphology, LGL populations are heterogeneous due to differences in the lymphoid lineages from which they are derived, ability to produce and respond to cytokines, ability to lyse target cells, variation in state of activation, and their surface phenotype. 3 28 34 44 51 Immunophenotypic analysis divides human LGLs into two lineages: CD3− natural killer (NK) cell LGLs that mediate nonmajor histocompatibility complex (MHC)-restricted cytotoxicity and CD3+ LGLs thought to be in vivo activated cytotoxic T-cells. 20 34
Little information is available on canine LGLs. In normal dogs, LGLs comprise 0–10% of the peripheral blood lymphocyte (PBL) population (estimated at 0–500/µl based on the upper normal limit for lymphocytes in canine blood). 48 49 Interleukin-2 stimulation of canine PBLs results in proliferation of LGLs with cytotoxic activity, 26 and treatment with anti-CD18 monoclonal antibody (mAb) markedly decreases LGL-mediated cytotoxicity. 19 Clinical disease associated with canine LGL lymphocytosis has been reported only rarely. Reactive LGL lymphocytosis occurs in some dogs with chronic ehrlichiosis. 48 In contrast, Wellman et al. describe three dogs with LGL lymphocytosis, two of which had progressive disease and infiltration of parenchymal tissues including spleen and lymph nodes. 49 Recently, we found that the leukointegrin αdβ2 is expressed by a minor population of canine PBLs. When sorted by immunomagnetic beads, approximately 35% of αd + PBLs have LGL morphology. 9 Tissue expression of αd is highly restricted to the red pulp of the canine spleen but is found on diverse lineages of canine leukocytes including macrophages, αβ T-cells, γδ T-cells, and non-T, non-B lymphocytes, thought to be NK cells (manuscript in preparation).
In order to characterize the immunophenotypic properties of canine LGLs and their associated clinical conditions, we solicited blood samples and clinical data from dogs with LGL lymphocytosis. Here we show that canine LGLs represent diverse lineages of lymphocytes that frequently express the leukointegrin αdβ2 regardless of lineage. In addition, canine LGL lymphocytosis is clinically heterogeneous, ranging from an indolent condition to aggressive malignancy. Finally, we conclude that lymphoproliferative diseases of canine LGLs frequently originate in the splenic red pulp, an environment normally highly enriched with αdβ2 + cells of diverse lineages.
Materials and Methods
Dogs
Wright-Giemsa stained blood films from dogs with lymphocytosis (>5,000/µl) were examined for the presence of lymphocytes with large granular morphology. Dogs were considered to have LGL lymphocytosis if greater than 10% of the PBLs had large granular morphology and the number of LGLs in peripheral blood exceeded 500/µl. Twenty-five cases were included in this study, and no dogs were identified with less than 2,000 LGLs/µl. Dogs were considered to have persistent LGL lymphocytosis if LGL counts remained elevated for at least 3 months.
Monoclonal antibodies
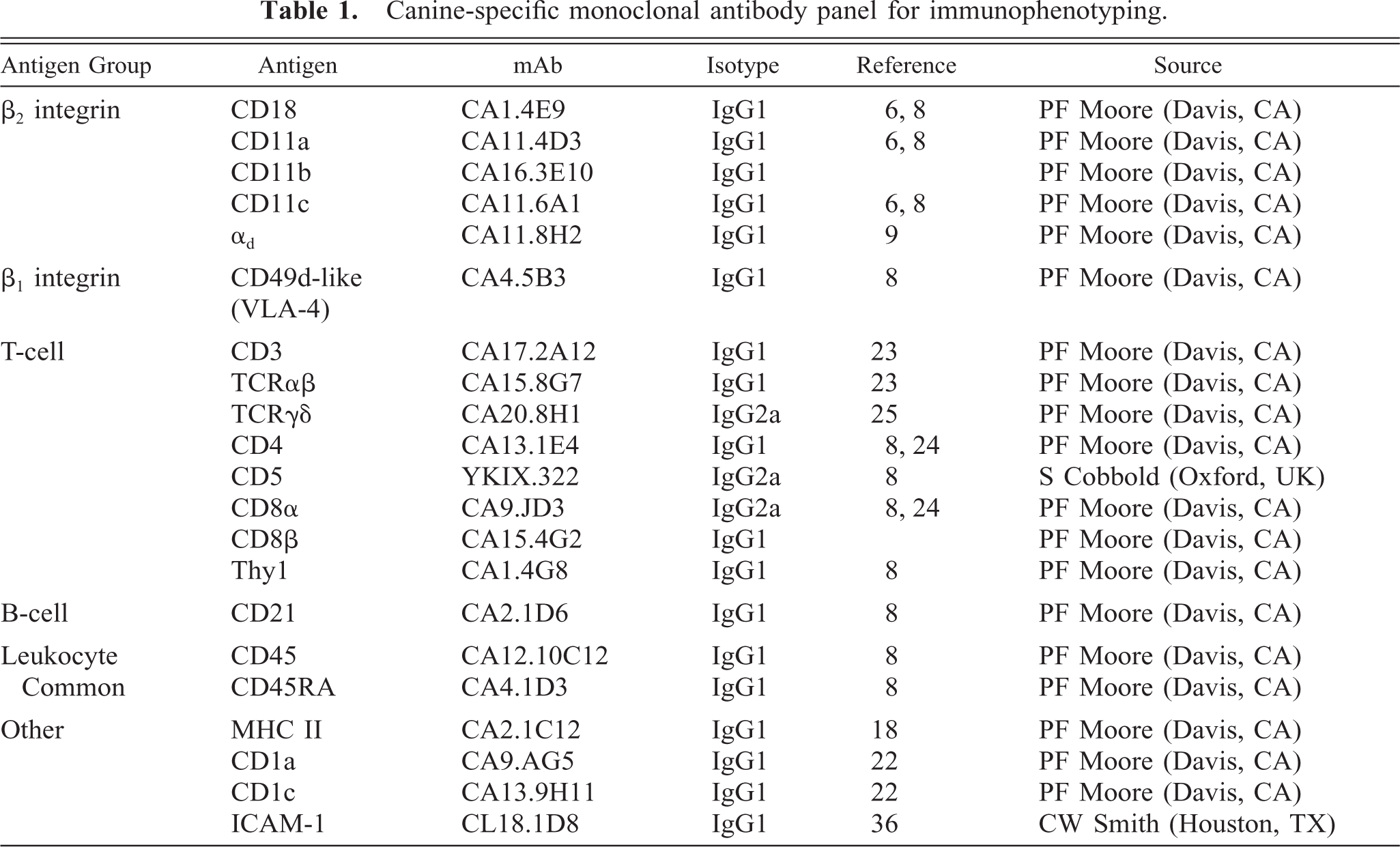
The source, specificity, and isotype of the canine-specific monoclonal antibodies (mAbs) used in this study are summarized in Table 1.
Canine-specific monoclonal antibody panel for immunophenotyping.
Immunohistologic staining
Representative samples of tissues available from three dogs were embedded in O.C.T. Compound (Miles, Elkhart, IN), snap frozen, and sectioned at 6 µm. Cytospins were made with 5 × 104 peripheral blood mononuclear cells isolated by density gradient centrifugation over Histopaque-1077 (Sigma, St. Louis, MO). Immunohistology was performed using a streptavidin-horseradish peroxidase technique according to manufacturer's instructions (Zymed, South San Francisco, CA) and previously described methods. 8 Primary mAbs were tissue culture supernatant diluted 1:10 for optimum reactivity as established by titration. Negative controls consisted of an isotype-matched irrelevant mAb or omission of the primary antibody.
Flow cytometry
Immunophenotyping was performed by incubating a 50-µl aliquot of whole blood with 25 µl of tissue culture fluid supernatant (except for FITC-conjugated YKIX 322, where 4 µl was used) for 30 minutes at room temperature. Erythrocytes were then lysed by addition of 3 ml ACK lysing solution. 15 MAb binding was detected by incubation with 50 µl of a 1:100 dilution of FITC-conjugated horse antimouse IgG (Vector Laboratories, Carpenteria, CA) for 20 minutes. If the stained cells could not be analyzed immediately, they were fixed with 2% (w/v) paraformaldehyde and stored at 4 C for no longer than 24 hours. Fluorescence was measured on 10,000 cells with a FACScan flow cytometer (Becton Dickinson, San Jose, CA). A lymphocyte gate was set on the basis of forward and side scatter light characteristics and data analyzed using Lysis II software (Becton Dickinson, San Jose, CA).
Results
Dog characteristics
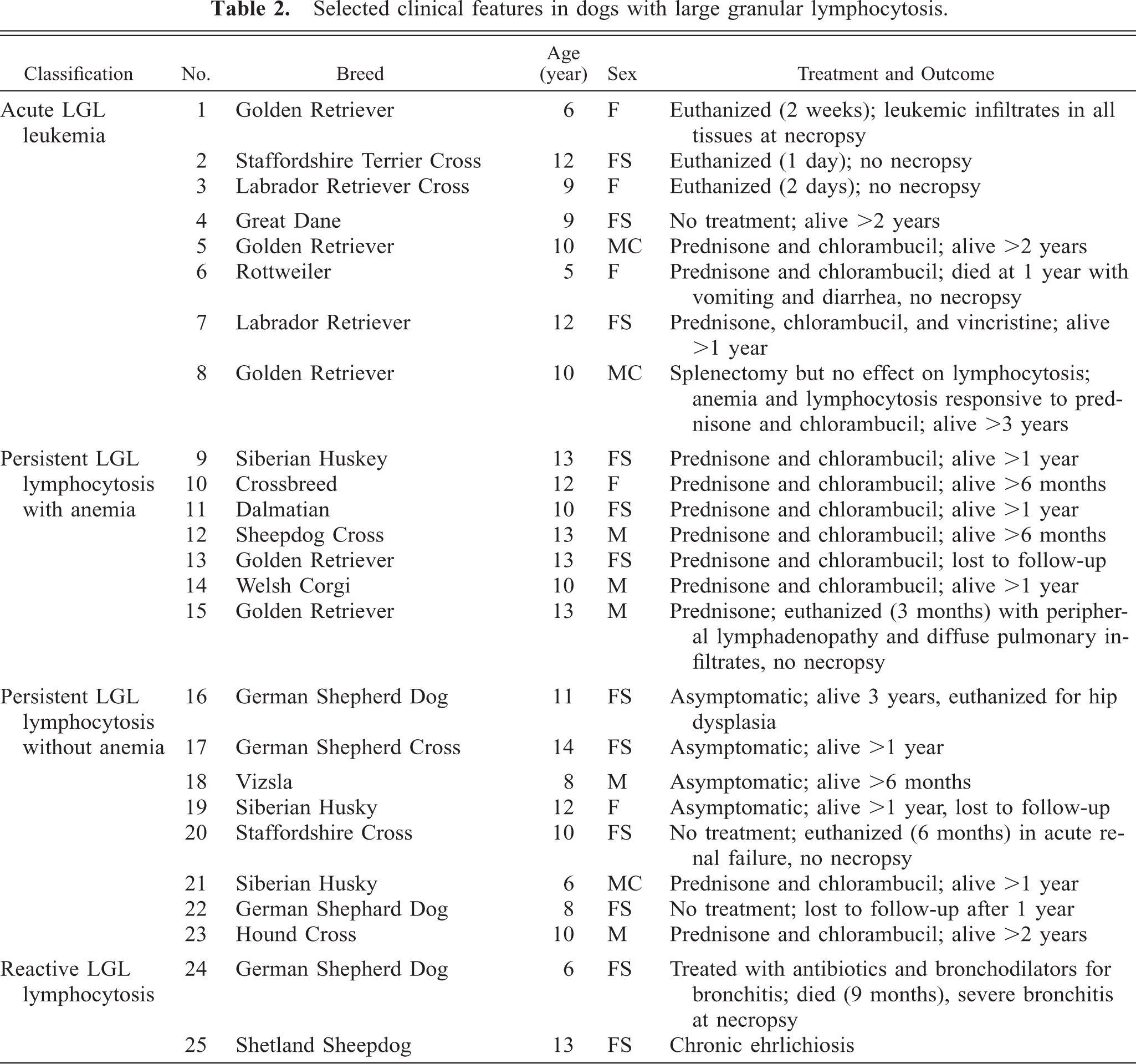
Affected animals were primarily middle aged and older large-breed dogs with an average age at initial diagnosis of 10 years (range 5–14 years). Females were affected almost twice as often as males (female:male ratio 1.8). Although 12 different breeds were represented, Golden Retrievers (n = 5) and German Shepherd Dogs (n = 4) accounted for over a third of all cases (Table 2). Except for three dogs that presented with acute aggressive clinical disease (dog Nos. 1–3), all dogs in this study had persistent (>3 months) LGL lymphocytosis.
Selected clinical features in dogs with large granular lymphocytosis.
Clinical and hematologic findings
Examination of Wright–Giemsa-stained blood films showed that in all but two instances (dog Nos. 24 and 25) over 50% of the lymphocytes were LGLs (average 88 ± 18.7%). Canine LGLs had abundant pale-staining cytoplasm with 3–20 azurophilic cytoplasmic granules. Granules were generally prominent, but granularity varied from case to case and often among cells from the same dog (Fig. 1). In four dogs, lymphocytes were infrequently granulated but otherwise had the cytologic characteristics of LGLs. Nuclear morphology also varied but was fairly homogeneous among cells from a single dog. Nuclei were usually round or reniform, although multilobular deeply cleaved nuclei with clover-leaf configurations were noted in two dogs. Nuclear chromatin was irregularly clumped and nucleoli were not visible.
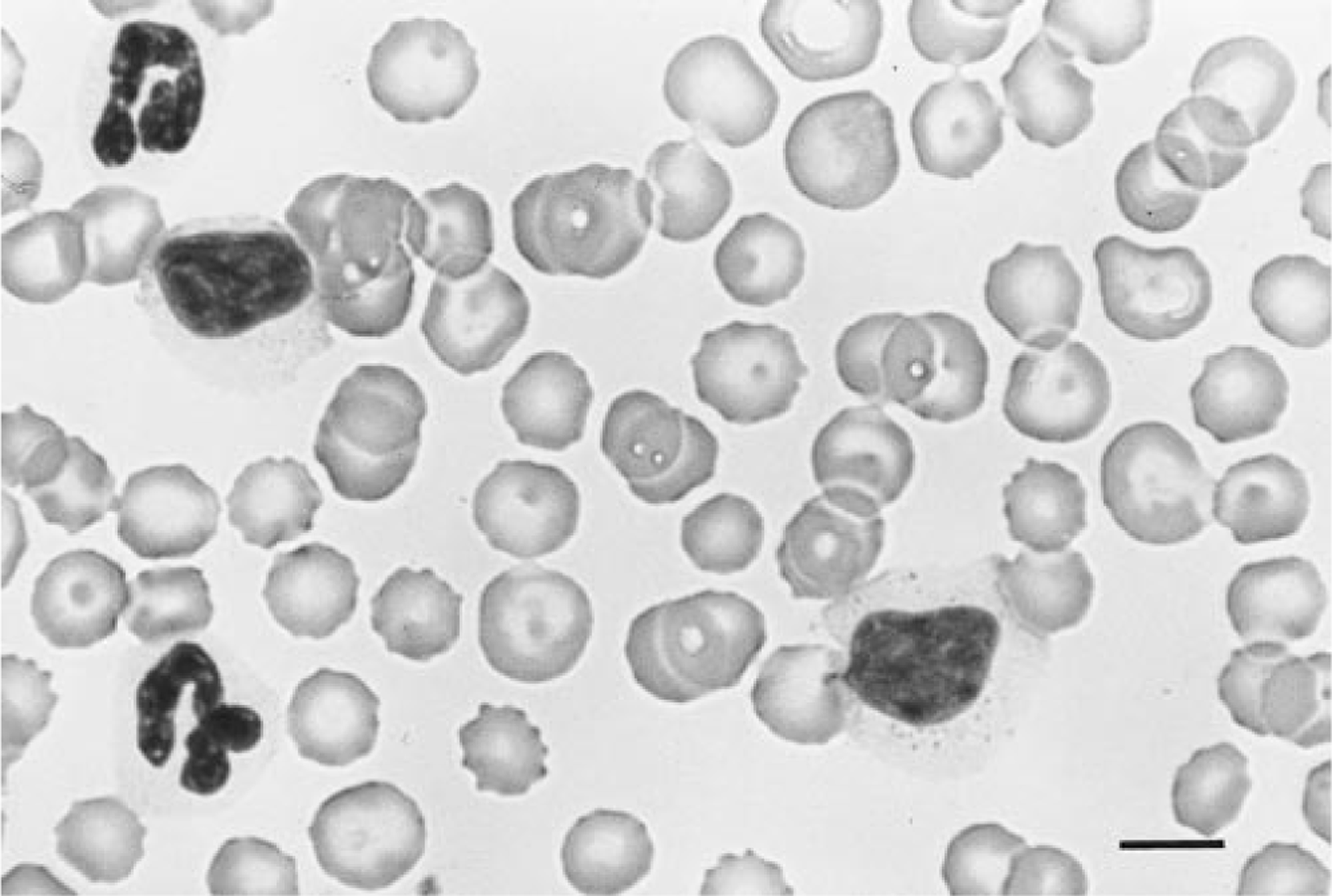
Blood smear, dog No. 1. Canine LGLs have abundant pale cytoplasm and 3–20 azurophilic cytoplasmic granules. Note that the size and number of granules varies between the two LGLs. Wright-Giemsa. Bar = 10 µm.
Of the 21 dogs that were clinically ill at initial diagnosis, 15 (71%) were anemic (dog Nos. 1–15). Bone marrow aspirates were evaluated from eight cases (dog Nos. 1, 3–7, 9, and 19). Three were normal (dog Nos. 4, 6, and 19), while the other five had concurrent myeloid hyperplasia, erythroid hypoplasia, and lymphocytosis. However, the degree of lymphocytic infiltration of bone marrow did not appear to be sufficient to cause myelophthisis. These results were supported by the hemograms. Neutrophil counts were within the expected range (3,000–11,500/µl) in seven dogs (28%), including four dogs that were asymptomatic (dog Nos. 16–19), and 17 (68%) were neutrophilic. The only neutropenic dog presented with an LGL count in excess of 200,000/µl accompanied by anemia and thrombocytopenia, compatible with myelophthisis (dog No. 10).
Splenic size was evaluated in 20 of 25 dogs, and splenomegaly was reported in 12 cases (60%). Palpable splenomegaly was found in nine dogs (dog Nos. 1–3, 7, 9–11, 15, 24), while an enlarged spleen was detected in three others by abdominal ultrasound (dog Nos. 4, 8, 13). The actual incidence of splenomegaly associated with LGL lymphocytosis was likely underestimated, as splenic size was not examined in five dogs and the eight dogs without palpable splenomegaly were not evaluated ultrasonographically.
The dogs were divided into four groups based on similar clinical and hematological findings (Table 3). Group 1 (acute LGL leukemia) consisted of dog Nos. 1–3, with rapidly progressive disease characterized by anemia, thrombocytopenia, and hepatosplenomegaly. Dog No. 3 had concurrent generalized lymphadenopathy. All were euthanized within 1 day to 2 weeks of initial diagnosis, and necropsy of dog No. 1 demonstrated lymphocytic infiltrates in all tissues. Group 2 included dog Nos. 4–15, with persistent (>3 months) LGL lymphocytosis accompanied by mild to moderate anemia and variable, but generally mild, thrombocytopenia. All 12 presented with lethargy, 5 had palpable splenomegaly, and 1 had mild generalized lymphadenopathy (dog No. 15). Three dogs had LGL counts in excess of 200,000/µl (dog nos. 6, 8, 9), while the other nine had LGL counts that averaged 51,500/µl (range 20,900–70,300/µl). Serology for Ehrlichia canis was negative in the five dogs that were tested. Ten dogs were treated with corticosteroids and chlorambucil with good to excellent results. Clinical signs, anemia, and increased LGLs recurred in seven dogs when the therapy was tapered or withdrawn, but all improved when treatment was reinstituted. The observed clinical remissions, characterized by increased activity levels and hematocrit, corresponded with decreased numbers of LGLs, suggesting a causative role for LGL lymphocytosis in the clinical symptomatology. All but two of these dogs are still alive, with survival times ranging from 6 months to 3 years.
Comparison of selected hematologic parameters from 25 dogs with large granular lymphocytosis.∗
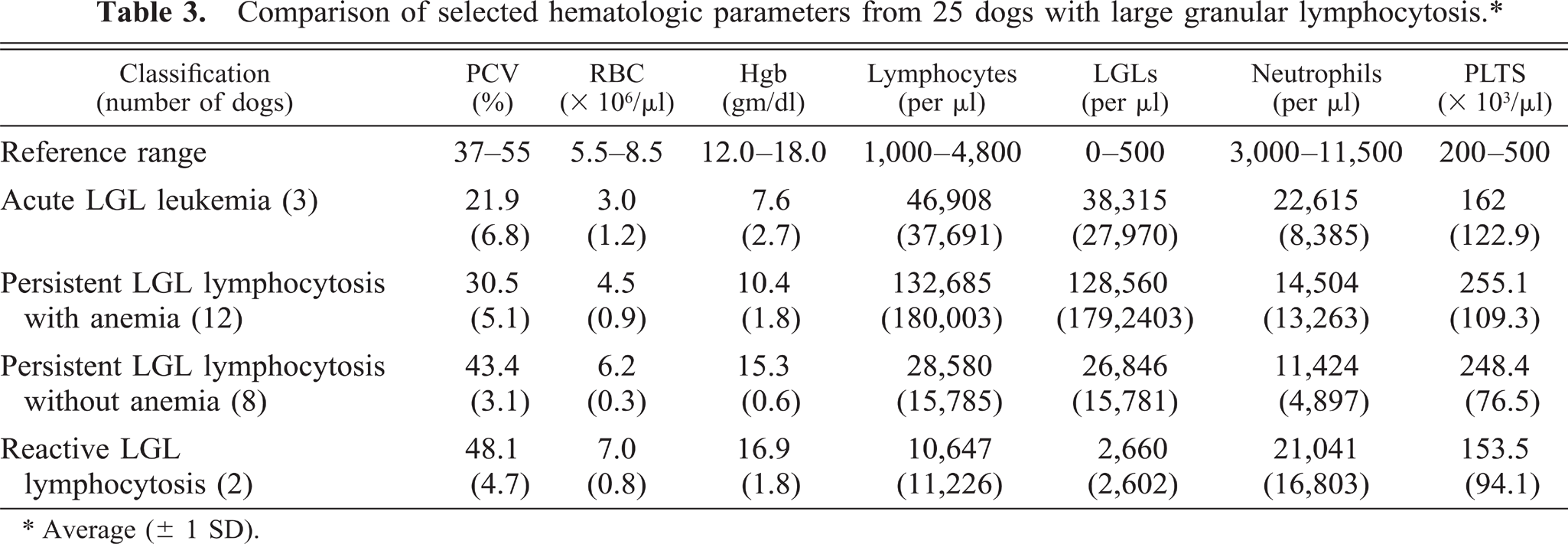
Average (± 1 SD).
Eight dogs in group 3 had persistent LGL lymphocytosis but were not anemic at initial diagnosis. Four dogs in this group (dog nos. 16–19) were asymptomatic, and their lymphocytosis was detected during routine hematological screening. These four dogs received no treatment, except dog no. 16, which was treated intermittantly with prednisone when mild thrombocytopenia developed. All four dogs remained in good health for periods ranging from 6 months to 3 years. Dog no. 16 was euthanized after 3 years, and immunohistology revealed the red pulp was diffusely infiltrated by large numbers of CD8+ cells (Fig. 2). The other four dogs in group 3 suffered an indolent clinical course characterized by lethargy, anorexia, and weight loss.
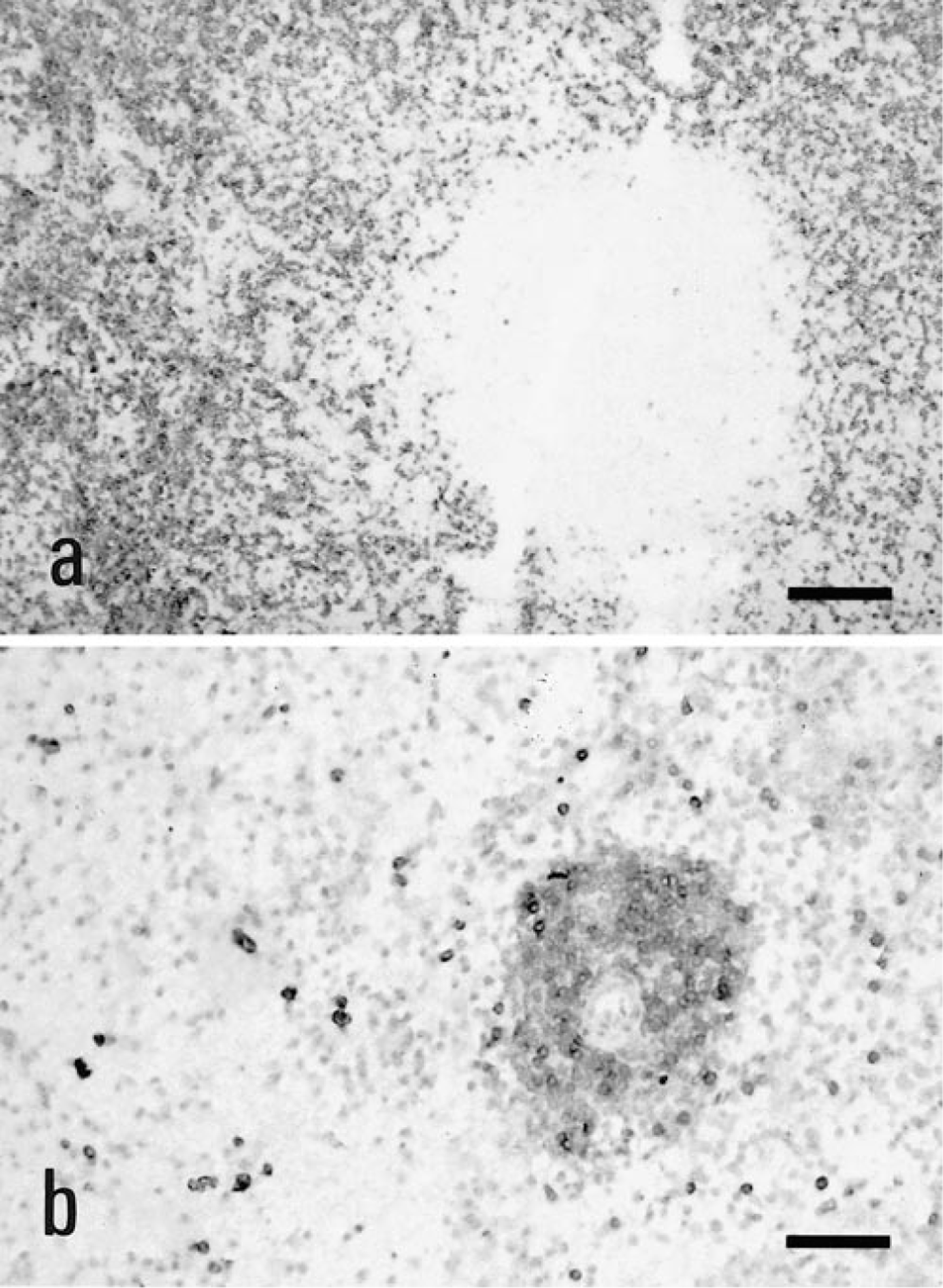
Spleen, dog No. 16. Immunohistology on frozen sections of spleen from a dog with persistent LGL lymphocytosis (A) compared with spleen from a normal dog (B). Both spleens were stained with monoclonal antibody CA9.JD3 specific for CD8α. Note the diffuse red pulp infiltration by large numbers of CD8+ cells, which distorted, but did not efface, normal splenic architecture. The clear space in panel A is a lymphoid follicle. Serial sections demonstrated these cells were immunophenotypically identical to the circulating LGLs. The chromogen was nickel-enhanced 3,3′-diaminobenzadine. Bar = 250 µm.
Group 4 consisted of two dogs with probable reactive LGL lymphocytosis. Both had increased numbers of circulating LGLs, but only 25–30% of the PBLs had LGL morphology. Dog no. 24 had chronic bronchitis and died after 9 months. Dog no. 25 had thrombocytopenia (87,000 platelets/µl) and chronic ehrlichiosis, confirmed by serology and polymerase chain reaction (D. Gebhard, personal communication).
Immunophenotypic analysis
Leukocyte antigen expression in canine LGL lymphocytosis is summarized in Fig. 3. Analysis of T-cell receptor (TCR) usage identified three lineages of canine LGLs. Most LGL lymphocytosis cases (60%) were characterized by proliferation of αβ T-cells, but an unexpectedly large proportion (32%) were associated with proliferation of γδ T-cells. Lymphocytes from two dogs (8%) with LGL lymphocytosis were CD3−. CD8α was expressed by LGLs in all 25 dogs, while CD8β was concurrently expressed in 19 cases (76%). LGLs from both CD3− cases were CD8α+/CD8β−. The high molecular weight isoform of leukocyte common antigen, CD45RA, was expressed by LGLs in all but one dog. In this instance, LGLs were also negative for CD45, suggesting they were devoid of any CD45 isoform.
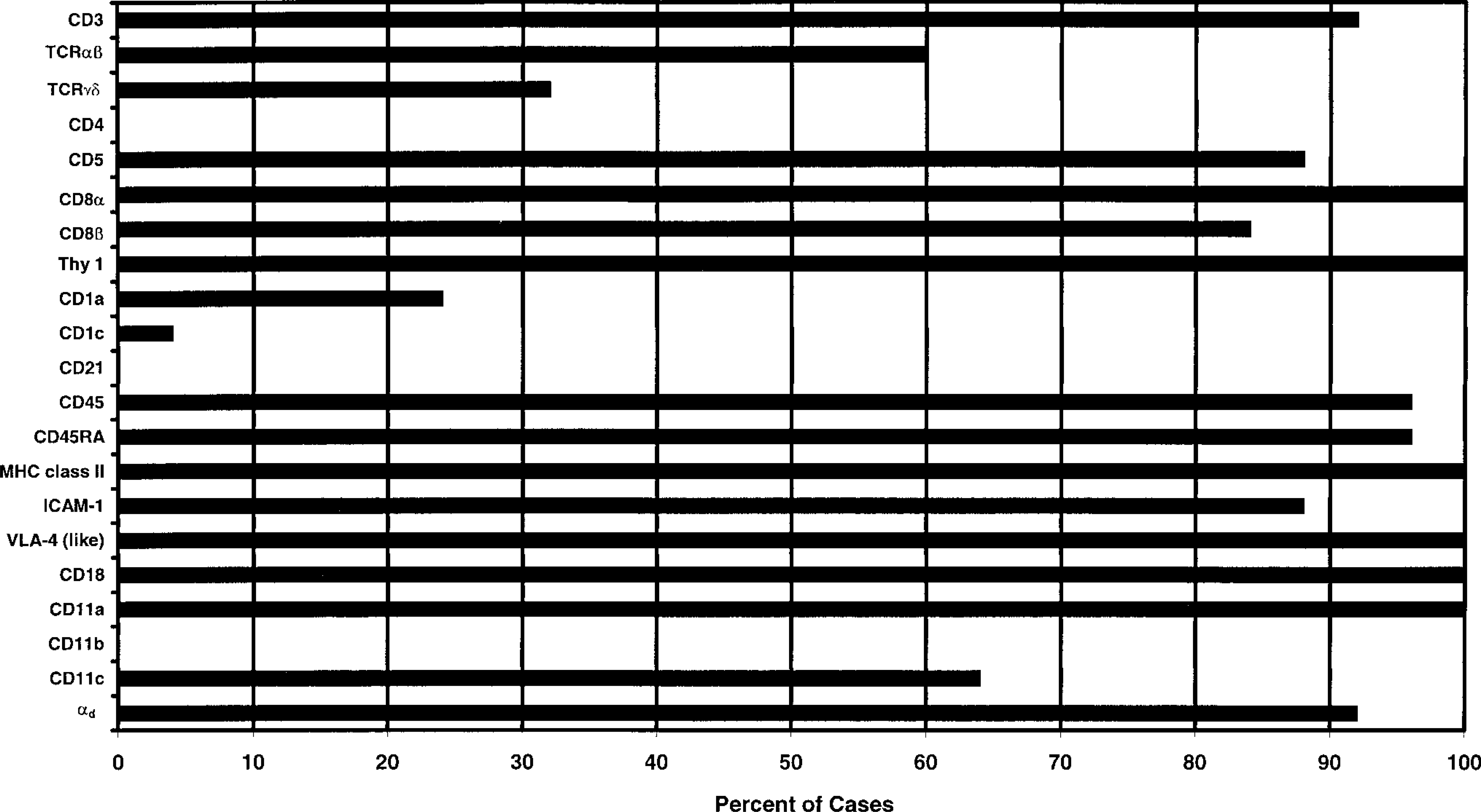
Summary of leukocyte antigen expression in canine LGL lymphocytosis.
The pattern of β2 integrin expression by canine LGLs was distinctive (Fig. 4). αd was expressed by LGLs in 92% of all cases, including 14 of 15 (94%) TCRαβ LGLs, 7 of 8 (88%) TCRγδ LGLs, and both CD3− LGL cases. As expected, LGLs in all cases expressed CD11a, but strikingly, CD11b expression was absent. Interestingly, CD11c was expressed by LGLs in 64% of all cases, but the intensity of expression was variable and weak compared with αdβ2. Immunocytology on cytospins found αdβ2 expression on the cell surface and in prominent cytoplasmic foci clustered in the nuclear indentation (Fig. 5). No evidence of cytoplasmic expression of CD11a, CD11b, or CD11c was found.
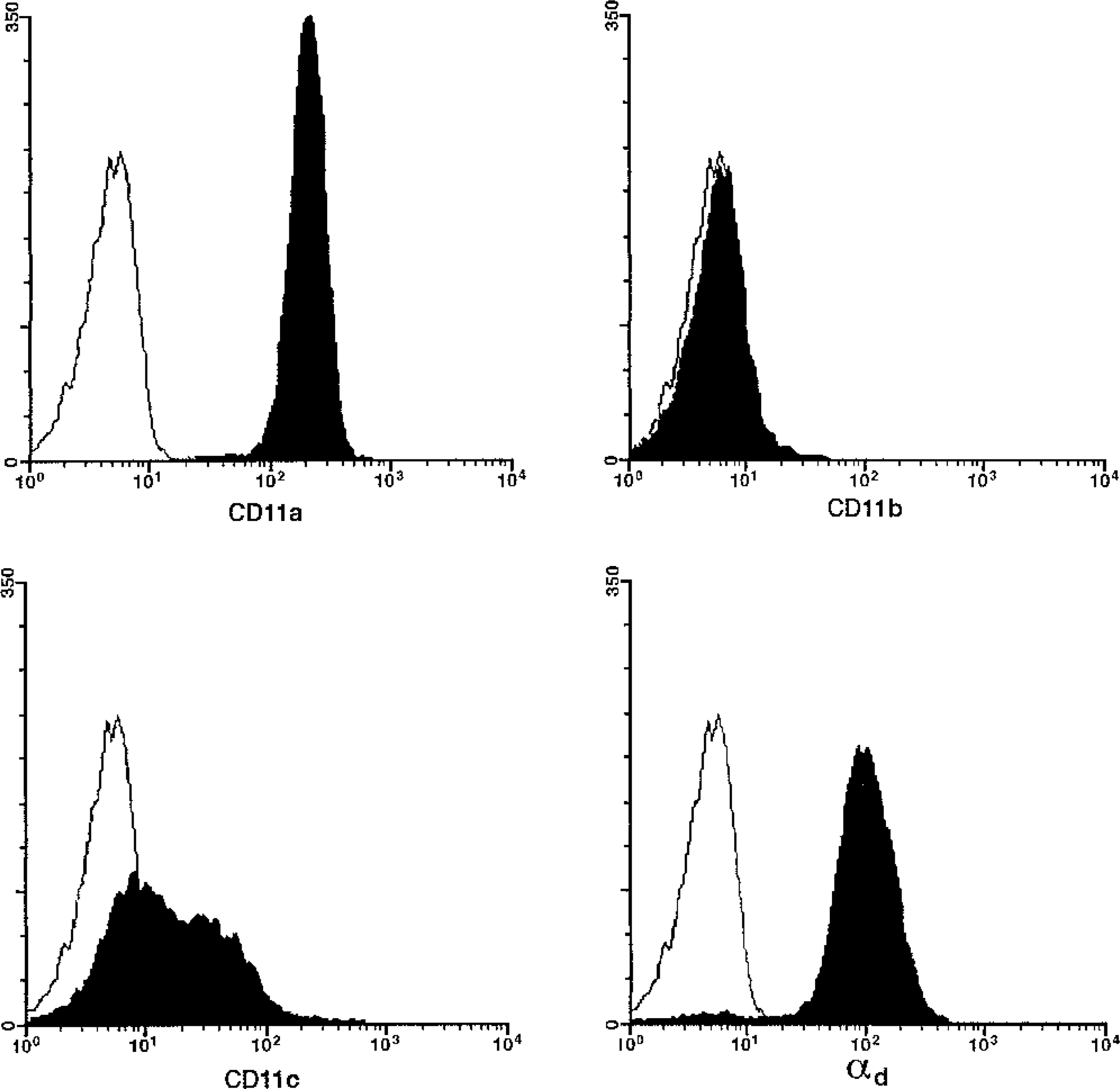
β2 integrin expression patterns, dog No. 16. Single-parameter FITC fluorescence histograms from a representative case of canine LGL lymphocytosis. The X-axis is relative fluoresence and the Y-axis is cell number. The histograms represent the lymphocyte gate established on the basis of forward and side scatter light characteristics. The unfilled histograms represent fluorescent staining with the isotype-matched irrelevant negative control, whereas the filled histograms represent staining with antibody specific for CD11a, CD11b, CD11c, and αdβ2, respectively. All cases expressed CD18/CD11a, while none expressed CD11b. CD11c was expressed by 64% of all cases, but the intensity of expression was usually broad and dim compared to the uniformly bright expression of the leukointegrin αdβ2.
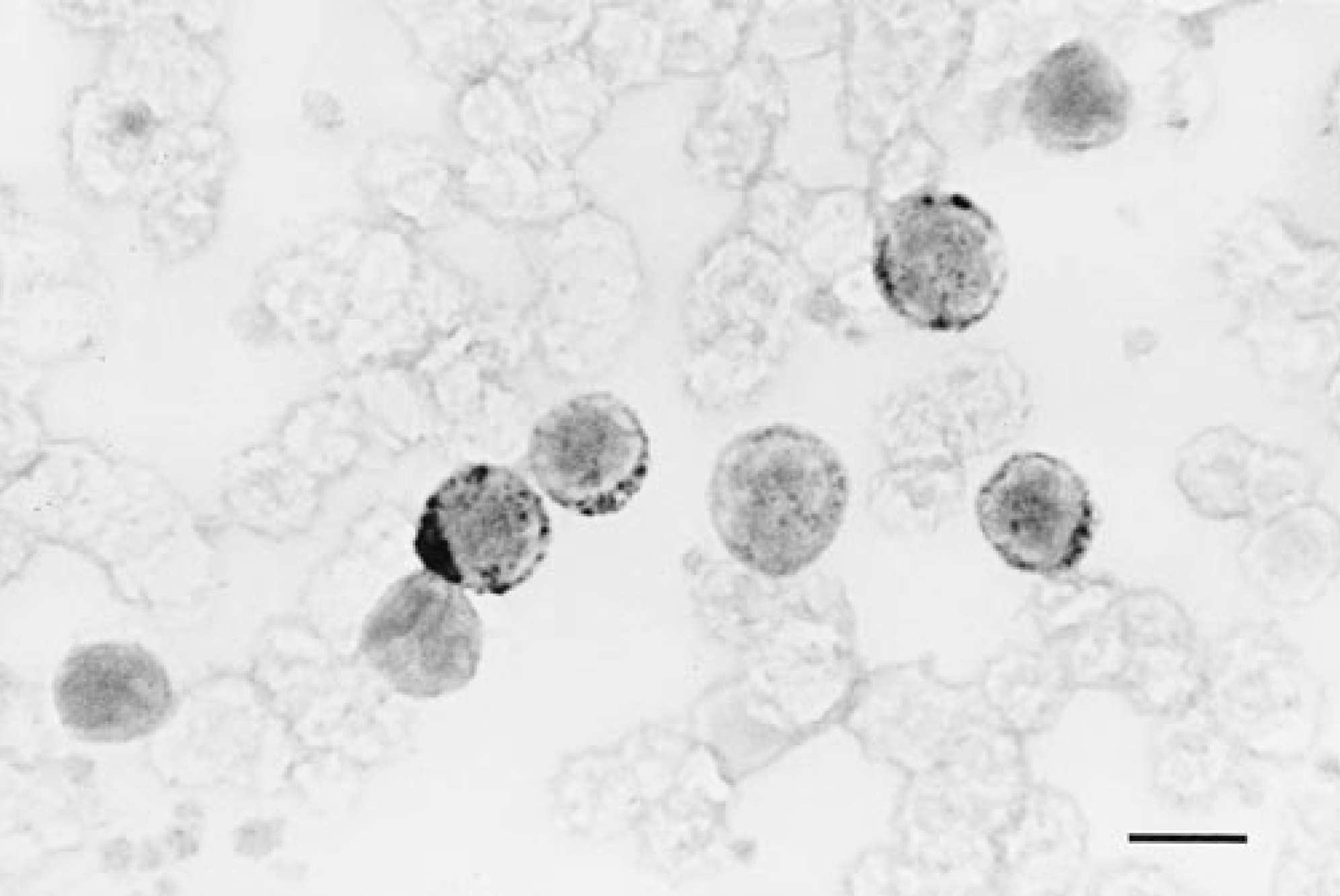
Peripheral blood, dog No. 8. Immunohistology on a peripheral blood smear from a dog with persistent LGL lymphocytosis stained with monoclonal antibody CA11.8H2, specific for the leukointegrin αdβ2. Note the intense reactivity in the cytoplasm often localized in clusters near the nucleus. The chromogen was aminoethylcarbozole and the slide was counterstained lightly with Gill's hematoxylin. Bar = 10 µm.
Immunophenotyes were homogeneous among cells from the same dog in all but the two cases of probable reactive LGL lymphocytosis (group 4). Since only 25–30% of the PBLs in dog nos. 24 and 25 had LGL morphology, immunocytology was performed on cytospins of peripheral blood mononuclear cells. LGLs from dog no. 24, with chronic bronchitis, were γδT-cells. Two-thirds of these LGLs expressed CD8α, while the remainder of the LGLs were negative for CD4, CD8α, and CD8β. The LGL immunophenotype from dog no. 25, with chronic ehrlichiosis, was also heterogeneous: 65% were γδ T-cells and the remaining 35% were αβ T-cells.
Discussion
The development of mAbs specific for canine leukocyte differentiation antigens has greatly aided the detailed analysis and histopathologic classification of lymphoproliferative diseases in dogs. 22 23 25 33 41 These studies confirm many similarities to human disease but also highlight important differences. 22 Using a similar approach, we found that the clinical, immunophenotypic, and pathologic features of canine LGL lymphocytosis resembled the human disease but with several distinctions. Like people, affected dogs displayed a broad clinical spectrum ranging from an indolent, occasionally asymptomatic condition, to aggressive malignancy. Canine LGLs represented diverse lineages of lymphocytes including αβ T-cells (60%), γδ T-cells (32%), and non-T, non-B lymphocytes, thought to be NK cells (8%). Regardless of lineage, the leukointegrin αdβ2 was expressed with high frequency (92%). αdβ2 + γδ T-cells represent less than 1% of circulating T-cells in normal dogs. In contrast, over 30% of T-cells in the normal canine spleen display this phenotype, and they localize almost exclusively in the red pulp (manuscript in preparation). The unexpectedly large proportion of γδ T-cell LGLs, the prominent expression of αdβ2, relative sparing of the bone marrow, and frequent splenomegaly suggest that canine LGL lymphocytosis arises in the splenic red pulp.
The large majority of human circulating LGLs are CD3− NK cells. 32 In contrast, most human LGL lymphocytosis cases are CD3+ while a smaller proportion are CD3− NK cell LGLs, 30 but NK cell LGL abnormalities may be more common than suggested in the literature. 34 Many distinctive immunophenotypes have been described, but Scott and Richards subdivide human T-cell LGL lymphocytosis into four main groups: 34 TCRαβ+/CD8+/CD4− (62%), TCRαβ+/CD8−/CD4− (2%), TCRαβ+/CD8− or dim/CD4+ (28%), and TCRγδ+/CD8− or +/CD4− (8%). The majority of canine LGL lymphocytosis patients also had CD3+ T-cell lymphocytosis (23/25) but displayed only two subtypes, TCRαβ+/CD8+/CD4− (65%) and TCRγδ+/CD8+/CD4− (35%). The lineage of the two CD3− cases was uncertain. Both expressed CD8α but not CD8β. No canine-specific NK cell markers are available, but we believe these are similar to a subset of human CD8α+ NK cells. 42 Alternatively, these CD3− LGLs may be T-cells that lost cell surface expression of the T-cell receptor complex during neoplastic transformation. 14
A polyclonal pattern of TCR gene rearrangement, compatible with a reactive process, is found in 10% of human T-cell LGL lymphocytosis patients, often in association with other diseases such as tuberculosis, rheumatoid arthritis, or neoplasia. 12 35 Canine TCR gene rearrangement analysis was unavailable, but a similar proportion of canine LGL lymphocytosis cases (group 4) were thought to be reactive due to the presence of immunophenotypically heterogeneous LGLs. Both dogs had expansions of γδ T-cell LGLs; in addition, one also had αβ T-cell LGLs. One dog had ehrlichiosis confirmed by serology and polymerase chain reaction (D. Gebhard, personal communication). Prolonged LGL lymphocytosis was previously described in a subset of dogs with chronic ehrlichiosis. 48 Interestingly, proliferation of a normally minor population of γδ T-cells occurs in people being treated for ehrlichiosis. 4 Taken together, these results provide strong circumstantial evidence that chronic canine ehrlichiosis can cause persistent γδ T-cell LGL lymphocytosis. The possibility that other infectious agents also cause LGL lymphocytosis in dogs awaits further study.
The other 90% of human T-cell LGL lymphocytosis cases are leukemias with monoclonal rearrangements of TCR genes. 18 20 These patients generally have an indolent clinical course with mild to moderate LGL lymphocytosis, neutropenia, anemia, and mild to moderate splenomegaly. 12 Bone marrow infiltration is usually sparse and accompanied by erythroid hypoplasia and myeloid maturation arrest. Morbidity is related to cytopenias rather than tumor burden. The largest group of dogs (group 2) in our study also had an indolent clinical course with moderate splenomegaly and anemia. Bone marrow aspirates showed erythroid hypoplasia and lymphocytosis, but the degree of lymphocytic infiltration was not sufficient to cause myelophthisis. The neutropenia that occurs in human LGL leukemia 12 30 is in striking contrast to the myeloid hyperplasia and neutrophilia seen in canine LGL lymphocytosis. These results suggest LGL infiltration of bone marrow in dogs leads to selective suppression of erythropoiesis, but not myelopoiesis, by an unknown mechanism. An inhibitory effect on erythropoiesis by human LGLs has been demonstrated in vitro, 17 most likely due to LGL-produced γ-interferon, which is a potent inhibitor of murine erythroid colony growth. 46 The neutropenia in some human patients has been corrected by granulocyte-macrophage colony-stimulating factor administration. 16
The eight dogs in group 3 also had persistent LGL lymphocytosis but were not anemic at initial diagnosis. Four dogs in this group had nonprogressive asymptomatic LGL lymphocytosis, and we speculate that these dogs had benign clonal expansions of LGLs analogous to older people with benign CD8+ T-cell clonal proliferation. 10 Except for the lack of anemia at initial diagnosis, the other four dogs in group 3 most resembled group 2. Comparison of the cytokine repertoire among canine LGLs and with human LGLs may shed light on possible mechanisms for the observed variation in hematological abnormalities.
Clonality in human NK cell LGL lymphocytosis has been assessed by demonstration of cytogenetic abnormalities or by X-linked DNA analysis, 5 40 but clonality is not proven in most cases. Some patients with CD3− NK cell LGL lymphocytosis have a chronic clinical course with no evidence of clonality, suggestive of a reactive process. 5 12 Human patients with clonal proliferation of CD3− LGLs generally have an acute clinical presentation with aggressive disease and massive hepatosplenomegaly associated with Epstein-Barr virus infection. 13 A small proportion of patients with clonal CD3+ LGL lymphocytosis also present with aggressive clinical disease. Interestingly, the LGLs from these patients are CD56+, similar to the more aggressive NK cell LGL leukemia, suggesting CD56 expression may be of prognostic importance. 11 One dog with putative NK cell LGL lymphocytosis presented with acute leukemia (group 1), while the other had persistent LGL lymphocytosis with anemia (group 2). Due to the lack of canine NK cell markers and the small number of cases studied to date, it is unknown if a distinct clinicopathologic entity similar to humans with clonal expansions of NK cell LGLs exists in dogs.
As classically defined, leukemia is malignant neoplasia of hematopoietic cells in the bone marrow with spillover into the blood. 7 While these dogs presented with leukemia, we do not believe they had primary marrow disease. The bone marrow findings imply canine LGL lymphocytosis arises from an extramedullary site, and the high frequency of splenomegaly and αdβ2 expression associated with LGL lymphocytosis may have relevance in this regard. Expression of αdβ2, which is highly restricted in normal dogs, is pronounced in the red pulp of the spleen on macrophages 9 and diverse lineages of lymphocytes including αβ T-cells, γδ T-cells, and perhaps NK cells (manuscript in preparation). Additionally, it is interesting to note the relatively high proportion of γδ T-cell LGLs found in this study. Although it is not known what proportion of canine γδ T-cells have LGL morphology, fewer than 1% of peripheral blood lymphocytes in normal dogs are αdβ2 + γδ T-cells. However, over 30% of canine splenic T-cells are αdβ2 + γδ T-cells (manuscript in preparation), and αdβ2 + lymphocytes localize almost exclusively in the red pulp. 9 Immunohistology on the spleen of two dogs revealed diffuse infiltration of the splenic red pulp while other lymphoid organs, including bone marrow, were less involved. Although leukemias can invade the spleen as a secondary site, it seems likely canine LGL lymphocytosis arises from αdβ2 + lymphocytes localized in the splenic red pulp. DNA content analysis in human cases shows that the proliferating LGLs are located in the spleen. 29 35 Splenic involvement is a prominent feature of the Fischer 344 rat model of LGL leukemia, and splenic origin of LGL proliferation has been suggested in this disease. 39 47 Interestingly, Fischer 344 rat LGLs are NK cells that express αdβ2 (G. Dietsch, personal communication).
β2 integrin expression patterns on canine LGLs differed from those reported for people and mice. All human and murine LGLs have CD11a, but CD11b (70–96%) and CD11c (47–66%) are less prevalent. 43 44 Expression of αdβ2 by LGLs in species other than the dog has not yet been reported. Similar proportions of canine LGLs expressed CD11a and CD11c, but CD11b expression was notably lacking while αdβ2 was expressed with high frequency (92%).
The significance of variation in β2 integrin expression patterns is uncertain but most likely reflects altered states of activation, with different activation signals inducing different cell surface antigens. 31 50 Cytokines increase β2 integrin surface expression by both de novo synthesis and mobilization of CD11b and CD11c from stores contained in cytoplasmic granules. 27 45 The signals regulating canine β2 integrin expression are largely unknown. In vitro canine LGLs down regulate αd expression, and supplementation with recombinant human IL-2 accelerates the decline (unpublished observations).
β2 integrins mediate specific but overlapping functions that contribute to the recruitment and retention of LGLs in tissue and are critical for stable conjugate formation between cytotoxic lymphocytes and targets. 2 β2 integrin-mediated functions include adhesion to vascular endothelium (CD11a, b), cell spreading, random migration and chemotaxis (CD11a, b, c), conjugate formation with targets (CD11a, b, c), and antibody-dependent cell cytotoxicity (CD11a, b, c). 1 The hierarchical use of these adhesion pathways is likely determined by the state of cellular activation. For example, CD11a is most important in endogenous and IL–2-stimulated killing. 43 In contrast, random migration of unstimulated LGLs is mediated by CD11b, but after stimulation with either IL-2 or ICAM-2, CD11a is the major receptor utilized in migration. 37 38 Treatment of canine peripheral blood mononuclear cells in vitro with an anti-CD18 mAb markedly decreases LGL lytic function due to defective effector-target conjugate formation. 19 Although the functions(s) and ligand(s) for canine αdβ2 are unknown, prominent αdβ2 expression compared with absent or weak CD11b and CD11c expression suggests this leukointegrin plays an important role in canine LGL biology and may reflect the origin of LGLs from the splenic red pulp.
Conclusion
Canine LGL lymphocytosis was a clinically heterogeneous disease that we believe arose in the red pulp of the spleen from diverse lineages of αdβ2 + lymphocytes including αβ T-cells, γδ T-cells, and perhaps NK cells. Assessment of clonality by T-cell receptor gene rearrangement analysis would be very helpful in assessing the nature of these cases of LGL lymphocytosis. Long-term follow-up with multiparameter analysis including clinical, hematologic, immunophenotypic, cytogenetic, functional, and TCR gene rearrangement studies will be useful in understanding the pathobiology of this disease and in prescribing treatment of affected dogs. Regardless of lineage, the β2 integrin αdβ2 was expressed with high frequency while CD11b and CD11c were absent or expressed only weakly. These results suggest that further research into the role this leukointegrin plays in canine LGL biology is warranted.
Footnotes
Acknowledgements
We gratefully acknowledge the assistance of Drs. Sonjia M. Shelly and William Vernau of IDEXX (Sacremento, CA). This study was supported in part by a grant from ICOS Corporation (Bothell, WA).
