Abstract
The combination of flow cytometric scatterplot analysis and specific monoclonal antibodies was used to evaluate the lineage of cells from six dogs with proliferative disorders of bone marrow. Scatterplot analysis was used to identify mature and immature myeloid and erythroid cells. The immunophenotype of cells in the immature myeloid gate was determined by labeling cells with four monoclonal antibodies. These results were compared to results of cytologic and cytochemical evaluation. The immunophenotype of a dog with a diagnosis of myelogenous leukemia was a cluster of differentiation–18 (CD-18) positive, CD-14 negative, Thy-1 negative, and a major histocompatibility complex (MHC) class II negative. The immunophenotype of a dog with a diagnosis of myelomonocytic leukemia was CD-18 positive, CD-14 positive, Thy-1 positive, and MHC class II positive. Although this phenotype clearly differentiated myelomonocytic leukemia from myelogenous leukemia, it was similar to the immunophenotype of dogs with a diagnosis of malignant histiocytosis or hemophagocytic syndrome. The immunophenotype of two dogs with myelodysplastic syndrome was CD-18 positive and CD-14 negative. Results for Thy-1 and MHC class II were variable. As additional lineage-specific monoclonal antibodies become available, immunophenotyping should become a valuable tool for determination of the lineage of cells in canine myeloproliferative disorders.
Proliferative disorders of canine bone marrow encompass a heterogeneous group of disorders, including acute and chronic myelogenous leukemia, acute and chronic myelomonocytic leukemia, acute and chronic lymphocytic leukemia, monocytic leukemia, acute undifferentiated leukemia, and myelodysplastic syndromes. 5,8,9,13 Other proliferative disorders of monocytes and macrophages in bone marrow include malignant histiocytosis and hemophagocytic syndrome. 12,15,20 The use of cytomorphologic criteria alone is inadequate to differentiate the lineage of the proliferating cells. 5,9 The specificity of diagnosis is improved by use of cytochemical stains. However, the value of cytochemical staining is limited by the low number of diagnostic stains available, staining of more than one cell lineage, and variable staining of malignant cells. 5,8,9
Monoclonal antibodies provide a powerful tool for detecting lineage-specific markers on the surface of hematopoietic cells. 6,10,11,16 Application of immunophenotyping to domestic animals has been limited by lack of appropriate monoclonal antibodies directed toward specific cluster of differentiation (CD) antigens. Although antibodies are available to detect major CD antigens on lymphoid cells, few antibodies are available to identify the various stages of myeloid, erythroid, and megakaryocytic differentiation. 7,10
In previous studies, we used a flow cytometric scatterplot technique to identify subpopulations of myeloid and erythroid cells in nonmalignant canine bone marrow specimens. 21 A template was developed to identify immature myeloid cells, metamyelocytes, mature myeloid cells, immature erythroid cells, and mature erythroid cells. Bone marrow differential cell counts, obtained by the scatterplot method, compared well with manual differential cell counts. In the present study, we used a combination of the scatterplot technique and four monoclonal antibodies to evaluate the lineage of bone marrow cells from six dogs with bone marrow proliferative disorders. Results were compared to those obtained by cytologic and cytochemical analysis.
Materials and Methods
Collection and processing of bone marrow specimens
Bone marrow samples were obtained from six dogs that had bone marrow proliferative disorders. To provide reference values for immunophenotyping, bone marrow samples were also obtained from six healthy dogs. Bone marrow samples were aspirated into syringes that had been rinsed with 2% EDTA solution. Wedge-type smears were prepared immediately, and the remainder of the marrow sample was refrigerated and processed within 2 hours after collection. Fifty microliters of marrow specimen was placed in a 6-ml sterile plastic tube and 2 ml of erythrocyte lysis buffer (Erythrolyse solution, Serotec USA, Washington, DC), containing 0.5% paraformaldehyde, was added. After 10 minutes of incubation at room temperature, tubes were centrifuged and the cell pellet was washed and resuspended in 700 µl of Dulbecco's phosphate buffered saline solution containing 1% sheep serum and 2 mM sodium azide. Previous studies have shown that only nonnucleated erythrocytes are lysed by this procedure. 21 A 100-µl aliquot of cell suspension was placed into each of seven 6-ml sterile plastic tubes. Tubes were incubated with one of four monoclonal antibodies or with irrelevant isotype-matched control antibodies. Antibodies included anti-CD-18 (R 15.7 provided by Mark Jutila, Montana State University, Bozeman, MT), anti-Thy-1 (DH 24A, VMRD Inc., Pullman, WA), antimajor histocompatibility antigen class II (MHC class II; MCA 1085, Serotec USA) and anti-CD-14 (TuK4, Serotec USA). The anti-CD-14 antibody was conjugated with phycoerythrin-Cy5. The staining characteristics of these antibodies for canine bone arrow have been defined by use of fluorescence-activated cell sorting (FACS) (unpublished observations). After a 60 minute incubation at room temperature, cells were washed and resuspended in 100 µl of Dulbecco's phosphate-buffered saline solution containing 1% sheep serum and 2 mM sodium azide. With the exception of samples incubated with anti-CD-14, samples were incubated with 20 µl of a 1:20 dilution of fluorescein-conjugated sheep anti-mouse IgG (MCA 1095, Serotec USA) or rat anti-mouse IgM (MCA 199, Serotec USA) for 30 minutes. Thereafter, samples were diluted to 1.0 ml and were analyzed within 2 hours.
Bone marrow specimens were also double labeled with anti-MHC class II and anti-CD-14. Samples were successively labeled with anti-MHC class II, sheep antimouse IgG, 5% mouse serum, and anti-CD-14.
Flow cytometric techniques
Flow cytometric analysis was performed using a FACS Calibur flow cytometer (Becton Dickenson Co., Pleasanton, CA). For all samples, cell populations were displayed by forward-angle versus side-angle light scatter properties as described. 21 Forward scatter was set at 0.1 A, side scatter was set at 444 V, amp gain was set at 9.54, and fluorescence intensity was set at 425. The forward scatter threshold was set at 50 to eliminate platelets and small particulate debris. A total of 30,000 cells were analyzed for each bone marrow specimen. Scatterplots were evaluated by use of a template previously developed for differential cell counting (Fig. 1). 21 This template enabled identification of mature and immature erythroid cells and mature and immature myeloid cells. The percentage of cells within each gate was recorded. Cells within each gate were also analyzed by use of a second scatterplot with forward-angle light scatter on the x-axis and fluorescence intensity on the y-axis. Double-labeled samples were analyzed by scatterplots with green fluorescence intensity on the x-axis and red fluorescence intensity on the y-axis (Fig. 2). Fluorescent cells from some bone marrow samples were backplotted onto the forward-angle versus side-angle light scatterplots.
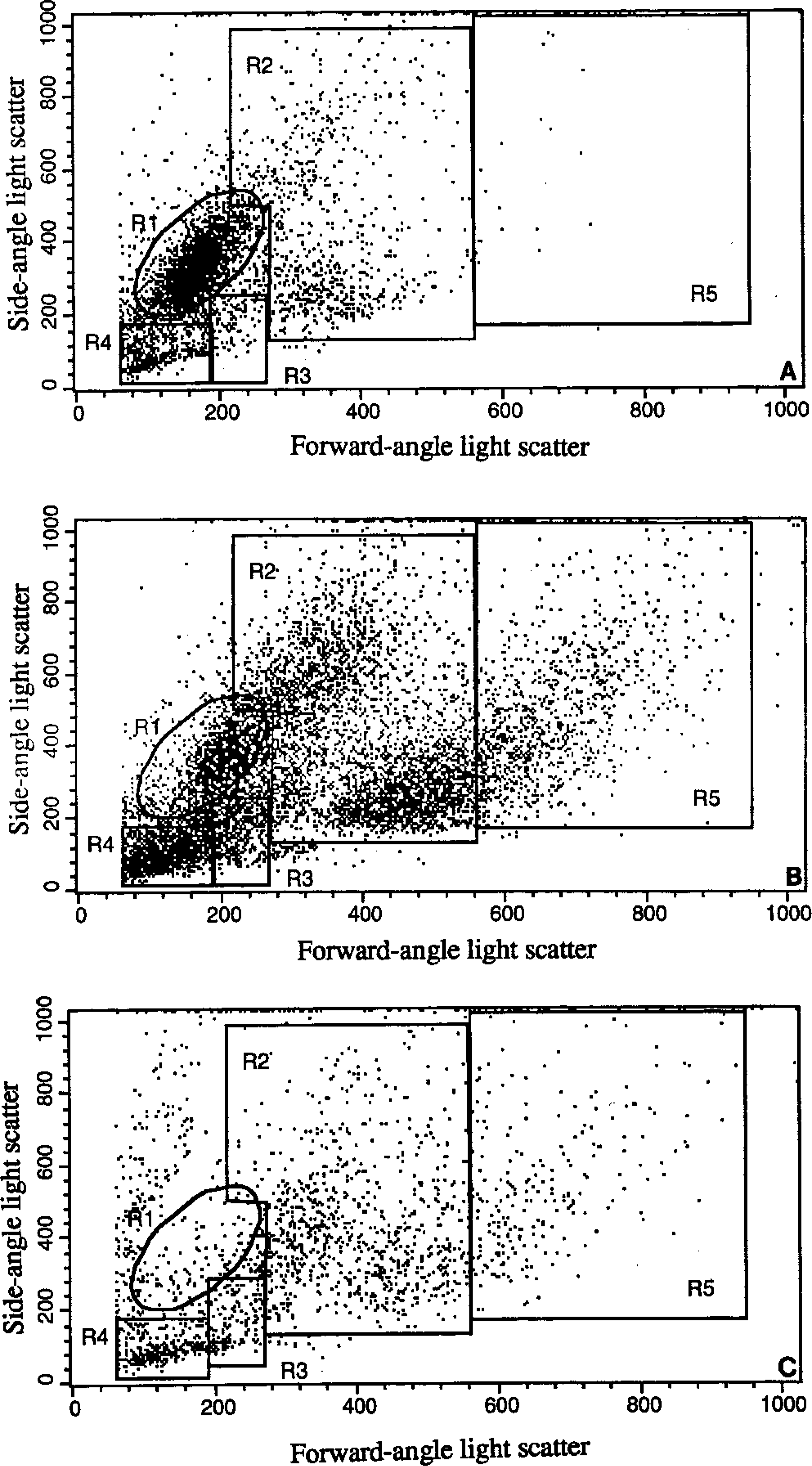
Flow cytometric forward-angle versus side-angle light scatterplot of canine bone marrow cells from a control dog (A), dogs with myelogenous leukemia (B), and malignant histiocytosis (C). Gates were set to identify mature myeloid cells (R1), immature myeloid cells (R2), immature erythroid cells (R3), mature erythroid cells (R4), and large cells (R5).
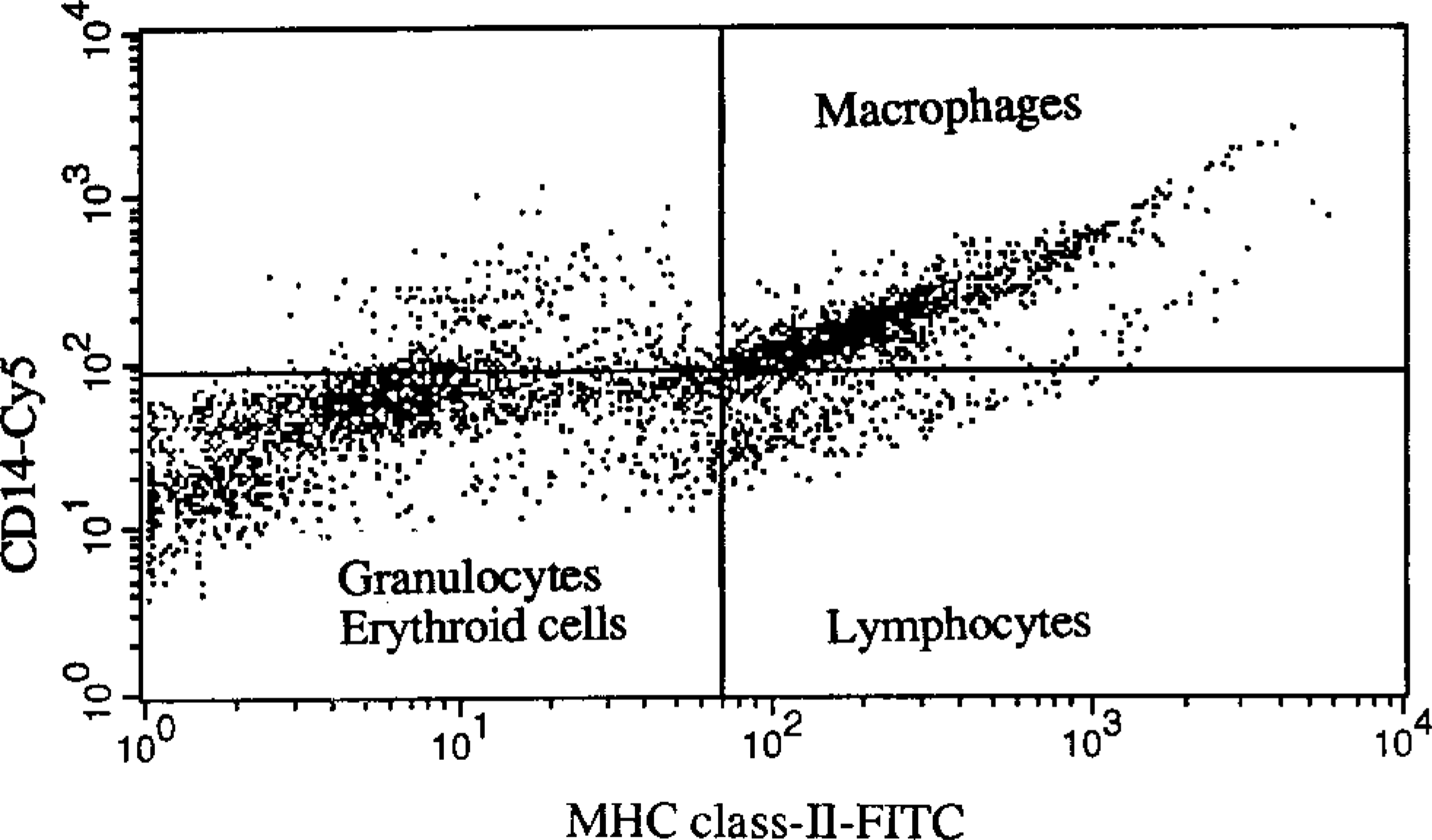
Flow cytometric scatterplot of bone marrow from a dog with malignant histiocytosis. Bone marrow cells were double labeled with anti-CD-14 phycoerythrin-Cy5 and anti-MHC class II labeled with fluorscein. Note distinct populations of monocyte and macrophages (CD-14 positive and MHC class II positive, upper left quadrant), lymphocytes (CD-14-negative and MHC class II positive, lower left quadrant), and granulocytes (CD-14 negative and MHC class II negative, lower right quadrant).
Cytologic evaluation
Bone marrow smears were Wright's stained, and 500 differential cell counts were done. The total number of blast cells and the myeloid to erythroid (M:E) ratio were determined.
Cytochemical evaluation
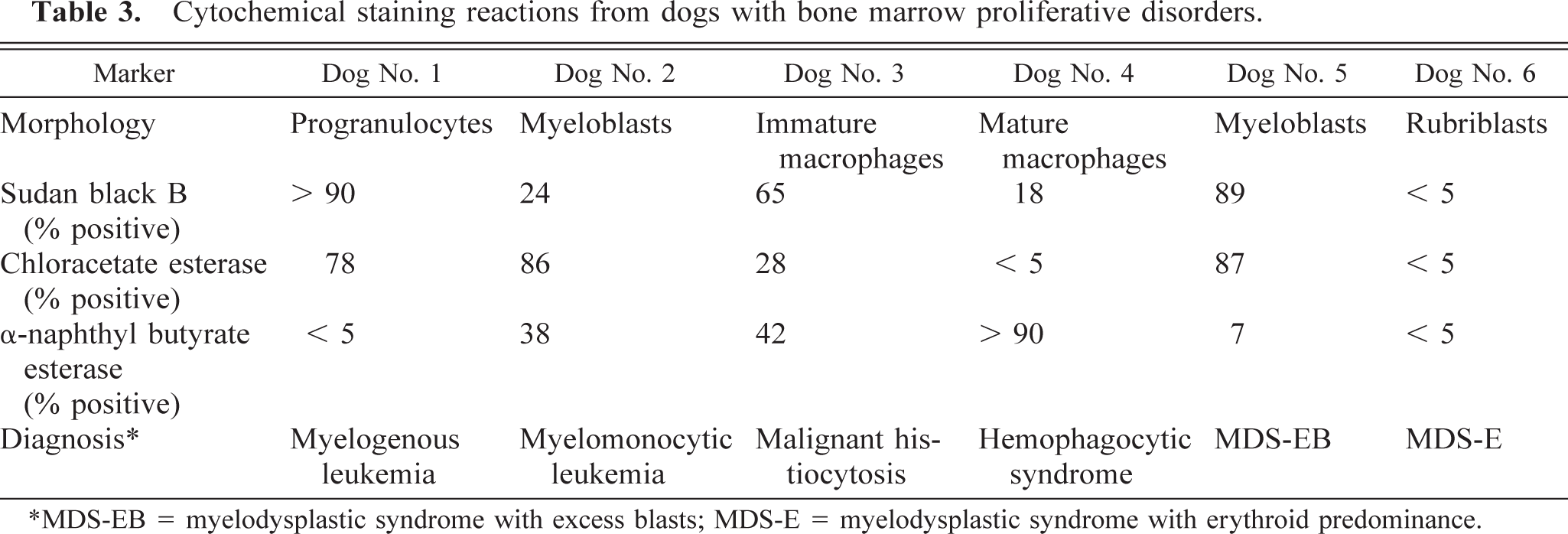
Cytochemical staining profiles of blast cells in bone marrow smears were evaluated by staining for Sudan black B, chloracetate esterase, and α-naphthyl butyrate esterase. Bone marrow smears were fixed in citrate-acetone-formaldehyde and stained using commercially available cytochemistry kits (Sigma Chemical Co., St. Louis, MO). The techniques used were those recommended by the manufacturer and have been previously described. 14
Results
Six dogs with an increase in immature or atypical cells in bone marrow were evaluated. A tentative diagnosis was based on results of blood and bone marrow cytology and cytochemical staining reactions. These diagnoses included myelogenous leukemia (dog No. 1), myelomonocytic leukemia (dog No. 2), malignant histiocytosis (dog No. 3), hemophagocytic syndrome (dog No. 4), and myelodysplastic syndrome (dogs Nos. 5 and 6; Table 1).
Clinical and laboratory features of dogs with bone marrow proliferative disorders.
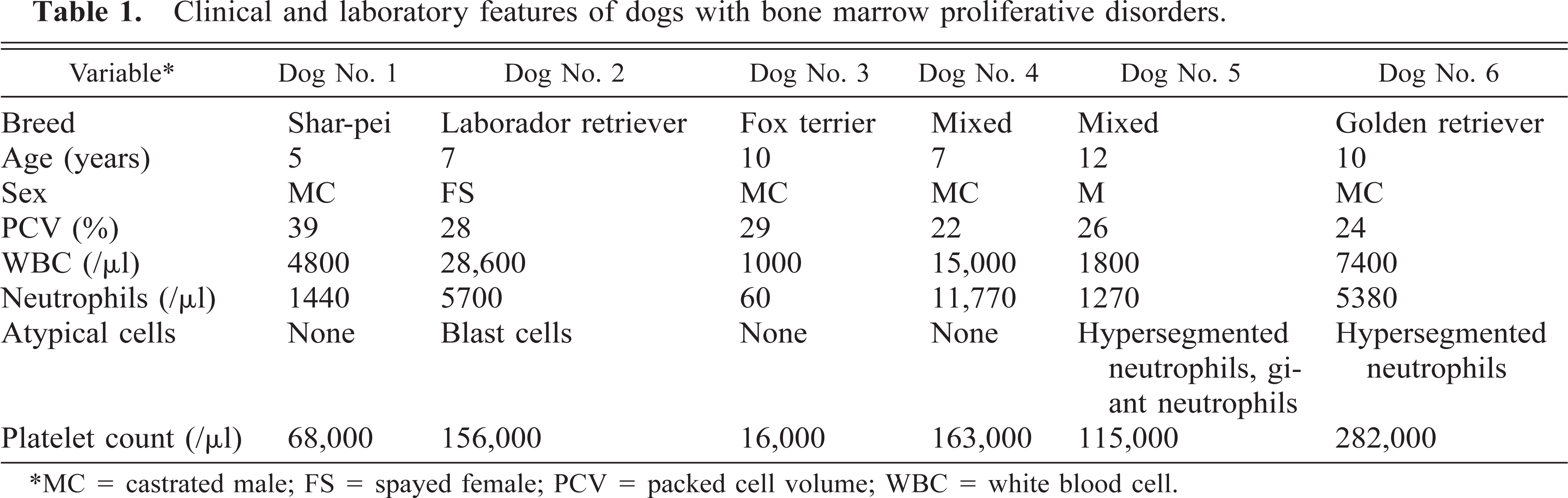
∗MC = castrated male; FS = spayed female; PCV = packed cell volume; WBC = white blood cell.
Myelogenous leukemia
Dog No. 1 had a diagnosis of myelogenous leukemia. Peripheral blood was characterized by mild anemia, neutropenia, and thrombocytopenia (Table 1). No atypical cells were observed in the blood. Bone marrow was characterized by 53% immature myeloid cells that resembled myeloblasts and progranulocytes (Table 2). Progranulocytes appeared to be hypergranular, and some had folded or reniform nuclei resembling cells described as M3 (acute promyelocytic leukemia) acute myeloid leukemia in human beings. 13 The M:t3E ratio was increased.
Manual and flow cytometric differential cell counts in dogs with bone marrow proliferative disorders.
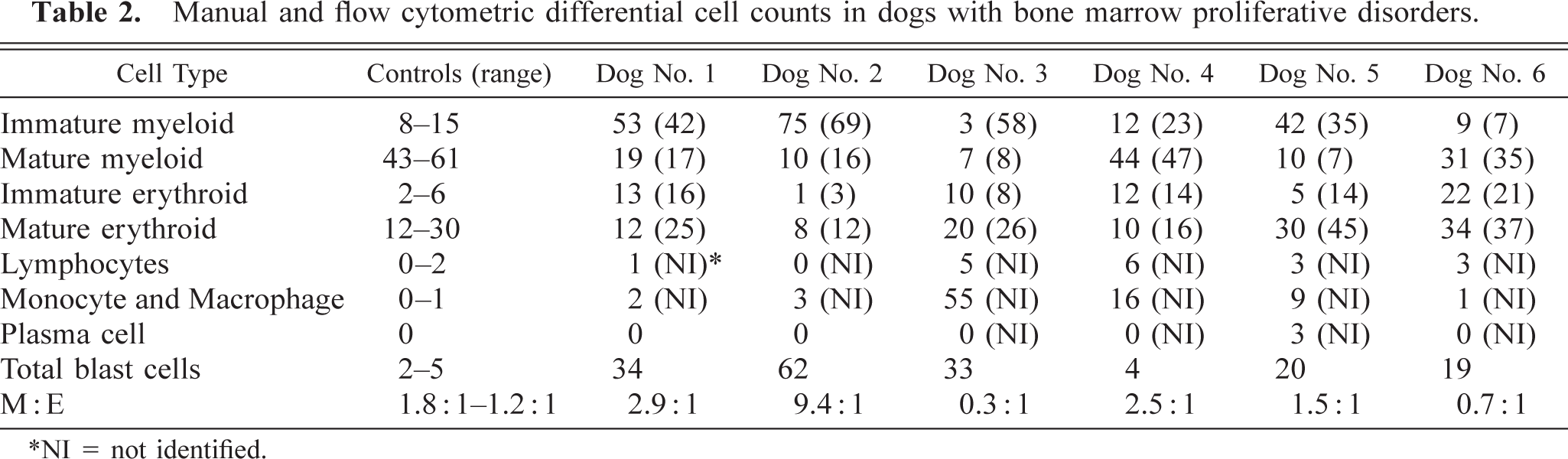
∗NI = not identified.
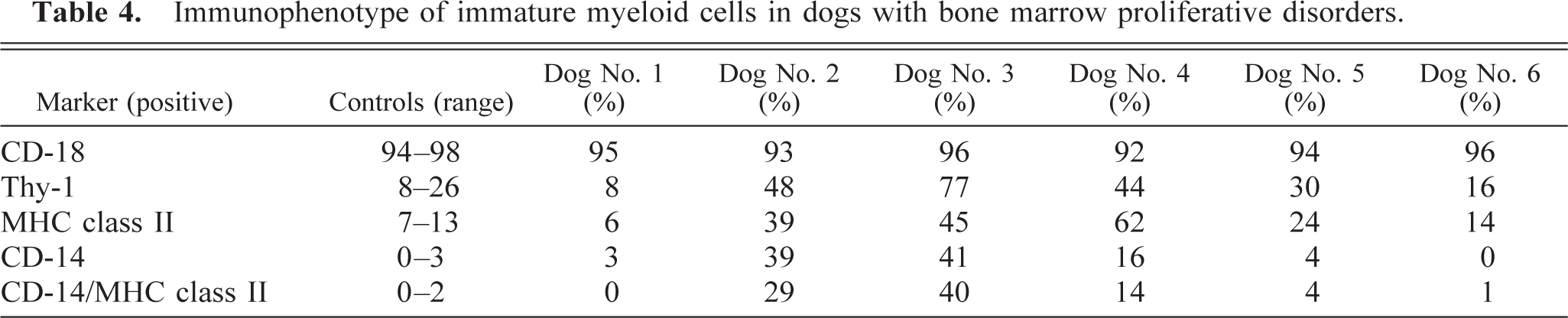
Cytochemical staining reactions indicated that most of the blast cells stained positively for Sudan black B and chloracetate esterase, but not for α-naphthyl butyrate esterase, which is consistent with a granulocytic origin of the malignant cells (Table 3). Immunophenotyping of immature myeloid cells indicated that most of the cells were CD-18 positive, but few were positive for MHC class II, Thy-1, or CD-14 (Table 4). When coexpression of MHC class II and CD-14 was examined, less than 1% of cells in the immature myeloid gate were positive for both markers.
Cytochemical staining reactions from dogs with bone marrow proliferative disorders.
∗MDS-EB = myelodysplastic syndrome with excess blasts; MDS-E = myelodysplastic syndrome with erythroid predominance.
Immunophenotype of immature myeloid cells in dogs with bone marrow proliferative disorders.
Examination of the scatterplots indicated an increase in cells in the lower part of the immature myeloid gate and a decrease in cells in the mature myeloid cells. No increase in the number of cells larger than immature myeloid cells was observed.
Myelomonocytic leukemia
Dog No. 2 had 58% blast cells in the peripheral blood that resembled myeloblasts (Table 1). Bone marrow contained 62% blast cells, which also resembled myeloblasts (Table 2). The M:E ratio was markedly increased.
Cytochemical staining reactions indicated that most blast cells were chloractetate esterase positive, and some were Sudan black B positive and α-naphthyl butyrate esterase positive, which was consistent with a diagnosis of myelomonocytic leukemia (Table 3). Immunophenotyping of cells in the immature myeloid gate indicated that most cells were CD-18 positive, whereas almost half the cells were Thy-1, MHC class II, and CD-14 positive (Table 4). Twenty-nine percent of cells were positive for both MHC class II and CD-14.
Examination of the scatterplots indicated an increase in cells in the immature myeloid gate (gate R2 in Fig 1B). A discrete population of cells were present in the lower part of the immature myeloid gate. Additionally, 16% of bone marrow cells were larger than cells in the immature myeloid gate (gate R5 in Fig 1B). When this population was analyzed, the immunophenotype was essentially the same as that for cells in the immature myeloid gate.
Malignant histiocytosis
In dog No. 3, blood was characterized by pancytopenia, and bone marrow was hypercellular, with 55% of cells being categorized as macrophages (Table 1,2). Cytologically, approximately half of these cells appeared to be mature macrophages, and half were immature macrophages. Immature macrophages were characterized by large size, cytoplasmic basophilia and prominent and multiple nucleoli. Thirty-four percent of macrophages were phagocytizing myeloid and erythroid cells and platelets. Rare multinucleate cells were present.
Cytochemical staining reactions indicated that 65% of the blast cells were positive for Sudan black B, 28% were positive for chloracetate esterase, and 42% were positive for α-naphthyl butyrate esterase (Table 3). Immunophenotyping of immature myeloid cells indicated that 96% of cells were CD-18 positive, 77% were Thy-1 positive, 45% were MHC class II positive, and 41% were CD-14 positive (Table 4). Forty percent of cells were positive for both MHC class II and CD-14.
Examination of the scatterplot indicated a diffuse increase in cells in the immature myeloid gate (gate R2 in Fig 1C). Additionally, 10% of cells exceeded the size of cells in the immature myeloid gate (gate R5 in Fig 1C). Backplotting CD-14–positive cells onto the scatterplot indicated that 75% of the cells were in the immature myeloid gate and 19% were in the larger cell population.
Hemophagocytic syndrome
In dog No. 4, peripheral blood was characterized by nonregenerative anemia and mild thrombocytopenia (Table 1). Bone marrow was characterized by hypercellularity, with large numbers of macrophages phagocytizing nucleated and nonnucleated erythroid cells, granulocytes, and platelets (Table 2). These macrophages varied in size but appeared mature. Lymphocyte numbers were also increased. The percentage of blast cells was less than 5%, and dysplastic features were not observed.
Cytochemical staining revealed that the phagocytic cells were α-naphthyl butyrate esterase positive, chloracetate esterase negative, and less than one third were Sudan black B positive (Table 3). Immunophenotyping of immature myeloid cells revealed 92% CD-18 positive, 44% were Thy-1 positive, 62% were MHC class II positive, and 16% were CD-14 positive (Table 4). Fourteen percent of cells were positive for MHC class II and CD-14.
Myelodysplastic syndromes
In dog No. 5, peripheral blood was characterized by nonregenerative anemia, leukopenia, and thrombocytopenia (Table 1). Giant neutrophils were present, and segmented neutrophils were large and hypersegmented. In bone marrow, the percentage of immature myeloid cells was increased, and 20% of cells were blast cells (Table 2). Marked dysgranulopoiesis, dyserythropoiesis, and mild dysthrombopoiesis was present. Dyserythropoiesis was characterized by asynchronous maturation and fragmented nuclei. Dysgranulopoiesis was characterized by giant forms, marked hypersegmentation (i.e., 8–20 lobe), and atypical lobing patterns. Dysthrombopoiesis was characterized by asynchronous maturation and anisokaryosis. Hemophagocytic macrophages were increased in number. The combination of pancytopenia in blood, hypercellular bone marrow with dysplastic features in all cell lineages, and myeloblasts > 5% was consistent with a diagnosis of myelodysplastic syndrome with excess blasts.
Cytochemical staining reactions indicated that most blast cells were Sudan black B and chloracetate esterase positive but were α-naphthyl butyrate esterase negative, which was consistent with a granulocytic origin of blast cells (Table 3). Immunophenotyping of immature myeloid cells indicated that most cells were CD-18 positive, but few were Thy-1, MHC class II, and CD-14 positive (Table 4).
Examination of the scatterplot indicated an increase in immature myeloid cells and a decrease in mature myeloid cells. Cells larger than immature myeloid cells were slightly increased (i.e., 1.5%).
In dog No. 6, peripheral blood was characterized by nonregenerative anemia without leukopenia or thrombocytopenia (Table 1). Hypersegmented neutrophils were present in blood smears. Bone marrow was characterized by an M:E ratio less than one and an increase in blast cells that resembled rubriblasts. Dyserythropoiesis was marked and was characterized by megaloblasts, fragmented nuclei, and asynchronous maturation. The combination of anemia without leukopenia or thrombocytopenia in blood, M:E ratio less than one, and prominent dyserythropoiesis in bone marrow was consistent with a diagnosis of myelodysplastic syndrome with erythroid predominance.
Cytochemical staining of bone marrow revealed that blast cells did not stain for Sudan black B, chloracetate esterase, or α-naphthyl butyrate esterase consistent with an erythroid or lymphoid lineage (Table 3). Immunophenotyping of bone marrow revealed that cells in the immature myeloid gate were CD-18 positive, but very few cells were positive for Thy-1, MHC class II, and CD-14 (Table 4). Immunophenotyping of cells in the immature erythroid gate indicated that <10% of cells were CD-18, Thy-1, MHC class II, or CD-14 positive, which was consistent with cells of erythroid origin.
Discussion
Classification of proliferative disorders in canine bone marrow has been problematic. Earlier studies established that cytomorphologic evaluation alone was inadequate for determining in the lineage of proliferating cells. 5,8,9,13 The use of cytochemical staining techniques has become a routine procedure for evaluation of cell lineage. However, cytochemical techniques are limited by the low number of lineage-specific stains available. 5,8,9
Immunophenotyping canine bone marrow is dependent on identifying an appropriate panel of lineage-specific markers. The First International Canine Leukocyte Antigen Workshop identified a variety of antibodies that reacted with canine leukocytes. 4 Most of these antibodies react with subpopulations of canine lymphocytes. However, a few antibodies were reported to react with granulocytes, eosinophils, erythrocytes, and platelets. A panel of these and other antibodies were used to immunophenotype leukemias in canine blood. 19 This panel permitted identification of acute myeloid leukemia, acute lymphocytic leukemia, acute large granular lymphocyte leukemia, acute undifferentiated leukemia, and chronic lymphocytic leukemia.
In the present study, we used the combination of scatterplot analysis and immunophenotyping to evaluate proliferative disorders of canine bone marrow. Cytologic evaluation of blood, bone marrow, or both and cytochemical staining techniques were used to tentatively identify the lineage of proliferating cells. Scatterplot analysis was used to identify mature and immature myeloid and erythroid cells and to detect alterations in cell distribution within these gates. The immunophenotype of cells in the immature myeloid gate was determined by labeling cells with four monoclonal antibodies. Previous studies, using a flow cytometric cell sorting technique, have documented that anti-CD-18 is a panleukocyte marker labeling granulocytes, monocytes and macrophages, and lymphocytes, but not erythroid cells; anti-CD-14 is a specific marker for monocyte and macrophages, and anti-Thy-1 and anti-MHC class II labels most lymphocytes and monocytes and macrophages. 22 The combination of anti-MHC class II and anti-CD-14 was used to differentiate monocytes and macrophages from lymphoid cells.
The CD-18-positive, CD-14-negative, Thy-1-negative, MHC class II-negative immunophenotypes identified the dog with a diagnosis of acute myelogenous leukemia. The immunophenotype of acute myelomonocytic leukemia was CD-18 positive, CD-14 positive, Thy-1 -positive, and MHC class II positive. This clearly differentiated myelomonocyte leukemia from myelogenous leukemia. However, the immunophenotype of malignant histiocytosis and hemophagocytic syndrome were similar to that for myelomonocytic leukemia. To immunologically differentiate these disorders, a specific marker for granulocytes would be needed. Anti-CD-66 has been used as a specific marker for mature granulocytes in human bone marrow, whereas anti-CD-13 has been used to detect immature granulocytes and monocytes in human bone marrow. 1 To the authors knowledge, anti-CD-66 and anti-CD-13 antibodies have not been described for use in dogs. However, one antibody that reacts with canine granulocytes has been described. 19 In the absence of a granulocyte-specific markers, malignant histiocytosis and hemophagocytic syndrome can usually be differentiated from myelomonocytic leukemia, cytologically. 2,20 Macrophages in these conditions are generally well differentiated and are frequently hemophagocytic, whereas cells in myelmonocytic leukemia are blast cells that most resemble myeloblasts.
The immunophenotype of myelodysplastic syndrome with excess blasts was CD-18 positive and partially Thy-1 and MHC class II positive. Lack of increase in CD-14-positive cells indicated that these cells were not monocytes and macrophages. The most probable explanation of these data is that some dysplastic granulocyte precursor cells expressed Thy-1 and MHC class II. Expression of antigens from another cell lineage on bone marrow cells has been documented in human myeloproliferative disorders. In one study, erythropoietin receptors were expressed on 60% of malignant cells in acute myeloid leukemia. 18 In another study, CD-4, a T-lymphocyte antigen, was expressed on erythroid cells of human patients with a variety of clinical disorders. 3
Lymphocytic proliferative disorders were not detected among the limited number of cases evaluated. However, we anticipate that the combination of CD-14 and MHC class II labeling would be effective in differentiating lymphocytic leukemias and lymphomas from myeloid leukemias, malignant histiocytosis, and hemophagocytic disorders. When bone marrow cells were labeled with anti-CD-14 and anti-MHC class II, distinct populations of CD-14- and MHC class II-positive cells (i.e., monocyte and macrophages), CD-14-negative and MHC class II-positive cells (i.e., lymphocytes), and CD-14- and MHC class II-negative cells (i.e., granulocytes and erythroid cells) were identified (Fig. 2). MHC class II antigens have been reported to be expressed on all canine bone marrow lymphoid cells, therefore, it appears to be a panlymphocyte marker. 7,17 The lymphoid origin of leukemias could be further evaluated by double labeling with CD-14 and Thy-1, or with T-lymphocyte- or B-lymphocyte-specific antibodies.
In conclusion, the combination of flow cytometric scatterplot analysis and four monoclonal antibodies were used to identify the lineage of immature myeloid cells in six dogs with bone marrow proliferative disorders. This combination of tests permitted differentiation of myelogenous leukemia from myelomonocytic leukemia, malignant histiocytosis, and hemophagocytic syndrome. However, because of a lack of a specific canine granulocyte marker, myelomonocytic leukemia could not be differentiated from malignant histiocytosis and hemophagocytic syndrome by use of immunophenotyping alone. As additional lineage-specific monoclonal antibodies are developed, immunophenotyping should become a valuable tool for determination of the lineage of cells in canine myeloproliferative disorders.
