Abstract
Among the intestinal tumors of hematopoietic cell origin, lymphoma is the most common in the dog. Herein, we characterized the clinical and pathologic features of 11 dogs (average age, 10.6 ± 2.5 years) with T-cell lymphoma of the intestinal tract with eosinophil infiltrates. No sex predominance was apparent. All had localized tumor masses in the small intestine. Grossly, the intestinal wall was thickened, and the lumen of the affected intestine was usually narrowed. Microscopically, we observed transmural diffuse invasion of round to pleomorphic tumor cells. Tumor cells showed varying morphology, from scanty to abundant cytoplasm, and round to ovoid nuclei with scattered to dense chromatin. In seven of the dogs, tumor cells had infiltrated into the epithelium. All showed infiltration of eosinophils and all 11 tumors had a T-cell phenotype (CD3+,CD79-). Only one tumor stained positive for the mast cell marker c-kit and none was positive for mast cell tryptase. We did not observe ultrastructurally apparent granules in any of the tumor cells. These results suggest that, in dogs, T-cell lymphomas of intestinal origin resemble mast cell tumors of intestinal origin with respect to cell structure and eosinophil infiltration. Therefore, in the absence of epitheliotropism, it is difficult to confirm the differential diagnosis without immunostaining for mast cell and lymphocyte markers, including mast cell tryptase, c-kit, CD3, and CD79.
Among the intestinal tumors of hematopoietic cell origin, lymphoma is the most common in dogs, 5 with primary intestinal lymphoma being less common than the multicentric form. 6 Although it was previously thought that primary intestinal lymphomas in dogs were of B-cell origin, recent reports have indicated that the majority of canine intestinal lymphoma are of T-cell origin. 3, 4, 9 Of 49 dogs with intestinal lymphoma, 38 cases were of T-cell phenotype with epitheliotropism, three cases were nonepitheliotropic lymphoma of B-cell origin, and eight cases could not be immunophenotyped. 3, 4, 9 T-cell lymphomas of intestinal origin have been divided into two types based on the morphological characteristics of the tumor cells: small- to moderate-sized lymphoma showing epitheliotropic behavior, and moderate- to large-sized lymphoma with or without epitheliotropism. The former type has been classified as intestinal T-cell lymphoma according to the World Health Organization (WHO) classification system 10 and is similar in cell morphology, immunophenotype, and epitheliotropism to previously reported cases. 3, 4, 9 Epitheliotropic T-cell lymphoma of the intestinal tract, also known as intestinal T-cell lymphoma, 10 has been described as a slowly progressive small cell lymphoma of the enteric tract, which appears to arise from a background of chronic inflammatory bowel disease. 4
We recently described mast cell tumors of the canine intestinal tract. 7 Cells of these tumors had moderate to abundant cytoplasm and round to ovoid nuclei with sparsely scattered granules showing metachromasia. These granules could not be seen with HE sections. In addition, mast cell tumors always had eosinophil infiltration. These morphological characteristics were very similar to those of moderate- to large-sized T-cell lymphoma of intestinal origin. During a retrospective reevaluation of small round cell tumors in dog intestinal tracts, we encountered 11 previously undescribed cases of T-cell lymphoma resembling mast cell tumor. The objective of this study was to characterize the clinical and pathologic features of these 11 tumors, and to establish a method for the differential diagnosis of T-cell lymphoma and mast cell tumor using immunohistochemical markers and electron microscopy.
Materials and Methods
Surgically-resected biopsy specimens of round cell tumors in gastrointestinal tract from 217 dogs had been removed and sent to Setsunan University and Marupi Lifetech Company for histopathologic examination from 1990 to 2004. Resected tumor masses were limited to the gastrointestinal tract. Of the total of 203 cases stored in the archive during this time period, 126 were lymphoma based on WHO classification using HE sections. 10 The remaining 77 dogs were diagnosed as lymphoma or mast cell tumor using HE sections. Of 77 dogs, 55 were excluded because they were mast cell tumors based on toluidine blue and immunohistochemical staining, 7 and 11 were excluded because of abundant nonspecific reaction in immunostaining. The remaining 11 cases were selected by immunostaining for CD3 and eosinophil infiltration. Specimens were fixed in 10% phosphate-buffered or neutral formalin (pH 7.4), dehydrated in a graded series of ethanol, and embedded in paraffin, after which 4-µm-thick sections were stained with HE. Cell size was classified according to the WHO classification, 10 with small cell nuclear size defined as 1.5 to 2 red blood cells in diameter; large cell nuclear size defined as 2 to 3 red blood cells in diameter; and moderate cell nuclear size defined as larger than small cell and smaller than large cell. Cell atypia was classified as small round (+); moderate in size, round to ovoid (++); and pleomorphic (+++). Epitheliotropism was defined as the presence of tumor cell infiltration among mucosal epithelial cells. The mitotic rate was the number of mitoses in 10 high power fields (400×). Eosinophilic infiltration was grouped as + (below 10 per 200× field), ++ (over 10 per 200× field), and +++ (eosinophils more numerous than tumor cells). Representative sections were also stained with toluidine blue and Alcian blue. For immunohistochemistry, the primary antibodies used were antihuman c-kit polyclonal antibody (1 ° 800, Dako, Kyoto, Japan), antihuman mast cell tryptase monoclonal antibody (1 ° 400, AAI, Serotec Ltd, Oxford, UK), anticanine CD45RA monoclonal antibody (CA21.4B3, 1:150, obtained from Dr. P. F. Moore), antihuman CD3 polyclonal antibody (1 ° 200, Dako), antihuman CD79acy monoclonal antibody (HM57, 1 ° 160, Dako), antivimentin monoclonal antibody (V9, 1 ° 200, Dako), and antihuman Ki-67 monoclonal antibody (MIB-1, 1 ° 50, Dako); for each, an appropriate normal canine tissue sample was used as a positive control. Tissue sections for assay of c-kit, mast cell tryptase, CD79acy, and vimentin were immersed in 0.01 M citrate buffer (pH 6.0) and microwaved at 600 W for 20 minutes to retrieve antigens; tissue sections for assay of CD45RA and CD3 were incubated at 37°C for 20 minutes in 0.4% pepsin for antigen retrieval; and tissue sections for assay of MIB-1 were immersed in 0.01 M citrate buffer (pH 6.0) and autoclaved at 120°C for 15 minutes to retrieve antigens. All slides were rinsed with 0.05 M Tris buffer saline (TBS, pH 7.6) and 0.01% Tween 20, treated with 1% hydrogen peroxide, and again rinsed with TBS plus Tween 20. Slides were incubated in bovine serum for 5 minutes and with primary antibodies overnight at 4°C. The slides were subsequently rinsed with TBS plus Tween 20, treated for 30 minutes at room temperature with N-Histofine MAX PO (M or R) (Nichirei, Tokyo, Japan), rinsed with TBS plus Tween 20, incubated in diaminobenzidine solution containing 0.01% hydrogen peroxide for the peroxidase coloring reaction, and counterstained with Mayer's hematoxylin. To evaluate cell proliferation activity using Ki-67 staining, we counted the number of strongly positive cells among more than 500 cells in 2 slides using image processing and analysis software (Ultimage pro Version 2.6.1., Graftek, Mirmande, France). Except for Ki-67 staining, positive staining of over 50% of tumor cells was judged as positive (+). For electron microscopy, three formalin-fixed tumor masses were further fixed with 2.5% glutaraldehyde in 0.1 M phosphate buffer, pH 7.4, post-fixed in 1% osmium tetroxide solution (pH 7.4) for 2 hours, and processed into resin. Semithin sections were cut and stained with toluidine blue. Ultrathin sections were cut and stained with uranyl acetate and lead citrate, and then examined under an electron microscope (JEM 1200EX, JEOL, Tokyo, Japan).
Results
Of the 11 dogs with tumors, three were Maltese, two were poodles, two were mongrels, and one each was a Shetland sheepdog, Pyrenean mountain dog, golden retriever, and English pointer. Their average age was 10.6 ± 2.5 years (range 5–11 years, median 8 years). Five were intact males, and two each were intact females, neutered males, and neutered females. All tumor masses were located in the small intestine. Eight tumors consisted of single masses, one in the jejunum and seven in the ileum; and three tumors were multiple masses, in both the jejunum and ileum. Clinical findings attributed to the intestinal tumor mass included vomiting (nine), diarrhea (five), and melena (two). Survival data were available for seven dogs. One dog died during surgery, one was killed humanely within 3 days of surgery, and two not treated with chemotherapy died 3 days after surgery. One dog treated with COAP protocol (cyclophosphamide, doxorubicin, vincristine, prednisone) died 20 days postoperatively. The two dogs not treated with chemotherapy survived 120 and 240 days postoperatively, respectively. Necropsy examinations were not performed on any of these animals.
On gross examination, in all cases, we observed a prominent thickening of the intestinal wall and a narrowing of the lumen of the intestine. Ulceration of the affected intestinal mucosa was often present, along with varying degrees of thickening of the mucosa. The cut surface of the tumor was colored white to gray and of variable consistency.
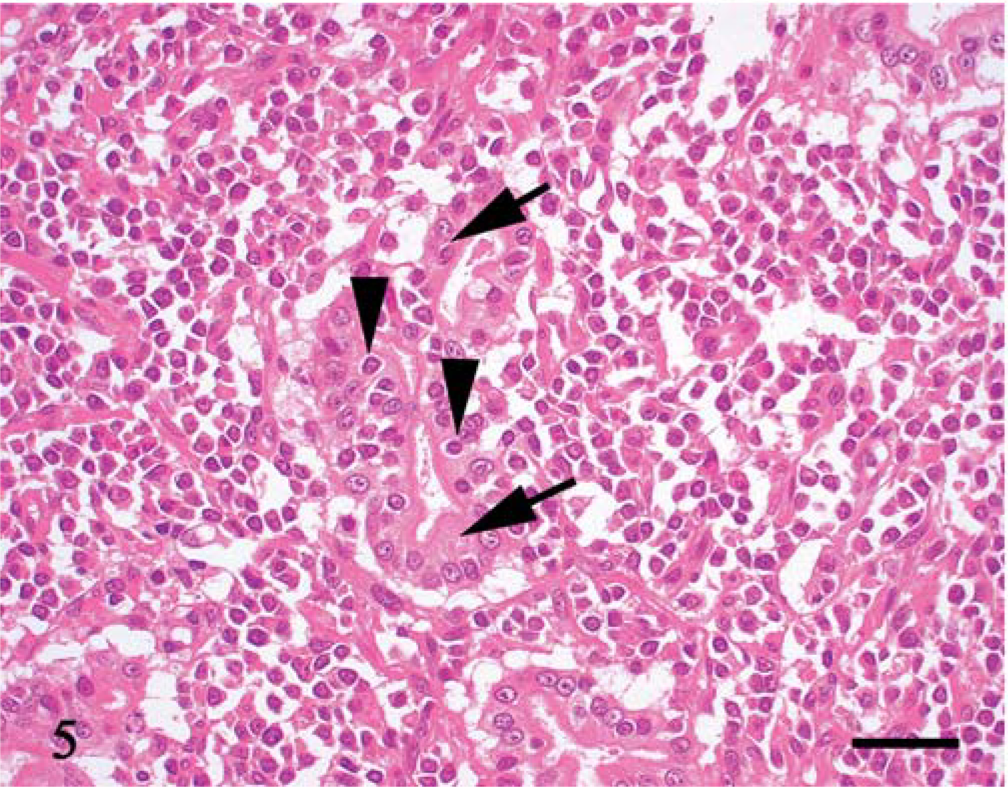
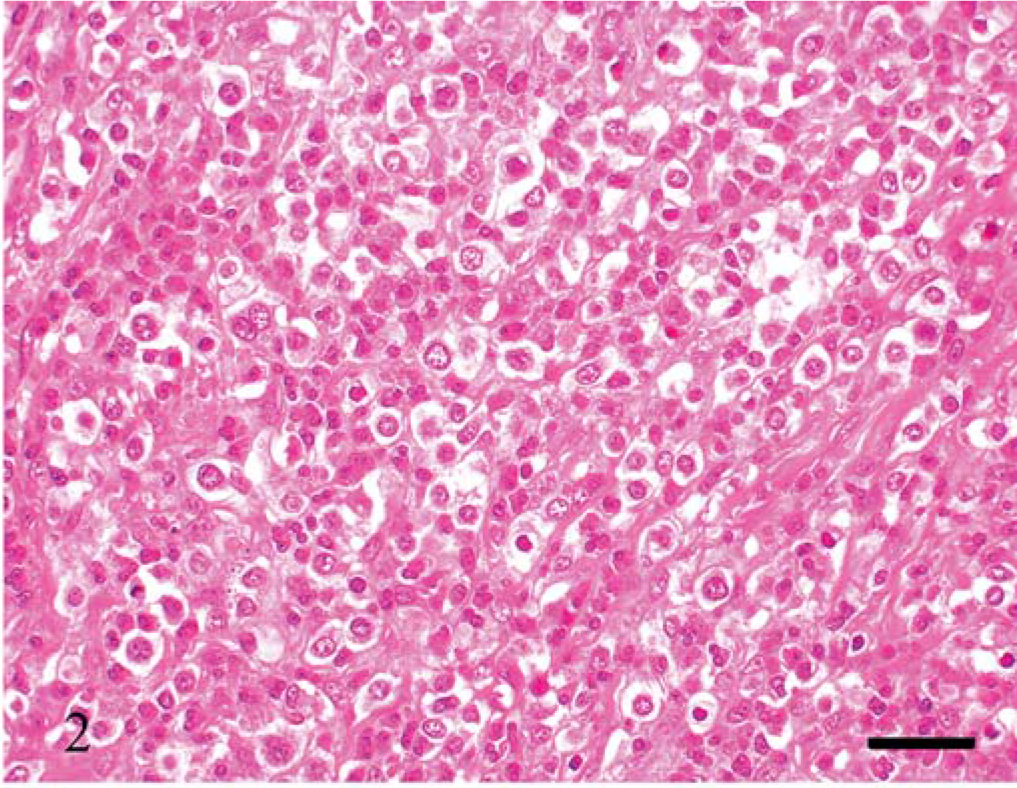
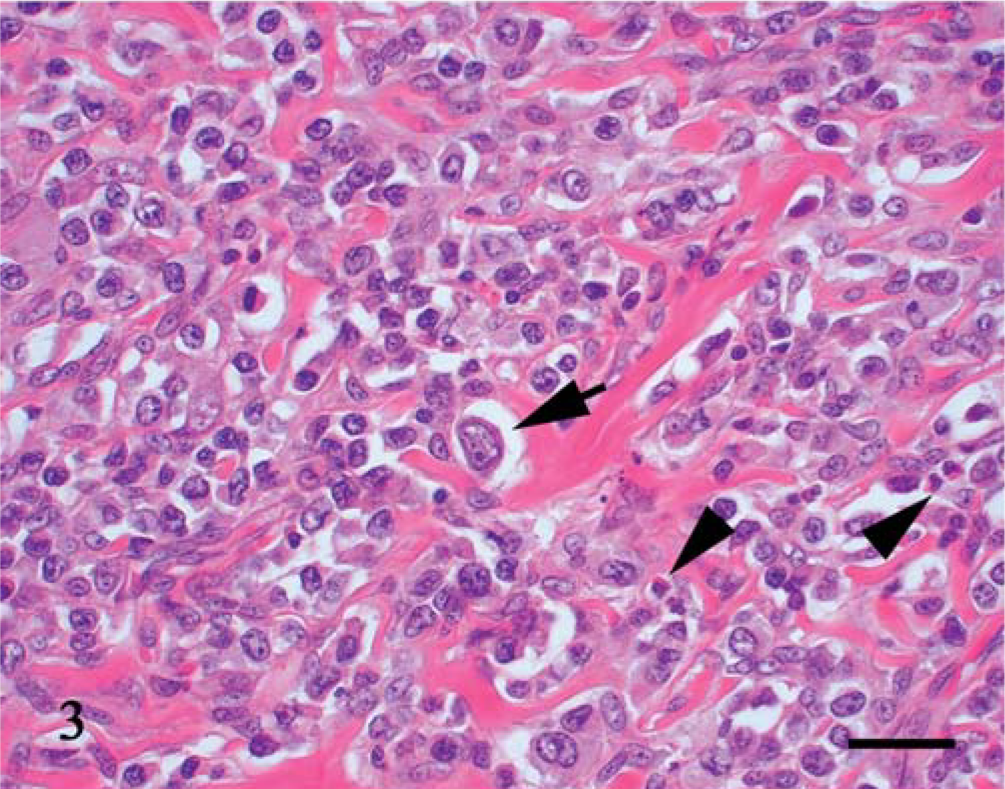
Under microscopic examination, we observed diffuse infiltration and proliferation of tumor cells from the mucosa to the tunica muscularis or serosa (Fig. 1). Morphologically, the tumor cells could be classified as small, round, and monomorphic in three dogs; moderate in size and round to ovoid in four dogs; and moderate to large in size and pleomorphic in four dogs (Figs. 2–4). Each tumor cell usually showed a distinct cell boundary. The nuclei also varied in shape and could be classified as small, round, and monomorphic in five dogs; moderate in size, round to ovoid in five dogs; and pleomorphic in one dog. The nuclei had scattered or dense chromatin with small distinct nucleoli. Mitotic rate was 14.0 ± 8.5 per 10 high power fields. The average MIB-1 index was 17.7 ± 18.6%. In eight cases, the tumor cells had moderate to abundant, slightly eosinophilic to pale staining cytoplasm, whereas the other three had small monomorphic tumor cells with scant to moderate cytoplasm. Epitheliotropic behavior was evident in 3/3 cases of small cell lymphoma, 3/4 tumors with moderate-sized cells, and 1/4 tumors with large cells (Fig. 5).
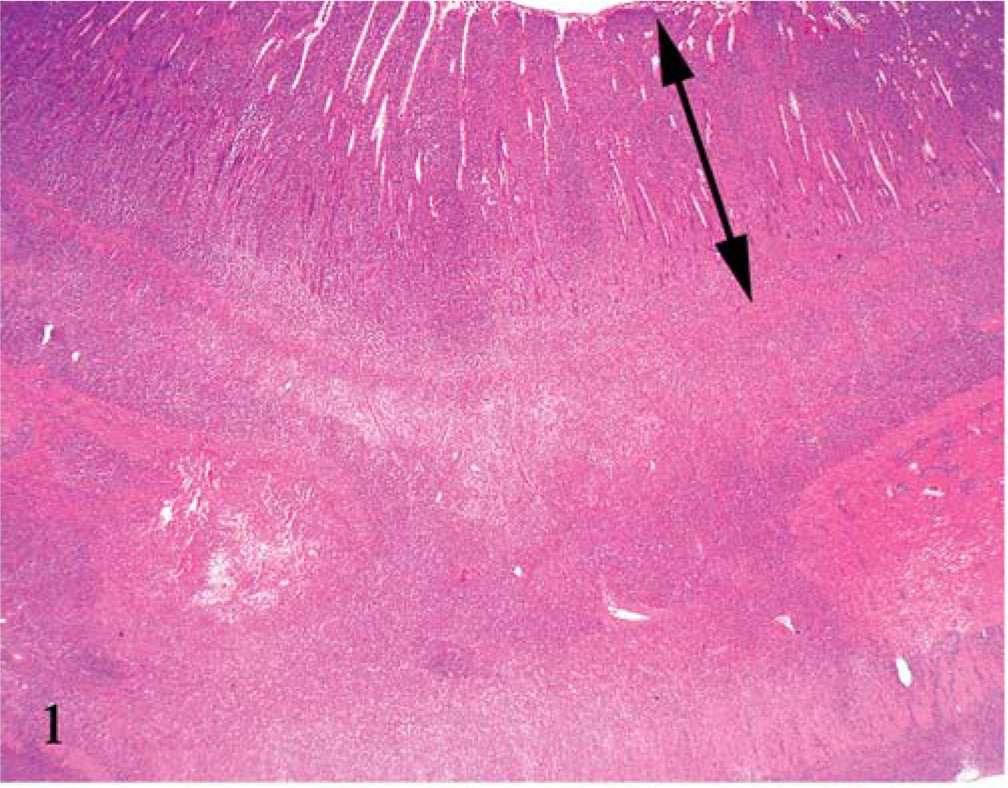
Jejunum; dog No. 8. Tumor cells diffusely invade from the mucosa to the serosa. Double arrow: mucosa HE.
Jejunum; dog No. 11. Tumor cells (arrowheads) infiltrate among crypt epithelial cells (arrows). HE. Bar = 30 µm.
Thin or thick fibrous stroma was observed in about half the tumors (Fig. 3). Stromal components were abundant, especially in tumors with moderate- to large-sized tumor cells. Eosinophil infiltration was present in all 11 tumors, varying from slight to heavy (Figs. 2, 4). In five tumors, in which intestinal mucosa was near the tumor tissue, there was lymphoplasmacytic infiltration of the lamina propria. The junctional region between tumor and lymphoplasmacytic inflammation was not sharply demarcated.
Jejunum; dog No. 6. Moderate-sized round tumor cells have round nuclei with scattered chromatin and small distinct nucleoli. The cell boundary is usually distinct. Many eosinophils infiltrate the tumor. HE. Bar = 30 µm.
Jejunum; dog No. 2. Pleomorphic tumor cells are observed with giant nuclei (arrow). Eosinophils (arrowheads) infiltrate between thick fibrous stroma. HE. Bar = 30 µm.
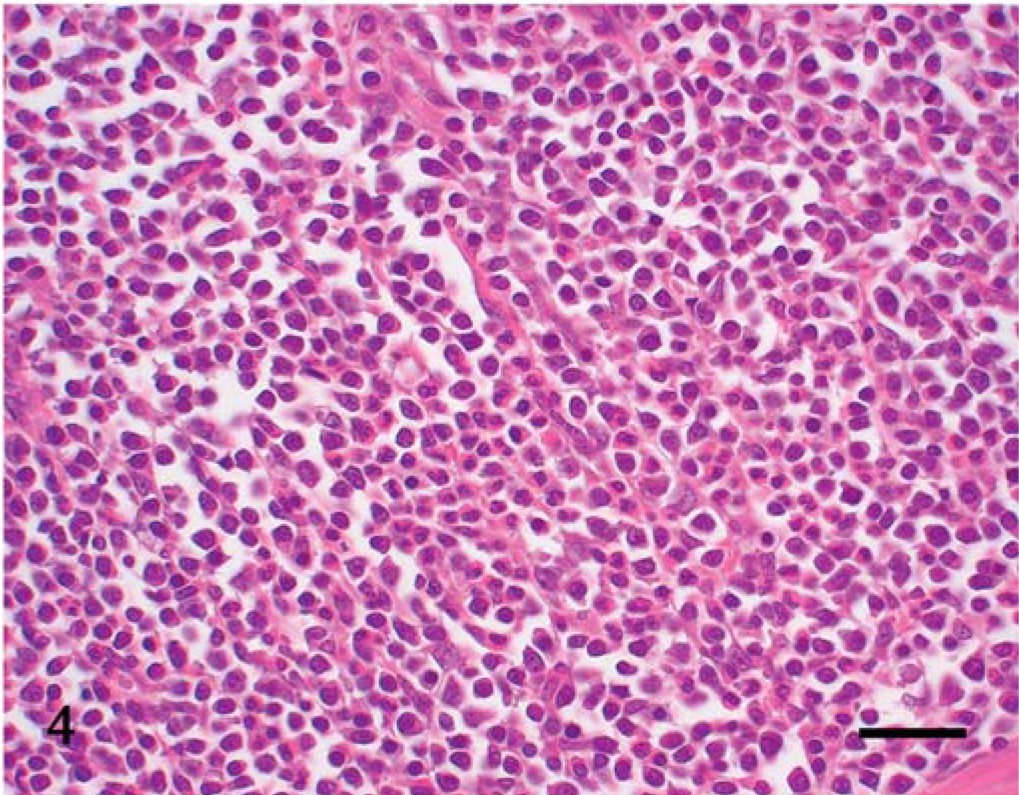
Jejunum; dog No. 7. Small round cells have round nuclei with dense chromatin. There is moderate infiltration of eosinophils. HE. Bar = 30 µm.
The results of immunohistochemistry and special staining are summarized in Table 1.
Summary of morphology, special staining, and immunohistochemistry of T-cell lymphoma in 11 dogs.∗
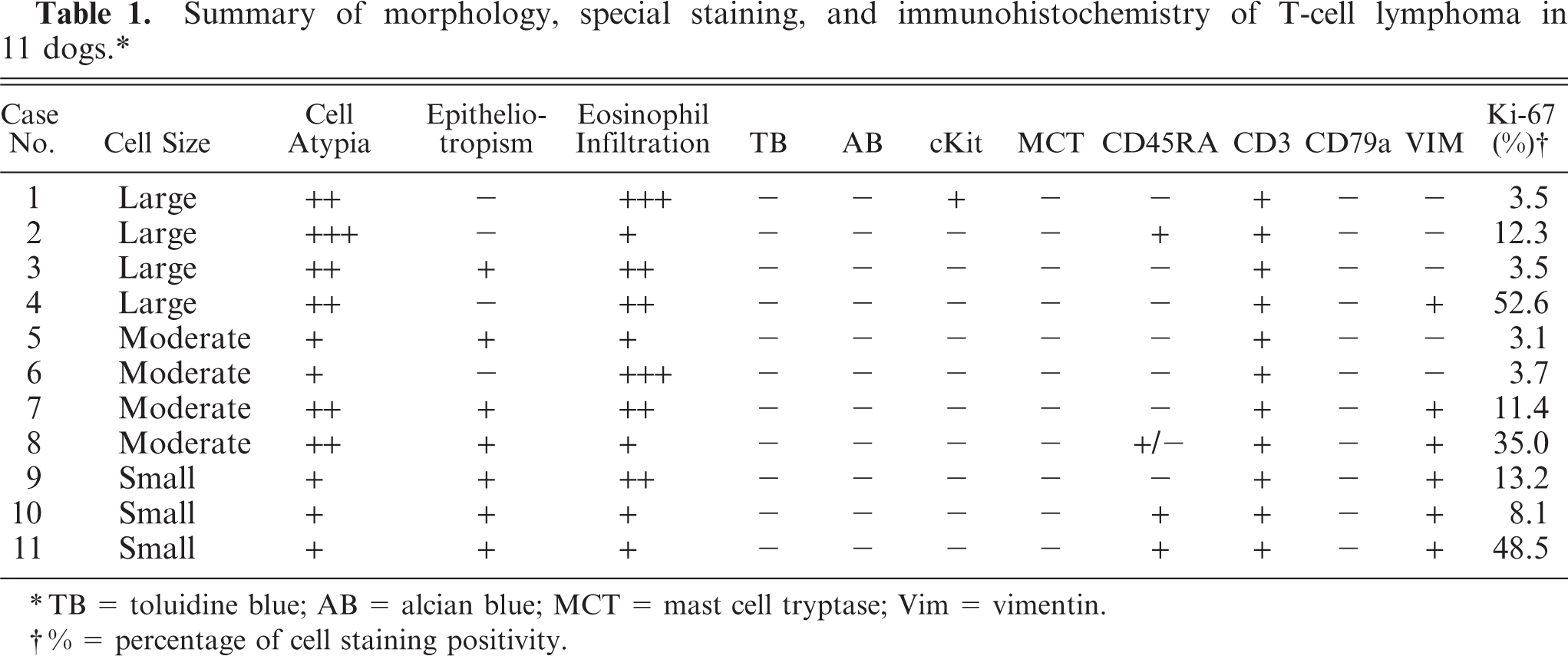
TB = toluidine blue; AB = alcian blue; MCT = mast cell tryptase; Vim = vimentin.
% = percentage of cell staining positivity.
All 11 tumors had a T-cell phenotype (CD3+, CD79−) (Fig. 6). All 11 tumors were negative for metachromasia by Alcian blue staining and for mast cell tryptase, although one tumor was positive for c-kit. Four of the tumors were positive for CD45RA and six were positive for vimentin.
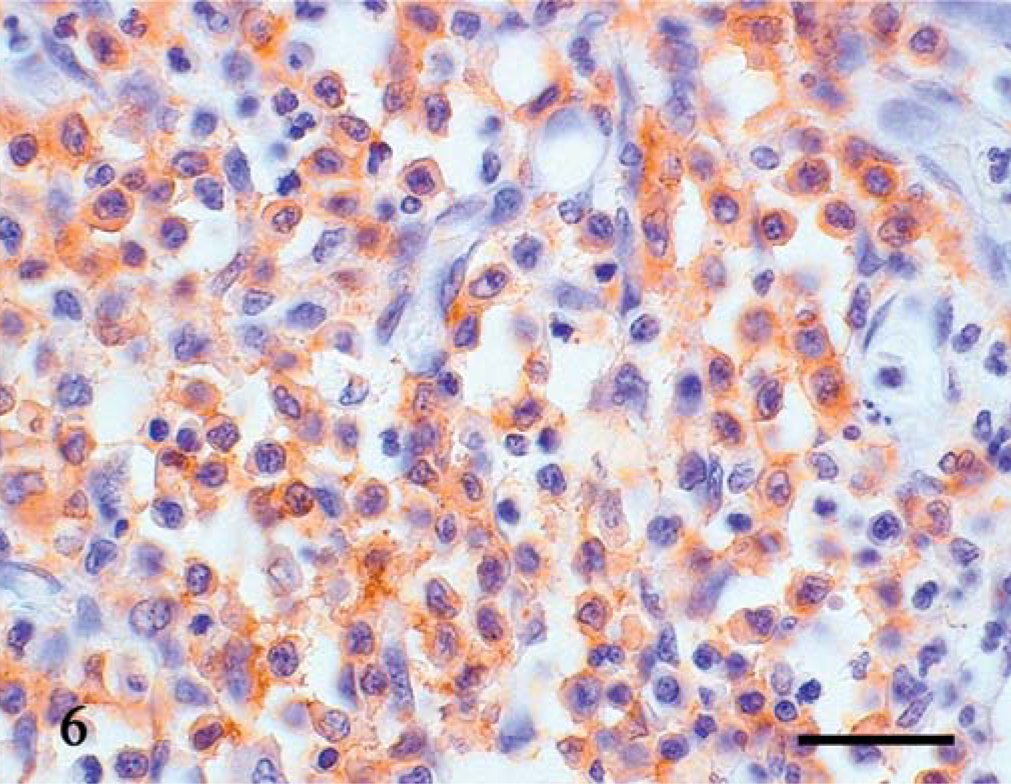
Jejunum; dog No. 3. Immunoreactivity for CD3 is detected. Universal Immuno-enzyme Polymer method, Mayer's hematoxylin counterstain. Bar = 30 µm.
Electron-microscopic examination of three of the tumors revealed that the tumor cells had smooth cell membranes, and cytoplasmic processes were not conspicuous (Fig. 7). In round to convoluted nuclei, heterochromatin was present along the nuclear membrane, with prominent euchromatin. Ribosomal rosettes, small rough endoplasmic reticulum, and lysosomes were frequently seen, but no granules were evident in the cytoplasm (Fig. 8).
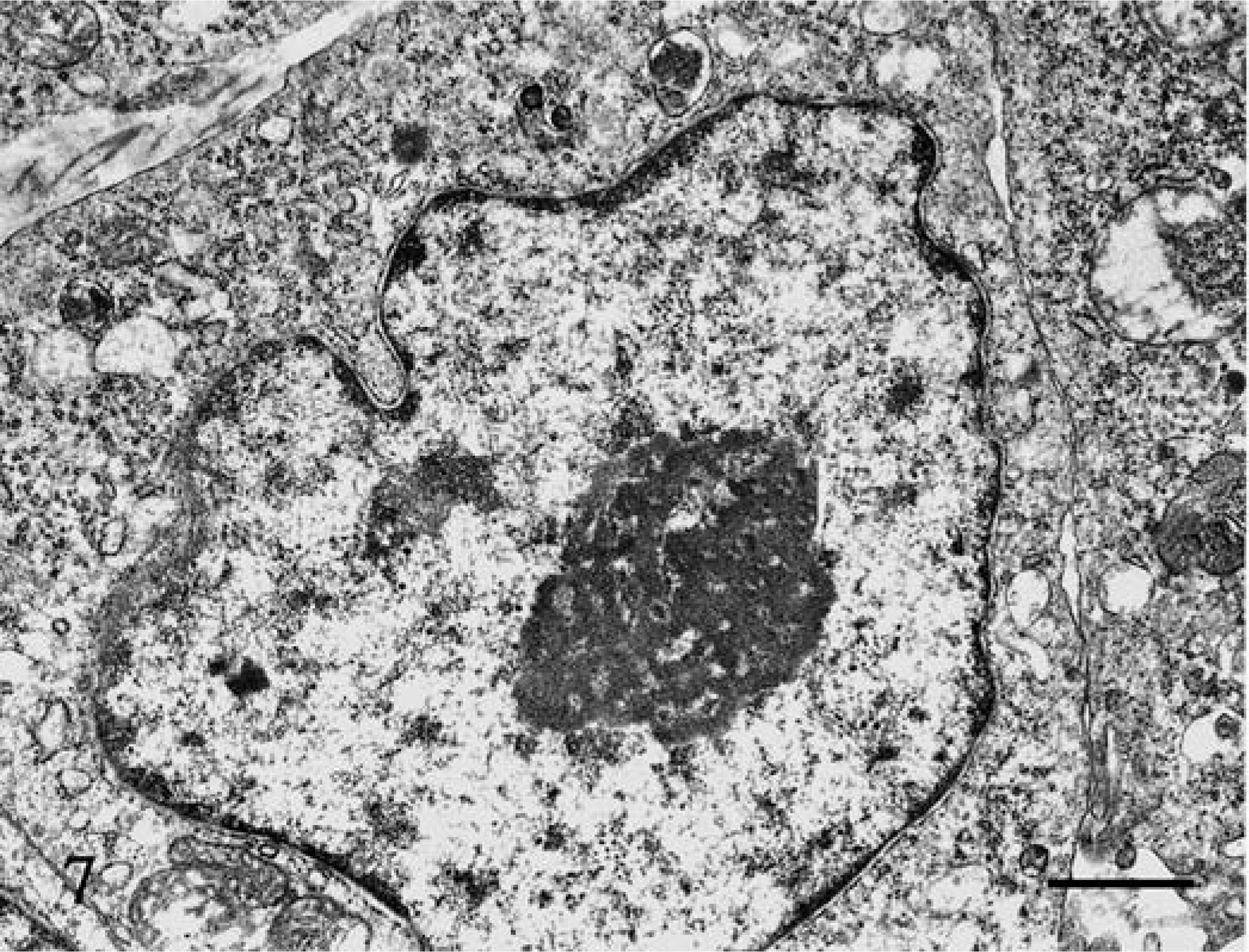
Jejunum; dog No. 6. Tumor cells have smooth cell membranes, and cytoplasmic processes are not conspicuous. Uranyl acetate and lead citrate. Bar = 1 µm.
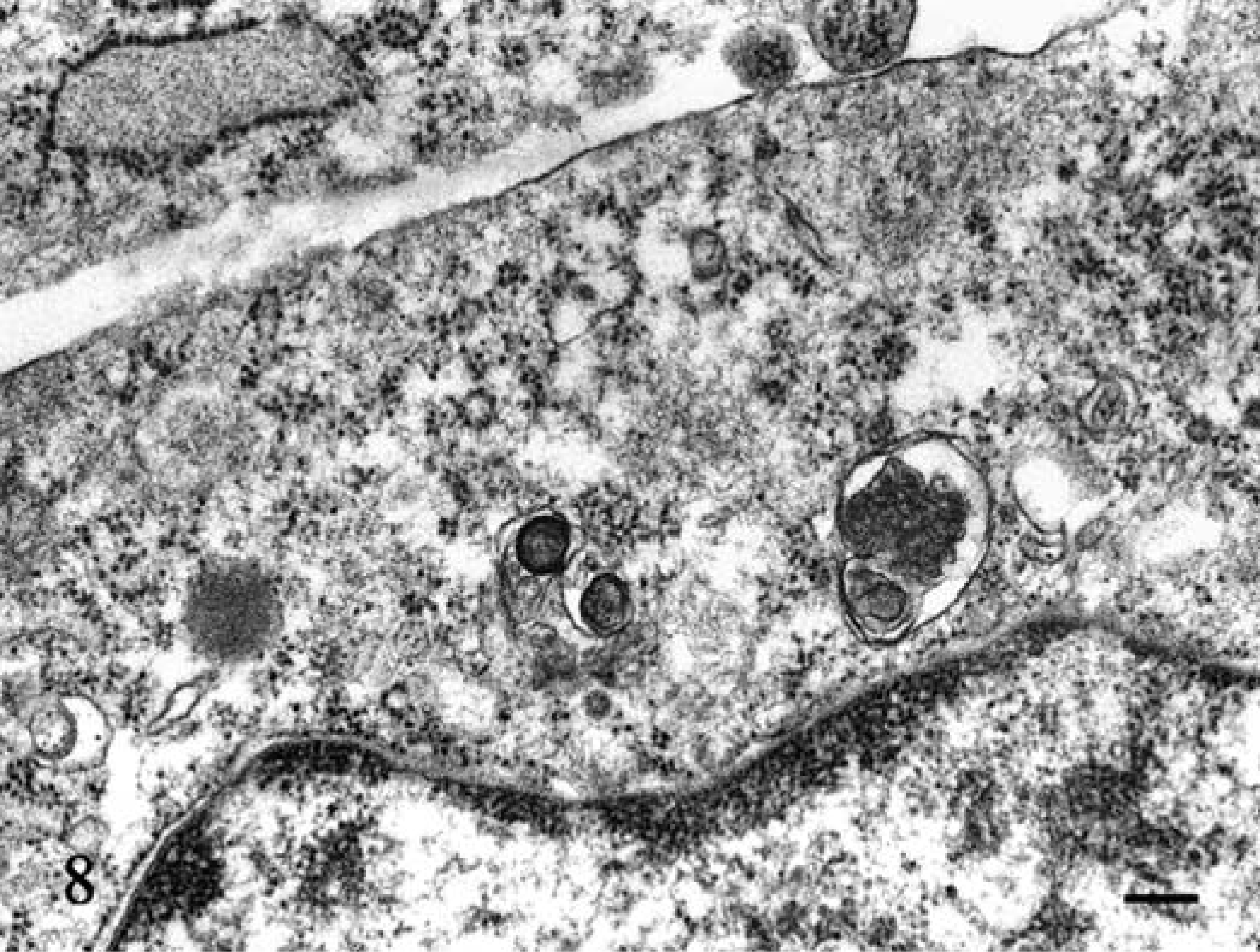
Jejunum; dog No. 6. Ribosomal rosettes, small rough endoplasmic reticulum and lysosomes were present, but there were no granules in the cytoplasm. Uranyl acetate and lead citrate. Bar = 200 nm.
All 11 tumors showed lymph vessel invasion. Lymph node metastasis was observed in all four cases in which mesenteric lymph nodes were examined.
Discussion
Although we found that eosinophil infiltration is a characteristic feature of the cases studied, to our knowledge it has never been reported previously in canine intestinal T-cell lymphoma. However, human intestinal T-cell lymphomas, especially the pleomorphic moderate and large cell types, are usually accompanied by eosinophil infiltration, 8 and eosinophil infiltration has been observed in peripheral/extranodal T-cell lymphomas in animals. 10 Eosinophil infiltration, however, has also been observed consistently in mast cell tumors of intestinal origin. 7 Thus, when a background of eosinophils is present in an intestinal round cell tumor, both T-cell lymphoma and mast cell tumor need to be considered in the differential diagnosis.
Epitheliotropism was a characteristic feature in canine T cell lymphoma and was not a feature in canine intestinal mast cell tumor. 3, 4, 7, 9 In our present cases, approximately 75% of intestinal T-cell lymphomas exhibited epitheliotropism. Epitheliotropism can be used to distinguish these two tumor types. However, approximately 25% of our present dogs did not have epitheliotropism. In previous study of canine intestinal T-cell lymphoma, the neoplastic cells always showed epitheliotropic behavior. 3, 4, 9 Canine intestinal T-cell lymphoma without epitheliotropism has not been reported. Therefore, these cases need immunostaining to confirm the differential diagnosis. In addition, lymphoplasmacytic infiltration, abundant fibrovascular stroma, and giant cells were seen in the present cases. These same features were seen in canine intestinal mast cell tumors. 7 Thus, these features were not able to help us to differentiate between the two tumor types.
Alimentary lymphoma of dogs is accompanied by lymphoplasmacytic infiltration. 2 An epitheliotropic alimentary form of canine T-cell lymphoma has also been found to be concomitant with lymphocytic-plasmacytic enteritis. 4 It has been suggested that lymphoplasmacytic enteritis is a premalignant phase of alimentary lymphoma in Basenji dogs. 1 In five of the cases reported here, there was no clear boundary between the tumor and lymphoplasmacytic inflammation. However, six of the cases reported did not show apparent lymphoplasmacytic inflammation. Two previous reports, which described 37 cases of epitheliotropic T-cell lymphoma, did not mention concomitant lymphoplasmacytic inflammation. 3, 9 Therefore, lymphoplasmacytic inflammation may not always be associated with T-cell lymphoma.
Alimentary lymphoma has aggressive behavior and a poor prognosis. 2 Only 1 of the 11 dogs in this series survived for as long as 240 days, whereas six others died or were killed humanely within 120 days. 2, 7 Epitheliotropism was not related to prognosis. Unfortunately, in our cases, necropsy was not performed on any of the animals. Therefore, we cannot exclude the possibility that some of these cases represented multicentric lymphoma with intestinal invasion.
In conclusion, we found that canine T-cell lymphomas, characterized by small to large cell types and eosinophil infiltration, developed in the small intestine. In 4 of 11 cases, tumor cells did not exhibit epitheliotropism. Diagnostic pathologists should be aware of T-cell lymphoma as a differential diagnosis for intestinal round cell tumors with eosinophilic infiltrates. Since these tumor cells were very similar to mast cell tumors of gastrointestinal origin, immunostaining for mast cell and lymphocyte markers was required for definitive diagnosis.
