Abstract
One hundred twenty-six cutaneous mast cell tumors obtained by excisional biopsy from 106 dogs were evaluated using immunohistochemical staining for the presence of p53 protein. A standard avidin–biotin immunohistochemical protocol was used incorporating a polyclonal antibody of rabbit origin (CM-1) as the primary antibody. Histopathologic grading of tumors was performed on hemotoxylin and eosin–stained samples. There was a significant difference in the percentage of cells staining positive for p53 for the histopathologic grades (
Survival of dogs with cutaneous mast cell tumors is related to the histopathologic grade of the tumor.3,13,18 The survival time of dogs with well-differentiated tumors (grade I) is twice that of dogs with intermediately differentiated tumors (grade II) and six times that of dogs with poorly differentiated tumors (grade III).18
Methods other than histopathologic grading have been evaluated in an attempt to more accurately determine the prognosis of dogs with cutaneous mast cell tumors. Such methods include the association between DNA ploidy and histopathologic grade of mast cell tumors, evaluation of chromosome nucleolar organizer regions stained with silver, and antiproliferating cell nuclear antigens.1,20 None of these methods predict survival time more accurately than histopathologic grade. However, using other methods in conjunction with histopathologic grade may have some prognostic benefit.
Wild-type p53 is an intranuclear, 53-kDa phosphoprotein that regulates the transition from the G1 phase to the S phase of the eukaryotic cell cycle.2 p53 functions as a transactivator of gene transcription by either promoting or repressing mRNA synthesis.11 This helps ensure accurate replication of DNA, preventing mutations of the cell's DNA from being propagated.2,24,25 p53 plays a role in maintaining genomic fidelity by orchestrating cell cycle arrest to enable DNA repair to occur prior to replication.2 When repair of the DNA is accomplished, the cell returns to the G1 phase and continues on with DNA replication and synthesis.11 If the damage to the DNA is irreparable, signals mediated through p53 may initiate apoptosis (programmed cell death).2
Mutation of the gene that encodes the wild-type p53 protein has been identified as one of the most common genetic changes associated with the development of many cancers of humans.2 The biological activity of mutant p53 may be altered, which can cause a decrease in or complete loss of function.2 Some mutant forms of p53 are oncogenic and stimulate cell division without screening the replicating genome for point mutations.11 As the concentration of mutant p53 in cells increases, the probability of neoplastic changes in the cell also increases.11,22
The purpose of this study was twofold: first, to determine if a difference exists in the percentage of cells staining positive for p53 between histopathologic grades of canine cutaneous mast cell tumors, and second, to determine if an association exists between the percentage of cells staining positive for p53 and prognosis (survival time and time to tumor recurrence) of dogs with cutaneous mast cell tumors.
Materials and Methods
Clinical data
Medical records at Louisiana State University School of Veterinary Medicine (LSU-SVM) were searched to identify cases of canine cutaneous mast cell tumor diagnosed and treated between January 1989 and December 1997. Criteria for inclusion specified that all samples were obtained by excisional biopsy with histopathologic confirmation of mast cell tumor. Fifty-five archival tissue samples of confirmed canine cutaneous mast cell tumors from the University of Georgia Veterinary Diagnostic Laboratory (UGVDL) were also acquired.
Data collected retrospectively from 71 LSU-SVM records included signalment, tumor location, clinical stage (Table 1), survival time following excisional biopsy, cause of death, recurrence of tumor (local vs. distant), and other treatments administered. Clinical stage of disease was determined based on information available in the medical records. Local recurrence of tumor was defined as any mast cell tumor that recurred within 2 cm of the original tumor. Since the distinction between distant recurrence and metastasis cannot be made in most cases, all distant appearances of mast cell tumor were considered to be tumor metastases. Follow-up information of LSU-SVM cases only was obtained through physical examination or telephone contact with dog owners or referring veterinarians.
World Health Organization17 staging system for canine mastocytomas.

Grading
All tissue samples were initially fixed in buffered formalin (not exceeding 4% formaldehyde) prior to embedding in paraffin. Samples of each section stained by hematoxylin and eosin (HE) were reviewed by one pathologist using light microscopy and assigned a histopathologic grade. Tumors were graded according to the histopathologic criteria defined by Patnaik et al. (Table 2).18 Information regarding the duration of formalin fixation prior to sectioning was not available for all samples.
Histopathologic grading system of mast cell tumors.18

Immunohistochemical staining
All steps in the immunohistochemical staining process were performed at room temperature unless otherwise noted. Tissue samples were sectioned to 2 μm, placed on silanized slides, and allowed to dry overnight in a 37°C oven. The following day, the slides were dried in a 55°C oven and then deparaffinized in xylene and rehydrated in distilled water. The slides were immersed in hydrogen peroxide in methanol. The slides were rinsed in distilled water followed by phosphate-buffered saline with Brij-35 (PBS-BRIJ) added. Slides were immersed in citrate buffer (BioGenex Laboratories, San Ramon, CA) and microwaved to retrieve antigens. After incubation in the citrate buffer, slides were cooled and rinsed in PBS-BRIJ. Slides were then incubated with normal goat serum (Vectastain Elite ABC Kit B, Vector Laboratories, Burlingame, CA) diluted in PBS-BRIJ. Polyclonal rabbit anti-p53 oncoprotein (Signet Laboratories, Bedham, MA) antibody (CM-1) diluted with normal goat serum in PBS-BRIJ was applied and incubated overnight.
The following day, slides were rinsed in PBS-BRIJ. Biotinylated goat anti-rabbit immunoglobulin G (IgG) (Vectastain Elite ABC Kit B, Vector Laboratories, Burlingame, CA) diluted in PBS-BRIJ was applied and allowed to incubate. The reagents for the avidin–biotin complex (ABC) method were prepared and added to the slides. The ABC reagent was removed with PBS-BRIJ. Slides were coated with diaminobenzidine (DAB) and incubated. Color development of the immunoprecipitate was monitored under a microscope during this reaction. When a brown immunoprecipitate was imparted to the slide, the reaction was halted. Copper sulfate was applied to the slides to enhance staining and toning of the DAB. Mayer's hematoxylin was applied as a counterstain. Slides were dehydrated through graded ethanol, cleared in xylene, mounted in a resinous mounting medium, cover-slipped, and labeled.
Controls
Negative controls consisted of sample tissue sections in which the primary antibody was replaced with rabbit serum IgG but otherwise stained similarly using immunohistochemical staining techniques. Paraffin-embedded canine tracheal epithelial cells transformed with the simian virus 40 (SV40) large T antigen (ACT8/SV40; Inhalation Toxicology Research Institute, NM) served as positive controls and were stained similarly to mast cell tumor samples.
Immunohistochemical scoring
Microscopic examination of slides was performed using 40× light microscopy. Eight to 10 fields covering at least 900 mast cells were counted per tissue section. Mast cells containing chromagen stain within their nucleus were considered to be positive for the presence of p53. For each slide, both positively and negatively stained mast cells were counted and the overall percentage of positively stained mast cells were calculated.
Statistical analysis
Histologic data
Each tumor was treated as an independent event. Since the intent of the study was to assess p53 staining of individual tumors, it was presumed that the staining of one tumor was distinct from another, regardless of whether it was obtained from the same dog. For each tissue sample examined, the percentage of cells staining positive for mutant p53 was recorded.
The data were tested for normality using the Shapiro-Wilk test with
Clinical data
Median and mean survival times and times to recurrence of tumor were estimated using a Kaplan-Meier analysis for the data acquired from medical records. Where an endpoint was unknown, the data were included as censored data (endpoint not reached). Death and euthanasia were considered to represent the same endpoint. Death or euthanasia was considered an endpoint if it was related to mast cell tumor disease. If the cause of death or euthanasia was unknown, it was assumed to be due to mast cell tumor disease. Dogs known to have died from or to have been euthanatized for other causes were included as censored data. Each dog was only included in the analysis once. To be conservative, for dogs with multiple tumors, the shortest time for recurrence and the highest histologic grade were used for that dog in the analysis.
Covariate analysis was used to determine any significant association of variables with survival time or time to recurrence. In exploring the data, each variable was tested independently. A log-rank test was used to test for significant association of each variable and survival time or time to recurrence. Significant variables were then added in forward stepwise modeling using the log-rank test to determine any gain in information by including more than one variable in the model. Variables used in the analysis were histologic grade, clinical stage, percentage of cells staining for p53, and tumor recurrence. The percentage of cells staining positive for p53 was evaluated as a continuous variable to avoid the loss of information that results from categorization of a continuous variable, particularly categorization to a binary result.4,8 However, for completeness, the analysis was also run using the p53 percentage in a dichotomous form: stain score of <10% or ≥10%.5,19,20 To be conservative, significance was determined at
Results
Histologic data
A total of 126 mast cell tumors were evaluated by light microscopy. Fifty-six tumors (44.4%) were determined to be grade I, 35 grade II (27.8%), and 35 grade III (27.8%).
The median percentage of cells staining positive for p53 for all tumors was 13.75% (range, 0.00–98.81%). The median percentage of cells positive for p53 (Fig. 1) for grade I tumors was 9.56% (range, 0.00–98.81%), 6.40% for grade II tumors (range, 0.00–92.65%), and 41.45% for grade III tumors (range, 0.00–98.28%). There was a significant difference in the percentage of cells staining positive for p53 among histopathologic grades (
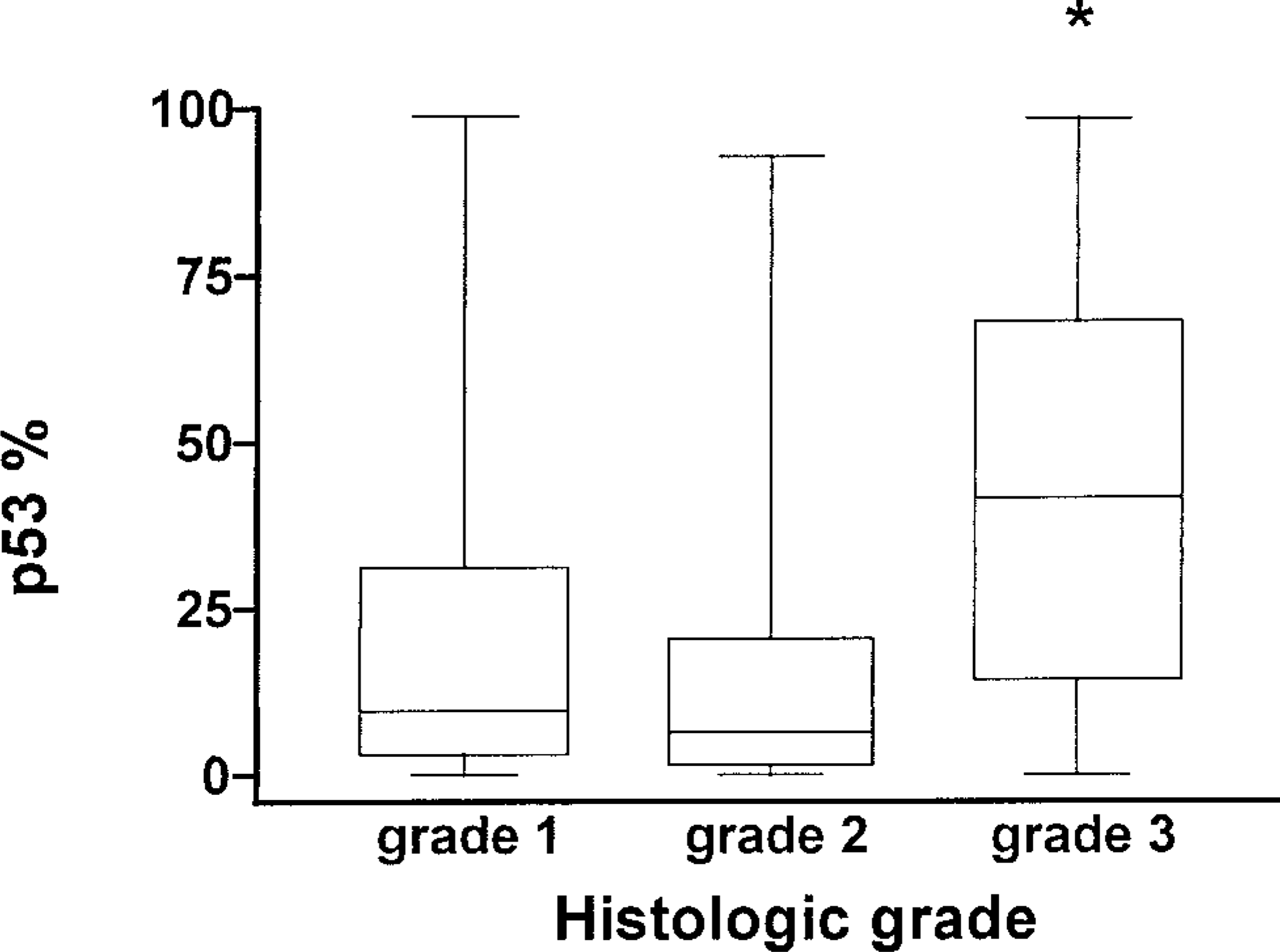
Box and whisker graph displaying the median percentage of cells positive for the presence of p53 (horizontal line) [p53%], range (whiskers), and 50% of data (box) for each histologic grade of mast cell tumor. An asterisk indicates that grade III is significantly greater than grades I and II at
Clinical data
Overall, 126 tumors from 106 dogs were evaluated. The breed was recorded for 103 dogs. Thirty-one breeds were represented, with Labrador Retrievers represented most often (
Location of tumor was recorded for 121 tumors. Fifty-five tumors (45.5%) occurred on the abdomen or thorax, 44 (36.4%) on the extremities, 10 (8.3%) on the head, 8 (6.6%) on the perineum, 3 (2.5%) on the scrotum, and 1 (0.8%) on the vulva.
The clinical stage of disease was determined for 70 dogs according to criteria in Table 1. Forty-six dogs (65.7%) had stage 1 disease, three (4.3%) had stage 2, 17 (24.3%) had stage 3, and four (5.7%) had stage 4 disease.
Of the 54 dogs for which follow-up information was available, tumor recurrence or metastasis occurred in 19 (35.2%). Eight of these 19 dogs (42.1%) had local recurrence of the tumor. Metastasis to regional lymph nodes occurred in 2 of 19 dogs (10.5%). The remaining dogs had distant metastases.
Twenty-five dogs had died or were euthanatized by the end of the study period. Four dogs diagnosed with mast cell tumors at necropsy were excluded from the Kaplan-Meier analysis. Nine of the 25 dogs (36.0%) were euthanatized because of direct effects of their mast cell tumor. The remaining 16 dogs died or were euthanatized because of other illnesses. Median survival time and time to recurrence estimates were variable. The estimates for all dogs were 1,924 days and 180 days, respectively.
Single covariate analysis using the percentage of cells staining positive for p53 in its continuous form showed a significant negative association of clinical stage and time to recurrence (
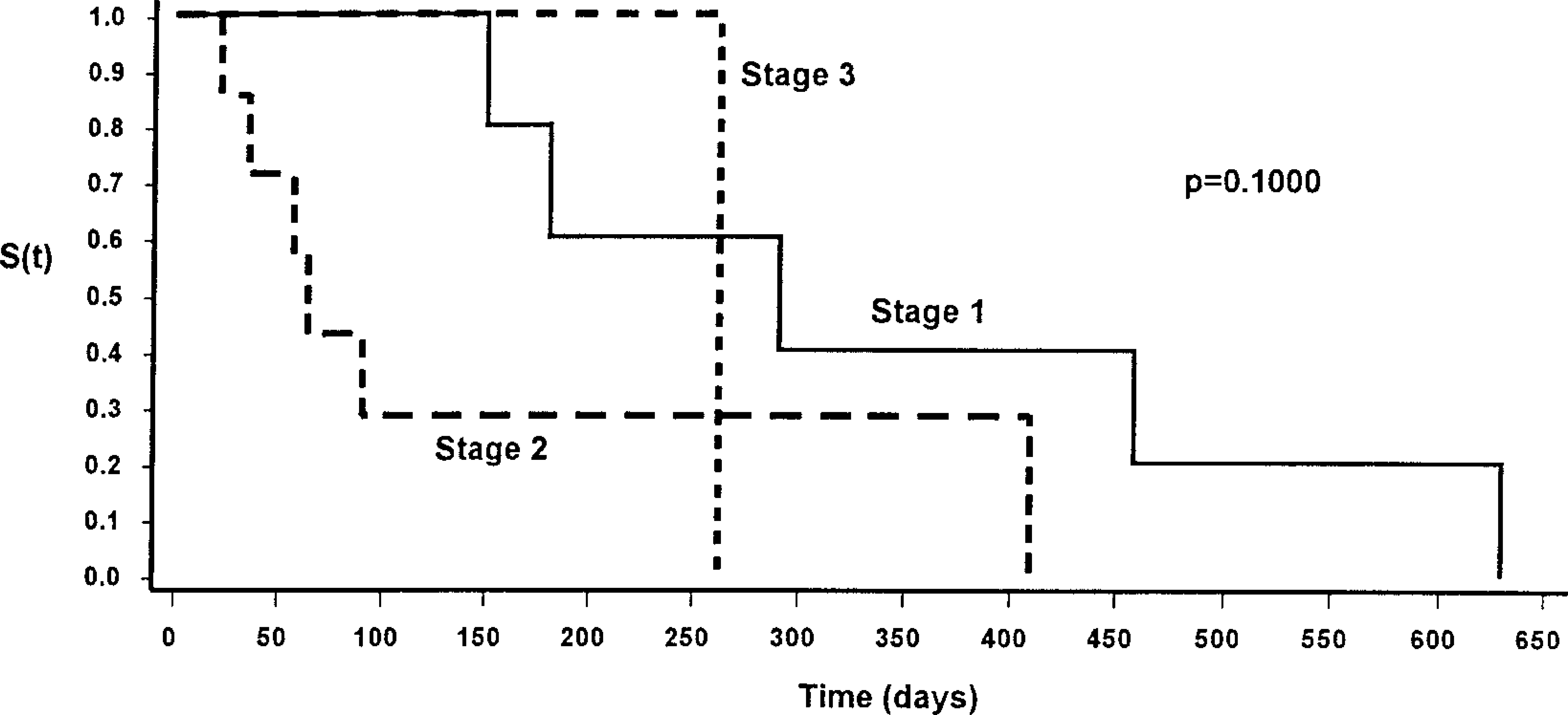
Kaplan-Meier survival function [
Single covariate analysis using the percentage of cells staining positive for p53 in its continuous form showed a significant negative association of tumor recurrence and survival time (
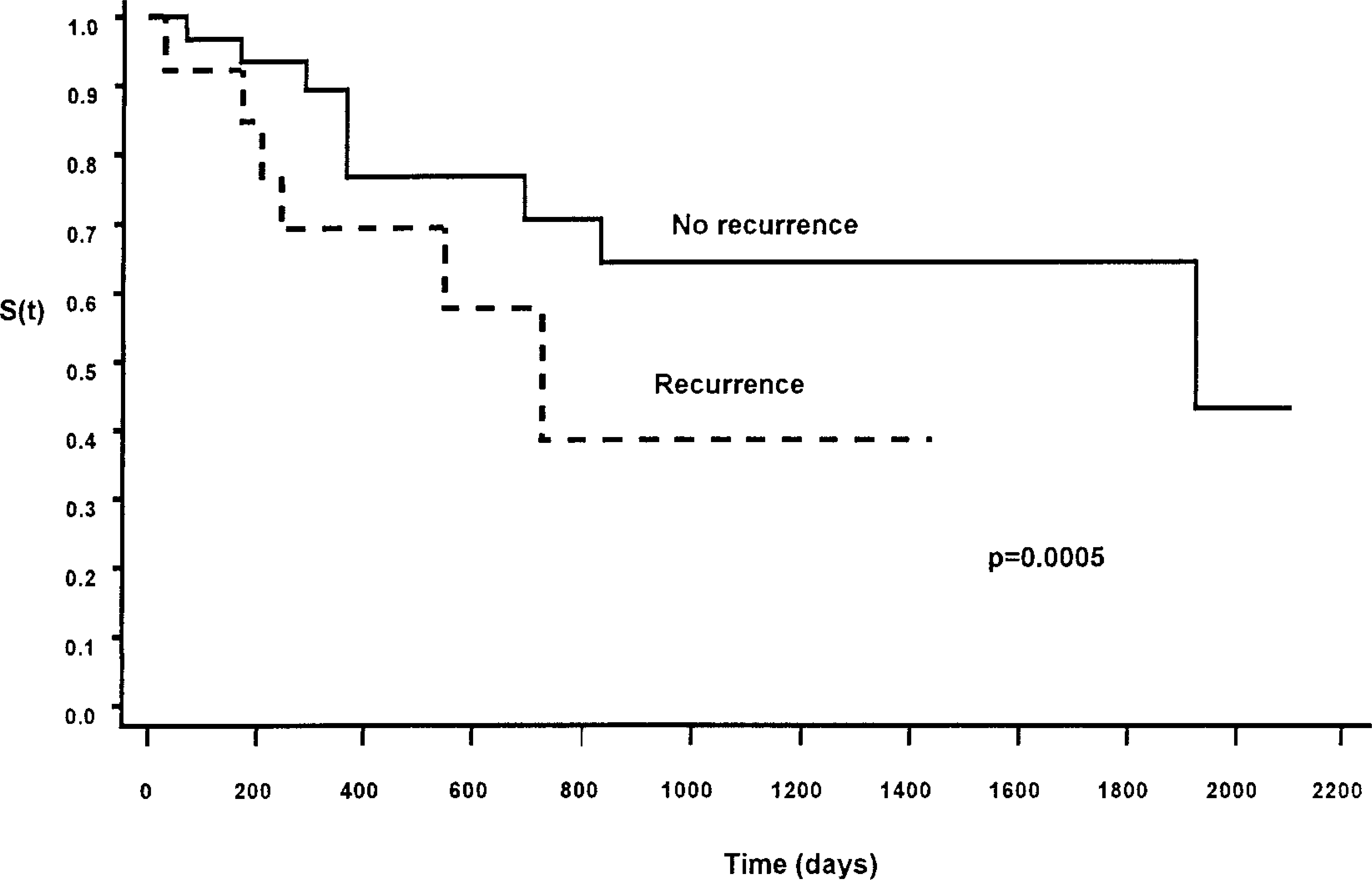
Kaplan-Meier survival function [
Discussion
Since grade III tumors had a significantly greater percentage of cells staining for p53 than grade I and II tumors, it would appear that the p53 staining is somewhat consistent with histologic grade. However, no difference was observed between grade I and II tumors, and the overlap of the ranges of the percentage of cells staining for p53 makes this criteria unreliable for discriminating between histologic grade. Some grade I mast cell tumors had a high percentage of cells staining for p53, whereas some grade III tumors had a very low percentage. We hoped this variability might add to the prognostic value of histopathologic grade in estimating survival time or time to tumor recurrence.
Based on previous work, histopathologic grade was used as the standard for prognosis estimation in this study.3,18 In the clinical situation, histopathologic grade is believed to be the most sensitive indicator of prognosis when coupled with completeness of tumor excision and clinical stage of disease. Anecdotal clinical observations have suggested that some grade I mast cell tumors behave malignantly while some grade III mast cell tumors behave benignly. This study shows that immunohistochemical staining for p53 offers no advantage over histopathologic grade with regard to association with survival time or time to recurrence. Similarly, a previous study showed the percentage of cells staining positive for p53 within tumor cells offered no advantage in the diagnostic confirmation or prognostication of colorectal neoplasms in dogs.26
Much of the clinical information derived from case records regarding signalment, history, and location of tumors was consistent with previous studies.6,9,10,12–14,16,21,23 As determined by the covariate analysis, tumor recurrence and histopathologic grade are negatively associated with survival time. This is consistent with the findings of other clinical studies.3,18 Clinical stage and histopathologic grade were shown to be negatively associated with time to tumor recurrence. It was noted, however, that the percentage of cells that stained positive for p53 in continuous or dichotomous form did not enhance the analysis of associations with survival time or time to tumor recurrence.
Tumor recurrence and survival times of dogs with cutaneous mast cell tumors are influenced by method of treatment. In all cases in this study, dogs were treated by surgical excision of the mast cell tumor. Adjunctive treatments following surgery included intralesional injections of deionized water (
The CM-1 polyclonal antibody used in our study binds with both mutant and wild-type p53. Polyclonal antibodies are generally less specific but have a greater stain sensitivity when compared to monoclonal antibodies.15 However, since the half-life of mutant p53 is long (up to 12 hours) and wild-type p53's half-life is comparatively short (30 minutes), it is assumed that the amount of wild-type p53 stained by the polyclonal antibody is negligible.2 The results of immunohistochemical staining of mast cell tumors using the CM-1 antibody in this study were similar to a previous study that evaluated p53 immunohistochemical staining prevalence of various canine tumors.7
Based on the information observed in our study, we conclude that immunohistochemical staining of canine cutaneous mast cell tumors for the estimation of the percentage of cells staining positive for p53 offers no improvement over histopathological grading or clinical staging for association with survival time or time to tumor recurrence. Although grade III tumors did show a significantly greater percentage of cells staining positive for p53, overall improvement in the grading of mast cell tumors by this method was not observed. The presence of p53 within individual cells of a mast cell tumor does not offer a consistent or reliable indication of the clinical course of the dog's disease. Tumors that had no cells positive for p53 or tumors with a very high percentage of cells staining for p53 were dispersed through all histologic grades of tumor evaluated. This indicated that even negative immunohistochemical findings are not reliable indicators of low histologic grade.
