Abstract
To elucidate the histopathological characteristics and immunophenotypes of canine transmural “mass-forming” gastrointestinal lymphomas and plasmacytomas, 83 surgically resected biopsy samples were examined. All lymphomas and plasmacytomas were located in the small or large intestine except for 1 plasmacytoma which was in the stomach. According to the World Health Organization (WHO) classification, B-cell neoplasms (17 cases) included lymphoplasmacytic lymphoma (6/17), plasmacytoma (5/17), follicular lymphoma (3/17), and diffuse large B-cell lymphoma (3/17). Based on nuclear sizes, T-cell neoplasms (66 cases) were broadly divided into large cell lymphoma (LCL; 48/66) and small cell lymphoma (SCL; 18/66). According to the WHO classification, T-cell neoplasms included anaplastic large T-cell lymphoma (ALCL; 10/66), angiotropic T-cell lymphoma (3/66), mixed inflammatory type peripheral T-cell lymphoma (mixed inflammatory type PTCL; 33/66), and PTCL-not otherwise specified (PTCL-NOS; 20/66). Mixed inflammatory type PTCLs were further divided into histiocyte- (27/33) and eosinophil- (6/33) dominant types. Immunohistochemically, lymphoplasmacytic lymphomas were positive for Pax5 (6/6) and IgM (5/6), while plasmacytomas were positive for IgG (5/6) and negative for Pax5. LCLs were immunopositive for granzyme B in 31/48 cases (65%) and CD8 in 9/48 cases (19%), while SCLs were immunopositive for granzyme B in 3/18 cases (17%) and CD8 in 3/18 cases (17%). Furthermore, 8/10 cases (80%) of ALCL and 19/27 cases (70%) of histiocyte-dominant PTCL were immunopositive for granzyme B, whereas 6/20 cases (30%) of PTCL-NOS, 1/6 cases (17%) of eosinophil-dominant PTCL, and no cases of angiotropic T-cell lymphomas were immunopositive for granzyme B. The present study describes the immunophenotypes in different histological types of transmural gastrointestinal lymphomas in the dog.
Gastrointestinal lymphoma is the most common extranodal lymphoma in dogs and accounts for 5% to 7% of all canine lymphomas. 20 Most canine intestinal lymphomas are of T-cell origin, whereas B-cell lymphomas, particularly diffuse large B-cell lymphoma (DLBCL) and mucosa-associated lymphoid tissue (MALT) lymphoma, are more common in humans. 5,33 Among canine B-cell lymphomas, MALT lymphoma is rare and has not been reported in the gastrointestinal tract. 38 On the other hand, DLBCL and follicular lymphoma (FL) occur as primary gastric or large intestinal lymphoma and less frequently as small intestinal lymphoma. 28,40 In addition to these relatively well-examined lymphomas of B-cell origin, B-cell lymphoma characterized by the proliferation of uniform medium-sized lymphocytes admixed with Mott cells and diagnosed as B-cell lymphoma with Mott cell differentiation has been reported. 6,11,31
In the histological classification of hematopoietic tumors of domestic animals, intestinal T-cell lymphoma (ITCL) is defined as a slowly progressive small cell lymphoma of the enteric tract, 39 which has also been called canine ITCL of the small cell type. 3,14,40 On the other hand, canine ITCL of the large cell type has often been classified as peripheral T-cell lymphoma (PTCL) or large granular lymphocyte (LGL) lymphoma, both of which are highly heterogeneous groups. 38,39,41 Feline ITCLs have recently been divided into mucosal and transmural types based on the extent of tumor infiltration in full-thickness biopsy samples. 19 In dogs, mucosal lesions of ITCLs have been classified into large cell lymphoma (LCL) and small cell lymphoma (SCL), which have different prognoses and proliferative activities. 14,21 However, a detailed classification of canine transmural ITCL has not yet been performed.
Previous studies have mostly focused on the mucosal lesions of T-cell lymphoma in the canine intestine. The present study examined full-thickness biopsy samples of localized, mass-forming or transmural gastrointestinal lymphomas in dogs. The aim of this study was to demonstrate the histological features and immunophenotypes of neoplastic lymphocytes, and also the breed predisposition and tumor localization of transmural gastrointestinal lymphomas in dogs.
Materials and Methods
Case Selection
Ninety-four full-thickness, surgically resected, formalin-fixed tissues of canine intestinal lymphoma and plasmacytoma were submitted for pathological examinations between 2009 and 2020 to the Laboratory of Veterinary Pathology, Graduate School of Agricultural and Life Sciences, the University of Tokyo. Among these cases, 83 were included in the present study. Ten cases were excluded because of the difficulties associated with reaching a detailed classification due to poor sample quality, and another case was excluded because of severe inflammation accompanied by nematode infection. Information on clinical signs, postmortem findings, and follow-up information was not available or insufficient and, thus, was not included. All samples were fixed in 10% neutral buffered formalin solution, processed routinely, and embedded in paraffin wax.
Histopathology
Four-micrometer-thick paraffin sections were deparaffinized, rehydrated through a graded series of alcohol, and stained with hematoxylin and eosin. A histopathological diagnosis was performed by 2 veterinary pathologists accredited by the Japanese College of Veterinary Pathologists (KU and JKC). In each case, the nuclear size of neoplastic cells was evaluated as small (<1.5× the size of a red blood cell), medium (1.5–2× the size of a red blood cell), or large (>2× the size of a red blood cell). An inflammatory background was defined by the presence of inflammatory cells accounting for more than 10% of all cells and areas of ulceration or necrosis were excluded in this assessment. For identification of histiocytes, immunohistochemistry for Iba-1 was also performed in all cases. Mucosal ulceration was not included in assessment of necrosis. Mitotic count was determined by manually counting and summing the number of mitotic figures in 10 fields using a microscope with 40× objective and ocular of FN 20 mm and multiplying by 1.17. 17
Immunohistochemistry
Details on primary antibodies and antigen retrieval procedures are summarized in Supplemental Table S1. Following antigen retrieval, endogenous peroxidase was inactivated with 3% hydrogen peroxide in methanol at room temperature for 5 minutes. Sections were blocked with 8% skim milk in Tris-buffered saline (TBS) at 37 °C for 40 minutes, and then incubated with primary antibodies at 4 °C overnight. After washing with TBS, each secondary antibody was applied. In the Dako Envision+ System (Dako), sections were incubated with an anti-rabbit or anti-mouse hydrogen peroxidase–labeled secondary antibody polymer at 37 °C for 40 minutes. In the Ventana OptiView DAB Universal Kit (Ventana Medical Systems), sections were incubated with Linker-HQ at 37 °C for 8 minutes, and then incubated with Multimer-HRP at 37 °C for 8 minutes. To detect dog IgM, IgG, IgA, or CD8α, 30 sections were incubated with biotin-conjugated anti-goat immunoglobulin G (1:400; KPL) or anti-rat immunoglobulin G (1:500; Dako) at 37 °C for 40 minutes, and then with horseradish peroxidase–conjugated streptavidin (1:400; Dako) at 37 °C for 40 minutes. Labeled complexes were visualized with 3,3′-diaminobenzidine (DAB) chromogen and 0.03% hydrogen peroxidase in Tris-HCl buffer and then counterstained with hematoxylin. Canine-activated lymph node or normal intestine was used as positive and negative tissue controls, and negative antibody controls were performed by applying irrelevant antibody and buffer instead of the primary antibodies. 26
Double-labeling immunohistochemistry for CD3 and Ki67 was performed on T-cell lymphoma sections to detect Ki67-positive neoplastic T cells. After heat-induced antigen retrieval (pH 6.0), endogenous peroxidase was inactivated with 3% hydrogen peroxide in methanol at room temperature for 4 minutes. Sections were blocked with 8% skim milk in TBS at 37 °C for 40 minutes and then incubated with the anti-Ki67 antibody at 4 °C overnight. After washing with TBS, sections were incubated with an anti-mouse hydrogen peroxidase–labeled secondary antibody polymer (Envision+ System; Dako) at 37 °C for 40 minutes. Labeled complexes were visualized with DAB chromogen and 0.03% hydrogen peroxidase in Tris-HCl buffer. Sections were then heated at 121 °C for 1 minute in citrate buffer (pH 6.0) to inactivate the anti-Ki67 antibody and anti-mouse secondary antibody. Sections were then blocked with 8% skim milk in TBS at 37 °C for 40 minutes and incubated with the anti-CD3 antibody at 4 °C overnight. After washing with TBS, sections were incubated with an alkaline phosphatase-conjugated anti-rabbit secondary antibody polymer (N-Histofine Simple Stain AP; Nichirei Biosciences), the immunoreaction was visualized using the N-Histofine New Fuchsin substrate (Nichirei Biosciences), and sections were counterstained with hematoxylin. Canine-activated lymph node was used as positive and negative tissue controls, and negative antibody controls were performed by buffer instead of the primary antibodies. 26 Representative images of double-labeling immunohistochemistry are shown in Supplemental Figures S1 and S2.
Immunohistochemical staining of the neoplasm in each case was considered to be positive when more than 30% of neoplastic cells were positive, except for Ki67. Ki67 labeling and CD3/Ki67 double labeling were examined at a medium magnification to select areas with the strongest proliferative activity. Within these areas, 5 fields at high magnification (40× objective, 0.0625 mm2) were photographed. The numbers of Ki67-positive and Ki67-negative nuclei were counted in each image using ImageJ software (National Institutes of Health) and the Ki67 index was calculated as the mean percentage of Ki67-positive nuclei. Regarding CD3/Ki67 double labeling, the number of Ki67-positive nuclei was assessed in CD3-positive cells in the same manner and the Ki67 index of CD3-positive cells was calculated. Ki67 index of 3 cases of B-cell neoplasms and 8 cases of T-cell neoplasms were 0% and thus were excluded from the Ki67 index assessment.
Classification Design
CD3-positive lymphomas were classified as T-cell lymphoma and CD3-negative tumors were classified as B-cell neoplasms if they were additionally positive for either CD20, Pax5, CD79α, or immunoglobulins. B-cell neoplasms were further classified based on the World Health Organization (WHO) histological classification for domestic animals. 39 T-cell lymphomas were broadly classified into LCL and SCL according to their nuclear size relative to red blood cells (RBC): LCL, >2 × RBC; SCL, <2 × RBC. 14 T-cell lymphomas were further subclassified based on the WHO histological classification 39 : anaplastic large T-cell lymphoma (ALCL), angiotropic T-cell lymphoma, PTCL, mixed inflammatory types, and PTCL-not otherwise specified (PTCL-NOS).
Statistical Analysis
The mean ages of dogs with B-cell neoplasms and T-cell lymphomas were compared with Welch’s t-test. The mean mitotic count and Ki67 index of LCLs and SCLs were compared using the Student’s t-test and Welch’s t-test, respectively. Variances were compared with the F test. The frequencies of histological features and immunohistochemical results were compared with Fisher’s exact test. ANOVA with post hoc Tukey HSD was used to compare the Ki67 index between each subtype of T-cell lymphoma. P values <.05 were considered to be significant. All statistical analyses were conducted with Statcel4 software (OMS Publishing Inc).
Results
Clinical Features and Tumor Location
Among the 83 cases examined, 17 were diagnosed as B-cell neoplasms and 66 as T-cell neoplasms according to the classification design. The age of dogs with transmural intestinal lymphomas ranged between 1 and 11.4 (mean 6.7) years for B-cell neoplasms and between 4 and 16.9 (mean 10.3) years for T-cell neoplasms. Mean age was significantly lower in dogs with B-cell neoplasms than in those with T-cell neoplasms (P < .0006). The proportion of Miniature Dachshunds in B-cell neoplasm cases (10/17, 59%) was higher than the proportion of Miniature Dachshunds in the canine population of Japan (6.6%; 2020 Japan Kennel Club survey). Among T-cell lymphoma cases, Pugs (12/66, 17%) were the most common, followed by Shiba dogs (6/66, 9%); the proportions of Pugs and Shiba dogs in T-cell neoplasm cases were higher than their proportions in the canine population of Japan (3.0% Shiba dog and 1.8% Pug). B-cell neoplasms were most commonly located in the large intestine (10/17, 59%), followed by the small intestine (6/17, 35%) and stomach (1/17, 6%). T-cell lymphomas were mostly located in the small intestine (64/66, 97%) and rarely in the large intestine (2/66, 3%). The age, sex, breed, and tumor location of each case are summarized in Supplemental Tables S2 and S3.
Histological Features and Tumor Classification
B-Cell Lymphomas
According to the WHO classification, 39 B-cell neoplasms were classified into lymphoplasmacytic lymphoma (LPL; 6 cases), FL (3 cases), DLBCL (3 cases), and plasmacytoma (5 cases).
The anatomical locations of LPLs were the small intestine in 5 cases and the large intestine in 1 case. Histopathologically, LPL was characterized by diffuse infiltration of small B cells and plasmacytoid cells in the submucosa and muscle layers, and mild infiltration in the lamina propria. Plasmacytoid cells had uniformly small densely stained nuclei with moderate amounts of densely stained cytoplasm, and nucleoli were inconspicuous (Fig. 1). In 2 cases (cases 4 and 5), a mixture of medium- to large-sized neoplastic lymphocytes were observed. Various numbers of Mott cells and mucosal ulceration were detected in all cases (Fig. 2). Mitotic count ranged from 7 to 48 with an average of 24.
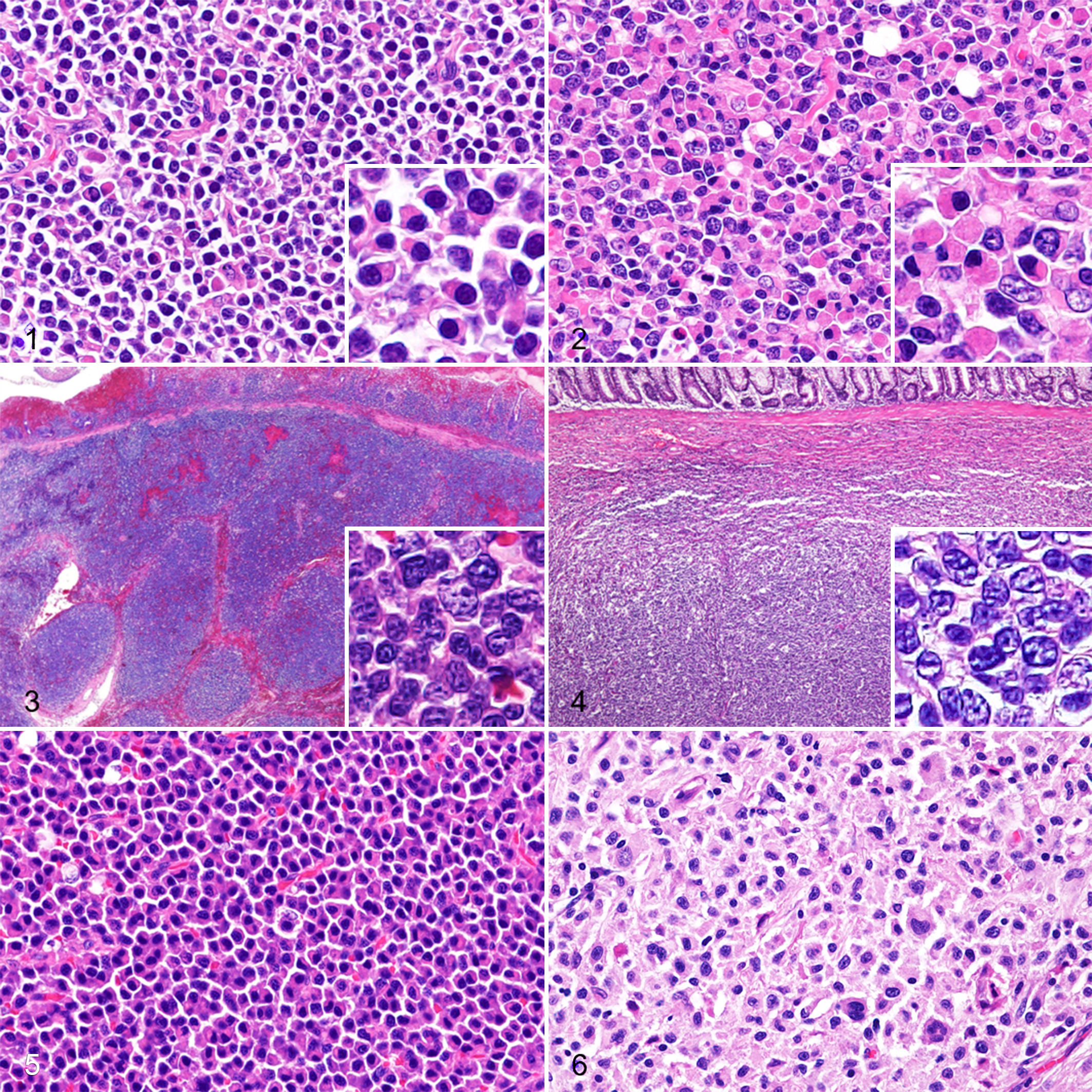
B-cell neoplasms, gastrointestinal tract, dog. Hematoxylin and eosin.
The anatomical location in all 6 cases of FL and DLBCL was the large intestine. FLs were characterized by follicles in the submucosa with expanded germinal centers and a fading mantle zone (Fig. 3). These germinal centers consisted of centrocytes with medium-sized nuclei and small nucleoli admixed with centroblasts with large nuclei and a prominent nucleolus. Neoplastic lymphocytes often infiltrated the lamina propria and focal to extensive erosion was noted in all cases. DLBCLs were characterized by diffuse proliferation of centroblasts with large nuclei and a prominent nucleolus infiltrating the submucosa and muscle layer (Fig. 4). In 3 cases, neoplastic lymphocytes also infiltrated the lamina propria, with mucosal erosion or ulceration. Mitotic count ranged from 19 to 73 with an average of 51 in FL and from 22 to 54 with an average of 42 in DLBCL.
Plasmacytomas were characterized by diffuse nodular proliferation of neoplastic cells in the submucosa and lamina propria, and the cells had eccentric nuclei and perinuclear clear zones. Mucosal erosion or ulceration was detected in all cases. Plasmacytomas were further subclassified into indolent plasmacytoma (2 cases) and anaplastic plasmacytoma (3 cases). Indolent plasmacytomas had a well-differentiated plasma cell morphology (Fig. 5); anaplastic plasmacytomas showed severe anisocytosis and anisokaryosis with cleaved nuclei in large cells (Fig. 6). Neoplastic cells in anaplastic plasmacytoma also had eccentric nuclei and occasional perinuclear clear zones, showing a plasmacytoid morphology. Mitotic count was 8 and 18 in the 2 indolent plasmacytoma cases, and ranged from 23 to 78 with an average of 48 in anaplastic plasmacytoma.
T-Cell Lymphomas
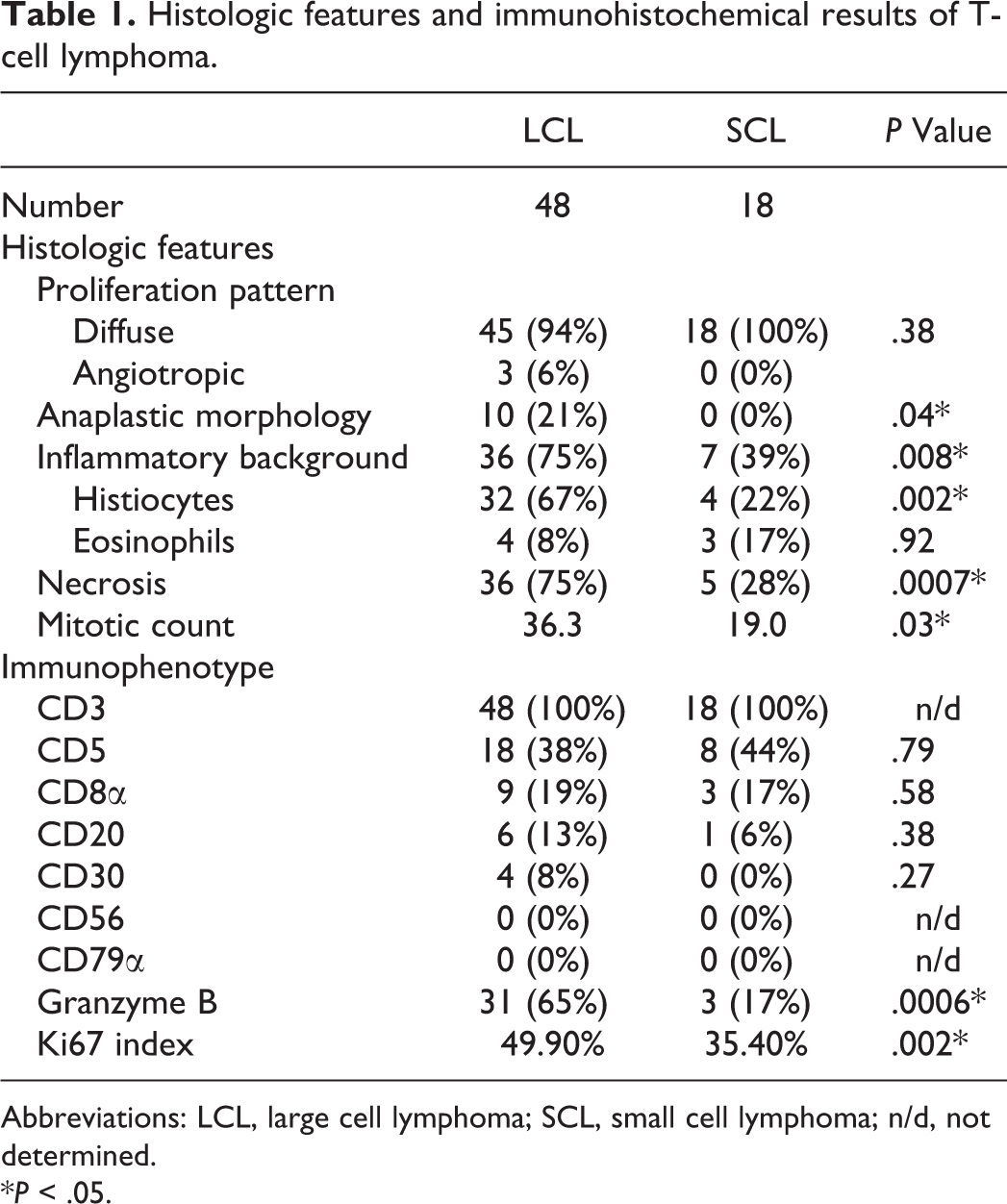
The histopathological features of ITCL are summarized in Table 1. ITCL cases showed transmural infiltration of neoplastic lymphocytes often involving mesenteric adipose tissue. Mucosal erosion or ulceration was observed in all cases. According to cell sizes, T-cell neoplasms were broadly classified into LCL (48 cases) and SCL (18 cases). LCLs were characterized by large cells (>2 × RBC) with pleomorphic, round to ovoid nuclei containing scattered chromatin and distinct nucleoli (Fig. 7). An anaplastic morphology, such as a horseshoe-shaped or pleomorphic nucleus, was noted in 10/48 LCL cases (Fig. 8). SCLs (<2 × RBC) were characterized by round and monomorphic nuclei with dense chromatin (Fig. 9).
Histologic features and immunohistochemical results of T-cell lymphoma.
Abbreviations: LCL, large cell lymphoma; SCL, small cell lymphoma; n/d, not determined.
*P < .05.
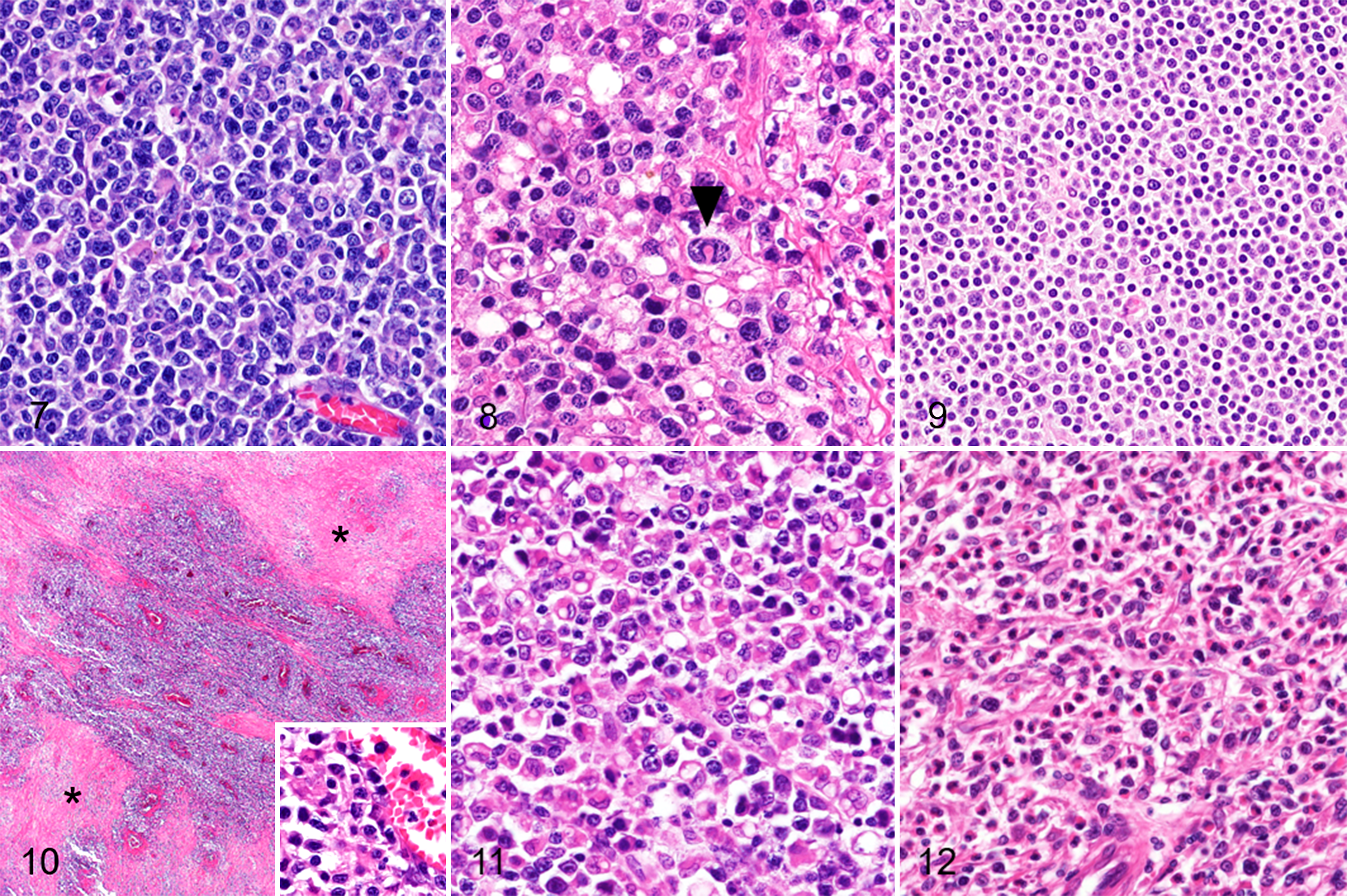
T-cell lymphoma, small intestine, dog. Hematoxylin and eosin.
Angiotropic infiltration was detected in 3/66 cases (5%) and extensive necrosis in 41/66 cases (62%). Transmural ischemic necrosis was also present in 3 cases with angiotropic infiltration (Fig. 10). Significant infiltration of histiocytes (Fig. 11) or eosinophils (Fig. 12) was observed in 43/66 cases (65%). Histiocytic infiltration was noted in 32/48 (67%) LCL cases and 4/18 (22%) SCL cases (P < .002). Necrosis was detected in 36/48 (75%) LCL cases and 5/18 (28%) SCL cases (P < .0007). The mean mitotic count was 36 in LCL cases and 19 in SCL cases (P < .03).
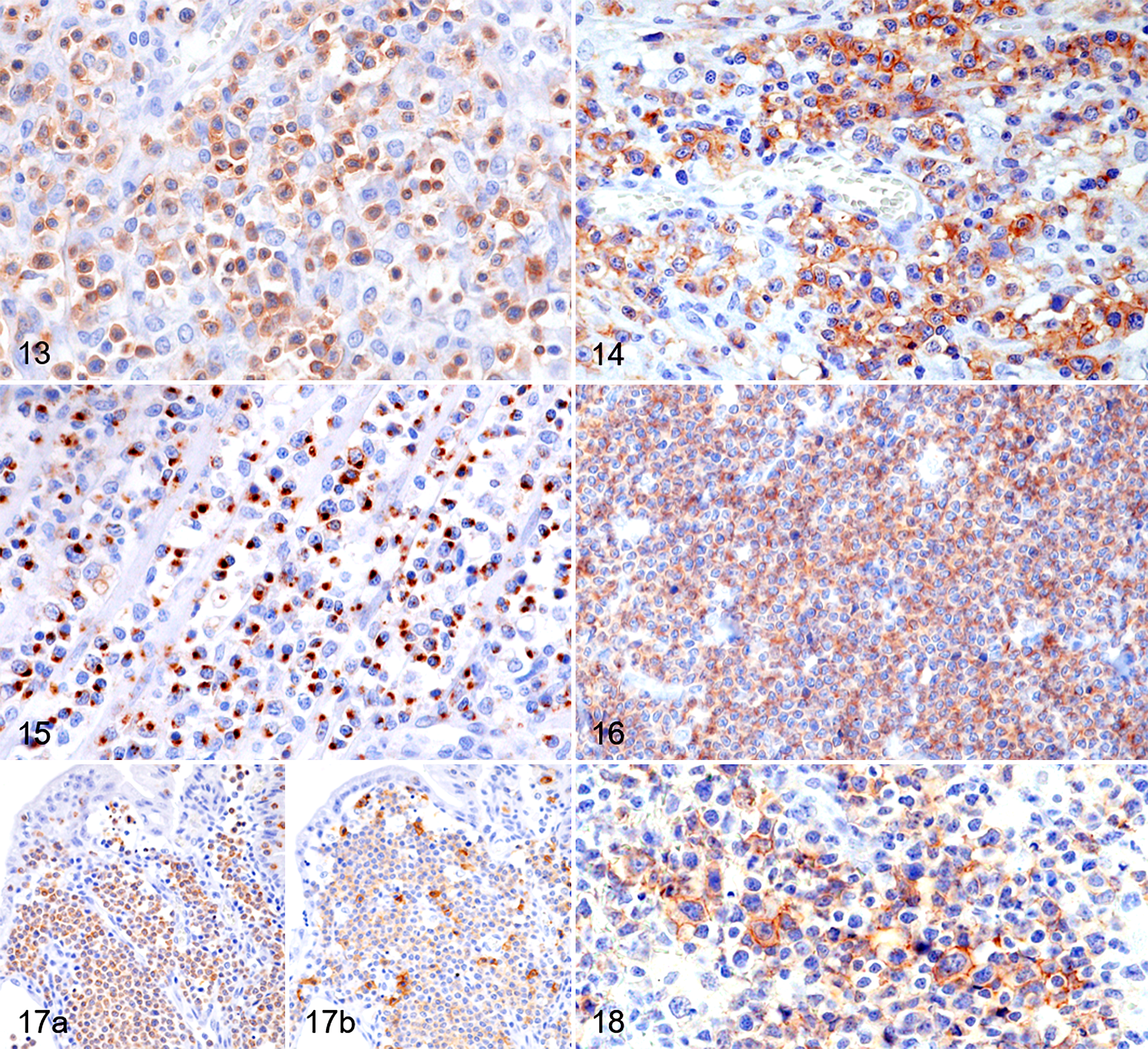
T-cell lymphoma, small intestine, dog. Immunohistochemistry.
According to the WHO classification, 39 the 66 cases of ITCL were classified based on their histological features as follows. LCLs with anaplastic morphology of the neoplastic lymphocytes were classified as ALCL (10/66, 15%) and those with angiotropism were classified as angiotropic T-cell lymphoma (3/66, 5%). Regarding the remaining cases, those with an inflammatory background were classified as “PTCL, mixed inflammatory types” (33/66, 50%), and further subclassified according to the predominant inflammatory cell as histiocyte-dominant (27 cases) and eosinophil-dominant (6 cases). Cases that did not fit into any subtype were classified as PTCL-NOS (20/66, 30%).
Immunohistochemistry
The immunohistochemical results of B-cell neoplasms are summarized in Table 2. All cases of LPL, FL, and DLBCL were positive for CD20, CD79a, and Pax5, except for 1 case of FL that was negative for CD79α. LPL, FL, and DLBCL cases were negative for MUM1. All cases of plasmacytoma were negative for CD20 and Pax5, whereas 1 case was positive for CD79α. Four out of the 5 cases of plasmacytoma were positive for MUM1. All cases of plasmacytoma were positive for immunoglobulin lambda light chain and negative for kappa light chain. Among LPL cases, 5/6 were positive for lambda light chain and negative for kappa light chain; 1/6 was negative for lambda light chain and positive for kappa light chain. Regarding the immunoglobulin heavy chain, all 5 plasmacytoma cases were positive for IgG. In addition, 1/5 cases was positive for IgM and 1/5 cases was positive for IgA. In LPL cases, 5/6 were positive for IgM and 1/6 was positive for IgG. All B-cell neoplasms were negative for CD3, CD5, CD30, CD56, and granzyme B (GRB).
Summary of immunohistochemical results of canine intestinal B-cell lymphoma.

Abbreviations: n, number of cases; n/d, not determined.
a Membrane and cytoplasmic staining was not observed in nonneoplastic B-cells in the canine lymph nodes.
The immunohistochemical features of T-cell lymphomas are summarized in Tables 1 and 3. All cases were positive for CD3 (Fig. 13) and negative for CD79α. Among 66 cases of T-cell lymphomas, 26 (39%) were positive for CD5 (Fig. 14), 34 (52%) were positive for GRB (Fig. 15), 12 (18%) were positive for CD8 (Fig. 16), 7 (11%) were positive for CD20 (Fig. 17a and b), and all cases were negative for CD56. Comparing LCL and SCL, 31/48 (65%) LCL cases and 3/18 (17%) SCL cases were positive for GRB (P < .0006). Comparing different histological subtypes, tumor cells were positive for GRB in 8/10 (80%) ALCL cases, 19/27 (70%) histiocyte-dominant mixed inflammatory PTCL cases, 6/20 (30%) PTCL-NOS cases, 1/6 (17%) eosinophil-dominant mixed inflammatory PTCL cases, and 0/3 (0%) of angiotropic lymphoma cases. Regarding CD30 immunohistochemistry, 3/10 (30%) ALCL cases and 1/27 (4%) histiocyte-dominant mixed inflammatory PTCL cases were positive (Fig. 18). Among the 7 cases that were positive for CD20, 6 (86%) were also positive for GRB; all of these cases were negative for CD8.
Summary of immunohistochemical results of canine intestinal T-cell lymphoma.
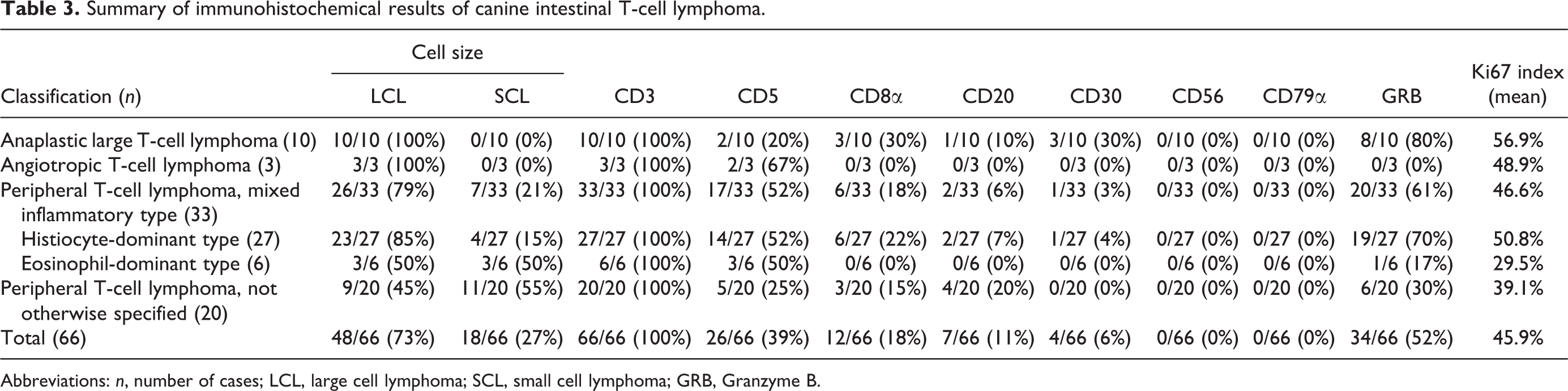
Abbreviations: n, number of cases; LCL, large cell lymphoma; SCL, small cell lymphoma; GRB, Granzyme B.
Ki67 Index
Among B-cell neoplasms, DLBCL showed the highest Ki67 index (mean 48.0%; range 32.7% to 56.6%); however, a statistical analysis was not performed due to the small number of cases.
Among T-cell neoplasms, the Ki67 index was significantly higher in LCL (mean 49.9%; range 24.7% to 82.9%) than in SCL (mean 35.4%; range 15.7% to 63.3%; P < .002). In comparing each histological subtype (Suppl. Fig. S3), ALCL showed a significantly higher Ki67 index (mean 56.9%; range 43.2% to 82.9%) than eosinophil-dominant mixed inflammatory PTCL (mean 29.5%; range 15.7% to 47.9%; P < .01) and PTCL-NOS (mean 39.1%; range 17.4% to 60.4%; P < .05). Histiocyte-dominant mixed inflammatory PTCL showed a significantly higher Ki67 index (mean 50.8%; range 25.9-69.0) than eosinophil-dominant mixed inflammatory PTCL (P < .01).
Discussion
The predominance of T-cell neoplasms (80%) over B-cell neoplasms (20%) in the present study was consistent with previous findings on canine intestinal lymphoma. 5 The present study revealed a high prevalence of B-cell neoplasms in young to middle-aged Miniature Dachshunds in Japan, suggesting a genetic predisposition in this breed. The high prevalence of T-cell lymphoma in Pugs and Shiba dogs was consistent with our previous study on the mucosal lesions of canine ITCLs. 16 The occurrence of T-cell lymphoma was almost exclusively confined to the small intestine (64/66, 97%), while FL and DLBCL were detected in the large intestine (6/6, 100%), which is consistent with previous findings. 28,40 LPL was more common in the small intestine (5/6, 83%) than in the large intestine (1/6, 17%).
DLBCL is the most common type of B-cell neoplasm in dogs. 25,38,41 However, LPL was the most frequent type of intestinal B-cell neoplasm in the present study. LPL was characterized by small- to medium-sized neoplastic lymphocytes admixed with plasmacytoid cells and Mott cells. Neoplastic cells were immunohistochemically positive for Pax5 and cytoplasmic IgM, consistent with human LPL, suggesting that the cell of origin may be B cells before the immunoglobulin class switch. 9,33 Although canine intestinal LPL has not been reported to date, cases that have been diagnosed as B-cell lymphoma with Mott cell differentiation are likely to represent the same disease entity. 6,11,31 In the present study, 2 LPL cases (cases 4 and 5) exhibited medium- to large-sized neoplastic lymphocytes and a high Ki67 index, which may be associated with increased malignancy. 7
Gastrointestinal plasmacytomas are uncommon in dogs and are mostly reported in the large intestine. 20,27,37 In the present study, plasmacytomas were located in the stomach (1/5, 20%), small intestine (1/5, 20%), and large intestine (3/5, 60%). Immunohistochemically, most plasmacytomas were positive for IgG, as shown in non-gastrointestinal plasmacytoma cases. 12,20 In addition to IgG, one case was positive for IgA and another case was positive for IgM; however, the significance of the expression of different immunoglobulin subtypes remains unknown. 12
In recent studies, the mucosal lesions of canine ITCLs have been classified into LCL and SCL. 3,14 In the present study, transmural ITCLs were classified into LCL and SCL and immunohistochemically examined. LCLs were positive for GRB exhibiting the cytotoxic phenotype, but were mostly negative for CD8. On the other hand, SCLs were mostly negative for both GRB and CD8, which is in contrast to the results showing a high frequency of GRB expression in both LCL and SCL in the mucosal lesions of canine ITCL cases. 14 Previous studies using flow cytometry revealed that most canine nodal T-cell lymphomas were CD4+CD8−, 8 whereas CD4−CD8+ or CD4−CD8− T-cell lymphoma predominated in extranodal lymphomas. 13 Immunohistochemically, canine cutaneous epitheliotropic lymphomas are consistently positive for CD8, 18 whereas canine hepatosplenic T-cell lymphomas are inconsistently positive for CD8, similar to the results on LCLs in this study. 10 Hepatosplenic T-cell lymphoma is characterized by the expression of GRB and γδ T-cell receptors; however, T-cell receptor expression was not investigated in the present study. Nevertheless, CD8 is a representative marker for cytotoxic T-cells that produce cytotoxic molecules including GRB, and thus a cytotoxic phenotype (ie, GRB-positive) without the expression of CD8 may be a characteristic phenotype of LCLs in canine transmural ITCLs. A non-cytotoxic phenotype of SCLs suggests a different origin of the neoplastic lymphocytes. Non-cytotoxic CD4+CD8− T-cells may be the origin of SCLs in canine transmural ITCL cases.
In the present study, 6 cases (13%) of LCL and 1 case (6%) of SCL were positive for CD20. CD20+ ITCL cases were consistently positive for GRB, but negative for CD8. T-cell lymphomas with expression of CD20 have been reported, including intestinal lymphoma in dogs and humans. 2,4,14,23,34 In our previous study of endoscopically collected mucosal lesions of canine ITCLs, the co-expression of CD3 and CD20 was immunohistochemically observed in 3/17 cases (18%) of LCLs and 21/33 cases (64%) of SCLs. 14 Different CD20 expression patterns of SCLs between the previous study (mucosal lesion) 14 and this study (transmural lesions) imply differences in the origins of neoplastic lymphocytes in these 2 groups.
In the 2017 WHO classification system, human ITCLs are classified into enteropathy-associated T-cell lymphoma (EATL, previously EATL type 1) and monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL, previously EATL type 2). 33,42 The mucosal lesions of canine LCL and SCL were recently compared to EATL and MEITL, respectively. 3,14 In the present study, most LCL cells showed the CD5−/+CD8−CD56−GRB+ immunophenotype, which is similar to human EATL that has a CD5−CD8−/+CD56−GRB+ immunophenotype. 33,42 Human EATLs commonly express CD30, particularly in cases with an anaplastic morphology; however, the majority of canine ITCL cases were negative for CD30, except in ALCL. Human EATL occurs in patients with celiac disease, which is an autoimmune inflammatory enteropathy against gliadin and other related prolamins. 29,33,42 A previous study implicated immune response against gliadin peptides in canine chronic enteropathy and intestinal lymphoma. 1,15 Neoplastic lymphocytes in canine SCL were mostly the CD5−/+CD8−CD56−GRB− immunophenotype, in contrast to human MEITL, which is commonly CD5−CD8+CD56+GRB+/−. 33,42 Differences in the immunohistochemical features of canine SCL and human MEITL indicate that these 2 groups are distinctively different diseases.
ALCL is a rare type of lymphoma in humans that is characterized by large cells with severe nuclear pleomorphism and CD30 expression. 33 A previous study revealed the expression of CD30 in canine intestinal ALCL 32 ; 3 cases of ALCL expressed CD30. In addition, ALCL showed a significantly higher Ki67 index than eosinophil-dominant mixed inflammatory PTCL and PTCL-NOS. Canine intestinal ALCL is reported to have a poor prognosis with an aggressive clinical course, 32 but information on clinical outcomes was not available in the present study.
Angiotropic T-cell lymphoma is histologically consistent with extranodal NK/T-cell lymphoma, nasal type (ENKL) in the WHO classification of humans. 33 ENKL is a predominantly extranodal lymphoma of mainly CD56+ NKT cells and sometimes a T-cell origin, and primary intestinal ENKL is rare. 43 Neoplastic lymphocytes of ENKL commonly express cytotoxic molecules and are positive for Epstein-Barr virus (EBV). 22,33,43 In the present study, angiotropic T-cell lymphoma exhibited the CD3+CD5+/-CD56- immunophenotype, which indicates a T-cell phenotype rather than that of NKT cells. Additionally, neoplastic lymphocytes were consistently negative for GRB.
Mixed inflammatory PTCL was the predominant type of canine intestinal lymphoma in the present study, and was further classified into histiocyte- and eosinophil-dominant subtypes. Although canine ITCL with eosinophil infiltration has been reported, a detailed immunophenotypical characterization was not performed. 24 In the present study, histiocyte-dominant cases showed the cytotoxic phenotype with the expression of GRB (70%) and CD8 (22%), whereas eosinophil-dominant cases mostly had a non-cytotoxic phenotype (17% GRB and 0% CD8 immunopositive). Furthermore, the Ki67 index was significantly higher in histiocyte-dominant than in eosinophil-dominant cases. In the process of lymphomagenesis, IL-5, which is a type 2 T-helper cell-associated cytokine produced by neoplastic lymphocytes, may recruit eosinophils. 20,35 Macrophages may be induced and activated by interferon-γ produced by cytotoxic T-cells, and interferon-γ is reportedly produced by intraepithelial lymphocytes in patients with celiac disease. 36 Therefore, the types of infiltrating inflammatory cells may reflect differences in proliferative activity and the immunological subset of neoplastic lymphocytes; histiocyte infiltration suggests neoplasms of cytotoxic T-cells, while eosinophil infiltration suggests neoplasms of T-helper cells.
In the present study, we histologically and immunohistochemically classified canine intestinal lymphocytic neoplasms using full-thickness biopsy samples. The results obtained revealed that the histological features possibly reflect the immunophenotype or cell of origin of neoplastic cells. For B-cell neoplasms, lymphoplasmacytic lymphomas appear to be derived from B cells before the immunoglobulin class switch. In T-cell lymphoma, LCL and SCL have distinctively different immunophenotypes, indicating different origins of the neoplastic lymphocytes. Furthermore, each histological classification of T-cell lymphoma correlates to a cytotoxic immunophenotype and the proliferative activity of tumor cells.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211030523 - Histopathological features and immunophenotyping of canine transmural gastrointestinal lymphoma using full-thickness biopsy samples
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211030523 for Histopathological features and immunophenotyping of canine transmural gastrointestinal lymphoma using full-thickness biopsy samples by Kazuhiro Kojima, James K. Chambers, Tatsuhito Ii, Kazumi Nibe, Takuya Mizuno and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by a Grant-in-Aid from the Japan Society for the Promotion of Science (19H03122).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
