Abstract
Increased or constitutive activation of nuclear factor kappa B (NF-kB) is a feature of many chronic disease processes, including cancer. While NF-kB overactivation has been documented extensively in human oncology, there is a relative paucity of data documenting the same phenomenon in veterinary medicine. To assess NF-kB activity, antibodies to p65 and p100/p52, which are components of NF-kB heterodimers, were first validated for specificity and canine cross-reactivity via Western blot and labeling of immortalized cell pellets. Then, nuclear labeling for these antibodies was assessed via QuPath software in over 200 tumor tissue samples (10 hemangiosarcomas, 94 histiocytic sarcomas, 71 lymphomas, and 28 mast cell tumors) and compared to immunolabeling in appropriate normal tissue counterparts. Greater than 70% of spontaneous canine tumors evaluated in this study had more nuclear p65 and p100/p52 immunoreactivity than was observed in comparable normal cell populations. Specifically, 144/204 (70.58%) of tumors evaluated had positive p65 nuclear labeling and 179/195 (91.79%) had positive p100/p52 nuclear labeling. Surprisingly, greater nuclear p100/p52 reactivity was associated with a longer progression-free survival (PFS) and overall survival (OS) in canine lymphomas. These results provide support and preliminary data to investigate the role of NF-kB signaling in different types of canine cancer.
Keywords
Disseminated mast cell neoplasia (MCT), hemangiosarcoma (HSA), and histiocytic sarcoma (HS) have poor prognoses in both humans and dogs, despite medical interventions available for these diseases. These neoplasms and more common diseases, such as lymphoma, share remarkable cross-species similarities.6,7,9,10,20,24,25,28 –31,33,41–43,48,53 Naturally occurring canine cancers can be used as a model to accelerate research for diseases that otherwise receive little attention in humans (so-called “orphan diseases”), and for diseases that are more common, but are biologically and molecularly similar.
Some canine and human cancer literature, as reviewed previously, supports constitutive nuclear factor kappa B (NF-kB) signaling as a therapeutic target in both canine and human tumors. 37 NF-kB proteins are a family of structurally related, eukaryotic transcription factors that have more than 400 genetic targets and are involved in many vital cellular processes, including innate immunity, inflammatory responses, development, cellular growth, and survival. As a transcription factor, NF-kB is typically held as a heterodimer in an inhibited state within the cytoplasm and, following activation, moves into the nucleus to initiate transcription of downstream target genes.11,21,40 Two proteins of interest in the current study are RelA/p65 (canonical pathway) and RelB/p52 (non-canonical or “alternative” pathway). p52 is produced as a precursor protein, p100, and proteasome-mediated selective degradation of its C-terminal region yields mature p52. The canonical NF-kB pathway is critical for immune response, as well as the survival and proliferation of immune cells, while the non-canonical pathway is important in lymphoid development. 8 Nuclear immunolabeling of NF-kB components, expressed as either a nuclear immunolabeling score or as a nuclear to cytoplasmic ratio, has been used previously as a surrogate of pathway activation status.23,26,47
This study evaluates NF-kB immunolabeling in 4 canine tumor types that were selected based on their sensitivity to an NF-kB inhibitor that is under study in our laboratory and, to the extent possible, correlates NF-kB immunolabeling data with existing information about these tumors, including outcome data, mitotic count (MC), and anatomic location.
Materials and Methods
Cell Lines
The canine cancer cell lines utilized in this study were generously provided by researchers at other institutions or purchased from the American Type Culture Collection (ATCC) (Table 1). Cells were maintained in appropriate media for the cell line (RPMI 1640 for lymphoma, leukemia, and mast cell lines, and DMEM—or Dulbecco’s minimum essential medium—for HS and HSA cells). Media was supplemented with 1× minimum essential medium (MEM) vitamin solution (Cellgro, Henderson, Virginia), 2 mM
List of cell lines used for validation of NF-kB pathway assessment.
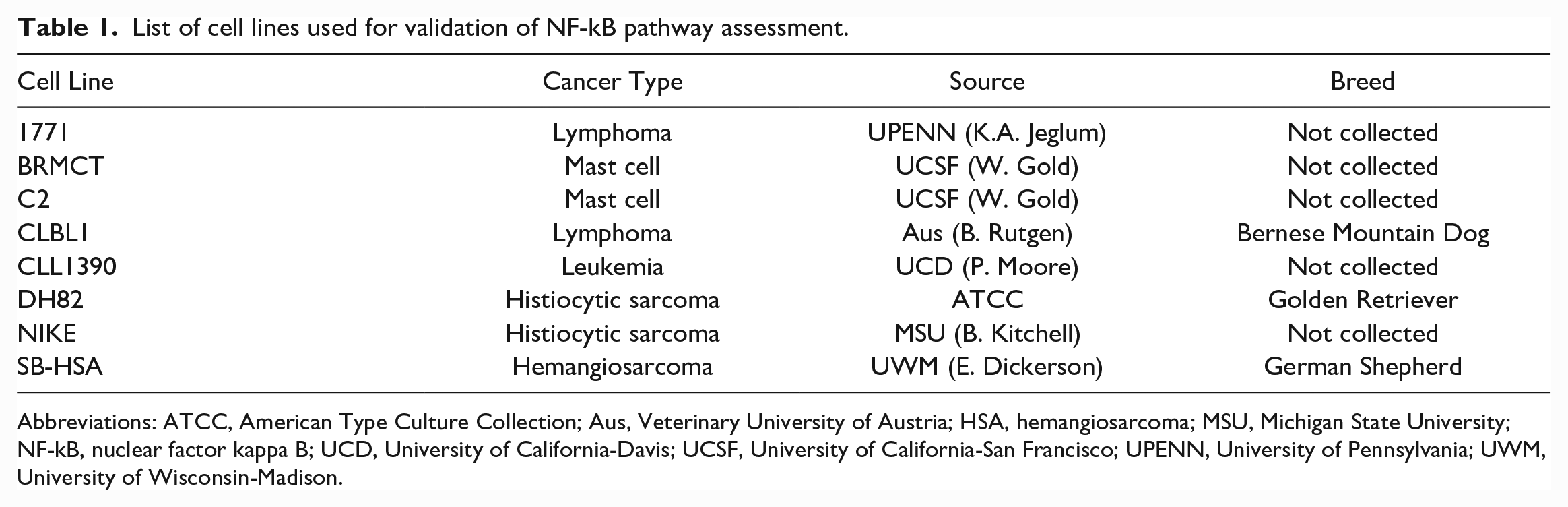
Abbreviations: ATCC, American Type Culture Collection; Aus, Veterinary University of Austria; HSA, hemangiosarcoma; MSU, Michigan State University; NF-kB, nuclear factor kappa B; UCD, University of California-Davis; UCSF, University of California-San Francisco; UPENN, University of Pennsylvania; UWM, University of Wisconsin-Madison.
Treatment and Preparation of Cell Blocks for Immunohistochemistry
Cells were grown to near confluence (~90%) in T175 culture flasks and were treated with DMSO (vehicle control) or 10 µM parthenolide (PTL), a documented canonical NF-kB inhibitor that prevents nuclear translocation of p65 in some cancer cells, 14 for 6 hours. Cells were harvested (via cell scraping or trypsinization, as appropriate) and then washed with phosphate-buffered saline (PBS). Cells were suspended in 10% buffered formalin in a microcentrifuge tube for 15 minutes to ensure fixation. Following fixation, cells were rinsed in PBS, centrifuged, and then resuspended in ~150 µL of 1% agarose gel. Pellets of gel containing cells were pipetted into the caps of fresh microcentrifuge tubes and allowed to cool on ice. Following cooling, gel disks were gently removed from microcentrifuge caps with a #10 blade. Gel disks were paraffin-embedded using standard methods, by the Colorado State University histopathology laboratory.
Antibodies and Immunohistochemistry
Table 2 details antibodies that were used in this study. Although neither of these antibodies have previously been evaluated in dogs, target epitopes have high identity between humans and dogs, and specificity and cross-reactivity were initially supported via Western blot.
Antibodies used for immunohistochemistry and Western blotting.
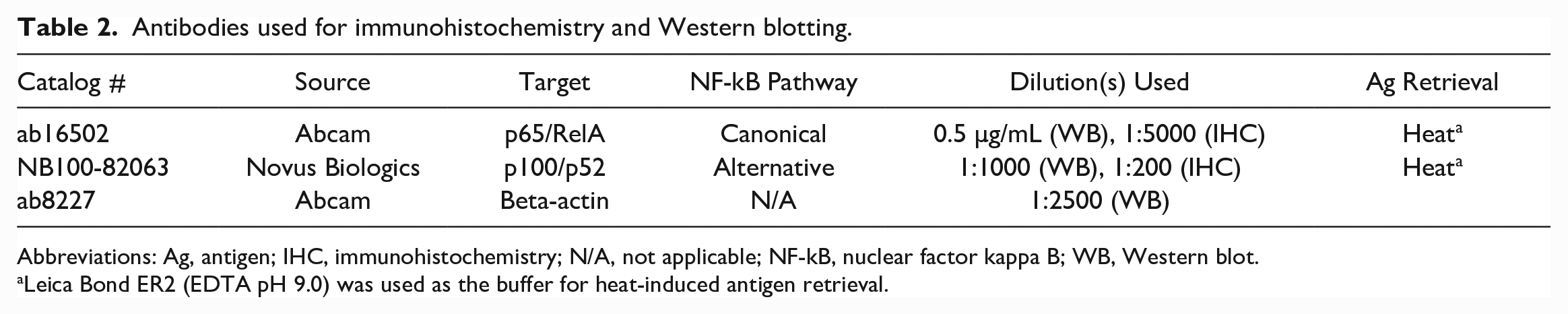
Abbreviations: Ag, antigen; IHC, immunohistochemistry; N/A, not applicable; NF-kB, nuclear factor kappa B; WB, Western blot.
Leica Bond ER2 (EDTA pH 9.0) was used as the buffer for heat-induced antigen retrieval.
Tissue blocks and paraffin-embedded cell pellets were sectioned at 5 µm, then mounted on Superfrost Plus slides (Fisher Scientific, Hampton, New Hampshire), and immunolabeling for p65 and p100/p52 was performed using standard methods. Briefly, tissue slides were de-paraffinized in xylenes and re-hydrated with a series of graded alcohols. Antigen retrieval was performed, as outlined in Table 2. Tissues were blocked for endogenous peroxidase by incubation in 3% hydrogen peroxide for 5 minutes. Sections were incubated with primary antibodies, as outlined in Table 2. Detection was performed using the universally labeled streptavidin-biotin2 system (Dako, Carpinteria, California), which uses a mixture of biotinylated goat anti-mouse and anti-rabbit IgG secondary antibodies followed by horseradish peroxidase (HRP)-labeled streptavidin. A universal negative control was used for each antibody evaluated. Positive labeling was visualized with DAB (3,3′-diaminobenzidine) chromogen substrates. Toluidine blue staining was performed to highlight metachromatic granules of mast cells in normal tissue controls. 17
Western Blot Analysis
When antibodies were being evaluated for use in canine cells and tissues, hematopoietic cell lines were initially evaluated, as these were believed to be potentially susceptible to the NF-kB inhibitor PTL. Cells were plated at ~75% confluency in T25 flasks with C/10 media 24 hours prior to treatment. The following day, cells were washed with PBS, collected either via cell scraping, when needed (1771, CLBL1, CLL1390, Br, C2 cells) or trypsinization (DH82, NIKE, SB-HSA cells), as appropriate. For DH82, NIKE, and SB-HSA, cells were washed with PBS and then exposed to 1 mL trypsin for 5 minutes to detach them from the culture plate. Two milliliters of C/10 media was added to neutralize trypsin. Collected cells were centrifuged at 1400 rpm for 5 minutes. The trypsin/media supernatant was removed from pelleted cells, and cell pellets were resuspended in 1 mL PBS in a 1.5-mL microcentrifuge tube. Cells were then centrifuged at 10,000 rpm at 4°C for 5 minutes. After discarding the PBS supernatant, cells were homogenized with 150 µL of buffer containing M-PER protein extraction reagent (Pierce), 1 mM NaVO4, 1 mM PMSF, Complete Mini Protease Inhibitor (Roche), and 1% SDS. The cell homogenate was placed on ice for 10 minutes, and lysates were passed through a 25-gauge needle 5 times. Following centrifugation at 10 000 rpm at 4°C for 5 minutes, the supernatant was used to measure cellular protein concentrations using the BCA protein kit (Bicinchoninic Acid Kit, Pierce, Rockford, Illinois). For concurrent evaluation of NF-kB p65 and B-actin, 25–30 µg samples of cells were electrophoresed into a denaturing 4% to 12% Bis-Tris gel (Invitrogen, Carlsbad, California) and were transferred electrophoretically to a polyvinylidene difluoride (PVDF) membrane. Membranes were cut directly below the 50 kDa marker (Precision Plus Protein Kaleidoscope Prestained Protein Standard, BioRad Laboratories, Hercules, California) and then blocked with SuperBlock (PBS) Blocking Buffer (Thermo Fisher Scientific, Waltham, Massachusetts) for 1 hour at room temperature, followed by incubation of primary antibody (as indicated in Table 2) overnight on a rocker, in blocking solution at 4°C. After 3 washes in tris-buffered saline with 0.1% Tween, membranes were incubated in a 1:20 000 dilution of HRP-conjugated goat anti-rabbit IgG for 1 hour at room temperature. Immunoreactive proteins were detected using SuperSignal West Pico Chemiluminescent Substrate (Pierce) and was analyzed via a ChemiDoc XES+ System (Bio-Rad Laboratories). In between evaluation for p65 and p100/p52, blots were stripped for 10 minutes on a rocker at room temperature, with Restore Stripping Buffer, followed by 3 washes in tris-buffered saline with 0.1% Tween. The protocol for primary antibody labeling was repeated with the p100/p52 antibody. Relative band intensity was measured in ImageLab computer software (Bio-Rad Laboratories).
Case Selection and Preparation of Tissue Microarrays
The Colorado State University (CSU) Veterinary Diagnostic Laboratory database from 2017 to 2021 was searched to identify formalin-fixed, paraffin-embedded (FFPE) tissue samples of HS and MCT cases. All available HS cases (n = 82) were included, which included cases from a variety of anatomical locations and breeds of dogs. Inclusion of the MCT cases (n = 30) required accompanying pre-existing data, including mitotic count (MC), Ki67 positivity, Patnaik and Kuipel grades (cutaneous tumors), KIT immunostaining pattern, and c-KIT mutation status. Cases with these available data were from various anatomic locations (cutaneous [n = 22] and non-cutaneous tumors [n = 6]), and the cutaneous MCTs were of various histologic grades (Grade I/Low [n = 2], Grade II/Low [n = 5], Grade II/High [n = 8], and Grade III/High [n = 7]). Lymphoma and splenic HSA tissue blocks were available from previous patient study populations, comprising 50 cases of lymphoma and 10 cases of splenic HSA.44,51 Supplemental Table S1 contains demographic data from patients. Details for the MCT cases are given in Supplemental Table S2. All parameters in the MCT cases were previously assessed by a board-certified CSU anatomic pathologist.
Twelve normal lymphoid tissues (spleen [n = 6] and lymph node [n = 6]) retrieved from the CSU Veterinary Diagnostic Laboratory database and 20 skin sections prospectively collected from dogs presented to the necropsy service were used to assess immunolabeling of normal cells. For comparison with the lymphomas, lymphocytes from germinal centers, mantle zones, and interfollicular regions were used, excluding cells that were larger and of non-lymphoid origin (histiocytic cells and mast cells) or were associated with stromal elements or vessels. As most HS are derived from antigen-presenting cell (APC) dendritic cells, normal dendritic cells were identified in these lymphoid tissues based on their location in the “light” staining area of the germinal centers, as well as cellular morphology and used as normal comparators for HS.13,52 For HSA, normal endothelial cells from the aforementioned normal spleen and lymph node tissues were used as controls. Normal mast cells were identified in lymphoid tissues, as well as perivascularly in the normal canine skin sections (32 tissues overall, 12 lymphoid tissues and 20 skin sections). At least 500 normal cells from all tissue types were evaluated, except for mast cells, in which 200 normal cells were confidently identified in the immunolabeled sections.
Tissue microarrays were prepared using EZ-TMA 120-well, 1.5 mm, recipient blocks (IHC World, Woodstock, Maryland). Tumor cores were taken with a 1.5 mm biopsy punch (one per tissue block) and were gently extruded with a metal stylette. Paraffin embedding was performed by the Colorado State University Veterinary Diagnostic Laboratory, as per recipient block manufacturer instructions.
Quantitative Grading of IHC Labeling
Immunohistochemical grading was performed using a quantitative scoring scheme for nuclear and cytoplasmic labeling with QuPath software. The nuclear and cytoplasmic scoring for NF-kB immunoreactivity were performed on tumor cells from tissue microarrays, using the total optical density for detection and the mean DAB optical density (nuclear or cytoplasmic) to identify positive cells. 4 Three thresholds for cell positivity (0.2, 0.4, 0.6) were used to classify cells as having no (blue), mild (yellow), moderate (orange), or strong (red) DAB labeling. Examples of the different levels of nuclear and cytoplasmic labeling of tumor cores are shown in Fig. 1. The H-score, as calculated by the software, is a numerical output of these data, derived from the following equation:
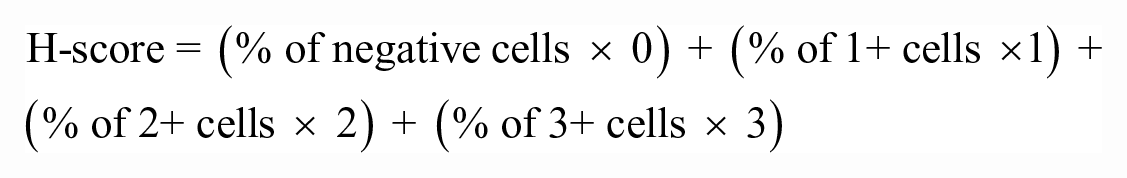
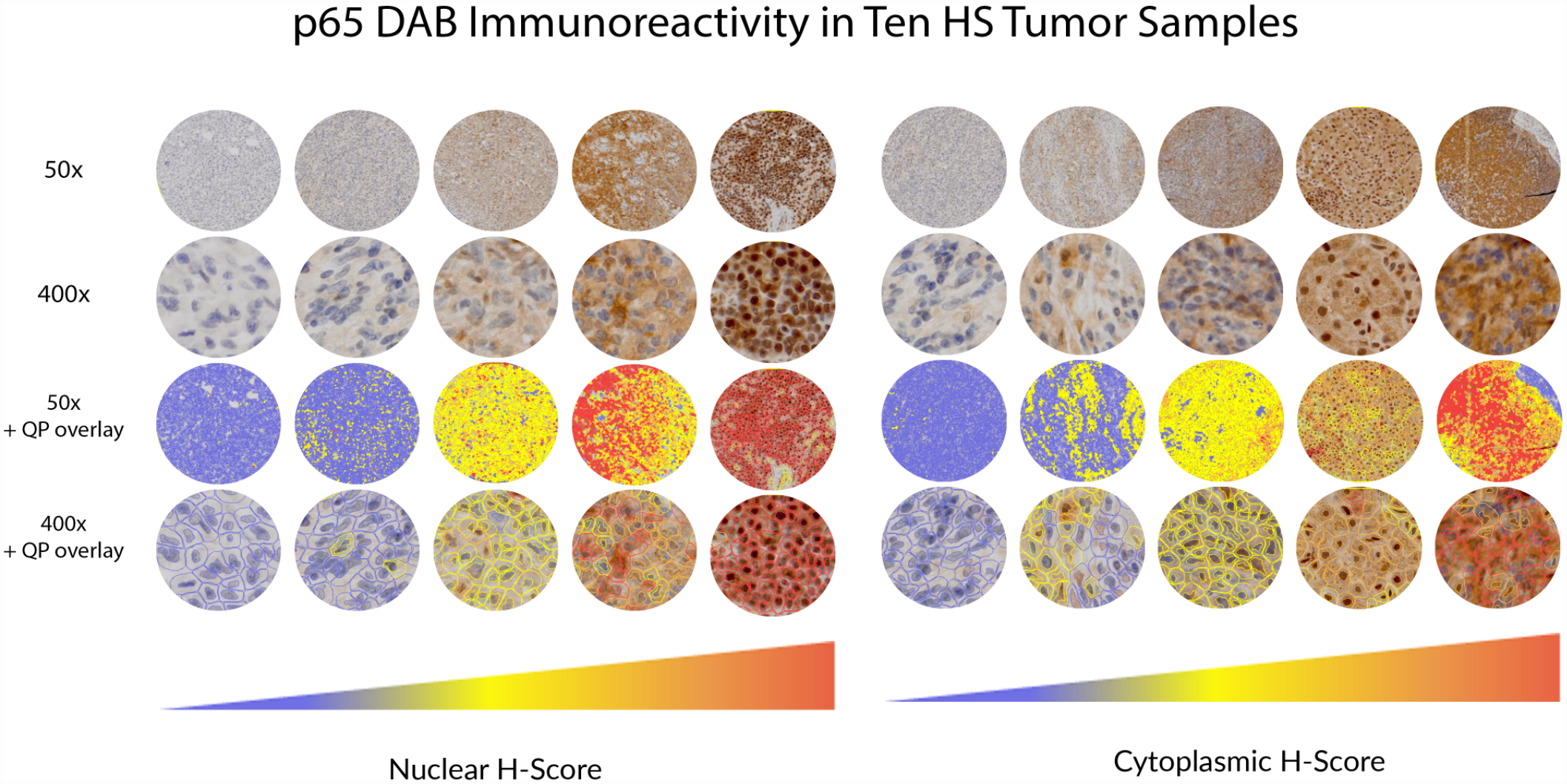
Neoplastic tissues graded by QuPath (QP) software, showing examples of HS (histiocytic sarcoma) tumor scores with different levels of nuclear and cytoplasmic p65 labeling. The H-score is a weighted average of all cells graded following manual exclusion of tissue folds, areas of necrosis, and non-neoplastic cells. The nuclear H-score and nuclear to cytoplasmic H-score ratio were used as surrogates of nuclear factor kappa B (NF-kB) activity.
To be eligible for inclusion, each microarray sample had to contain at least 500 cells for grading with the software, without significant crush artifact or necrosis in the area evaluated. The number of tumor samples available for evaluation varied due to loss of some tissue cores during embedding.
Areas of tissue folding and those with non-neoplastic cells were manually excluded prior to grading. If the nuclear H-score for NF-kB labeling exceeded the maximum established by evaluation of normal control cells for that tumor type (Figs. 4, 5), the tumor core was graded as “positive.” The nuclear H-score was divided by the cytoplasmic H-score to determine the nuclear to cytoplasmic (N:C) ratio for each tumor sample; the nuclear H-score and N:C ratio were evaluated independently in statistical calculations, as both nuclear scoring and the N:C ratio have been used as surrogates for NF-kB activity in other studies.
Correlation With Clinicopathologic Data
The relative immunolabeling intensities of tumors for NF-kB p65 and p100/p52 H-scores, as well as calculated N:C ratios, were compared with factors that may influence a tumor’s biologic behavior, either in theory or as previously demonstrated in the literature or by patient outcomes in our data sets.
To create the lymphoma microarrays, tumor cores were taken from 50 dogs with naïve multicentric T-cell or B-cell lymphoma that were subsequently treated with a standardized, 19-week, CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) chemotherapy protocol, and outcome data were recorded for these patients. 51 HS and HSA samples were taken from diagnostic laboratory biopsy and necropsy samples, and thus, follow-up data were largely unavailable. When available, IHC grading was evaluated with respect to the anatomic location of the tumor and/or the mitotic count. Outcome data were similarly not available with respect to the MCTs used in this study; however, as previously noted, there was access to KIT IHC localization data, Kuipel and Patnaik grades (for cutaneous tumors), MC (number of mitoses in 10 high-power, 40× objective fields, or a total area of 2.37 mm2), and anatomic location of the tumor.5,22,35 In addition, c-KIT mutation status, assessed as previously described, was available for all tumors. 50
Statistical Analyses
Correlations between continuous variables (MC and nuclear p65 and p100/p52 scores, as well as nuclear to cytoplasmic score ratios) were computed by means of the Spearman rank correlation coefficient. The Kruskal-Wallis nonparametric analysis of variance was used to determine the significance levels of differences between groups of continuous variables after stratification on categorical variables (such as tumor grade or anatomical site of origin). When 2 groups of continuous variables were used after stratification on categorical variables, a 2-tailed, Mann-Whitney nonparametric test was used. Pre- and post-CHOP treatment immunoreactivity was compared in lymphoma cores using a 2-tailed, paired Mann-Whitney test. The Kaplan-Meier method was used to estimate progression-free interval (PFI) and overall survival time (OST). Impact of immunoreactivity score and ratio on outcome (PFI and OST) were assessed using log-rank and Cox proportional hazards methods. Statistical analyses were performed using SPSS v28.0.1.0 (IBM, Armonk, New York) and Prism v9.0 (GraphPad, San Diego, California).
Results
Western Blot Analysis
Western blots using the antibodies employed in this study are shown in Fig. 2. Based on this initial screening experiment, all hematopoietic cell lines evaluated had evidence of specific p65 protein expression, and most had evidence of p100 and p52 expression, to varying degrees.
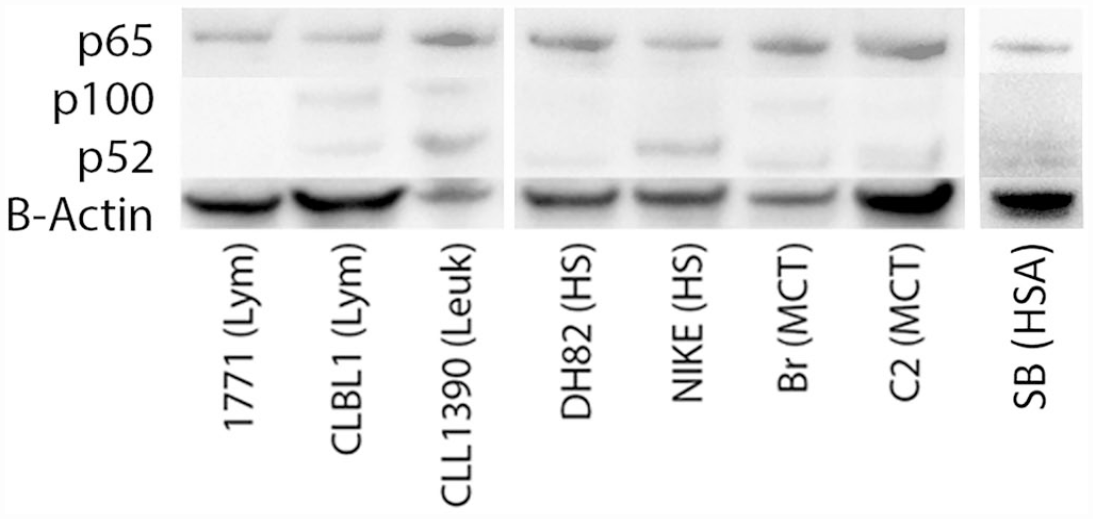
Western blot evaluating nuclear factor kappa B (NF-kB) antibodies for specificity in multiple neoplastic cell lines of hematopoietic origin in dogs. Specific bands were observed at molecular weights of 65, 100, and ~52 kDa, consistent with the molecular weights of NF-kB p65, p100, and p52, respectively. β-actin was used as a loading control. Lym, lymphoma; Leuk, leukemia; HS, histiocytic sarcoma; MCT, mast cell tumor; HSA, hemangiosarcoma.
Cell Pellet Immunohistochemistry
Cell pellets from various canine cell lines (as shown in Fig. 3) were evaluated for expression of NF-kB RelA/p65 and p100/p52 via IHC. To further validate the p65 antibody for use in dogs, we utilized PTL, a drug that inhibits NF-kB activity by interfering with p65-DNA binding and to a lesser extent by inhibiting nuclear translocation of p65.12,14,27 There was a subtle decrease in nuclear p65 immunolabeling intensity with PTL treatment in DH82 (HS) cells and a potentially slight difference in SB (HSA) cells. Measuring this difference in nuclear labeling intensity with QuPath software, the p65 N:C ratio in untreated cells was 1.0 for DH82 HS cells and 1.11 for SB HSA cells. This ratio decreased, following PTL treatment, to 0.66 for DH82 HS cells and 0.88 for SB HSA cells. This difference in nuclear localization is also present in a pilot immunocytochemistry experiment in DH82 HS cells that was presented in previous work.36,37 There was not a detectable difference in pre- and post-treatment p65 nuclear labeling intensity in the other cell lines. All cell lines evaluated appeared to have strong nuclear labeling localization of p100/p52. IHC pellets from cells treated with PTL appeared similar to vehicle control cells.
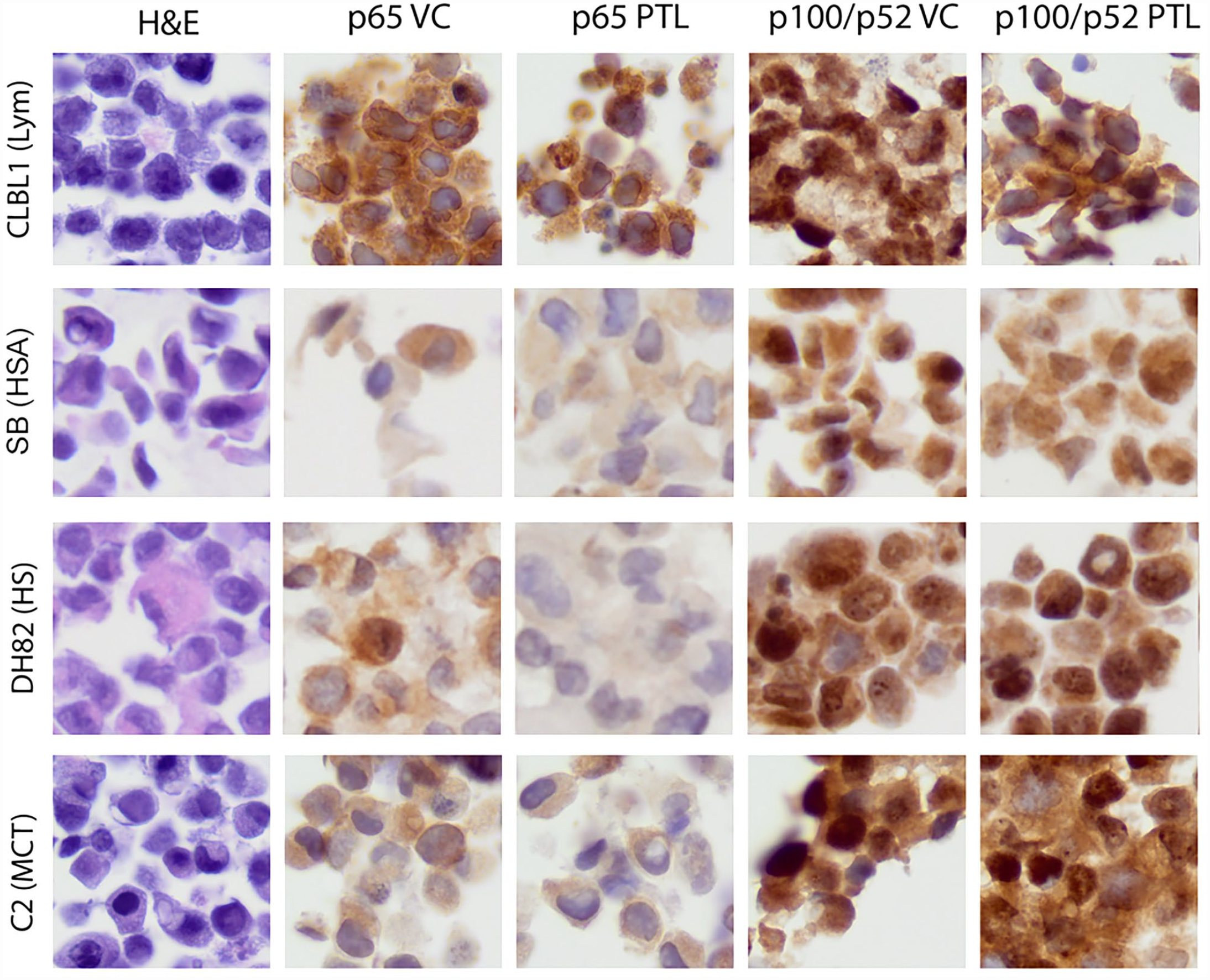
Hematoxylin and eosin (left-most panel) and p65 and p52 immunohistochemistry (remaining panels) of canine cell line cell pellets treated with vehicle control (VC) or the nuclear factor kappa B (NF-kB) inhibitor parthenolide (PTL). Inhibition of nuclear translocation of the canonical NF-kB signaling molecule p65 is subtly evident in DH82 (histiocytic sarcoma, HS) and possibly in SB (hemangiosarcoma, HSA) cells. As quantified by QuPath software, minor changes are measurable. Labeling for non-canonical NF-kB signaling members (p100/p52) appears similar between VC- and PTL-treated cells, as expected (as PTL is only documented to inhibit the canonical pathway).
IHC Grading of Tissue Microarrays
Examples of normal tissue labeling with NF-kB p65 and p100/52 antibodies are presented in the left-hand columns of Figs. 4 and 5. The middle and right-hand columns of Figs. 4 and 5 show examples of tumor tissues that had relatively low and high nuclear H-scores, as computed by QuPath software. For each tumor sample, 50× and 200× images (top and bottom) are provided. An unlabeled image and images labeled for nuclear and cytoplasmic scoring by QuPath are included (left, middle, right). Figure 6 shows the numbers of tumors that were ultimately classified as having positive nuclear p65 or p100/p52 labeling. Relative to control cells, many tumor cell types exhibited increased nuclear labeling for both p65 and p100/p52.
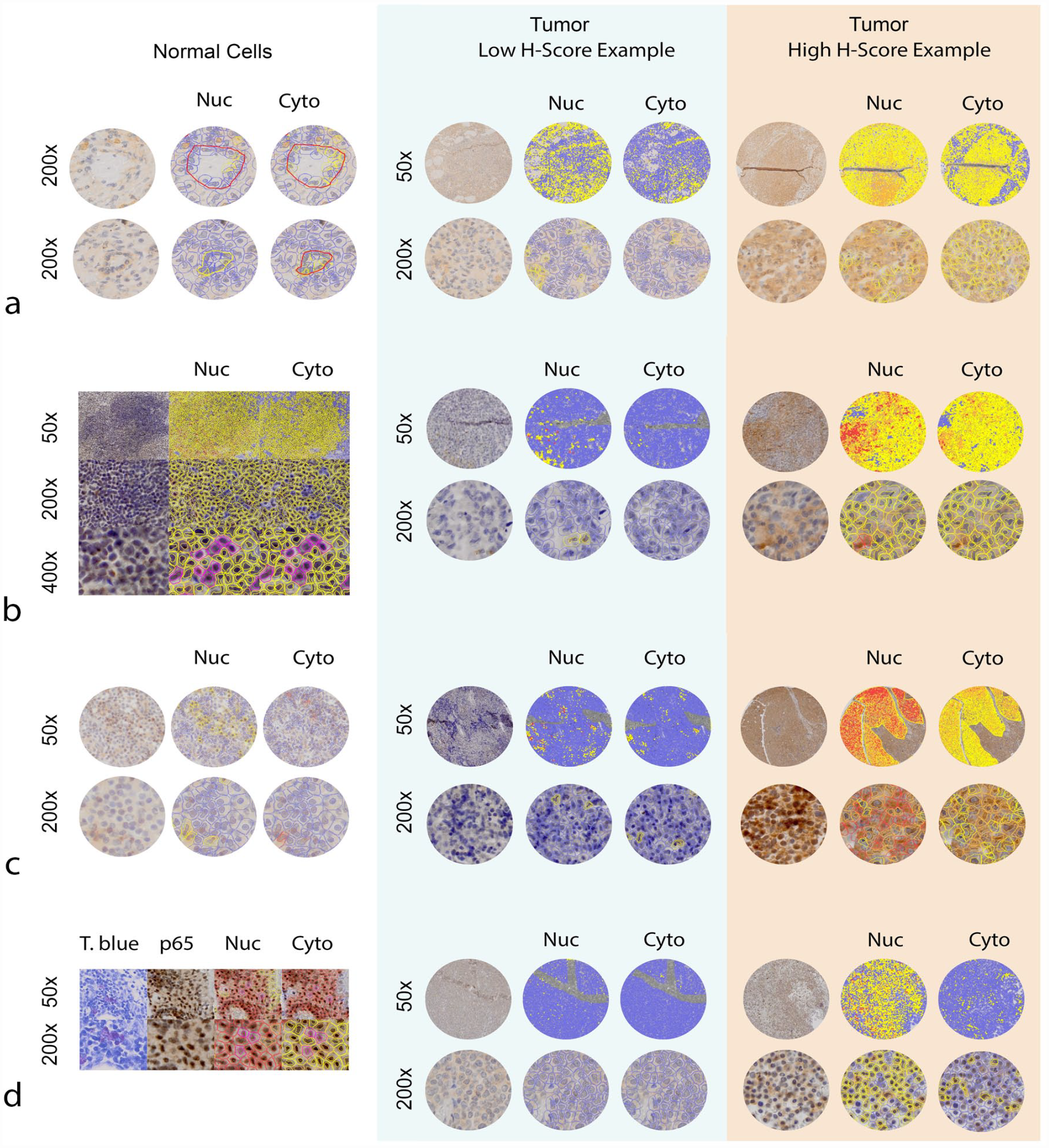
Examples of normal p65 tissue immunolabeling and application of the nuclear and cytoplasmic grading system by QuPath software. The left panel shows representative labeling of p65 in normal tissues, while the middle and right panels show examples of low and more strongly positive nuclear labeling in neoplastic tissues. (a) Left: Cells within the line are representative of those that were manually gated and counted as normal endothelial cells across normal lymphoid tissue samples (2 examples are shown). Middle and right: Hemangiosarcoma samples. (b) Normal lymph node germinal center at 3 magnifications. Cells that have been outlined with pink were counted as probable dendritic cells because they appeared in the light staining area of the germinal center, contained large, round to reniform nuclei, and had more abundant cytoplasm, as compared to surrounding lymphocytes (shown with and without pink outlines). Middle and right: Histiocytic sarcoma samples. (c) Left: Normal lymph node. Middle and right: Lymphoma samples. (d) Left: Normal mast cells were identified in non-neoplastic lymph nodes (example shown) and skin. Following confirmation of mast cells within normal tissues with toluidine blue stain, immunolabeled tissue samples were evaluated in the same regions in which mast cells had been observed. Cells with a low nuclear to cytoplasmic ratio and abundant cytoplasm with a slightly granular character were counted as probable mast cells (shown with and without pink outlines). Note that in the sample tissue shown here, which contains moderate to large numbers of mast cells, there is greater immunolabeling of p65 in neighboring lymphocytes and endothelial cells, consistent with antigenic stimulation in this tissue. Middle and right: mast cell tumor samples. Nuc, nuclear H-score; Cyto, cytoplasmic H-score; T blue, toluidine blue.

Examples of normal p100/p52 tissue immunolabeling and application of the nuclear and cytoplasmic grading system by QuPath software. The left panel shows representative labeling of p100/p52 in normal tissues, while the middle and right panels show examples of low and more strongly positive nuclear labeling in neoplastic tissues. (a) Left: Cells within the line are representative of those that were manually gated and counted as normal endothelial cells across normal lymphoid tissue samples (2 examples are shown). Middle and right: Hemangiosarcoma samples. (b) Normal lymph node germinal center at 3 magnifications. Cells that have been outlined with pink were counted as probable dendritic cells because they appeared in the light staining area of the germinal center, contained large, round to reniform nuclei, and had more abundant cytoplasm, as compared with surrounding lymphocytes (shown with and without pink outlines). Middle and right: Histiocytic sarcoma samples. (c) Left: Normal lymph node. Middle and right: Lymphoma samples. (d) Left: Normal mast cells were identified in non-neoplastic lymph nodes (example shown) and skin. Following confirmation of mast cells within normal tissues with toluidine blue stain, immunolabeled tissue samples were evaluated in the same regions in which mast cells had been observed. Cells with a low nuclear to cytoplasmic ratio and abundant cytoplasm with a slightly granular character were counted as probable mast cells (shown with and without pink outlines). Middle and right: mast cell tumor samples. Nuc, nuclear H-score; Cyto, cytoplasmic H-score; T blue, toluidine blue.
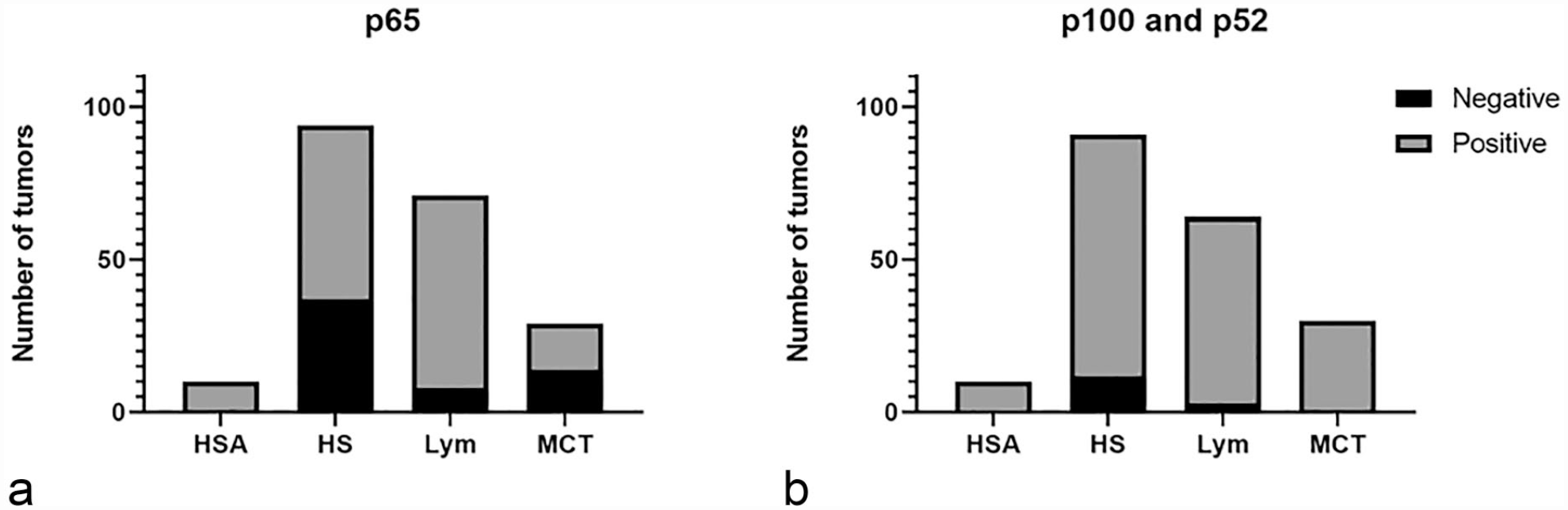
Graphical representation of tumor positivity for (a) p65 and (b) p100/p52. HSA, hemangiosarcoma; HS, histiocytic sarcoma; Lym, lymphoma; MCT, mast cell tumor.
NF-kB Expression and Correlation With Clinicopathologic Data
Lymphoma
50/86 lymphoma cases had outcome data available. Inadequate numbers of T-cell lymphoma cases (n = 10) were available to make statistical comparisons between NF-kB scoring metrics (nuclear H-score and N:C ratio) and survival data. There was no difference in nuclear H-score or the N:C ratio in p65 or p100/p52 for pre- vs post-treatment lymphoma patients in this study. There was no significant correlation between nuclear H-score or the N:C ratio and survival metrics (PFS, OST). For B-cell lymphoma cases with outcome data (n = 40), there were significant correlations between a higher p100/p52 nuclear H-score, cytoplasmic H-score, and the N:C ratio and increased progression-free survival and overall survival. The hazard rate (HR) is an estimate of the ratio of the hazard rate in patients with a greater nuclear or cytoplasmic H-score, for each metric.
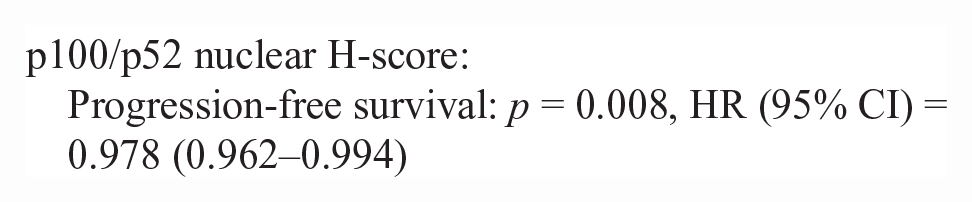
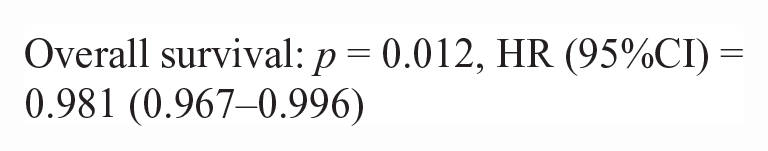
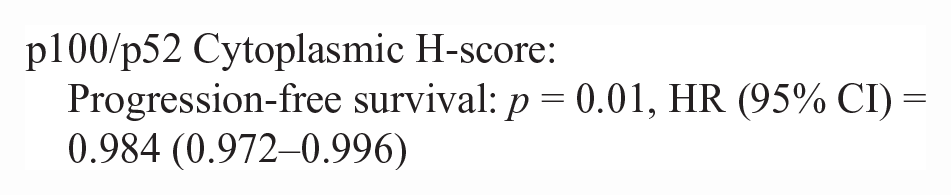
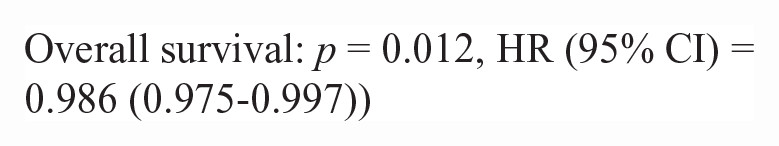
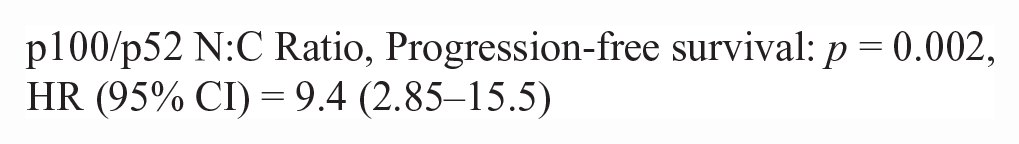
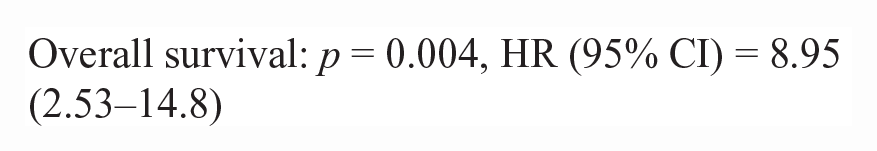
Histiocytic sarcoma
There was no statistically significant difference in the MC that could be predicted by a tumor’s anatomical site of origin in this data set. There was no significant correlation (Spearman’s r) between a tumor’s MC and nuclear p65 or p100/p52 labeling intensity, nor between the MC and the N:C labeling ratio.
Mast cell tumor
There was no statistically significant difference in the MC that could be predicted by a tumor’s anatomical site of origin in this data set (22 cutaneous, 6 visceral). There was no significant correlation (Spearman’s r) between a tumor’s MC and the nuclear p65 or p100/p52 labeling intensity, nor between the MC and the N:C labeling ratio.
There were no statistically significant correlations between cutaneous tumor grade and NF-kB immunolabeling identified, although there was a trend toward a greater N:C p65 ratio with higher Kuipel grade tumors (Fig. 7).
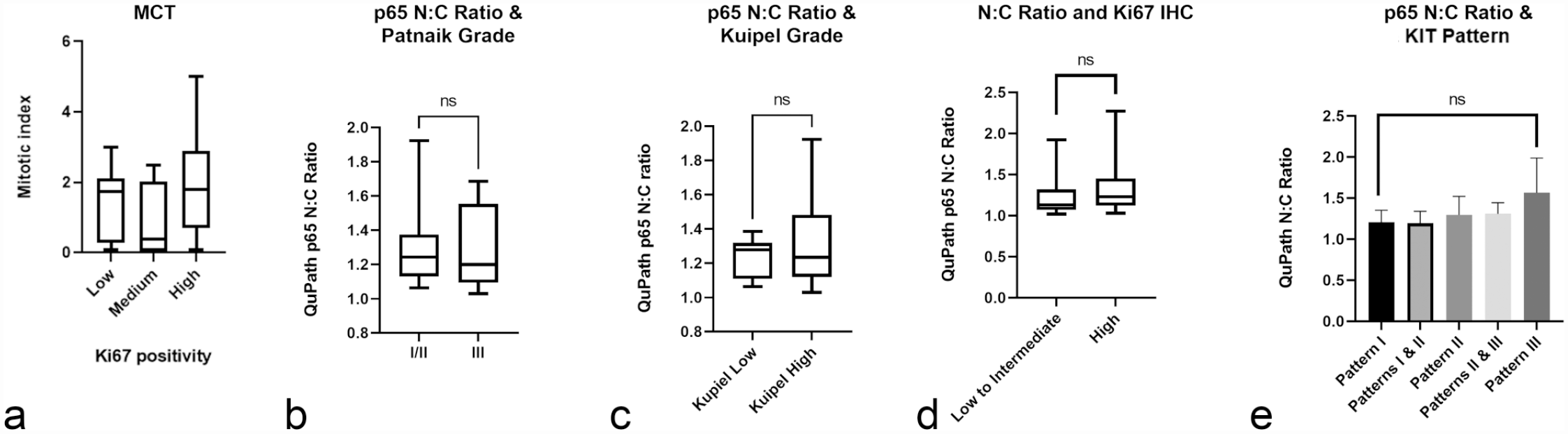
Canine mast cell tumor (MCT) summary data. Selected data show (a) trend toward increased mitotic count with Ki67 positivity, (b) no apparent association between p65 nuclear to cytoplasmic ratio (N:C) and Patnaik grade, (c) a trend toward a higher p65 N:C ratio with Kuipel grade, (d) a trend toward higher p65 N:C ratio with Ki67 positivity, and (e) a trend toward tumors with a higher p65 N:C ratio exhibiting KIT pattern III immunolabeling.
Although not statistically significant, there was a trend toward higher Ki67 positivity with increased MC, as expected (Fig. 7). A statistically significant correlation was not observed between Ki67 positivity and p65 or p100/p52 nuclear labeling in this study. There was, however, a trend toward tumors with a greater p65 N:C ratio having greater Ki67 positivity (Fig. 7).
A statistically significant correlation was not identified between c-KIT mutation status and NF-kB p65 or p100/p52 IHC labeling intensity; however, only a small number of tumors (n = 5) in this study had detectable mutations. No statistically significant associations were identified between KIT immunolabeling pattern and NF-kB p65 or p100/p52 IHC labeling results, although again, there was a trend toward tumors with a higher p65 N:C ratio having KIT staining pattern III (Fig. 7).
Hemangiosarcoma
Most splenic HSAs had greater IHC labeling intensity of both p65 and p100/p52, as compared to normal endothelial cells. There was no significant correlation (Spearman’s r) between a tumor’s MC and nuclear p65 or p100/p52 labeling intensity, nor between the MC and the N:C labeling ratio.
Discussion
Overall, findings from this study support altered NF-kB signaling as a common finding in a variety of tumor types, and importantly, in types of cancer that are overrepresented in some dog breeds but are considered orphan diseases in humans. Data from this study could be used to support further study of canonical and alternative NF-kB signaling in these tumors in both species.
The lymphoma data for p100/p52 immunolabeling are difficult to interpret. When considered in isolation, it is tempting to view the p100/p52 N:C labeling ratio as a potential predictor of PFS and OS in dogs with B-cell lymphoma. In fact, previous literature has shown that constitutive activation of the alternative NF-kB signaling pathway occurs in dogs with diffuse large B-cell lymphoma (DLBCL) and contributes to neoplastic cell survival. 39 However, this may also be a statistical anomaly, as most samples had both strong nuclear and cytoplasmic reactivity, the ratio is less meaningful. According to this data set, increased p100/p52 nuclear and cytoplasmic labeling is associated with improved outcomes. Additional study is warranted to further explore this phenomenon.
Although lymphoma is a heterogeneous disease with variable biologic behavior, inadequate numbers of tumors were available to assess NF-kB p65 and p100/p52 labeling with respect to histologic subtype. In addition, inadequate sample numbers were available to assess labeling differences with respect to T-cell versus B-cell immunophenotype. Ideally, data from this preliminary study could be used to further evaluate the NF-kB activation status of a larger number of lymphomas for which additional data are available for correlation (immunophenotype, histologic subtype, and outcome).
Cutaneous MCTs are one of the most common neoplasms to affect dogs, but their biologic behavior is quite variable, and various grading schemes and molecular markers have been studied to attempt higher precision in prognostication and optimization of treatment regimens. 49 One substantial challenge in histologic grading of MCTs is that in the Patnaik system, more than 40% of tumors are classified as being of “intermediate” grade, yet these tumors demonstrate significantly variable biologic behavior, and problematically, there is significant pathologist interobserver variability in this grading scheme.22,29 Many of the studies evaluating additional biologic markers for MCTs, including proliferative indices, have been aimed at overcoming the limitations that are evident in existing histologic grading schemes.
The KIT protein is a tyrosine kinase receptor and a product of the c-KIT proto-oncogene. Of many potential markers, KIT has been consistently identified as a key player in MCT development, and its constitutive activation is associated with receptor tyrosine kinase-mediated cellular survival, proliferation, and motility via multiple downstream pathways, including the RAS/mitogen-activated protein kinase pathway, JAK-STAT, PI3-kinase, and Src family of kinases (SFK) pathways. 15 Because these pathways are downstream of KIT signaling, we had hoped to see more evidence of p65 nuclear labeling in MCTs overall.1,3,19,34,38,46 The significance of this seeming discrepancy is unknown at the time of writing. As this was a relatively small data set, perhaps the data are not fully representative of p65 activation status. In addition, there are other mechanisms of KIT activation aside from exon 8 and 11 mutations (additional mutations that were not assayed for, and for which the biological significance is currently unknown; paracrine or autocrine production of stem cell factor). Evaluation of a direct measure of KIT activation, such as KIT phosphorylation status, may also correlate with NF-kB activation status, for example.2,18,45 Alternatively, this seemingly discordant finding could be real, as MCT cell lines are relatively more resistant to the NF-kB inhibitor PTL, as compared to leukemia, lymphoma, and HS cell lines, in our experience (manuscript in preparation). 36 Ideally, larger numbers of MCTs would be assessed to make this determination with more confidence.
For HS, HSA, and MCT, this study suffers from the usual problems affecting studies that utilize biopsy and necropsy specimens, which include variable time of formalin fixation prior to embedding (which can, in theory, negatively affect nuclear IHC labeling properties of cells 16 ), incomplete information regarding disease presentation (for biopsy specimens), and lack of outcome data. In this work, 10 cell pellet samples that were placed in formalin for an extended time (7 days) did not demonstrate noticeably different IHC labeling properties as compared to samples that were fixed for 24 hours prior to embedding. In addition, although we cannot know the time from biopsy collection to embedding, as many of these tissues were submitted from clinicians outside of the university, there was not a noticeable difference in labeling intensities between samples obtained via necropsy or biopsy.
The use of tissue microarrays introduces inherent bias into any study that uses them, as small pieces of tissue may not be fully representative of the sampled tissue or have patchy distribution of a marker of interest. This is an inherent limitation of histopathology generally, as biopsy samples or tissues trimmed from whole organs may not be fully representative. However, tissue microarrays allow researchers to assess immunostaining across many tumors in the same immunohistochemical procedure, which minimizes error in comparing results. Supplemental Figs. S1–S4 show patterns of generally uniform immunostaining with the antibodies used in this study, using large tissue sections of the tumor types assessed. These data indicate that the results derived from microarrays are likely truly representative of the tumors evaluated in this study. In addition, this high-throughput method reduces the cost and materials used in traditional immunostaining, and materials from this study can be used for others in the future.
Overall, data from this study demonstrate that increased NF-kB p65 and p100/p52 nuclear labeling (representative of both the canonical and alternative signaling pathways) is present in a wide variety of canine tumor types and may be a viable therapeutic target. Additional studies are needed to understand the biological implications of these signaling abnormalities, as well as the genetic bases of mutations or non-mutational processes that may give rise to NF-kB signaling in these tumors. Such studies may ultimately provide necessary data for improving our understanding of tumor biology, as well as an improved understanding of therapeutics that may be useful for these tumors.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231180484 – Supplemental material for Immunohistochemical evidence of NF-kB activation in canine lymphomas, histiocytic sarcomas, hemangiosarcomas, and mast cell tumors
Supplemental material, sj-pdf-1-vet-10.1177_03009858231180484 for Immunohistochemical evidence of NF-kB activation in canine lymphomas, histiocytic sarcomas, hemangiosarcomas, and mast cell tumors by Lisa J. Schlein and Douglas H. Thamm in Veterinary Pathology
Footnotes
Acknowledgements
We gratefully acknowledge the CSU histopathology laboratory, particularly Todd Bass and Laura Ashton, for their assistance with immunolabeling, and Drs Travis Meuten and Daniel Regan for assistance in review of IHC grading methodology.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to thank the Morris Animal Foundation (fellowship training grant D20CA-402), the CSU Cancer Biology Scholars Program (Morris Animal Foundation), and National Institutes of Health (grant number 1TL1TR002533-01) for funding.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
