Abstract
Mast cell tumors (MCTs) of gastrointestinal origin that had been surgically removed from 39 dogs were examined to evaluate their pathologic features. Miniature breeds, especially Maltese, were most frequently affected. The average age of affected dogs was 9.7 ± 2.6 years. No sex difference was apparent. The most frequently affected sites were in the upper digestive tract, and the prognosis was very poor. Grossly, the gastrointestinal wall was prominently thickened, and the lumen of the affected gut was usually narrowed. Microscopically, there was diffuse transmural invasion of round to pleomorphic tumor cells. Tumor cells had moderate to abundant cytoplasm, round to ovoid nuclei with scattered chromatin, and mitotic figures. Fibrous stroma was observed in about half of the tumors. There was variable infiltration of eosinophils. In all tumors, cytoplasmic granules showed weak metachromasia, but the number of granules was very small. Immunohistochemical staining for c-kit and mast cell tryptase was positive in 77% and 62% of tumors, respectively. All tumors were positive for at least two of these markers. Immunohistochemical staining for p53 was positive in 13% of the tumors. Reactivity for staining markers and p53 was unrelated to cell pleomorphism, vessel invasion, or survival time. Gastrointestinal MCTs have histologic and immunohistochemical features completely different from those of other primary or metastatic gastrointestinal tumors. The combination of immunostaining for mast cell tryptase and c-kit and histochemical staining for metachromasia appears to be a powerful tool for the diagnosis of gastrointestinal MCTs.
Keywords
Cutaneous mast cell tumors (MCTs) are very common neoplasms; however, MCTs of gastrointestinal origin are less common, and their pathologic features remain unclear.4,12,17 Malignant lymphoma is the most common gastrointestinal tumor of hematopoietic cell origin in dogs. Recently, we encountered tumor masses consisting of small round individual cells in miniature breed dogs, especially Maltese. The majority of these cells had moderate to abundant cytoplasm with round to ovoid nuclei similar to those of some types of lymphoma cells. A small number of these tumor cells contained sparsely scattered granules showing metachromasia with toluidine blue stain. Therefore, these tumors were thought to be most appropriately diagnosed as gastrointestinal MCTs. However, a definitive diagnosis was sought because only a small number of cells in some tumors contained metachromatic granules.
The objective of this study was to characterize the clinical and pathologic features of this MCTs and to establish criteria for definitive diagnosis. We employed evaluation of metachromasia, immunostaining for c-kit and mast cell tryptase,1,3,13 and electron microscopic examination. We also investigated the relationship between p53 tumor-suppressor protein expression and cell morphology and survival time.
Materials and Methods
Biopsy specimens of gastrointestinal MCTs collected from 1990 to 2000 from 39 dogs were sent to the Research Institute of Drug Safety (Setsunan University) and to the Marupi Lifetech Company for histopathologic examination. All specimens were from the gastrointestinal tract of dogs that were free of cutaneous MCTs and systemic mastocytosis. Specimens were fixed in 10% phosphate-buffered or neutral formalin (pH 7.4), dehydrated in a graded series of ethanol, and embedded in paraffin, after which 4-μm-thick sections were stained with hematoxylin and eosin (HE). Representative sections were also stained with toluidine blue and alcian blue. For immunohistochemistry, the primary antibodies used were anti-human c-kit (1:800 polyclonal antibody; DAKO Japan, Kyoto, Japan; mast cells of normal canine duodenum used as positive control), anti-human mast cell tryptase (1:400 monoclonal antibody AAI; Serotec Ltd., Oxford, UK; mast cells of normal canine duodenum used as positive control), anti-canine CD45RA (1:50 monoclonal antibody CA21.4B3; from Dr. P. F. Moore; B cells and mast cells of normal canine lymph node used as positive control), anti-human CD3 (1:200 polyclonal antibody; DAKO, Glostrup, Denmark; T cells of normal canine lymph node used as positive control), anti-human CD79αcy (1:25 monoclonal antibody HM57; DAKO, Denmark; B cells of normal canine lymph node used as positive control), anti-human chromogranin A (prediluted monoclonal antibody DAK-A3; DAKO EPOS, Glostrup, Denmark; medulla cells of normal canine adrenal gland used as positive control), anti-human synaptophysin (prediluted polyclonal antibody; DAKO EPOS; medulla cells of normal canine adrenal gland used as positive control), anti-human cytokeratin (1:200 monoclonal antibody MNF116; DAKO Denmark; mucosal epithelial cells of normal canine duodenum used as positive control), anti-vimentin (1:200 monoclonal antibody V9; DAKO Denmark; submucosal cells of normal canine duodenum used as positive control), anti-human proliferating cell nuclear antigen (PCNA) (1:400 monoclonal antibody PC10; DAKO Denmark; mucosal epithelial cells of normal canine duodenum used as positive control), and anti-human p53 (1:200 rabbit polyclonal antibody CM-1 against wild and mutant forms of p53; Signet Laboratories, Dedham, MA; tumor cells of canine mammary adenocarcinoma used as positive control). After all tissue sections were deparaffinized and rehydrated, slides for c-kit, mast cell tryptase, CD79αcy, vimentin, chromogranin A, synaptophysin, PCNA, and p53 were immersed in citrate buffer (pH 6.0) and microwaved for 20 minutes to retrieve antigens. Tissue sections for CD45RA, CD3, and cytokeratin were incubated in pepsin (37 C, 20 minutes) for antigen retrieval. All slides were rinsed in Tris-buffered saline with Tween 20 (TBS-T), treated with 1% hydrogen peroxide, and rinsed again using TBS-T. Slides were incubated in bovine serum for 5 minutes and with primary antibodies overnight at 4 C. The next day, slides (except for chromogranin A and synaptophysin) were rinsed using TBS-T and treated for 30 minutes at room temperature with N-Histofine MAX PO (M or R) (Nichirei, Tokyo, Japan), which is a labeled polymer prepared by combining amino acid polymers with peroxidase and goat anti-mouse immunoglobulin and goat anti-rabbit immunoglobulin reduced to Fab fragment. Slides were rinsed using TBS-T, and diaminobenzidine (DAB) solution containing 0.01% hydrogen peroxide was used for color development. Slides for chromogranin A and synaptophysin were rinsed using TBS-T, and DAB solution containing 0.01% hydrogen peroxide was used for the peroxidase coloring reaction. The slides were then counterstained with Mayer's hematoxylin. For the evaluation of PCNA staining, the percentage of strongly positive cells in >500 cells was determined by image processing and analysis software (Ultimage pro version 2.6.1; Graftek, Mirmande, France). For electron microscopy, seven formalin-fixed samples were further fixed with 2.5% glutaraldehyde in 0.1 M phosphate buffer, pH 7.4, postfixed in 1% osmium tetroxide solution (pH 7.4) for 2 hours, and processed into resin. Semithin sections were cut and stained with toluidine blue. Ultrathin sections were cut and stained with uranyl acetate and lead citrate and examined under an electron microscope (JEM 1200EX; JEOL, Tokyo, Japan).
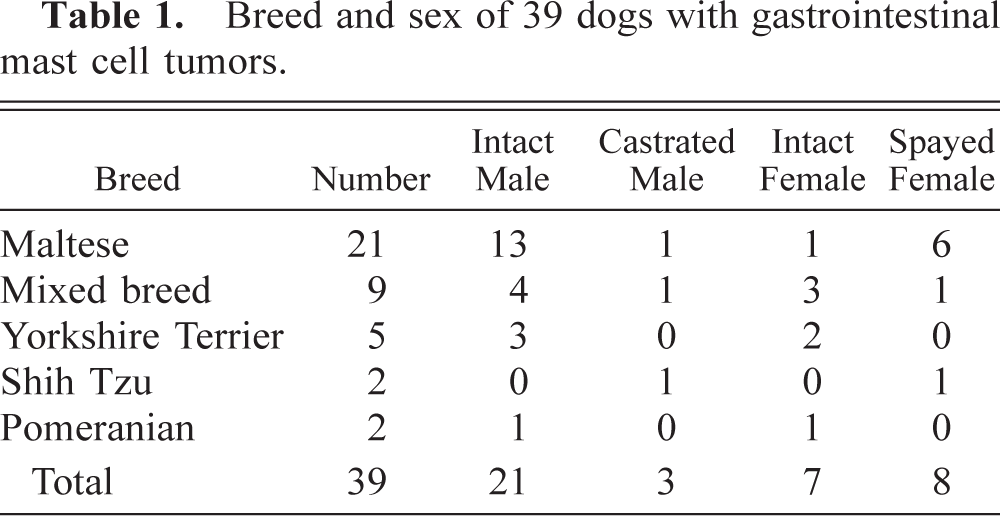
Breed and sex of 39 dogs with gastrointestinal mast cell tumors.
Immunohistochemical and special staining results of mast cell tumors of the gastrointestinal tract.∗
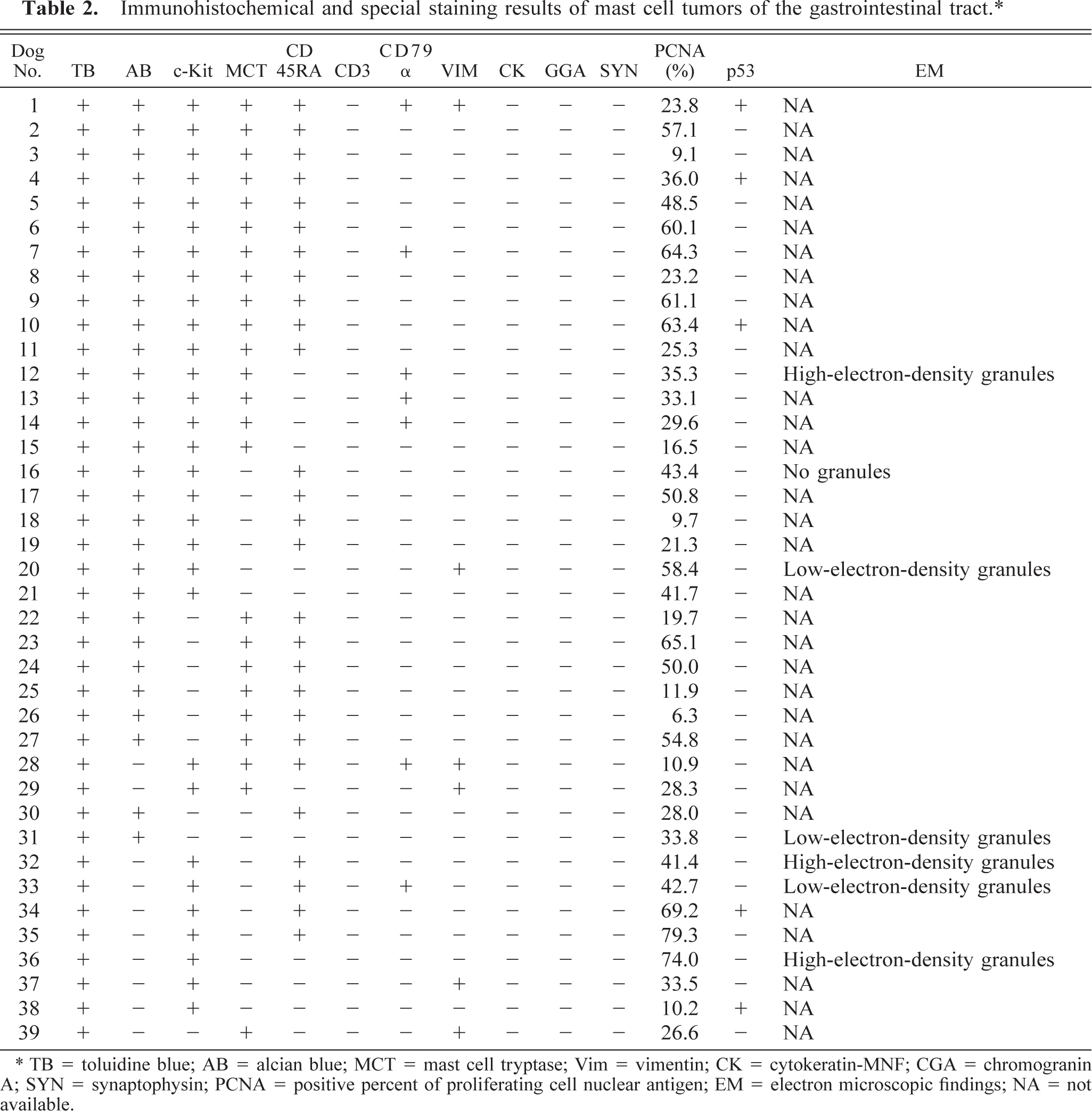
TB = toluidine blue; AB = alcian blue; MCT = mast cell tryptase; Vim = vimentin; CK = cytokeratin-MNF; CGA = chromogranin A; SYN = synaptophysin; PCNA = positive percent of proliferating cell nuclear antigen; EM = electron microscopic findings; NA = not available.
Result
About 82% of the 39 affected dogs were miniature breeds: Maltese (21, 53.8%), mixed breed (9, 23.1%), Yorkshire Terrier (5, 12.8%), Shih-Tzu (2, 5.1%) and Pomeranian (2, 5.1%). The average age was 9.7 ± 2.6 years (range, 2–15 years; median, 10 years), and there were intact males (53.8%) and females (17.9%) and neutered males (7.7%) and females (20.5%). Tumors were located in the stomach (20.5%), duodenum (33.3%), small intestine (28.2%), and large intestine (10.3%) and in multiple areas (7.7%). Clinical findings were vomiting, diarrhea, and melena in almost all cases. Survival data were available for 23 dogs. The survival rate for the first 30 days after first admission was 39.1%, and only two dogs survived over 180 days (Fig. 1).
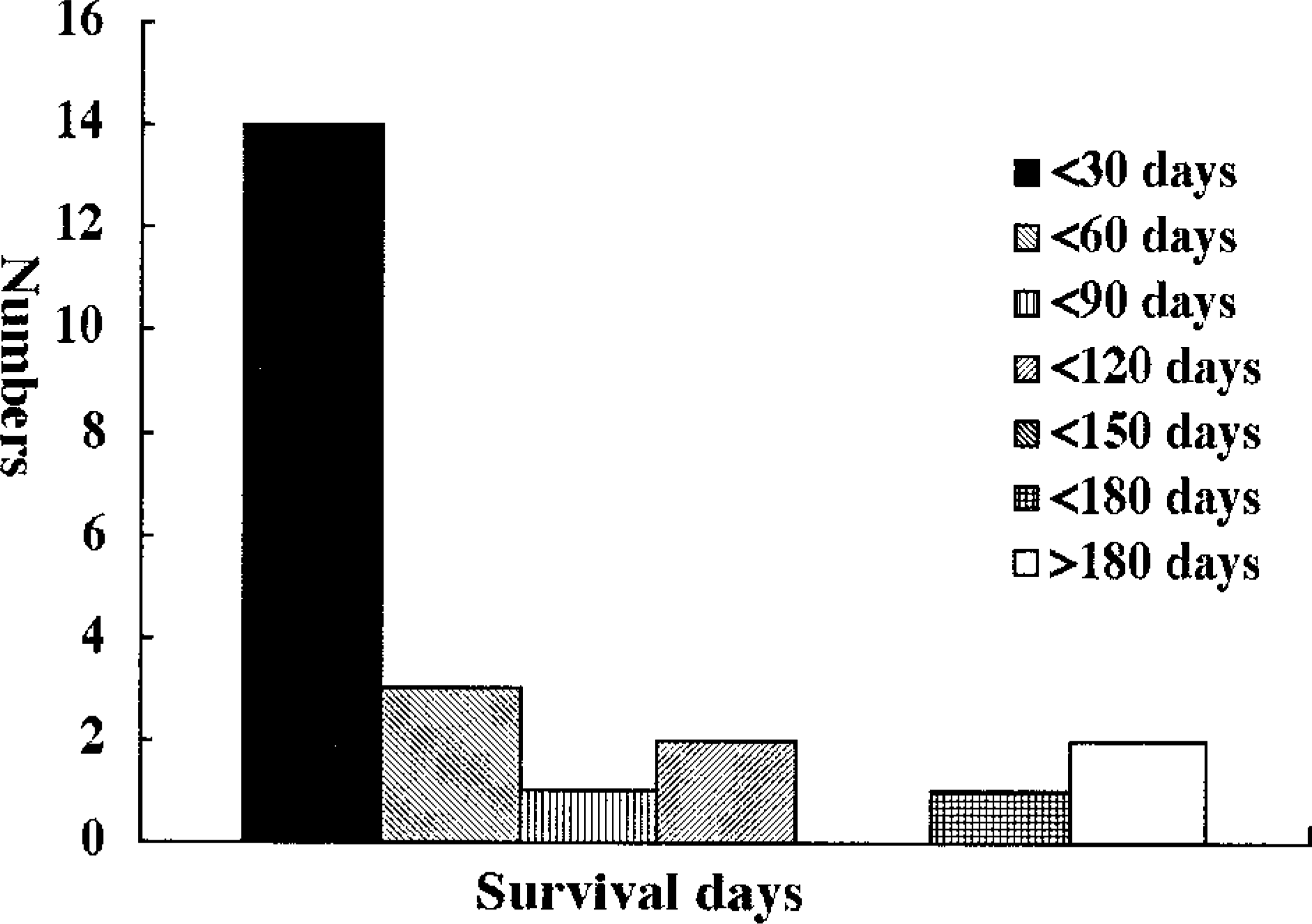
The survival (in days) of dogs with gastrointestinal mast cell tumors.
Grossly, the gastrointestinal wall in all dogs was prominently thickened, and the lumen of the gut was usually narrowed. After dissection of the gastrointestinal wall, ulceration was often seen in the thickened mucosa. The cut surface of the tumor was white to gray and of variable consistency. Microscopically, there was diffuse tumor cell invasion from the mucosal cell layer to the tunica muscularis or serosa, sometimes replacing all layers of the gastrointestinal wall (Fig. 2). Mucosal ulceration was usually associated with tumor cell infiltration. Fibrous stroma was observed in about half of the tumors (Fig. 3), but fine fibrovascular stroma did not form a lobular pattern. Tumor cells were round and monomorphic (26%), round to ovoid with slight pleomorphism (56%), or pleomorphic (18%) (Figs. 4–6). Each tumor cell usually showed a distinct cell boundary. The nuclei were round and monomorphic (26%), round to ovoid with slight pleomorphism (56%), or pleomorphic (18%). Nuclei had scattered chromatin with small distinct nucleoli. Mitotic figures were seen in 77% of tumors and were especially numerous in 23% of tumors; 38.4% ± 20.2% of the cells were positive for PCNA. Occasional cells with giant nuclei were present in 64% of tumors, and multinucleated giant cells were observed in 31% of tumors. Cells had moderate to abundant, slightly eosinophilic, and granular cytoplasm in 80% of tumors. However, basophilic granular cytoplasm, which is observed in differentiated cutaneous type mast cells (CTMCs), was present in only two tumors.
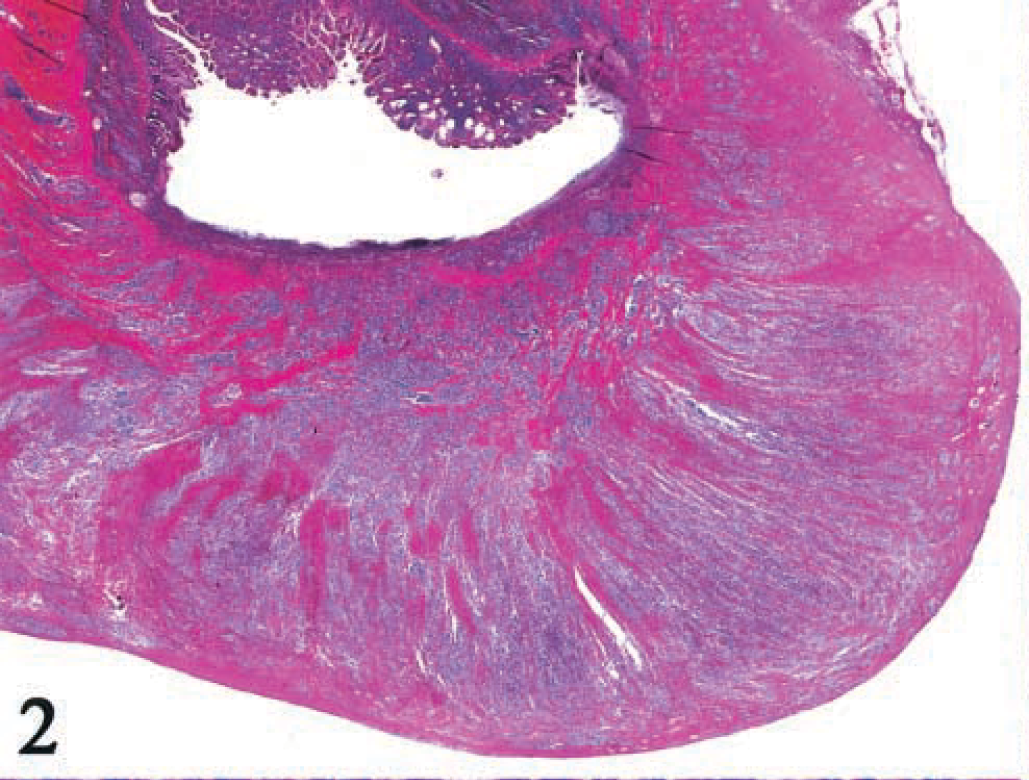
Jejunum; dog No. 22. Tumor cells arediffusely invading from the mucosal cell layer to the serosa. Mucosal ulceration is visible with tumor cell infiltration. HE.
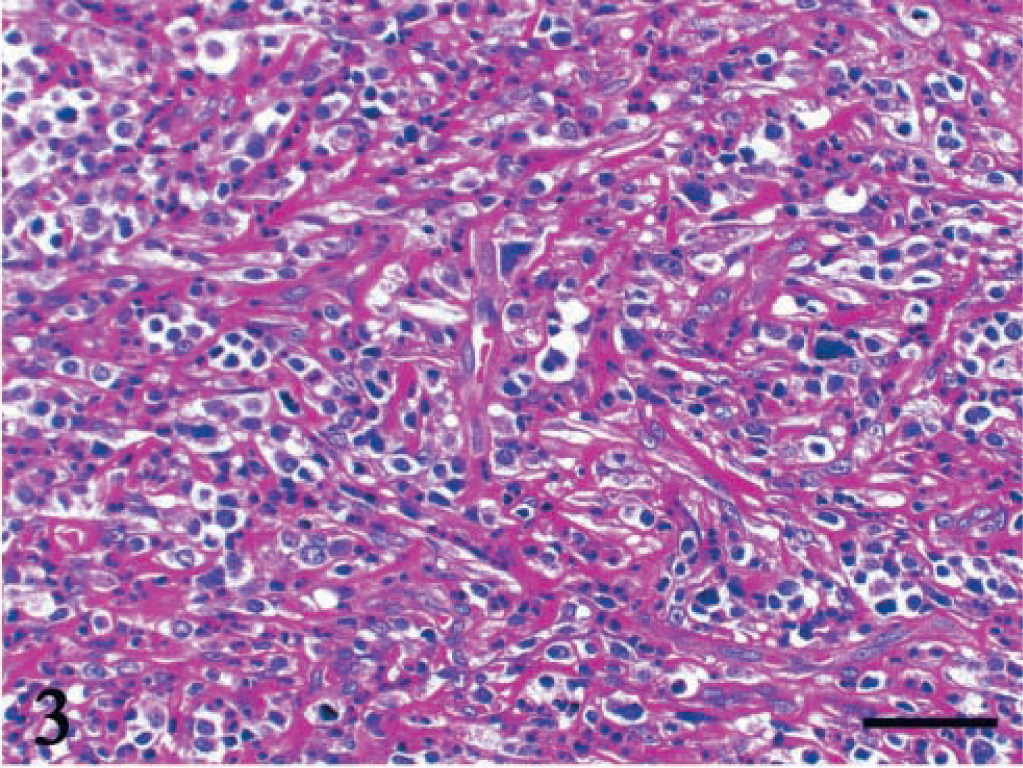
Duodenum; dog No. 4. Thick fibrous stroma has formed among tumor cells. HE. Bar = 50 μm.
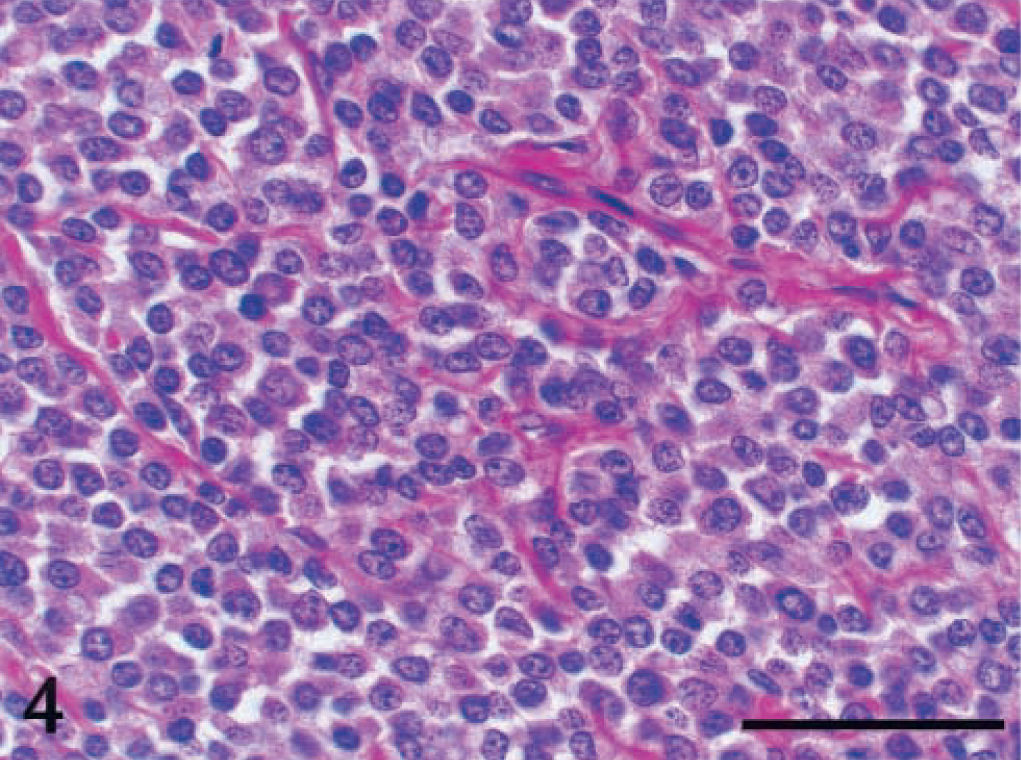
Ileum; dog No. 2. Round tumor cells have round nuclei with scattered chromatin and small distinct nucleoli. Cell boundary is usually indistinct. HE. Bar = 50 μm.
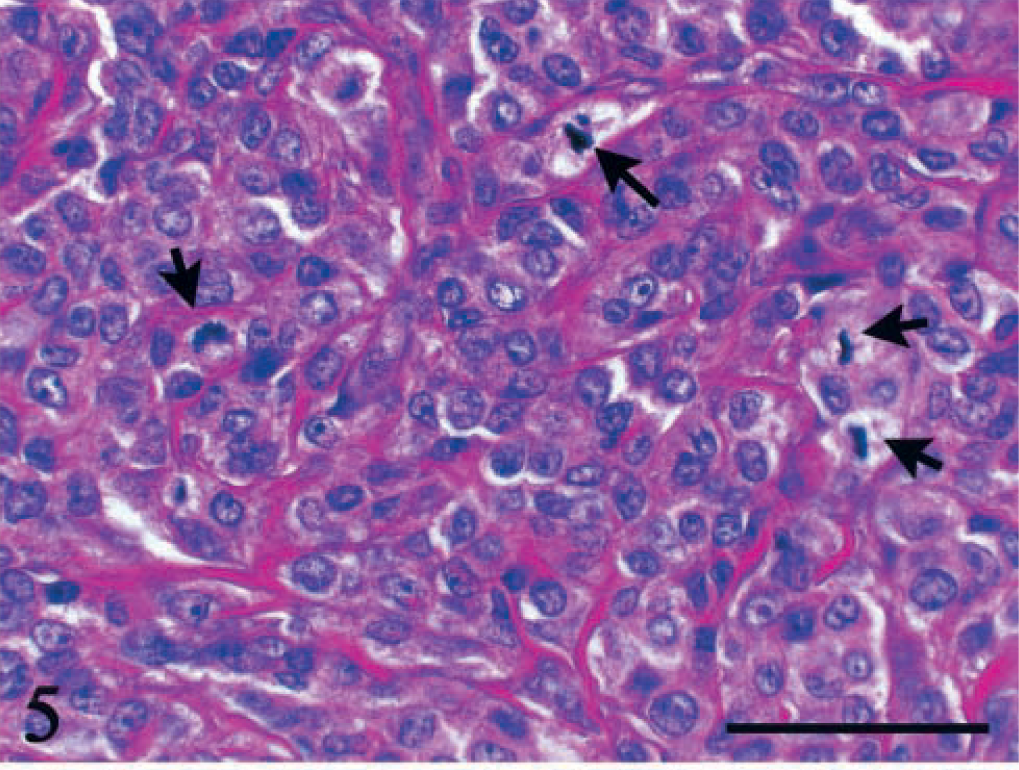
Stomach; dog No. 27. Slight pleomorphic tumor cells include round to ovoid nuclei with some pleomorphism. Numerous mitotic figures are visible (arrows). HE. Bar = 50 µm
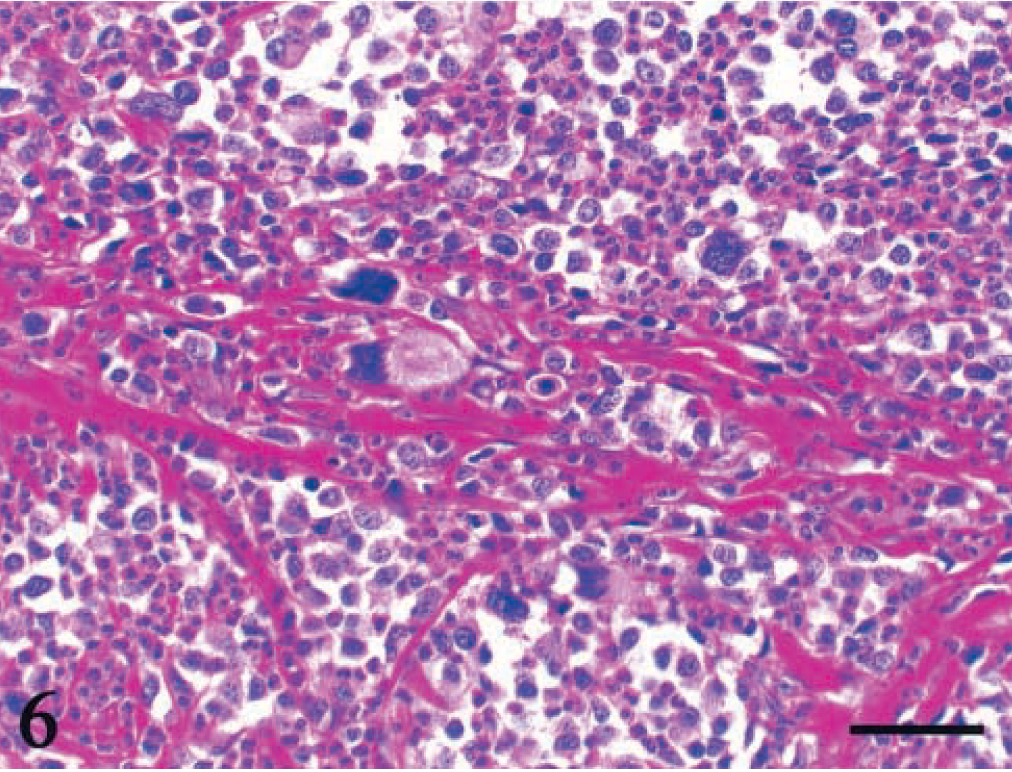
Duodenum; dog No. 19. Pleomorphic tumor cells have giant nuclei and eosinophil infiltration. HE. Bar = 50 μm.
In all tumors, cytoplasmic granules showed metachromasia after toluidine blue staining, but the number of granules was relatively small (Figs. 7, 8). In 74% of tumors, cells were stained with alcian blue (Figs. 8, 9). Immunohistochemical staining for c-kit and mast cell tryptase was positive in 77% and 62% of tumors, respectively; c-kit was detected in the cell membrane and cytoplasm, and mast cell tryptase was localized to the cytoplasm (Figs. 8, 10, 11). Staining was positive for at least one of the markers (metachromasia, alcian blue, c-kit, or mast cell tryptase) in all tumors. Positivity for CD45RA, CD79αcy, and vimentin was demonstrated in 69%, 18%, and 15% of tumors, respectively. However, tumor cells were negative for CD3, cytokeratin, chromogranin A, and synaptophysin. Necrosis and edema were not conspicuous except in the area near mucosal ulcers. Eosinophil infiltration was slight to heavy in 92% of tumors. Lymph and blood vessel invasion was seen in 31% and 13% of tumors, respectively. Many tumors also showed lymph node metastasis (11 of 14 dogs for which the lymph nodes were examined). Local metastasis was detected in two of the 23 dogs for which survival data were available; tumor cells metastasized to the liver, kidney, adrenal gland, and intestine. Immunohistochemical staining for p53 was positive in 13% of tumors (Fig. 12). Reactivity for all staining markers and p53 was not related to cell pleomorphism, vessel invasion, or survival time.
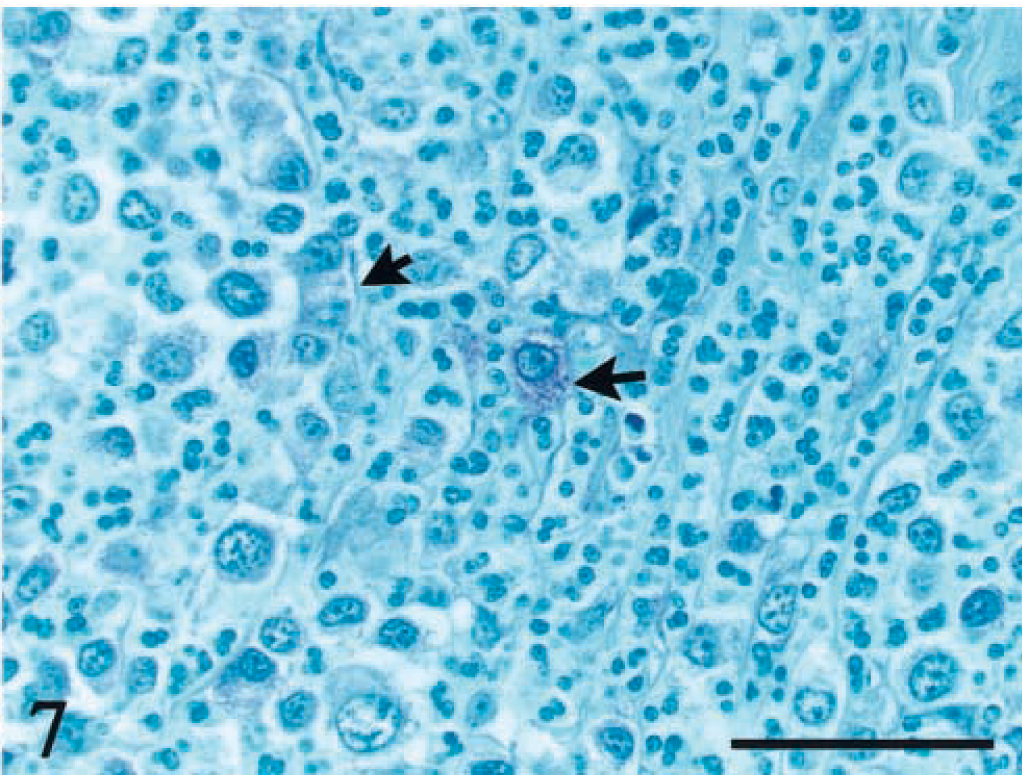
Duodenum; dog No. 19. Metachromatic granules are present in some tumor cells but are few in number (arrows). Toluidine blue. Bar = 50 μm.
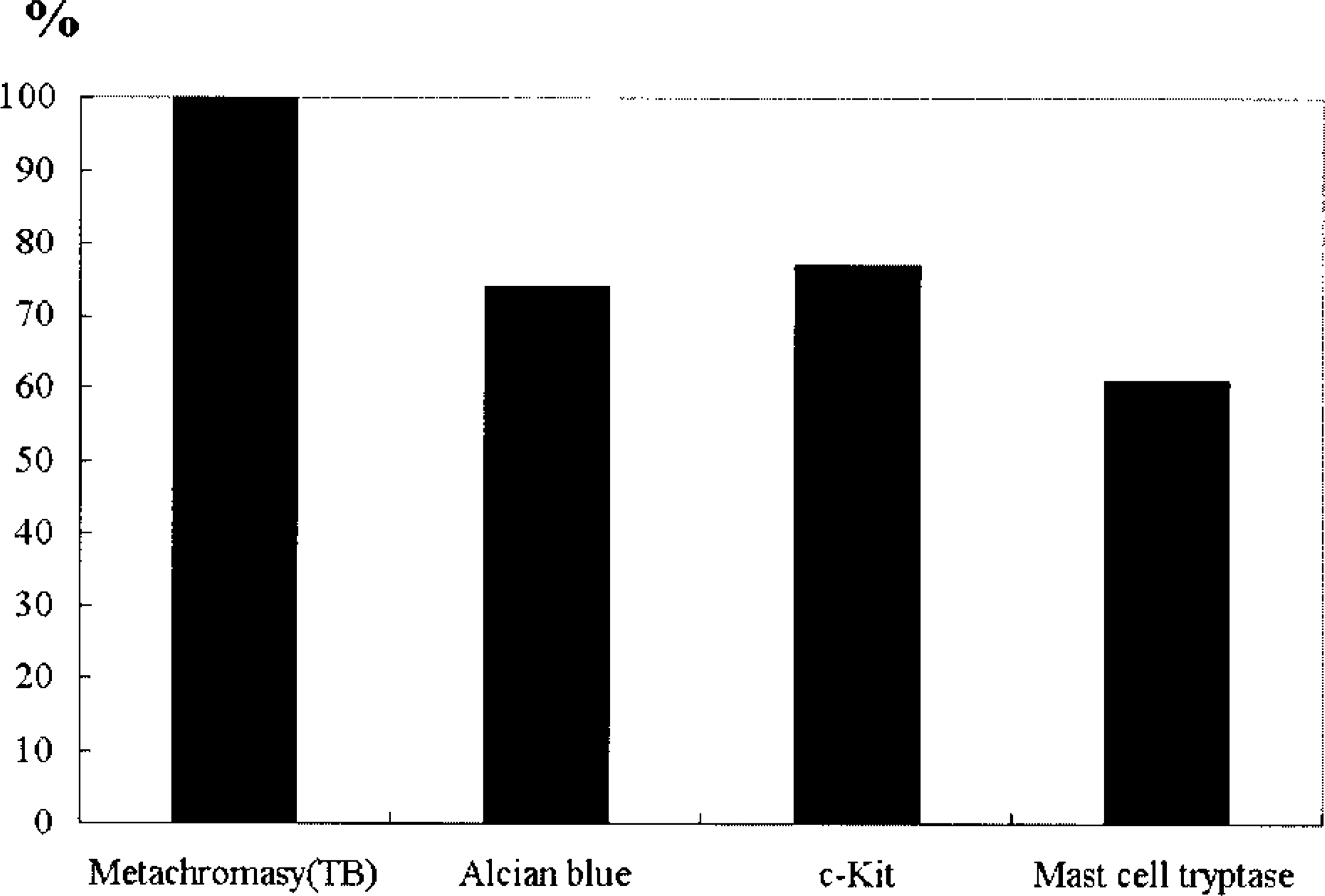
Positive staining of mast cell tumor cells for metachromasia with toluidine blue (TB) and for other markers with alcian blue, anti–c-kit, and anti–mast cell tryptase.
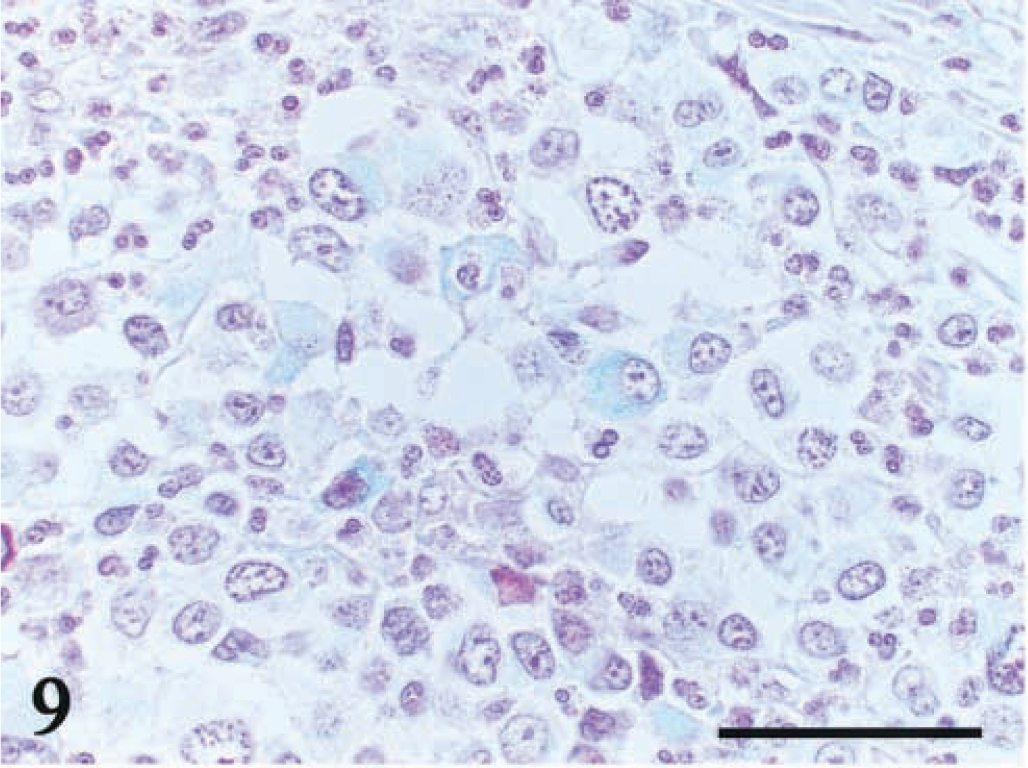
Duodenum; dog No. 19. Positive straining with alcian blue is visible in some tumor cells. Bar = 50 μm.
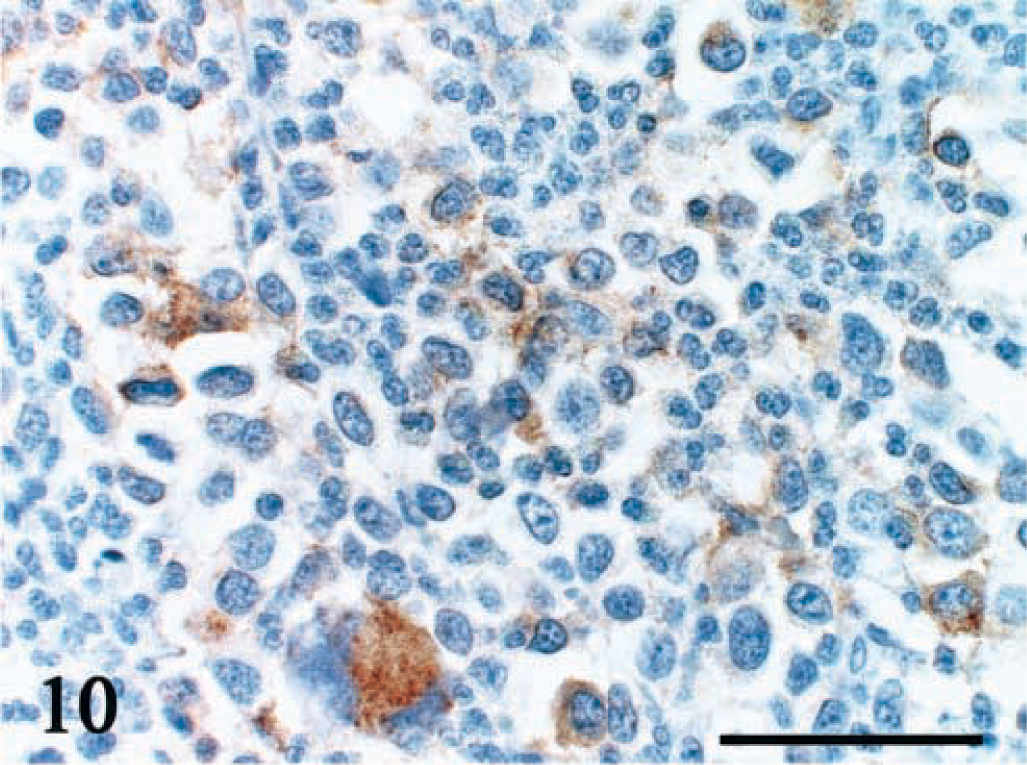
Duodenum; dog No. 19. Immunoreactivity for c-kit is visible in the cell membrane and cytoplasm. Universal immuno-enzyme polymer method, Mayer's hematoxylin counterstain. Bar = 50 μm.
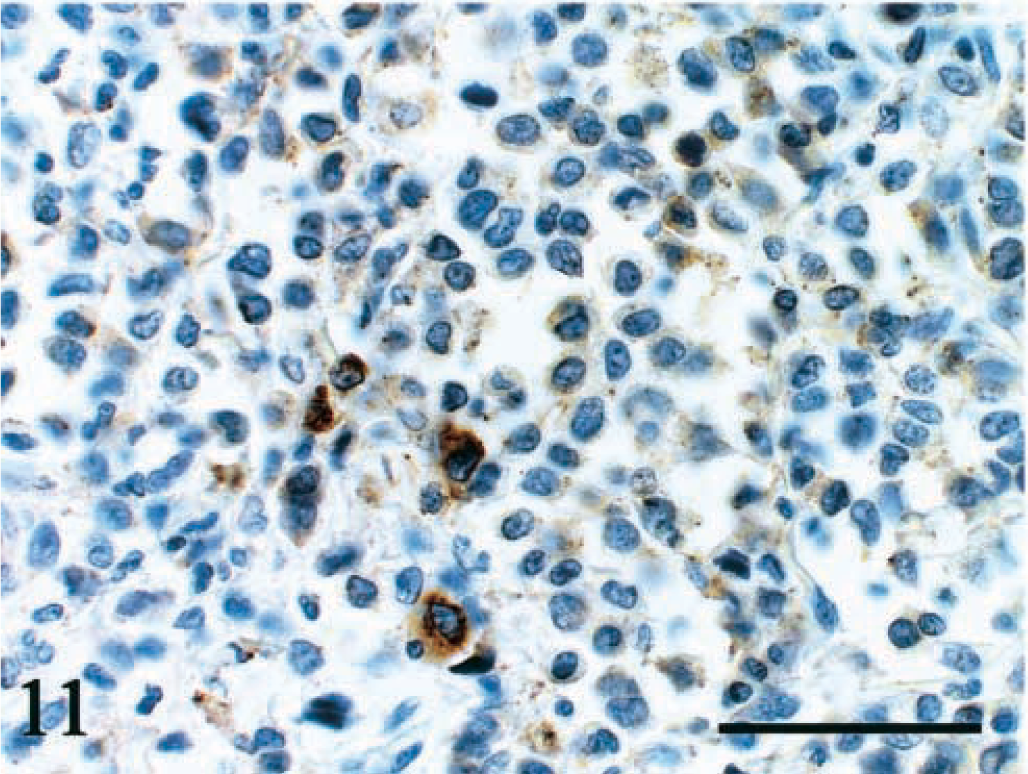
Duodenum; dog No. 6. Immunoreactivity for mast cell tryptase is visible in the cytoplasm. Universal immuno-enzyme polymer method, Mayer's hematoxylin counterstain. Bar = 50 μm.
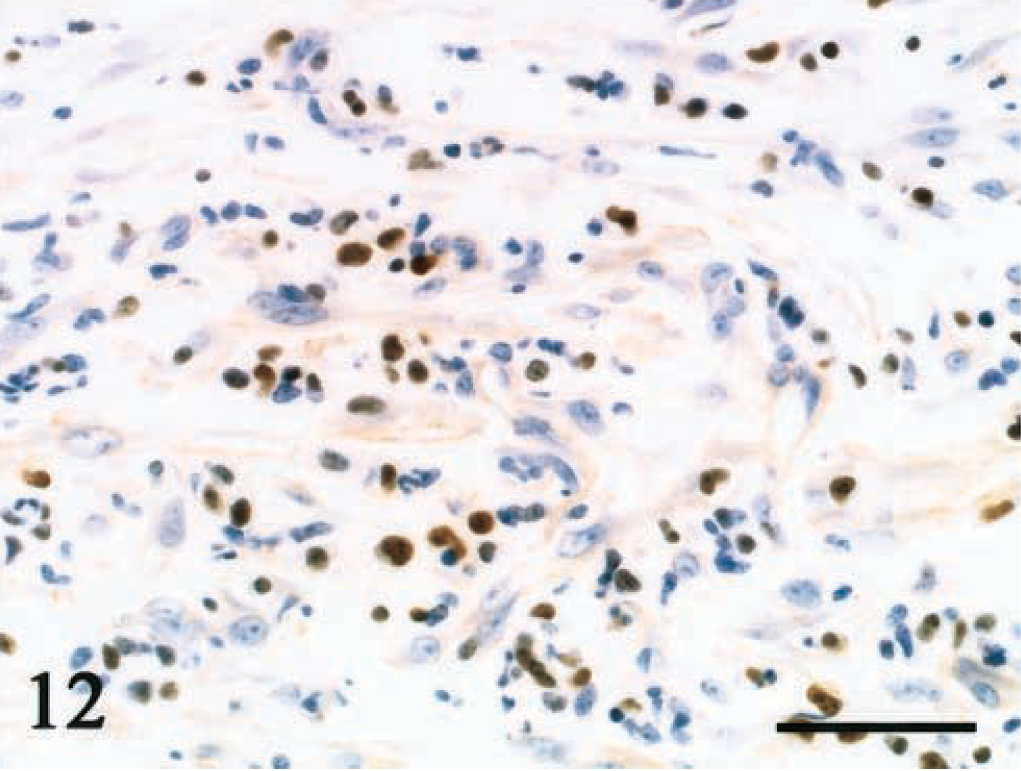
Ileum; dog No. 1. Immunohistochemical staining for p53 is positive in many tumor cells. Universal immuno-enzyme polymer method, Mayer's hematoxylin counterstain. Bar = 50 μm.
Electron microscopic examination was conducted in seven of the 39 tumors. The tumor cells had smooth cell membranes, and cytoplasmic processes were not conspicuous. Nuclei were round to lobulated, and heterochromatin was recognized along the nuclear membrane. Euchromatin was prominent. The number, shape, and electron density of the cytoplasmic granules varied among tumors, but almost all cells included a small number of cytoplasmic granules. The granules in three tumors were membrane bound, round, 0.5–1.5 μm in diameter, and of low density. The granules were microgranular or empty and sometimes had a lamellar structure (Fig. 13). The granules in two tumors contained high-density homogeneous material with crystalline structure. Cell organelles were more prominent in the cells with scarce granules. There was one tumor in which the granules could not be seen by electron microscopy despite the metachromasia they showed with light microscopy.
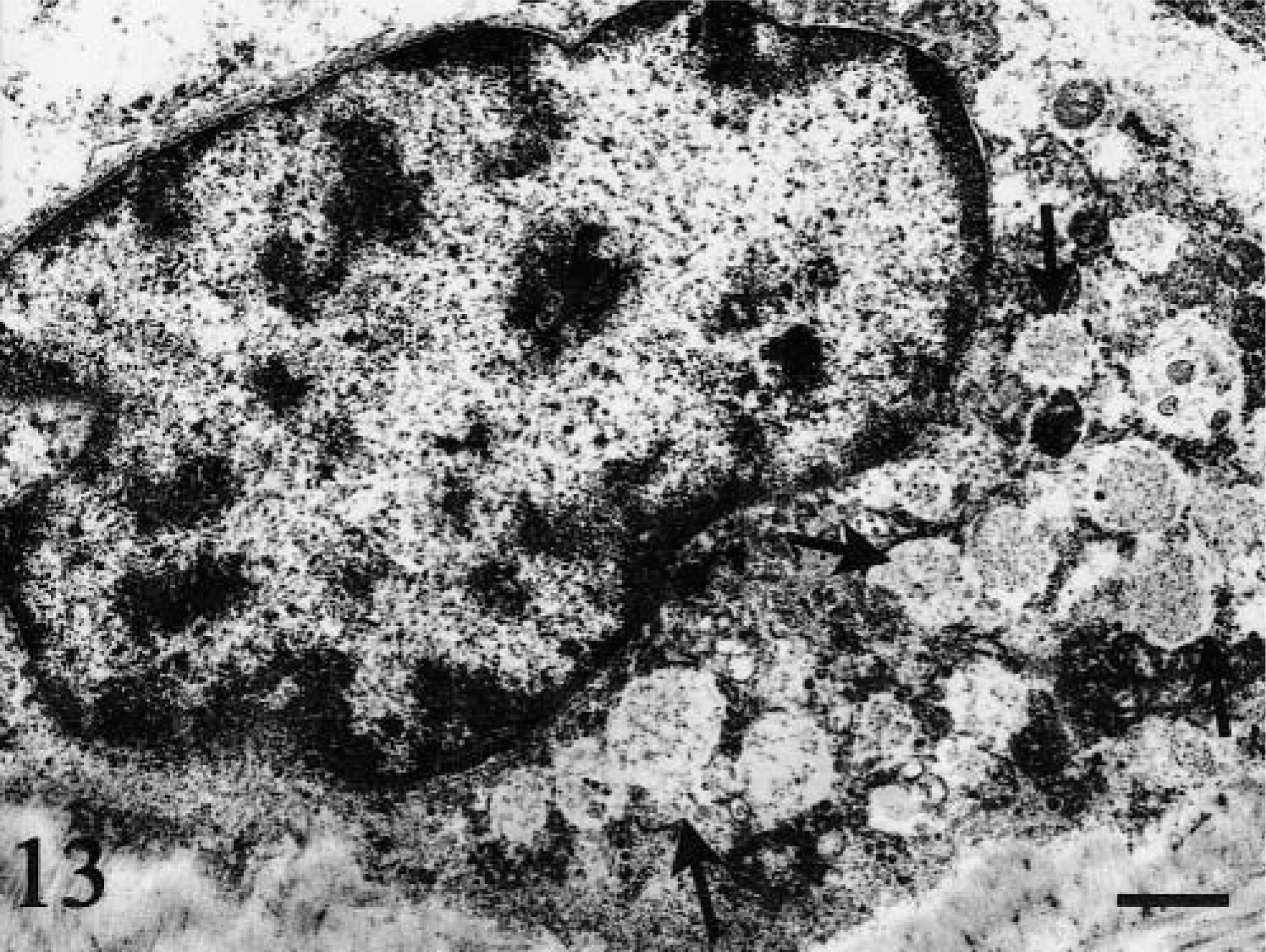
Duodenum; dog No. 31. Granules are of low density and are microgranular to empty. Uranyl acetate and lead citrate. Bar = 1 μm.
Discussion
The histopathologic features of tumor cells in gastrointestinal MCTs were similar to those associated with certain types of malignant lymphoma. Furthermore, gastrointestinal MCTs had to be differentiated from metastatic lesions of cutaneous MCTs. Tumor cells of malignant lymphoma are characterized by round nuclei with dense chromatin, distinct large nucleoli, and scanty cytoplasm. In contrast, the histologic characteristics of the MCT cells in these affected dogs were moderate to abundant cytoplasm, round nuclei with scattered chromatin, fibrous stroma, and eosinophil infiltration. Little necrosis was seen. Metachromasia (observed with toluidine blue staining) was required to confirm the diagnosis because some malignant lymphoma cells had morphologic features similar to those of gastrointestinal MCT cells. Since gastrointestinal MCTs had scarcely any metachromatic granules,4,17 metachromasia alone did not provide definitive diagnosis of gastrointestinal MCT. The diagnostic panel, which included immunostaining for mast cell tryptase and c-kit, electron microscopy, and staining for metachromasia, allowed definitive diagnosis of MCT.Negative staining for CD3 distinguished MCT from malignant T-cell lymphoma. In 32 dogs, MCT was differentiated from malignant B-cell lymphoma by negative staining of MCT for CD79αcy, but seven cases of MCT showed positive staining. However, all seven CD79αcy–positive tumors had metachromatic granules and were positive for at least one of the immunohistochemical mast cell markers (c-kit or mast cell tryptase). CD45RA could not be used to discriminate between B-cell lymphoma and MCT because it is expressed by all B-cell lymphomas and by MCTs.
Because some of these tumors resembled gastrointestinal stromal tumors (GISTs), carcinomas, and neuroendocrine tumors, differential diagnosis was necessary. GIST constitutes the largest category of primary nonepithelial neoplasms of the stomach and small bowel. GIST have spindle cells and/or epithelioid cells, which are usually positive for c-kit.14 MCTs also showed immunoreactivity for c-kit, but MCT cells were round to pleomorphic and usually positive for metachromasia and/or mast cell tryptase. Negative staining for cytokeratin is extremely useful in distinguishing MCTs from carcinomas, which express cytokeratin. MCTs did not stain with chromogranin A and synaptophysin, differentiating them from neuroendocrine tumors.
Cutaneous MCTs resembled gastrointestinal MCTs with respect to cell shape, collagenous stroma, and eosinophil infiltration. The greatest difference was the presence of abundant granules with metachromasia in cutaneous MCTs. In addition, tumor cells with mitotic activity and giant nuclei or multinucleated giant cells were seen more frequently in gastrointestinal MCTs than in cutaneous MCTs. However, it is difficult to accurately distinguish gastrointestinal MCTs from metastatic cutaneous MCTs in animals with both cutaneous and gastrointestinal disease. For this study, animals with cutaneous MCTs were excluded.
Mast cells are divided into two subtypes: CTMCs and mucosal type mast cells (MMCs).2 CTMCs are found mainly in skin, various connective tissues, and the nonmucosal portion of the gastrointestinal tract. These cells show metachromasia in formalin-fixed tissue, contain the serine proteases chymase and tryptase, and uniformly possess electron-dense granules.2 In contrast to CTMCs, MMCs are located in the lamina propria of the gastrointestinal tract and lung, show no metachromasia in formalin-fixed tissues, contain only tryptase, and include variably electron-dense granules.2 Among previous reports on MCTs originating in the gastrointestinal tract, only one report specified the origin of the mast cells.4 In that case, the MCT may have originated from CTMCs, based on heparin immunostaining and electron microscopic features. In another previous report,17 certain characteristics of intestinal MCTs were similar to those of MMCs, but the derivation of mast cells was not clearly specified. In the present study, tumor cells in some dogs may have arisen from CTMCs, based on the results of electron microscopy and the finding of metachromasia. In other dogs, the tumors were judged to be from MMCs or of unknown origin. Both CTMCs and MMCs are derived from common bone marrow precursors and can transform into one another, has demonstrated in experiments in which mature CTMCs transformed into MMCs after injection into mucosa and MMCs transformed into CTMCs after injection into the abdominal cavity.6,9,10,16 Thus, the phenotypic characteristics of mast cell populations may alter in response to changes in the microenvironment.7
Miniature breeds, especially Maltese, were most often affected by gastrointestinal MCTs, as reported previously.17 As with cutaneous MCTs, no gender differences were found with gastrointestinal MCTs.18 Takahashi et al.17 reported that males were more susceptible than females to intestinal MCTs. However, the number of dogs in their study was too small to evaluate sex differences for this tumor type. The results of the present study indicate that gastrointestinal MCTs are characterized by nonspecific clinical manifestations, a poor prognosis, and a propensity for involvement of the upper digestive tract. Common clinical signs in cutaneous MCTs were digestive signs attributed to the effects of excessive release of histamine associated with the degranulation of tumor cells.8 Therefore, gastric and duodenal ulcers in the dogs with cutaneous MCTs were considered part of a paraneoplastic syndrome. However, digestive signs of gastrointestinal MCTs could also be attributable to the ulceration accompanying direct invasion of tumor cells. Clinical signs due to ulceration were only manifested in the advanced stage, which may account for the poor prognosis for MCTs. PCNA labeling (mean = 38%) in these gastrointestinal MCTs was higher that (mean = 21%) for cutaneous MCTs.15 The high proliferation capacity in these tumors may also contribute to the poor prognosis.
In cutaneous MCTs, the prognosis is related to the histopathologic grade of the tumor,11 and a poor prognosis is associated with more pleomorphic tumor cells. The prognosis was poor in nearly all cases of gastrointestinal MCTs, demonstrating that there is no relation between pleomorphism and prognosis for this condition. Thus, histopathologic grading systems for cutaneous MCTs were not useful in establishing the prognosis for gastrointestinal MCT.
Pleomorphic tumor cells in cutaneous MCTs (grade III tumor) showed the highest expression of c-kit, and differentiated tumor cells (grade I tumor) showed the lowest c-kit expression.13 Therefore, there appears to be an inverse correlation between the degree of differentiation of cutaneous MCTs and the expression of c-kit.13 However, not all MCTs were c-kit positive in the present study, and only a small number of tumor cells were immunoreactive, even in positive cases. There was no relation between cell pleomorphism and the expression of c-kit in gastrointestinal MCTs. The reason for negative immunoreactivity in some tumor cells was uncertain. Although c-kit is expressed mainly on the cell membrane in normal canine mast cells, it accumulated primarily in the cytoplasm of many MCT cells.13
As for cutaneous MCTs, immunohistochemical expression of p53 was not useful as a prognostic indicator in gastrointestinal MCTs.3,5 The relationship between histopathologic grade and the expression of p53 in cutaneous MCTs is uncertain. Ginn et al.3 reported that immunoreactivity for p53 in cutaneous MCT was not associated with the histopathologic grade. However, Jaffe et al.5 showed that there was a significant difference in the percentage of p53-positive cells according to histopathologic grade, with grade III tumors having a significantly greater p53 immunoreactivity than grade I or grade II tumors. However, both groups of researchers agreed that p53 immunoreactivity was not related to survival times.
Footnotes
Acknowledgements
We thank H. Takagi (Marupi Lifetech Co., Ltd.) for technical assistance.
