Abstract
Sex steroid-producing adrenocortical adenomas and carcinomas occur frequently in neutered ferrets, but the molecular events underlying tumor development are not well understood. Prepubertal gonadectomy elicits similar tumors in certain inbred or genetically engineered strains of mice, and these mouse models shed light on tumorigenesis in ferrets. in mice and ferrets, the neoplastic adrenocortical cells, which functionally resemble gonadal steroidogenic cells, arise from progenitors in the subcapsular or juxtamedullary region. Tumorigenesis in mice is influenced by the inherent susceptibility of adrenal tissue to gonadectomy-induced hormonal changes. The chronic elevation in circulating luteinizing hormone that follows ovariectomy or orchiectomy is a prerequisite for neoplastic transformation. Gonadectomy alters the plasma or local concentrations of steroid hormones and other factors that affect adrenocortical tumor development, including inhibins, activins, and Müllerian inhibiting substance. GATA-4 immunoreactivity is a hallmark of neoplastic transformation, and this transcription factor might serve to integrate intracellular signals evoked by different hormones. Synergistic interactions among GATA-4, steroidogenic factor-1, and other transcription factors enhance expression of inhibin-α and genes critical for ectopic sex steroid production, such as cytochrome P450 17α-hydroxylase/17,20 lyase and aromatase. Cases of human adrenocortical neoplasia have been linked to precocious expression of hormone receptors and to mutations that alter the activity of G-proteins or downstream effectors. Whether such genetic changes contribute to tissue susceptibility to neoplasia in neutered ferrets and mice awaits further study.
Introduction
Sex steroid–producing adrenocortical adenomas and carcinomas occur frequently in neutered ferrets and cause significant morbidity. 60, 97 It has been proposed that competent cells in the ferret adrenal cortex undergo neoplastic transformation and adopt features of gonadal steroidogenic cells in response to the hormonal changes that follow gonadectomy. 23 Chronically elevated serum luteinizing hormone (LH) levels have been implicated in the pathobiology of these neoplasms, 111, 125 but the molecular events underlying tumorigenesis remain poorly characterized. Although the ferret is a valuable experimental animal for studies of reproductive endocrinology, 4 it is not standardized with regard to genotype and therefore not ideal for studies of tumorigenesis. Moreover, the latency of adrenocortical neoplasia in neutered ferrets, which averages more than 3 years, 31, 60, 110 hinders prospective studies of tumor development in this species.
The phenomenon of gonadectomy-induced adrenocortical neoplasia also has been observed in mice, rats, guinea pigs, and hamsters. 104 Of these, the laboratory mouse is the most useful experimental model because of its well-characterized genetics. In certain inbred 21 or genetically engineered 35, 72, 89 strains of mice, adrenocortical neoplasms develop within weeks to months of gonadectomy with near complete penetrance. Although the similarities in adrenocortical neoplasia between inbred mice and ferrets are mentioned in veterinary medicine textbooks, 23, 98 no concerted effort has been made to apply knowledge gleaned from studies of tumorigenesis in mice to ferrets. This comparative review highlights the histologic changes that accompany gonadectomy-induced adrenocortical neoplasia in both species and presents updated information on the molecular mechanisms of tumorigenesis deduced from the mouse models. The relevance to human adrenocortical tumors is also discussed.
Overview of the Structure, Function, and Development of the Adrenal Cortex
The adrenal cortex is a major source of steroid hormones that are synthesized from cholesterol through the sequential activities of a series of cytochrome P450 enzymes (Fig. 1). In most mammals, the adrenal cortex is divided into three main layers: the zona glomerulosa, zona fasciculata, and zona reticularis (Table 1). Mineralocorticoids are produced in the outermost layer, the zona glomerulosa, whereas glucocorticoids and androgen precursors are synthesized in the zona fasciculata and zona reticularis. Two less prominent layers, the zona intermedia and zona juxtamedullaris, have also been described in the ferret and other carnivores, although the functional significance of these layers is unclear. 33 Cortisol is the principal glucocorticoid secreted by the adrenal cortex of the ferret. 23 Under physiologic conditions, the ferret adrenal gland produces only limited amounts of the androgenic steroids dehydroepiandrosterone (DHEA), DHEA sulfate (DHEA-S), or androstenedione. 14, 99 In the mouse adrenal, the zona glomerulosa and zona fasciculata are well defined, but there is no discernable zona reticularis. The adrenal cortex of the young mouse contains an additional layer, the X-zone, which is adjacent to the medulla and is analogous to the fetal zone of the human adrenal cortex. 45 The function of the X-zone, which regresses at puberty in males and during the first pregnancy in females, is unknown. No correlate of the mouse X-zone or human fetal zone has been described in the ferret. Adrenocortical cells in the adult mouse lack cytochrome P450 17α-hydroxylase/C17–C20 lyase (P450c17), a dual-function enzyme required for synthesis of cortisol and sex steroids. 46 Consequently, corticosterone is the principal glucocorticoid secreted by the mouse adrenal cortex, and under normal conditions, androgenic steroids are not produced in this tissue.
Comparative anatomy and physiology of the adrenal cortex.
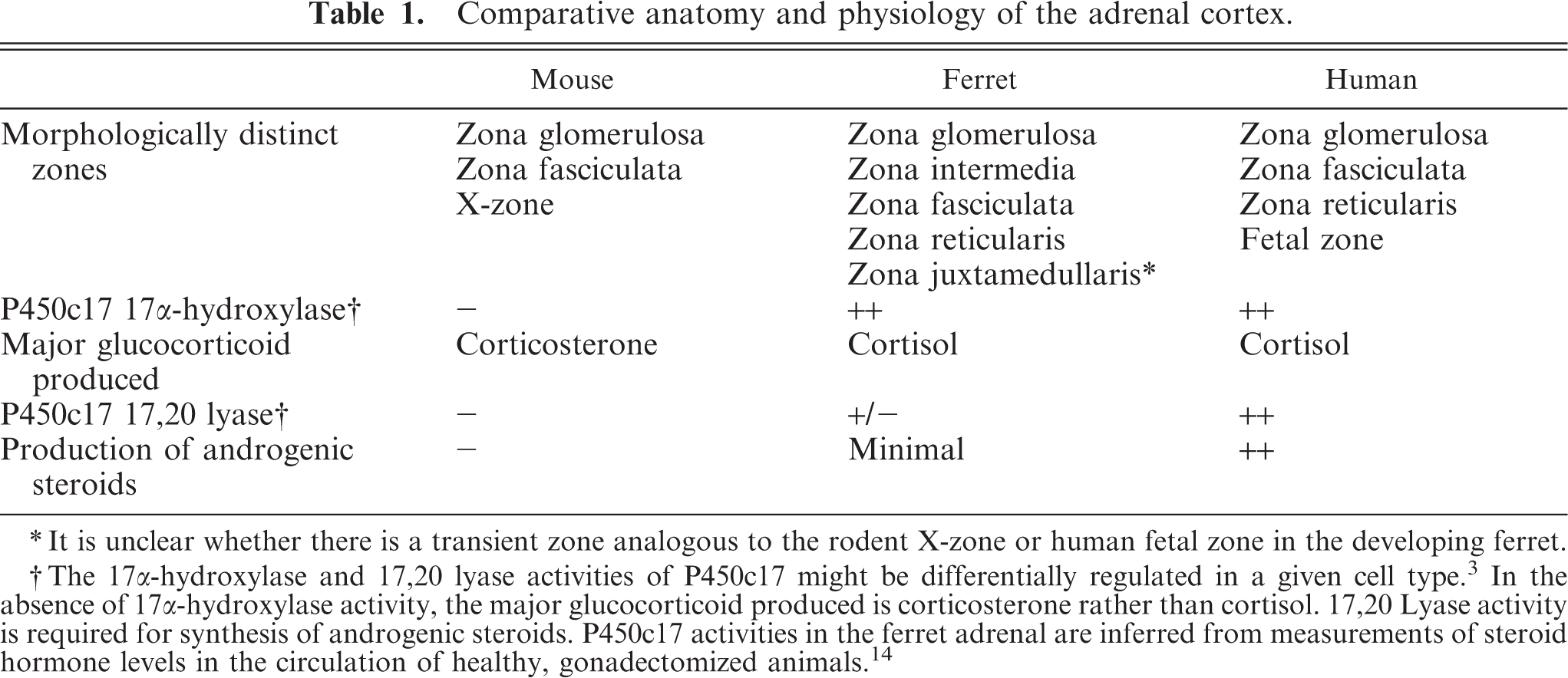
∗ It is unclear whether there is a transient zone analogous to the rodent X-zone or human fetal zone in the developing ferret.
† The 17α-hydroxylase and 17,20 lyase activities of P450c17 might be differentially regulated in a given cell type. 3 In the absence of 17α-hydroxylase activity, the major glucocorticoid produced is corticosterone rather than cortisol. 17,20 Lyase activity is required for synthesis of androgenic steroids. P450c17 activities in the ferret adrenal are inferred from measurements of steroid hormone levels in the circulation of healthy, gonadectomized animals. 14
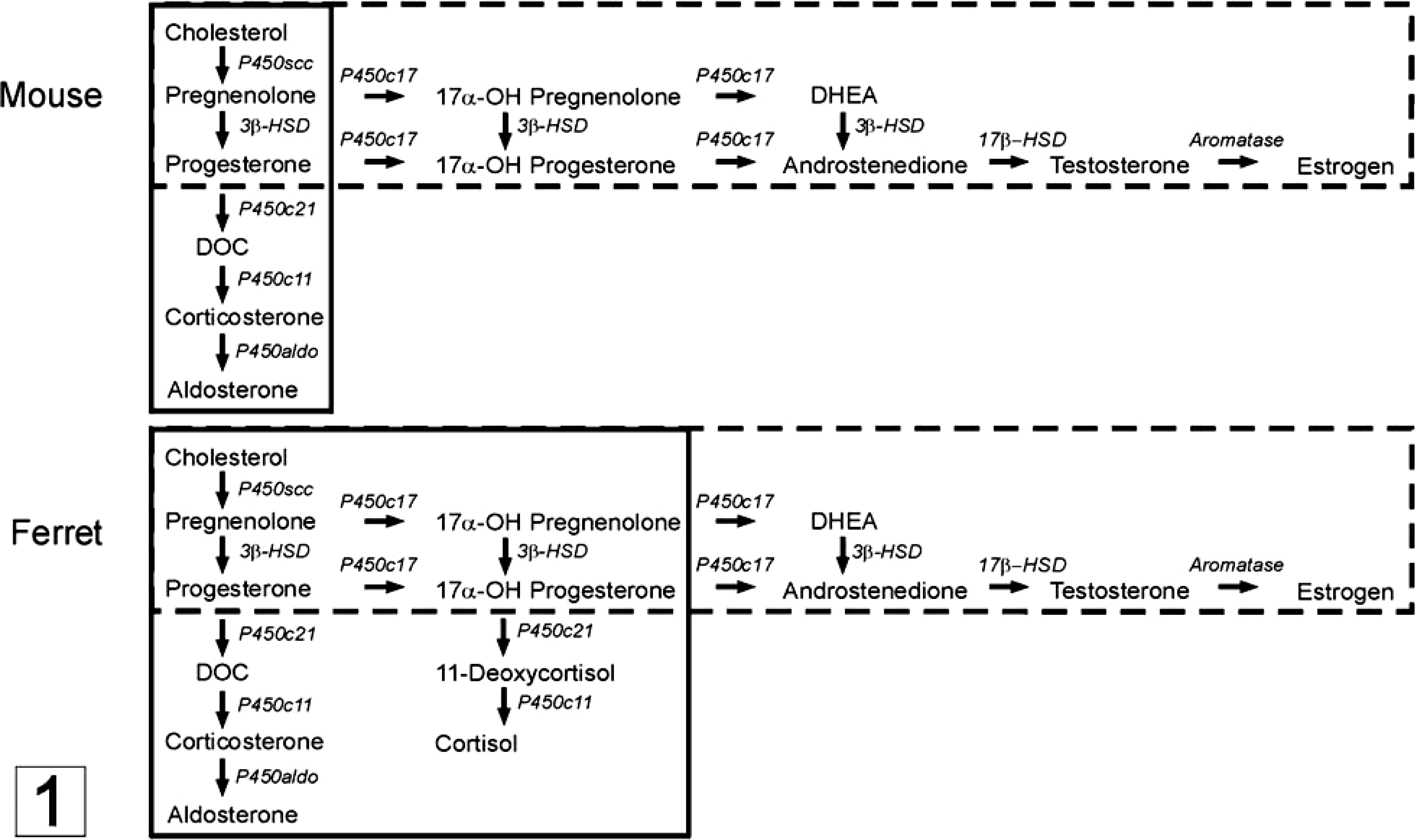
Steroid hormone biosynthesis in normal (solid lines) versus neoplastic (dashed lines) adrenocortical cells of the mouse and ferret. The repertoire of enzymes distal to the common pathway determines the steroidogenic capacity of a given cell.
During embryonic development, steroidogenic cells in the adrenal cortex and gonads arise from progenitors in the urogenital ridge (Fig. 2). 45 Differentiation of the so-called adrenogonadal primordium is thought to be influenced by growth factors secreted by the underlying mesonephros. 76 Progenitors of adrenocortical cells migrate medially and rostrally, eventually associating with neural crest derivatives that will give rise to the adrenal medulla; cells destined to become gonadal stroma move laterally and associate with primordial germ cells (Fig. 3). Mutations in certain genes expressed in the urogenital ridge or adrenogonadal primordium, such as Wilms'tumor-1 (Wt-1), Wnt4, and steroidogenic factor-1 (Sf-1) disrupt development of both gonadal and adrenal steroidogenic cells, underscoring the close relationship between these two lineages. 45, 76, 80 The further differentiation, growth, and survival of steroidogenic cells in the adrenal gland are controlled by a diverse array of hormones, including adrenocorticotropic hormone (ACTH; Fig. 4), angiotensin-II, endothelin-1, vasopressin, and insulin-related growth factors. 27, 102, 127 In certain instances, endocrine and paracrine factors traditionally associated with the function of gonadal steroidogenic cells, such as LH (Fig. 4), inhibins, and activins, can also influence the differentiation, proliferation, and function of adrenocortical progenitor cells. 6 These reproductive factors are of particular relevance to gonadectomy-induced tumorigenesis.
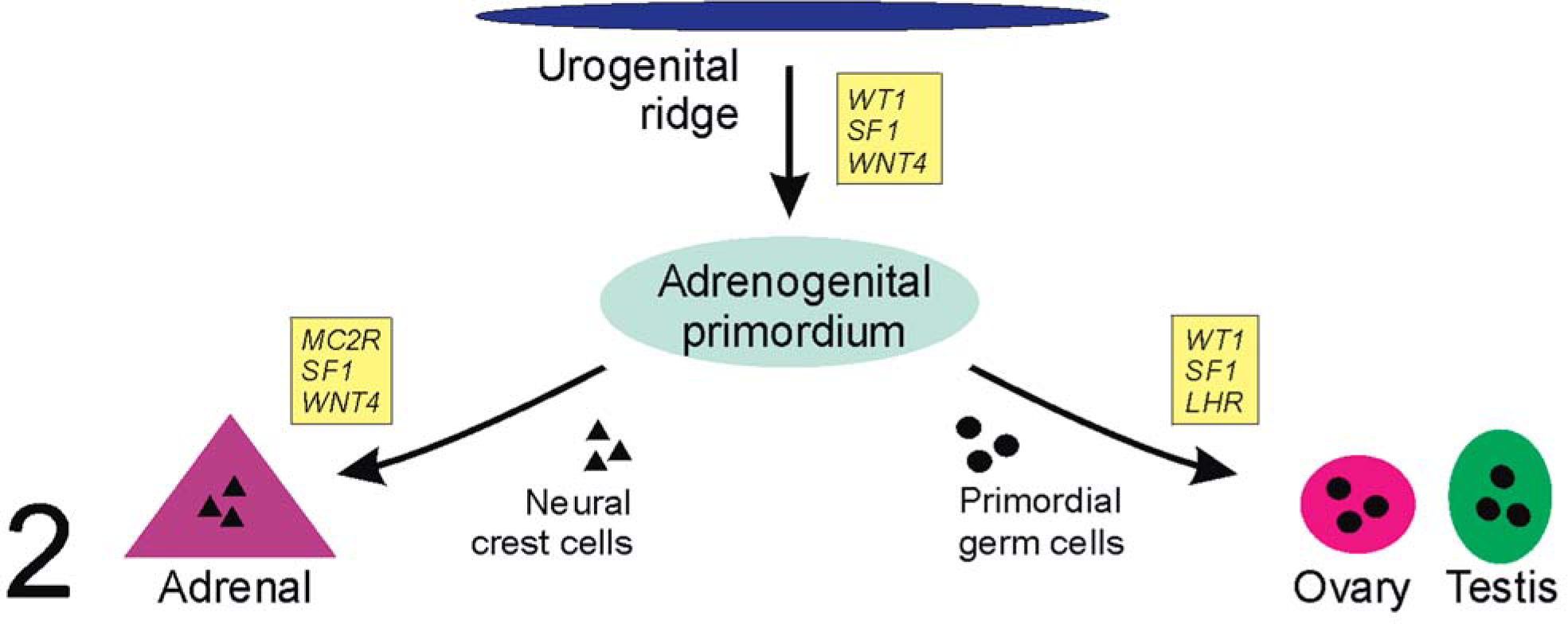
Development of the adrenal gland and gonads from the urogenital ridge. Key regulatory genes at the various developmental steps are shown in boxes.
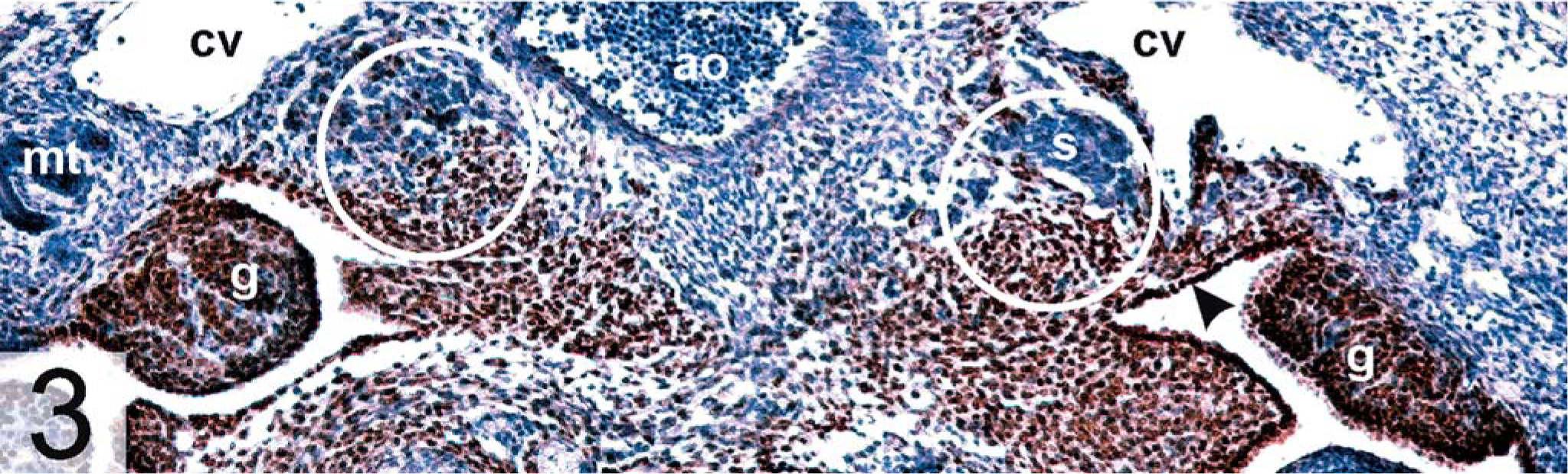
Gonadal ridge derivatives; DBA/2J mouse. Embryonic day 13.5. Arrowhead points to epithelium lining the medial coelomic bay. Dorsal aorta (ao), cardinal vein (cv), gonad (g), mesonephric tubules (mt), sympathoblasts (s). Circles indicate the nascent adrenals. GATA-4, avidin–biotin complex peroxidase method. Bar = 100 µm.
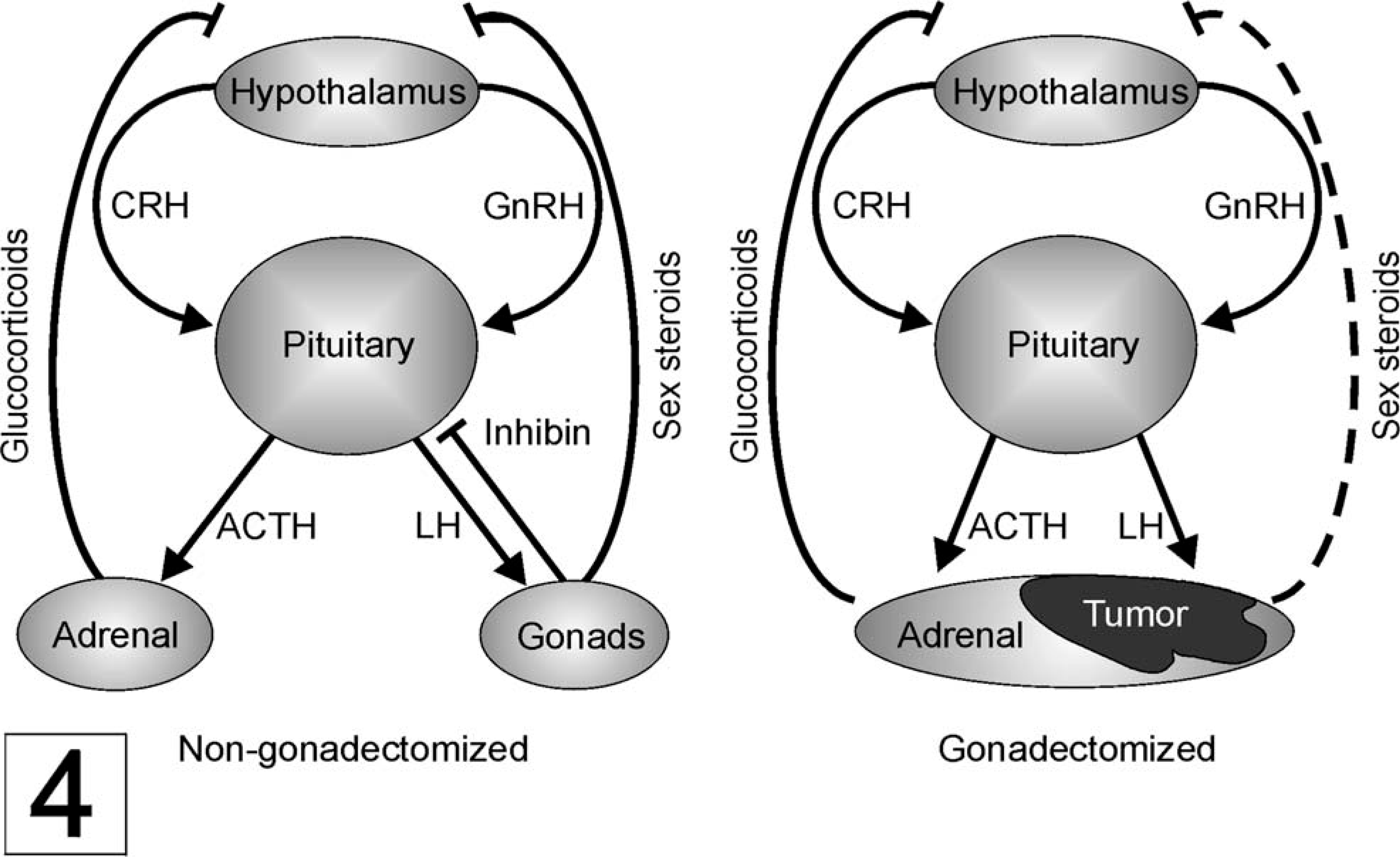
Hormone secretion and regulatory pathways in the hypothalamo–pituitary–adrenal/gonadal axes of nongonadectomized and gonadectomized animals. → and —| indicate positive and negative regulation, respectively. Following gonadectomy, feedback inhibition of the hypothalamus by sex steroids is reduced, as indicated by the dashed line.
Gonadectomy-associated Adrenocortical Neoplasia in the Ferret
In ferrets, adrenocortical neoplasia affects both genders but is more easily recognized in jills. 60, 97, 129 The prevalence of this condition has been attributed to early gonadectomy. 110 Adrenocortical tumors are common in ferrets living in the United States, where most commercially raised ferrets are castrated or ovariohysterectomized before 6 weeks of age. In countries in which preadolescent neutering of domestic ferrets is not routine, such as the United Kingdom, the incidence of adrenocortical neoplasia is markedly lower. 110 Other factors hypothesized to predispose ferrets to neoplasia include inbreeding at commercial facilities, unnatural photoperiodic stimulation, and diet. 23, 98
The neoplastic cells that accumulate in the ferret adrenal after prepubertal gonadectomy functionally resemble gonadal steroidogenic cells. The ectopic production of sex steroids by these tumor cells causes a syndrome known as adrenal-associated endocrinopathy (AAE) or hyperadrenocorticism. Clinical signs of this syndrome attributable to sex steroids include bilateral symmetric alopecia, enlargement of the vulva (in jills), squamous metaplasia of prostatic ductular epithelium (in hobs), and resumption of mating behavior (in hobs). 23 The diagnosis of AAE is confirmed by documenting elevated plasma concentrations of 17α-hydroxyprogesterone, androstenedione, DHEA-S, or estradiol. 23, 99 Ferrets with AAE can also exhibit Cushingoid features, such as thin skin, muscular atrophy, and pot-bellied appearance, 23 raising the possibility that excess glucocorticoid production contributes to the syndrome. Plasma concentrations of cortisol are infrequently elevated in ferrets with AAE, 23, 99 but in some cases, the urinary corticoid : creatinine ratio is elevated and resistant to suppression by dexamethasone, 113 consistent with ACTH-independent cortisol production by the neoplastic adrenal gland.
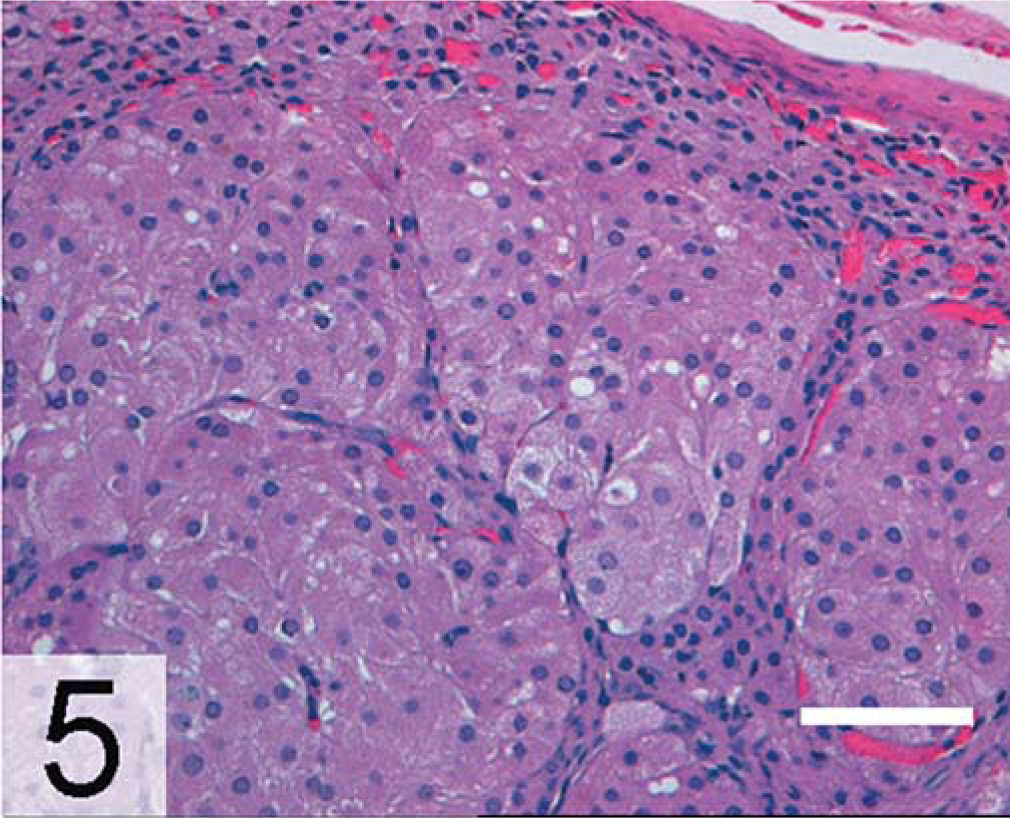
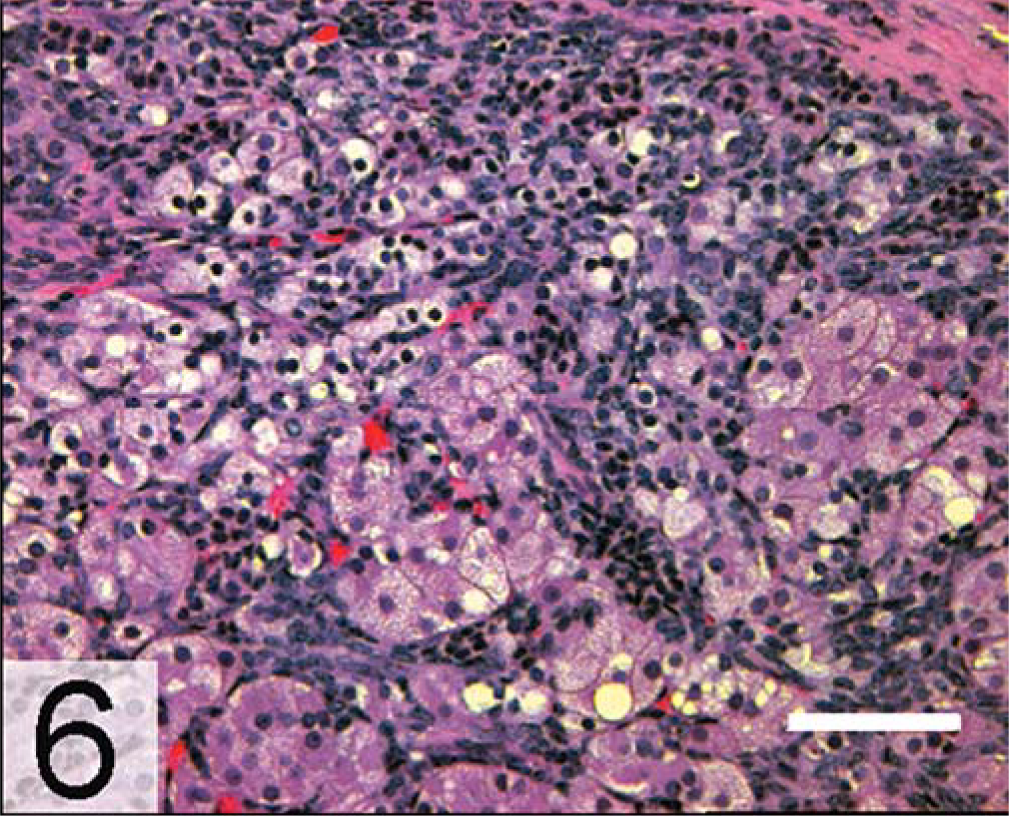
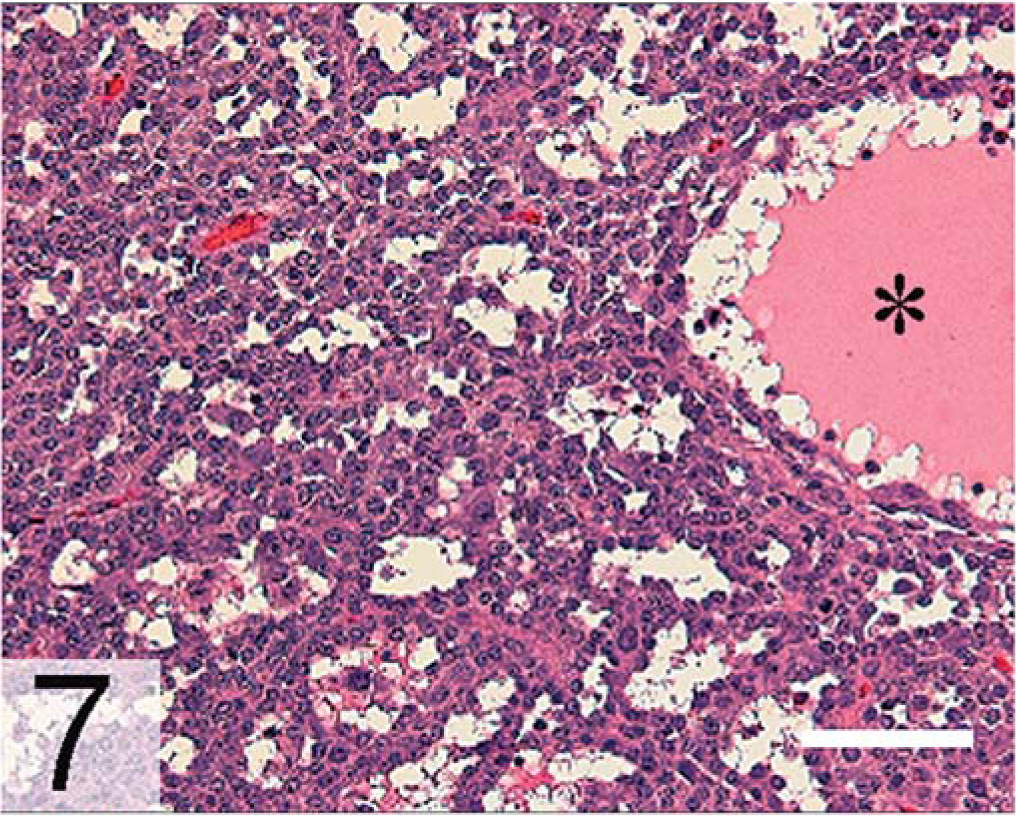
In approximately 85% of ferrets with AAE, only one adrenal gland is enlarged. 100, 128 Histologic examination might reveal nodular hyperplasia, adenoma, or carcinoma of the adrenal cortex. Adenomas are well-demarcated lesions composed mainly of polyhedral cells with small, hyperchromatic nuclei (Fig. 5). 101 Adrenocortical carcinomas are generally large, proliferative lesions that invade beyond the adrenal capsule. 101 Carcinomas often contain mixtures of small basophilic ovoid cells, large polyhedral cells, and large clear cells with small dense nuclei and vacuolated cytoplasm (Fig. 6). Anaplastic variants of adrenocortical carcinoma exhibit myxoid differentiation (Fig. 7). 85 A separate spindle cell component might be seen in benign or malignant adrenocortical tumors in ferrets (Fig. 8), although its prognostic importance is unclear. 26, 81 Unlike adjacent normal adrenocortical tissue, gonadectomy-associated tumors in ferrets express markers characteristic of gonadal steroidogenic cells. Among these markers are inhibin-α and two nuclear factors that regulate gene expression in gonadal somatic cells, transcription factor GATA-4 (Fig. 9) and estrogen receptor-α (ERα; Fig. 10). 81, 84, 85 The pathophysiologic significance of these markers is discussed below.
Adrenal gland, adenoma; neutered ferret. Note the polyhedral cells with hyperchromatic nuclei. HE. Bar = 100 µm.
Adrenal gland, adrenocortical carcinoma; neutered ferret. Note the mixture of small basophilic ovoid cells, large polyhedral cells, and clear cells with small dense nuclei and vacuolated cytoplasm. HE. Bar = 100 µm.
Liver, metastatic myxoid adrenocortical carcinoma; neutered ferret. The asterisk indicates a mucin pool. HE. Bar = 100 µm.←
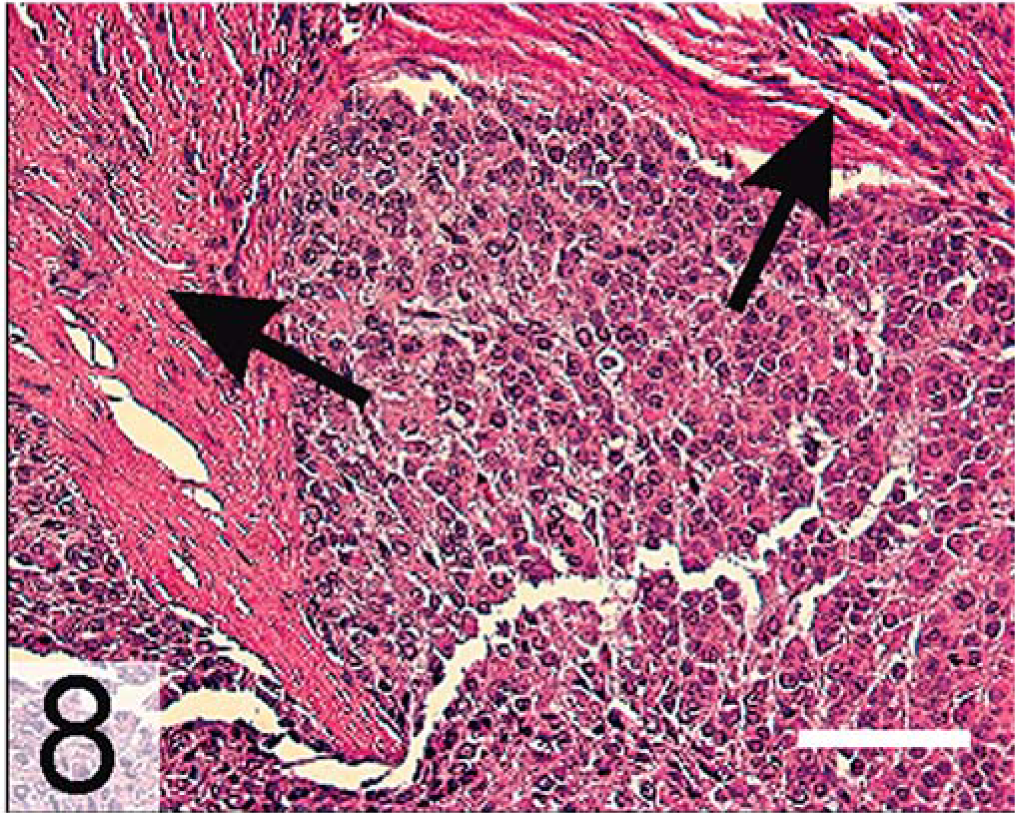
Adrenal gland, anaplastic adrenocortical carcinoma; neutered ferret. Note the spindle cell component (arrows). HE. Bar = 100 µm.
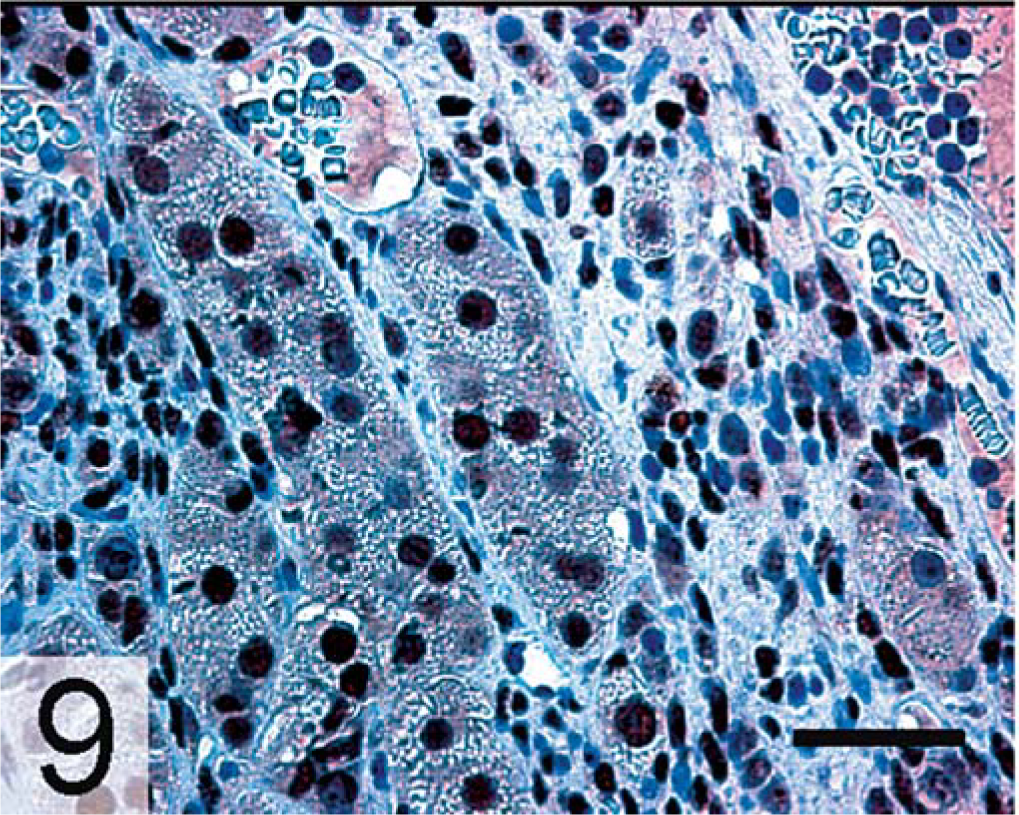
Adrenal gland, adrenocortical carcinoma; neutered ferret. Nuclear GATA-4 expression is evident in both large and small tumor cells. GATA-4, avidin–biotin complex peroxidase method. Bar = 100 µm.
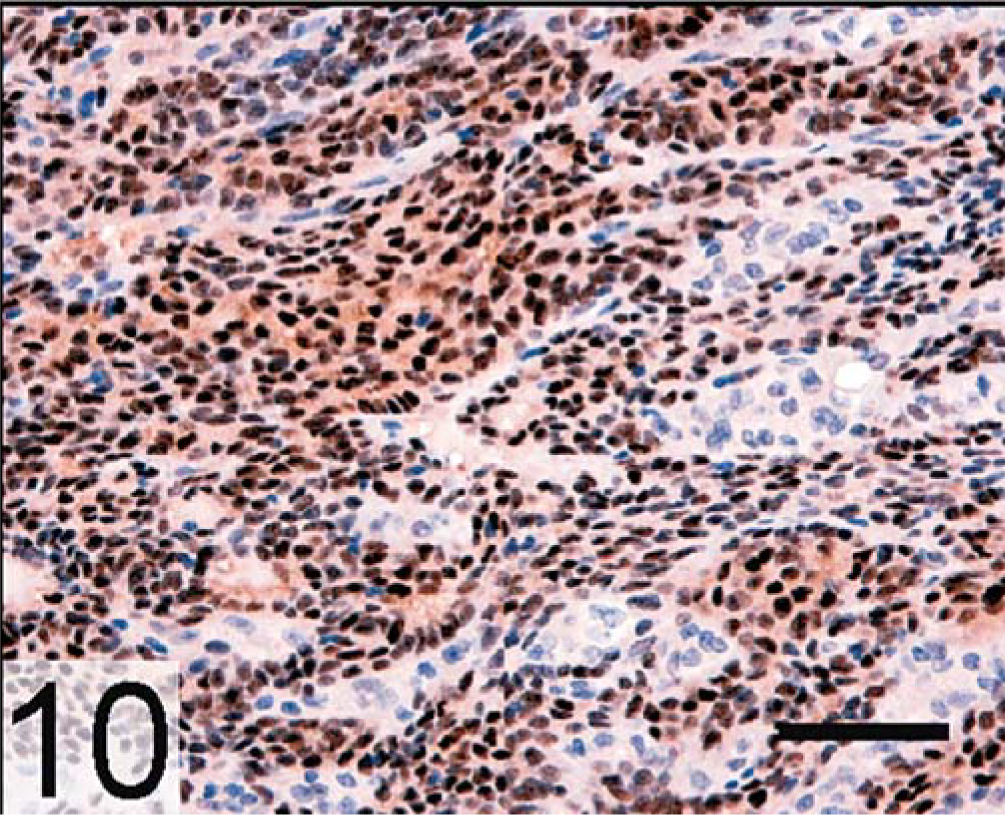
Adrenal gland, adrenocortical carcinoma; neutered ferret. Nuclear ERα expression is evident in the neoplastic cells. ERα, avidin–biotin complex peroxidase method. Bar = 250 µm.
Mouse Models of Gonadectomy-induced Adrenocortical Neoplasia
Inbred mouse strain
Prepubertal gonadectomy induces sex steroid–producing adrenocortical tumors in certain inbred mouse strains: CE mice develop carcinomas, whereas strains C3H and DBA/2J develop adenomas. 9, 21 These adrenal neoplasms arise in the subcapsular region and invade the deeper layers of the cortex, displacing normal adrenocortical tissue (Figs. 11a, b, 12a, b). The neoplasms are composed of two populations of cells: small, basophilic cells, known as A cells, and large, lipid-laden cells, termed B cells. 9, 21 Type A cells express two markers of gonadal stroma, GATA-4 and the receptor for Müllerian inhibiting substance (MIS), but lack expression of steroidogenic markers. 8, 9 The related transcription factor, GATA-6, which is found in steroidogenic cells of the normal adrenal cortex, is downregulated in the neoplastic A cells. 8, 9 Type B cells express markers of gonadal steroidogenic cells, including GATA-4, SF-1, LH receptor (LHR), inhibin-α, MIS, ERα, P450c17, and an “ovarian-specific” exon 1 variant of aromatase. 8, 9 Type B cells produce androgens, estrogens, or both. Although the terms “A cell” and “B cell” are vague and lackluster, they have been in use for 60 years and have not been supplanted by more precise or descriptive wording. Functionally, type A cells resemble stromal cells of the postmenopausal ovary, which can metabolize cholesterol into oxysterols but have limited capacity for the synthesis of steroid hormones, particularly sex steroids. 39 Type B cells resemble follicular theca cells from women with polycystic ovary syndrome, a condition characterized by chronically elevated LH levels. 43 Traditional adrenocortical cell markers, such as cell surface hormone receptors (e.g., the ACTH receptor [melanocortin-2 receptor, MC2R]) or enzymes dedicated to the synthesis of corticosterone or aldosterone (e.g., steroid 21-hydroxylase [P450c21], P450 11β-hydroxylase [P450c11, Fig. 11b, 12b], aldosterone synthase [P450aldo]), become excluded from the neoplastic tissue. 8, 9, 130
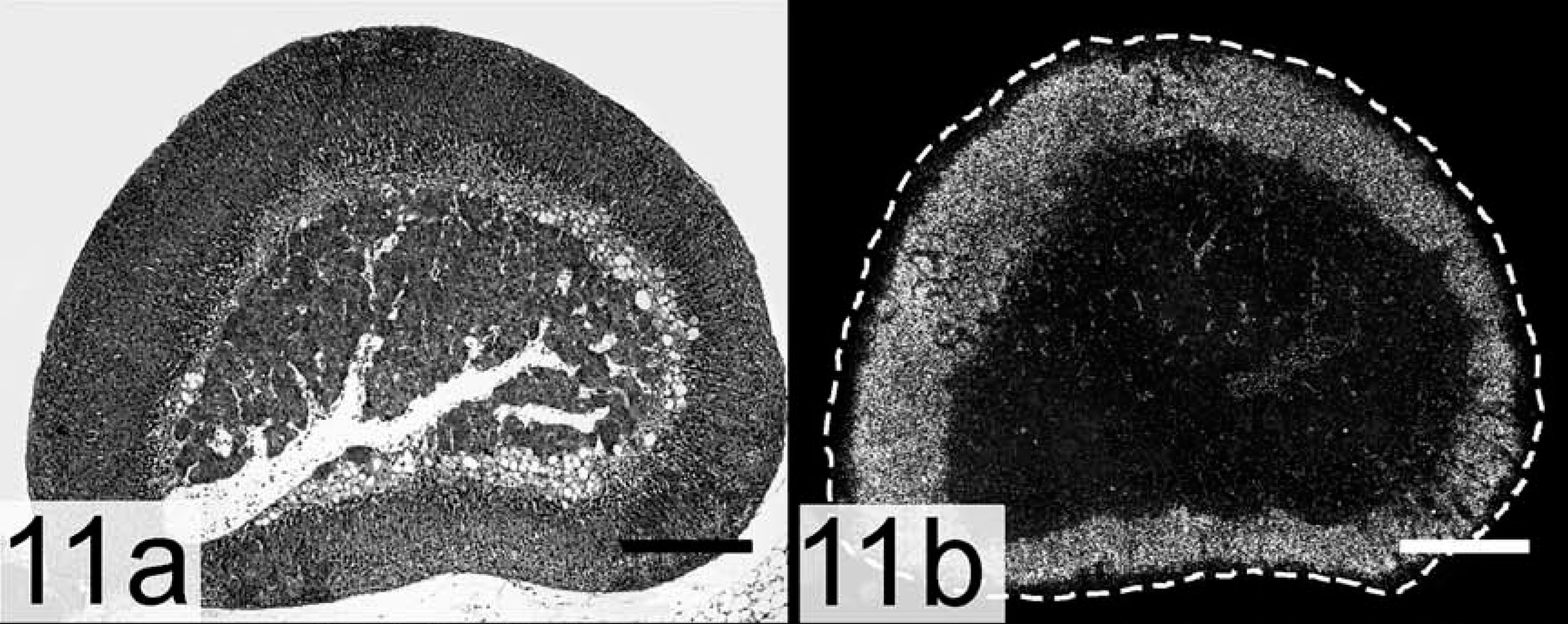
Adrenal gland; intact DBA/2J mouse. A dashed line marks the capsule. P450c11, an enzyme required for corticoid synthesis, is expressed throughout the cortex.
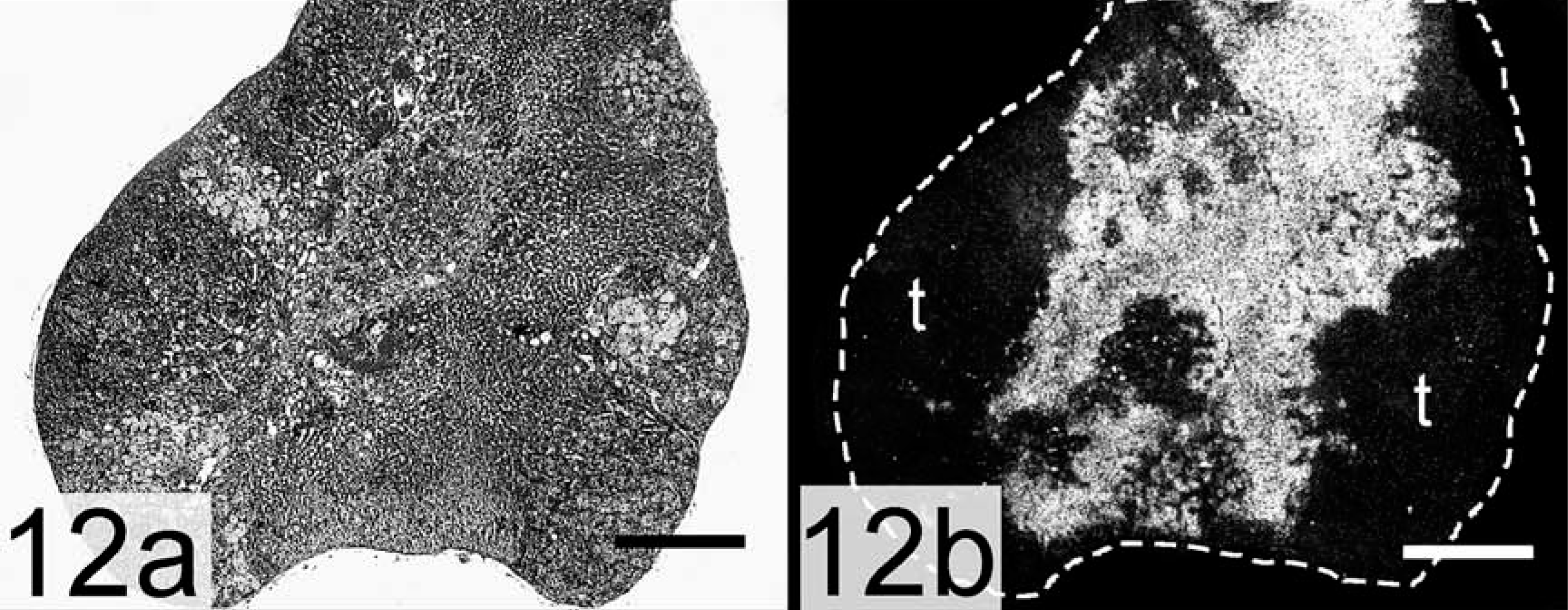
Adrenal gland, adenoma; ovariectomized DBA/2J mouse. A dashed line marks the capsule. The cortex is infiltrated by tumor that lacks the capacity to produce corticoids.
Inhibin-α promoter–SV40 T-antigen transgenic mice
Intact inhibin-α promoter–SV40 T-antigen transgenic mice develop gonadal sex cord stromal tumors. 44, 90 When these mice are subjected to prepubertal gonadectomy, they develop sex steroid–producing adrenocortical carcinomas by 5–7 months of age. These adrenocortical carcinomas generally arise in the X-zone, but subcapsular patches of type A and B cells can accompany the X-zone lesions (Fig. 13a, b). 44, 90 The molecular markers of adrenocortical neoplasia in these mice, LHR, inhibin-α, GATA-4 (Fig. 13c), and P450c17, are similar to those described in gonadectomized DBA2/J mice. 9 The adrenocortical tumors in inhibin-α promoter–SV40 T-antigen transgenic mice secrete large amounts of estrogen. 79, 91 GATA-6, a factor associated with growth inhibition in some cell types, 77 is downregulated during adrenocortical tumorigenesis in these transgenic mice (Fig. 13d). 52
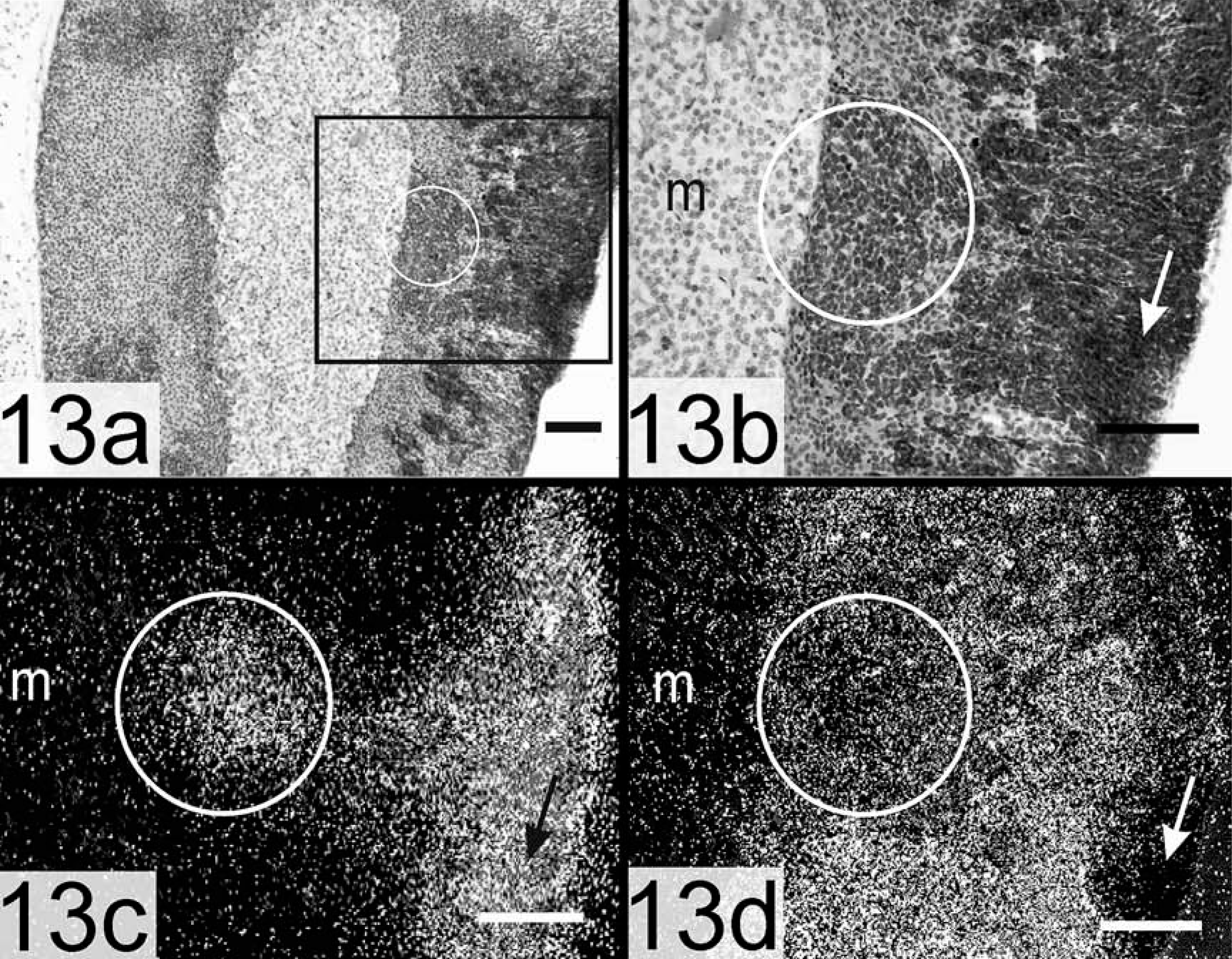
Adrenal gland, adrenocortical carcinoma; gonadectomized inhibin-α promoter–SV40 T-antigen transgenic mouse. The boxed region in panel a is shown in panel b. Subcapsular A cells (arrows) and juxtamedullary carcinoma cells (circles) express GATA-4 but little GATA-6.
Inhibin-α null mice
Mice lacking inhibin-α develop gonadal stromal cell tumors that secrete large amounts of activin, which cause a cancer cachexia-like wasting syndrome and death by 18 weeks of age. 72, 74 Gonadectomized inhibin-α null mice develop sex steroid–producing tumors of the adrenal cortex with a latency of 5–7 months. 72 As in the foregoing model, the adrenocortical tumors appear to arise from the X-zone, but subcapsular cells with histologic features of type A and B cells have been observed. Tumor development is accompanied by upregulated expression of LHR, P450c17, and aromatase, and the neoplastic cells secrete both androgen and estrogen. 5, 11
Adrenocortical Tumorigenesis is Similar in Ferrets and Mice
Salient features of adrenocortical tumor development in ferrets and each of the mouse models are compared in Table 2. The similarities are striking; in each case, gonadectomy leads to an increase in circulating LH, ectopic expression of LHR, and differentiation toward a gonadal phenotype. In ferrets, ectopic sex steroid production contributes to alopecia and other characteristic signs of AAE. In mice, the side effects of estrogen or testosterone production by the adrenal are more subtle but are present in reproductive tissues (e.g., vaginal mucosa, uterine epithelium, seminal vesicles) and nonreproductive tissues that exhibit sexual dimorphism (e.g., Bowman's capsule and submaxillary glands). 21 Thus, gonadectomy-induced adrenocortical tumorigenesis appears to follow similar, if not identical, molecular pathways in ferrets and mice.
Comparison of gonadectomy-induced adrenocortical neoplasia in domestic ferrets and three mouse models.

Controversies Regarding the Origin and Interrelatedness of Neoplastic Adrenocortical Cells
The cell of origin for gonadectomy-induced adrenocortical neoplasms in ferrets and mice remains elusive. It might be a committed gonadal progenitor that is ectopically located in the adrenal. Rare cells with morphologic features of Leydig cells have been reported in the adrenal cortex of other mammals, 64 and in gonadectomized ferrets sex steroid–producing tumors histologically indistinguishable from adrenocortical tumors have been observed at another extragonadal site, the ovarian pedicle. 83 On the other hand, the cell of origin could be a multipotential progenitor capable of differentiation into either adrenocortical or gonadal-like steroidogenic cells, depending on the hormonal milieu and other environmental factors. 83 Another theoretical, albeit less likely, possibility is that neoplastic progenitors are of extra-adrenal origin and invade the adrenal capsule by direct contact or via the blood stream. 16
An additional source of controversy is the relationship of subcapsular A and B cells in mouse adrenocortical neoplasms (or their histologic counterparts in ferret tumors—the small basophilic cell and large clear cell). Early investigators speculated that B cells might be descendants of A cells, but direct evidence of this is lacking. 21 It is equally plausible that A and B cells arise from separate progenitors. Their temporal appearance raises the possibility that type A cells produce factors that promote B cell differentiation. Stromal cells of the postmenopausal ovary synthesize growth factor–binding proteins. 39 Assuming that A cells are the functional equivalent of postmenopausal stromal cells, 39 these neoplastic cells might serve to insulate sex steroid–producing B cells from the effects of growth factors that promote differentiation into corticosteroid-producing cells. Conversely, B cells might affect the proliferation or survival of A cells through secretion of paracrine factors (see below). Lineage-tracing studies and tests of clonality are needed to definitively establish the origin and interrelationship of A and B cells.
Signaling Molecules and Pathways Implicated in Gonadectomy-induced Neoplasia
Given the similarities between neoplastic adrenocortical cells and normal gonadal somatic cells, it comes as no surprise that reproductive hormones along with their receptors and intracellular effectors play prominent roles in adrenocortical tumorigenesis in ferrets and mice (Table 3). The binding of hormones to cell surface or nuclear receptors triggers signaling cascades that affect the proliferation, differentiation, and survival of steroidogenic cells. Often these signaling pathways culminate in the binding of transcription factors to cis-regulatory elements in genes critical for cell growth or function. Hormonal signaling can also modulate the activity of cell cycle regulatory proteins.
Signaling molecules and cell cycle regulatory proteins implicated in gonadectomy-induced adrenocortical neoplasia.

Pituitary hormones
Luteinizing hormone
This dimeric glycoprotein hormone is composed of a common gonadotropin α-subunit and hormone-specific β-subunit. LH is secreted from the pituitary in a pulsatile fashion in response to a surge of gonadotropin releasing hormone (GnRH; Fig. 4). LH binds to a G-protein–coupled surface receptor, LHR, which is present on gonadal steroidogenic cells. Activation of this receptor has pleiotropic effects on cell growth, differentiation, and steroid production, which are mediated through several signaling pathways, including cAMP/protein kinase A (PKA; Fig. 14), mitogen-activated protein (MAP) kinase, and phosphatidylinositol-3-kinase. 35, 120 This in turn leads to phosphorylation/activation of transcription factors and enhanced expression of steroidogenic enzyme genes. The net effect is increased production of sex steroids, which feedback on the hypothalamic–pituitary axis and decrease LH secretion (Fig. 4). LHR has also been detected in lesser amounts on extragonadal cells, although the physiologic significance of this extragonadal LHR is uncertain. 63
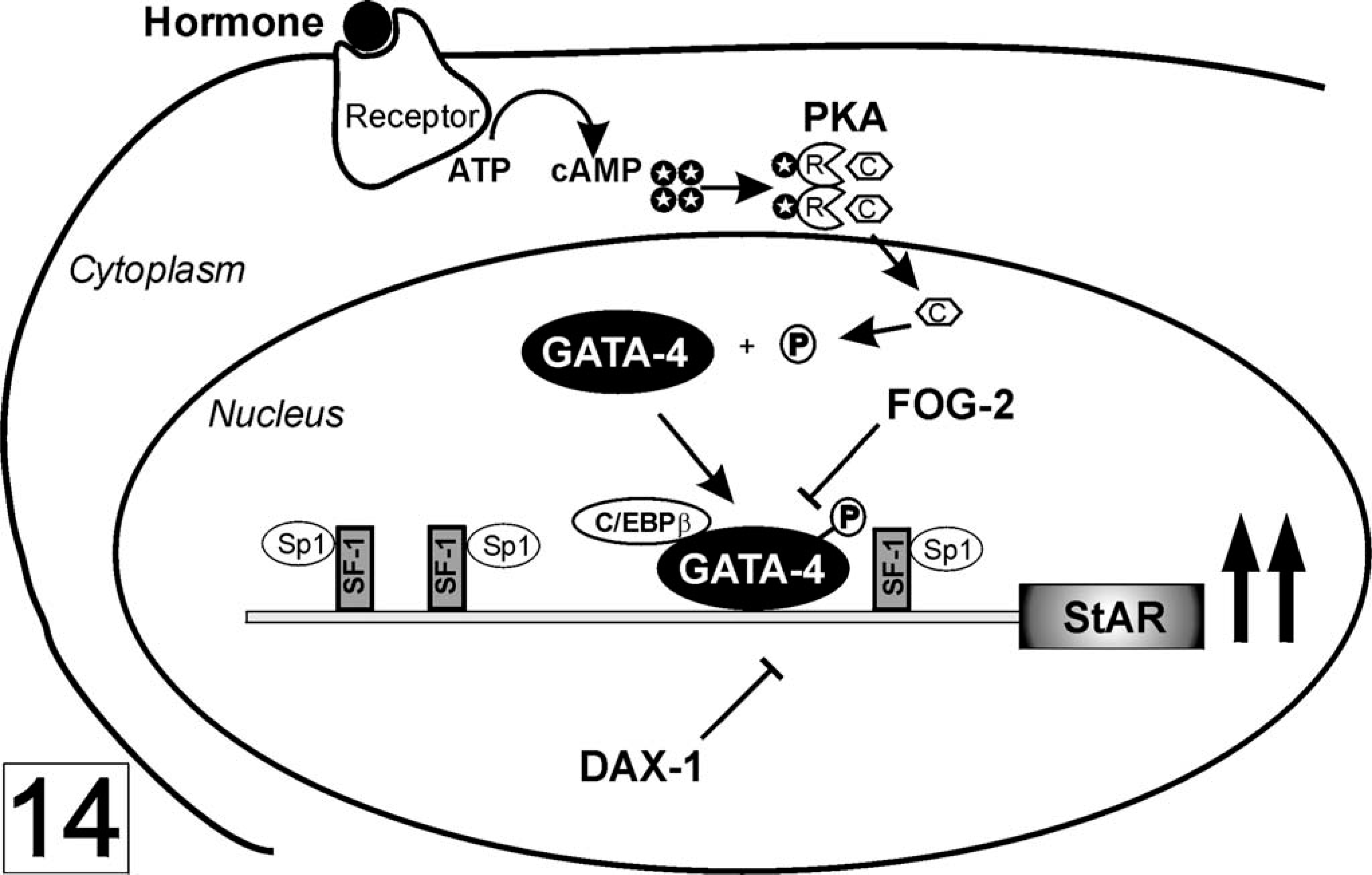
Regulation of StAR expression. The binding of LH to its receptor leads to increases in the level of cAMP (black circle with white star). Binding of cAMP to the regulatory subunit (R) of PKA allows dissociation of the catalytic subunit (C) and its translocation to the nucleus, where it phosphorylates target transcription factors, including GATA-4. SF-1 is essential for basal StAR expression and binds another widely expressed transcription factor, Sp1. C/EBPβ (CCAAT-enhancer binding protein) acts synergistically with GATA-4. DAX-1 and FOG-2 function as repressors. Modified from Manna et al. 69 and Tremblay and Viger. 120
Several lines of evidence suggest that LH signaling plays a central role in adrenocortical tumorigenesis in neutered ferrets. A highly regulated system of hormonal and physical cues ensures that LH is produced at the appropriate time and concentration in the intact ferret. 4 Gonadectomy disrupts feedback inhibition of the hypothalamic–pituitary axis, leading to increased numbers of gonadotrophs in the pars distalis adenohypophysis 112 and chronically elevated plasma LH levels. 15, 105, 107 In the mature ferret, LH levels can be modulated further by photoperiodic stimulation. 4 In neutered ferrets with adrenocortical tumors, plasma sex steroid levels are higher during the breeding season (March–August in the northern hemisphere), when prolonged daylight augments LH secretion by the pituitary. 40, 97, 111 Conversely, alopecia and other signs of AAE can remit during autumn and winter in response to the decline in LH associated with a shortening photoperiod. 23 Administration of GnRH increases plasma sex steroid levels in ferrets with adrenocortical tumors, 111 whereas inhibition of gonadotropin secretion with leuprolide acetate or deslorelin acetate can decrease sex steroid production by tumors and temporarily ameliorate clinical signs of AAE. 125, 126
Pharmacologic and genetic studies in mice support the premise that LH has a central role in gonadectomy-induced adrenocortical neoplasia. Chronic gonadotropin elevation is a prerequisite for tumor development in all three of the mouse models. Hypophysectomy abrogates gonadectomy-induced adrenocortical tumorigenesis in inbred mice. 37 Likewise, pharmacologic suppression of gonadotropin secretion with a GnRH antagonist precludes tumor development in inhibin-α promoter–SV40 T-antigen mice. 93 Gonadectomy-induced adrenocortical neoplasia is prevented by introduction of an hpg (GnRH null) mutation into either inhibin-α promoter–SV40 T-antigen transgenic mice or inhibin-α null mice. 56, 89 Studies on NU/J nude mice, another inbred strain that is susceptible to gonadectomy-induced adrenocortical neoplasia, suggest that chronic LH elevation can induce adrenocortical tumorigenesis in the absence of gonadectomy. 8 With the use of xenografts of human chorionic gonadotropin (hCG)–secreting cells, persistent gonadotropin stimulation was found to induce adrenocortical tumors in nongonadectomized NU/J nude mice.
Transgenic mice with constitutively elevated LH/hCG secretion have also been used to study the effects of chronic gonadotropin stimulation on adrenocortical function and tumorigenesis. Three such transgenic models have been generated: bLHβ-CTP mice, which express an analog of LH in females, 67 and two transgenic lines that overexpress hCG in both sexes. 34, 71, 103 Female mice that bear both the bLHβ-CTP and inhibin-α promoter–SV40 T-antigen transgenes develop adrenal tumors in the absence of gonadectomy. 75 Interestingly, nongonadectomized female inhibin-α null mice bearing a bLHβ-CTP transgene fail to develop adrenocortical carcinomas. 5 In this instance, the secretion of activin by the gonadal tumors is postulated to inhibit adrenal tumor formation by accelerating the apoptosis of cells in the X-zone. 5
For LH to have a direct effect on cells, its cognate receptor must be present. LHR is expressed in ovarian theca, granulosa, and luteal cells and in testicular Leydig cells. This receptor is also evident in the fetal but not adult adrenal of mice, a developmental expression pattern resembling those of GATA-4 and P450c17. 2, 9, 51, 90 Low-level expression of LHR mRNA and protein can be detected in the adrenal cortex of intact ferrets, but these receptors appear to be nonfunctional. 86, 111 Adrenocortical tumorigenesis in mice and ferrets is accompanied by dramatic and sustained increases in the levels of LHR mRNA and protein. 84, 111 This differs from the situation that exists in most LH-responsive cells, wherein chronic exposure to LH results in downregulation or desensitization of its receptor. The mechanism responsible this paradoxical upregulation of LHR in the neoplastic tissue is unknown. It is conceivable, albeit unproven, that the subcapsular or juxtamedullary regions contain rare cells that express functional LHR and transform or proliferate in response to chronic gonadotropin stimulation. Another possibility is that adrenocortical tumor formation is accompanied by increased expression of GATA-4, which then activates transcription of LHR in a feed-forward amplification loop. 90 Concomitant with the upregulation of LHR in sex steroidogenic adrenocortical tumor cells, the ACTH receptor, MC2R, is downregulated. 9 Thus, a switch in signaling accompanies adrenocortical tumorigenesis; the neoplastic tissue responds to LH but is no longer responsive to ACTH.
Prolactin
Another pituitary hormone, prolactin, can affect steroidogenesis in gonadal and adrenocortical cells of some species by modulating LHR expression. 18, 47, 68 In bLHβ-CTP mice, prolactin appears to enhance LHR expression in normal adrenocortical cells, leading to expansion of the zona fasciculata and excess corticosterone production, which in turn is associated with retroperitoneal fat deposition and elevated serum leptin levels. 47 Prolactin can potentiate LH-induced androgen production in ovarian interstitial cells. 66, 82 Given the established role of LHR in gonadectomy-induced adrenocortical tumorigenesis, it is tempting to speculate that changes in prolactin influence either tumor development or ectopic sex steroid production in ferrets. Like LH, prolactin secretion in ferrets is controlled by complex hormonal and physical cues (e.g., photoperiod) that are disrupted by gonadectomy. 106 After neutering, 30–40% of the adenohypophyseal cells in healthy ferrets exhibit prolactin immunoreactivity. 112 In female ferrets, prolactin has been shown to mediate the induction of puberty by long days. 106 There is no direct evidence, however, that gonadectomy-induced changes in prolactin affect adrenocortical tumor formation in ferrets.
Factors produced by gonadal and neoplastic adrenocortical tissue
Activins and inhibins
Activin and inhibin are dimeric glycoproteins belonging to the TGFβ superfamily. They function as endocrine hormones or paracrine factors that act within the pituitary, gonads, and other tissues, including the adrenal glands. 6, 17, 94 These signaling molecules can elicit various effects on the growth and function of steroidogenic cells, depending on the cell type, developmental stage, receptors present, and the overall hormonal milieu. Activin promotes the differentiation of granulosa cells, but in theca cells it inhibits LH-dependent androgen production. 17, 122
A variety of gonadal and extragonadal cell types produce activin. In normal mammals, the serum levels of activin do not fall significantly after orchiectomy or ovariectomy, suggesting that serum levels of this factor mainly reflect extragonadal production. 61 Nevertheless, local concentrations of activin might change in response to gonadectomy. Activin uses the TGFβ signaling pathway, which includes transmembrane receptor kinases and their downstream mediators, the Smad proteins. 115 Activin receptors are expressed in the murine adrenal gland, 6 and activins have been shown to inhibit adrenocortical cell growth and steroid production and to enhance apoptosis of X-zone cells. 5, 56 Activin secreted by tumorous adrenals in gonadectomized inhibin-α–deficient mice inhibits carcinoma formation in the contralateral adrenal, possibly through effects on the X-zone. 6
Inhibins are produced primarily by gonadal somatic cells. Consequently, gonadectomy leads to a profound and rapid fall in circulating inhibin levels. Inhibins have no direct effect on steroidogenesis or cell survival but can antagonize activin signaling by competing for activin receptors. 122 The synthesis of inhibins also limits the availability of βA- and βB-subunits for activin synthesis. 122 Although studies of inhibin-α null mice have established that this gene might function as a tumor suppressor, the inactivation of this locus is not essential for adrenocortical tumorigenesis. Indeed, robust expression of inhibin-α is evident in adrenocortical tumors from inbred mice, 9 ferrets, 85 and humans. 122
Müllerian inhibiting substance
MIS, also known as anti-Müllerian hormone, is a distinctive member of the TGFβ superfamily. Whereas most factors in this family have a range of functions in different organs, MIS acts only on reproductive organs. MIS is produced by gonadal somatic cells but not by normal adrenocortical cells. In the testis, MIS is produced in Sertoli cells, whereas in the ovary, it is secreted by granulosa cells. Binding of MIS to its receptor, MISIIR (MIS receptor type II), initiates downstream signaling via Smad proteins, analogous to the activin signaling pathway. 25, 124 MIS causes Müllerian duct regression during sexual differentiation and regulates postnatal Leydig cell development through its effects on cell proliferation and gene expression. 131 MIS decreases the expression of P450c17 in Leydig cells and aromatase in granulosa cells. 13, 88 Male MIS-deficient mice are predisposed to Leydig cell hyperplasia, suggesting that MIS can inhibit Leydig cell proliferation. 73 In rats, MIS inhibits rat adult Leydig cell regeneration after ethylene dimethanesulfonate ablation. 108
Circumstantial evidence suggests a possible role for MIS in gonadotropin-induced adrenocortical neoplasia. MIS immunoreactivity has been observed in sex steroid–producing adrenocortical neoplasms of the ferret. 83 In the gonadectomy-induced adrenocortical neoplasms that arise in inbred mice, MIS immunoreactivity localizes to A and B cells, whereas transcripts encoding MISIIR are found exclusively in A cells. 8 It is tempting to speculate that MIS, acting in an autocrine or paracrine fashion, can inhibit proliferation or steroidogenesis in A cells, facilitate their centripetal migration, or both. Genetic studies suggest that inhibin-α and MIS exert a synergistic effect on tumorigenesis in gonads. In inhibin-α null mice, the absence of MIS causes earlier onset and more aggressive development of testicular tumors. 73 Whether the loss or overexpression of MIS can affect gonadectomy-induced adrenocortical tumorigenesis is unknown.
Wnt proteins
Wnt proteins have been implicated in a variety of normal and pathologic developmental processes, including neoplasia. 87 Members of the Wnt family bind to Frizzled receptors on the cell surface and initiate a cytoplasmic signaling cascade that results in the stabilization of β-catenin and its subsequent translocation to the nucleus. 87 Once in the nucleus, β-catenin can enhance gene expression through interactions with various transcription factors or coactivators, including TCF/LEF family members, Smad factors, and CREB-binding protein (CBP). Wnt-2b is expressed in subcapsular cells of the adrenal 62 and Wnt-4 in the zona glomerulosa. 30 Targeted mutagenesis of Wnt4 leads to aberrant development of both the adrenal cortex and gonads; Wnt4 null mice have decreased expression of P450aldo in the adrenal and ectopic expression of the adrenal-specific marker P450c21 in gonadal tissue. 30, 41 In the adrenal glands of gonadectomized inbred NU/J nude mice, β-catenin immunoreactivity has been observed in the nuclei of B cells (and to a lesser extent A cells), suggesting that the Wnt signaling pathway might participate in adrenocortical tumorigenesis. 8 β-catenin has been shown to synergize with SF-1 in the transcriptional activation of inhibin-α in steroidogenic cells, which could account for the robust expression of this gene in type B cells. 28
Sex steroids
In mouse models of gonadectomy-associated adrenocortical neoplasia, the administration of sex steroids can inhibit tumor formation or ectopic sex steroid production. 56, 92 Besides decreasing the number of pituitary gonadotrophs and suppressing pituitary gonadotropin secretion (Fig. 4), sex steroids can directly inhibit steroid production by normal or neoplastic adrenocortical cells. 42, 65 In the setting of elevated LH/hCG and testosterone levels, as is the case in intact NU/J mice bearing hCG-secreting xenografts, neoplastic cells accumulate in the adrenal cortex, but these cells express little P450c17, suggesting that they have limited capacity for sex steroid production. 8 Expression of ERα in adrenocortical tumors of both mice 8 and ferrets 81 (Fig. 10) supports the notion that the neoplastic steroidogenic cells can respond directly to estrogen.
Transcription factors
GATA transcription factors and their coactivators
GATA factors are a group of DNA-binding proteins that recognize the consensus motif (A/T)GATA(A/G). One member of this family, GATA-4, is expressed in the genital ridge and in gonadal somatic cells of fetal and adult mammals. 29, 48, 123 This transcription factor and its putative target gene, P450c17, are also transiently expressed in the fetal adrenal of the mouse but are absent from the adrenal cortex of postnatal animals. 46, 51, 84 In gonadal steroidogenic cells of rodents, Gata4 is expressed during periods of active proliferation 29, 49, 120 and can be induced by gonadotropins. 52
Ectopic expression of GATA-4 in the adrenal heralds the appearance of neoplastic cells. 9, 84 In the ferret adrenal, GATA-4 immunoreactivity can be seen in both small and large neoplastic cells (Fig. 9) and seems to correlate with the degree of nuclear and cytoplasmic atypia. 84 GATA-4 is particularly abundant in anaplastic myxoid adrenocortical carcinomas but is absent from the spindle cell component of adrenocortical tumors. 84 In the mouse models of gonadectomy-induced adrenocortical neoplasia, GATA-4 is expressed in A cells, B cells, and juxtamedullary carcinoma cells (Fig. 13c). 9
GATA-4 is a transcriptional activator of genes involved in sex steroid biogenesis, such as the genes for the steroidogenic acute regulatory protein 32 (STAR; Fig. 14), P450c17, 22 and aromatase. 116, 119 In addition, GATA-4 has been shown to drive expression of the inhibin-α and -β subunit genes 19, 119 as well as LHR. 90 Studies of genetically engineered mice suggest that fully functional GATA-4 is required for the proper differentiation of steroidogenic cells in the fetal testis. 117 Several posttranslational modifications can modulate the activity of GATA-4. For example, phosphorylation of GATA-4 by PKA or MAP kinase enhances its ability to activate steroidogenic enzyme genes. 120 Sumoylation of GATA-4 limits cooperative interactions between this factor and others that compose transcriptional complexes. 55 In addition, there is evidence that GATA-4 acts as an antiapoptotic factor in various cell types, including gonadal steroidogenic cells. 53 In neoplastic cells, upregulation of this factor can lead to enhanced expression of “gonadal-specific” genes harboring GATA binding sites in their promoters. Among the upregulated genes, LHR could play an important role in the phenotypic switch from ACTH responsiveness to LH responsiveness and sex steroid production. Thus, GATA-4 is presumed to play a role in the induction, maintenance, or both of the neoplastic phenotype.
Another member of the GATA family, GATA-6, is expressed in steroidogenic cells of the testis, ovary, fetal adrenal, and adult adrenal. On the basis of its tissue distribution and in vitro studies, GATA-6 appears to regulate steroidogenesis in adrenal and gonads rather than merely gonads, as is the case with GATA-4. 22, 59, 120 In addition to activating genes critical for the differentiation and function of steroidogenic cells, 70, 109, 120 GATA-6 might limit cellular proliferation by modulating the expression of cell cycle regulatory proteins. 77 Increased expression of GATA-6 correlates with growth arrest and upregulation of the cell cycle inhibitor p21Cip1 in various nonsteroidogenic cell types. 77 Adrenocortical tumor development in inbred mice and inhibin-α promoter–SV40 T-antigen transgenic mice is accompanied by a decrease in GATA-6 in subcapsular A cells (Fig. 13d) and a contemporaneous increase in GATA-4 expression in these same cells (Fig. 13c). 52 Gata6 expression persists in B cells, underscoring its importance in sex steroidogenesis. The mechanisms underlying the reciprocal changes in GATA-4 and GATA-6 in A cells are unknown; studies on knockout embryonic stem cells suggest that GATA-4 might directly or indirectly inhibit GATA-6 expression. 78
GATA factors often execute their activity through interactions with the friend-of-GATA (FOG) family of coactivators or corepressors. 1, 95 One member of the FOG family, FOG-2, is essential for steroidogenic cell differentiation in the testis, 117 and Fog2 is expressed in neoplastic B cells that accumulate in the adrenal of gonadectomized inbred mice. 8 Another coactivator, CBP, can also augment the activity of GATA factors. 36 . CBP serves a critical role in the activation of cAMP-inducible promoters or enhancers, which typify most steroidogenic enzyme genes.
SF-1 and DAX-1
SF-1 is an orphan nuclear receptor expressed in steroidogenic cells of both the adrenal cortex and gonads. 135 SF-1–deficient mouse embryos exhibit aberrant adrenal and gonadal development, 135 and haplo-insufficiency for this factor impairs the compensatory adrenocortical cell growth that follows unilateral adrenalectomy. 7 SF-1 activates a wide array of genes involved in both adrenal and gonadal steroidogenesis. This transcription factor cooperates with GATA-4, GATA-6, or both in the activation of genes expressed in gonads, including Mis and genes encoding steroid biosynthetic enzymes (Fig. 14). 118, 135 Similarly, SF-1 cooperates with GATA-6 in the transcriptional activation of genes in normal adrenocortical tissue. 59 Like GATA-4, SF-1 undergoes posttranslational modifications that modulate its activity, including phosphorylation and sumoylation. 55 Because SF-1 is expressed in steroidogenic cells found in both normal and neoplastic tissue, it is not useful clinically as a specific marker of gonadectomy-induced adrenocortical neoplasia. In light of its established role in gonadal cell steroidogenesis, SF-1 probably is an essential factor in the production of ectopic sex steroids by the adrenocortical tumors. The absence of SF-1 from type A cells suggests that this transcription factor might not be essential for the induction of tumorigenesis but rather is essential for maintenance of the steroidogenic cell phenotype.
The orphan nuclear hormone receptor DAX-1 has an expression pattern resembling that of SF-1. 38 DAX-1 binds to SF-1 in vitro and functions as a repressor of steroidogenesis, generally suppressing the same genes that SF-1 activates (Fig. 14). 58, 134 In Dax1 knockout mice, the adrenal cortex develops normally and produces steroid hormones, but X-zone degeneration is impaired. 132, 133 DAX-1 is downregulated in the neoplastic cells that accumulate in the adrenal cortex of gonadectomized inbred mice. 8
Cell cycle regulatory proteins
There is evidence that adrenocortical tumorigenesis involves changes in cell proliferation, which are controlled by cyclins and their associated cyclin-dependent kinases (CDKs). 114 Cyclins bind and activate CDKs and these cyclin–CDK complexes phosphorylate critical intracellular substrates, such as the product of the retinoblastoma tumor suppressor gene, thereby facilitating cell cycle progression. 114 Mice bearing null mutations in both inhibin-α and Ccnd2, which encodes cyclin D2, develop gonadal and adrenocortical tumors more slowly than inhibin-α null mice. 10 Thus, cyclin D2 can antagonize the suppressive effects of inhibin on adrenocortical tumorigenesis. In inhibin-α null mice, the absence of the CDK inhibitor p27Kip1 causes earlier onset and more aggressive gonadal tumors. 10, 12 However, the loss of p27Kip1 has little effect on adrenocortical tumor progression in gonadectomized inhibin-α null mice. As noted above, increased expression of GATA-6 correlates with growth arrest, downregulation of cyclin A, and the upregulation of another CDK inhibitor, p21Cip1, in various nonsteroidogenic cell types. 77 Of note, this same correlation has not been observed in transplanted adrenal tissue or human adrenocortical tumors. 50
Genetic Basis of Strain Susceptibility to Gonadectomy-induced Adrenocortical Neoplasia
Adrenocortical neoplasms develop in only a minority of neutered ferrets, suggesting that genetic changes, perhaps inadvertently selected through inbreeding, influence susceptibility of adrenal tissue to neoplastic transformation. The same is true in mice, as borne out by the inbred and genetically engineered mouse models of adrenocortical neoplasia. Studies of inhibin-α promoter–SV40 T-antigen mice demonstrate that forced expression of a potent oncogene in the adrenal can alter tissue susceptibility to gonadectomy-induced tumor formation. Similarly, the phenotype of inhibin-α null mice shows that decreased expression of a tumor suppressor gene (inhibin-α), presumably in adrenal tissue per se, can affect susceptibility. Organ transplantation experiments with inbred mice suggest that strain susceptibility to gonadectomy-induced neoplasia resides in adrenal tissue; prepubertal adrenal glands transplanted from a susceptible strain into a gonadectomized nonsusceptible strain still develop tumors. 37, 121 However, the genetic changes (in the transplanted tissue) that account for strain-specific differences in susceptibility have not been elucidated.
Inbred mouse strain susceptibility to gonadectomy-induced adrenocortical tumorigenesis appears to correlate with the in vivo and in vitro capacity of normal gonadal tissue to produce steroid hormones in response to gonadotropin stimulation. In general, “low-steroid” strains (e.g., CE, DBA/2J), which exhibit poor fertility, are susceptible to gonadectomy-induced tumorigenesis, whereas “high-steroid” strains (e.g., C57BL/6), which display superior reproductive performance, are resistant to gonadectomy-induced tumorigenesis. 9 Intriguingly, a polymorphism in the Sf1 coding sequence appears to discriminate between low- and high-steroid strains. 24 Although the physiologic consequence of this polymorphism is not yet known, such a mutation could alter the capacity of SF-1 to regulate adrenal versus gonadal-type lineage specification, either directly or through synergistic interactions with other transcription factors. Alterations in the level of SF-1 have been shown to disrupt compensatory adrenal growth response to unilateral adrenalectomy, 7 so an inhibitory SF-1 mutation might preferentially affect adrenal cell proliferation and shift the balance in favor of ectopic gonadal cell differentiation or proliferation.
It seems plausible that inherited genetic changes can alter the sensitivity of adrenal tissue to reproductive hormones or paracrine factors like LH and activin. Such changes might enhance cell proliferation or survival and thereby predispose cells to acquire additional (i.e., secondary) mutations that activate oncogenes or inhibit tumor suppressor genes. The genetically engineered mouse models of gonadectomy-induced tumorigenesis lend credence to such secondary events. Cells derived from adrenocortical tumors that arise in inhibin-α null mice are unresponsive to activin in vitro. 6 This acquired property might explain how these cells are able to proliferate and secrete sex steroids despite high local concentrations of activin.
Ambiguities in Adrenocortical Tumor Classification Confound the Discussion of Pathogenesis
When categorizing adrenocortical lesions, it can be difficult to separate cell proliferation, often termed “hyperplasia,” from so-called “adenomas.” Traditionally, pathologists have used the term “hyperplasia” to describe localized replacement of cortical tissue by abnormal cells, with an increase in the width of the cortex but minimal extension beyond the capsule. When the proliferation of the abnormal tissue is more extensive and localized, the term “adenoma” has been used. Actually no sharp distinctions can be made between the two. In the mouse, both types of lesions will progress and eventually become transplantable neoplasms provided the animal lives long enough, 21 and the same could hold true for the ferret. Alternatively, the word “metaplasia” has been used to describe the transformation of adrenocortical tissue into cells resembling gonadal stroma. 9 Although each of these terms has merit, we have elected to discuss gonadectomy-induced adrenocortical lesions under the rubric of “neoplasm,” acknowledging that tumorigenesis might be a multistep process that begins as hyperplasia or metaplasia.
Human Adrenocortical Tumors
As in ferrets and mice, adrenocortical tumor formation in humans appears to be influenced by both the hormonal milieu and the inherent susceptibility of the adrenal gland. Subcapsular adrenocortical neoplasms with histologic features resembling luteinized ovarian stroma (“thecal metaplasia”) have been reported in postmenopausal women 20 and men with acquired testicular atrophy, 96 suggesting that the hormone changes associated with gonadal failure (e.g., elevated LH, decreased sex steroids, etc.) might contribute to tumor development in humans. Analyses of heritable and spontaneous types of human adrenocortical tumors have documented alterations in either cell surface receptors or their downstream effectors that make adrenocortical tissue more susceptible to hormone-associated neoplastic transformation (Table 4). 54, 57 In a majority of cases, these genetic alterations lead to adrenocortical neoplasms that produce excess glucocorticoids, but in a few cases, notably those involving young children, the adrenocortical tumors produce large amounts of sex steroids and inhibin-α, like the tumors in gonadectomized ferrets and mice. Whether such genetic changes contribute to tissue susceptibility to neoplasia in neutered ferrets or mice awaits further study. The inbred mouse models, coupled with the rapidly expanding database of genomic DNA sequences and single nucleotide polymorphisms in various strains, afford a means to characterize the alleles and genetic modifiers influencing gonadectomy-induced adrenocortical neoplasia. In addition, these models will facilitate studies of the signaling pathways that control cell fate decisions in normal and neoplastic steroidogenic lineages.
Genetic alternations observed in heritable and spontaneous types of human adrenocortical tumors.

Footnotes
Acknowledgements
We thank Drs. Richard Peterson and Charles Capen (The Ohio State University) and Dr. Matti Kiupel (Michigan State University) for providing tumor specimens from neutered ferrets. We thank Drs. Nafis Rahman (University of Turku) and Ilpo Huhtaniemi (Imperial College London) for providing tissue sections of gonadectomized inhibin-α promoter–SV40 T-antigen transgenic mice. Rosie Wilson provided technical assistance. The Finnish Pediatric Research Foundation; Juselius Foundation; NIH HL61006; and DK52574, MOD 1-FY02-203, and AHA 0455623Z supported this work.
