Abstract
Adrenocortical neoplasms are a common cause of morbidity in neutered ferrets. Recently we showed that gonadectomized DBA/2J mice develop adrenocortical tumors that express transcription factor GATA-4. Therefore, we screened archival specimens of adrenocortical neoplasms from neutered ferrets to determine whether GATA-4 could be used as a tumor marker in this species. Nuclear immunoreactivity for GATA-4 was evident in 19/22 (86%) of ferret adrenocortical carcinomas and was prominent in areas exhibiting myxoid differentiation. Normal adrenocortical cells lacked GATA-4 expression. Two other markers of adrenocortical tumors in gonadectomized mice, inhibin-α and luteinizing hormone receptor, were coexpressed with GATA-4 in some of the ferret tumors. No GATA-4 expression was observed in three cases of nodular hyperplasia, but patches of anaplastic cells expressing GATA-4 were evident in 7/14 (50%) of tumors classified as adenomas. We conclude that GATA-4 can function as a marker of anaplasia in ferret adrenocortical tumors.
Keywords
Adrenocortical adenomas and carcinomas occur frequently in ferrets, especially those neutered at a young age. 3,7,10 Often these tumors produce sex steroids that result in pathophysiologic changes such as alopecia, vulvar hypertrophy, or squamous metaplasia of prostatic ductular epithelium. 4,8 Adrenocortical neoplasia in gonadectomized ferrets is theorized to be due to chronic stimulation by luteinizing hormone (LH). 9,11 Pharmacologic inhibition of gonadotropin production can reduce ectopic sex steroid production and ameliorate clinical signs in affected animals. 11 In addition to neoplastic epithelial cells, ferret adrenocortical tumors often contain a spindle cell component that expresses smooth muscle actin. 2 An aggressive variant of adrenocortical carcinoma has been described that exhibits inhibin-α expression and prominent mucin production, interpreted as myxoid differentiation. 6
Recently we showed that GATA-4, a transcription factor normally expressed in somatic/interstitial cells of the ovary and testes but not in cells of the adult adrenal gland, is expressed in sex steroid–producing tumors that accumulate in the adrenal cortex of DBA/2J mice after gonadectomy. 1,5 Besides GATA-4, these neoplastic cells express inhibin-α and LH receptor (LHR). 1 Based on these observations in mice, we hypothesized that changes in GATA-4 expression might accompany adrenocortical tumorigenesis in neutered ferrets.
We performed immunohistochemistry on surgical biopsy and necropsy specimens of ferret adrenocortical neoplasms obtained from archives of The Ohio State University Department of Veterinary Biosciences (24 cases), the Michigan State University Animal Health Diagnostic Laboratory (15 cases), and the Washington University Department of Pediatrics (1 case). 6 All of these ferrets had been gonadectomized. As controls, we examined normal adrenal tissue specimens from intact and gonadectomized adult ferrets. Formalin-fixed, paraffin-embedded tissues were sectioned (5 µm) for hematoxylin and eosin staining or immunohistochemistry. 6 The following primary antibodies were used: 1) GATA-4 (#sc-1237, Santa Cruz Biotechnology, Santa Cruz, CA), 2) inhibin-α (Serotec, Inc., Raleigh, NC), or 3) LHR (courtesy of I. Huhtaniemi, University of Turku, Finland). Sections were exposed to the antibody at dilutions of 1 : 200 for 30 minutes at room temperature or overnight at 4 C. An avidin–biotin–immunoperoxidase system (Vectastain Elite ABC Kit, Vector Laboratories, Burlingame, CA) and diaminobenzidine (Sigma Chemicals, St. Louis, MO) were used to visualize the bound antibody; slides were counterstained with 100% hematoxylin.
Our retrospective analysis included cases of anaplastic adrenocortical carcinoma (18), well-differentiated adrenocortical carcinoma (5), adenoma (14), and nodular hyperplasia (3). Histologic criteria for classification of these tumors are given elsewhere. 6 Several of these neoplasms contained residual normal cortex and four had direct hepatic invasion or metastasis. Consistent with observations on other mammalian species, 1 there was a lack of GATA-4 expression in normal adrenocortical cells of the intact or gonadectomized adult ferret (not shown). On the other hand, nuclear immunoreactivity for GATA-4 was observed in ferret adrenocortical carcinomas (Fig. 1). GATA-4 immunostaining was particularly robust in anaplastic adrenocortical carcinoma cells, including those exhibiting myxoid differentiation or metastasis to the liver (Fig. 2). Positive staining also was evident in well-differentiated adrenocortical carcinomas. Overall, 19/22 (86%) of neoplasms classified morphologically as adrenocortical carcinomas expressed GATA-4, and there were no sex differences in expression of this marker. Minimal to no nuclear GATA-4 staining was present in the spindle cell component of the tumors. In individual adrenocortical carcinomas, cells staining positively for GATA-4 also displayed nuclear atypia (Fig. 3a, b)) and cytoplasmic inhibin-α immunoreactivity (Fig. 4a, b)). Moreover, cytoplasmic LHR was coexpressed with GATA-4 in some of the tumor cells (Fig. 5a, b)).
Adrenal gland, adrenocortical carcinoma; ferret. The densely arranged neoplastic cells that express nuclear GATA-4 are reminiscent of cells seen in gonadectomized DBA/2J mice.
1
GATA-4, avidin–biotin–peroxidase complex method. Bar = 100 µm.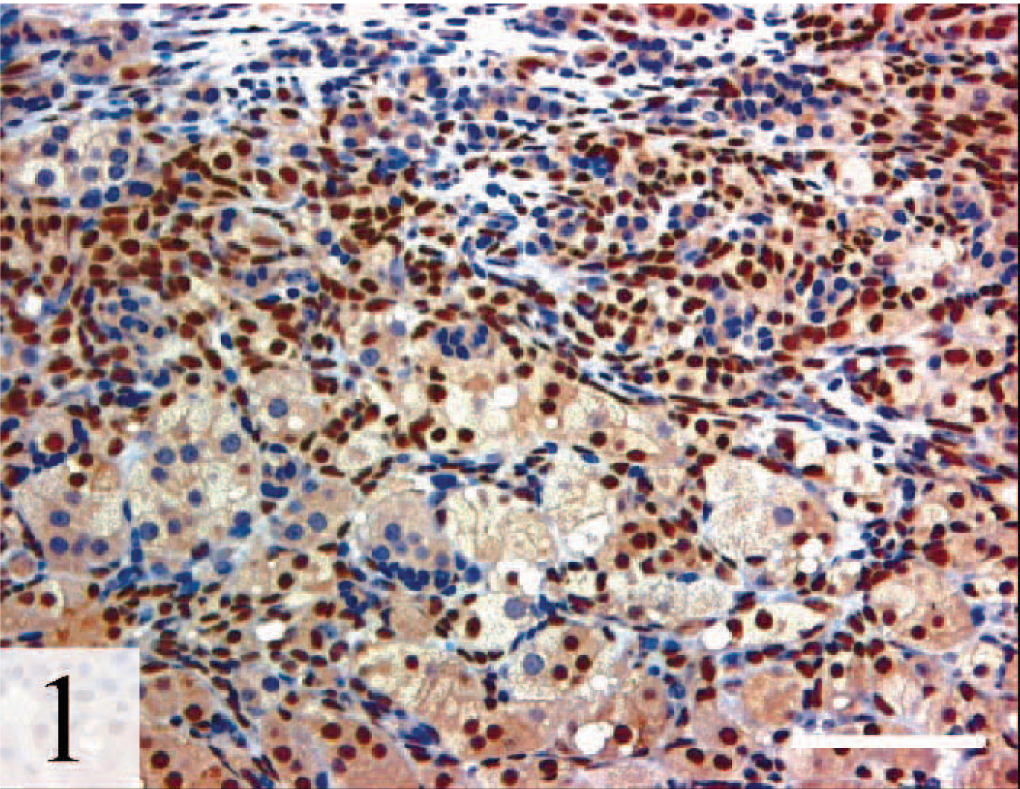
Liver, metastatic myxoid adrenocortical carcinoma; ferret. Note the presence of mucin pools. Strong nuclear GATA-4 expression is evident in these anaplastic cells exhibiting myxoid differentiation. GATA-4, avidin–biotin–peroxidase complex method. Bar = 100 µm.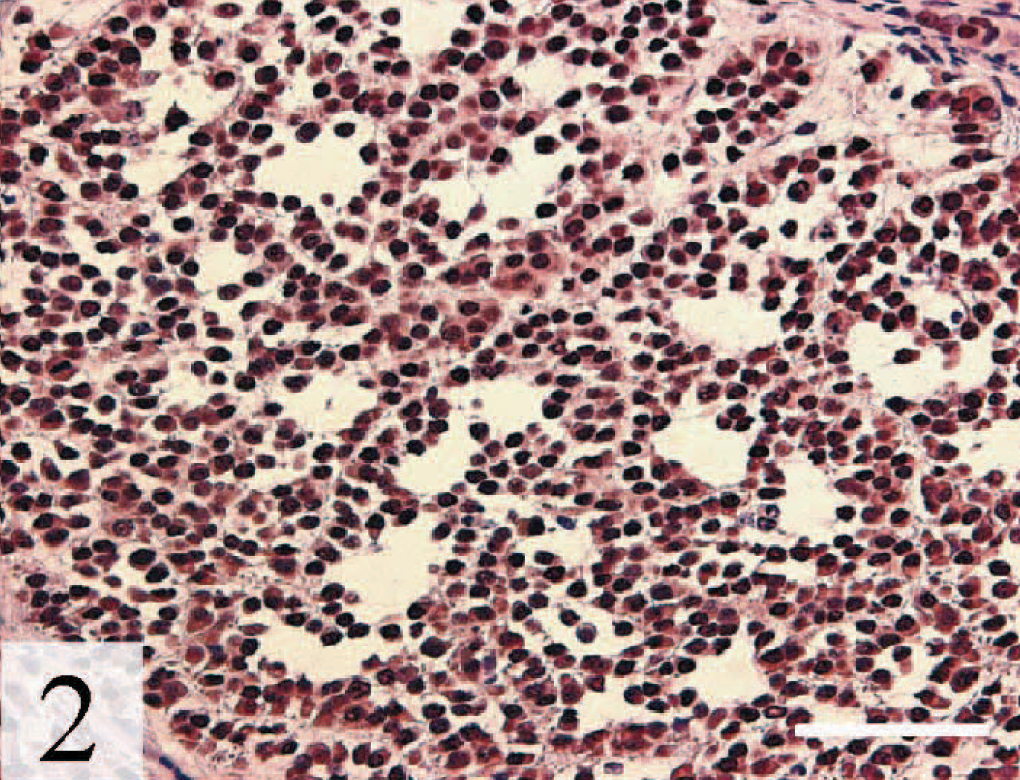
Adrenal gland, adrenocortical carcinoma; ferret. GATA-4 is expressed in the subset of cells exhibiting nuclear and cellular atypia. 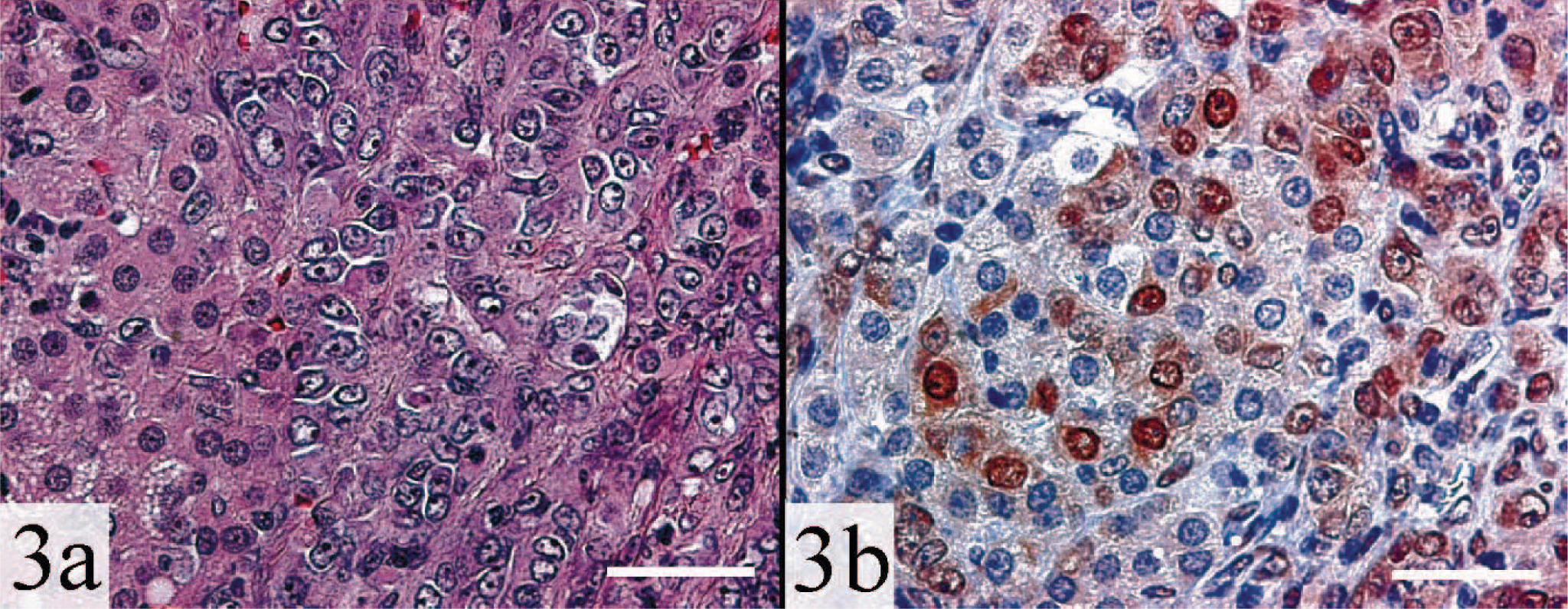
Adrenal gland, adrenocortical carcinoma; ferret. Regions expressing nuclear GATA-4 also express cytoplasmic inhibin-α. 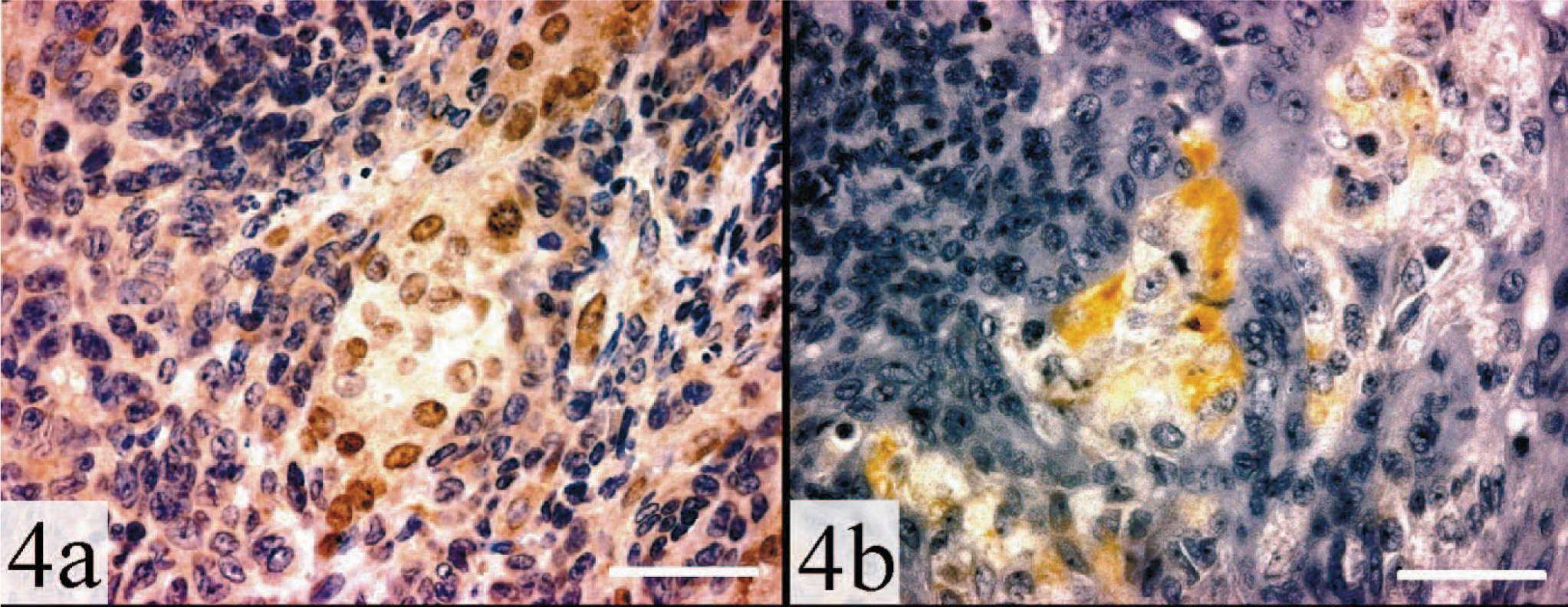
Adrenal gland, myxoid adrenocortical carcinoma; ferret. The asterisk highlights a mucin pool. Regions expressing nuclear GATA-4 also express LHR. 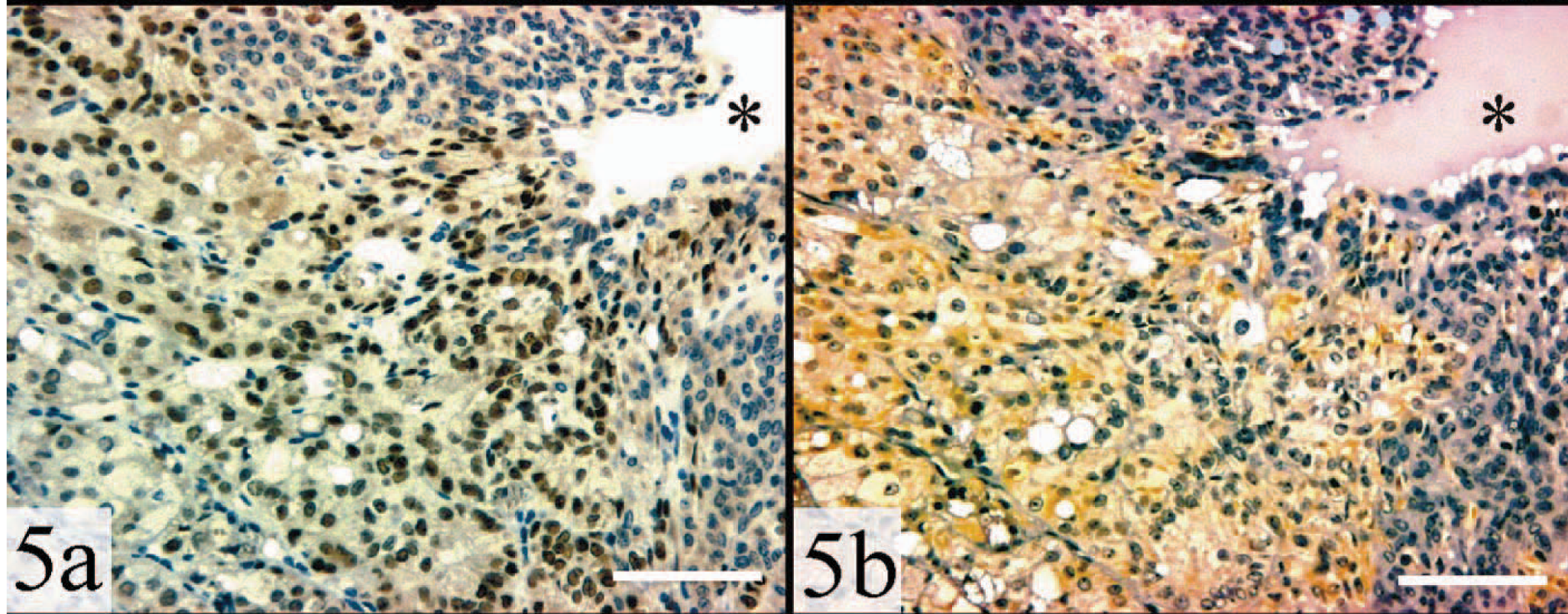
In contrast to adrenocortical carcinomas, no nuclear GATA-4 staining was observed in the three cases of nodular hyperplasia (Fig. 6). Isolated patches of GATA-4–positive cells were evident in 7/14 (50%) of tumors classified as adenomas (Fig. 7). In retrospect, these GATA-4–expressing cells, which exhibited atypia and were rich in extracellular matrix, may reflect foci of malignant transformation. Some of these GATA-4–expressing adenomas also expressed LHR.
Adrenal gland, nodular hyperplasia; ferret. Note the absence of nuclear staining. GATA-4, avidin–biotin–peroxidase complex method. Bar = 100 µm.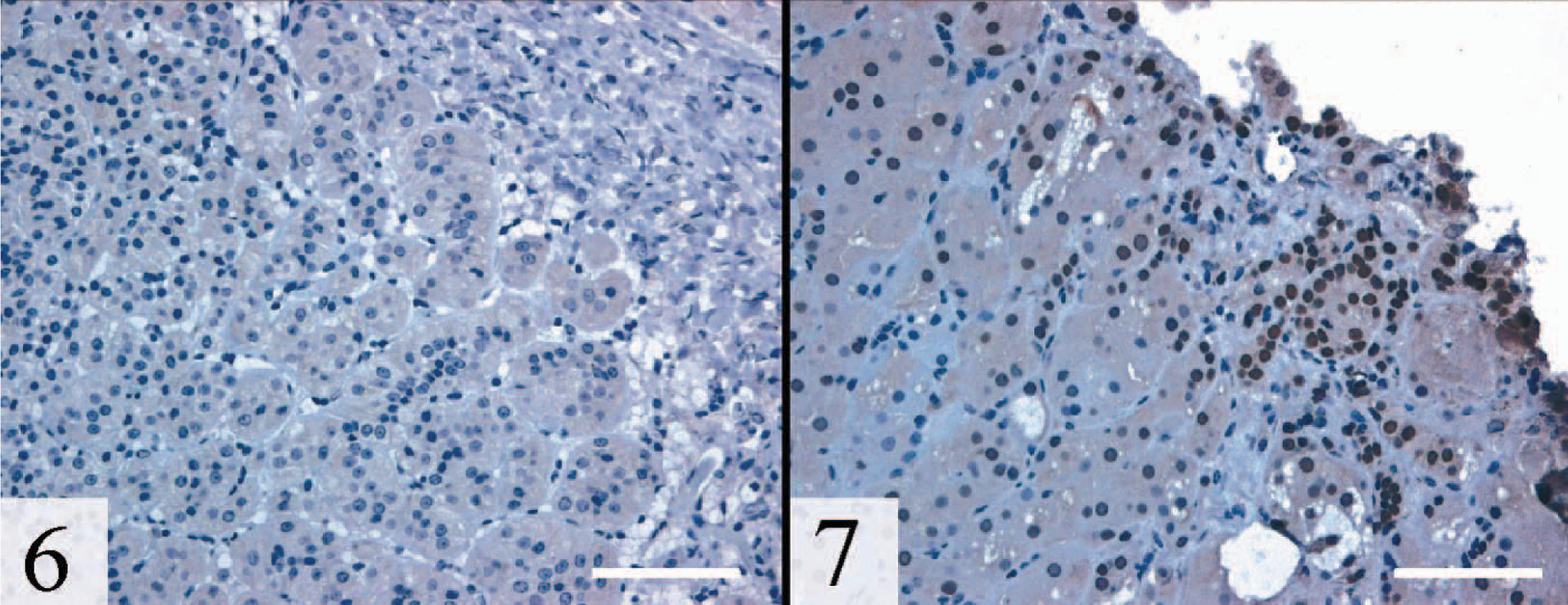
Adrenal gland, adenoma; ferret. Both positively and negatively staining cells are evident. Nuclear staining is more prominent in regions rich in stroma. GATA-4, avidin–biotin–peroxidase complex method. Bar = 100 µm.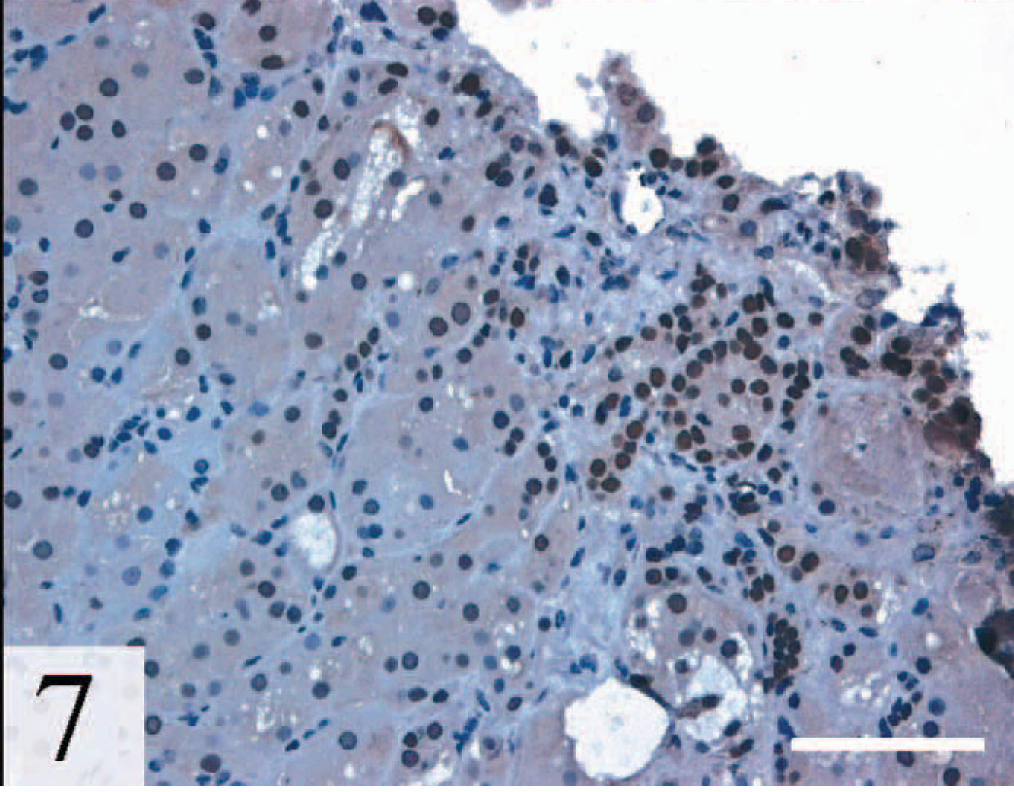
Traditionally pathologists have relied on morphologic features, such as cellular atypia, tumor encapsulation, or inva-siveness, to distinguish adrenocortical carcinomas from adenomas or cases of nodular hyperplasia. Recently, we demonstrated that myxoid differentiation often accompanies adrenocortical carcinoma and is indicative of a highly malignant lesion based on the rate of invasion into adjacent tissue and propensity to metastasize. 6 Another characteristic of aggressive tumors is expression of inhibin-α. 6 In this study, we have shown that GATA-4 is a marker of anaplasia in adrenocortical tumors. Nuclear GATA-4 immunoreactivity was observed in the vast majority of cases of adrenocortical carcinoma, and GATA-4 was detected in focal areas of anaplasia in tumors previously classified as adenomas. We propose that GATA-4 immunohistochemistry may serve as a diagnostic tool for detecting adrenocortical metastases and for discriminating potentially aggressive tumors from more benign variants.
Footnotes
Acknowledgements
Portions of this manuscript have been submitted in abstract form to the 54th ACVP Meeting. We thank Susan Porter-Tinge and Rosie Wilson for technical assistance. The Juselius Foundation, NIH HL61006, March of Dimes 1-FY02-203, Mallinckrodt Foundation, the Barnes-Jewish Hospital Foundation, and DDRCC P30 D52574 supported this work.
