Abstract
An epizootic infection was observed in a colony of 80 New World monkeys consisting of various species including a group of marmosets and
Although pox virus infections are not common among primates, spontaneous outbreaks of pox diseases are occasionally observed in captive nonhuman primate populations. In particular, four members of the pox virus family belonging to the Orthopox virus (OPV) genera, Yabapox virus and Mollusk pox virus, cause disease in nonhuman primates. 4 The most important disease is monkeypox (MP), an infection with the MP virus (MPV), an OPV closely related to the smallpox virus of humans. 1 Old World monkey species seem to have a higher risk of becoming infected than do New World monkeys. Nevertheless, outbreaks have also been observed among cotton-top tamarins and squirrel monkeys. 18, 19 Infection leads to a generalized disease characterized by circular expanding lesions of the skin that undergo vesiculation and ulceration or develop into pustules that crust over and drop off, leaving small scars.
Other poxviruses, belonging to the molluscum contagiosum group, also have been identified as causative agents of subcutaneous tumor-like lesions. 12 For example, natural infections with Yabapox virus in rhesus monkeys and baboons were reported. 13 The disease is characterized by rapidly growing subcutaneous nodules on the head and limbs. 5 This infection does not seem to occur in New World monkeys. 4 This also applies to Tanapox viruses, which are unrelated to the smallpox virus, but can induce a contagious skin disease in macaques and humans. The infection leads to multiple, crusted macules on face and arms that heal in 3 to 4 weeks and resemble MP arrested at the papular stage. Pathologically, the lesions are characterized by epidermal hyperplasia. Swollen epithelial cells contain eosinophilic, cytoplasmic inclusion bodies. The disease is not transferable to cebids or callithrichids. 16
Another poxvirus that is serologically closely related to the Tanapox virus and is infective for New World monkeys was isolated from a spontaneous outbreak in
To date and to the authors' knowledge, published information is not available concerning cowpox infection in nonhuman primates. Therefore, the purpose of the study reported here was to provide a detailed pathologic description of an uncommon OPV epizootic outbreak among various New World monkey species.
An unsuspected and atypical epizootic poxvirus infection was observed in a colony of 80 New World monkeys consisting of various primate species in Lower Saxony, Germany. A group consisting of
The owner of the six animals observed fever, depression, apathy, and seromucous nasal discharge. In the course of the febrile disease, all six animals developed severe erosive-ulcerative lesions of the oral mucous membranes. Three tamarins (No. 1–3) and one marmoset (No. 4) died at this early stage of disease, after a period of 1–2 days of illness. The other animals were treated empirically with antibiotics (Baytril) and Baypamun (Bayer, Leverkusen, Germany). All treated animals died with typical hemorrhagic skin lesions distributed randomly over the body, but most relevantly, on the face, scrotal region, and the soles and palms (Fig. 1, 2). Some lesions developed into erythematous papules. Only individual papular lesions became incrusted (Fig. 2). Other clinical findings were mild-to-moderate facial edema and severe lymphadenopathy of the mandibular and axillary regions (Fig. 1).
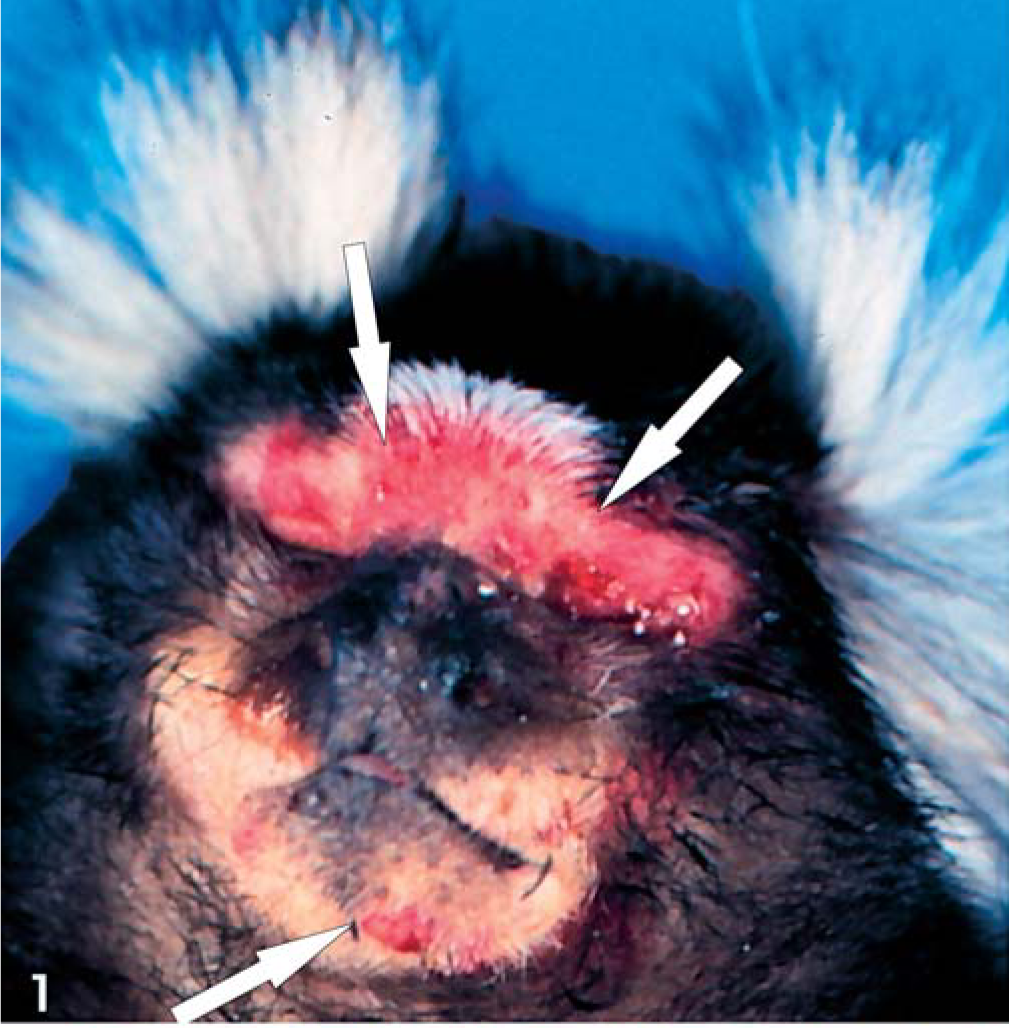
Skin; animal No. 5; marmoset. Notice hemorrhagic skin lesions and erythematous papules (arrow) with predilection for the face.
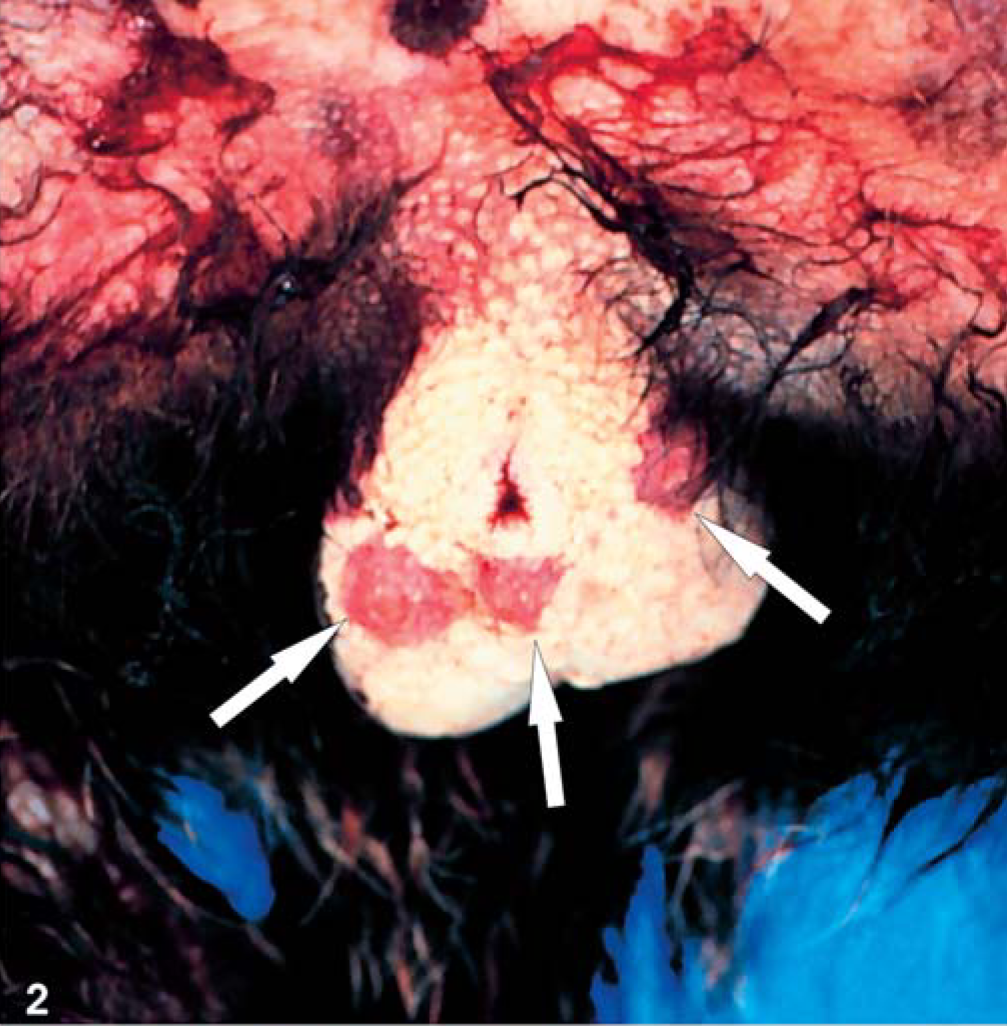
Skin; animal No. 5; marmoset. Notice severe hemorrhagic skin lesions and erythematous papules (arrow) with predilection for the scrotal region.
At necropsy, a spectrum of various focal dermal and mucosal lesions was seen. In all animals, the most prominent and consistent findings were massive erosions and ulcerations of the oral mucous membranes affecting the gingiva, tongue, and pharyngeal region. Four animals developed focal hemorrhagic-to-pustular skin lesions randomly spread over the body. These findings were accompanied by lymphadenopathy of the mandibular and axillary lymph nodes and several alterations of other organs, which differed from case to case (Table 1). The lymph nodes were hyperplastic with necrotizing lymphadenitis of various degrees. The histopathologic pattern depended on stage of development, degree of severity, and secondary bacterial infections, especially for the mucosal lesions. These patterns were classified as severe, epidermal hemorrhage and vesiculation; epidermal acanthosis and acantholysis; and full-thickness, epidermal necrosis and ulceration (Fig. 3).
Signalment, clinical signs of disease, and pathologic findings in the New World monkeys with pox virus infections.

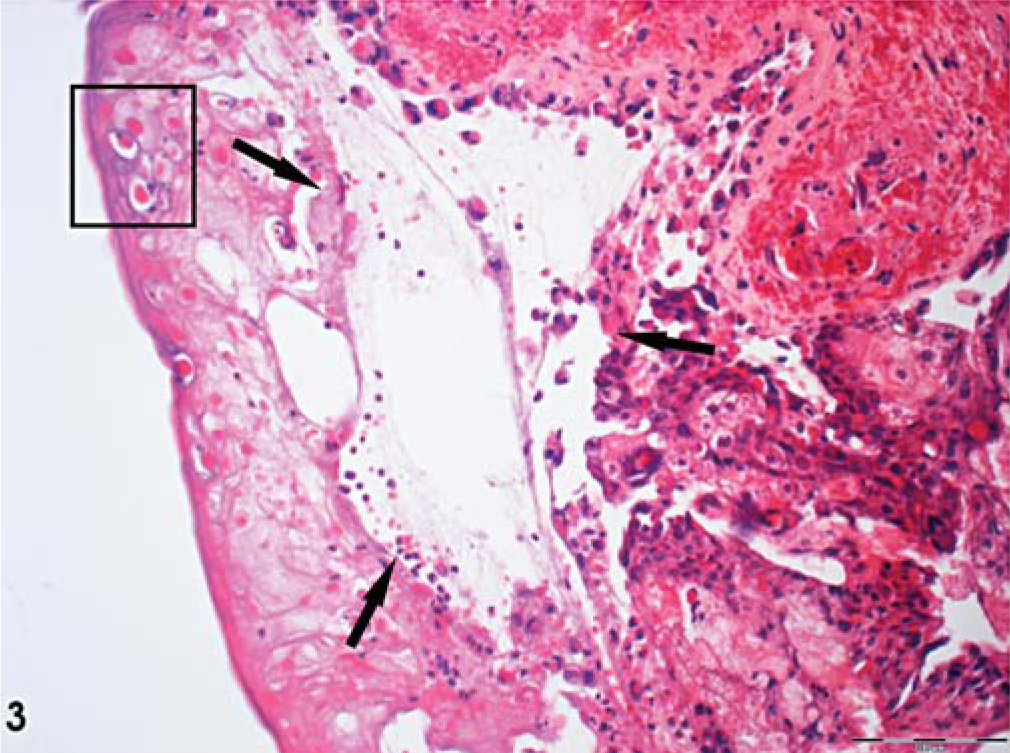
Skin; animal No. 5; marmoset. Severe hemorrhagic skin lesions, with erythematous papules characterized by severe vesiculation (arrow) in the epidermis accompanied by severe acanthosis and acantholysis. Intracytoplasmic inclusion bodies (inset) were evident in single epidermal cells. HE. Bar = 100 µm.
The bacterial superinfection was caused by various bacterial species and
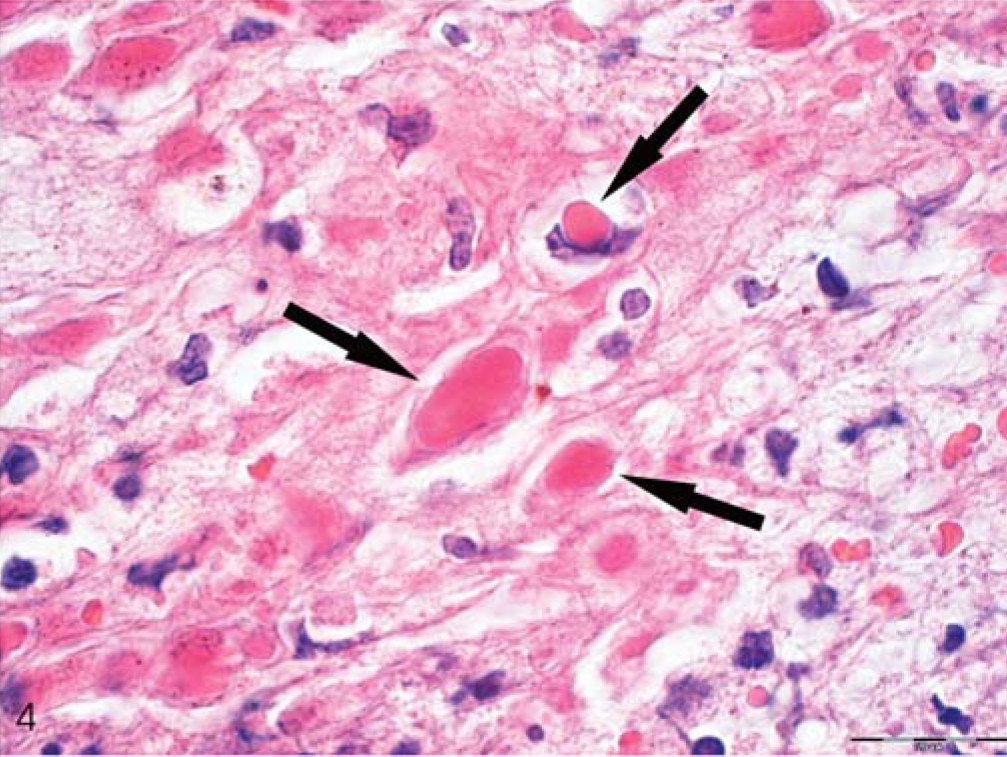
Skin; animal No. 5; marmoset. Notice intracytoplasmic Guarnieri bodies of various size randomly distributed in altered epithelium. Depicted is the inset region of Figure 3. HE. Bar = 20 µm.
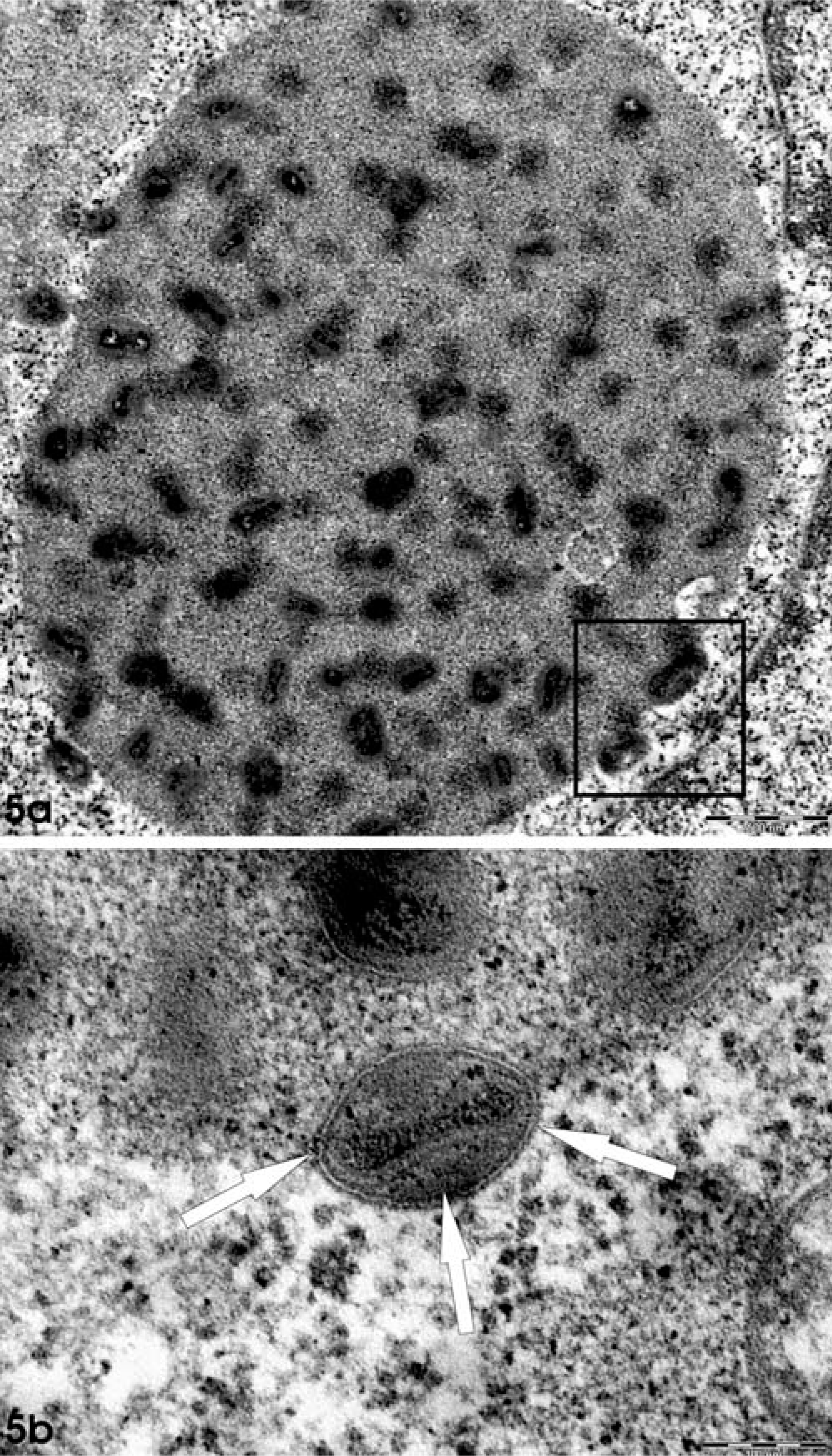
Skin; animal No. 5; marmoset. Fig. 5a. Transmission electron microscopy reveals intracytoplasmic inclusion bodies (small virus factories) containing several virus particles. Bar = 500 nm. Fig. 5b. Inset region of Fig. 5a. Single enveloped viral particles (arrow) were ovoid to brick-shaped, with a pale central core, presenting characteristic pox-like ultrastructural features. Bar = 100 nm.
For virus propagation, homogenates of liver tissue and skin samples from one marmoset (animal No.5) and one saddleback tamarin (animal No.2) were incubated with the permanent African green monkey kidney cell lines, Vero and MA 104, and were cultured using typical viral culture techniques. After 24 hours, the first signs of a cytopathogenic effect became visible in the Vero/MA 104 cell monolayer. Three days later, all cell cultures from the first passage had a rapidly increasing number of plaques, confirming the presence of infective virions in the tissue specimens (Fig. 6). The species-specific detection and differentiation of the virus isolates obtained from tissue specimens was performed by use of a monoclonal antibody/antigen-capture ELISA, and results were compared with a large panel of (OPV) reference strains and isolates. 11 The ELISA was based on 8 genus- and species-specific monoclonal capture antibodies raised against modified vaccinia virus Ankara (MVA), cowpox virus (CPV) KR2 Brighton, MPV Copenhagen, and ectromelia virus Munich 1. Reactivity exists with antigenic sites localized on the 14-kd fusion protein, the 32-kd adsorption protein, and the A-type inclusion body (ATI) protein of OPV encoded by the vaccinia virus open reading frames (ORF) A27L, D8L, and A25L. 7, 10 In this assay, the monkey isolates proved to be identical to key group I, consisting of vaccinia virus and CPV strains. To determine the species within the genus OPV, isolates of this special group were further differentiated by D8L polymerase chain reaction (PCR) analysis and cycle sequencing of this ORF.
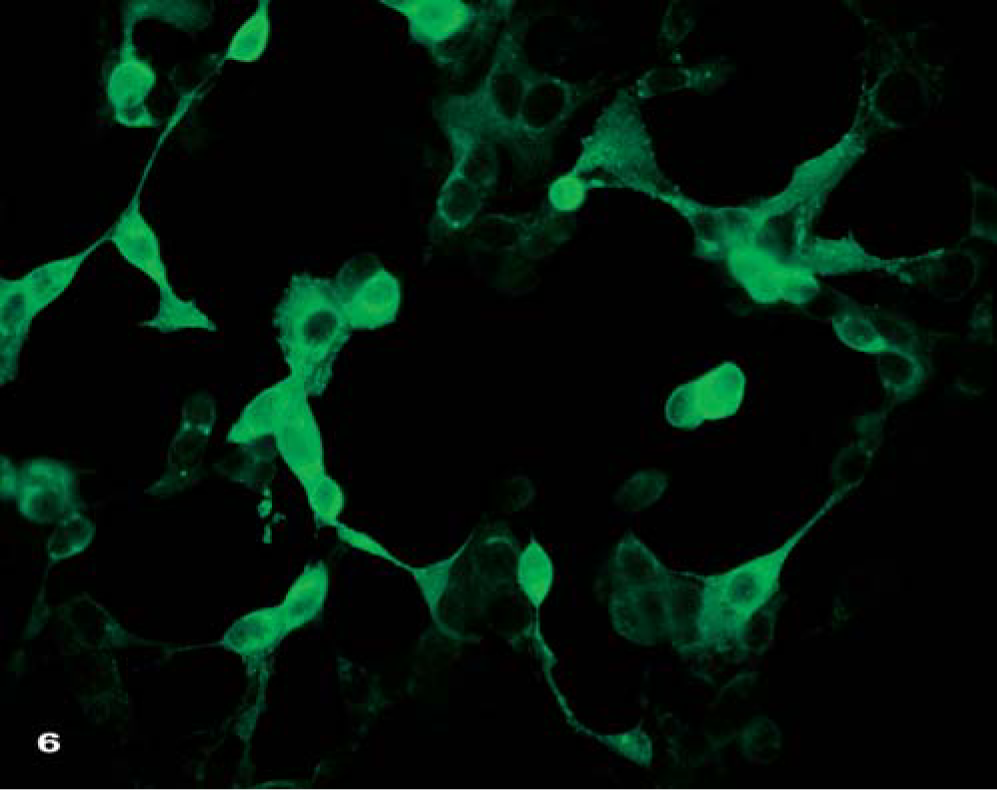
Vero cell culture with strong cytopathogenic effect. Green fluorescence indicates poxvirus replication within the cells. Indirect immunofluorescence method.
For this purpose, DNA was prepared from various tissues. The entire D8L genes of the monkey isolates encoding the 32-kd adsorption protein were amplified by genus-specific primers binding in the highly conserved N- and C-termini of the D8L ORF gene homologues of OPVs.
8
Gel electrophoretic analysis of the PCR products was performed with the gel image-analyzing system Eagle Eye II (Stratagene, Amsterdam, Netherlands) and revealed amplicons of 915 bp, identical to that of the corresponding reference strain—a vaccinia virus (Fig. 7). The DNA was also extracted from cell-free supernatants of virus-inoculated Vero/MA 104 cells. It was analyzed by use of a crmB-specific real-time PCR analysis that yielded approximately 106–107 viral genome copies/ml of each supernatant. These data confirmed the replication of infective OPV in cell culture. The DNA sequences from the monkey isolates in our study were aligned to published sequences and to the corresponding sequences of 31 OPV reference strains and recent isolates from humans, cats, elephants, pigs, foxes, and tapirs by the DNA Star Lasergene Computer program (GATC, Konstanz, Germany). To identify a species-specific relationship and to elucidate the origin of the virus, a phylogenetic tree was constructed and deduced from the amino acid sequences of the 32-kd adsorption protein using the Neighbor Joining method. Results of this analysis confirmed that the virus isolates of the monkeys represented a typical CPV belonging to a group of well-known CPV isolates derived from cows, cats, horses, and humans. The presence of the variola virus could be excluded according to our experimental protocols. The CPV was detected in various organs of the monkeys including liver, spleen, lung, small intestine, esophagus, skin, and mucosa. Sequence data confirmed all 6 animals to be infected with the same virus (Fig. 7). Additionally, on the basis of the sequence of CPV Brighton red (gene bank Acc. No. AF482758), the overlapping primer pairs were selected to amplify and to sequence the entire
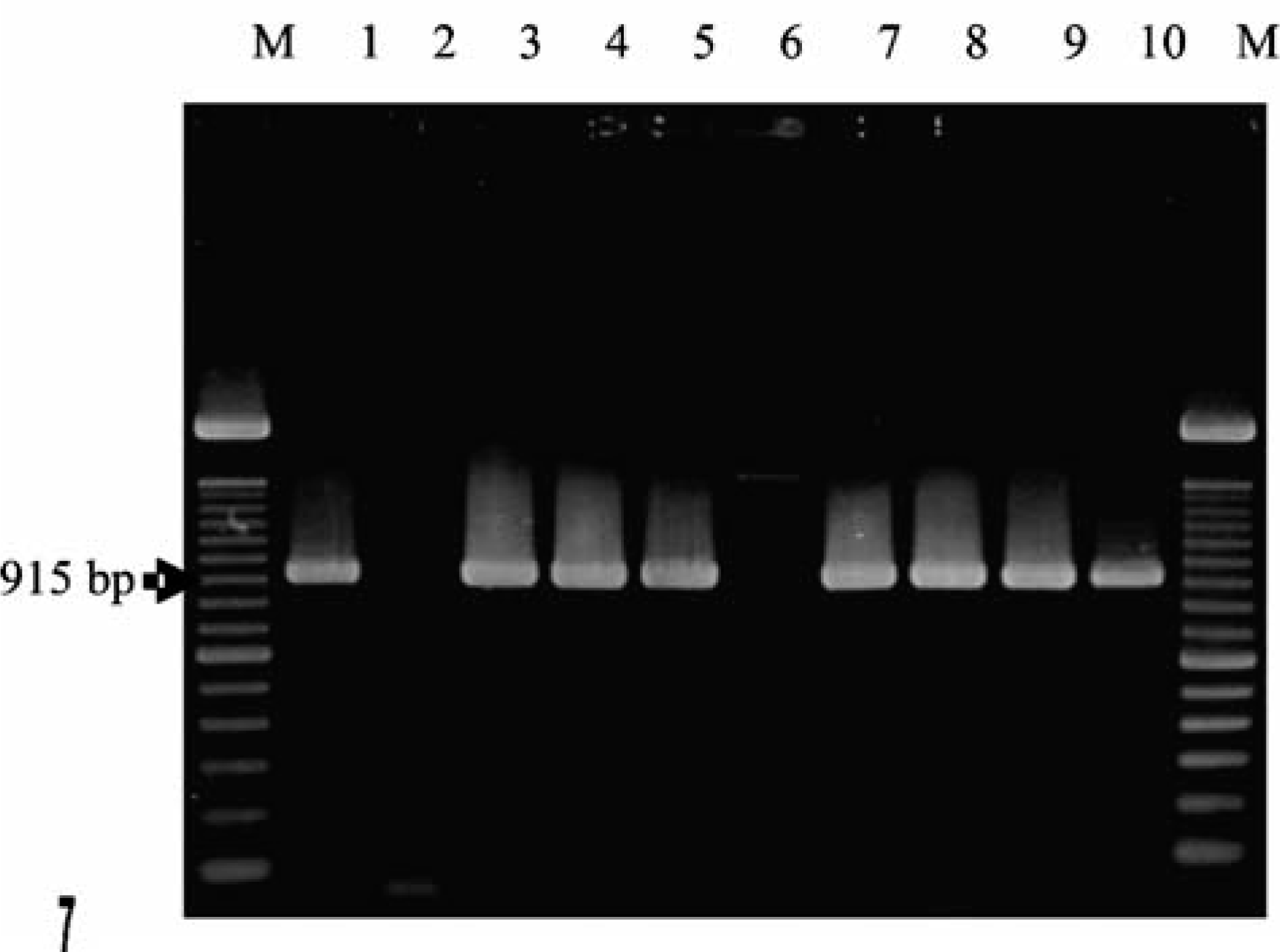
Sequence data of the D8L-amplicon. Sequence data confirmed the presence of orthopox virus within various organs from various animals. All animals were infected with the same virus. M = molecular weight marker. Lanes: 1, vaccinia virus Elstree (positive control); 2, water (negative control); 3, lymph node from the skin of animal No. 1; 4, esophagus, animal No. 2; 5, liver, animal No. 6; 6, spleen, negative-control animal; 7: adrenal gland, animal No. 4; 8: gingiva, animal No. 5; 9: lung, animal No. 6; and 10: small intestine, animal No. 6.
Since vaccinia vaccination currently is not being undertaken, the possibility that the human population, especially people <25 years old, is susceptible to poxvirus infections cannot be ruled out. Therefore, and bearing in mind that single MB cases in the USA have been observed, zoonotic poxvirus infections are of great interest. The current outbreak of an OPV infection in a group of various New World monkeys raises a question about the possible risk of this infection for other species including humans. Cowpox virus, a member of the OPV genus, was identified as the agent responsible for this fatal infection among New World monkeys. During this outbreak, all infected animals developed a severe systemic infection, with the most prominent lesions found on the oral mucous membranes, which is in line with the typical epithelial manifestation of the virus. The most advanced lesions consisted of multifocal epidermal and mucosal vesicle formation, necrosis, and ulceration attended by intense inflammatory response. Interestingly, only some of the animals developed typical papular skin lesions. Classic pustule formation was observed in only one animal. The other animals died before reaching this stage of illness. The detection of hemorrhagic papular skin diseases in nonhuman primates should focus attention on a contagious, possibly zoonotic disease. For diagnosis of pox virus infections, the presence and detection of typical intracytoplasmic inclusion bodies within the altered epidermis is vital. These bodies represent viral inclusions. However, electron microscopic and viral isolation techniques are adequate means of confirming a poxvirus infection.
Phylogenetic analyses of the isolated poxvirus revealed closest homology to CPV. Cowpox virus is a rodent virus that can infect cats, cows, zoo animals, and through contact with these, humans. 3 Cowpox virus infections are endemic among cattle, although clinical cases in the European cattle population are rare. 2 Field and experimental studies have indicated that CPV has a broad host range, and rodents and foxes are wildlife reservoirs. 6 Furthermore, the virus is often isolated from domestic cats, which should be regarded as important vectors in urban areas. 3, 6, 17 In recent years, several human cases, usually with localized skin lesions, were observed. Lesions in humans can often be found on the hands, forearms, face, and neck. 14 Predisposition, such as immunosuppression, may lead to a severe or fatal course of infection, as has been reported in the case of a glucocorticoid-treated asthma patient after contact with a cat. 9, 11 Careful evaluation of the epidemiologic situation of CPV infection suggests that CPV has a low virulence and contagiousness for humans. 2 Cowpox virus infection is most often diagnosed in domestic cats. It is a zoonosis, but human cases are rare. Therefore, it is not expected that CPV infection will become a major problem for human health in the future. 2 As for nonhuman primates, the situation is still unclear. The described cases indicated that nonhuman primates of various species are susceptible. All infected animals were in good condition beforehand, and predisposing immunosuppressive cofactors were not reported. According to these results, the isolated virus has to be considered contagious for nonhuman primates.
The infection was not transmitted to the animal owner, animal keepers, clinical veterinarians, or pathologists during the outbreak being studied. Thus, the zoonotic potential of this pox virus could not be recognized. The epidemiologic situation in this epizootic is still under investigation. The exposed animal owner and his family, with history of smallpox vaccination, had low titers of cross-reacting antibodies to CPV. One unvaccinated child did not have anti-CPV titer. Several precautions were taken to prevent spread to animal keepers. The contact with infected animals was reduced to only a few people, who were strongly encouraged to wear protective clothing, gloves, and a protective mask. There was no evidence of a specific vector in this case, but it is assumed that rodents carried the virus into the colony. After the flood, more rodents were seen in the husbandry area and even in the cages. In total, 32 mice were trapped and investigated. Antibodies against OPVs were detected by immunofluorescence in 41% of rodents caught in this area, indicative of a previous OPV infection in mice. The infected monkeys had access to outdoor areas. They tended to hunt small animals and, therefore, most likely, were exposed to infected rodents.
Rodents are thought to be a reservoir for CPV and persistent infections, in addition, recombination events among pox viruses might occur. 20 Cowpox viruses are known to infect various species, including cats, humans, and other primates. They cause only localized, self-limiting infections. To our knowledge, CPV infections have not been observed in New World monkeys. However, a recombination event between different viral strains could have generated a virus with the newly observed properties, leading to fatal systemic infection of New World monkeys. The virus was spread to secondary organs, with high viral titers in liver and spleen, and many symptoms associated with smallpox infections in humans were seen. The infected animals were in direct contact via a climbing rope among various cages. Furthermore, two infected animals were moved from one stable to another where two more marmosets became infected and died. As a result of this observation, it seems that the virus is transmitted from animal to animal either by respiration or by skin contact establishing a route of infection under natural conditions. So far, it is not clear whether the virus is also capable of infecting humans or other primate species. Orthopox virus infections in exotic animals may have a transmission potential for humans; however, those viruses are unlikely to be highly contagious for humans, since the animal care workers having closest contact with the infected animals did not manifest signs of infection. The source of this poxvirus outbreak remains to be discovered, and the origin and species of the virus needs to be precisely determined to assess the risk of transmission to humans.
Footnotes
Acknowledgements
We are grateful to W. Henkel, K. Kaiser-Jarry, N. Knöchelmann, E. Lischka, and H. Zuri for excellent technical assistance and to W. Bodemer for his generous advice and support.
