Abstract
Cowpox virus infections have been described in various domestic and exotic animal species. This report is the first on an outbreak of fatal generalized cowpox virus infection among captive banded mongooses (Mungos mungo, suborder Feliformia). All animals of a colony of 8 mongooses showed a fulminant course of disease. The whole population died (n=7) or was euthanized (n=1) within 11 days. Postmortem examinations were performed on 4 animals. All animals showed extensive necrotizing inflammation of retropharyngeal lymph nodes, typical poxviral skin lesions, and multiple necrotic foci in liver and spleen. Three animals exhibited an ulcerating stomatitis. Pulmonary lesions, a common feature of fatal cowpox virus infections in other feliform species, were not obvious. Histopathologically, characteristic cytoplasmic inclusion bodies were detected in all affected organs but the spleen. Based on transmission electron microscopy and cell culture, Orthopoxvirus was identified as the etiology. The virus was further characterized by polymerase chain reaction and sequence analysis, identifying it as cowpox virus. A survey in the habitat suggests wild brown rats (Rattus norvegicus) as the most likely source of infection.
Cowpox virus (CPXV), a member of the genus Orthopoxvirus of the family Poxviridae, is endemic in parts of Europe and Western Asia. 6,10 Natural infections have been documented in a broad range of species, 4, 5, 7, 10, 12–14, 20, 21, 23, 24, 27 including various captive exotic mammals 14,16,18,19,21 in particular also in several wild species of the family Felidae, such as ocelots (Leopardus pardalis), black panthers (Panthera pardus), lions (Panthera leo), pumas (Puma concolor), Far Eastern cats (Felis bengalensis), jaguars 18 (Panthera onca), Eurasian lynx 25 (Lynx lynx), and cheetahs 1,18 (Acinonyx jubatus). Wild rodents are generally regarded as the natural reservoir of CPXV. 6 At present, the most common source of human CPXV infection, a rare zoonosis, is the domestic cat, probably originally infected by rodents. 2,4,12 However, several recent cases of human CPXV infection have been reported as transmitted by infected pet rats. 5 Domestic cats infected with CPXV usually develop mild clinical signs consisting of typical poxviral cutaneous lesions, which usually heal spontaneously. 4 Mild pyrexia, inappetence, and oral ulcerations are occasionally seen. 4,24 Severe illness or even fatal courses of the disease with necrotizing pneumonia are rare. 2,4,12,13,24 However, in exotic feline species, severe CPXV infections with lethal outcome are well known. 1,18,19 During severe outbreaks in the Moscow Zoo 18 and in British Whipsnade Park, 1 fatal pulmonary, milder dermal, and, less frequently, mixed forms of the disease were observed even within the same species. Animals affected by the pulmonary form exhibited a fibrino-necrotic, partially hemorrhagic pneumonia and sero-fibrinous pleuritis with lethal outcome. Dermal courses of the disease were of varying severity, and fatalities were rare. Mixed forms were characterized by cutaneous, oral, and esophageal papules and vesicles as well as fibrino-necrotic pneumonia and laryngitis. Here we report an outbreak of fatal generalized CPXV infection in captive banded mongooses (Mungos mungo). Mongooses are members of the family Herpestidae and belong, as Felidae, to the suborder Feliformia (order Carnivora).
Materials and Methods
In August 2008, the whole population of 8 banded mongooses in a zoological park in Germany died (n=7) or was euthanized in agony (n=1) within 11 days. The mongooses were housed in an outdoor enclosure, without contact to any other animal of the zoo. Three animals died without clinical symptoms. The other mongooses exhibited anorexia, lethargy, imbalance, and severe dyspnea with wheezing. Three animals showed a prominent swelling in the intermandibular region. There was no response to antibiotic and anti-inflammatory treatment. Four mongooses died within 1 to 2 days after onset of the disease. One animal was euthanized in agony. No other animal in this zoo of any other species showed obvious signs of illness. On 4 mongooses (case Nos. 1–4), as well as on 2 wild brown rats (Rattus norvegicus) trapped near the enclosure of the dead mongooses for epidemiologic reasons (rats Nos. 1 and 2), a full gross examination was performed, and organs from all animals were collected for further investigations. Tissue samples were fixed in 10% formalin and routinely processed for histological assessment. Homogenates of organ samples from liver, spleen, kidney, and skin from all mongooses were routinely incubated on mammalian cell lines, including CRFK cells (Crandell-Rees feline kidney cells; ATCC CCL-94) and FCWF cells (felis catus whole fetus cells), for 5 to 7 days with daily microscopic examination. 9 Cultures without evidence of cytopathic effect or with questionable cytopathic effect were subcultivated and kept until a cytopathic effect was seen. Supernatant was harvested from cultures exhibiting a cytopathic effect and subjected to transmission electron microscopy (TEM; negative staining technique). Intestinal samples of all mongooses were routinely prepared for negative staining by TEM as previously described, with a prolonged incubation time of 15 to 30 minutes. 3 For TEM of ultrathin sections, formalin-fixed samples of skin (case No. 3) and liver (case Nos. 2–4) were postfixed with 1% osmium tetroxide, dehydrated, and embedded in a mixture of Epon and Araldite. Resin embedded samples were cut at 60 to 90 nm and stained with uranyl acetate and lead citrate. Semithin and ultrathin sections were routinely prepared and examined. DNA extraction of blood and skin samples of case No. 1, as well as of blood, liver, and skin samples of rat No. 1, was performed using QIAamp Viral RNA Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. A 110–base pair region of the 14-kDa fusion protein gene was amplified by use of artus Orthopox LC PCR Kit (Qiagen) according to the manufacturer’s instructions. 22 Orthopox virus–positive samples were taken for amplification, and sequencing of the cytokine response modifier B (crmB) region was performed using consensus primers VL2N and VL33. 15 Furthermore the hemagglutinin (HA) gene was amplified and sequenced. 8
Results
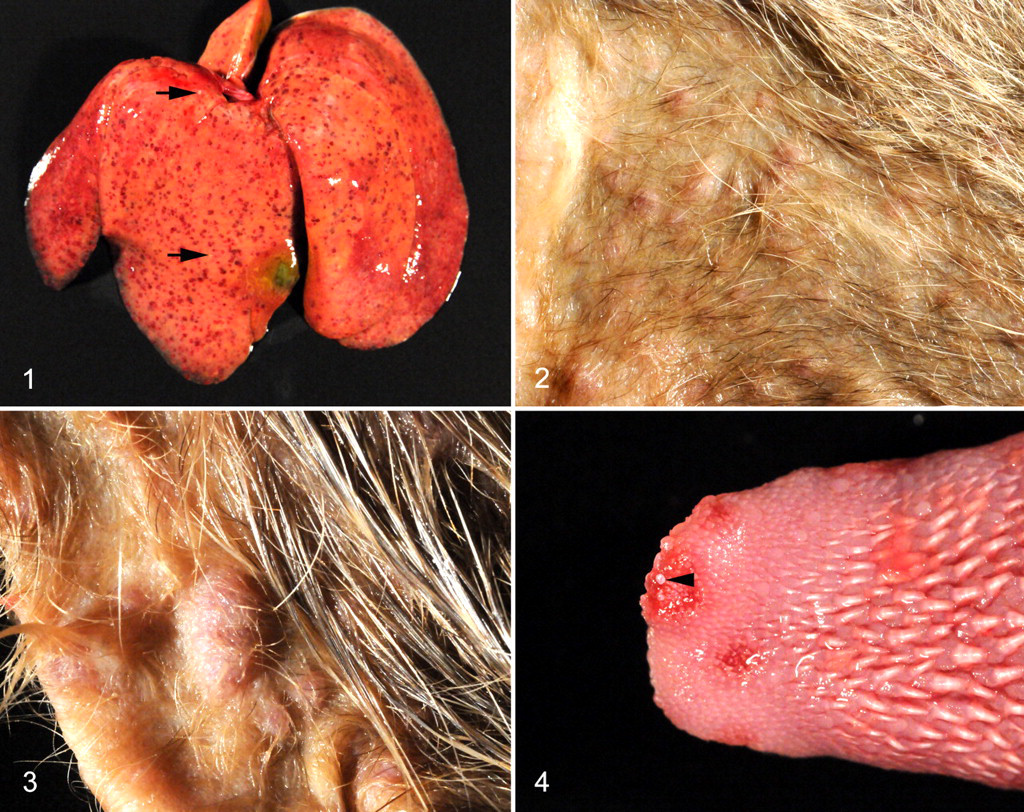
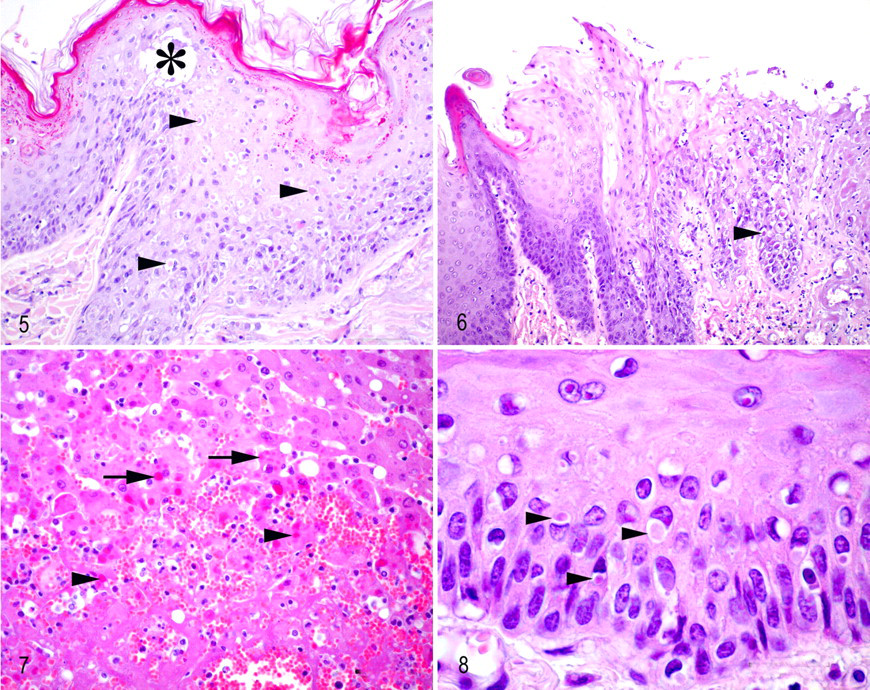
All mongooses were adult (2 male, 2 female) and in good body condition. Postmortem examination revealed disseminated petechiae and miliary foci of necrosis in the liver of all animals (Fig. 1 ). Additionally, all animals showed multiple, sometimes confluent, papular, vesicular, or pustular cutaneous lesions, measuring up to 0.5 cm in diameter, sometimes covered by small crusts (Figs. 2, 3). The lesions were especially numerous in the sparsely haired skin of ventral trunk, in medial parts of limbs, in ears, around nose and eyes, and in the area of the ventral neck. In the location of the retropharyngeal lymph nodes, a compact, poorly demarcated, expansile, homogeneous white mass was seen in all animals, measuring up to 5.0 × 2.8 × 2.8 cm, which severely compressed the larynx. Case No. 1 additionally exhibited fibrino-necrotic tonsillitis and laryngitis. In case Nos. 2–4, multiple erosions, ulcers, and few vesicles were found in the mucosa of the oral cavity, most pronounced on the tongue (Fig. 4 ). Case No. 1 showed a multifocal fibrino-necrotic enteritis. Wild rat No. 1 exhibited a single crusted vesicular cutaneous lesion near the muzzle. Microscopic examination of the skin lesions of the mongooses revealed marked epithelial hyperplasia, ballooning degeneration, and acantholysis of the keratinocytes (Fig. 5). Formation of tiny intraepidermal vesicles and serocellular crusts was occasionally observed (Fig. 5). Many lesions showed extensive necrosis of keratinocytes with ulceration, sometimes extending deeply into the dermis, with infiltration of lymphocytes, macrophages, and neutrophils. Numerous individual or multiple variably sized homogeneous eosinophilic cytoplasmic inclusion bodies were seen in keratinocytes (Fig. 5). The mucosal lesions showed similar histologic features, including cytoplasmic eosinophilic inclusion bodies in epithelial cells (Fig. 6 ). The liver exhibited multiple foci of coagulative necrosis with numerous cytoplasmic eosinophilic inclusion bodies in hepatocytes, acute intralesional hemorrhage, and mild infiltration of neutrophils (Fig. 7 ). The retropharyngeal lymph nodes showed an extensive necrotizing inflammation that extended into the surrounding skeletal muscle and connective tissue (necrosuppurative myositis and cellulitis with edema) with eosinophilic inclusion bodies in cells with histiocytic and fibroblastic morphology. The spleen showed multiple foci of necrosis and a mild infiltration with neutrophils. Case Nos. 1, 3, and 4 had multiple small foci of necrosis in the bone marrow. Mongoose No. 1 exhibited a multifocal fibrino-necrotic enteritis with multiple eosinophilic inclusion bodies (enterocytes). Postmortal alterations did not allow for histological assessment of the intestine of mongooses Nos. 2–4. The histopathologic examination of the cutaneous lesion of rat No. 1 revealed severe ulceration of the epidermis with hydropic degeneration of keratinocytes and cytoplasmic inclusion bodies (Fig. 8 ).
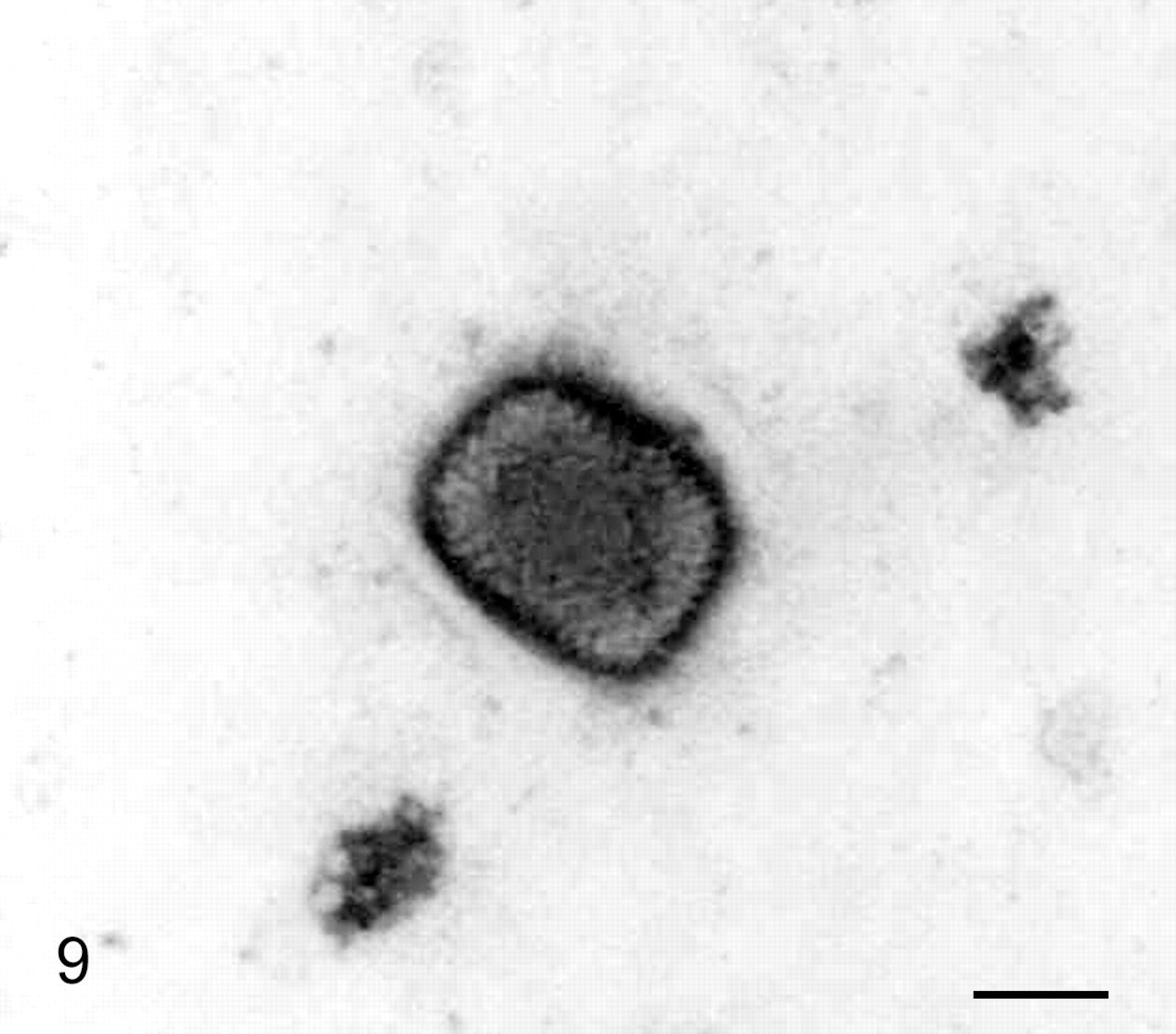
Intestine; banded mongoose No. 1. Note the characteristic mulberry formed viral particle with short, randomly arranged surface tubules. Transmission electron microscope, negative staining. Bar = 130 nm.
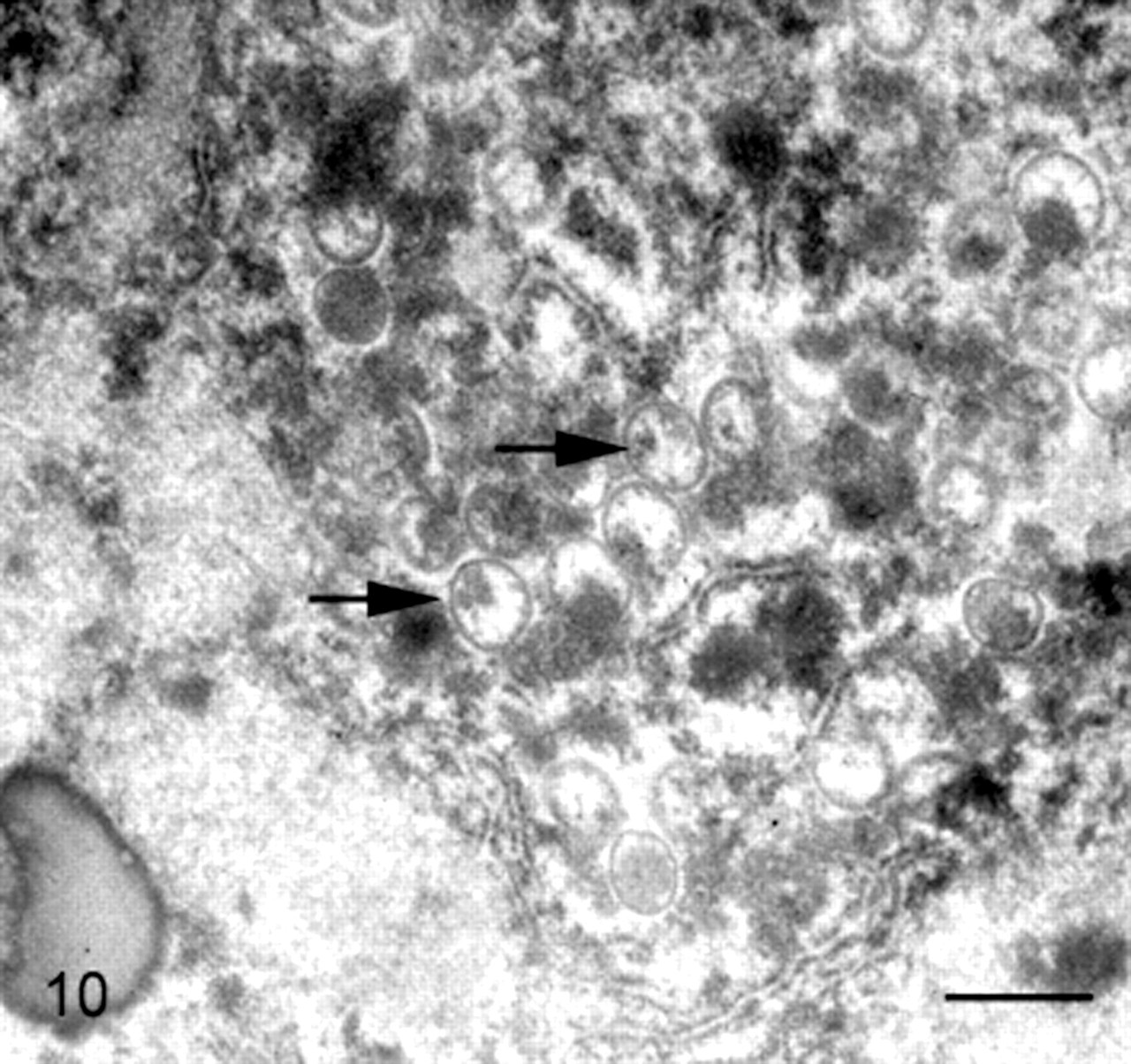
Liver; banded mongoose No. 3. Virus factories with immature viral particles (arrows) were detected in hepatocytes. Transmission electron microscope, uranyl acetate and lead citrate. Bar = 500 nm.
Cell cultures showed cytopathic effects 24 to 48 hours after inoculation. TEM examination of intestinal samples and culture supernatants identified virus particles of approximately 220 × 280 nm with characteristic brick-shaped morphology and tubular surface projections (Fig. 9 ) in all cases. Examination of ultrathin sections of liver (case Nos. 2–4) and skin (case No. 3) revealed so-called virus factories 10,16 with crescent-shaped structures and immature virus particles in hepatocytes (Fig. 10 ), Kupffer cells, and keratinocytes. In the subcutaneous connective tissue, extracellular mature virus particles were detected, and virus particles in different stages of maturation were found in fibroblasts. Orthopox DNA could be amplified from extracts of blood and skin (case No. 1). Melting point analyses identified the virus as genus Orthopoxvirus (Poxviridae). 22 Amplification and sequencing of the crmB and HA gene confirmed this observation and classified it as a member of the CPXV by blast analysis (http://blast.ncbi.nlm.nih.gov/Blast.cgi). Comparison with published sequences revealed a homology of 94% to the reference strain Brighton Red (accession No. U07767) and a homology of 96% to a strain isolated from a rat in France (FJ843101). Polymerase chain reaction failed to identify Orthopox virus-specific DNA in the cutaneous lesion, blood or liver of rat No. 1.
Discussion
To our knowledge, this report is the first on fatal generalized CPXV infection in banded mongooses. Like other members of the suborder Feliformia, such as lions, banded mongooses appear highly susceptible to CPXV infection with a morbidity and mortality of 100%, showing a highly fulminant course of disease. Compared to other CPXV outbreaks, also among Feliformia, this outbreak in banded mongooses is exceptional, mainly because of its uniform course and mortality rate of 100%. 1,18 The latter has, so far, been only reported in the population of lions and cheetahs in the Moscow Zoo. 18 In Whipsnade Park, not all cheetahs that were in contact with diseased animals became infected. 1 Although the mongooses exhibited a fatal generalized form of disease, the postmortem and histopathological examinations revealed no sign of pneumonia, which is usually reported as a major feature in other exotic members of the suborder Feliformia 1,18 and less frequently in domestic cats 4,12,13,24 affected by a highly fulminant, generalized, and fatal CPXV infection (inclusion bodies in bronchiolar and alveolar epithelial cells, 13,18 endothelial cells, 12 mesothelial cells 13,24 ). However, the mongooses showed severe lesions in several internal organs. Whereas affection of internal organs other than the lungs is uncommon in domestic cats, pinpoint foci of necrosis in the liver and foci of follicular necrosis in the spleen have also been described in lions, cheetahs, and a black panther affected by the pulmonary form of the disease. 18 Lesions in the oral and pharyngeal mucosa owing to generalized CPXV infection, as seen in the mongooses, have been described in species as various as elephants (Elephas maximus), 14,19 domestic cats, 4,24 nondomestic cats, 1,18 horses, 23 saddlebacked tamarins (Saguinus fuscicollis), 16 and common marmosets (Callithrix jacchus). Although lymphadenopathy is reported to occur in humans affected by CPXV infection 1,5,23,24 and although necrotizing lymphadenitis of the mandibular lymph nodes has been observed in tamarins and marmosets, 16 the extensive necrotizing lymphadenitis of retropharyngeal lymph nodes seen in this case is a noteworthy finding; it even caused severe wheezing respiratory distress in all mongooses owing to compression of the larynx. In the present study, epithelial cells (skin, oral and intestinal mucosa, hepatocytes), macrophages (Kupffer cells, histiocytes), and fibroblasts were identified as target cells for the virus, which is in accordance with the known epitheliotropism of CPXV in several species. 14,16,18,24 Viral replication in fibroblasts and cells of the macrophage lineage has also been reported in cats. 12,13 However, inclusion bodies in hepatocytes have so far not demonstrated. 18
The distinctive histopathologic findings in the cutaneous lesion at the muzzle of rat No. 1 strongly indicate an infection with CPXV. 17,20 The only other poxvirus known to cause characteristic cutaneous lesions with cytoplasmic inclusion bodies in rodent species is ectromelia virus (ECTV; Orthopoxvirus). No data are available concerning the prevalence of ECTV in wild rats, and experimental studies have shown that rats are relatively resistant to ECTV infection. 11 Thus, despite the negative polymerase chain reaction result, the characteristic histopathologic findings may suggest the wild brown rat as the most probable source of CPXV infection in the mongooses. Although bank voles (Clethrionomys glareolus), field voles (Microtus agrestis), and wood mice (Apodemus sylvaticus) are generally regarded as the main host reservoirs of CPXV in Europe, 6 rats have been shown to play a role as source of infection for captive exotic animals 14,18,19,21 and even humans. 5,27 Because no data are available concerning the seroprevalence of CPXV antibodies in wild rats in Germany, it remains unclear whether brown rats are accidental or reservoir hosts. 27
Concerning the route of virus transmission, it is still unknown whether repeated direct contact between infected rats and mongooses was necessary or whether mongoose-to-mongoose transmission occurred too. Observations during outbreaks in zoological parks as well as among experimentally infected brown rats showed that animals also got infected without having had direct contact to infected animals before. 1,17,18 Experimental CPXV infection of brown rats revealed that the virus is excreted for more than 1 month in urine and feces by surviving animals. 17 These findings indicate that indirect transmission (eg, via infected excreta as urine or feces, contaminated cages or food) may also have played a role, especially as virus particles were demonstrated in intestinal samples of the mongooses. Thus, this report underlines the difficulty to eliminate the risk of CPXV transmission by wild rodents entering the enclosures of the zoo animals while foraging. The fatal outbreak described in our report indicates the importance of opportune vaccination of potentially susceptible exotic animals. Management plans in case of future outbreaks should be considered as poxvirus crosses species barriers and zoonotic infections of keepers by exotic zoo animals have already occured. 14,18 Those plans will rely on prompt diagnosis and strict segregation of affected animals. Apart from the zoologic value of the exotic species, this is important with regard to the zoonotic potential of CPXV. Whereas human infections are usually mild and self-limiting, severe and even lethal infections have been documented in immunocompromised patients. 2,7 As a consequence of a decreasing immunity against orthopoxviruses in society, owing to cessation of vaccination against smallpox and in light of the circulation of CPXV in wild and captive animals, the risk of infection is probably increasing for keepers, animal trappers, and owners of cats and pet rats. 5,21,26
Footnotes
Acknowledgements
Excellent technical assistance was provided by Sabine Wack and Ute Zeller.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
