Abstract
Six New World primates, including 2 golden lion tamarins (Leontopithecus rosalia), 2 cotton-top tamarins (Saguinus o. oedipus), 1 black howler monkey (Alouatta caraya), and 1 black-handed spider monkey (Ateles g. geoffroyi), were diagnosed with unilateral (4/6) or bilateral (1/6) adrenal or extra-adrenal (1/6) pheochromocytoma by light microscopy and immunohistochemical staining for chromogranin A. Overt invasive behavior or metastases were not observed in any primate, and thus these neoplasms were considered benign. All primates either died spontaneously (4/6) or were euthanatized (2/6) as a result of concurrent malignant neoplasia, infection, renal disease, or a combination of several disease processes. Although we did not determine whether these pheochromocytomas were functional, all 6 primates had myocardial fibrosis, and some had arteriosclerosis.
Keywords
Endocrine and, specifically, adrenal neoplasia is infrequently described in nonhuman primates 1,9 ; however, endocrine neoplasms were the most frequent neoplasms in 2 retrospective studies in Old and New World primates, 4,7 and some surveys of neoplasia in nonhuman primates list a number of endocrine tumors. Adrenal tumors other than cortical adenomas appear to be rare in primates. 1,4,9 To the authors' knowledge, fewer than 20 cases of pheochromocytoma have been mentioned or less frequently described in nonhuman primates in the literature. 1,4,9,11,16
We present the clinicopathologic and immunohistochemical data of 6 cases of pheochromocytoma in New World primates, including 2 golden lion tamarins (Leontopithecus rosalia), 2 cotton-top tamarins (Saguinus o. oedipus), 1 black howler monkey (Alouatta caraya), and 1 black-handed spider monkey (Ateles g. geoffroyi). Table 1 details animal data and clinical findings. Ages ranged 13 to 32 years old in 5 primates; case No. 4 was an adult cotton-top tamarin of unknown age. Four of 6 primates died spontaneously, and 2 were euthanized as a result of concurrent disease. Three tamarins had concurrent malignant neoplasia diagnosed before (No. 2) or at necropsy (Nos. 1 and 4). Primate No. 5 was diagnosed with chronic protein-losing nephropathy 4 years before death.
Animal data, clinical findings, and concurrent disease processes in 6 New World primates with pheochromocytoma.∗

∗unk = unknown; d = died; e = euthanatized.
Necropsies were performed on all 6 primates by referring veterinarians (5 cases) or a pathologist (1 case), and representative samples of selected tissues were obtained for histopathology, fixed in 10% neutral buffered formalin, routinely embedded in paraffin, and stained with hematoxylin and eosin. Chromogranin A (6 cases) and synaptophysin (3 cases) were demonstrated with immunohistochemistry following previously reported protocols. 12
Grossly, adrenal lesions consisted of unilateral (Nos. 2–4) or bilateral (No. 6) enlargement in 4 primates. In 2 primates, adrenal masses were not mentioned in the gross necropsy reports submitted by the referring veterinarians. In primate No. 3, there was a nodule 7 mm in diameter adjacent to the cranial pole of the left adrenal (Fig. 1). Both adrenal glands of primate No. 6 were 4–5 cm in greatest dimension, and the right adrenal was adhered to the caudal right liver lobe. Primate No. 4 had a bilobed mass in the dorsal body wall between the kidneys. Primate No. 1 was reported to have red to black discoloration of 1 adrenal gland. Adrenal glands were yellow on the cut surface in primate No. 5. Gross lesions in other organs are summarized in Table 2.
Gross and microscopic findings not pertaining to the adrenal gland in 6 New World primates with pheochromocytoma.

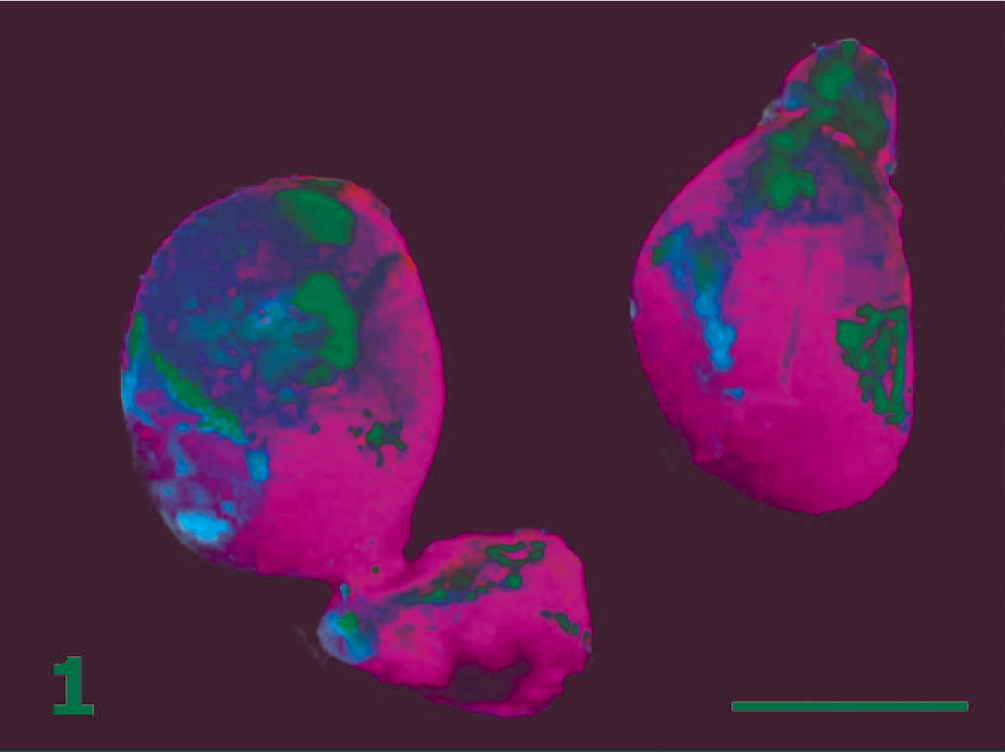
Kidneys and adrenal glands; cotton-top tamarin No. 3. Note the presence of a large mass adjacent to 1 adrenal gland. The right adrenal and kidney are normal (right). Bar = 1 cm.
Histologically, 5 primates had an adrenal neoplasm consisting of a variably well-circumscribed and encapsulated mass expanding the medulla and causing variable compression and atrophy of the surrounding cortex (Fig. 2), which was severe in primate No. 4. In 1 tamarin (No. 3), the neoplasm was adjacent to one of the adrenal glands in the planes of section examined (Fig. 3). In the spider monkey (No. 6), both adrenal glands were affected. Typically, neoplastic cells were arranged in cords, nests, or packets and supported by delicate fibrovascular stroma (Fig. 4). Neoplastic cells occasionally palisaded around dilated capillaries (Figs. 3, 4). Neoplastic cells were polygonal and characterized by a large central, round or oval nucleus with reticular chromatin, small or unapparent nucleoli, and usually moderate amounts of basophilic and granular cytoplasm (Fig. 4). Anisocytosis, anisokaryosis, pleomorphism, and anaplasia were mild. In all cases, the mitotic activity was unapparent or very low. Mild invasive growth into the adjacent cortical tissue and blood vessels was observed in the howler monkey (No. 5). Metastases were not observed in any case.

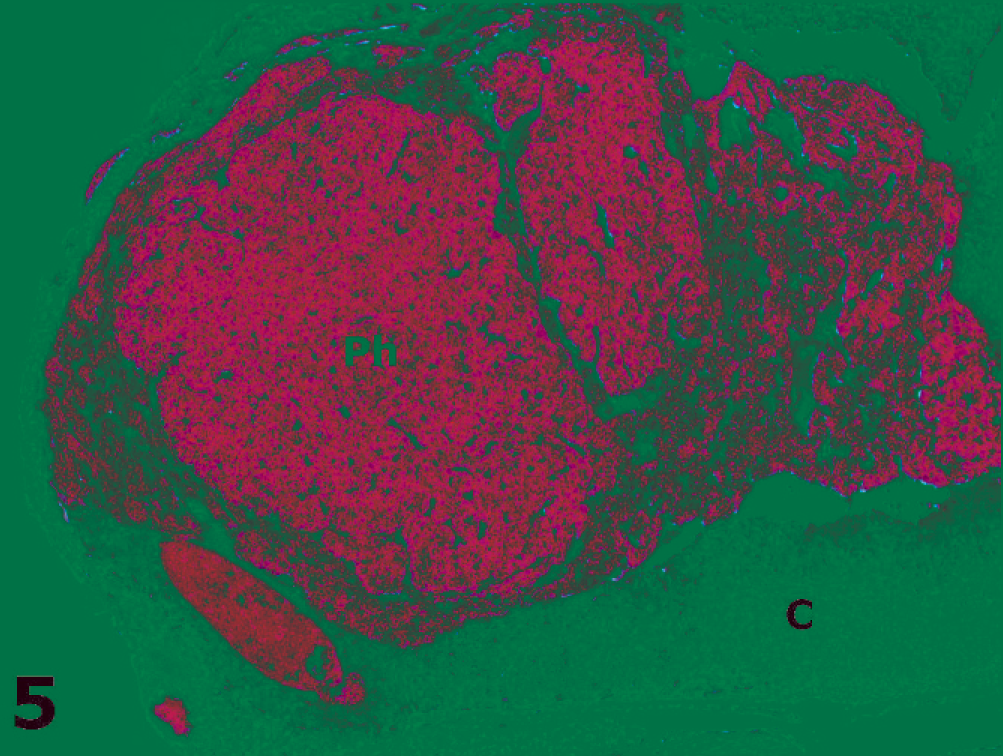
Adrenal gland; golden lion tamarin No. 2. Note a well-circumscribed pheochromocytoma (Ph) expanding the adrenal medulla and compressing (arrows) the cortex (C). HE.
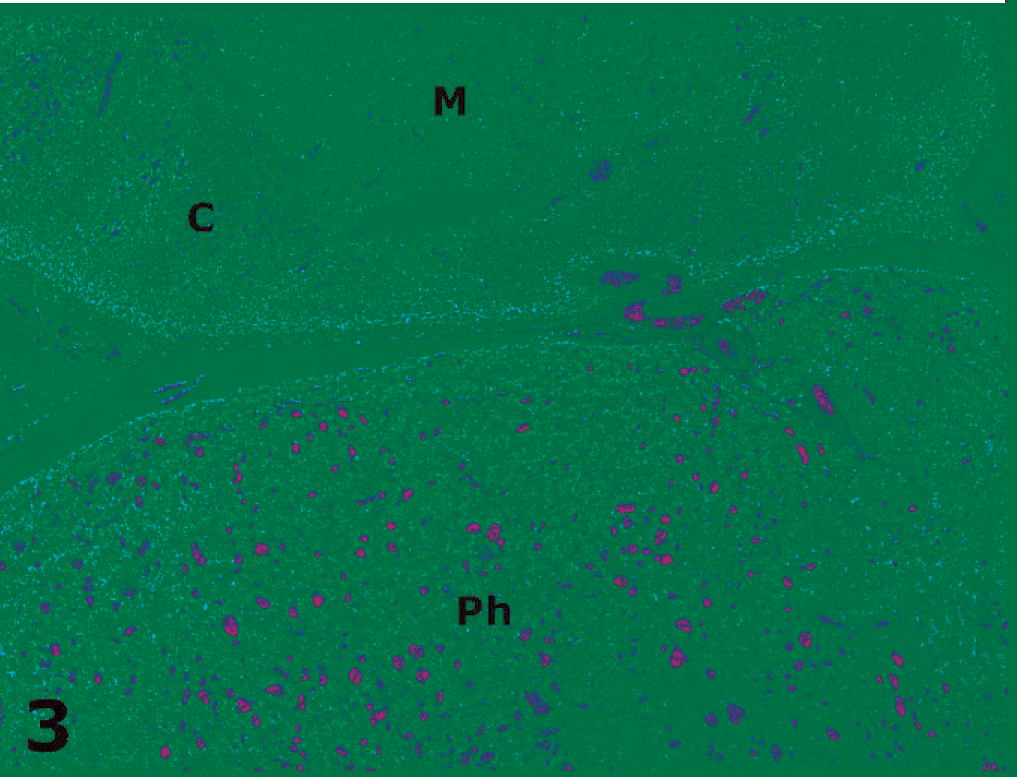
Adrenal gland; cotton-top tamarin No. 3. Note an extra-adrenal, well-circumscribed pheochromocytoma (Ph) adjacent to 1 adrenal gland (C = cortex, M = medulla). HE.

Adrenal gland; cotton-top tamarin No. 3. Neoplastic cells are arranged in cords and nests and supported in delicate fibrovascular stroma; note multifocal palisading of neoplastic cells around dilated sinusoidal vessels. HE.
All 6 primates had either cardiac or vascular lesions or both, which included: mild or moderate multifocal myocardial fibrosis (6/6) with edema and hemorrhage (No. 5) or hypertrophy of cardiomyocytes (No. 3); mild multifocal lymphocytic interstitial myocarditis (No. 3); severe aortic atherosclerosis (Nos. 4 and 5) with aortic thrombosis (No. 5); moderate segmental to circumferential, partial to full-thickness hyaline degeneration with occasional mineralization and intimal fibrosis of myocardial and meningeal vessels (No. 1); marked intimal and medial fibrosis and focal mineralization in the great vessels and mural vessels of the myocardium (No. 4); and severe medial hyperplasia and atherosclerosis of renal arteries associated with glomerular atherosclerosis (No. 5). Primate Nos. 4 and 6 had mild interstitial fibrosis in the kidneys. Additional relevant microscopic findings and concurrent disease processes are summarized in Table 2.
Strong, diffuse cytoplasmic labeling with antibody against chromogranin A was found in the neoplastic cells of all adrenal tumors (Figs. 5, 6). Positive immunostaining for synaptophysin was found in the 3 primates tested (Nos. 2, 4, and 6).
Adrenal gland; golden lion tamarin No. 2. Note diffuse immunostaining for chromogranin A. Immunoperoxidase-DAB, hematoxylin counterstain.

Adrenal gland; cotton-top tamarin No. 3. Prominent cytoplasmic immunostaining for chromogranin A is present in neoplastic cells. Immunoperoxidase-DAB, hematoxylin counterstain.
Pheochromocytoma was diagnosed in 6 primates based on light microscopy and immunohistochemistry. Although the microscopic features of these pheochromocytomas were fairly typical, immunohistochemistry for chromogranin A and synaptophysin was helpful to confirm this diagnosis. These markers are commonly used to characterize pheochromocytomas in human beings and other species. 3,13 All primates either died spontaneously (4/6) or were euthanatized (2/6) as a result of concurrent malignant neoplasia, infection, renal disease, or a combination of several disease processes. Pheochromocytoma was not considered the cause of death in any case but possibly contributed to it in primate No. 3.
Endocrine neoplasms are infrequently described in nonhuman primates. However, endocrine neoplasms were the most frequent neoplasms in 2 retrospective studies in Old 7 and New 4 World primates. A survey of 388 tumors in 363 nonhuman primates included 5 endocrine tumors in 3 New World primates and 34 endocrine tumors in Old World primates. 9 Adrenocortical adenoma appears to be the most frequent endocrine neoplasm in several surveys, particularly in macaques and howler monkeys. 7,9,11 To date, the 2 largest case series of pheochromocytomas in primates consist of 5 cases out of 13 endocrine neoplasms in a survey of 1,106 necropsies of New World primates 4 and 7 cases in a survey of 388 tumors. 1,4,9 Pheochromocytomas have been reported in 9 macaques, 9,11,16 3 golden lion tamarins, 4 2 mantled howler monkeys, 4,9 1 cotton-top tamarin, 1 1 brown spider monkey (Ateles fusciceps), 4 and 1 patas monkey (Erythrocebus patas). 9 Both tamarin species (common in zoos) are also represented in our survey, and new species records of pheochromocytoma in this report are the black howler monkey and black-handed spider monkey. However, there are still few descriptions of endocrine neoplasia, including pheochromocytomas, which appear to be rare in nonhuman primates. To the authors' knowledge, the occurrence of bilateral and extra-adrenal pheochromocytomas has not been previously described in New World primates. Immunohistochemical findings of pheochromocytomas have only been previously described in 1 macaque. 16
There are no reliable morphologic markers of malignancy for pheochromocytomas other than overt invasive behavior into periadrenal structures or the presence of metastases. 2,13 All 6 animals in our series had pheochromocytomas that were considered benign. Mild local invasive behavior was observed in 1 primate (No. 5), but no metastases were observed in any case. Bilateral adrenal involvement and extra-adrenal pheochromocytoma were each present in 1 primate. Bilateral and extra-adrenal pheochromocytomas are infrequent in animals. 2
Lesions reported to be associated with functional pheochromocytomas in other species 2,8 were observed in all 6 primates of this report, particularly myocardial fibrosis (6 animals), arteriosclerosis (3 animals), and renal fibrosis (2 cases). However, in primate No. 5, the arterial lesions consisted of renal and glomerular atherosclerosis, to which howler monkeys appear to be particularly susceptible, and were attributed to a high cholesterol diet. 5 Arteriosclerosis and myocardial fibrosis can result from hypertension caused by catecholamines released from functional pheochromocytomas. 2 Myocardial fibrosis with cardiomyocyte degeneration/necrosis and/or cardiomyopathy with congestive heart failure and arteriosclerosis have been reported in a cotton-top tamarin, a golden lion tamarin, a brown spider monkey, and a macaque with pheochromocytoma. 1,4,16 However, we could not determine whether these pheochromocytomas were functional. The most common method to diagnose functional pheochromocytomas is by detection of catecholamines or their catabolites in the urine. 2,14,15
Unlike previous reports, 4,9 associations with other endocrine neoplasms or adrenal ganglioneuroma were not observed in any primate in this series. However, the spider monkey (No. 6) presented with marked islet hyperplasia, which may have been caused by increased insulin resistance associated with pheochromocytoma and/or pregnancy. 6,17 Endogenous catecholamine excess caused by functional pheochromocytomas can induce or aggravate insulin resistance in patients with type II diabetes and patients with normal glucose tolerance. 17 Pregnant nonhuman primates can develop islet hyperplasia with insulin overexpression and gestational diabetes, which are attributed to increased insulin resistance under progesterone influence. 6,10 Islet hyperplasia was also observed in a male tamarin (No. 3) in this series.
In conclusion, this series of 6 cases of pheochromocytoma in New World Primates adds to the literature 2 new species records (black howler monkey and black-handed spider monkey) and the occurrence of bilateral and extra-adrenal pheochromocytomas in New World primates. Unlike previous reports, 4,9 associations with other endocrine neoplasia and adrenal ganglioneuroma were not found, but 2 primates had pancreatic islet hyperplasia. To date, immunohistochemistry had been only used to support the diagnosis and further characterize nonhuman primate pheochromocytomas in 1 macaque.
Footnotes
Acknowledgements
We thank Jamie Kinion and Christie Buie (Northwest ZooPath) for data retrieval and image editing, respectively, and Roy Brown (Histology Consulting Service) for slide preparation.
