Abstract
The pathogenesis of five different Newcastle disease virus (NDV) isolates representing all pathotypes was examined in commercial and specific pathogen-free (SPF) turkeys. Experimentally-infected birds were monitored clinically and euthanatized, with subsequent tissue collection, for examination by histopathology, by immunohistochemistry for the presence of NDV nucleoprotein, and by in situ hybridization for the presence of replicating virus. Clinically, the lentogenic pathotype did not cause overt clinical signs in either commercial or SPF turkeys. Mesogenic viruses caused depression in some birds. Turkeys infected with velogenic neurotropic and velogenic viscerotropic isolates showed severe depression, and neurologic signs. Histologic appearances for all strains had many similarities to lesions observed in chickens inoculated with the various isolates; that is, lesions were present predominantly in lymphoid, intestinal, and central nervous tissues. However, in general, disease among turkeys was less severe than in chickens, and turkeys could be considered a subclinical carrier for some of the isolates.
Keywords
Newcastle disease is a recurring concern to poultry industries internationally, and outbreaks caused by virulent strains of Newcastle disease virus (NDV) are referred to as Exotic Newcastle disease (END) in the United States. The disease can result in significant economic impact from morbidity and mortality accompanied by the inability to export products from flocks not known to be infected. There are numerous examples throughout the world in recent years of END creating sizable trade barriers. 11, 15 Additionally, when commercial poultry are infected with NDV strains of low virulence, there may also be a negative economic impact, because of a reduction in productivity of infected birds. 17
NDV, synonymous with avian paramyxovirus type 1, is a nonsegmented, single-stranded, negative-sense enveloped RNA virus, belonging to the Paramyxoviridae family, and a member of the genus Avulavirus. 18 The virus genome is 15,186 nucleotides long with six major genes encoding the structural proteins. 10 The fusion protein is known to be a major determinant of NDV virulence; 14 however, recent in vitro studies have demonstrated that the hemagglutinin-neuraminidase protein can contribute significantly to viral spread in the host. 6
The most widely used standard methods for determining virulence of NDV isolates are by intracerebral pathogenicity index (ICPI) and/or intravenous pathogenicity index, and embryonating egg inoculation to determine mean death time. 2 Based on those results, virus strains are classified into three major pathotypes, to include lentogenic (low virulence), mesogenic (moderate virulence), and velogenic (high virulence). In chickens lentogenic strains produce mild or inapparent respiratory infections. Mesogenic strains are associated with low mortality, acute respiratory disease, and neurologic signs in some birds. The velogenic strains are further divided into either neurotropic velogenic NDV (causing respiratory and neurologic signs with high mortality) or viscerotropic velogenic NDV (acute lethal infections with necro-hemorrhagic lesions most obvious in the gastrointestinal tract). 1, 2, 6 The World Organisation for Animal Health (formerly known as the Office International des Epizooties, and still recognized by the abbreviated designation OIE), defines Newcastle disease as notifiable if the virus has an ICPI greater than 0.7 or possesses multiple basic amino acids at the fusion cleavage site. NDV infects a wide range of avian species, with more than 250 species of birds known to be susceptible to NDV infection. 7, 19 The disease in chickens has been well studied; however, the pathogenesis in turkeys is poorly understood and the development of control programs in an outbreak in a turkey-producing area will require a more thorough appreciation of the pathogenesis of the virus in this species. The purpose of this study was to characterize clinicopathologic features and tissue tropism of NDV in turkeys, using five different strains representing all pathotypes.
Materials and Methods
Turkeys
Sixty 4-week-old commercial Medium White tom turkeys, from British United Turkeys of America, and 45 6-week-old specific pathogen–free (SPF) Beltsville white turkeys, 12 from the Southeast Poultry Research Laboratory (SEPRL) flock, were housed in negative pressure isolators at the facilities of the SEPRL (ARS, USDA) under BSL-3 agriculture conditions. Feed and water were provided ad libitum. Commercial turkeys were sampled weekly to test for antibody to NDV, using hemagglutination inhibition. In commercial turkeys, the experiment was not begun until maternal antibodies were no longer detectable, which was at 4 weeks of age. Prior to the experiment, SPF turkeys were also tested for NDV antibody, and they were all negative.
Turkeys were inoculated via instillation bilaterally in the conjunctival sac with 50 µl/eye of inoculum with one of five NDV strains. The control group was inoculated with phosphate buffered saline (PBS) by the same route. The birds were visually examined daily. Two birds of each group were euthanatized at 2, 5, 10, and 14 days postinoculation (dpi), or when severe morbidity was evident. Euthanasia was by intravenous injection of sodium pentobarbital in the brachial vein, and necropsies were performed immediately postmortem. The remaining birds were observed clinically through 14 dpi, with exceptions for those groups that had severe morbidity prompting euthanasia prior to that point.
The following tissues were collected immediately postmortem and fixed by immersion in 10% neutral buffered formalin: eyelid, Harderian gland, bursa of Fabricius, thymus, spleen, crop, esophagus, pharynx, turbinate, proventriculus, duodenum with pancreas, small intestine at Meckel's diverticulum, cecal tonsil, large intestine, caudal thoracic air sac, trachea, lung, heart, liver, kidney, bone marrow, and brain. After 24 hours the formalin was changed to new formalin and approximately 28 hours later the formalin was removed and replaced with PBS, until the tissues could be processed to paraffin. Four µm sections were cut for hematoxylin and eosin staining (HE), immunohistochemistry (IHC), and in situ hybridization (ISH).
Viruses
Five different isolates of NDV were studied: LaSota (lentogenic), first isolated in 1946; 16 Roakin (mesogenic), originally isolated from chickens in New Jersey in 1946; 16 Turkey North Dakota-92-43084 (Turkey ND, velogenic neurotropic), isolated from a turkey flock in North Dakota in 1992; 16 Iowa 1519 (velogenic neurotropic), isolated from a turkey during an ND outbreak in Iowa in the early 1970s; 5 California 1083 (CA 1083, velogenic viscerotropic), isolated from a large outbreak in poultry in southern California in 1971. 16 All the isolates were obtained from the SEPRL repository. The virus was propagated in 9- to 10-day-old embryonated chicken eggs inoculated by the chorioallantoic route, and a dilution of approximately 105.0 embryo infectious doses (EID50) was inoculated intraconjunctivally (0.1 ml/bird). The amounts of inoculum delivered per bird were as follows: LaSota, 104.9 EID50 for commercial turkeys and 103.5 EID50 for SPF turkeys; Roakin, 105.1 EID50 for commercial turkeys and 105.5 EID50 for SPF turkeys; Turkey ND, 103.5 EID50 for commercial turkeys and 105.1 EID50 for SPF turkeys; Iowa 1519, 104.5 EID50 for commercial turkeys and 105.7 EID50 for SPF turkeys; and CA 1083, 104.5 EID50 for commercial turkeys and 105.1 EID50 for SPF turkeys.
Virus isolation and serology
Immediately prior to necropsy, oropharyngeal and cloacal swabs were collected for virus isolation. The swabs were placed in tubes containing 1.5 ml of brain-heart infusion broth with antibiotics (2000 units/ml penicillin G, 200 µl/ml gentamicin sulfate, and 4 µg/ml amphotericin B; Sigma Chemical Co., St. Louis, MO). The swab fluids were centrifuged at 1000 × g for 20 minutes, and undiluted supernatant was inoculated (0.2 ml/egg) into three 9- to 10-day-old SPF embryonated chicken eggs. Inoculated eggs were incubated for 7 days. The infectivity of each inoculum was calculated from the results of the embryonated eggs, and virus-infected dead or surviving embryos were identified by hemagglutination (HA) activity in amnioallantoic fluid harvested from chilled eggs. The verification of NDV in HA-positive samples was assayed by hemagglutination inhibition (HI) test with NDV-specific antiserum. 8 At 10 and 14 dpi, blood for serum was collected for the HI test. Both HA and HI tests were performed by conventional microtiter methods. 4, 8
IHC
Tissues samples of all birds were examined using an IHC protocol to detect viral nucleoprotein (NP) with an antipeptide viral NP antibody. 9 Briefly, after deparaffinization, tissues were subjected to antigen retrieval by microwaving (10 minutes at full power) in Vector antigen unmasking solution (Vector Laboratories, Burlingame, CA). Sections were treated with universal blocking reagent for 7 minutes at room temperature (Biogenex, San Ramon, CA). Incubation with the primary antibody (anti-NP protein), diluted 1 : 8000, was conducted for 2 hours at 37°C (or overnight at 4°C). After washing, sections were incubated with biotinylated goat anti-rabbit antibody and then with either avidin-biotin-alkaline phosphatase or elite-peroxidase (Vector Laboratories). Substrate development was with Vector Red or diaminobenzidine. Sections were counterstained with hematoxylin and coverslipped with Permount for a permanent record.
ISH
Selected sections were processed for ISH. 3 For each strain utilized in our experiment, approximately 640 bases of cDNA from the 5′ end of the matrix gene were cloned into pGEM transcription vectors (Promega, Madison, WI). Then, negative sense riboprobes were generated using RNA polymerase (either SP6 or T7) in the presence of labeled digoxigenin nucleotides. Incorporation of digoxigenin was verified by dot blot. Tissue sections were deparaffinized, rehydrated, digested with 30 µg/ml (or 3 µg/ml for CA 1083 strain) of Proteinase K for 15 minutes at 37°C. Hybridization was conducted overnight (42°C) with approximately 20 ng of each specific probe in hybridization solution consisting of sodium dodecyl sulfate, N-lauroyl-sarcosine, blocking reagent, formamide and standard sodium citrate. After stringent washes, antidigoxigenin alkaline phosphatase was added to the sections to detect bound probes, followed by the chromogen/substrate nitroblue tetrazolium and 5-bromo-4-chloro-3-indolyl-phosphate. Tissues were counterstained lightly with hematoxylin and coverslipped.
Results
Clinical disease, virus isolation, and serology
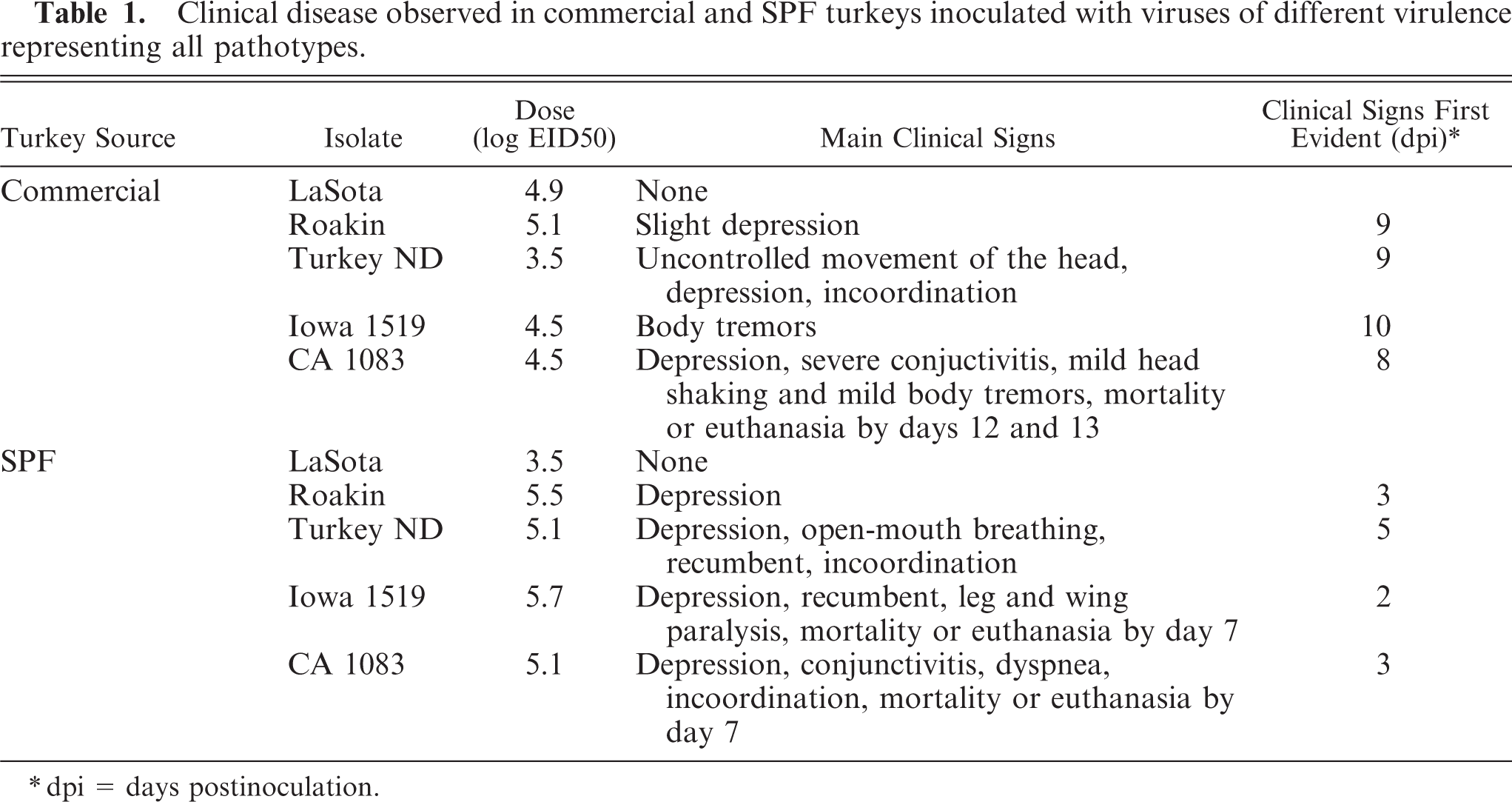
Clinical observations are summarized in Table 1. No abnormal clinical signs were observed in the PBS-inoculated control groups, or in the groups inoculated with the LaSota isolate (lentogen). In both commercial and SPF turkeys inoculated with the Roakin isolate, there was depression noted, but it occurred much earlier in the SPF turkeys than in the commercial birds. Similarly, in both groups of turkeys, predominant clinical signs of central nervous system (CNS) disorders were observed with Turkey ND, but onset was earlier in the SPF birds. The Iowa 1519 isolate induced striking differences between the commercial and SPF turkeys. Commercial turkeys had minimal disease, with body tremors being the only clinical sign noted, and only in one bird, and not until 10 dpi. However, in the SPF birds infected with Iowa 1519, almost all birds were very ill, with CNS signs evident (Fig. 1), and euthanasia of all birds was completed by 7 dpi because of severe morbidity. The CA1083 isolate induced similar clinical signs in both groups, but onset was earlier in the SPF birds. Severe depression and severe conjunctivitis (Fig. 2) were observed in commercial turkeys at 12 and 13 dpi. All SPF turkeys inoculated were dead or euthanatized by 7 dpi.
Clinical disease observed in commercial and SPF turkeys inoculated with viruses of different virulence representing all pathotypes.
∗ dpi = days postinoculation.

Paresis; SPF turkey inoculated with Iowa 1519 isolate.
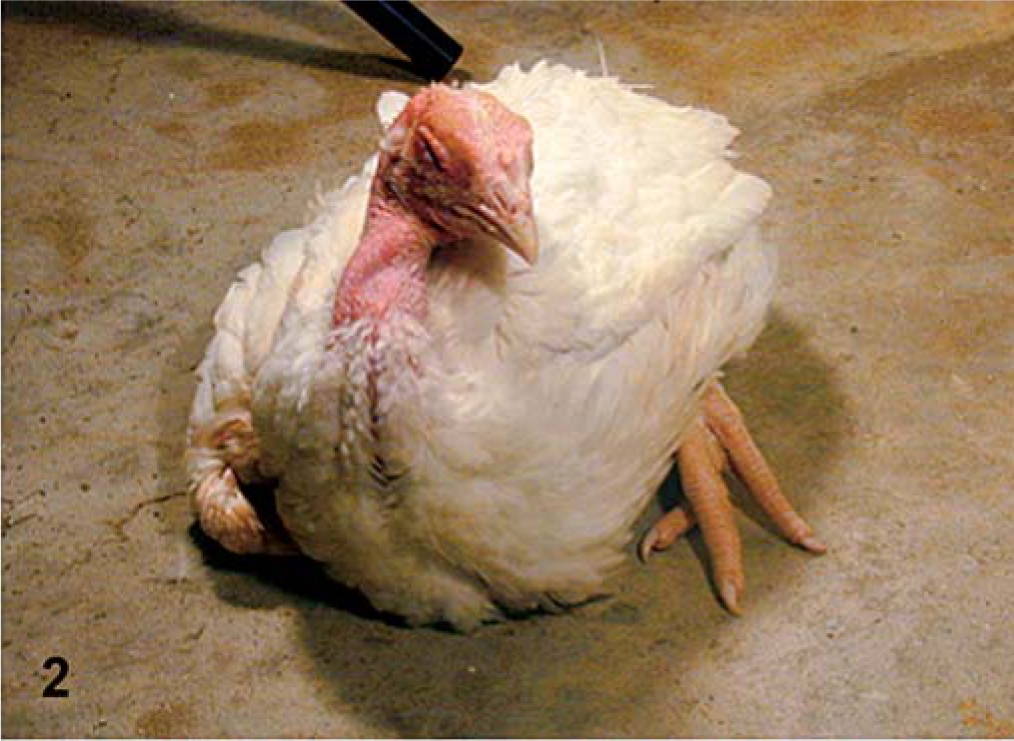
CA 1083–infected commercial turkey. Severe conjunctivitis and depression at 12 dpi.
Viral infection was confirmed by virus isolation and serology in all groups of infected turkeys. In general, virus isolation from oral swabs preceded virus isolation from cloacal swabs. Seroconversion was detected beginning with day 10.
Gross findings
There were petechiae found in the eyelids with all isolates and in both commercial and SPF turkeys. For the LaSota isolate, there were no other gross abnormal findings. For the Roakin isolate, Commercial and SPF turkeys had enlarged and mottled spleens, beginning at 5 dpi. For the Turkey ND isolate, in commercial and SPF turkeys, spleens were mottled and enlarged beginning at 5 dpi. In addition, in the SPF birds, there were multifocal white areas in the pancreas at 14 dpi. For the Iowa 1519 isolate, there were no lesions (other than eyelid petechiae as mentioned above) in commercial turkeys. In SPF turkeys, discrete multiple white foci in the pancreas (2 dpi), perithymic petechiae, cloudy air sacs, and pulmonary hemorrhage were present at 7 dpi. For the CA 1083 isolate, by 2 dpi, in commercial turkeys, spleens were mottled, there were multifocal to coalescent white foci throughout the pancreas at 10 dpi (Fig. 3), opacity of the air sacs, and severe conjunctival edema, with some hematoma formation at 12 and 13 dpi. In SPF turkeys, spleens were enlarged and mottled by 2 dpi, and at 6 dpi, air sacs were cloudy. and there were scattered hemorrhages in the trachea, pharynx, crop, esophagus, and intestine.
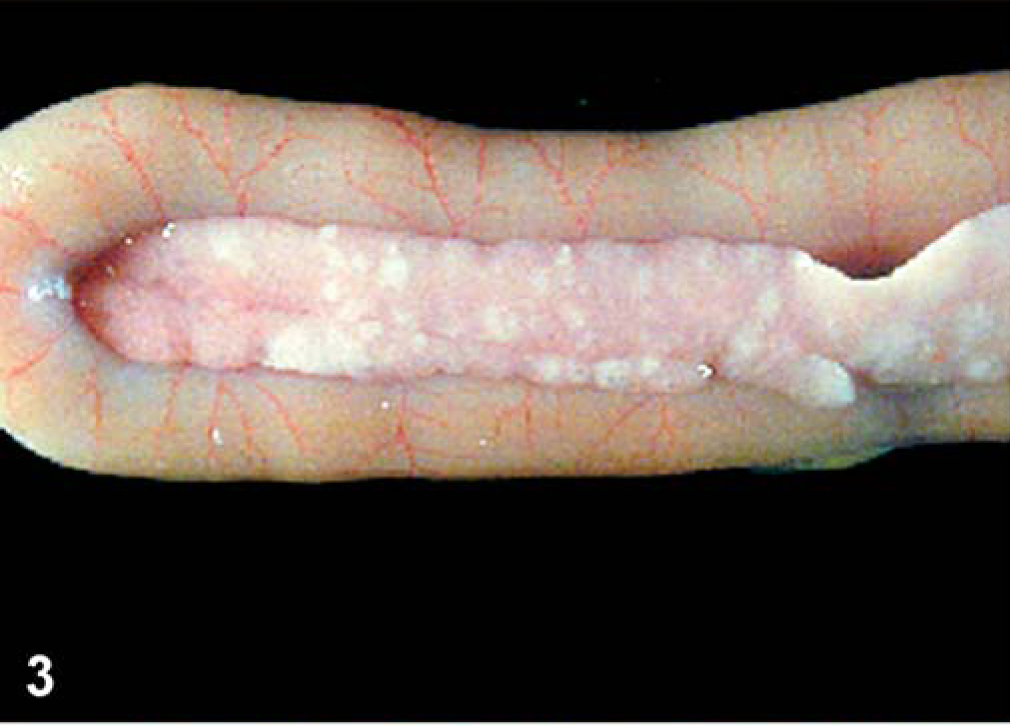
Pancreas; commercial turkey inoculated with CA 1083 isolate; 12 dpi. Multifocal areas of white foci, representing necrosis.
Histopathology
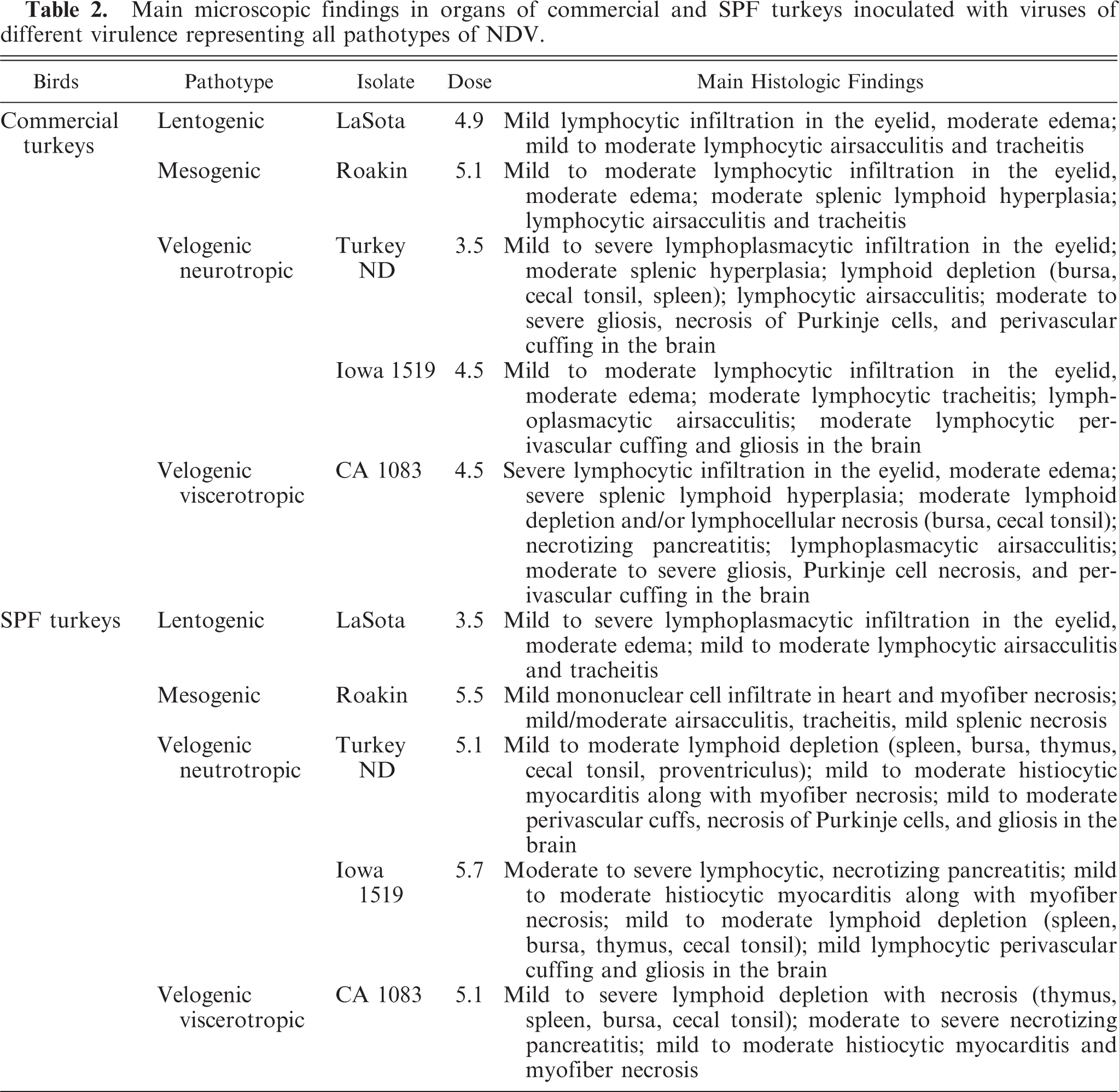
Major histopathologic findings are summarized in Table 2.
Main microscopic findings in organs of commercial and SPF turkeys inoculated with viruses of different virulence representing all pathotypes of NDV.
Commercial turkeys
All infected birds had mild to severe mononuclear inflammatory cell infiltration in the loose connective tissue of the eyelid. For the LaSota isolate, mild lymphocytic infiltrates were present in the submucosa of trachea and pharynx beginning at 5 dpi. Splenic lymphoid hyperplasia was detected at 10 and 14 dpi. For the Roakin strain, there was minimal lymphocytic infiltration in the trachea beginning at 5 dpi, and also in the Harderian gland at 10 dpi. Splenic lymphoid hyperplasia was observed at 14 dpi. For the Turkey ND isolate, the most remarkable lesions observed were in the CNS at 10 and 14 dpi. The lesions were more prominent in the molecular layer of the cerebellum, with hypercellular areas of gliosis and inflammatory infiltrates, accompanied by Purkinje cell necrosis and loss. In the cerebrum and brainstem, there were neuronal necrosis, gliosis, and perivascular cuffing. In nonneural tissue, by 5 dpi, there was modest lymphoid depletion in the spleen, bursa of Fabricius, and cecal tonsil. Individual exocrine cell necrosis with lymphocytic infiltration was observed in the pancreas at 10 dpi. Also, mild lymphocytic inflammation was noted in the trachea, turbinates, pharynx, and kidney. For the Iowa 1519 isolate, mild lymphocytic tracheitis was present in many birds, and one bird had modest lymphocytic infiltration around small capillaries and gliosis in the brainstem and cerebellum. For the CA 1083 isolate, the main lesions were in lymphoid organs and in the brain. Lymphoid depletion with some fibrin deposition in the bursa of Fabricius and cecal tonsil was detected at 5 dpi, and this was followed by scattered necrotic cells randomly distributed through the spleen and then mild splenic lymphoid hyperplasia. There was moderate epithelial cell degeneration with attendant mononuclear inflammatory cell infiltration, usually in submucosal areas, in a number of organs, including the intestinal tract, pharynx, esophagus, turbinates, and pancreas. By 10 dpi, distinct lesions were present in the brain, consisting of perivascular cuffing, gliosis, and neuronal necrosis predominantly in the cerebrum and cerebellum. Purkinje cell necrosis and loss were also observed.
SPF turkeys
All infected birds had mild to severe mononuclear cell infiltration in the eyelid. For the LaSota isolate, mild epithelial cell degeneration, necrosis, and heterophils were present in air sacs, and some lymphocytic infiltrates were present in the trachea, pharynx, turbinates, and Harderian gland. Splenic lymphoid hyperplasia was observed at later time points. For the Roakin isolate, there was mild epithelial cell degeneration, and necrosis with heterophils observed in the pharynx, crop, proventriculus, and air sacs by 5 dpi. Mild to moderate lymphocytic infiltration was observed in the Harderian gland and throughout the gastrointestinal tract. Also at this time, there was moderate necrosis in the spleen, occasionally with fibrin deposits replacing lymphoid sheaths. Individual exocrine pancreatic cell necrosis with lymphocytic infiltration was present at 5 dpi. In the same interval, the heart had mild to moderate monocytic inflammatory cell infiltrates, with some myofiber necrosis. For the Turkey ND and Iowa 1519 isolates, both velogenic neurotropic isolates induced lesions similar in character. However, lesions observed were slightly more severe with Turkey ND than with Iowa 1519. There was destruction of lymphoid areas, specifically spleen, bursa of Fabricius, thymus, proventriculus, and cecal tonsil, and often fibrin was present. Necrosis extended from lamina propria and submucosa through to the epithelium with ulceration and hemorrhage in the cecal tonsil of one Turkey ND–infected bird. In the trachea, pharynx, esophagus, crop, and air sacs, mononuclear inflammatory cell infiltrates were present around vessels, and there was regular epithelial cell degeneration or necrosis. Some birds also had mild mononuclear inflammatory cell infiltration in the kidneys and liver, often associated with necrosis of selected parenchymal cells. In Iowa 1519–infected birds, there was moderate to severe multifocal to coalescent lymphocytic necrotizing pancreatitis at 5 and 7 dpi. In SPF turkeys infected with either of the two velogenic neurotropic viruses, there was mild to moderate mononuclear inflammatory cell infiltration in the heart with myofiber necrosis. The earliest brain lesion detected was with Turkey ND–infected birds at 5 dpi, becoming more severe at 10 dpi; the lesions consisted of mild multifocal lymphocytic perivascular cuffs, and neuronal necrosis with moderate gliosis in the cerebrum, brainstem, and cerebellum. Lack of Purkinje cells was also observed. In some birds, vacuolation and gliosis in the cerebellar white matter was present. In birds infected with CA 1083 isolate, the main lesions were in the lymphoid organs. From 5 dpi, lymphoid necrosis, with abundant fibrin deposition, and hemorrhage were evident in multiple lymphoid tissues at 5 dpi (Fig. 4). This was followed directly by necrosis of overlying intestinal epithelium and severe ulceration. Epithelial cell degeneration and necrosis, often extensive, were present in pharynx, esophagus, crop, nasal mucosa, air sacs, and proventriculus; these conditions were usually accompanied by a mononuclear cell infiltrate. In addition, the tracheitis, and pharyngitis were hemorrhagic in character. Mononuclear inflammatory cell infiltration with parenchymal necrosis was observed in kidneys, liver, and heart (Fig. 5). Focal areas of necrosis were observed in the bone marrow by 5 dpi. CNS lesions were prominent, and were first observed at 5 dpi, with moderate to severe infiltration of mainly lymphocytes surrounding small capillaries, gliosis, and neuronal degeneration in the cerebrum and brainstem. In the molecular layer of the cerebellum, there were moderate gliosis, perivascular cuffing, and Purkinje cell necrosis and loss. Vacuolation was observed in the cerebellar white matter, and mild lymphocytic meningitis was observed in some of these turkeys.
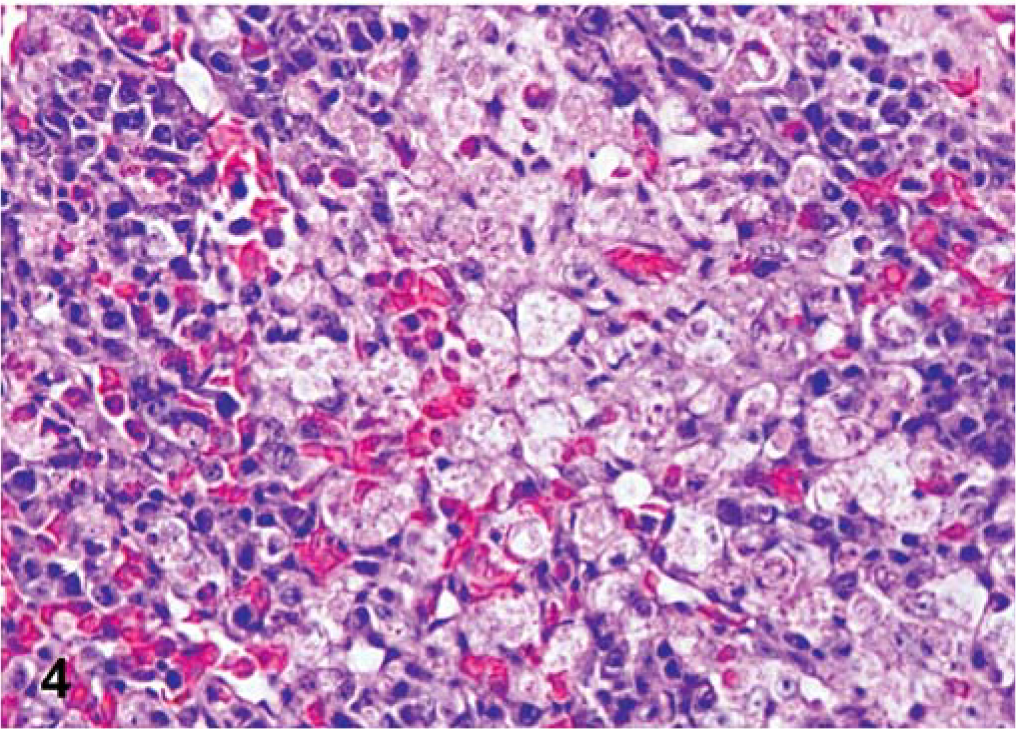
Spleen; SPF turkey inoculated with velogenic viscerotropic CA 1083 at 5 dpi. Splenic necrosis with significant amount of fibrin. HE.
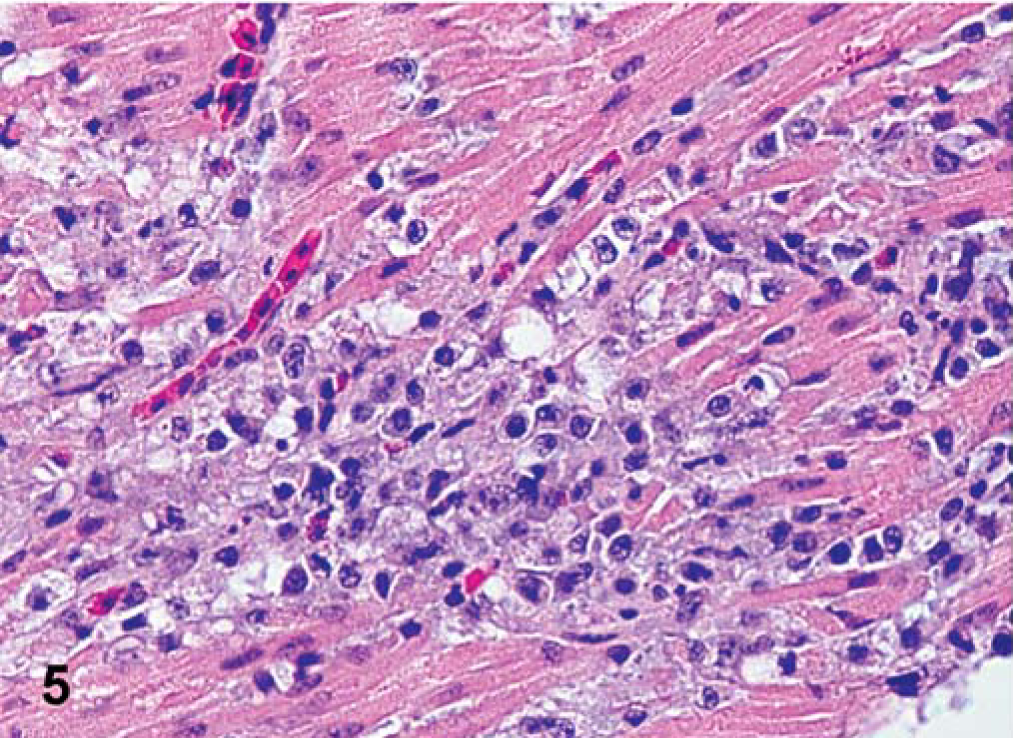
Heart; SPF turkey inoculated with CA 1083 isolate at 7 dpi. Multifocal areas of mononuclear cell infiltration and myofyber necrosis. HE.
IHC
IHC results are presented in Table 3. The tissues of the PBS-inoculated turkeys were all consistently negative for NDV with IHC. For all those tissues with positive staining, the signal was invariably restricted to the cytoplasm. For LaSota (lentogen), there was minimal IHC staining in commercial turkeys, limited to epithelial cells in the trachea, and no staining was detected in tissues of SPF birds. For Roakin (mesogen), in commercial turkeys, at 5 dpi, there were few scattered positive epithelial cells in the trachea of infected birds. In SPF-infected turkeys infected with this isolate, there was discrete positive staining in the esophagus (periglandular large cells) and in the heart (myofiber, and inflammatory cell) at 5 dpi. For velogenic neurotropic isolates, in commercial birds infected with the Turkey ND strain, IHC positive cells were detected in macrophage-type cells within periarteriolar and periellipsoid sheaths of the spleen and in Purkinje cells of the brain as well as in neurons and glial cells in both cerebrum and brainstem at 10 dpi (Fig. 6). Commercial turkeys infected with Iowa 1519 isolate had discrete positive staining in the epithelial cells of the trachea by 5 dpi. For SPF-infected turkeys, both groups of birds inoculated with velogenic neurotropic strains had similar positive staining in multiple organs; however, a lesser number of organs was positive in birds inoculated with Iowa 1519. With Turkey ND isolate, at 5 dpi, viral antigens were detected in epithelial and lymphoid aggregates of the eyelids, esophagus, intestine, lung, spleen, and kidneys (interstitial lymphocytes, tubular cells). In many organs, such as pancreas, proventriculus, duodenum, cecal tonsil, bursa of Fabricius, and others, the majority of positive staining was detected in infiltrates of lymphocytes and macrophages in the muscularis and/or smooth muscle of vessels of these organs. Less intestinal involvement was observed in the Iowa 1519–inoculated SPF turkeys. In the heart, multiple macrophages and myocytes were positive in both groups (Fig. 7). Also, moderate amounts of viral antigen were present in pancreatic exocrine cells and infiltrates. By 10 dpi, in Turkey ND-infected SPF birds, there were moderate numbers of positive cells present in the brain (Purkinje cells, neurons, and glial cells).
Extent of viral infection characterized by the presence of NDV detected by IHC in tissues of commercial and SPF turkeys.∗
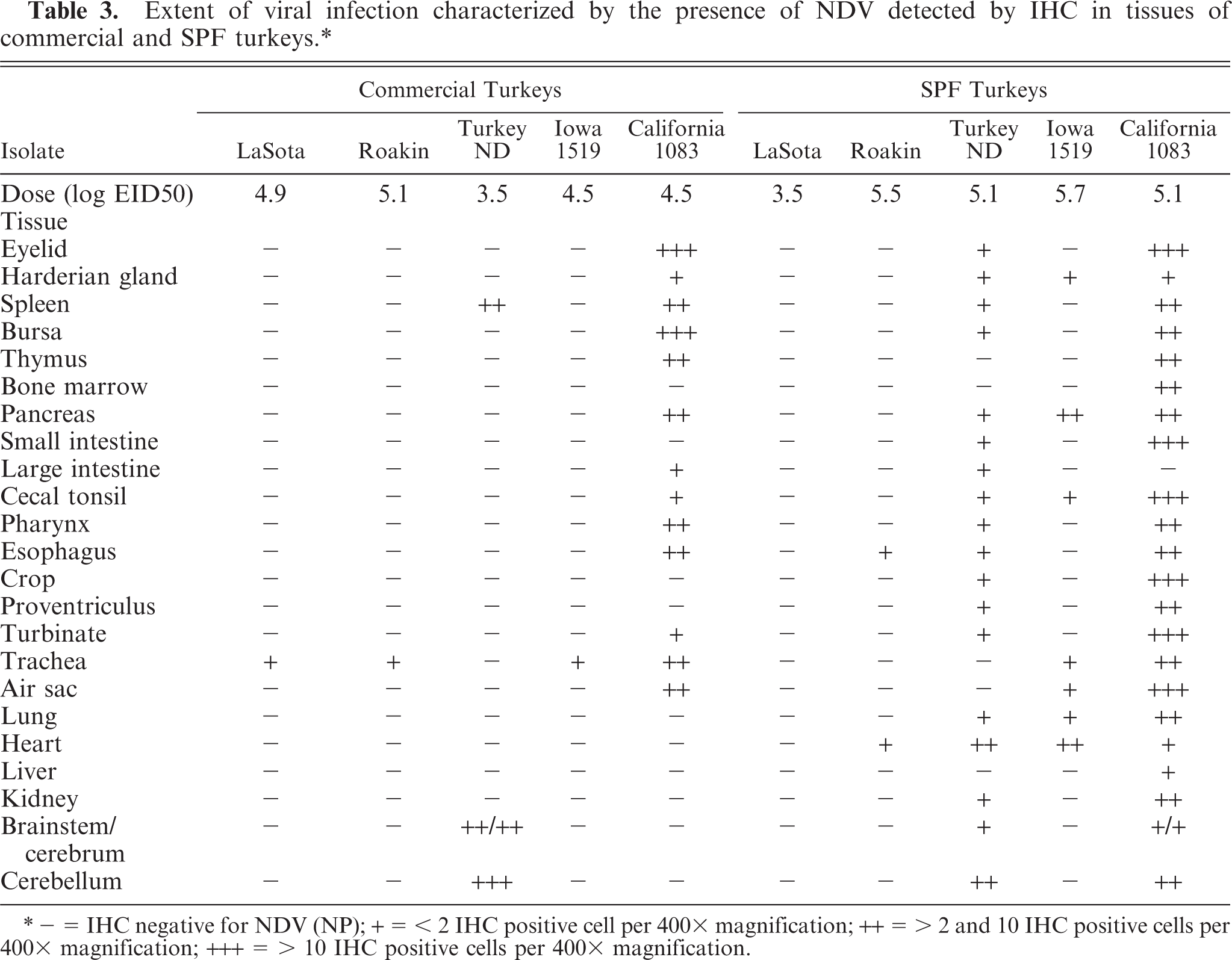
∗ − = IHC negative for NDV (NP); + = < 2 IHC positive cell per 400× magnification; ++ = > 2 and 10 IHC positive cells per 400× magnification; +++ = > 10 IHC positive cells per 400× magnification.
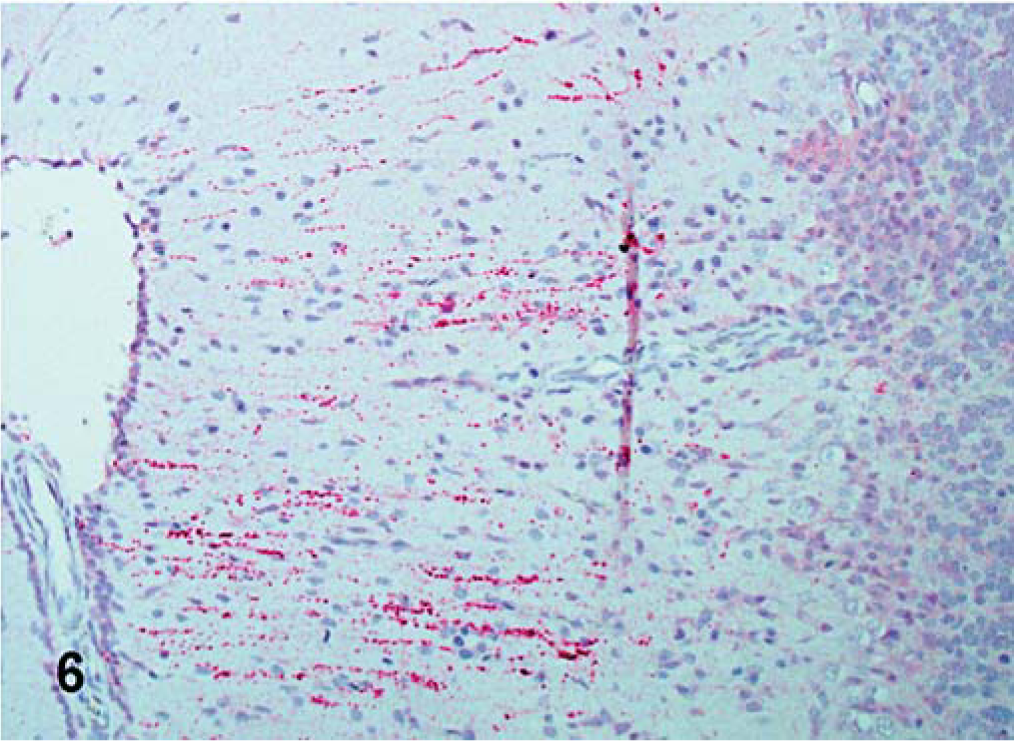
Cerebellum; commercial turkey inoculated with velogenic neurotropic Turkey ND isolate at 10 dpi. Dendrites of Purkinje cells, and glial cells are positive for viral NP (red-stained) in the molecular layer of the cerebellum. IHC, avidin-biotin alkaline phosphatase, hematoxylin counterstain.
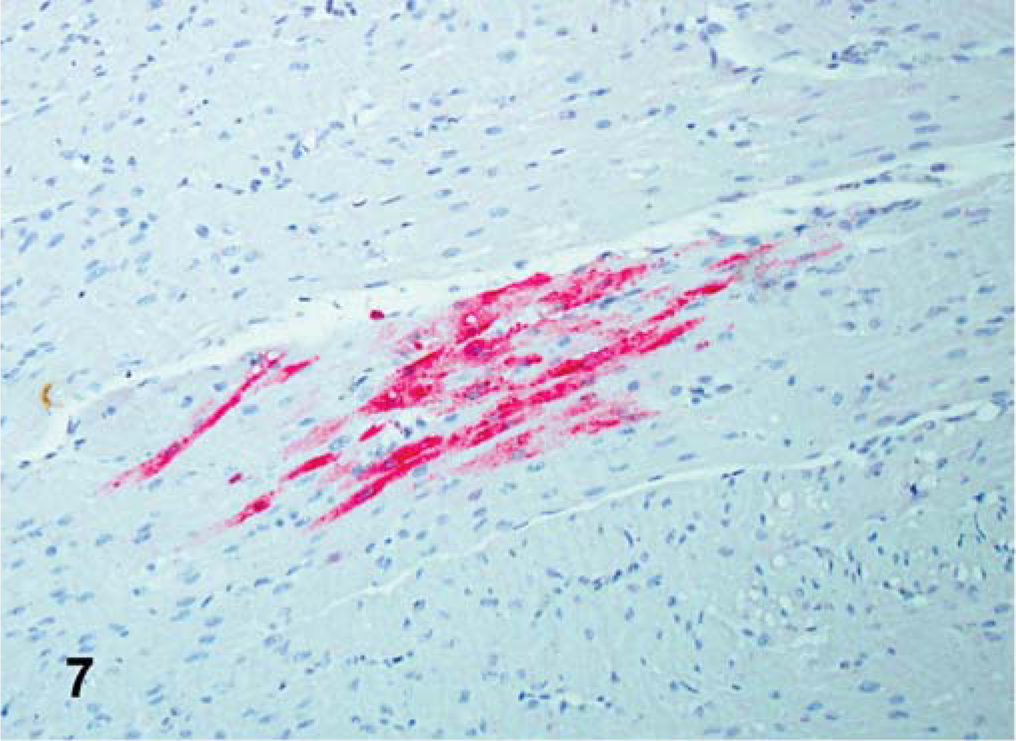
Heart; SPF turkey infected 5 days previously with the Turkey ND isolate. Presence of viral NP in myocytes and monocytes (red stained). IHC, avidin-biotin alkaline phosphatase, hematoxylin counterstain.
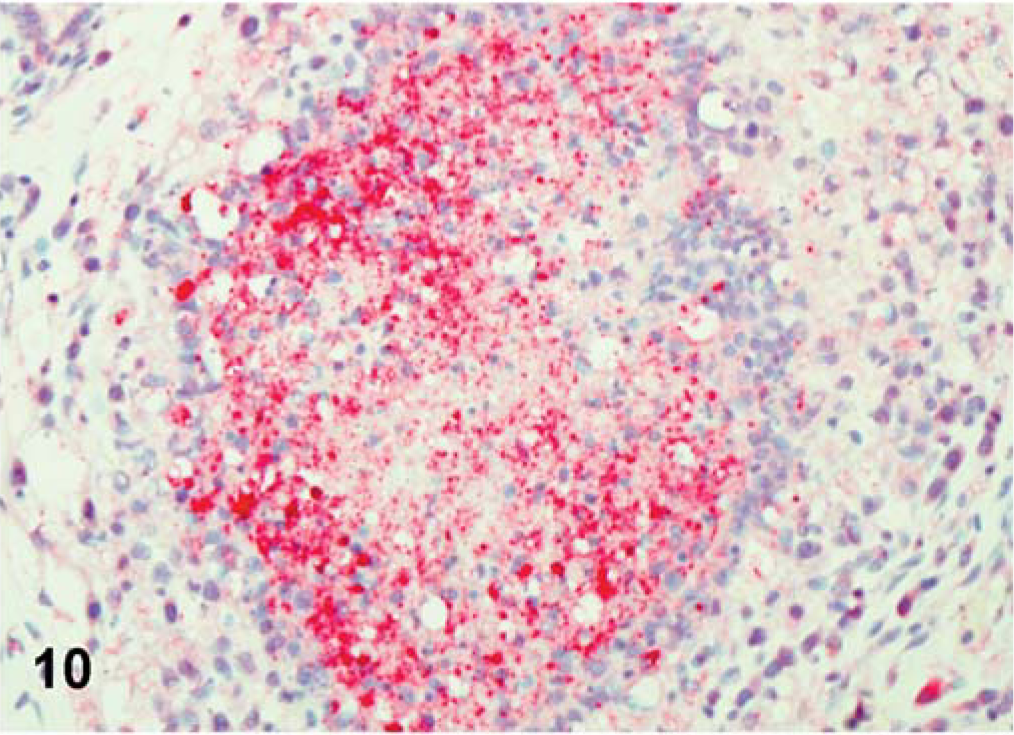
For CA 1083 (velogenic viscerotropic), the viral antigen distribution was similar between commercial and SPF turkeys. However, the number of infected tissues and the amount of viral protein detected in organs varied greatly within both groups. Both commercial and SPF turkeys had abundant viral NP antigen in the lymphoid aggregates and/or epithelium of the conjunctiva of eyelid at 2 dpi (Fig. 8). By 5 dpi, viral NP-positive cells were present mainly in lymphoid organs (Fig. 9) and in respiratory and digestive tracts, along with multiple visceral organs (Fig. 10). Epithelial cells were strongly positive in the trachea, pharynx, turbinates, air sacs, esophagus, crop, pancreas, and mucosal epithelial cells of the proventriculus. In SPF turkeys, there was extensive viral staining in the brain (glial cells and neurons of cerebrum and brainstem) at 7 dpi, which was the last day of the experiment in this group. In contrast, staining in the brain was present at a later time period, 10 and 12 dpi, among commercially obtained turkeys.
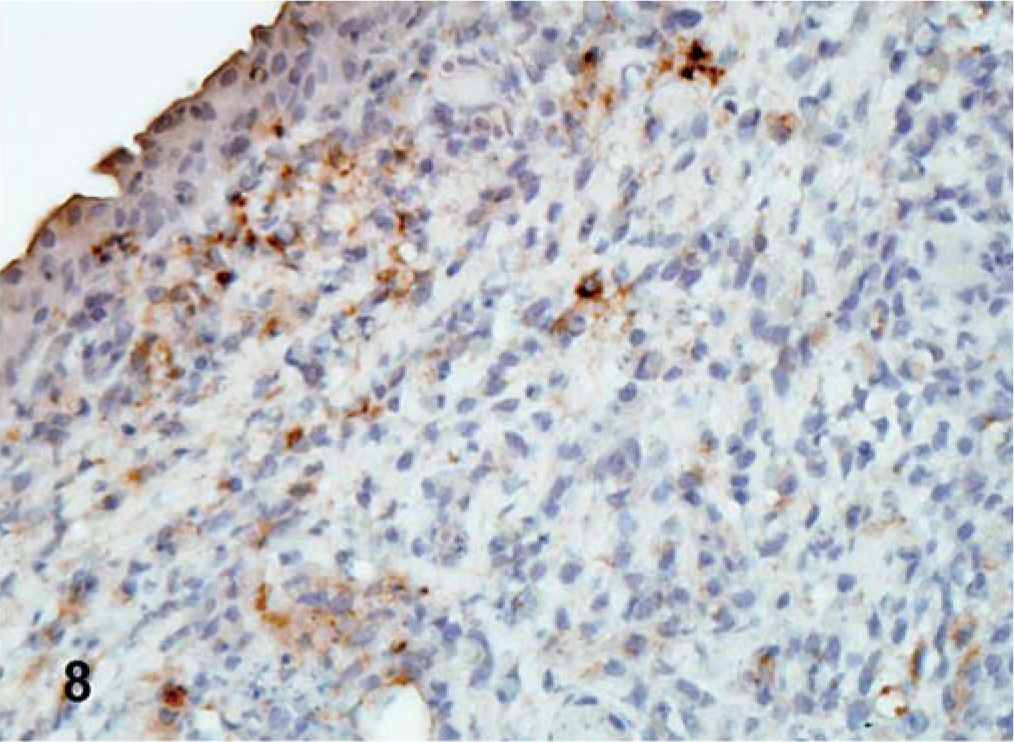
Eyelid; commercial turkey inoculated with CA 1083 isolate at 2 dpi. Viral NP-positive lymphocytes in the lymphoid aggregates of the eyelids. IHC, avidin–biotin–peroxidase, hematoxylin counterstain.
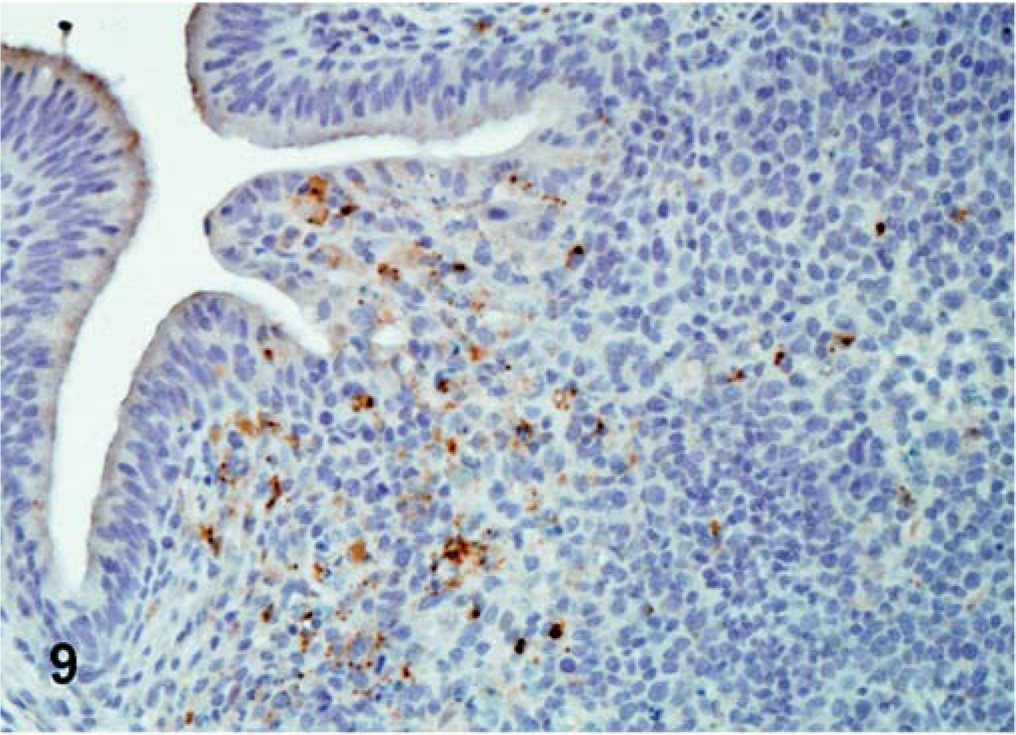
Bursa; commercial turkey infected with CA 1083 isolate at 5 dpi. Multiple lymphocytes positive for viral NP (brown-stained) in the bursa. IHC, avidin–biotin–peroxidase, hematoxylin counterstain.
Bursa of Fabricius; SPF turkey inoculated with CA 1083 isolate at 5 dpi. Viral NP-positive cell (red-stained) was detected in necrotic follicles of the bursa. IHC, avidin–biotin alkaline phosphatase, hematoxylin counterstain.
ISH
Commercial turkeys
The ISH performed in selected tissues detected some additional indications of viral replication. In all cases, ISH staining was cytoplasmic. In Turkey ND–infected birds, positive staining was detected in the lymphoid aggregates of the cecal tonsil. In birds inoculated with Iowa 1519, tracheal epithelial cells were positive at 5 dpi. In birds inoculated with the CA 1083 isolate, moderate epithelial staining was present in the air sacs (Fig. 11), and at 10, 12, and 13 dpi, minimal numbers of positive cells were detected in the cecal tonsil, esophagus, turbinates, pharynx, and brain. In SPF turkeys inoculated with Iowa 1519 isolate, small amounts of replicating virus were detected in the bursa of Fabricius, small intestine, and pharynx. There was extensive staining in exocrine pancreatic cells and in lymphoid aggregates (Fig. 12). At 5 dpi, in CA 1083–infected birds, there were moderate numbers of positive cells in the large intestine, and cecal tonsil (6 dpi).
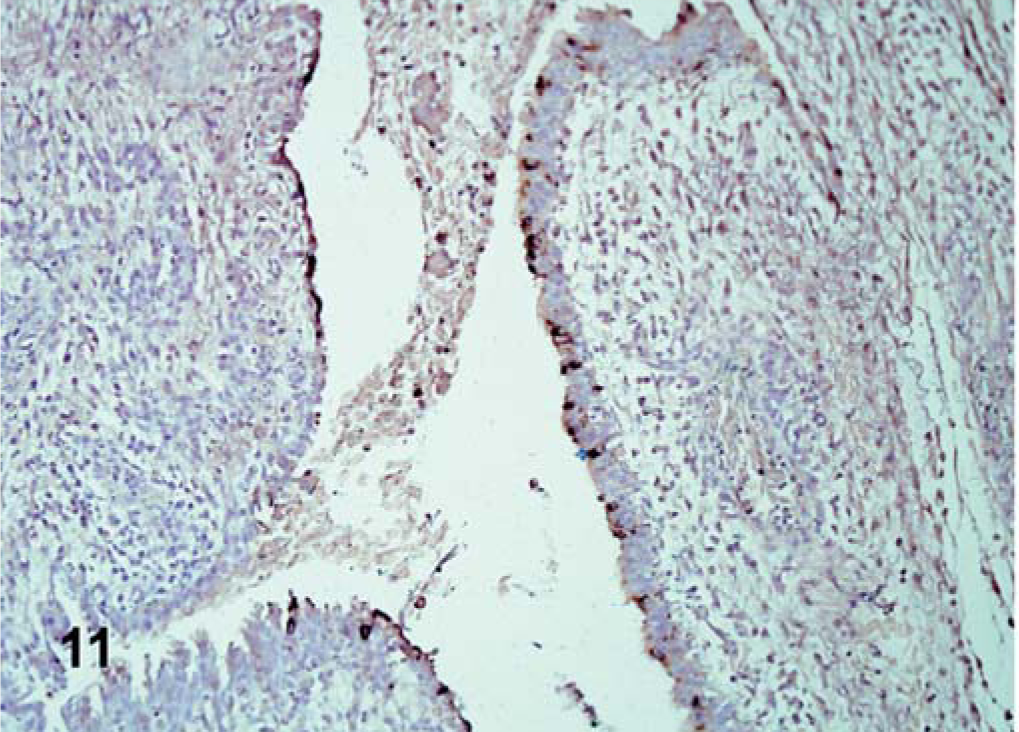
Air sac; CA 1083–infected commercial turkeys. Positive epithelial cells for mRNA in the air sac. ISH, hematoxylin counterstain.←
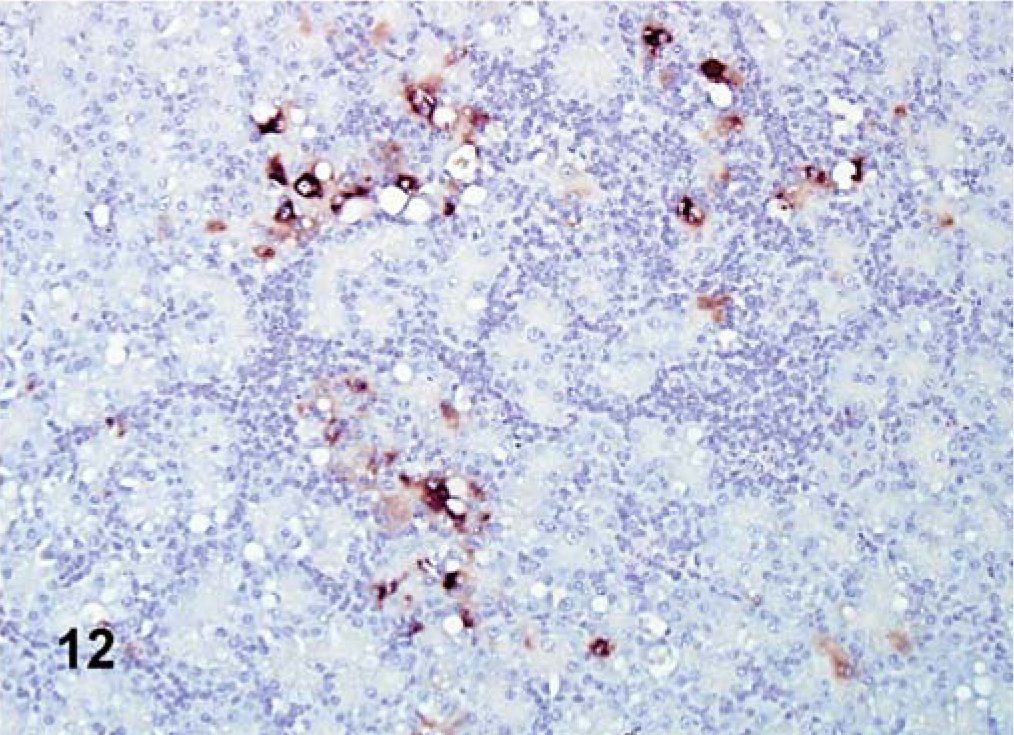
Pancreas; SPF turkey inoculated with Iowa 1519 at 5 dpi. Modest mRNA detected in degenerative exocrine pancreatic cells and lymphocytes (brown stained). ISH, hematoxylin counterstain.
Discussion
Newcastle disease is regarded as a very significant pathogen for domestic chickens. The virus is capable of infecting a large number of avian species, but relatively little is known about the pathogenesis of viruses of different pathotypes in turkeys. Results from this study demonstrate that commercial and SPF turkeys can be infected with various isolates of NDV, and the differences in susceptibility to viral infection may be related to breed and strain differences.
With lentogenic strains, clinical disease in both commercial and SPF turkeys was insignificant. Histopathologic lesions were minimal and nonspecific, and so it is unlikely that infection would be diagnosed during routine diagnostic pathology surveillance. In SPF turkeys, the inoculum dose per bird was less than the inoculum dose given to commercial turkeys, which might explain the lack of any tissues positive by IHC. However, it was apparent that replication occurs readily, as demonstrated by viral isolation following inoculation with lentogenic NDV. These results are similar to what has been reported in chickens experimentally inoculated in a similar manner with B1, another lentogenic NDV strain. 4
The mesogenic Roakin strain caused mild clinical disease, consisting primarily of depression, in both commercial and SPF turkeys. Microscopically lesions were minimal. An unusual feature detected in the SPF birds was the presence of mild myocarditis. Such a feature might alert a diagnostic laboratory to undertake further testing to identify an etiologic agent. The sites of replication, with the exception of the myocardium, were primarily restricted to mucosal surfaces, specifically trachea and esophagus. Presence of virus in the myocardium of SPF turkeys indicates a systemic spread and a potential for more severe disease in this group of birds. However, drawing conclusions about breed variability should be done with caution, because doses of the inocula varied between the two groups. With the Roakin isolate, the SPF birds received a slightly higher dose (100.4 EID50 greater). In comparison to results in chickens experimentally infected with mesogenic isolates, findings in SPF turkeys were similar, with evidence of systemic infection, including myocardial replication. 4
With velogenic viscerotropic and neurotropic strains, there were striking differences between commercial and SPF turkeys. In general, clinical signs in commercial turkeys were less severe and of later onset than those observed in SPF birds. The amount of virus dose per bird in the commercial turkeys was less than in SPF turkeys, which may be the reason for the difference.
With one of the velogenic neurotropic isolates, Turkey ND, clinical signs were similar in both commercial and SPF turkeys, however with an earlier presentation of the disease observed in SPF turkeys. Once again, this may be attributable to the difference in inocula, with the SPF birds receiving 101.6 more EID50. Histologically, the main site of damage in commercial turkeys was the brain. This Turkey ND strain was capable of producing substantial brain lesions with large amounts of viral protein and nucleic acid present in the neural tissue. In SPF turkeys inoculated with the Turkey ND isolate, histologic lesions were also observed in other organs and as visualized by IHC. There was an abundant amount of virus in multiple organs, as well as in brain. Previous studies involving experimental infection of chickens with the Turkey ND isolate revealed mild histologic damage of brain, but very minimal amounts of virus in neural tissue. 4 It is worth noting that this NDV strain was first isolated from turkeys, and it may be that it is more adapted to turkey tissues, allowing for greater ease of replication in turkeys than in chickens. 16
With the Iowa 1519 isolate, the second velogenic neurotropic strain used in this study, clinical signs of disease varied greatly between commercial and SPF turkeys. All the SPF turkeys had severe clinical disease and died or were euthanatized by 7 dpi. In contrast, commercial turkeys presented with mild clinical illness, not nearly of the same severity as that observed in the SPF birds. Although discrete brain lesions were observed at 7 dpi in SPF turkeys, there was no virus detected in this tissue by either IHC or ISH. It is presumed that the absence of detectable virus in the brain is because of the early stage of the disease, and that although the virus was already causing damage, the antigen or nucleic acid was not present in sufficient amount in the particular 4-µm sections that were examined histologically. In contrast to the SPF birds, in commercial turkeys, virus distribution was restricted to the trachea. There are no existing reports of disease in chickens infected with Iowa 1519 so an interspecies comparison for this strain was not possible.
In commercial and SPF turkeys inoculated with the velogenic viscerotropic strain, CA 1083, there were enormous differences in clinical disease. The clinical disease was much more severe in SPF than in commercial turkeys. The most striking microscopic lesions detected in commercial turkeys were in lymphoid organs, pancreas, and brain, where necrosis and inflammation were evident. In comparison, SPF turkeys had extensive microscopic lesions in all lymphoid organs and the brain, as well as a variety of other organs, including the heart, pancreas, liver, kidney, as well as respiratory and digestive tracts. In commercial and SPF turkeys, the presence of viral antigen or replicating virus was observed in a number of parenchymatous organs and not just at mucosal surfaces, reflecting systemic spread. However, the number of infected organs and the amount of positive IHC or ISH staining was less in commercial than in SPF turkeys. The pathogenesis of CA 1083 isolate has been studied in chickens and in general, when inoculated into chickens, there is wide dissemination of virus, with death by 5 dpi. 4, 13 From our results, it appears that this virus is also quite virulent for turkeys, but in comparison to chickens, turkeys survive longer after infection, presumably with correspondingly longer periods of viral shedding prior to succumbing to the infection.
In summary, our results indicate that all pathotypes of NDV can infect turkeys. In general, disease is milder than in chickens, but of similar character. This study confirms that NDV can cause a wide range of disease, depending on a multiplicity of factors, including strain of the virus, and species of bird.
Footnotes
Acknowledgements
A. M. Piacenti was supported by a scholarship from Fundação Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES- Brazil). This research was funded by Southeast Poultry Research Laboratory (USDA-ARS) and the US Poultry & Egg Association Project #368. The authors acknowledge the technical support of Phillip Curry and the animal care support of Roger Brock.
