Abstract
Pigeon paramyxovirus serotype 1 (PPMV-1) is a globally distributed, virulent member of the avian paramyxovirus serotype 1 serogroup that causes mortality in columbiformes and poultry. Following introduction into the United States in the mid-1980s, PPMV-1 rapidly spread causing numerous mortality events in Eurasian collared-doves (Streptopelia decaocto) (ECDOs) and rock pigeons (Columba livia) (ROPIs). The investigators reviewed pathological findings of 70 naturally infected, free-ranging columbiforms from 25 different mortality events in the United States. Immunohistochemistry targeting PPMV-1 nucleoprotein was used to determine the tissue distribution of the virus in a subset of 17 birds from 10 of the studied outbreaks. ECDOs (61 birds) and ROPIs (9 birds) were the only species in which PPMV-1-associated disease was confirmed by viral isolation and presence of histologic lesions. Acute to subacute tubulointerstitial nephritis and necrotizing pancreatitis were the most frequent histologic lesions, with immunolabeling of viral antigen in renal tubular epithelial cells and pancreatic acinar epithelium. Lymphoid depletion of bursa of Fabricius and spleen was common, but the presence of viral antigen in these organs was inconsistent among infected birds. Hepatocellular necrosis was occasionally present with immunolabeling of hypertrophic Kupffer cells, and immunopositive eosinophilic intracytoplasmic inclusion bodies were present in hepatocytes of 1 ECDO. Immunopositive lymphocytic choroiditis was present in 1 ECDO, while lymphocytic meningoencephalitis was frequent in ROPIs in absence of immunolabeling. This study demonstrates widespread presence of PPMV-1 antigen in association with histologic lesions, confirming the lethal potential of this virus in these particular bird species.
Keywords
Viruses belonging to the class II genotype VI of the avian paramyxovirus serogroup 1, also referred to as pigeon paramyxovirus serotype 1 (PPMV-1), are globally distributed, virulent members of the genus Avulavirus, family Paramyxoviridae, that cause mortality in columbiforms and poultry. 10 PPMV-1 has caused more than 23 Newcastle disease (ND) outbreaks in poultry in Great Britain 3,17 and Canada. 34 In the majority of these events, the source of infection was thought to be contact with feral rock pigeons (ROPIs) or their feces. 2,17 Based on the F glycoprotein cleavage site deduced amino acid motif, all PPMV-1 viruses are considered virulent in poultry, and most strains are phenotypically mesogenic as assessed through intracerebral pathogenicity index in 1-day-old chickens (Gallus gallus). 12
The first PPMV-1-associated cases in the United States were described in domestic and feral ROPIs from New York State in 1984. 31 Since then, mortality events caused by PPMV-1 have been confirmed in multiple other locations in both ROPIs and Eurasian collared-doves (Streptopelia decaocto) (ECDOs). 5,31,32
PPMV-1 has been repeatedly isolated from oropharyngeal or cloacal swabs from numerous columbiforms, including ROPIs, ECDOs, and mourning doves (Zenaida macroura), 14,22 and occasionally from birds belonging to the orders Accipitriformes, Suliformes (gannet [Morus species]), and Galliformes (chicken [G. gallus], pheasant [Phasianus colchicus], turkey [Meleagris species], and chukar partridge [Alectoris chukar]. 14 It is not known whether these occasional isolations are the result of spillovers from the natural reservoirs (columbiforms) or whether intermediate hosts are involved. However, to date, there have been no published reports of PPMV-1 causing ND outbreaks in commercial poultry in the United States. Nevertheless, some strains of PPMV-1 are not detected by the regular real-time reverse transcription polymerase chain reaction (RT-PCR) for the fusion protein gene. 21 Therefore, it is possible that low-level mortalities might have gone undiagnosed. 24
In published descriptions of lesions associated with PPMV-1 infection in columbiforms in North America, confirmation of disease was based on viral isolation from 1 or more internal organs (brain, liver, kidney, or spleen) and the presence of suggestive histologic changes such as tubulointerstitial nephritis, pancreatitis, lymphoid depletion of the spleen and bursa of Fabricius, multifocal hepatocellular necrosis or lymphocytic choroiditis, and meningoencephalitis. 5,19,31,32
Paired comparison of microscopic lesions and virus antigen distribution by immunohistochemistry (IHC) has proven useful to help characterize the pathogenesis in galliformes experimentally infected with PPMV-1. 14,25,26,35 Conversely, IHC has been rarely used for this purpose in columbiforms. Specifically, PPMV-1 nucleoprotein (NP) immunolabeling has been associated with renal tubular necrosis and splenic lymphoid depletion in 1 ECDO in the United States 32 and 1 case of renal tubular necrosis in a feral ROPI in Japan. 30 The objective of the present study is to demonstrate the lethal potential of PPMV-1 infections in certain species of columbiforms in the United States, to help illuminate the pathogenesis of the disease in its natural host, and to suggest the most likely pathways of viral shedding.
Methods
We reviewed pathology reports and hematoxylin and eosin (HE)–stained histologic slides of 70 birds from 25 PPMV-1-associated mortality events in 10 states of the United States from 2009 to 2016. Postmortem investigation of 24 events took place at the USGS National Wildlife Health Center (NWHC) (Madison, Wisconsin), while the postmortem study of 1 event in Kansas was carried out at the Southeastern Cooperative Wildlife Disease Study (SCWDS) (Athens, Georgia). Confirmation of PPMV-1 infection in each submitted carcass was based on the following: (1) presence of suggestive histologic changes consistent with PPMV-1 infection, (2) viral isolation from 1 or more internal organs (brain, liver, kidney, spleen) of at least 1 bird per event, followed by (3) APMV-1 identification by RT-PCR. Subsequent confirmation of PPMV-1 by fusion gene (F gene) sequencing was pursued in at least 1 isolate per event in 21 of 25 (84%) events. ECDOs (61 birds) and feral ROPIs (9 birds) were the only species in which PPMV-1-associated disease was confirmed. We excluded from this study 9 additional mortality events in columbiforms investigated by the NWHC in which PPMV-1 was isolated from at least 1 submitted bird but specimens did not meet the criteria for pathology review (poor postmortem preservation or lack of sufficient tissue available).
Fresh chilled specimens were received by the laboratory within 24 to 36 hours of collection or death. When rapid shipment was not possible, the birds were frozen, kept frozen during shipment, and thawed at 4°C upon arrival. Each carcass was shipped in a separate sealed plastic bag to prevent cross-contamination of individual specimens.
Diagnostic necropsies were performed using standard protocols; tissues from each bird including brain, esophagus, proventriculus, ventriculus, intestine, heart, liver, kidney, spleen, pancreas, trachea, bursa of Fabricius, thymus, adrenal gland, kidney, skeletal muscle, peripheral nerve, skin, and gonads were fixed in 10% neutral-buffered formalin, processed routinely, sectioned at 5 μm, and stained with HE. Virus isolation in embryonating eggs was conducted using fresh tissue aseptically collected during necropsy as previously described. 27 Allantoic fluid from each egg was tested for the presence of hemagglutinating viruses using chicken and turkey erythrocytes. Hemagglutination-negative samples were passaged at least once more and retested before the original samples were considered negative. APMV-1 was identified from hemagglutination-positive samples by RT-PCR. Viral RNA was extracted (Applied Biosystems MagMax AI/ND 96 RNA extraction kits; Ambion, Austin, Texas) according to the manufacturer’s instructions. RT-PCR was performed using the US Department of Agriculture (USDA)–validated matrix and fusion gene primers. 37 APMV-1-transcribed RNA was used as positive control. Any sample with a cycle threshold value (Ct) below 35 was considered positive. The sequence of the fusion protein cleavage site was determined as previously described 23 (data to be published elsewhere).
Cloacal and tracheal swabs were collected in viral transport media and tested for the presence of avian influenza virus (AIV) by matrix gene RT-PCR screening followed by isolation of viable AIV in embryonating eggs and identification by hemagglutination (HA) and matrix gene RT-PCR as previously described. 16 The presence of West Nile virus (WNV) was determined according to previously described methods by viral isolation in Vero cell line followed by RT-PCR when cytopathic effect was observed. 11
IHC detection of PPMV-1 NP was carried out at the University of Georgia (UGA) Department of Pathology (Athens, Georgia) on lungs, heart, pancreas, kidney, spleen, liver, brain, gastrointestinal tract, and bursa of Fabricius from 17 carcasses in excellent postmortem preservation (14 ECDOs and 3 ROPIs) from 10 of the 25 events. The selection of these 17 birds was based on optimal preservation of tissues and temporal and geographic separation of the events. Bursa of Fabricius was present in only 3 IHC-evaluated ECDOs, while pancreas and spleen were unavailable in 5 and 3 ECDOs, respectively. Viral antigen was detected in tissues (UltraVision LP Large Volume Detection System: AP Polymer, ThermoFisher Scientific, Waltham, MA) according to the manufacturer’s protocol. Briefly, unstained, 5-μm-thick paraffin sections were deparaffinized (Hemo-De, Fisher Scientific, Waltham, MA). Epitopes were unmasked by microwaving (20 minutes at minimum power) in preheated unmasking solution (Vector Laboratories, Burlingame, California, Fremont, CA). After blocking nonspecific binding (1× Power Block; Biogenex) for 10 minutes at room temperature, slides were incubated overnight at 4°C with a 1:8000 dilution (in phosphate-buffered saline with 0.02% Tween 20) of a polyclonal rabbit anti-Newcastle disease virus (NDV) NP antibody (raised against the synthetic peptide TAYETADESETRRIC and cross-reactive with PPMV-1). 25 All steps after the incubation with the primary antibody were conducted per the manufacturer’s directions. Antigen-antibody complexes were visualized with Fast Red (Fisher Scientific). Sections were counterstained with hematoxylin, coverslipped (Permount; Fisher Scientific), and graded by a board-certified veterinary pathologist (M.I.A.) using the following grading system: – (no staining); + (<30% of area staining); ++ (30%–50% of area staining); +++ (>50% of area staining). The same set of organs from an ECDO and a ROPI negative for PPMV-1 and with an unrelated cause of death were used for negative control of the technique. Brain from a White Leghorn chick experimentally infected with NDV was used as positive control.
For the 17 carcasses selected for IHC, the overall extent and distribution of the histologic lesions on HE slides, and the extent and distribution of specific histologic changes (including lymphocytic depletion, cellular degeneration-necrosis, and inflammation), were assessed by a board-certificated veterinary pathologist (M.I.A.). For the remaining 53 birds, the presence or absence of these histologic lesions was determined by the same pathologist. Finally, the occurrence of every one of these lesions was calculated for the 70 birds included in the study.
Results
Epidemiological Data From Outbreaks
Relevant epidemiological information from the 25 confirmed PPMV-1 events is summarized in Supplemental Table 1 along with the diagnostic results used for confirmation. Briefly, 25 mortality events attributed to PPMV-1 in 10 states of the United States have been confirmed since 2009. In 18 events, the only species involved were ECDOs, while in 4 of them, mortalities were restricted to ROPIs. In 3 events, other species of columbiforms, including white-winged doves (Zenaida asiatica), common ground doves (Columbina passerina), and mourning doves, in addition to ECDOs and ROPIs, were found dead in the field, but postmortem confirmation of PPMV-1-associated disease was successful only in ECDOs and ROPIs. Clinical signs often reported in these PPMV-1 outbreaks were lethargy, weakness, docility, incoordination, and difficulty flying. Birds were often found dead under roosts. Supplemental Table 2 summarizes epidemiological data of 9 additional mortality events of columbiforms in the United States investigated by the NWHC in which specimens did not meet the criteria for pathology review (poor postmortem preservation or lack of sufficient tissue available) but PPMV-1 was isolated from at least 1 submitted bird.
Viral Isolation and Identification
Viable APMV-1 was isolated from 62 of the 70 accessions (89%). Forty-six of these isolates (74%) were further identified as PPMV-1 by F gene sequencing. Avian influenza virus was not isolated or detected by AIV matrix RT-PCR in cloacal or tracheal swabs of any of the 70 birds. West Nile virus isolation was negative from pinfeathers or pooled spleen-kidney in all 27 accessions from which it was attempted.
Necropsy Findings
All birds had completely grown feathers. Of the 70 birds, 25 (36%) were estimated to be younger than 6 months and 45 (64%) older than 6 months based on the presence or complete involution of the bursa of Fabricius. 8 Thirty-nine (56%) birds were in poor body condition with depletion of fat stores. There were no gross abnormalities attributed to PPMV-1 infection. In 15 birds (21%), concomitant pathological processes were observed; these included bone fractures and liver rupture due to blunt trauma, soft tissue lacerations caused by predation, focal necrotizing stomatitis, pharyngitis and ingluvitis consistent with trichomoniasis, focal fibrinous to granulomatous fungal airsacculitis, and egg-yolk peritonitis.
Histopathological and Immunohistochemical Evaluation in 17 Selected Birds
The following lesions were observed in tissues with immunolabeling for PPMV-1 NP: tubulointerstitial nephritis (16/17), necrotizing pancreatitis (9/12), lymphoid depletion in the spleen (4/14) and bursa of Fabricius (3/3), hepatocellular necrosis (4/17), lymphocytic choroiditis (1/17), and pinfeather pulpitis (1/1). The comparative assessment of these lesions with the PPMV-1 NP immunolabeling is summarized in Table 1.
Severity of Microscopic Lesions (HE)a and Distribution of Pigeon Paramyxovirus Serotype 1 Nucleoprotein (IHC)b in Tissues From 17 Naturally Infected Wild Columbiforms in the United Statesc.
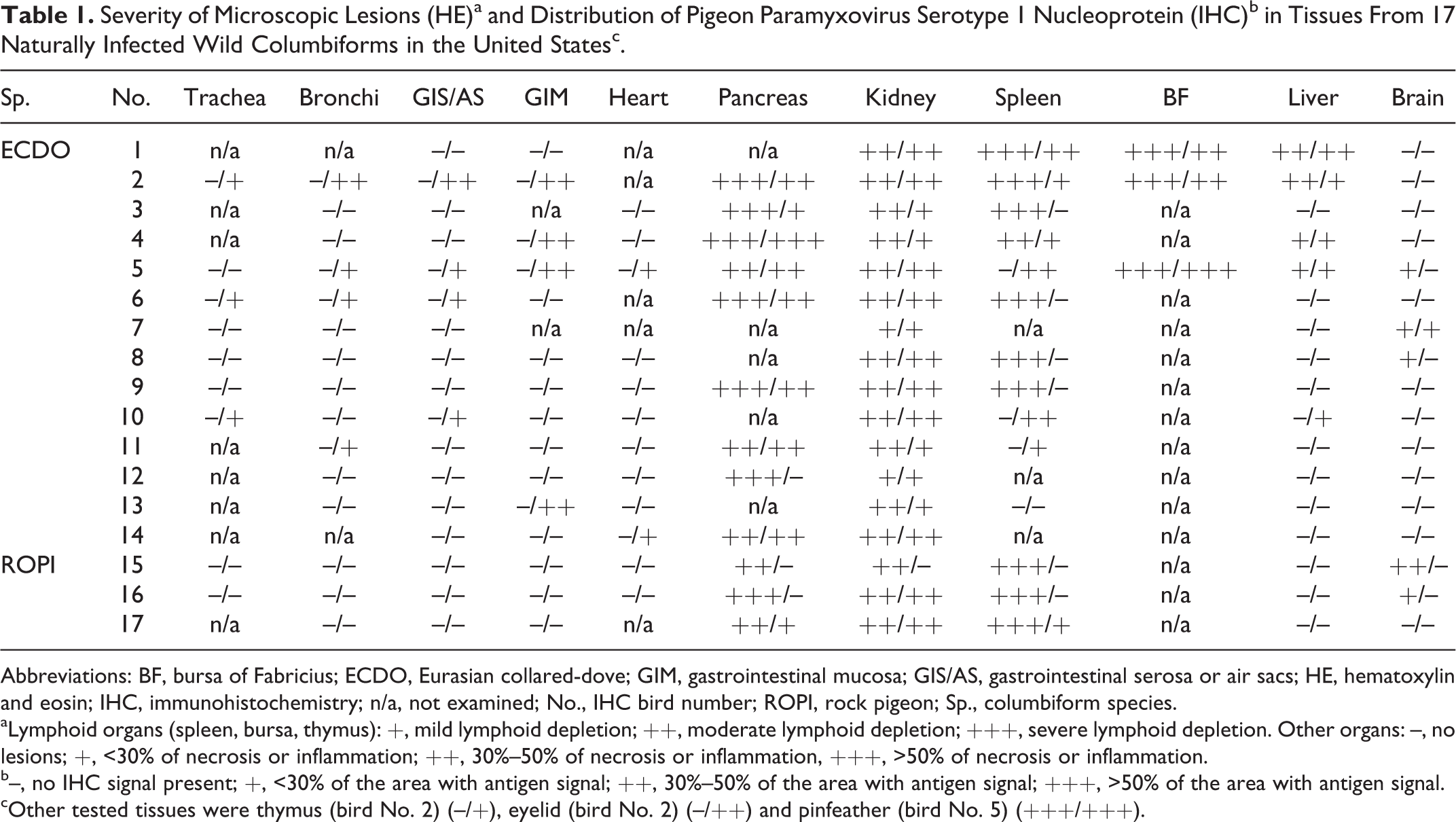
Abbreviations: BF, bursa of Fabricius; ECDO, Eurasian collared-dove; GIM, gastrointestinal mucosa; GIS/AS, gastrointestinal serosa or air sacs; HE, hematoxylin and eosin; IHC, immunohistochemistry; n/a, not examined; No., IHC bird number; ROPI, rock pigeon; Sp., columbiform species.
aLymphoid organs (spleen, bursa, thymus): +, mild lymphoid depletion; ++, moderate lymphoid depletion; +++, severe lymphoid depletion. Other organs: –, no lesions; +, <30% of necrosis or inflammation; ++, 30%–50% of necrosis or inflammation, +++, >50% of necrosis or inflammation.
b–, no IHC signal present; +, <30% of the area with antigen signal; ++, 30%–50% of the area with antigen signal; +++, >50% of the area with antigen signal.
cOther tested tissues were thymus (bird No. 2) (–/+), eyelid (bird No. 2) (–/++) and pinfeather (bird No. 5) (+++/+++).
All sections of kidney examined exhibited tubulointerstitial nephritis with tubular epithelial necrosis (Fig. 1). Necrosis of less than 30% (in 4 birds) to 30%–50% (in 13 birds) of the medullary and to a lesser extent cortical renal tubules was accompanied in 11 birds by lymphoplasmacytic inflammatory infiltrates that occupied up to 30% of the tissue, replacing lost parenchyma and expanding the interstitium. In 2 ROPIs, the inflammatory infiltrates occupied over 30% of the kidney. Glomeruli were spared in all cases. Viral antigen immunolabeling was detected in the cytoplasm and rarely in the nucleus of degenerating and necrotic tubular epithelial cells in proportion to the extent of the tubular damage.
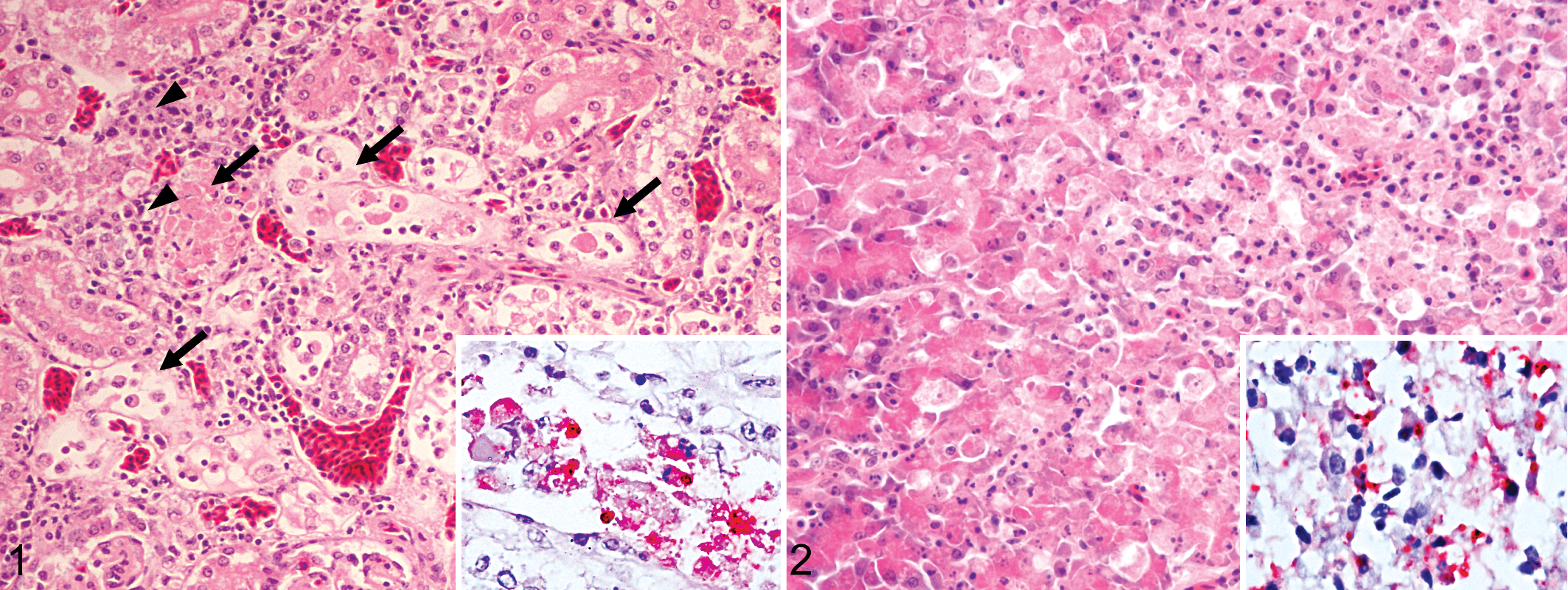
All sections of pancreas examined had multifocal to coalescing necrotizing pancreatitis, with acinar cell degeneration and necrosis of less than 30% (in 4 birds), 30%–50% (in 3 birds), and over 50% (in 5 birds) (Fig. 2). Mild to moderate multifocal saponification of the peripancreatic fat was observed in all cases. In 5 birds, 30%–50% of the pancreatic tissue was occupied by lymphocytes and plasma cells that expanded the interstitium and replaced the lost parenchyma. Two of these 5 birds with lymphoplasmacytic inflammation had multifocal mild pancreatic fibrosis. Independent of the extent of the lesions, variable amounts of viral antigen were observed in the cytoplasm of degenerating and necrotic acinar cells.
In the spleen, moderate to severe diffuse lymphoid depletion of both periarteriolar lymphoid sheaths (PALS) and periellipsoidal lymphoid sheaths (PELS) was observed in 10 birds (Fig. 3). In 4 of these birds, variable amounts of fragmented cells with condensed and pyknotic nuclei located around arterioles and ellipsoids were interpreted as necrotic lymphoid cells (lymphocytolysis). Low to moderate amounts of protein-rich exudate replaced the depleted PALS and PELS in 4 birds. Viral antigen signal was scattered throughout the spleen within the cytoplasm of round cells that were up to 20 μm in diameter with an eccentric nucleus and vacuolated cytoplasm (macrophage-like cells). Occasionally, the antigen was also present in areas of lymphocytolysis.
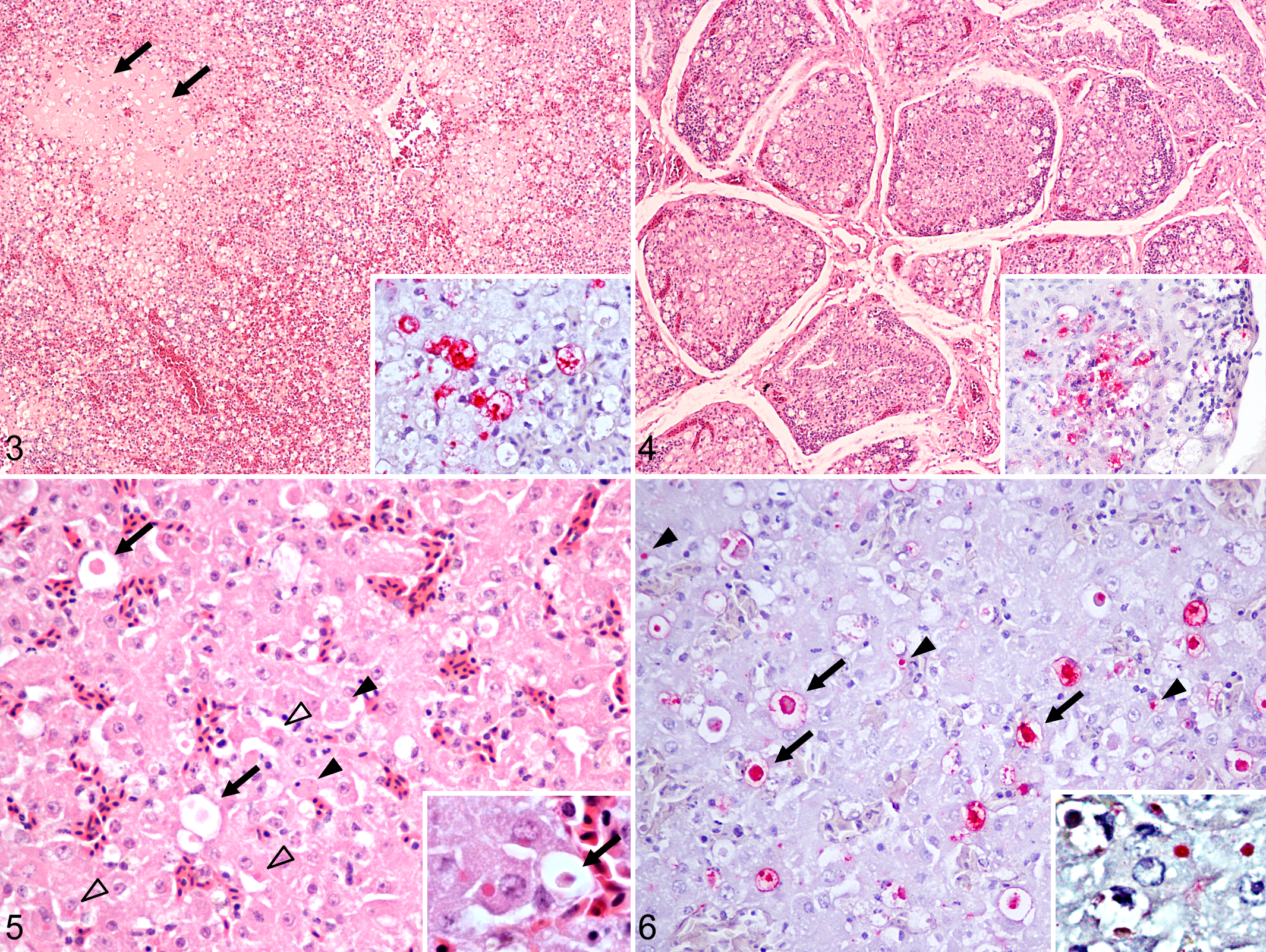
In the 3 birds in which the bursa of Fabricius was assessed, there was diffuse, severe lymphoid depletion of the follicular medulla and to lesser extent of the cortex (Fig. 4). Lymphocytolysis was present in the medulla of multiple follicles in 1 bird. Medullary lymphocytolytic debris and scattered medullary and cortical cells morphologically compatible with macrophages were immunopositive.
In 4 ECDOs, small clusters or individual randomly distributed hepatocytes showed condensed, hypereosinophilic cytoplasm and pyknotic, karyorrhectic, or karyolytic nuclei interpreted as hepatocellular necrosis (Fig. 5). In 1 of these birds (No. 1), numerous intact and degenerating hepatocytes contained one and rarely two, 4-μm-diameter, pale, bright, eosinophilic intracytoplasmic inclusion bodies. In these 4 birds, hepatocellular necrosis was accompanied by increased numbers of prominent (up to 20 μm in diameter) cells scattered in the sinusoids and/or the space of Disse (enlarged Kupffer cells) that contained aggregates of antigen in the cytoplasm and on the cell membrane (Fig. 6). In bird No. 1, viral antigen was also detected in discrete small clusters with similar location and size as the inclusion bodies within the cytoplasm of numerous hepatocytes. An increased number of enlarged Kupffer cells was observed in the absence of immunolabeling in 9 birds.
Histologic changes in the central nervous system associated with the presence of antigen were observed in only 1 bird with focal lymphocytic choroiditis, characterized by focal degeneration and necrosis of choroid plexus cells and multiple 1-cell-thick lymphocytic perivascular cuffs. Abundant PPMV-1 antigen immunolabeling was observed in the cytoplasm of the affected choroid plexus cells. Focally extensive mild to moderate lymphocytic meningoencephalitis characterized by mild gliosis and multiple, 1- to 2-cell-thick, perivascular lymphocytic cuffs in the neuroparenchyma and leptomeninges were observed in the brains of 4 birds without associated immunolabeling.
A pinfeather examined from ECDO No. 5 had moderate histiocytic pulpitis. The interstitium of the pulp was diffusely infiltrated by macrophages and fewer lymphocytes and rare plasma cells. Abundant antigen signal was multifocally distributed in the feather pulp within the cytoplasm of endothelial cells, macrophages, and unidentified interstitial cells and in scattered keratinocytes of the basal and spinous stratum of the inner feather shaft. In ROPI No. 15, the feather pulp was multifocally infiltrated by macrophages and fewer lymphocytes and plasma cells. Multiple pulp capillaries had plump endothelial cells that occasionally were lost and fibrin thrombi attached to the subendothelial tissue partially or completely obliterating the vessel. Immunohistochemical evaluation was not performed on the pulp feather of this last bird due to a technical problem.
Cytoplasmic viral immunolabeling was observed in the following cells in the absence of significant histologic changes: macrophage-like cells in spleen and thymus; interstitial cells of the palpebral mucosa; respiratory epithelial cells of trachea and bronchi and associated parabronchial epithelium; unidentified interstitial cells of the bronchus-associated lymphoid tissue (BALT); mesothelial cells and unidentified interstitial cells of the intestinal serosa and abdominal air sacs; esophageal squamous epithelium; unidentified interstitial cells of the lamina propria of the small and large intestine and cloaca; and myocardial myocytes and fibrocytes of the semilunar valve of the heart.
Occurrence of Lesions Suggestive of PPMV-1 Infection
The occurrence of lesions suggestive of PPMV-1 in 70 confirmed naturally infected birds (including the 17 IHC-tested birds and 53 IHC-untested birds) is summarized in Table 2. Briefly, bursal lymphoid depletion (24/26, 92%) and splenic lymphoid depletion (40/49, 82%) were the most frequent histologic changes, followed by tubulointerstitial nephritis (51/69, 74%), necrotizing pancreatitis (18/30, 60%), hepatocellular necrosis (14/43, 33%), and nonsuppurative meningoencephalitis (9/45, 20%). Lymphoid depletion of the bursa of Fabricius was commonly observed in ECDOs, but no specimens were available for histologic assessment in ROPIs. Meningoencephalitis was frequently observed in ROPIs but was a rare finding in ECDOs.
Lesions in 70 Wild Columbiforms With Confirmed Pigeon Paramyxovirus Serotype 1 Disease From 25 Events in the United Statesa.

Abbreviations: BF, bursa of Fabricius; ECDO, Eurasian collared-dove; ROPI, rock pigeon; LD, lymphoid depletion; n/a, not examined.
aHistopathological results expressed as number of birds in which the lesion was present/number of birds in which the organ was examined (percentage).
Discussion
Since the first reported PPMV-1-associated mortality event in 1984 in domestic pigeons in the State of New York, 31 PPMV-1 associated disease has been detected in multiple regions of the United States. 5,32 Mortality events caused by this virus have been confirmed since 2009 by the NWHC affecting mostly ECDOs and less often feral ROPIs. Interestingly, PPMV-1 infection has not been confirmed in columbiforms native to North America such as white-winged doves, common ground doves, and mourning doves, despite being found with ECDOs and ROPIs during mortality events. Estimated mortalities from tens to thousands of birds were reported in 23 of 25 events (92%). Consistent with what has been previously reported in columbiforms, gross lesions were not present 5,19 with the exception of poor body condition in 39 of 70 (56%) birds. Poor body condition is common in wild birds submitted for necropsy and can be due to a wide range of factors, including disease, breeding cycle, migration, adverse weather events, or lack of food availability. Stressors such as these have been proposed as predisposing factors for infection with APMV-1. 14 Infection with PPMV-1 in ECDO normally has an acute course, and death occurs within 1 to 2 days of the onset of clinical signs, 32 suggesting that other factors may have been responsible for the suboptimal body condition. Concomitant infectious processes were observed at the time of death in 9 of 70 (13%) birds, but these were only minor and localized, and therefore PPMV-1 was considered the cause of death in most birds. Occasionally, severe trauma and predation (5/70, 7%), possibly secondary to PPMV-1-induced debilitation, were considered the cause of death.
The histologic lesions most commonly associated with PPMV-1 infection in this study were similar to those noted in previous reports: (1) acute to subacute and rarely chronic tubulointerstitial nephritis; 5,19,31,32 (2) acute to subacute and rarely chronic necrotizing pancreatitis; 5,19,31 (3) lymphoid depletion of the spleen 31,32 and bursa of Fabricius; 32 (4) random acute multifocal hepatocellular necrosis 5,19 with proliferation of enlarged Kupffer cells; and (5) lymphocytic choroiditis and meningoencephalitis. 5,19,31
Similar microscopic lesions may also be observed in other wild bird species infected with highly pathogenic AIV (HPAI) 20 and WNV. 13 Although viremia of these 2 viruses has been confirmed in columbiforms, they seem particularly resistant to overt disease. 1,4 All 70 birds included in this study were negative for AIV. In addition, viral culture and isolation in Vero cells attempted in 27 birds were unrewarding, suggesting that coinfections with WNV or other flaviviruses were unlikely.
Tubulointerstitial nephritis varied in presentation depending on the stage of the renal infection. In acute stages, we observed the cytolytic effect of the virus on infected cells. These were predominantly tubular epithelial cells of medullary tubules and to a lesser extent cortical tubules. In subacute stages, there was an influx of lymphocytes and fewer plasma cells, while the presence of necrotic tubules was rare. 5 Occasionally, exuberant inflammation replaced tubules and expanded the interstitium, but there was minimal tubular damage and absence of antigen. These may represent chronic stages in which the virus had been already cleared but the tissue changes still persisted. Extensive tubulointerstitial nephritis might result in impaired ability to concentrate urine and defective resorption or secretion of metabolites. Nevertheless, since the extension of the renal lesion rarely reached 50% of the parenchyma, it is unlikely to be the cause of death in most cases but could be a contributor to the loss of homeostasis. 7 In addition, the high viral load in tubules suggests that urine may be an important route of viral excretion in most PPMV-1-infected birds.
Necrotizing pancreatitis was most often observed as an acute change in which virus-infected exocrine cells underwent vacuolar degeneration and necrosis. Interstitial expansion and replacement of the lost parenchyma by inflammatory cells dominated over degeneration and necrosis in subacute stages of the pancreatic infection. 6 Chronic pancreatitis without detection of viral antigen was occasionally observed and may be due to a cleared pancreatic infection. Acute pancreatitis might cause death of the animal by the release of activated proteolytic enzymes and proinflammatory cytokines into the bloodstream, causing a systemic inflammatory response syndrome and multiorganic failure. 18 The stage of chronicity of pancreatitis was usually concurrent with that of tubulointerstitial nephritis.
As previously described in infections with certain velogenic and mesogenic strains of NDV in chickens, we observed moderate to severe lymphoid depletion with virus-induced lymphocytolysis in PALS (thymus-derived naïve T lymphocytes) and PELS (bursa-derived naïve B lymphocytes) of the spleen and in the follicular medulla and to lesser extent cortex of the bursa of Fabricius. 24,29 Occasionally, depleted PALS and PELS were replaced by mild to extensive deposits of protein-rich exudates resembling fibrin. 6,31,32 Since lymphocytolysis was only reliably confirmed in few cases, we preferred to avoid the term “necrotizing splenitis or bursitis,” which has been used for similar findings by other authors. 31,32 However, significant changes in lymphoid organs have not always been described in PPMV-1 outbreaks in columbiforms in North America. 5,19 In consonance with this, in the present study 3 systemically infected ECDOs had an apparently normal population of lymphocytes in the spleen with presence of viral antigen-laden macrophages. In addition, in 6 of 14 (43%) birds for which IHC was performed on the spleen, severe lymphoid depletion of this lymphoid organ was observed in absence of viral antigen. In 3 of these 6 infected animals, body condition was poor, which may explain the severe lymphoid atrophy. 15 Although the present IHC study indicates that on some occasions PPMV-1 may induce lymphoid depletion by lymphocytolysis, the prevalence of this finding in PPMV-1 cases may be inflated by other concomitant factors such as nutritional deprivation. The low availability of thymic tissue for the present study prevents us from drawing conclusions about the impact of PPMV-1 on this lymphoid organ.
Random, multifocal hepatocellular necrosis is a less common but striking change occasionally reported in PPMV-1-infected columbiforms 19,32 and was also present in ECDOs included in this study. Here we use the term necrosis to indicate dead cells in histologic sections, regardless of the pathway by which the cells died (ie, we cannot distinguish necrosis from virus-induced apoptosis). 26 Prominent immunopositive enlarged Kupffer cells were invariably observed in all the birds in which hepatocellular necrosis was diagnosed. Proliferation of macrophage-like cells, predominantly Kupffer cells in the liver, has been described in chickens infected with NDV. 33 They enlarge and accumulate vacuoles and lysosomal debris during acute inflammatory responses and lead the phagocytic removal of circulating infected cells. In overwhelming viral infections, many of the Kupffer cells and adjacent uninfected hepatocytes may undergo necrosis. 9 In 1 bird, viral antigen was also present in viable and degenerating hepatocytes, indicating that in some instances hepatocellular necrosis may be induced by direct infection of these cells by PPMV-1.
In this study, mild focal to multifocal meningoencephalitis was relatively frequent in ROPIs but rare in ECDOs. However, of 17 brains examined, PPMV-1 antigen was only observed in a focus of lymphocytic choroiditis in 1 ECDO. In this bird, numerous necrotic and apparently normal choroid cells contained abundant cytoplasmic viral antigen. In poultry, NDV reaches the choroid plexus and endothelial cells of vessels in the brain within 3 days post infection, and viral antigen remains in neurons until day 8. 36 Choroiditis with abundant intracytoplasmic viral antigen in choroid cells but without major inflammatory changes or antigen presence in the rest of the brain may represent an early stage of brain infection. An experimental infection of White Leghorn chicks with PPMV-1 showed that meningoencephalitis usually persists up to 10 days after the loss of viral antigen. 24 Hence, the lack of immunolabeling in the brain of birds with subacute to chronic lesions and minimal immunolabeling in other organs might represent late stages of the disease in which the virus has been cleared from the brain. However, for those birds in which an acute PPMV-1 infection is suggested by acute lesions and abundant immunolabeling in other target organs, these small foci of nonsuppurative meningoencephalitis without accompanying viral antigen may represent residual inflammation from unrelated processes. Overall, the prevalence of central nervous system (CNS) involvement in overt PPMV-1 disease was unexpectedly low, particularly given the neurologic clinical signs often described, and might be dependent on the duration of the infection before death and the tropism of the infecting strain for the nervous tissues. 29
In this study, we described for the first time the presence of significant histologic lesions in the pinfeathers from 2 different birds associated with the presence of viral antigen in one of them. Mononuclear inflammation was diffuse through the feather pulp matrix. Endothelial damage and thrombosis of pulp vessels were prominent in 1 feather. The viral antigen was abundant in the cytoplasm of numerous macrophages and other interstitial cells and in numerous endothelial cells of capillaries. Viral antigen was also scattered in the flat squamous epithelium of the inner root sheath of the feather. Therefore, feathers from infected animals may be another source of virus shedding.
Focal to multifocal necrotizing and lymphoplasmacytic myocarditis has been described in chickens infected with PPMV-1. 25 We detected viral antigen in the sarcoplasm of apparently normal myocardiocytes and the cytoplasm of fibroblasts in 1 semilunar valve of ECDOs. This confirms that some strains of PPMV-1 also have, under certain circumstances, tropism for the myocardium of ECDOs.
The capacity of PPMV-1 to infect the respiratory epithelium in trachea, bronchi, parabronchi, and abdominal air sacs indicates that as described for NDV in chickens, 28 PPMV-1 may be efficiently excreted via aerosolized respiratory secretion. For the same reason, inhalation may be an important pathway of PPMV-1 infection. In addition, the presence of antigen in superficial epithelium of the esophagus indicates that regurgitation of the crop content may be another important way of transmission of the virus. This may have implications during parental feeding of nestlings due to the feeding of sloughed esophageal mucosa (“crop milk”) in columbiforms.
Although chronicity of infection cannot be determined from these opportunistic samples, based on these results, a proposed course of PPMV-1 infection involves acute infection of the kidney and pancreas, and less often infection of lymphoid tissue, liver, and CNS, with resultant degeneration and necrosis of those organs. In birds surviving the acute phase of the disease, a subacute phase with lymphoplasmacytic inflammation in kidney and pancreas, gliosis, and lymphocytic inflammation of the CNS and depletion of spleen and bursa of Fabricius may be observed with no or minimal detectable antigen.
Although the virulence of PPMV-1 in chicken was already proven 12 and it is an important concern for the industry of poultry in the United States, little was known about the virulence of this group of viruses in their natural hosts. Regardless of the limitations intrinsic to a study based on natural infections, we provided evidence of the systemic distribution and lethal outcome of PPMV-1 infections in ECDOs and ROPIs.
Large mortalities in free-ranging columbiforms across the United States are connected to PPMV-1-associated disease. The large majority of these events involve ECDOs that in many cases present in suboptimal body condition without other remarkable gross findings. Acute to subacute necrotizing tubulointerstitial nephritis and necrotizing pancreatitis are common histopathologic changes caused by viral infection of the renal tubular epithelium and pancreatic exocrine epithelium. Although lymphoid depletion of the bursa of Fabricius and spleen is a frequent finding, the proportion of animals in which this change is exclusively attributable to PPMV-1 is uncertain. Hepatocellular necrosis and lymphocytic encephalitis are other histologic changes occasionally observed, but whether they are caused by PPMV-1 needs further research. Viral shedding in urine is likely in most actively infected animals. Respiratory tract aerosols, pinfeathers, and regurgitation of the crop content during parental feeding are other probable sources of infection. This study represents a detailed and broad pathologic analysis of PPMV-1-associated disease in columbiforms in the United States, representing an important step forward for the understanding of PPMV-1 in its natural hosts.
Footnotes
Acknowledgments
We acknowledge Corrie Brown, Valerie Shearn-Bochsler, and Jennifer Chipault for their help with data collection, synthesis, and interpretation. We also recognize the technical assistance provided by all the personnel of the USGS-NWHC, SCWDS, UGA Department of Pathology, and Kansas Department of Wildlife Parks and Tourism, particularly Renee Romaine Long, Stephanie Steinfeldt, Dottie Johnson, Jian Zhang, Rebecca Poulson, and Shane Hesting. The use of trade, product, or firm names is for descriptive purposes only and does not imply endorsement by the US government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
