Abstract
The pathogenesis of six pigeon-origin isolates of Newcastle disease virus (NDV) was investigated in chickens. Four isolates were previously defined as the variant pigeon paramyxovirus 1 (PPMV-1), and two isolates were classified as avian paramyxovirus 1 (APMV-1). Birds inoculated with PPMV-1 isolates were euthanatized, and tissue samples were collected at 2, 5, and 10 days postinoculation (DPI). Birds inoculated with APMV-1 isolates died or were euthanatized, and tissue samples were collected at 2, 4, and 5 DPI. Tissues were examined by histopathology, immunohistochemistry (IHC) for the presence of NDV nucleoprotein, and in situ hybridization (ISH) for the presence of viral mRNA for the matrix gene. Spleen sections were stained by the terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling (TUNEL) assay and by IHC using an anti-active caspase-3 antibody (IHC-Casp) to detect apoptotic cells. Brain sections of PPMV-1-infected birds were examined by IHC to detect T and B lymphocytes and glial fibrillary acidic protein (GFAP). Histologically, birds inoculated with PPMV-1 isolates had marked lesions in the heart and brain. Presence of viral nucleoprotein and viral mRNA in the affected tissues was confirmed by IHC and ISH, respectively. Numerous reactive astrocytes were observed in brain sections stained for GFAP. Among all the isolates, the IHC-Casp demonstrated that apoptosis was very prominent in the ellipsoid-associated cells of the spleen at 2 DPI. Results of the TUNEL assay indicated that apoptotic cells were prominent at 5 DPI and were more randomly distributed. The clinical signs and gross and histopathologic changes observed in the APMV-1-infected birds were characteristic of an extensive infection with highly virulent NDV evident by IHC.
Keywords
Newcastle disease (ND) is a globally distributed avian disease that can cause severe economic losses in commercial poultry. According to the Office International des Epizooties, ND belongs to the reportable List A diseases (International Animal Health Code, E-mail: http://www.oie.int). Newcastle disease virus (NDV) is synonymous with avian paramyxovirus 1 (APMV-1), a nonsegmented, single-stranded, negative-sense RNA virus belonging to the family Paramyxoviridae, subfamily Paramyxovirinae, genus Rubulavirus.3 Recent studies based on phylogenetic analysis have suggested that NDV should be assigned to a new genus within the subfamil Paramyxovirinae.15,42
Pigeon paramyxovirus 1 (PPMV-1), a variant of NDV, is characterized by unique monoclonal antibody (Mab)-binding profiles.7,20,31 After its emergence in the Middle East (late 1970s),32 pigeon NDV spread worldwide,2,32,45 and dissemination from pigeon populations into commercial poultry has been documented in Great Britain6 and Austria.44 Newcastle disease virus has been recovered from pigeons in the USA9,20,40 with further characterization of the isolates as APMV-1 and PPMV-1 using Mab panels.20,40 To date, there have been no reports of PPMV-1 spread with overt clinical disease among commercial poultry in the USA. However, because of the frequent isolation of PPMV-1 from pigeons across the USA, there is a need to assess the risk potential of these viruses causing disease in chickens. Previously, investigators demonstrated virulence increase based on NDV pathotyping tests after PPMV-1 isolates were serially passaged through chickens, particularly using the intramuscular route of infection.5,6,22,23 Another concern regarding PPMV-1 is its ability to spread from infected pigeons to nearby chickens.4 In one study, the contact infection rate of two different PPMV-1 strains in 4-week-old specific pathogen–free (SPF) chickens reached 100%.22
Further characterization of most PPMV-1 isolates demonstrated the presence of multiple basic amino acids at the fusion protein cleavage site and an intracerebral pathogenicity index equal to or greater than 0.7.12 Infections of birds with APMV-1 isolates typical of most PPMV-1 are now defined as Newcastle disease,38 a reportable disease that may result in the initiation of international trade restrictions depending on the species involved or the epidemiologic association of birds with ND, or both. There is a concern that these PPMV-1 strains, if allowed to circulate among chickens, could evolve greater disease-causing capability. The potential for such an occurrence has already been demonstrated for NDV. The 1998–2000 NDV outbreaks reported in Australia,17,21,46 with clinical disease and mortality observed among commercial chickens, were caused by a virulent APMV-1 strain that evolved from a low-virulence strain that was circulating among chickens apparently for a long time. Consequently, it is important to determine the real threat for domestic chickens represented by PPMV-1 isolates, which are usually characterized as NDV of mild to moderate virulence.
In this study, six pigeon-origin NDV isolates that were previously passaged in 2-week-old chickens23 were inoculated into 4-week-old White Leghorns to assess clinical disease and pathogenesis through 14 days postinoculation (DPI). Infected birds were examined for gross and microscopic lesions, and tissue samples were evaluated using immunohistochemistry (IHC), in situ hybridization (ISH), and two different assays for apoptosis.
Materials and Methods
Eggs and chickens
The source of embryonated chicken eggs and chickens was the Southeast Poultry Research Laboratory (SEPRL, ARS, USDA) SPF White Leghorn flock. Embryonated eggs were utilized for virus amplification of chicken-passaged virus. Chickens were inoculated for the pathogenesis study and housed in negative pressure isolators under BSL-3 agriculture conditions at SEPRL and provided food and water ad libitum.11,23
Viruses
Six pigeon-origin isolates of NDV were previously characterized by NDV Mab binding profiles using the hemagglutination-inhibition (HI) test.20,23 Four of these isolates were characterized as PPMV-1: Pigeon TX, from Texas outbreak, 1998; Pigeon GA, from Georgia outbreak, 1998; 84-44407, early US PPMV-1 isolate, Northeastern, 1984;40 and Pigeon 84, early US PPMV-1 isolate, New York, 1984.20 The other two isolates were classified as APMV-1: P1307, from imported quarantine lots of fruit and crown pigeons, 1975;16 and P5658, from racing pigeons, 1975.16 All isolates were provided by the USDA APHIS National Veterinary Services Laboratories with exception of Pigeon 84.20
Pathogenesis study
The viruses utilized in this experiment were passaged in chickens as previously described.23 Six groups of six 2-week-old SPF White Leghorns were inoculated intramuscularly with six pigeon-origin NDV isolates. At 2 DPI, three or four chickens from each group were euthanatized, and their spleens were removed aseptically and homogenized. The homogenate was inoculated intramuscularly into another group of 2-week-old SPF White Leghorns. This procedure was repeated for a total of four passages with the PPMV-1 isolates. Only one passage was performed with the two APMV-1 isolates because of the severity of the disease and high mortality observed during the first passage.
Infective amnioallantoic fluid from the first (P1307 and P5658), third (Pigeon 84, chosen because of higher titers than fourth passage virus), or fourth (Pigeon TX, Pigeon GA, and 84-44407) chicken passages was prepared for inoculation into six groups of 10 4-week-old SPF White Leghorns. After dilution in brain-heart infusion broth, approximately 105.0 EID50 was inoculated intraconjunctivally (0.1 ml/bird). One group of 10 4-week-old birds served as noninfected controls.
The disease was followed serially by examining tissues from birds infected with the PPMV-1 isolates that were euthanatized (2/day) at 2, 5, and 10 DPI.11,23 The remaining four birds were observed clinically through 14 DPI, tested for the presence of NDV antibodies using the HI test,3 and euthanatized. Because of the severity of the clinical signs observed in birds inoculated with the APMV-1 isolates (P1307 and P5658), these birds were examined at 2, 4, and 5 DPI. Necropsies were performed immediately postmortem, and the following tissues were collected and fixed by immersion in 10% neutral buffered formalin for approximately 48 hours: spleen, thymus, bursa, lower eyelid (including conjunctiva and skin), Harderian gland, proventriculus, pancreas, small intestine, cecal tonsils, large intestine, caudal thoracic air sac, trachea, lung, heart, liver, kidney, and brain. All sampled tissues were routinely processed into paraffin, and 3-μm sections were cut for hematoxylin and eosin (HE) staining. Selected sections were processed for IHC,23 ISH,11 and the terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assay.37
Immunohistochemistry
The sampled tissues of all the infected birds were examined by IHC using the following generic protocol to detect viral nucleoprotein (N) and apoptotic cells. After deparaffinization, tissue sections were treated with 3% hydrogen peroxide and then subjected to antigen retrieval by microwaving for 10 minutes at full power in antigen unmasking solution (Vector Laboratories, Burlingame, CA) followed by blocking (only for IHC-N) with universal blocking reagent (Biogenex, San Ramon, CA) as recommended by the manufacturer. Tissues were incubated with the primary antibody overnight at 4 C or for 2 hours at 37 C. After washing, sections were incubated with biotinylated antibody against the species in which the primary antibody was made and then with either avidin–biotin–alkaline phosphatase or elite-PO (Vector Laboratories). Substrate was Vector Red or diaminobenzidine (DAB) (Vector Laboratories). Sections were counterstained lightly with hematoxylin and coverslipped for a permanent record.
To detect NDV nucleoprotein (IHC-N), the primary antibody was an anti-peptide antibody23 made in rabbit and used at 1:8,000 dilution. Sections were incubated with avidin–biotin–alkaline phosphatase, and the substrate was Vector Red. Sections of the spleen were examined by IHC using a rabbit polyclonal anti-active Caspase-3 (IHC-Casp) antibody (Promega, Madison, WI) at 1:350 dilution. The enzyme was elite-PO, and the substrate was DAB.
Selected sections of the brain of PPMV-1–infected birds were examined for the presence of T lymphocytes using anti-CD3 rabbit polyclonal antibody (Dako, Carpinteria, CA), for the presence of B lymphocytes using the Mab BLA-36 (Biogenex), and for the presence of astrocytes expressing glial fibrillary acidic protein (GFAP) using an anti-GFAP Mab (Biogenex). The following generic protocol was used with CD3 (IHC-CD3), BLA-36 (IHC-BLA-36), and GFAP (IHC-GFAP) antibodies. Only the steps that differ from the previous protocol are mentioned. Deparaffinized sections were treated with 5% hydrogen peroxide. The antigen retrieval was performed using antigen retrieval Citra Solution (Biogenex) incubated in a steamer for 25 minutes followed by blocking with 5% bovine serum albumin. Sections were incubated with the primary antibody for 30 minutes at 37 C. Sections were then incubated with biotinylated antibody and with extra-avidin™ peroxidase conjugate (Sigma, St. Louis, MO). Substrate was DAB. The antibody dilutions for CD3, BLA-36, and GFAP were 1:100, 1:50, and 1:400, respectively.
In situ hybridization
Selected tissue sections of the PPMV-1–infected birds were stained with a negative-sense digoxigenin-labeled 850-base riboprobe representing the 5′ end of the matrix gene of the NDV Fontana (CA 1083) strain as previously described.11 The matrix gene from the Fontana strain was cloned into pCRII transcription vectors (Invitrogen, Carlsbad, CA). Anti-sense digoxigenin-labeled riboprobes were generated using RNA polymerase in the presence of labeled nucleotides. For ISH, tissue sections were deparaffinized, rehydrated, and digested with 30 µg/ml Proteinase K for 15 minutes at 37 C. Hybridization was conducted overnight at 42 C with approximately 20 ng of probe in prehybridization solution. After stringent washes, anti-digoxigenin alkaline phosphatase was added to the sections. The development was with chromogen/substrate nitroblue tetrazolium (NBT) and 5-bromo-4-chloro-3-indoylphosphate (BCIP). Tissues were counterstained lightly with hematoxylin and coverslipped.
TUNEL assay
The TUNEL assay37 (Boehringer Manheim, Indianapolis, IN) was utilized in addition to the IHC-Casp to detect apoptosis in sections of the spleen. The tissue sections were treated with 20 µg/ml Proteinase K for 15 minutes at 37 C. After washing, the sections were incubated with TUNEL reaction mixture for 1 hour at 37 C. Converter alkaline phosphatase was added to the slides for 30 minutes at 37 C. The development was with chromogen/substrate NBT and BCIP. Tissues were counterstained lightly with hematoxylin and coverslipped with Permount.
The TUNEL and IHC-Casp results were analyzed by comparing the number of apoptotic cells detected at 2, 5, and 10 DPI in spleen sections of the infected versus control birds. Apoptotic cells were counted in five high-power fields (400×), and the final result was based on the average between the fields.
Results
Clinical findings
PPMV-1 isolates
Several chickens (10–20%) from each experimental group had mild periocular edema and bilateral conjunctivitis at 2 DPI. With the Pigeon TX isolate, two birds had slight depression by day 7, and one bird was very listless by day 8 postinoculation. At 10 DPI, all remaining Pigeon TX–infected chickens had mild to severe depression. With the Pigeon GA isolate, one bird was walking on its hocks and had head tremors, drowsiness, and incoordination by day 7 postinoculation. One bird infected with isolate 84-44407 was depressed at 7 DPI. Two birds inoculated with the Pigeon 84 isolate had slight depression by day 8, and one bird was walking on its hocks by day 9 postinfection.
APMV-1 isolates
The clinical signs presented by chickens inoculated with the two pigeon-origin APMV-1 isolates (P1307 and P5658) were similar. However, birds inoculated with P1307 were more severely ill. With both APMV-1 isolates, all infected birds had severe bilateral conjunctivitis and periocular edema by 3 DPI. Only one bird infected with P1307 was depressed at that time. At 4 DPI, all APMV-1–infected chickens were severely depressed and inactive and had ruffled feathers. By 5 DPI, one P1307-infected bird was comatose and three were dead. Of the P5658-infected birds, one was dead and the remaining three were severely depressed and prostrate, and two of them had open-mouth breathing. The remaining birds were euthanatized in extremis at day 5 postinfection. No clinical signs were observed in the noninfected control birds.
Gross findings
PPMV-1 isolates
With all PPMV-1 isolates, the gross findings were similar. Some infected birds had petechial hemorrhages and edema in the conjunctiva of the lower eyelid at 2 DPI. At 5 DPI, spleens were large and mottled, whereas one bird infected with Pigeon 84 had petechial hemorrhages in the thymus. By 10 DPI, two Pigeon GA–infected birds had large mottled spleens, and one 84–44407–infected bird had petechiae in the eyelid.
APMV-1 isolates
In birds inoculated with both APMV-1 isolates, the macroscopic lesions were severe and widespread. At 2 DPI, the lesions were confined to the eyelids (edema and petechial hemorrhages) and spleen (large, friable, and mottled). By 4 DPI, the grossly affected tissues were eyelids (edema and petechial hemorrhages), Harderian gland (edema), spleen (small, pale or mottled), bursa (very small), and thymus (small, edematous or with petechial hemorrhages). The gastrointestinal tract was empty in all infected birds, and the carcasses were thin and dehydrated. Multifocal hemorrhages were observed in the mucosa of the proventriculus (P1307-infected birds) and small intestine and ceca (P5658-infected birds). At 5 DPI, gross lesions were observed in the eyelids (edema and petechial hemorrhages), spleen (small), bursa (very small), thymus (edema and petechial hemorrhages), and kidneys (urate deposits). Multifocal hemorrhages were observed in the mucosa of the proventriculus (P1307- and P5658-infected chickens) and small intestine (only P5658-infected chickens). Noninfected control chickens did not have any gross lesions.
Histopathology, ISH, and IHC
PPMV-1 isolates
The microscopic lesions observed in the infected birds were similar among all the PPMV-1 isolates. Viral nucleoprotein and mRNA were detected in the affected tissues by IHC and ISH, respectively. Mild conjunctivitis was observed at 2 DPI in birds inoculated with Pigeon TX, Pigeon GA, and 84–44407 isolates. Consistent among all the isolates was the presence of multifocal infiltrations of mononuclear inflammatory cells (lymphocytes, plasma cells, and macrophages) in the heart, sometimes associated with necrosis or disruption of cardiac myofibers or both, observed at 5 DPI (Fig. 1) and 10 DPI. Figure 2 illustrates the presence of viral mRNA in the myofibers at 5 DPI. Inflammatory cells, found in the affected areas of the heart, were sometimes also positive for viral mRNA. There was multifocal necrosis with fibrin deposits and apoptotic cells in the spleen of two birds infected with the 84-44407 isolate by 5 DPI. Lymphoid depletion, individual cell necrosis, and apoptotic cells were observed in the spleen, bursa, and thymus by 5 and 10 DPI, mostly in birds inoculated with the Pigeon GA and 84-44407 isolates.
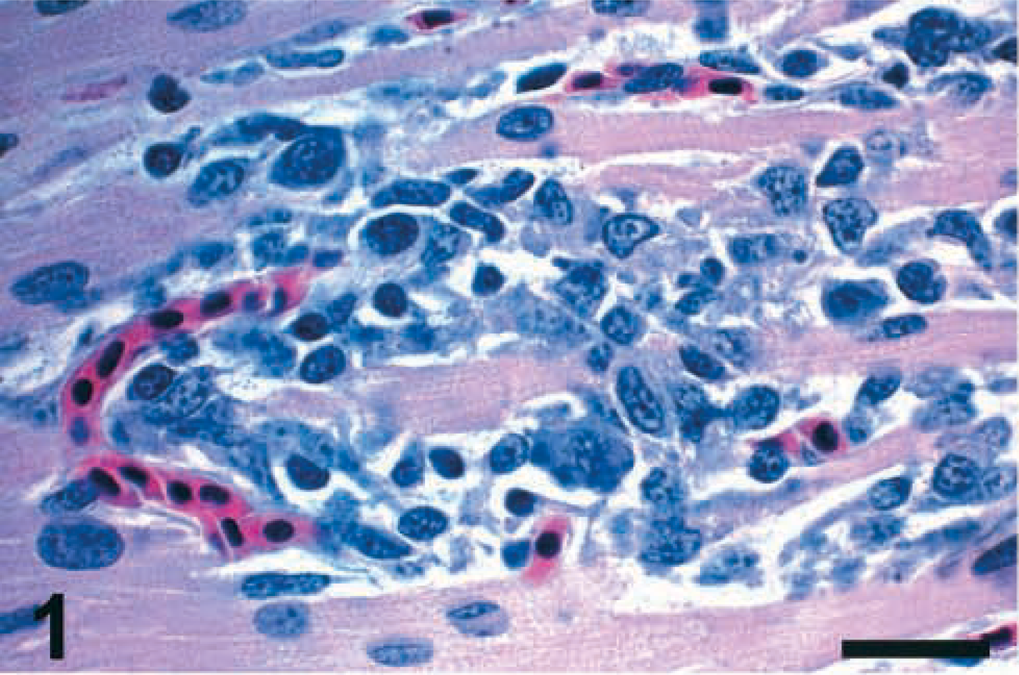
Heart; chicken inoculated with Pigeon TX isolate (PPMV-1); 5 DPI. The myofibers are necrotic or disrupted, and there is infiltration of lymphocytes, plasma cells, and macrophages. HE. Bar = 30 μm.
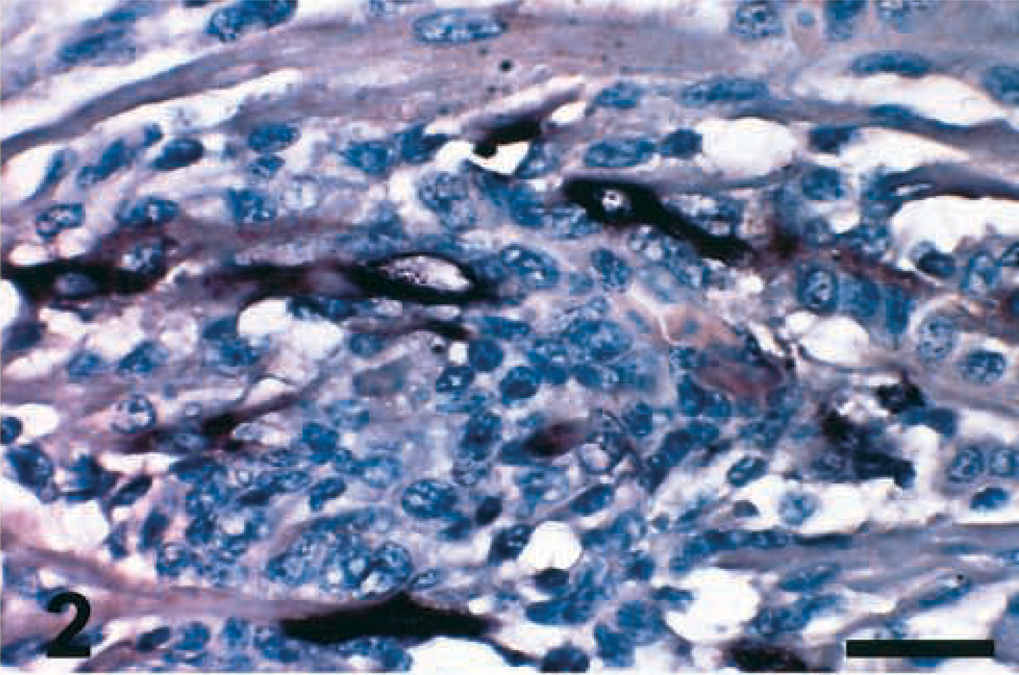
Heart; chicken inoculated with Pigeon TX isolate (PPMV-1); 5 DPI. NDV mRNA (matrix gene) is present in the myofibers (dark brown staining). ISH, hematoxylin counterstain. Bar = 30 μm.
The most remarkable histologic finding with all PPMV-1 isolates was observed in the brain. At 2 DPI, a few cerebral neurons with central chromatolysis were observed in one bird inoculated with the 84-44407 isolate. At 5 DPI, mononuclear inflammatory infiltrations in the neuropil, endothelial cell hyperplasia, and central chromatolysis in the cerebral neurons characterized birds infected with the 84-44407 isolate. Hypercellular areas, interpreted as gliosis and inflammatory reaction, were observed in the molecular layer of the cerebellum associated with Purkinje cell necrosis or loss or both. Vacuolation in the cerebellar white matter was observed in one Pigeon 84–infected bird at 5 DPI.
Brain lesions were severe by 10 DPI and similar among all PPMV-1–infected birds. Multifocal infiltrations of mononuclear inflammatory cells, mostly lymphocytes and plasma cells, were observed surrounding the small capillaries of the molecular layer of the cerebellum (Fig. 3). Hypercellular areas, interpreted as multifocal gliosis, were associated with Purkinje cell loss in some birds at 10 DPI (Fig. 3, inset). Mild vacuolation was sometimes observed in the molecular layer, and focal lymphoplasmacytic meningitis was also observed in a few birds. Severe multifocal lymphoplasmacytic perivascular cuffs extending to the adjacent neuropil (Fig. 4), perivascular edema, endothelial cell hyperplasia, central chromatolysis, neuronal necrosis with neuronophagia, multifocal gliosis, and vacuolation of the neuropil were observed in the cerebrum and brain stem of PPMV-1–infected chickens.
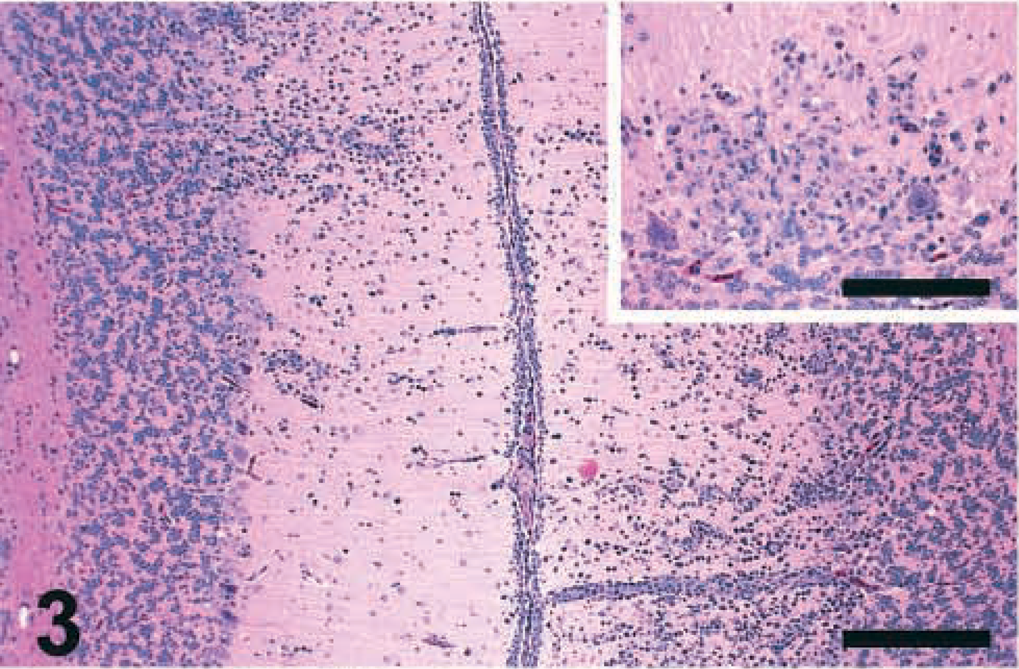
Cerebellum; chicken inoculated with Pigeon GA isolate (PPMV-1); 10 DPI. Multifocal infiltrates of mononuclear inflammatory cells are surrounding the small capillaries of the molecular layer. HE. Bar = 250 μm. Inset: Purkinje cell loss and hypercellular area (gliosis and inflammatory reaction) in the molecular layer. HE. Bar = 150 μm.
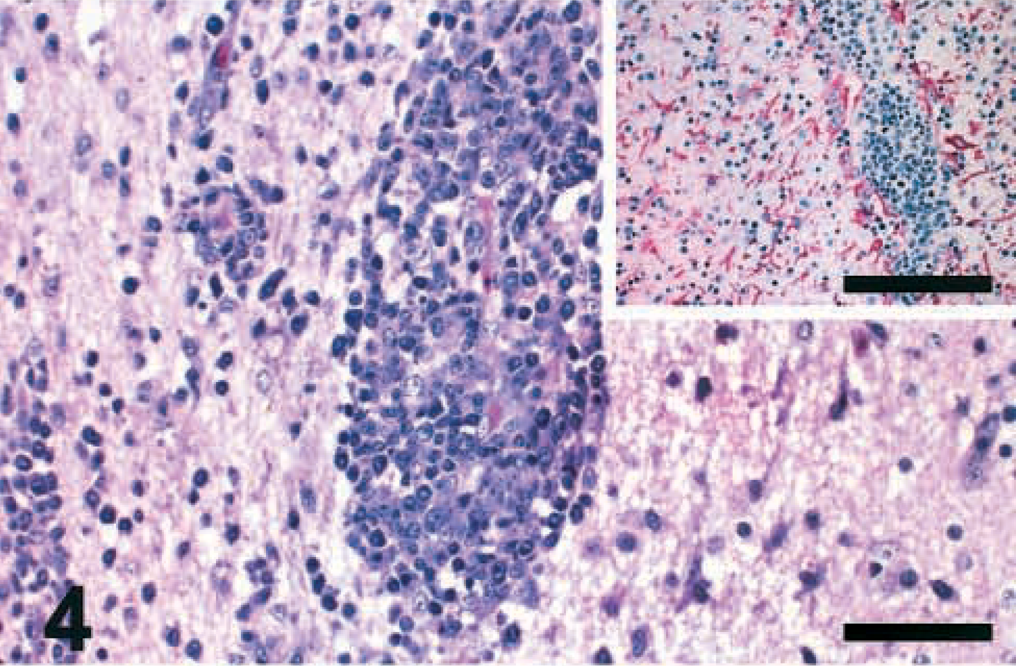
Cerebrum; chicken inoculated with 84–44407 isolate (PPMV-1); 10 DPI. Severe multifocal lymphoplasmacytic perivascular cuffs are extensive to the adjacent neuropil. HE. Bar = 100 μm. Inset: Numerous GFAP-positive astrocytes (red stained) are present in the areas of intense perivascular inflammatory response. IHC, avidin–biotin–alkaline phosphatase, hematoxylin counterstain. Bar = 250 μm.
Numerous GFAP-positive astrocytes were observed in the cerebrum, cerebellum, and brain stem. These changes were more prominent surrounding areas of intense perivascular inflammatory response (Fig. 4, inset) and neuronal loss. A few GFAP-positive cells and cell processes were detected in the multifocal hypercellular areas of the cerebellar molecular layer, including the Bergmann-glia, at 10 DPI. The presence of GFAP-positive cells in the brain of the negative controls was within normal limits. A few lymphocytes were positively stained with IHC-CD3 (T cells) and IHC-BLA-36 (B cells) in the hypercellular areas of the cerebellar molecular layer at 5 DPI. By 10 DPI, many T and B lymphocytes were detected primarily surrounding the small capillaries and in the adjacent neuropil of the molecular layer. Numerous T and B lymphocytes were also observed in the perivascular cuffs and in the adjacent neuropil in other affected regions of the brain at 10 DPI.
Viral N protein was detected in Purkinje cells, other neurons, and glial cells at 5 DPI. One Pigeon 84–infected bird had large amounts of viral N protein and mRNA (Fig. 5) in the cytoplasm of cerebral neurons at 10 DPI. The amount of N protein was quantitatively less in the affected tissues from birds inoculated with the PPMV-1 isolates than in those infected with the APMV-1 isolates. Staining was more often observed in sections of the heart and brain but less often in lymphoid organs at 5 DPI.
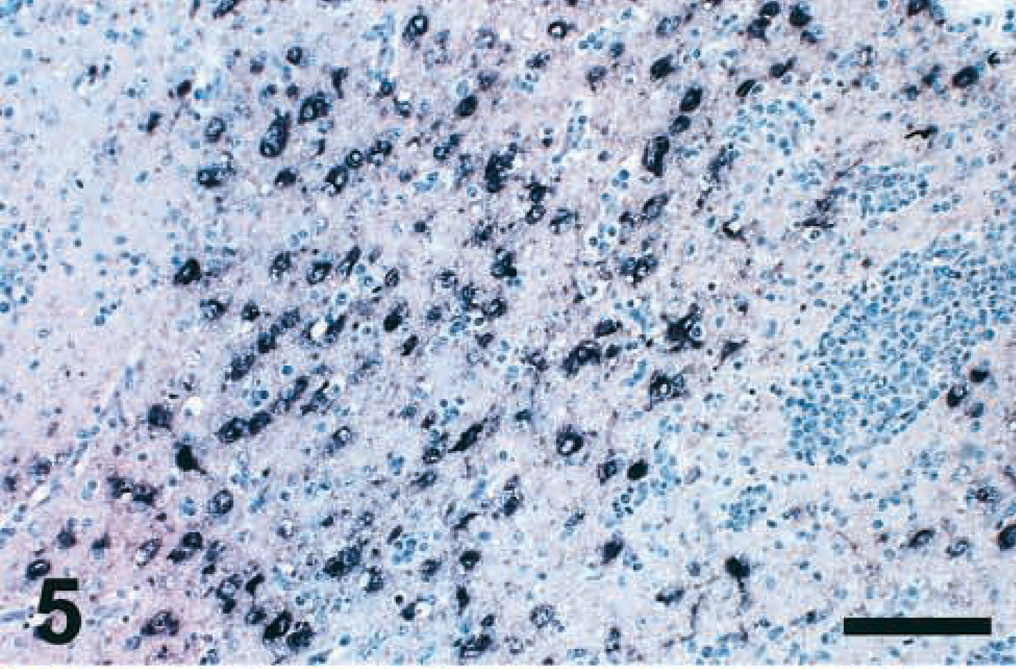
Cerebrum; chicken inoculated with Pigeon 84 isolate (PPMV-1); 10 DPI. Abundant viral mRNA is present in the cytoplasm of neurons. Note intense inflammatory reaction in this area. ISH, hematoxylin counterstain. Bar = 200 μm.
APMV-1 isolates
One P5658-infected bird had multifocal hemorrhages in the cecal tonsils at 2 DPI. Severe conjunctivitis, characterized by edema and lymphoplasmacytic infiltrates, was observed at 2, 4, and 5 DPI among the APMV-1–infected birds. At 4 and 5 DPI, severe lymphoid depletion and necrosis were observed in the lymphoid organs (thymus, bursa of Fabricius, and spleen). The presence of fibrin deposits was marked in the necrotic areas of the spleen. Similar lesions, sometimes associated with multifocal hemorrhages, were observed in the gut-associated lymphoid tissues (GALT), mostly in the cecal tonsils. Necrosis of the superficial epithelium and hemorrhages in the lamina propria were observed in the proventriculus and small intestine. Mild multifocal necrosis was observed in the pancreas and Harderian gland. In the heart, there was mild necrosis of the myofibers associated with mild multifocal infiltrates of macrophages and lymphocytes. Focal areas of necrosis and lymphoplasmacytic infiltrates were observed in the liver of a few birds. There was epithelial hyperplasia with necrosis and fibrin deposits in the trachea and severe pulmonary congestion in a few P1307-infected birds.
The brain lesions were similar to but less frequent than those described for PPMV-1–infected chickens at 5 DPI and were more often observed in birds inoculated with the P1307 isolate. At 2 DPI, only P1307-infected birds had very mild multifocal gliosis in the cerebellar molecular layer. Glial nodules and vacuolation were noticed in the cerebrum and brain stem at 4 and 5 DPI. By 5 DPI, one P1307-infected bird had intracytoplasmic eosinophilic inclusion bodies and distinct accumulations of eosinophilic granular material in neurons of the nucleus reticularis in the brain stem (Fig. 6) and in Purkinje cells. The affected neurons were strongly positive for N protein by IHC. The staining appeared as multiple granules in the cytoplasm (Fig. 6, inset) and the cell processes. No microscopic lesions were observed in the noninfected control chickens.
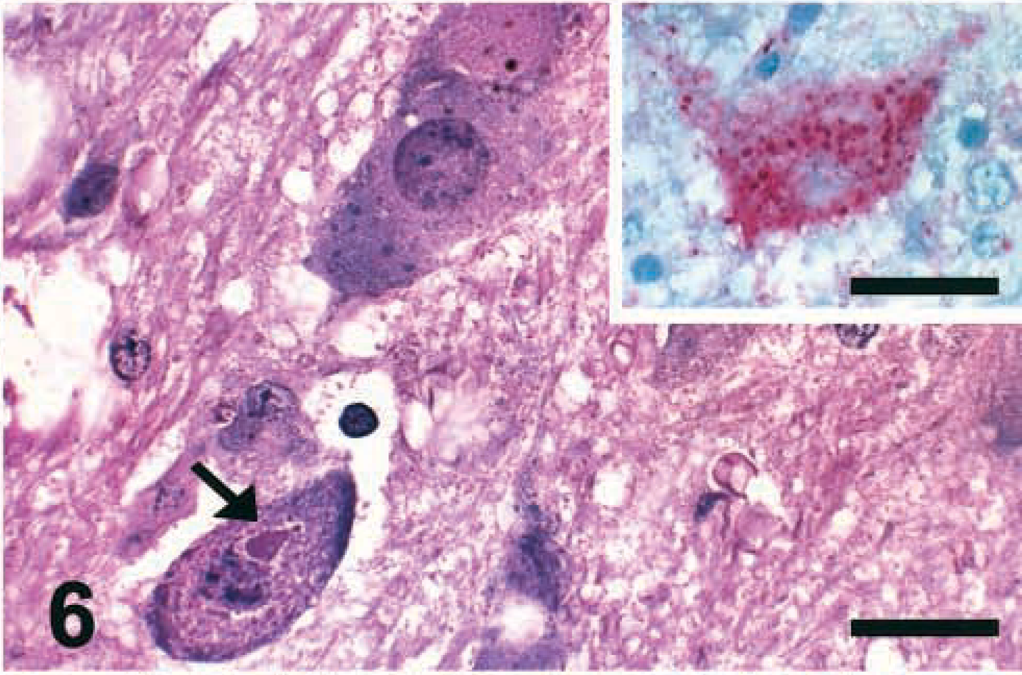
Brain stem; chicken inoculated with P-1307 isolate (APMV-1); 5 DPI. Intracytoplasmic eosinophilic inclusion bodies (arrow) and distinct accumulations of eosinophilic granular material are present in neurons of the nucleus reticularis. Vacuolation of the neuropil is also visible. HE. Bar = 30 μm. Inset: Affected neuron of the nucleus reticularis is strongly positive for viral nucleoprotein (intracytoplasmic red granules). IHC, avidin–biotin–alkaline phosphatase, hematoxylin counterstain. Bar = 30 μm.
Large amounts of viral N protein were observed in all affected tissues. At 2 DPI, N protein staining was observed infrequently in the conjunctiva of the lower eyelid (in epithelial and lymphoid cells), cecal tonsils (lymphoid cells), heart (myofibers), thymus (more prominent in the medulla), and spleen (mostly in large macrophage-type cells). By 4 and 5 DPI, N protein–positive cells were more often seen in spleen (randomly distributed cells), brain (neurons of the cerebrum and brain stem, Purkinje cells, glial cells in the cerebellar molecular layer, and individual cells in the lymphoid aggregates of the choroid plexus), heart (mostly myofibers), thymus (widespread), bursa (widespread), proventriculus (superficial epithelial cells and lymphoid cells), and GALT (small intestine and ceca). Viral N protein was also detected in smaller amounts in epithelium of the conjunctiva and in associated lymphoid follicles, trachea (epithelial cells), lung (epithelial cells and bronchus-associated lymphoid tissue), liver (Kupffer cells), pancreas (lymphoid aggregates), kidney (tubular epithelial cells), feather follicles (epidermis of the follicle wall) and in the feather pulp. IHC-N was negative in all tissue sections of the noninfected control birds.
Apoptosis assays
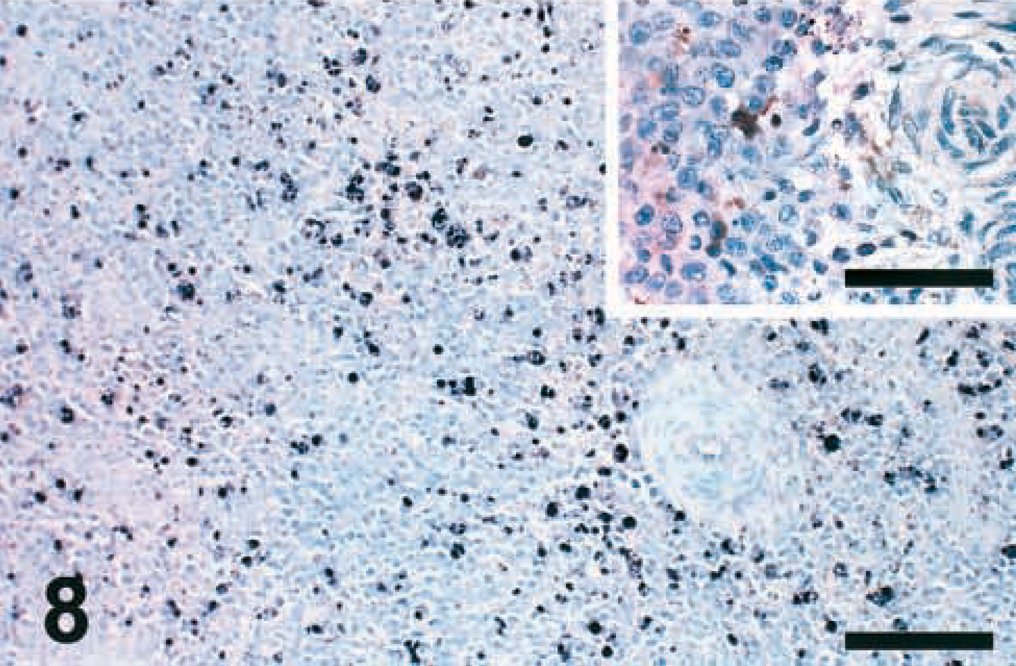
Apoptotic cells were detected by two different assays, TUNEL and IHC-Casp, in spleen sections of chickens infected with the six pigeon NDV isolates (PPMV-1 and APMV-1) and of control chickens. At 2 DPI, the number of IHC-Casp–positive cells in the spleens of infected chickens was 3–5 times higher than that in the spleens of control chickens. IHC-Casp–positive reaction was found mostly in the area of the ellipsoid-associated cells (EACs) and was characterized by dark brown staining of dendrite-shaped cells surrounding the penicilliform capillaries (Fig. 7). This distribution pattern was very consistent among all the isolates. At 2 DPI, the number of randomly distributed apoptotic cells in TUNEL-stained spleen sections of infected birds was small (Fig. 7, inset) and equal to or slightly higher than that detected in the noninfected controls. At 5 DPI, the number of TUNEL-positive cells increased 2–3 times (Fig. 8), and the number of IHC-Casp–positive cells decreased markedly (Fig. 8, inset) in spleen sections of birds inoculated with almost all isolates. At 10 DPI, no differences were observed in the number of positive cells (stained by IHC-Casp and TUNEL) found in the spleen sections of infected chickens versus the noninfected controls.
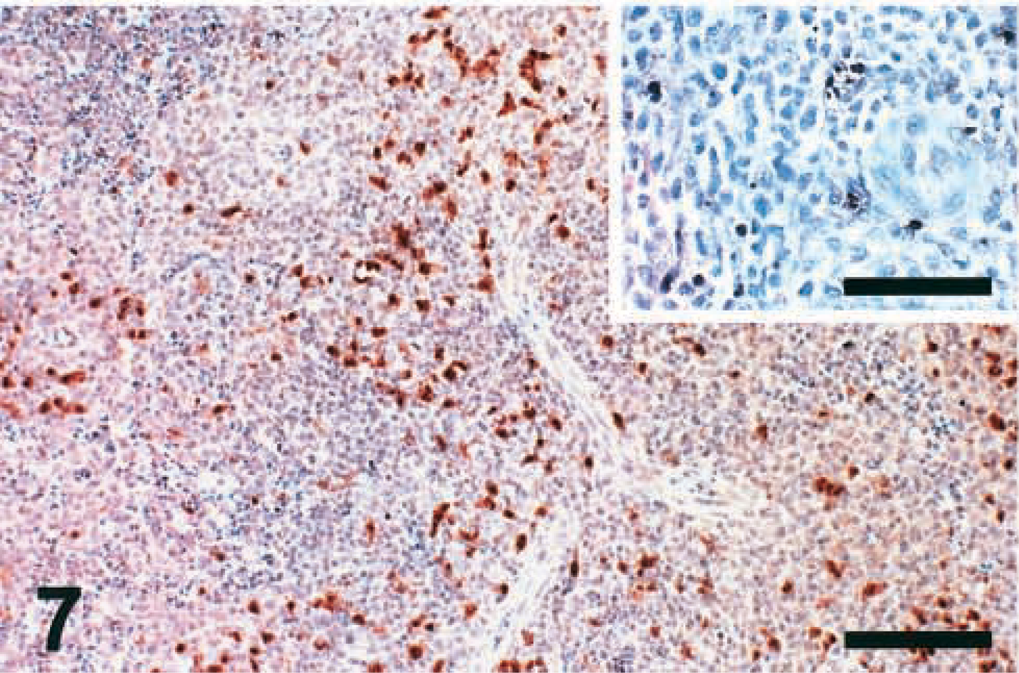
Spleen; chicken infected with Pigeon TX isolate (PPMV-1); 2 DPI. Large amounts of apoptotic ellipsoid-associated cells (EACs) are positive for active caspase-3 surrounding the penicilliform capillaries. IHC, avidin–biotin–peroxidase, hematoxylin counterstain. Bar = 230 μm. Inset: There are few randomly distributed TUNEL-positive apoptotic cells. TUNEL, hematoxylin counterstain. Bar = 100 μm.
Spleen; chicken infected with P-1307 isolate (APMV-1); 5 DPI. A large number of TUNEL-positive cells are evenly scattered throughout the spleen. TUNEL, hematoxylin counterstain. Bar = 230 μm. Inset: The number of active caspase-3–positive cells is markedly small. IHC, avidin-biotin-peroxidase, hematoxylin counterstain. Bar = 100 μm.
Discussion
Six pigeon-origin isolates of NDV, previously passaged through 2-week-old chickens,23 were inoculated intraconjunctivally into 4-week-old SPF White Leghorns. Four isolates (Pigeon TX, Pigeon GA, 84–44407, and Pigeon 84) were previously classified by Mab binding profiles (using the HI test) as PPMV-1, and two isolates (P1307 and P5658) were defined as APMV-1.20,23
Although some birds presented with severe neurologic signs, overt clinical disease was not a major finding in chickens inoculated with PPMV-1 isolates, and no mortality occurred during the 14-day observation period. However, all four PPMV-1 isolates caused similar tissue damage in the infected chickens. Microscopic lesions were observed in the heart, brain, and lymphoid organs. The most remarkable and consistent histopathologic changes were observed in the brain at 5 DPI and more so by 10 DPI. The lesions were characterized by severe multifocal lymphoplasmacytic encephalitis with neuronal necrosis, perivascular edema, endothelial hyperplasia, gliosis, and less often neuropil vacuolation and meningitis. Multifocal perivascular lymphoplasmacytic inflammatory reaction and gliosis associated with areas of Purkinje cell loss were consistently observed in the cerebellar molecular layer. Sometimes vacuolation of the adjacent neuropil occurred. Of the 11 PPMV-1–infected birds with distinct brain lesions, only seven had overt neurologic signs.
Discordant temporal relationship between virus replication in the brain and occurrence of clinical disease has been reported with a neurotropic APMV-1 strain in chickens. In that study,47 central nervous system disease developed several days after maximal virus replication had occurred in the brain. The occurrence of clinical disease, sometimes followed by death, was interpreted as a culmination of the brain damage associated with viral infection.47 Although mortality was not observed in our PPMV-1–infected birds, the brain lesions probably would be severe enough to cause significant neurologic deficit and affect normal development of commercial chickens.
Significant lesions observed in the heart consisted of multifocal lymphoplasmacytic and histiocytic myocarditis associated with myofiber necrosis/disruption. Similar lesions were also described in a previous passage study with the same PPMV-1 and APMV-1 isolates23 and in several other studies with APMV-1 strains.8,28 Because NDV-infected chickens have altered electrocardiograms35,36 and because of the functional importance of the heart, bird performance, including weight gain and growth rate, could be reduced among commercial chickens infected with pigeon NDV.
The use of IHC and ISH to detect APMV-1 infections in tissues of chickens has been reported several times.11,24,33 However, to our knowledge, studies reporting the use of those methods to define tissue distribution of PPMV-1 infection in chickens are scarce.23 In our study of PPMV-1–infected birds, viral replication was characterized by the presence of viral N and mRNA in the same sites of damage among the affected tissues.
Large amounts of virus were detected in the brain of one Pigeon 84–infected bird at 10 DPI. In comparison, in chickens infected with two APMV-1 neurotropic strains (Texas GB and Turkey North Dakota) Brown et al.11 detected only very little viral mRNA by ISH at 10 DPI. In birds infected with another neurotropic APMV-1 strain (Missouri-(H) Len 1950), NDV could not be recovered by virus isolation from pooled brain tissue after 9 DPI.47 The abundant viral replication observed in the brain of PPMV-1–infected birds appears to be somewhat unusual compared with that of previously described APMV-1 isolates, which have a tropism for brain.11,47
In brain sections of PPMV-1–infected birds, GFAP-positive astrocytes were detected in remarkably high numbers, mostly surrounding the areas of perivascular inflammatory response and neuron degeneration/necrosis in cerebrum and brain stem. GFAP-positive reaction was observed in the areas of gliosis adjacent to Purkinje cells at 10 DPI. Normally in chickens, the Bergmann-glia, a highly specialized astrocytic cell type found in the cerebellar molecular layer, is GFAP negative.19 Reactive gliosis following a stab wound lesion was described in the cerebellar molecular layer in chickens. In that case, the Bergmann-glia showed a marked immunopositivity to GFAP around the lesion site.1 Similar GFAP staining was observed in the Bergmann-glial cells inside or adjacent to the multifocal cerebellar reactive gliosis of infected chickens in our study.
On HE-stained sections, it was not easy to identify all the cell types involved in the hypercellular areas observed in the cerebellar molecular layer mostly at 5 DPI. For this reason, brain sections were also stained for lymphocyte subset (CD3 and BLA-36 antibodies) to detect T and B lymphocytes, respectively. Only a few of the cells seen in the molecular layer were positive by IHC-CD3 and IHC-BLA-36 at 5 DPI. Both lymphocyte cell types were abundant in those areas at 10 DPI, surrounding the walls of small capillaries and in the adjacent neuropil. T and B lymphocytes were also detected as cell components of the perivascular cuffs and inside the adjacent neuropil, mostly in the affected cerebral sections.
With the two pigeon-origin APMV-1 isolates, severe clinical disease and mortality were observed in the inoculated chickens. Not all NDV isolates coming from pigeons are the variant PPMV-1. Pigeons infected with highly virulent NDV of the APMV-1 type represent a very serious threat to commercial poultry flocks. P1307 and P5658 were characterized as velogenic isolates in previous tests.16,23 In the present study, both isolates produced gross and histopathologic changes characteristic of highly virulent NDV, typical of the velogenic viscerotropic NDV pathotype11,18,34,39 Viral replication, detected by IHC-N, in APMV-1–infected birds was abundant and much greater than that in birds inoculated with the PPMV-1 isolates.
Intracytoplasmic inclusion bodies and distinct accumulations of eosinophilic granular material, as observed in the brain of one P1307-infected bird, are not often reported for NDV-infected birds. Case reports of ND include the presence of intranuclear and intracytoplasmic eosinophilic inclusion bodies in epithelial cells of esophageal glands and in neurons of the ganglia subjacent to the adrenal gland in infected pheasants and in hepatocytes of cuckoo doves.43 Transmission electron microscopy revealed intracytoplasmic inclusion bodies suggestive of paramyxovirus in endothelial cells in blood vessel from the esophagus of an NDV-infected game chicken.14
Several investigators have detailed many different techniques26–28,30 to detect NDV-induced apoptosis. Most of the studies were performed in cell culture or in vivo with the APMV-1 Texas GB strain. Texas GB strain, a neurotropic virus, causes both necrosis and apoptosis of infected chicken peripheral blood lymphocytes,30 apoptosis of chicken embryo fibroblasts,26 and apoptosis of peripheral blood mononuclear cells,27 including virus-infected macrophages.29 In contrast, during this study, apoptosis assays were confined to those that could be used in formalin-fixed, paraffin-embedded tissues.
In the lymphoid organs and tissues examined in this study, the most striking results were obtained from APMV-1–infected birds, where apoptosis was followed by extensive lymphocellular necrosis. Less prominent lesions were observed in lymphoid organs of the PPMV-1–infected birds and were characterized by apoptosis, depletion, and less often, by necrosis of lymphoid cells. The occurrence of apoptosis in lymphoid organs was further analyzed in sections of the spleen by two different approaches, the TUNEL assay and IHC-Casp staining. Among all the NDV isolates studied here, apoptotic cells were detected earlier (at 2 DPI) and in greater numbers mostly in the area of the EACs of the spleen using the IHC-Casp staining as compared with the TUNEL assay. At 5 DPI, TUNEL staining was more extensive than IHC-Casp, and the apoptotic cells were randomly distributed in the spleen sections. These results could be explained by the fact that active caspase-3 is an enzyme that functions early, effecting apoptosis,13 and thus its detection in a “preapoptotic” cell would occur before DNA breakdown, which is detected by the TUNEL assay.25,37 The role of caspase-3 in apoptosis during NDV infection might be similar to that found in another study, where Sendai virus (another paramyxovirus) induced apoptosis through activation of caspase-3 and caspase-8.10 NDV infection followed by early NDV-induced apoptosis of the EACs in the spleen might have severe consequences for the bird immune response because EACs play such a crucial role in antigen processing and presentation.41
We demonstrated that PPMV-1 causes severe lesions among infected chickens, mostly affecting the heart and brain. Pigeons must be considered seriously as a potential source of NDV infection and disease for commercial poultry flocks. Preventive NDV vaccination of domestic pigeons and strict biosecurity measures should be taken to reduce the risk of pigeon ND outbreaks in chickens.
Footnotes
Acknowledgements
G. D. Kommers was supported by a scholarship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (Brasília, DF, Brazil). This work was funded by US Poultry and Egg Association grant 406 and USDA-ARS-CRIS project 6612-32000-021-00D. We acknowledge the excellent technical support of Phillip Curry and Erica Behling-Kelly.
