Abstract
Selected lymphoid and reproductive tissues were examined from groups of 3-week-old chickens and 62-week-old hens that were inoculated choanally and conjunctivally with 106 EID50 of a virulent Newcastle disease virus (NDV) isolate from the California 2018–2020 outbreak, and euthanized at 1, 2, and 3 days postinfection. In the 3-week-old chickens, immunohistochemistry for NDV and for T and B cell lymphocytes, as well as in situ hybridization for IL-1β, IL-6, IFN-γ, and TNF-α revealed extensive expression of IL-1β and IL-6 in lymphoid tissues, often coinciding with NDV antigen. IFN-γ was only expressed infrequently in the same lymphoid tissues, and TNF-α was rarely expressed. T-cell populations initially expanded but by day 3 their numbers were below control levels. B cells underwent a similar expansion but remained elevated in some tissues, notably spleen, cecal tonsils, and cloacal bursa. Cytokine expression in the 62-week-old hens was overall lower than in the 3-week-old birds, and there was more prolonged infiltration of both T and B cells in the older birds. The strong pro-inflammatory cytokine response in young chickens is proposed as the reason for more severe disease.
Keywords
Newcastle disease (ND), caused by Newcastle disease virus (NDV, also known as avian paramyxovirus-1), in the new genus Orthoavulavirus of the family Paramyxoviridae, represents a serious threat to the poultry industry as well as to backyard chickens the world over. The virulent forms of the virus are reportable to the World Organisation for Animal Health (OIE) if they meet the criteria for virulence, and outbreaks of this disease may result in serious trade restrictions on poultry or poultry products. 12 Infection with highly virulent strains in naïve chickens results in a rapid course of disease and high mortality. Multiple pathogenesis studies in birds have been conducted with various strains of NDV. These studies point to a key difference between the highly virulent and lesser virulent viruses. The virulent NDV (vNDV) possess a fusion protein configuration that allows for cells to enter beyond trypsin-coated mucosal surfaces. With the subsequent viremia, the virulent viruses infect macrophages preferentially, resulting in necrosis in multiple lymphoid sites and organs. 1,2,10,18,19
There are a few studies indicating that an over-vigorous innate immune response may be occurring with vNDV infection. 6,8,15 The innate immune response consists of a constellation of factors dominated by pro-inflammatory cytokines that serve to decrease infection in the early stages and initiate the development of the specific immune response. When this innate immune response is hyperresponsive, it can create severe disease in the host and has been referred to as a “cytokine storm.” 4 Macrophages are the key targets of vNDVs and also the major conductors of the innate immune response.
Previous studies proposing that pro-inflammatory mediators are overexpressed in vNDV infections have used molecular methods on tissue extracts. 6,8,15 It was the aim of this study to examine cytokine expression in situ, in tandem with visualization of NDV and T and B cell lymphocytes, to chronicle cytokine expression and compare it to the presence of NDV and the relative amounts of T and B cell lymphocytes. This study focused only on those tissues that demonstrate the most severe effects subsequent to inoculation of NDV, that is, the lymphoid area at the portal of entry, spleen, cecal tonsils, cloacal bursa, and thymus. Additionally, as part of the study involving adult hens, the reproductive tract was also examined.
Materials and Methods
Bird Tissues
Paraffin blocks of tissue were obtained from the USDA’s National Poultry Research Center, from a previously reported pathogenesis study in chickens with the virus that was circulating in California in 2018 (chicken/California/D1806566/2018; CA18 is used hereafter). This virus was designated as belonging to sub-genotype V.1 (former Vb). In that study, morbidity, mortality, shedding, and viral titers were monitored in 2 large group of birds, and presence of the virus was assessed by immunohistochemistry on formalin-fixed tissues taken at 4 and 5 days of infection. 5 Birds examined included 3-week-old chickens and 62-week-old hens. All birds were infected with CA18, through both choanal and left eye drop inoculation of approximately 106 embryo infectious doses50 (EID50) in 0.1 ml of brain heart infusion (BHI) broth. The virus inoculum had an ICPI of 1.76. Negative control birds received a BHI broth mock inoculum and were housed separately. All tissues were collected immediately after euthanasia in 10% NBF, and then embedded within paraffin before 72 hours. It is these tissues (1, 2, and 3 days postinfection [dpi]) that are used in this current study. Tissues from 2 birds at each time point were examined in the various assays. The experimental protocol was approved by the National Poultry Research Laboratory IACUC, Protocol #USNPRC-2020-018.
For the 3-week-old birds, the focus was on the tissues most impacted by the virus based on many previous studies demonstrating that lymphoid tissues are preferentially damaged. These are also the tissues most likely to produce cytokines. These tissues included nasal turbinates around the area of the choana (site of inoculation), spleen, lymphoid areas at proximal ceca (known as “cecal tonsils”), thymus, and cloacal bursa. For the 62-week-old hens, the following tissues were assessed using the same techniques: nasal turbinates, spleen, cecal tonsils, and reproductive tract.
Histopathology and Immunohistochemistry
Paraffin sections were cut at 3 to 4 µm, stained routinely with hematoxylin–eosin (H&E), and examined microscopically for evidence of pathologic changes. Immunohistochemistry (IHC) was used for detection of NDV, B cells, and T cells using a similar protocol for all 3 antibodies. Unstained slides were deparaffinized and subjected to antigen retrieval by microwaving for 20 minutes at minimum power in Vector antigen unmasking solution (Vector Laboratories), followed by application of a universal blocking reagent (Biogenex). The primary antibodies were as follows: anti-NDV was a polyclonal antibody raised in rabbits inoculated with an NDV nucleoprotein synthetic peptide (TAYETADESETRRIC) and used at 1:8000 dilution; for T cells, anti-CD3 was a rabbit polyclonal antibody (DAKO), which was designed for detection of human T cells but has been shown to work in numerous species, and used at 1:1000 dilution; for B cells, a mouse monoclonal antibody to B cells, anti-Chicken CD268/BAFF-R (Bio-Rad), was used. The detection system for NDV was an alkaline phosphatase–labeled polymer specific for the Fc portion of rabbit immunoglobulin (LabVision) and chromogen was a naphthol-based dye, Fast Red (DAKO). A biotin-free probe and polymer was used for CD3 and B cell marker, by using a specific probe to detect mouse antibody followed by HRP-polymer that binds to both probe and rabbit antibodies. For these protocols, chromogen was DAB (Biocare Medical). Positive controls included tissues from previous studies, and negative controls were chickens inoculated with BHI.
In Situ Hybridization
Probes were designed by and obtained from Advanced Cell Diagnostics as follows: interleukin (IL)-1β in C1 channel, IL-6 in C2 channel, tumor necrosis factor (TNF)-α in C1 channel, and interferon (IFN)-γ in C2 channel. Manufacturer’s directions guided procedures. Briefly, 4 µm paraffin-embedded sections were deparaffinized, rehydrated, and digested with Protease Plus (supplied by manufacturer) at 40 °C for 30 minutes. Hybridization occurred at 40 °C for 2 hours with both C1:C2 channel probes at 1:50 dilutions. This was followed by applying a series of compounding chromogenic amplifiers (AMP1 to AMP6) to detect the red color for the C2 channel probes. After a brief blocking, AMP8 to AMP10 were applied for detection of the green color for the channel 1 probes. Sections were counterstained with 50% hematoxylin for 30 seconds and cover-slipped with VectaMount (Vector Laboratories). Both positive control probe (Gg-UBC) and negative control probe (2-plex) for chicken were used with the same procedure (supplied by the manufacturer). Positive cells with the C1 probe had green signal, and the signal with the C2 probe was red.
For all IHC and RNAScope procedures, tissues from both birds at each time point were assayed. Ten 400× fields were examined for each tissue to arrive at semiquantitative scores designated for IHC and cytokine amounts.
Results
Three-Week-Old Birds
Results are presented in Tables 1 and 2. A complete description of all results at all time points is included as Supplemental Material.
Scoring of Newcastle disease virus (NDV) and cytokine expression in 3-week-old chickens after inoculation with virulent NDV. NDV and cytokines were detected by immunohistochemistry and in situ hybridization, respectivelya.
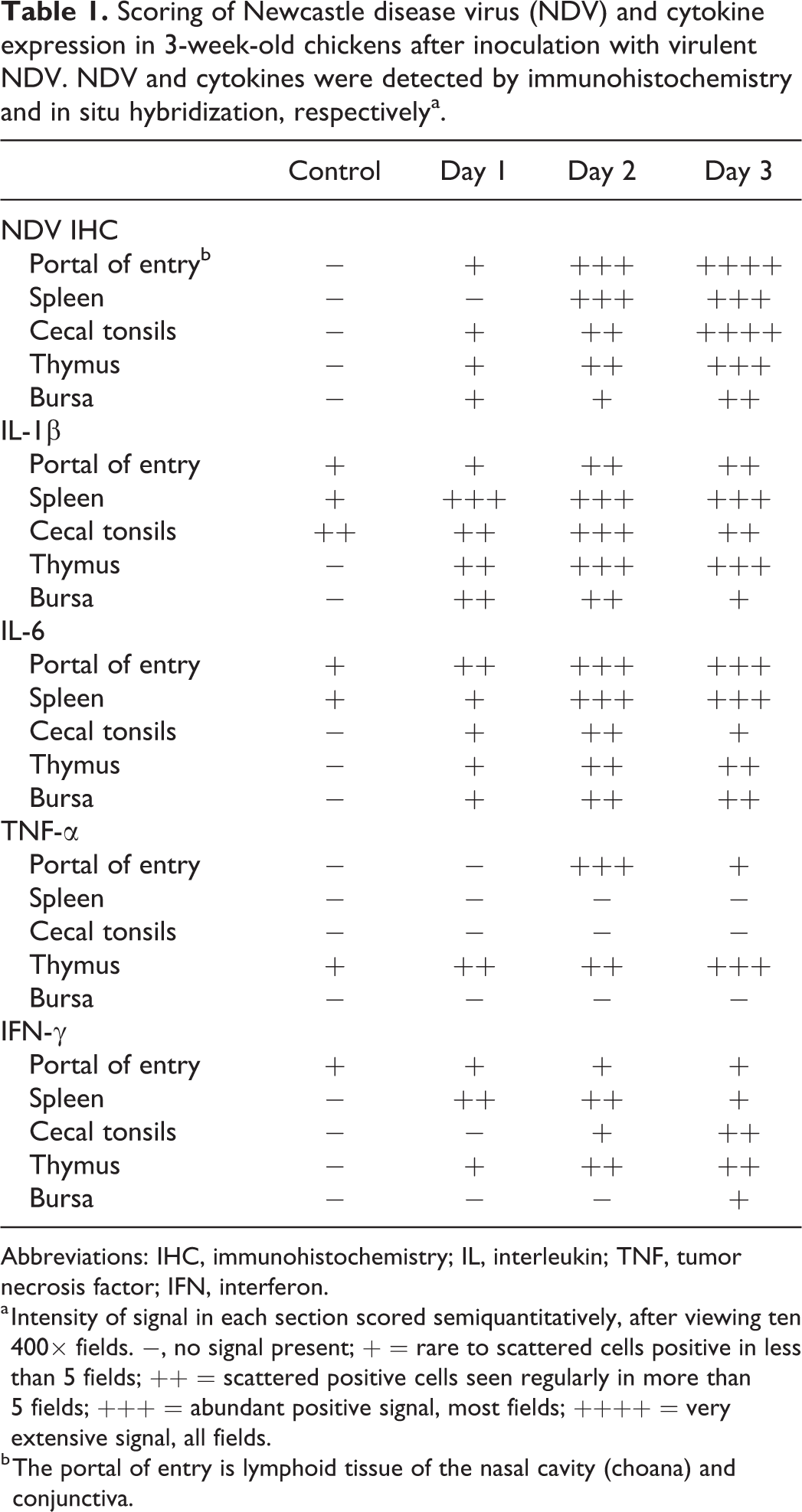
Abbreviations: IHC, immunohistochemistry; IL, interleukin; TNF, tumor necrosis factor; IFN, interferon.
a Intensity of signal in each section scored semiquantitatively, after viewing ten 400× fields. −, no signal present; + = rare to scattered cells positive in less than 5 fields; ++ = scattered positive cells seen regularly in more than 5 fields; +++ = abundant positive signal, most fields; ++++ = very extensive signal, all fields.
b The portal of entry is lymphoid tissue of the nasal cavity (choana) and conjunctiva.
Relative numbers of T and B cells in specific organs, as detected by IHC, 3-week-old chickensa.

Abbreviation: IHC, immunohistochemistry.
a=, same as negative controls; < or >, less or more (1–2×) than in negative control; << or >>, less or more (>2×) than in negative control.
Briefly, NDV signal generally increased with time in all tissues and correlated with the amount of damage seen histologically (Figs. 1a, 2a, 3a). In infected birds, T cell amounts were initially greater than in noninfected controls, but by 3 dpi were decreased below levels seen in the noninfected controls. B cell amounts increased and remained elevated over control levels, except at the portal of entry where they were decreased at 3 dpi (Fig. 2c). For the cytokines, IL-6 and IL-1β expression had similar patterns to each other, with increasing amounts through the infection period, and some tissues having extensive signals at 3 dpi (Figs. 1b, 2b, 3b). The amount of IFN-γ signal was generally less than those seen for IL-6 or IL-1β, and it was most consistent in spleen and thymus (Figs. 1c, 3c). TNF-α was absent in 3 of the 5 tissues sampled, at every time point.
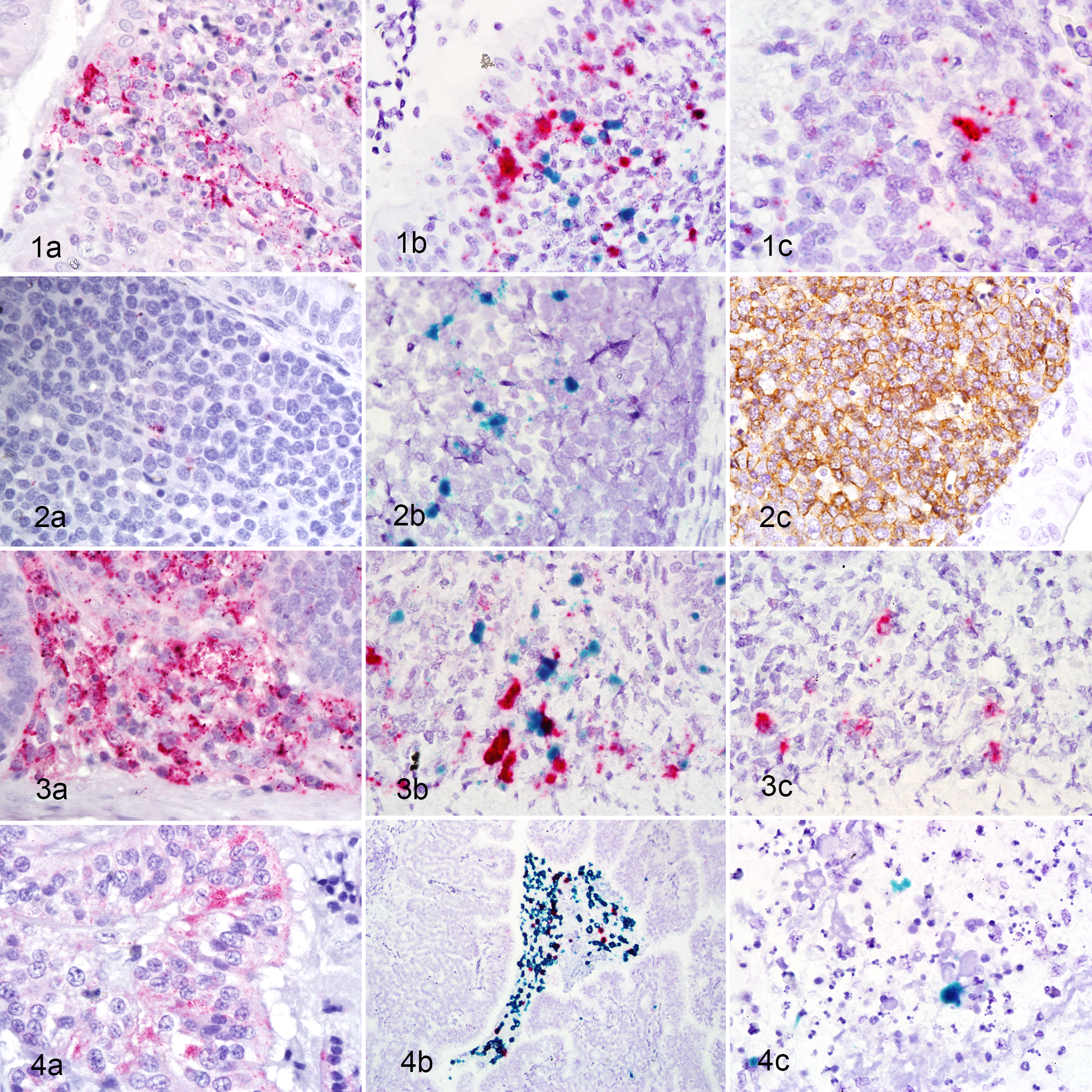
Newcastle disease (experimental infection), chickens.
62-Week-Old Hens
For the 62-week-old birds, the focus of investigation was on the portal of entry, spleen, cecal tonsils, and reproductive tract. For the latter, 5 areas were examined initially: ovary, infundibulum, magnum, isthmus, and shell gland. Only infundibulum and ovary are described here because signals were more pronounced in these tissues, with the other areas usually lacking any evidence of NDV, lymphocytes, or cytokines.
Results are presented in Tables 3 and 4. More details are presented in the Supplemental Material.
Scoring of Newcastle disease virus (NDV) and cytokine expression in 62-week-old hens after inoculation with virulent NDV. NDV and cytokines were detected by immunohistochemistry and in situ hybridization, respectivelya.
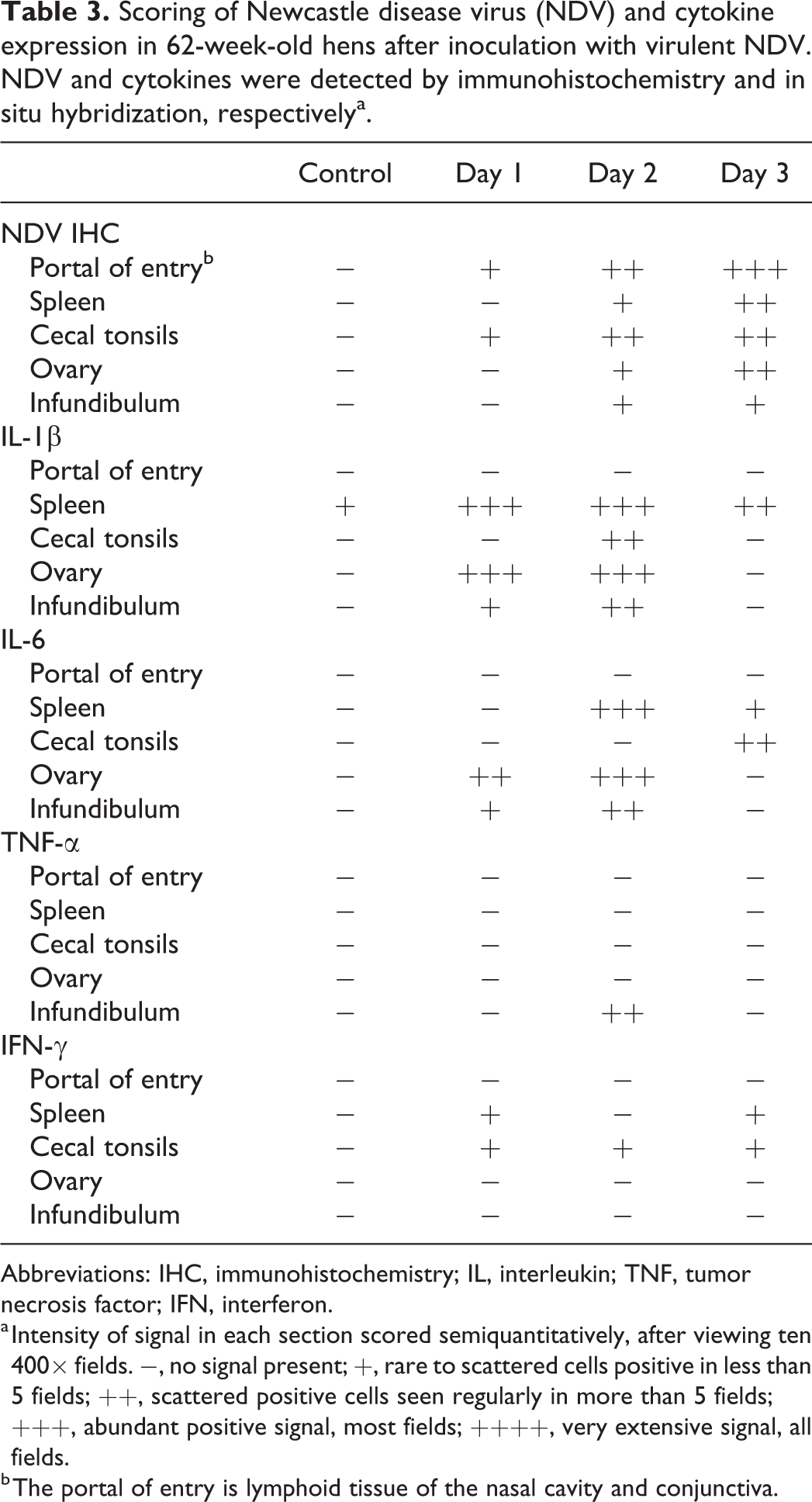
Abbreviations: IHC, immunohistochemistry; IL, interleukin; TNF, tumor necrosis factor; IFN, interferon.
a Intensity of signal in each section scored semiquantitatively, after viewing ten 400× fields. −, no signal present; +, rare to scattered cells positive in less than 5 fields; ++, scattered positive cells seen regularly in more than 5 fields; +++, abundant positive signal, most fields; ++++, very extensive signal, all fields.
b The portal of entry is lymphoid tissue of the nasal cavity and conjunctiva.
Relative numbers of T and B cells in specific organs, as detected by IHC, 62-week-old hensa.

Abbreviation: IHC, immunohistochemistry.
a=, same as negative controls; < or >, less or more (1–2×) than in negative control; << or >>, less or more (>2×) than in negative control.
Briefly, amount of NDV signal increased with time and correlated with the extent of histologic damage seen (Fig. 4a). For the lymphocyte IHC, in general there were increases in both T and B cell signal through the time course, with at least double the negative control levels at 3 dpi, in most of the tissues examined. For cytokines, expression of IL-1β, and to a lesser extent IL-6, were prominent and 1 and 2 dpi (Fig. 4b). TNF-α signal was rarely seen, and IFN-γ expression was scant at all time points (Fig. 4c).
Discussion
This study provided insight into the early immune responses in chickens infected with vNDV. In situ detection of NDV, T cells, B cells, and various cytokines allowed for a larger glimpse into the early cascading of the host response subsequent to infection, in 2 different age groups.
Lymphocytes, especially T cells, are key players in both innate and specific immune responses. In the 3-week-old birds, there was a notable rise in T cell numbers in tissues early in infection but then a general decrease, to below control levels. For B cells, the pattern was similar except that increased presence of B cells remained in spleen, cecal tonsils, and bursa. This is similar to what has previously been reported in experimental infections with vNDV, where there was a decrease in overall numbers of T cells but a general increase in B lymphocytes. In the 3-week-old birds, the decrease in T cells after an initial increase was noted in multiple tissues, and in general, there was significant T cell depletion by 3 dpi. The reasons are unclear but are thought to be due to apoptosis, as suggested by other authors. 7,13
In the older birds, there was no marked decrease in T cells from infection through to 3 dpi. Instead, there was an overall marked increase in T cells, compared to the controls. Similarly, B cells were abundant in the key lymphoid organs of the older birds at 3dpi, in striking contrast to the modest levels seen in the 3-week-old birds.
Gene expression of inflammatory cytokines, especially IL-1β and IL-6, occurred early in NDV infection. Prominent IL-1β and I-L6 expression was seen in multiple tissues. The amounts of these 2 cytokines often correlated well to the amount of positive signal for NDV in the same tissue. Also, the signals for these cytokines and for NDV were often in proximity, indicating that NDV replication and the stimulus for promotion of these mediators are occurring in the same area of the tissue.
For the 3 pro-inflammatory cytokines produced primarily by macrophages, that is, IL-1β, IL-6, and TNF-α, the cytokines were often expressed by cells in areas where there was also positive NDV signal. The stimulus for production of pro-inflammatory mediators includes viral pathogen-associated molecular patterns (PAMP) recognizing pattern recognition receptors (PRR) that in turn cause Toll-like receptors (TLR) to trigger the activation of innate antiviral and pro-inflammatory genes. The pathway is well-recognized in mammalian systems, and although there are some minor differences in poultry, overall the system is remarkably similar. 3
Unregulated and overvigorous activation of the innate immune response has been termed “cytokine storm” and is often noted as the main cause of disease in some infections. 4 It is likely that this also occurs in acute ND and is responsible for the severe clinical disease noted in early infection, in the absence of any obvious organ system failure.
Previous studies, using RNA extracted from spleens in paraffin blocks, identified that IL-6 was produced at high levels as early as 2 dpi, which corresponds with the present study. 6 Levels of both IL-6 and IL-1β were prominent and generally increasing, with highest levels generally at 2 dpi. IL-6 is known to come from activated macrophages as well as from fibroblasts and endothelium, which fits with the pattern of signal which was seen in multiple tissues in the study reported here. In another study focusing on spleens, in which birds were infected with vNDV and spleens harvested at 1 or 2 dpi, IL-6 was expressed at very high levels at 1 dpi, and both IL-6 and IL-1β were elevated at 2 dpi. 15 In a third study in which chickens were infected with vNDV, spleens taken at 2 dpi and subjected to Affymetrix technology, there were markedly elevated levels of IL-6 and IL-1β. 8
The role of IFN-γ in NDV infection remains uncertain. This cytokine, made by T cells and NK cells subsequent to stimulation, has pleiotropic effects, but serves to promote both innate and adaptive responses. It is thought to especially promote Th1 immune responses, which are so essential in controlling intracellular pathogens. Ecco et al examined spleen tissue from birds experimentally infected with vNDV and found that at day 2 there were very high levels of IFN-γ. 6 Then it was hypothesized that if IFN-γ were present earlier in vNDV infection, perhaps it could provide the type of response needed to thwart the infection. Consequently, Susta et al created an infectious clone with the gene for IFN-γ inserted. This allowed for IFN-γ production earlier, that is, at the same time as the earliest viral replication, and found there was decreased disease severity and less mortality. 17 However, subsequent studies incorporating IFN-γ with various vaccine formulations did not improve immune response. 16 In the present study, IFN-γ expression was seen most reliably in the 3-week-old chickens with relatively minimal evidence of its presence in the group of older infected birds. For the younger chickens, greatest amounts were noted in spleen and thymus. A previous study on bursas collected from chickens experimentally infected with vNDV demonstrated increased levels of IFN-γ at 1 to 3 dpi. 11 The exact role of IFN-γ in vNDV, in either protection from viral infection and/or enhancing the cytokine storm aspect of infection, remains unknown.
Earlier studies involving cytokine production in vNDV infection did not analyze TNF-α because the gene had not yet been documented in chickens. The avian ortholog was recently discovered. 14 Biological activity of the protein expressed from this gene was confirmed. 14 In chickens, TNF-α is known to be produced by both macrophages and T helper cells. In our study, it appears that TNF-α may not play a major role in the early response to NDV, as its presence was not consistently noted, and amount of signal was generally low.
Virulent ND viruses are known to cause clinical disease in laying hens, with lesions and virus noted in the reproductive tract. Marked decreases in egg production have been noted at 1 to 2 weeks postinfection. 9 In this study, which was very early in infection, virus was found in the reproductive tract, along with associated lesions. Cytokine production, including IL-1β, IL-6, and TNF-α, was noted, as well as marked increases in B cells. This acute inflammatory response likely creates alterations which lead to permanent impairment of parts of the tract.
Comparing viral replication and cytokine transcription in the 2 different age groups demonstrates that both viral replication and cytokine production tend to be greater in the younger group of birds. It should be noted that in the original experiment, all inoculated birds died, or were euthanized in extremis, although the 62-week-old hens had a slightly longer survival time, whereas the 3-week-old chickens succumbed much more rapidly. 5 The very robust innate response in the younger birds, which could be construed as a “cytokine storm,” might have contributed to their early demise. Conversely, the older birds, with a more moderated cytokine response, were able to muster more specific immunity in the form of expanding T and B cells, before succumbing. There are likely other age-related factors that play a role in the overall response to infection.
In summary, IL-1β and IL-6 are prominent cytokines early in infection of chickens with vNDV. In contrast to mammalian systems, TNF-α appears to play only a minor role in the innate immune response. In comparing the 2 age groups, the cytokine response and evidence of viral replication were more robust in the younger chickens. T cell expansion, seen initially in both groups, then decreased to below control levels whereas in the older birds the T cells remained elevated.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211045945 - In situ cytokine gene expression in early stage of virulent Newcastle disease in chickens
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211045945 for In situ cytokine gene expression in early stage of virulent Newcastle disease in chickens by Corrie Brown, Jian Zhang, Mary Pantin-Jackwood, Kiril Dimitrov, Helena Lage Ferreira and David Suarez in Veterinary Pathology
Footnotes
Acknowledgements
We wish to thank Iryna Goraichuk, Tonya Taylor, Tim Olivier, Suzanne DeBlois, and Dawn Williams Copland for their technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by USDA CRIS Project 6040-32000-072.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
