Abstract
The laboratory records from 384 dogs with a diagnosis of either melanoma or melanocytoma were selected for study. Significant negative determinants of patient survival for melanocytic tumors were: 1) metastasis, 2) mitotic index (MI), 3) nuclear atypia, 4) tumor score, 5) increasing size/volume, 6) the presence of deep inflammation, and/or 7) intralesional necrosis. In addition to these attributes, age was a significant determinant for tumors of the skin. For the feet and lips, 8) age and 9) junction activity negatively impacted survival. Mathematic models were constructed based on these significant determinants to predict the postsurgical outcome of melanocytic neoplasia. Melanocytic oral neoplasms comprised 19% (73/384) of the neoplasms; 92% of these were classified as malignant in the biopsy report, but malignant behavior (i.e., metastasis or recurrence) was observed in only 59% of cases. The prognostic model for oral tumors based on nuclear atypia provided the most accurate (89%) prediction of overall behavior. Melanocytic tumors of the feet and lips were also 19% (73/384) of the total population. Seventy-four percent were reported malignant, whereas only 38% actually demonstrated malignant behavior. The prognostic models based on both MI or nuclear atypia had an overall correct behavioral classification of 81%. Melanocytic tumors in the skin comprised 59% (227/384) of study specimens. Although 39% were reported as malignant, only 12% exhibited malignant behavior. A satisfactory predictive model that employed MI could not be constructed, but one using nuclear atypia gave an overall correct classification in 93.3% of the cases.
Information correlating histologic features of canine melanocytic neoplasms with biologic behavior offers diverse perspectives and little consensus. 1– 5, 8, 9, 11, 13, 15, 16– 20, 22– 25 Because of the prevalence and aggressive nature of oral cavity tumors in dogs, the available information is clearly weighted toward those tumors. The consequences attending diagnosis and treatment of melanocytic neoplasia in human medicine have stimulated a large volume of data specifically focused on the accurate prognosis of cutaneous melanocytic neoplasms. Information related to canine melanocytic disease is often supplemented by and modeled on the abundant and accessible human literature. 9, 20, 22 The obvious anatomic and biologic differences that distinguish the human disease from its canine counterpart, however, suggest that attempts to arrogate the human data for veterinary purposes may obscure a real understanding of the canine disease. In the foundational descriptive literature correlating behavior and pathology in melanocytic disease of dogs, 4, 5 the tumors were anatomically classified and prospectively designated as benign or malignant based on subjective anatomic criteria contained in the World Health Organization (WHO) Bulletin. 28 These dogs were then followed to document the correlation between the diagnosis and observed behavior. Over the years, this published information, as well as earlier data, 6– 10, 14, 22 has enjoyed dissemination and validation primarily via textbooks, resulting in “melanoma dogma” that is widely utilized as a generalized prognostic tool by diagnostic pathologists and as teaching material in the training of pathology residents.
In this study, we have retrospectively followed a series of cases to document the behavior of melanocytic neoplasms. With this information at hand, the histologic characteristics of each neoplasm were evaluated for morphologic features, in aggregate, that might predict benign and malignant behavior of melanocytic disease in dogs. The purpose of this study was to examine histologic characteristics of selected melanocytic neoplasms in 384 dogs in which ultimate outcomes were known and to: 1) expand the available data related to prevalence, metastasis, and survival of canine melanocytic neoplasia; 2) identify morphologic characteristics of melanocytic neoplasia that might distinguish malignant from benign behavior; and 3) compare the prognosis reported to the referring veterinarian for each melanocytic neoplasm with the actual outcome.
Materials and Methods
The canine accessions considered in this study were retrospectively selected from surgical cases originally examined by one or more members of a group of eight board-certified veterinary pathologists who were working in one location and who had extensive interaction. Cases were selected from the laboratory database when the computerized diagnosis field contained either of the words melanoma or melanocytoma. Eight hundred accessions were selected for follow-up. Approximately 100 cases per month from September 1993 through December 1993 were followed up in the same months of 1997, producing 400 cases with a follow-up interval of 4 years. Approximately 100 cases per month from January 1996 through April 1996 were followed up in the same month of 1998, producing 400 cases with a follow-up interval of 2 years.
To obtain follow-up information, we sent a single-page questionnaire to the veterinarians who were responsible for the submission of the 800 cases. The questionnaire included: 1) verification of the dog's age, sex, and breed taken from the original submission; 2) the date of death/euthanasia and reason (if known) and whether death was a direct result of the neoplasm under inquiry; 3) the anatomic site from which the specimen was obtained and the size and/or diameter of the tumor (in those cases, where possible, the diameter was verified or corrected based on measurements of the histologic sections); 4) whether the tumor recurred locally or metastasized and the time interval between initial treatment and recurrence or metastasis (either local recurrence or distant metastases were considered indicators of malignancy); and 5) whether additional tissue (related to this case) had been submitted and any narrative comments that might clarify the outcome of the case.
Almost all cases were treated by surgical excision of the tumor, with submission of tissue for pathologic evaluation. A few of the animals ( <10) whose tissues were submitted from specialty practices received adjunctive therapies of varying type and duration following surgery that, for the purposes of this article, were considered inconsequential.
Cases were excluded from the study when completed follow-up questionnaires contained conflicting information (e.g., when it was indicated that the dog was alive at the time of follow-up but a date of death was given, when the date of death was reported to have occurred prior to the submission of the specimen in question, after the actual receipt of the questionnaire). If a date of death were unknown, the case was similarly excluded from consideration. In some instances, a “last-seen” date was used as a survival period for a patient that was designated “alive at follow-up” when that date was at least 12 months following biopsy submission.
Because there were numerous examples of poorly differentiated tumors presumed by the reporting pathologists to be melanocytic (especially in the oral cavity), the cases of melanocytic neoplasms considered by this article were restricted to those that the majority of veterinary pathologists could agree were melanocytic in origin. With few exceptions (11/384 dogs in which the file slide was missing), the histologic slides from each case were reviewed (W. L. Spangler), and those cases that did not conform to the following histologic criteria were excluded: 1) at least minimal melanin pigment was required in cells of the neoplasm considered to represent melanocytes; 2) a population of “melanocytic cells” identified and differentiated from other present cell types; 3) neoplastic cellular proliferation that tended to occur in the form of “packets” was positively considered a melanocytic trait; 4) neoplasms arising from or resulting in a proliferative disruption of the basal epidermal cells (junctional activity) and/or intraepidermal proliferation of melanocytes was considered characteristic for melanocytic neoplasia; 5) neoplasms were excluded when it was felt by the reviewer that the tissue had originally been misdiagnosed or would, by current convention, not be reasonably considered a neoplasm of melanocytic origin; 6) when melanocytic tumors were simultaneously submitted from more than one location on the body, none was considered except when it occurred in close proximity and possessed similar histologic properties; and 7) when the only submitted surgical specimen was obviously a metastatic lesion (e.g., present in a lymph node), it was excluded from analysis.
Based on these criteria, 405 of approximately 800 follow-up cases initially qualified for study inclusion. These 405 cases were arbitrarily grouped by anatomic location into the following categories: 1) Oral; 2) Lips (involving mucosal tissues); 3) Lips (involving cutaneous tissues); 4) Feet; 5) Head; 6) Body (torso, neck, limbs, back); 7) Eyelids; and 8) Eye/Globe. Eleven of these cases were ocular tumors (five intraocular, six scleral/limbal). These were arbitrarily excluded from further consideration because the numbers were too small to accurately represent the full range of canine ocular melanocytic tumors and would not contribute useful information to extant literature regarding these neoplasms. 25
As the study progressed, 10 more dogs were ultimately excluded for various reasons (as previously described). There were 384 dogs and 389 neoplasms considered in this study. Five of the dogs each had two melanocytic neoplasms located in relative close proximity and of similar histologic appearance, and these five dogs were alive at the end of follow-up. The dogs in the existing seven anatomic-location groups were ultimately consolidated into three larger groups (i.e., oral, feet and mucosal surface of lips, and cutaneous) based on statistic analysis (chi-square) of the mortality/cause of death. The status of each dog at follow-up was categorized as alive at follow-up, dead (euthanatized or naturally occurring) from causes related to the neoplasm, or dead (euthanatized or naturally occurring) from causes not related to the neoplasm (competing causes).
To evaluate potential malignancy of melanocytic tumors, each case was microscopically examined to determine: 1) mitotic index (MI), 2) degree of nuclear atypia, 3) nuclear diameter (10 randomly selected nuclei from the 10 high-power fields) and the qualitative presence/absence of 4) intralesional inflammation, 5) intraepithelial melanocyte proliferation, 6) junctional activity, 7) the presence of giant cells, 8) the presence of intralesional necrosis, and 9) the cell type(s). All these criteria were analyzed against the actual outcome of each case (alive or dead) based on the questionnaire survey. The cell type in each tumor was classified as: 1) mixed, 2) epithelioid, 3) spindle, 4) signet ring, 5) balloon, or 6) dendritic.
MI was the number of mitotic figures counted in 10 consecutive, nonoverlapping high-power fields (500 µm in diameter). Mitotic activity varied within specimens from one location to another, so the entire specimen was scanned to find mitotic figures and the counts were commenced from the point of highest mitotic density. Nuclear atypia, indicating the relative degree of cellular maturation, was scored only in epithelioid cells and was determined as follows. Well-differentiated or typical melanocytic tumor cells were characterized by a small nucleus with a single, centrally oriented nucleolus and minimal clumping of chromatin material. Often, condensed strands of nuclear chromatin extended from the nucleolus to the nuclear membrane, with condensation of chromatin along the inner surface of the membrane. In sections of cells lacking a nucleolus, the chromatin was fine and evenly dispersed at the periphery of the nucleus (Figs. 1, 2). More undifferentiated neoplastic cells (atypical) were characterized by larger nucleoli of less-regular shape. They were eccentrically located in the nucleus and often multiple. In some cases, multiple nucleoli haphazardly connected to the inner surface of the nuclear membrane by thin strands of chromatin and gave the appearance of a coarsely vacuolated nucleus (Figs. 3, 4). Nuclear atypia was evaluated on an incremental scale from 1 to 10, signifying the subjectively estimated percentage of nuclei involved (i.e., 0 = no nuclear atypia, 1 = 1–9% involved nuclei, 2 = 10–19% involved nuclei, 3 = 20–29% involved nuclei, etc.). Described nuclear atypia criteria could not be easily defined among those neoplasms predominantly composed of spindle, dendritic, or signet ring cells, and those tumors were excluded from the scoring system.
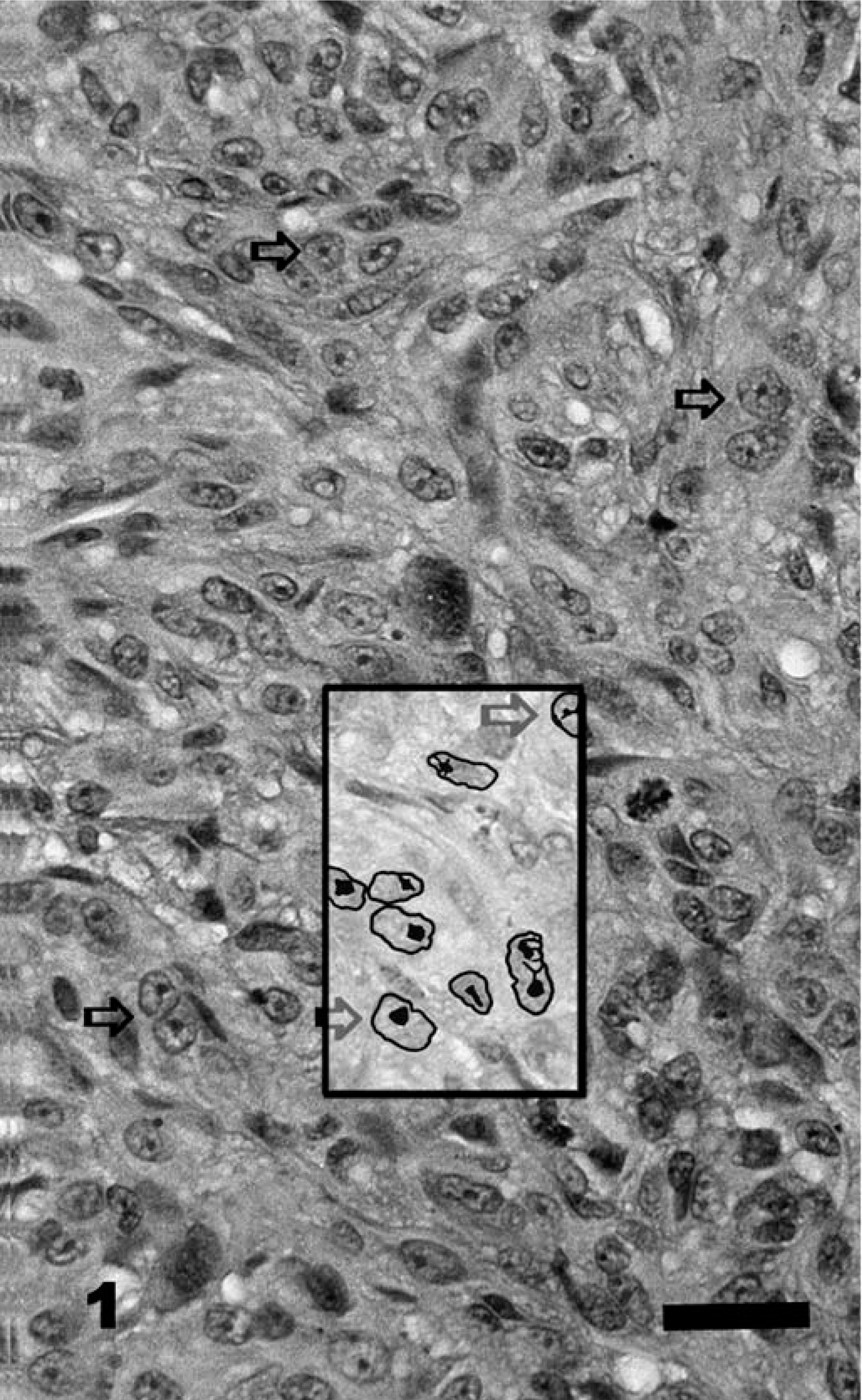
Cutaneous melanocytic neoplasm (low-grade malignancy), left tarsus; dog No. 267. MI = 15, nuclear atypia = 1, tumor score = 17. Alive at the end of follow-up (1,473 days). The preponderance (90%) of nuclei in this section are typical melanocytic with a single, centrally oriented nucleolus (horizontal arrows). There is little variation in size and shape of individual nuclei. The inset represents a pen-and-ink enhancement of nuclear detail. HE. Bar = 100 µm.
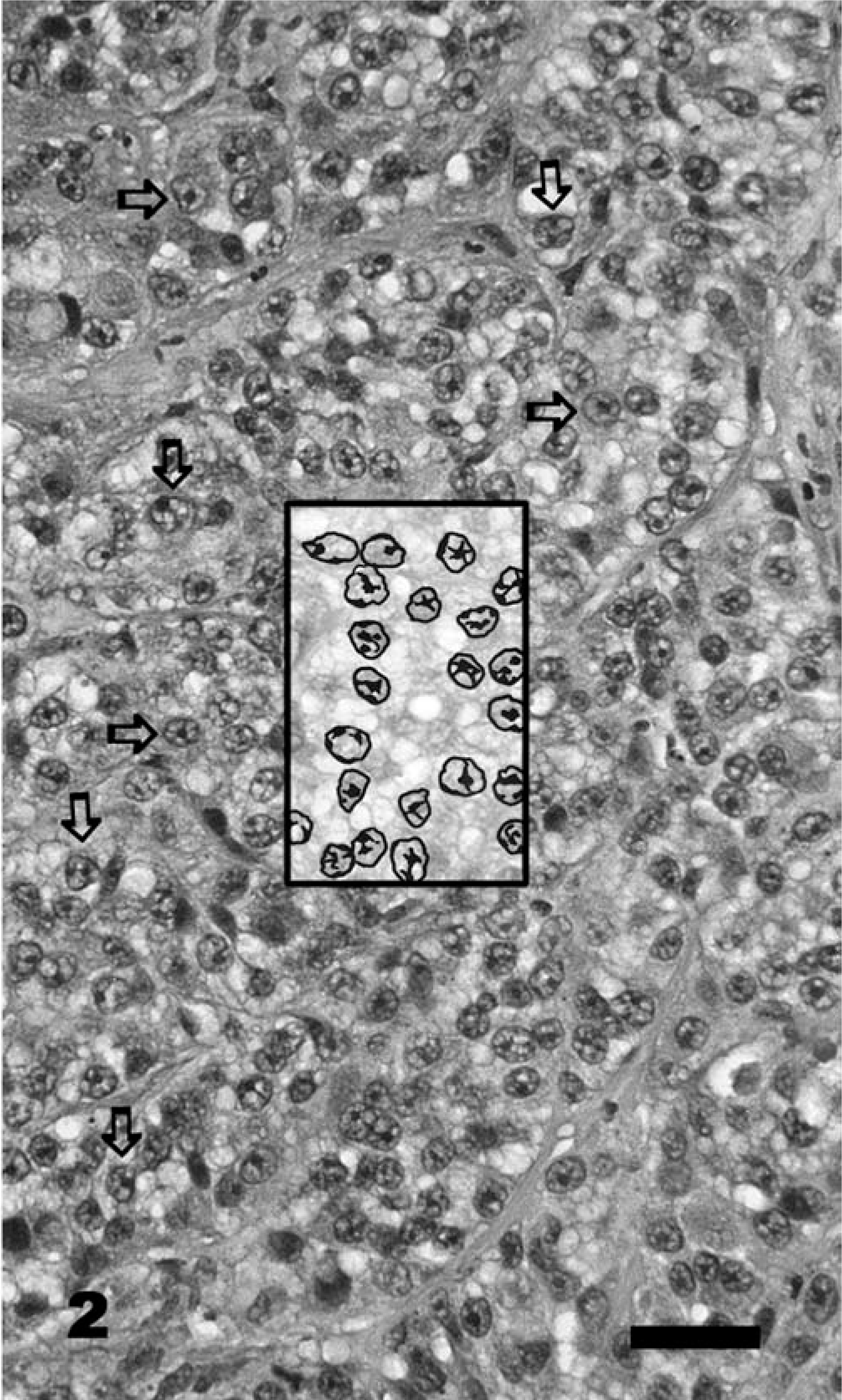
Cutaneous melanocytoma, lip; dog No. 368. MI = 2, nuclear atypia = 2, tumor score = 7. Alive at 733 days after surgery. The preponderance of nuclei are typical of melanocytic cells (horizontal arrows), but some show atypical eccentric nucleoli, multiple nucleoli, and mild anisokaryosis (vertical arrows). The inset represents a pen-and-ink enhancement of nuclear detail. HE. Bar = 100 µm.
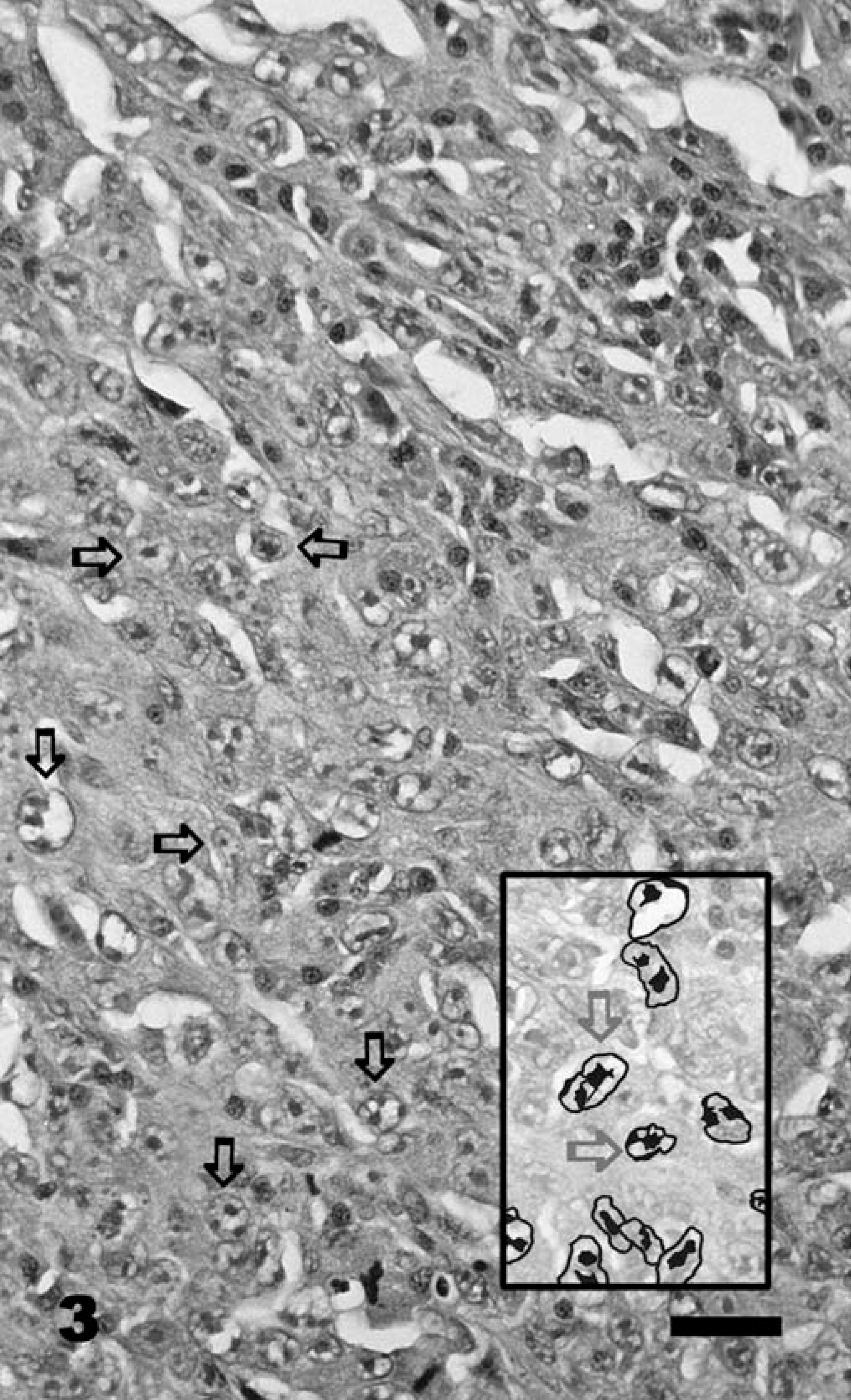
Mucosal lip melanoma; dog No. 325. MI = 23, nuclear atypia = 9, tumor score = 34. Alive at 735 days postoperative recurrence or metastasis at 23 months postoperative. Typical melanocytic cells are indicated by horizontal arrows. The inset represents a pen-and-ink enhancement of nuclear detail. HE. Bar = 100 µm.
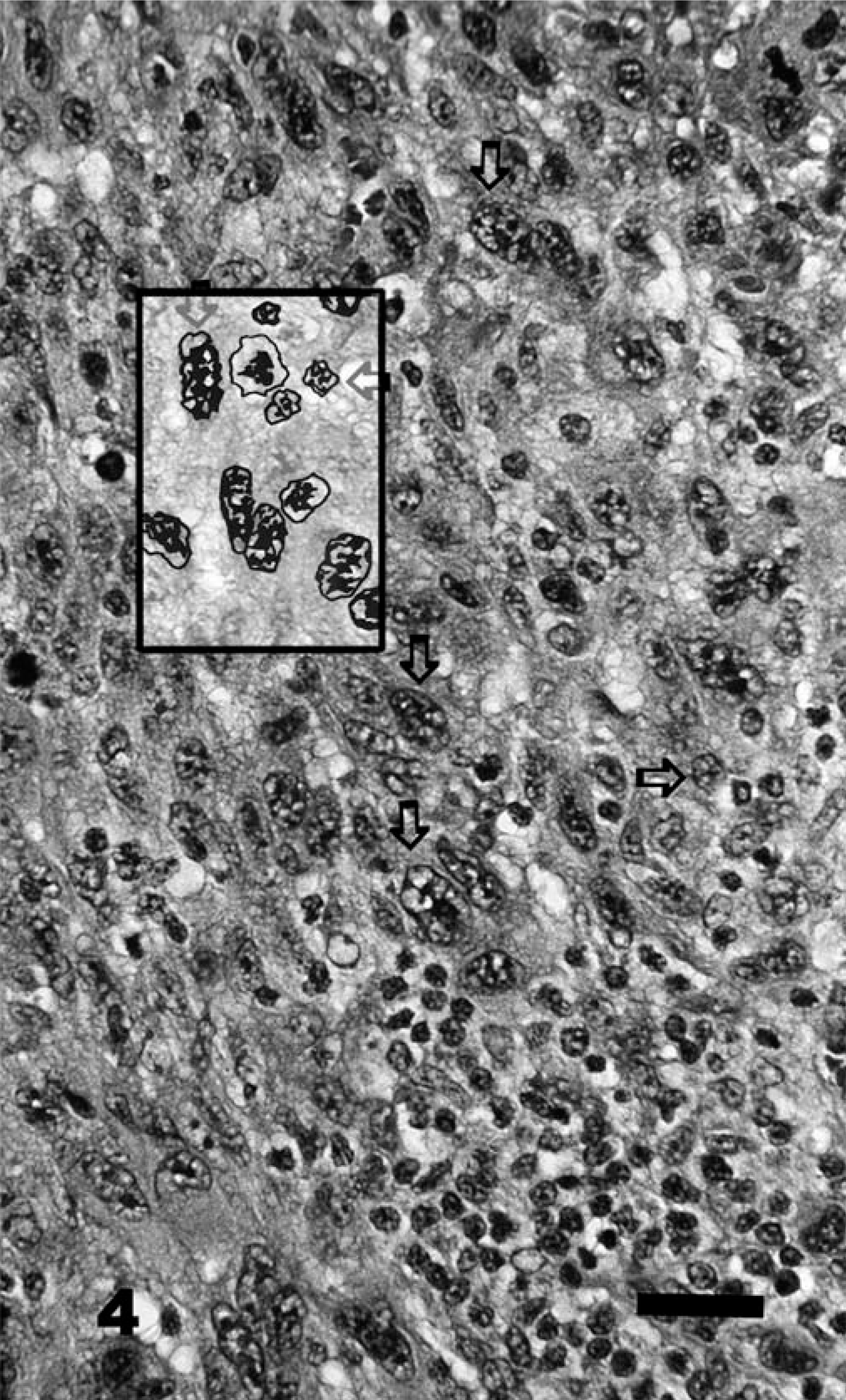
Oral melanoma; dog No. 23. MI = 72, nuclear atypia = 9, tumor score = 84. Dead of related causes 101 days after surgery. The preponderance of nuclei ( >90%) are atypical with multiple nucleoli, prominent anisokaryosis, and pleomorphism (vertical arrows), with only occasional well-differentiated (typical) nuclei that have a single, centrally located nucleolus indicated by horizontal arrows. Focal, deep lymphoid inflammation (lower right) is present. The inset represents a pen-and-ink enhancement of nuclear detail. HE. Bar = 100 µm.
Tumor attributes that did not contribute to mortality (i.e., cell types, nuclear diameter, intraepithelial melanocyte proliferation, junctional activity, giant cells) were excluded from the determination of “tumor score.” Each of the tumor attributes found to significantly and positively affect mortality (i.e., MI, nuclear atypia, deep inflammation, intralesional necrosis of tumor cells, tumor size/volume) were combined to form a tumor-scoring system. The “tumor score” was calculated by adding the numeric values obtained or assigned for each individual attribute; for example, [(numerical value of MI) + (numerical value of nuclear atypia) + (deep inflammation {+1 when present, 0 when absent}) + (intralesional necrosis of neoplastic cells {+1 when present, 0 when absent}) + (size/volume factor { <0.5 cm = 0, 0.5–1.0 cm = +1, >1.0 cm = +2})] = “tumor score.”
Median survival was calculated for each of the three tumor groups using the product-limit method of survival function estimation. The influence of age, sex, and anatomic tumor characteristics on survival was evaluated using a proportional-hazards regression model. Continuous variables were fit using fractional polynomials to obtain the best concordance between the model and data. The assumption of constancy of the proportional effect over time was assessed using likelihood ratio tests of improvement in model fit by adding an interaction between each factor in the model and log time. Results are presented as hazard rate ratios and 95% confidence intervals.
Dogs were treated as censored observations if they were either lost to follow-up or died from competing causes (i.e., for reasons unrelated to melanoma-induced illness). Dogs that either died from the tumor or were followed up for a minimum of 1 year were included in a logistic-regression analysis to develop a model to predict patient status (alive versus dead) after 1 year. Cut points for continuous variables were established to maximize the overall correct classification while ensuring that a minimum of 75% of both alive and dead dogs were correctly classified. Findings are presented as model sensitivity, specificity, positive predictive value (the proportion of dogs predicted to die and those that actually die in the first year following diagnosis), and negative predictive value (the proportion of dogs predicted to live at least 1 year following diagnosis and those that actually do). In the material reviewed for this study, it was generally not feasible to definitively evaluate the surgical margins; thus, incomplete excision of benign melanocytic neoplasms may result in reported recurrence of the tumor and, therefore, functional classification of malignancy.
Results
The mortality characteristics associated with tumors in each of the eight topologically oriented tumor groups of this study were compared in a chi-square analysis to detect differences among groups. Based on this analysis, three distinct mortality groups emerged from the eight originally used: 1) neoplasms that originated in the oral cavity and included the gingiva, tongue, palate, and pharynx, which were consistently the most lethal; 2) neoplasms arising from either the feet (distal to the carpal or hock joints) or mucosal (buccal) surfaces of the lips, which were similar in their mortality characteristics but not as lethal as those in the palate or gingival; and 3) cutaneous melanocytic tumors of all other body sites (i.e., torso, eyelids, ears, face, legs, back, neck, perineum), which had the lowest mortality rates.
Table 1 gives the cell-type distribution of melanocytic neoplasia in the various body locations. Epithelioid cells were the most common type in all locations, whereas a mixture of cell types (consisting primarily of epithelioid and spindle types) was seen with less frequency. Melanocytic tumors entirely comprised of signet ring cells, spindle cells, balloon cells, or dendritic cell types were even less frequently seen. Table 2 provides the distribution of oral melanocytic tumors in this study compared with the site predilections reported in previous articles.
Predominant cell type in canine melanocytic tumors.
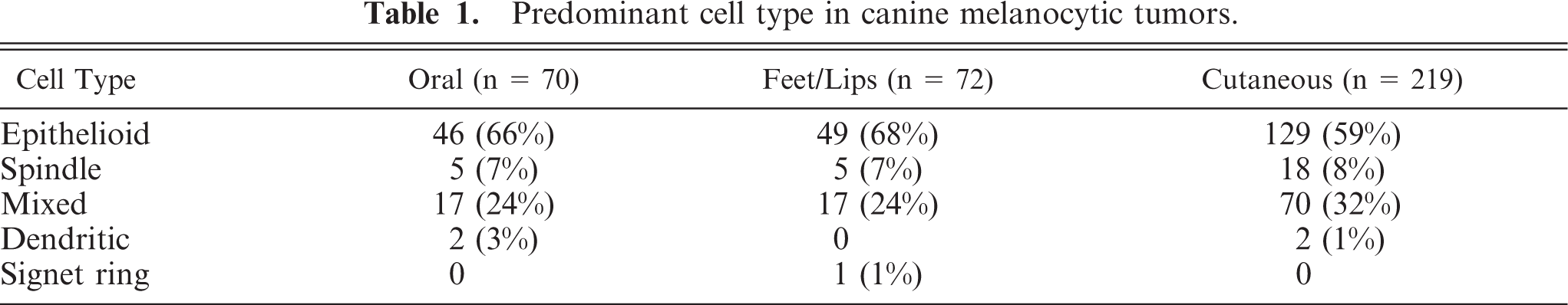
∗ Some cases included in this table were excluded from analysis in other areas because of missing or incomplete data.
Tables 3, 4, and 5 provide an evaluation of the anatomic variables used as potential determinants of survival for melanocytic neoplasia occurring in the oral cavity, feet, lips (mucosal), and skin, respectively. MI (P ≤ 0.001), nuclear atypia (P ≤ 0.001), tumor score (P ≤ 0.001), and the presence of metastasis or recurrence (P ≤ 0.001) provided a strong indication of eventual outcome of the disease for melanocytic tumors in general. Deep inflammations, increasing size/volume of the tumor, and intralesional necrosis varied in P value among the different sites but were significant in all cases. In addition, increasing age and the presence of junctional activity were significant in the case of melanocytic tumors of the feet and mucosal surfaces of the lips. Similarly, age was a significant determinant of mortality in melanocytic tumors of the skin. Sex, intraepithelial proliferation, and the presence of neoplastic giant cells were not significantly related to mortality in any of the three topologic categories. The less subjective but more labor-intensive measurement of nuclear size (diameter) was determined only for the oral neoplasms and was dropped from consideration because it was a cumbersome and time-consuming procedure not significantly related to mortality in that group of tumor-bearing dogs.
Determinants of survival for oral melanocytic neoplasms in dogs.∗
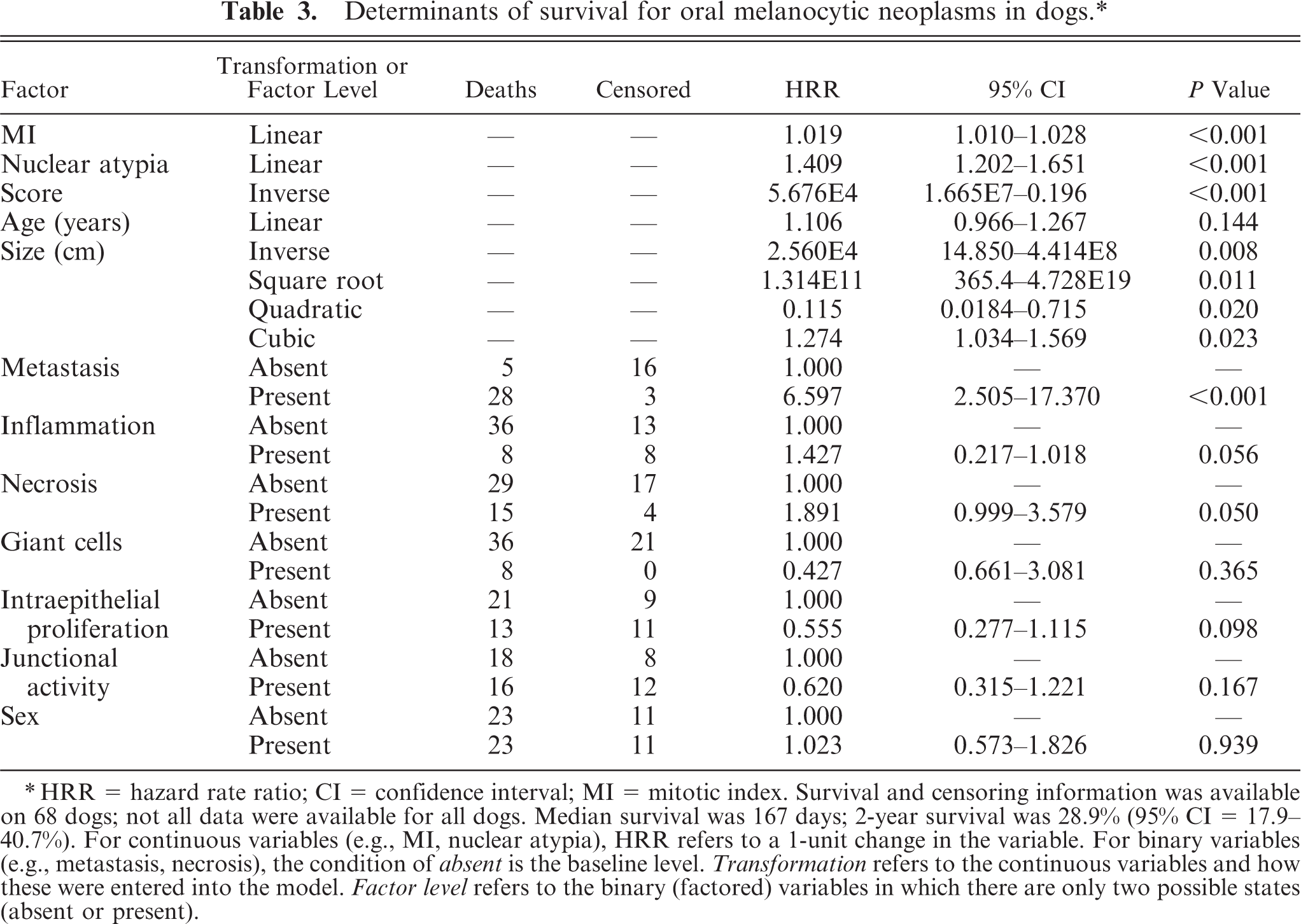
∗ HRR = hazard rate ratio; CI = confidence interval; MI = mitotic index. Survival and censoring information was available on 68 dogs; not all data were available for all dogs. Median survival was 167 days; 2-year survival was 28.9% (95% CI = 17.9–40.7%). For continuous variables (e.g., MI, nuclear atypia), HRR refers to a 1-unit change in the variable. For binary variables (e.g., metastasis, necrosis), the condition of absent is the baseline level. Transformation refers to the continuous variables and how these were entered into the model. Factor level refers to the binary (factored) variables in which there are only two possible states (absent or present).
Determinants of survival for feet or lip melanocytic neoplasms in dogs.∗
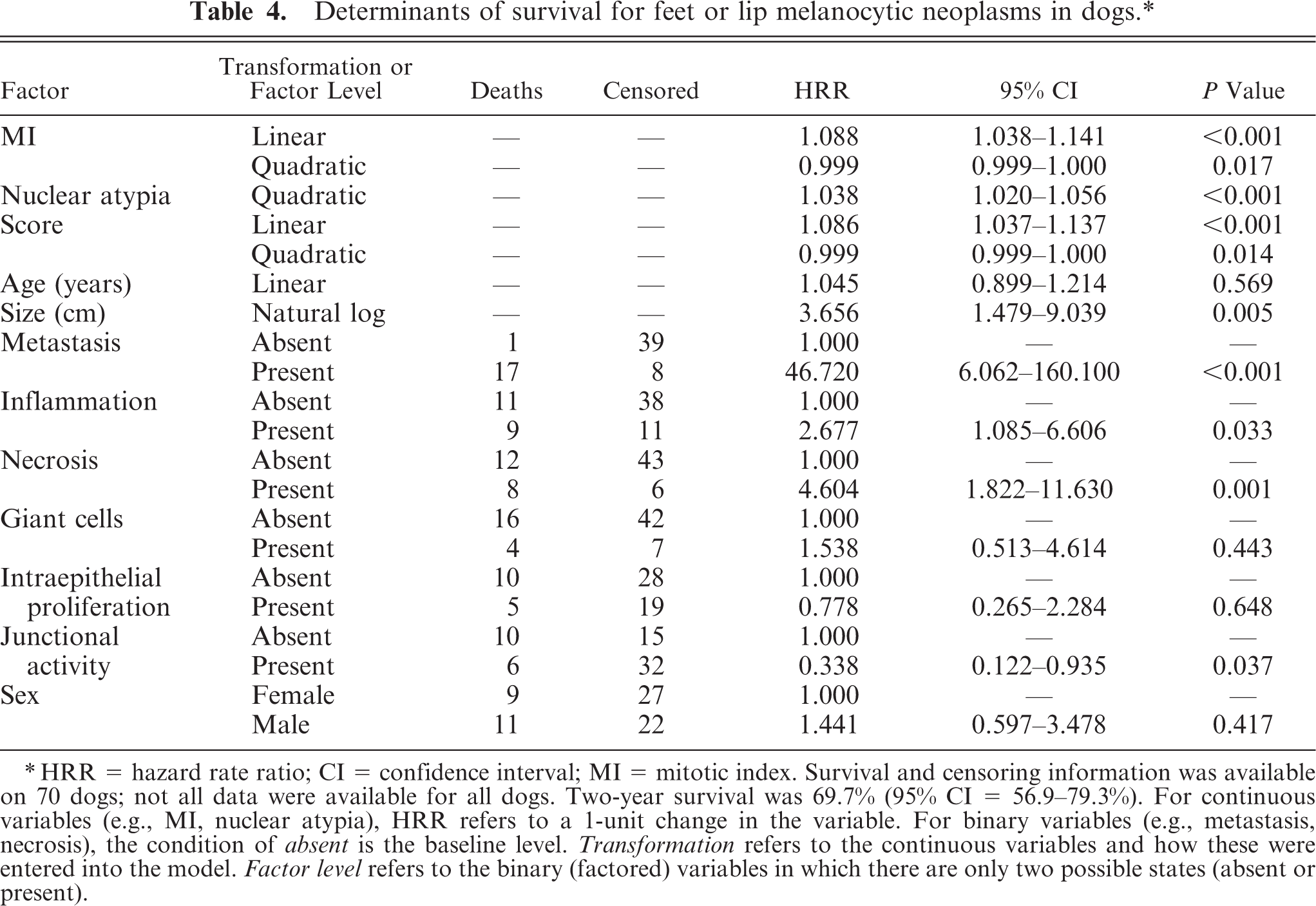
∗HRR = hazard rate ratio; CI = confidence interval; MI = mitotic index. Survival and censoring information was available on 70 dogs; not all data were available for all dogs. Two-year survival was 69.7% (95% CI = 56.9–79.3%). For continuous variables (e.g., MI, nuclear atypia), HRR refers to a 1-unit change in the variable. For binary variables (e.g., metastasis, necrosis), the condition of absent is the baseline level. Transformation refers to the continuous variables and how these were entered into the model. Factor level refers to the binary (factored) variables in which there are only two possible states (absent or present).
Determinants of survival for cutaneous melanocytic neoplasms in dogs.∗
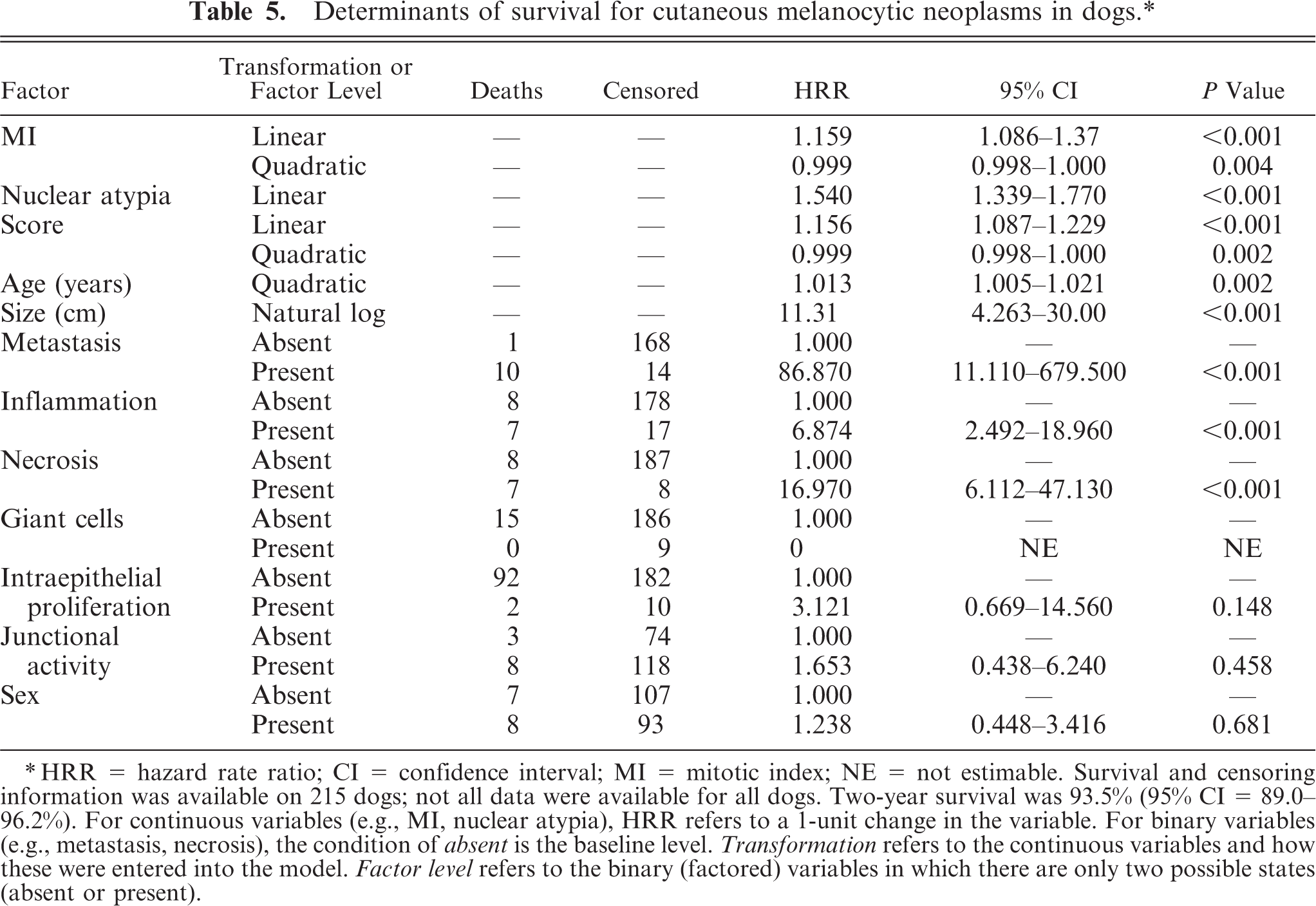
∗ HRR = hazard rate ratio; CI = confidence interval; MI = mitotic index; NE = not estimable. Survival and censoring information was available on 215 dogs; not all data were available for all dogs. Two-year survival was 93.5% (95% CI = 89.0–96.2%). For continuous variables (e.g., MI, nuclear atypia), HRR refers to a 1-unit change in the variable. For binary variables (e.g., metastasis, necrosis), the condition of absent is the baseline level. Transformation refers to the continuous variables and how these were entered into the model. Factor level refers to the binary (factored) variables in which there are only two possible states (absent or present).
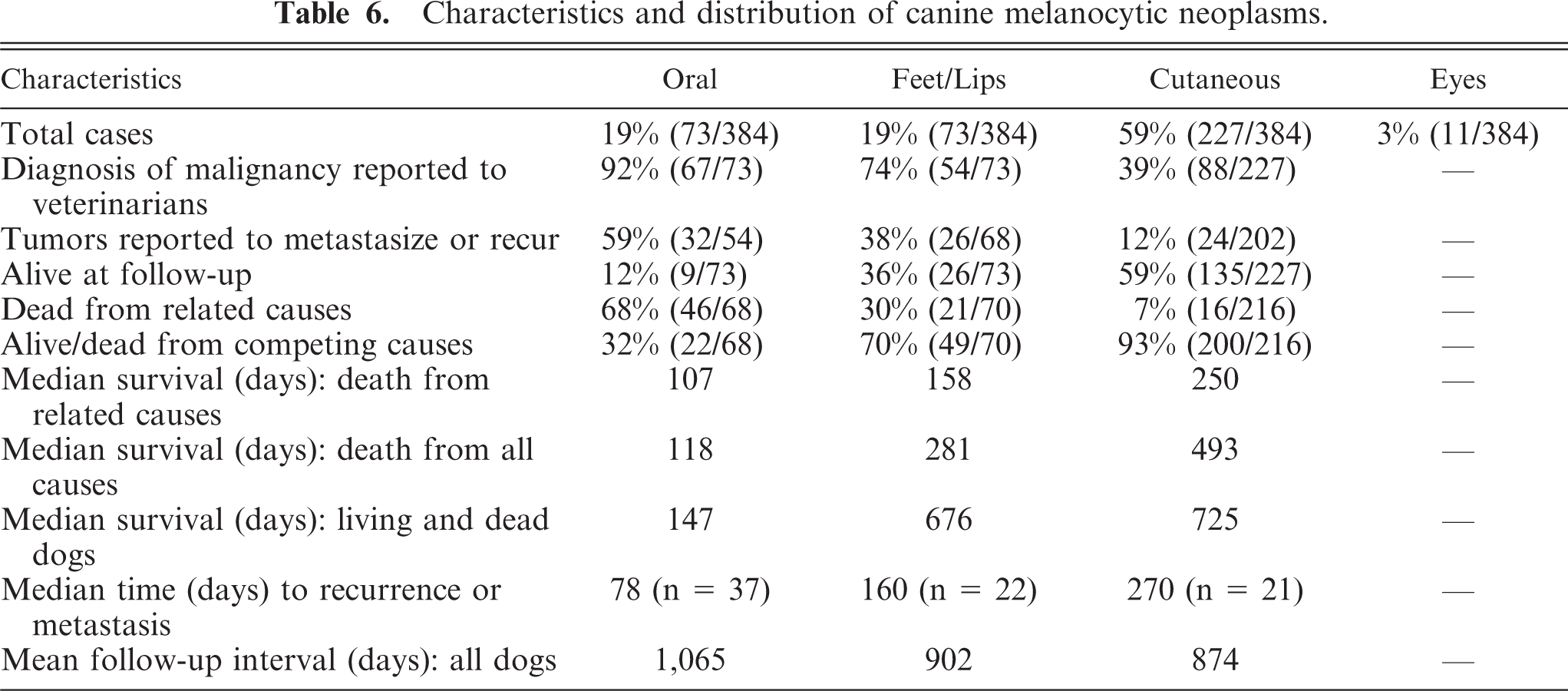
Different mathematic models were developed for melanocytic neoplasms occurring in the three topologic categories, minimizing prognostic misclassification while constraining the sensitivity and specificity of the model to be at least 75%. Table 6 gives the general characteristics and distribution of 384 canine melanocytic neoplasms that met the criteria for study consideration. Nineteen percent of all melanocytic tumors occurred in the mouth/oral cavity, 19% fell into the group derived from the feet or mucosal surface of the lips, and 59% were found in the skin at a variety of specific sites. The remaining 3% involved the eye.
Characteristics and distribution of canine melanocytic neoplasms.
Oral melanocytic neoplasia
The neoplasms from this group were disqualified from the study at the highest rate because of the proportion of poorly differentiated (anaplastic) tumors that lacked the specific criteria used in this study for the identification of melanocytic tumors. Dogs with oral melanocytic tumors also had the highest mortality rate (68%), resulting in the shortest median survival time (147 days) and median time to recurrence or metastasis (78 days) when compared with the other two groups (Table 6). These results are compatible with those observed in other studies. 4, 17, 24
The 68% mortality experienced from oral melanocytic neoplasia agrees well with the 59% of oral tumors that were actually reported to metastasize or recur. However, the degree of malignancy reported to submitting veterinarians (92%) was disproportionate when compared with the 59% of tumors that were reported to manifest malignant behavior (Table 6). Interestingly, 32% of dogs (with oral melanocytic tumors) were either alive at follow-up or had died of competing causes, and 12% remained alive to the end of the 2- to 4-year follow-up period (mean follow-up period = 1,065 days). Although 12% survival correlates well with the reported diagnosis of malignancy in 92% of cases (Table 6), these numbers also suggest a subset (8%) of oral tumors that were easily distinguished as benign or inconsequential neoplasms. If the percentage of tumors that resulted in related deaths (68%) or those reported to recur or metastasize (59%) were subtracted from the percentage originally reported to veterinarians as malignancies (92%), the differences argue in favor of a method to anatomically distinguish the 24% or 33% of melanocytic neoplasms for which the consequences were more favorable.
When the scoring method was applied to dogs with oral tumors, among 9/73 dogs alive at the end of follow-up, 33% (3/9) had tumors graded ≥10 and 67% (6/9) had tumors graded <10. Only 1 (2%) of the 46 dogs that died of causes related to the melanocytic tumors during the study period had a tumor grade <10. Thirty-two of the 36 dogs (89%) with documented metastatic or recurrent neoplasms had tumors that were graded >10. In the construction of predictive models for oral melanocytic neoplasia, 60 dogs were available with qualifying 1-year follow-up data. The overall correct classification of malignancy and subsequent death in this group of dogs was 89% (Table 7). The best prognostic model was based on nuclear atypia scores of ≥5. The models constructed using tumor score, based on the other individual indicators of a poor clinical outcome (Tables 3–5), could not remain within the 75% constraint established for sensitivity and specificity of the model.
Characteristics of prognostic models for oral melanocytic neoplasms.∗

∗ MI = mitotic index.
Melanocytic neoplasia of the tongue appears to represent another exception to the oral melanoma dogma; 2 unfortunately, the information is based on relatively few dogs. In one report, 43% (3/7) of the dogs died of competing causes, and in 57% (4/7) the local disease responded favorably to treatment. The mean survival of all dogs with melanocytic disease associated with the tongue was >1 year (459 days). 2 The dogs with melanocytic tumors of the tongue in the current study presented similar results. In eight dogs with oral melanocytic tumors (8/73), the neoplasm was in the tongue (11%). Of the eight dogs, three (38%) were alive at the end of the study or died of competing causes. The grading system used in this study predicted 2/3 of these results. On the other hand, 62% (5/8) of the dogs died of related causes, all of the five had neoplasms of high grade ( >10), and 3/5 were Chow Chows. The problems inherent in citing these data are the relative paucity of dogs in the groups and the likelihood that the numbers do not represent the overall population of dogs with lingual melanocytic neoplasia.
Melanocytic neoplasms of the feet and lips (mucosal)
In this group of dogs, 32 had melanocytic tumors involving the feet and 41 had neoplasms that involved the mucosal surfaces of the lip. Among the tumors of the feet, 2/32 were taken from the cutaneous tissue of the toe (or between the toes), 7/32 from the footpads, 14/32 from the skin of metacarpus or metatarsus, and 9/32 from the nail bed. One of the tumors removed from the skin of the toe was a small, circumscribed, and completely excised melanocytoma. This dog was alive at the end of the follow-up period. The other neoplasm was a large, ulcerated melanoma that recurred or metastasized and eventually resulted in the death of the dog.
Melanocytic tumors of the foot pads (7/32) showed a range of behaviors. Three of these tumors showed evidence of malignancy, but only one resulted in the death of the patient, one dog was alive at the end of the study period, and five others had died of competing causes. The cutaneous neoplasms of the metatarsal/metacarpal region were generally benign in behavior; one recurred or metastasized but the dog died from competing causes, as did another dog in that subgroup. Ten of these dogs were alive at the end of the follow-up, and the cause of death in two dogs was unknown. The death of nine dogs with nail bed tumors was split between those related to the neoplasms (4/9) and competing causes (5/9), but the outcomes of these cases were not necessarily reflected by the tumor scores. In dogs that died of competing causes, 2/5 had scores <10, and the remaining three scores ranged from 32 to 78. All of the tumor scores in the four dogs that died from related causes ranged from 32 to 74.
Melanocytic tumors of the combined location of feet and mucosal surfaces of the lips comprised 19% of all melanocytic neoplasms, the same as oral tumors, and possessed intermediate mortality characteristics. Intermediate mortality is reflected in the reduced number of “malignant” tumors in this group (74%) reported to referring veterinarians (Table 6). This number, however, substantially overstates the actual percentage of neoplasms that were reported to recur or metastasize (38%) and “related” deaths that occurred during the follow-up period (30%). Eighty-one percent (21/26) of the dogs alive at the end of the follow-up interval had scores <10, and only 4% (1/26) of those dogs experienced serious consequences. Among dogs with “related” deaths, 90% (19/21) had tumors scored >10. Follow-up data for 1 year, including MI, were available for 61 dogs. Three predictive models in which specificity and sensitivity were simultaneously constrained to exceed 75% are illustrated in Table 8. Regardless of which of the three models is used, a model value in the malignant range would have predicted death within 1 year with 66% accuracy, whereas a “benign” model value would correctly predict survival for a least 1 year in 90% of cases.
Characteristic of prognostic models for melanocytic neoplasms of feet and lips.

∗ MI = mitotic index.
Cutaneous
Skin over the remainder of the body was the site of the majority (59%) of canine melanocytic tumors (Table 6). Fifty-nine percent of the dogs with cutaneous melanocytic tumors were alive at follow-up. Although this agrees closely with the 39% of tumors actually reported as malignancies to the submitting veterinarians, it grossly exceeds the 12% of cutaneous melanocytic tumors that actually expressed malignant behavior (recurrence or metastasis). Only 7% (16/227) of the dogs with cutaneous melanocytic neoplasms died of related causes, further illustrating the substantial disparity between those tumors perceived histologically to represent dangerous neoplasms from those deemed benign. Among the 59% of living dogs at the end of follow-up, 96% (128/134) had tumors with a score <10. Of the remaining 4% (6/134), 1% (2/135) were spindle cell types not amenable to scoring and the remaining 3% (4/135) were tumors with scores >10. Only one dog in that small group suffered serious consequences (local recurrence). Among the dogs with deaths related to skin tumors, 69% (11/16) had scores >10. Only 3% of the living dogs had tumor scores greater than 10, but 27% (4/11) of the dogs that died as a result of the tumors in the skin had a tumor score <10 (which, based on the score, would have been predicted benign). There were no attributes of these tumors, in retrospect, that would allow one to predict malignant behavior. Completeness of excision in these cases may have been a factor in the final outcome.
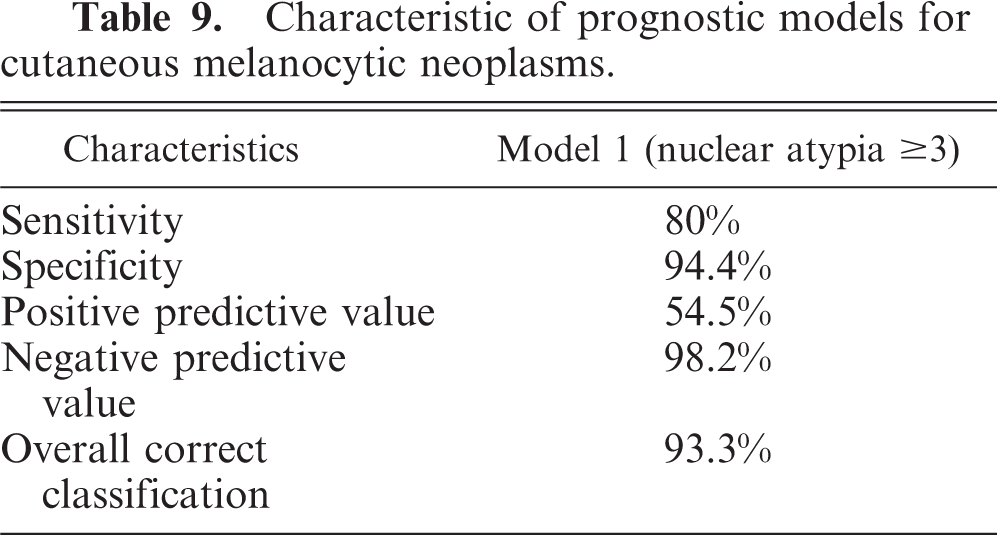
A predictive 1-year follow-up mathematic model for cutaneous melanocytic neoplasia was available on 193 dogs. It was not possible to construct a model using MI that maximized sensitivity or specificity while constraining the other to be no less than 75%. Only one model could be constructed to the previously mentioned constraint (Table 9). By using a nuclear atypia cutoff of ≥3, the model has a sensitivity of 80%, a specificity of 94.4%, a positive predictive value of 54.5% (reflecting that only 15/193 dogs died from the melanocytic tumor within 1 year of diagnosis), and a negative predictive value of 98.2%. The overall correct classification with the model was 93.3%. This model suggests that using the nuclear atypia metric, predicting that a dog with cutaneous melanoma will die from the disease within 1 year of diagnosis, will be wrong nearly as often as correct. On the other hand, predicting survival to at least 1 year will almost always be correct. Attempting to improve this positive predictive value would come at a cost. The model sensitivity would become so low that the majority of dogs that died within 1 year would have been predicted to live to 1 year and beyond.
Characteristic of prognostic models for cutaneous melanocytic neoplasms.
Discussion
The literature dealing with canine “melanoma” teems with conflicting information. In one instance, when tumors were arbitrarily classified as malignant or benign based on histopathologic features, 4, 26 the behavior of oral melanocytic tumors could be effectively predicted, but the clinical outcomes for melanocytic neoplasms of the skin were predicted accurately in <50% of cases. When MI was included as a prognostic guide, the clinical course of cutaneous melanocytic neoplasms was much more accurately predicted, but that technique was not effective with oral tumors. 4, 24 In another report 3 using the same criteria of malignancy, 28 the authors were able to accurately predict the clinical course in almost 90% of those animals. Some authors, 17 have found that tumor size, MI, and tumor location were useful in predicting clinical outcomes, whereas others 1, 18, 23 found similar criteria to bear no statistic relationship to patient survival. Even the terminology for melanocytic neoplasia in dogs, until recently, had not been solidly established. 12, 14 In this article, the convention established by the most recent WHO report 14 has been followed: the term melanoma indicates a melanocytic malignancy, and melanocytoma indicates a benign tumor of melanocytic origin regardless of its topologic location.
The most basic classification of melanocytic tumors is the prevalence of malignancy or mortality of neoplasms from different body sites. 4, 5, 9 Because the current study and others 3, 5, 9, 26 have shown a distinct difference in mortality based on location of the tumor, this subdivision seems appropriate as an initial categorization for this class of neoplasia. Overall, the most predominant cell type found in canine melanocytic neoplasms was epithelioid (Table 1). 27 Melanocytic tumors composed of mixed cell types were next in abundance and most often comprised varying numbers of epithelioid and spindle cells. Although cell type appears to play some role in the prognosis of ocular melanocytic neoplasms, 25 this feature lacked significance in prognoses (i.e., prediction of mortality) of melanocytic tumors occurring in the mouth, feet, buccal mucosa, and skin of dogs in this and other studies. 1, 3, 4, 16– 18, 22, 24
MI, nuclear atypia, and tumor score were all highly correlated (P ≤ 0.001) with malignancy. Other features of melanocytic tumors statistically associated with malignancy were: 1) deep lymphocytic inflammation, 2) intralesional necrosis, 3) size of the tumor (volume), and 4) metastasis/recurrence. Tables 3, 4, and 5 provide the P values obtained for each of the neoplastic features significantly associated with malignancy (defined by death of the dog from causes related to the neoplasm). MI has been found useful to predict malignant behavior in some reports 1, 4, 17 and lacked significance in others. 18, 24 Although it was not possible to predict with 100% assurance the outcome for any specific neoplasm, when MI was used in conjunction with nuclear atypia, it was possible to model a system to provide relative probabilities of experiencing the outcome predicted by the diagnosis. In addition, it has recently been shown that proliferative index 21 and DNA ploidy, 3 as assessed by immunohistochemistry and flow cytometric analysis, respectively, where they are available, can be expected to provide an accurate prediction of clinical outcome. Profiles of genetic changes within tumor cells may eventually complement all the currently available methods to predict malignancy. 22
The statement frequently encountered in the literature that all (or nearly all) melanocytic oral neoplasms should be considered malignant, 12, 13, 15, 18 or that benign oral melanocytic tumors are extremely rare, 8 is misleading. Unfortunately, this concept is so deeply embedded in the canine “melanoma” literature (thus “dogma”) that it impacts clinicians as well as pathologists. The effect on pathologists is the emphasis of potential malignancy of these neoplasms (Table 6), and the effect on clinicians or oncologists is overreaction in some cases where minimal intervention would suffice. Although a high proportion of oral melanocytic neoplasms behave with malignant characteristics, as seen in Table 6, 32% of all the dogs with melanocytic oral neoplasia were either alive at follow-up or had died of causes unrelated to the neoplasm. Similar results have previously been reported in a different context. 3, 11, 17, 24 In the current study, 12% of dogs with neoplasms in the oral cavity were alive at the end of the follow-up period and had not experienced difficulties associated with the neoplasm. Although this fact alone may not be interpreted as an absolute indication of benign behavior, it is the best indicator available. Information in Table 6 indicates that truly benign melanocytic tumors (melanocytomas) are indeed recognized in the oral cavity on a routine basis and have now been reported elsewhere. 11
The scoring method used in this study was highly correlated with deaths actually caused by the tumors and accurately identified those animals likely to die as a result of their disease. However, when modeling the data to obtain the most consistent prognostic information, MI and nuclear atypia were simpler to obtain and gave the best overall result for tumors arising in the mouth, feet, or mucosal surfaces of the lip. The data presented on oral melanocytic tumors in Table 6 illustrates that, with adequate numbers of cases, a well-defined group of oral tumors with benign behavior emerges. This observation is supported by information from other follow-up studies of oral melanocytic neoplasia 18, 24 in which recurrence or metastasis was observed in 71% and 73% of cases, respectively, leaving 29% and 27% of the oral melanocytic tumors with no observed consequences during the follow-up period. Previous studies have been unable to make a strong correlation between MI and malignancy (or death of the animal) with oral melanocytic tumors. 4, 18, 24 In the current study, MI is strongly correlated with death due to the neoplasm (P ≤ 0.001).
The scoring system used in this study for melanocytic tumors of the feet and lips was highly correlated with deaths actually caused by the tumors and was able to identify neoplasms likely to metastasize or recur with about 90% accuracy. The prognostic model using an MI ≥ 5 (Table 8) provided an overall correct classification of 82%. Longer survival times in this group of dogs (Table 6) may, in part, be due to the fact that tumors of the lips and feet are likely treated earlier in their development because they are more readily detected by owners than those in the oral cavity. Also, 10% (7/73) of “foot tumors” were amputated toes in which invasive, poorly differentiated, melanocytic neoplasms were invading and destroying osseous tissue of the digits (nail bed tumors). These tumors are histologically malignant, with high scores, but they can be effectively treated by early amputation and are, therefore, more survivable and carry a better overall prognosis.
Correlation of subjective histologic characteristics and biologic behavior in melanocytic tumors, specifically of the extremities (feet), was previously documented in 28 dogs. 1 In that study, 50% of the neoplasms were histologically designated benign and 50% were histologically designated malignant. Survival times for dogs with malignancies were not different from those with designated benign melanocytic neoplasms. Although there may be a variety of ways to explain these disappointing results, it likely has to do with the effectiveness of amputation when the tumor is peripherally located on an extremity. In this case, a better overall clinical result or lengthened survival time positively skews the survival of those dogs with anatomically designated malignancy.
In this study, 92% of dogs with oral melanocytic tumors were reported to veterinarians with a diagnosis and/or comment indicative of malignancy. This correlates with the fact that 12% of the dogs with oral neoplasms were alive at follow-up (Table 6). However, only 59% of dogs with oral melanocytic tumors were reported to have developed metastasis or recurrence, and only 68% of the dogs actually died from causes related to their oral tumors. Similar proportions are evident (Table 6) for the other anatomic sites (i.e., feet, lips, skin) and suggest that, where melanocytic tumors are concerned, malignancy is consistently overstated by about 30%.
The prognostic record for the histologic evaluation of cutaneous melanocytic neoplasms (i.e., torso, head, neck, proximal extremities) was not so clear cut. Although only 7% of dogs died of related causes and 12% of neoplasms were reported to recur or metastasize, there was a subset of cutaneous melanocytic tumors with malignant behavior that would have been predicted benign using the criteria so far indicated. Because the mortality was so low among cutaneous tumors, the positive predictive value of the model was a virtual coin toss (54.5%), even though the overall correct classification was 93% (Table 9). The median survival times for dogs in the cutaneous category were the longest. Median survival for all dogs with cutaneous melanocytic tumors was 88% of the total follow-up time period in the study, whereas survival for dogs with oral tumors or tumors of the feet and lips was 30% and 67%, respectively (Table 6).
Grave consequences associated with melanocytic neoplasms may be defined as death of the animal caused directly or indirectly by the neoplasm, metastasis, or perhaps even local recurrence. If death is used as an end point and euthanatized animals are included in that end point, it is important to distinguish those dogs euthanized as a medical necessity to avoid unacceptable pain and discomfort from those whose disease inception or progression merely coincides with euthanasia performed as a convenience or economic expediency to the owner. 5 Unfortunately, this is a difficult or perhaps impossible statistic to derive from any source. The results of this study should be interpreted with that in mind.
Necropsy examinations were generally not available for any of the dogs in this study, so the causes of death (i.e., related, competing, or unknown) were obtained from the questionnaires returned by the submitting veterinarians or, in some instances, directly from the dog's owner.
The reason for disparity in sex distribution between earlier reports 6, 8, 16, 23 (male dominance) and those published more recently 3, 24 (equal sex distribution) has no obvious explanation, but is an example of the “melanoma dogma” that has developed around the canine disease. 6, 16, 23 The prevalence of castrated male dogs today, as opposed to several decades ago, may provide some explanation.
The mean age of melanocytic tumor–bearing dogs has consistently increased in the 4 decades covered by the reviewed literature, from something more than 9 years of age to about 12 years of age. 6– 9, 16, 17, 24 This probably reflects favorably on both owners and veterinarians and on more effective management of neoplastic diseases in general. In the correlation of histologic appearance and behavior, a number of variables should be considered. An apparently aggressive neoplasm with a high proliferation index may be identified and completely excised before it has an opportunity to extend beyond the local area, thus exhibiting putative benign behavior. Similarly, the peripheral location (e.g., digit) of such a neoplasm, readily removed, likely enhances the probability of a successful long-term outcome for cancer in general, whereas those originating in the mouth are much more difficult given the traditional and most widely used treatment for neoplasia of this type (surgery). On the other hand, a neoplasm that begins life as a virtually static or slow-growing entity, over an extended time, may take on more aggressive and malignant characteristics and behavior. This variability in time and space necessarily complicates the relationship between histology and observed behavior. Along with the evidence of the current study, more recent reports 11, 24 suggest that, although a serious threat, oral melanocytic neoplasia is not the uniformly fatal disease that it was once reported to be, denying the “dogma” that has developed around this class of tumor. 4, 5, 9, 13, 16, 26
Although it is impractical to accurately predict, on an individual basis, the final outcome of melanocytic neoplasia by applying numeric criteria to certain histologic features (e.g., MI, nuclear atypia), it is possible to maximize the percentage of correctly classified tumors. In human medicine, the documented biology of the neoplasm drives the selection of a treatment regimen that is favorably known to alter the projected outcome of the disease. In veterinary medicine, the decision to recommend an expensive (and sometimes painful) treatment of neoplastic disease, especially in an older dog, is often influenced by a cost–benefit relationship that must be based on a similar knowledge of the projected outcome. An alternate and viable decision in canine medicine may be to simply withhold treatment altogether and recommend euthanasia. When making these decisions, it is essential to have the best available information.
Footnotes
Acknowledgements
We acknowledge the expert editorial assistance of Robert Clark in Greensboro, North Carolina; the photographic and artistic contribution of Jeff Childress in Winters, California; the financial assistance and case material from IDEXX corporation; the IDEXX pathologists in Sacramento, California, who are responsible for the initial evaluation of the cases used in this report; and the veterinarians who submitted the cases to the laboratory and then took the time to complete the questionnaires.

