Abstract
Melanoma diagnosis in dogs can be challenging due to the variety of histological appearances of canine melanocytic neoplasms. Markers of malignancy are needed. Receptor for activated C-kinase 1 (RACK1) was found to characterize melanomas in other mammals. We investigated the value of RACK1 detection in the classification of 19 cutaneous and 5 mucosal melanocytic neoplasms in dogs. These tumors were categorized as melanocytomas or benign and melanomas or malignant after evaluation of their morphology, mitotic index, and Ki-67 growth fraction. Using immunofluorescence, we confirmed microphthalmia-associated transcription factor (MITF) as a marker of normal and transformed melanocytic cells in dog tissues. All control (n = 10) and tumoral (n = 24) samples stained positively for MITF (34/34, 100%). Whereas RACK1 was not detected in healthy skin melanocytes, melanocytic lesions were all positive for RACK1 signal (24/24, 100%). RACK1 cytoplasmic staining appeared with 2 distinct distribution patterns: strong, diffuse, and homogeneous or granular and heterogeneous. All melanoma samples (13/13, 100%) stained homogeneously for RACK1. All melanocytomas (11/11, 100%) stained heterogeneously for RACK1. Immunohistochemistry was less consistent than immunofluorescence for all labelings in melanocytic lesions, which were often very pigmented. Thus, the fluorescent RACK1-MITF labeling pattern helped to distinguish melanomas from melanocytomas. Furthermore, RACK1 labeling correlated with 2 of 11 morphological features linked to malignancy: cell and nuclear size. These results suggest that RACK1 may be used as a marker in dog melanomas.
Canine melanomas account for 3% of dog neoplasms and 7% of malignant tumors. 30 Preferential sites are, in descending order, the oral cavity, lips, skin, and toes. 30 Cellular pleomorphism is the main characteristic of these tumors. 22 Differential diagnosis should first include melanocytomas. 12 Herein, the term melanoma refers to a melanocytic malignancy, and melanocytoma indicates a benign tumor of melanocytic origin regardless of its topologic location, according to the current World Health Organization nomenclature. 10 Setting a definitive diagnosis based on anatomopathological examination alone can still be challenging. Biomarkers of malignancy are needed to confirm diagnosis to adapt therapy to improve prognosis. Early identification of malignant melanocytic lesions is crucial for dog survival since melanomas possess high metastatic capacities. 4
Immunodetection of RACK1 protein could be considered one such marker. Indeed, RACK1 is strongly and homogeneously detected in melanoma cells of primary tumors and metastases developed in human patients, MeLiM minipigs, 7 and horses. 5 This pattern of RACK1 detection is specific for malignant melanocytic tumors. Nonetheless, RACK1 protein is ubiquitously expressed, but its levels are cell type dependent. 1 Functionally, RACK1 is regarded as a signaling hub allowing crosstalk between various pathways contributing to both physiological and tumorigenic cellular functions.1,19,27 The aim of this study was to assess the value of RACK1 detection in canine melanoma diagnosis.
Material and Methods
Dog Tissues
Dog (Canis familiaris) tissues in paraffin blocks came from biopsy specimens at the Alfort Veterinary Medicine School (n = 17) (Maisons-Alfort, France) and at the IDEXX Diagnosis Laboratory (n = 17) (Alfortville, France). Samples consisted of previously diagnosed cutaneous (n = 8) and mucosal melanomas (n = 5), including 2 nonpigmented melanomas; cutaneous melanocytomas (n = 11); and normal skin (n = 5) and lip tissues (n = 5) as controls. Plasmocytomas (n = 2), cutaneous histiocytoma (n = 1), cutaneous nonepitheliotropic T-cell lymphoma (n = 1), peripheral nerve sheath tumor (n = 1), and low-grade fibrosarcoma (n = 1) were included as control cutaneous canine tumors.
Routine diagnosis between melanocytoma and melanoma relied on morphologic criteria such as invasiveness and architectural symmetry of the tumor as well as cellular characteristics: anisocytosis, anisokaryosis, nucleolar prominence, and atypical mitoses. Cases for which differentiation between melanocytomas and melanomas was difficult were excluded. Mitotic index and growth fraction were later assessed as they have been demonstrated to provide a strong indication of eventual outcome of the disease for melanocytic tumors in general.3,31 No information on the history of tumors was available. Dark skin and dark mucous membrane dogs were overrepresented. Mean age was 9.5 years. Ten melanomas of 13 (77%) were localized in the oral cavity or at the lips or toes. In contrast, 8 melanocytomas of 11 (73%) were localized on furry parts of the body (Table 1). Epidemiological analysis of our samples indicated that our cohort was representative of reported dog melanoma.8,12,20,21,31
Epidemiological Data From Dogs With the Melanocytic Neoplasms Examined.
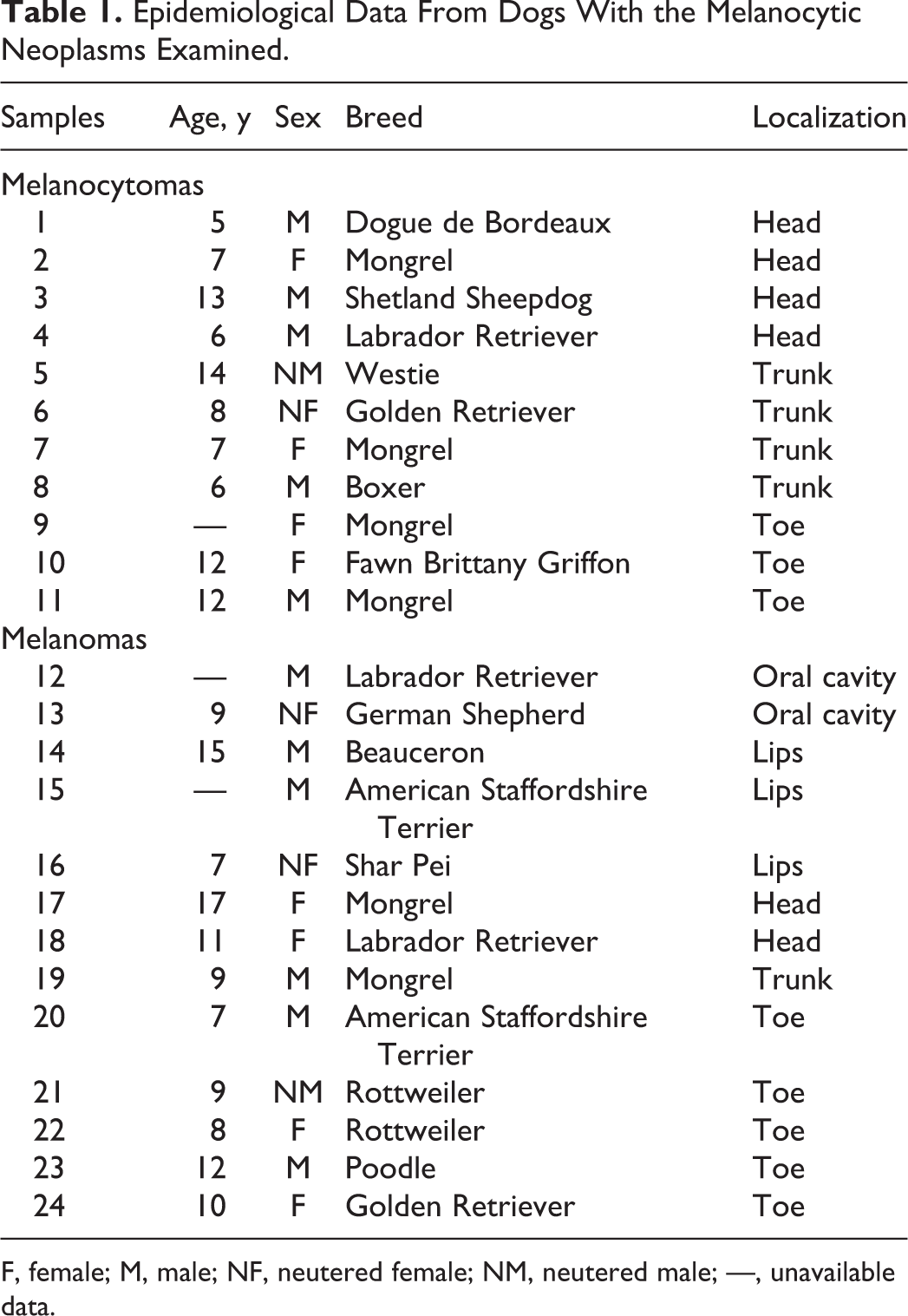
F, female; M, male; NF, neutered female; NM, neutered male; —, unavailable data.
Immunostaining, ApoTome Microscopy Analysis of RACK1 Distribution, and Ki-67 Index
Immunostaining was performed without bleaching the sections. The immunofluorescence protocol for microphthalmia-asso-ciated transcription factor (MITF) and receptor for activated C-kinase 1 (RACK1) was previously described. 5 Briefly, mouse monoclonal antibodies anti-RACK1 (Transduction Laboratories, 1:150; BD Biosciences, Franklin Lakes, NJ) and anti-MITF (Zymed, around 3 μg/ml; Invitrogen, Carlsbad, CA) and rabbit polyclonal antibody anti–cytokeratin 5 (Covance; 1:1000; Eurogentec, Seraing, Belgium) were incubated on 4-μm sections after antigen retrieval in Tris-EDTA (pH 9) for 30 minutes in a water bath at 98°C to 100°C. Primary antibodies were incubated overnight at 4°C. Respective secondary antibodies were Alexa Fluor labeled (1:400; Invitrogen). Alternatively, a biotinylated anti–mouse IgM antibody (Vector Labs; Eurobio Abcys, Courtaboeuf, France) was used for RACK1 and revealed with Alexa Fluor–labeled streptavidin (1:400). 4′,6-Diamidino-2-phenylindole (DAPI) was used for nuclear counterstaining.
Immunofluorescence for Ki-67 was performed on serial sections with mouse monoclonal MIB-1 antibody (Dako-Cytomation, 1:75; Dako, Glostrup, Denmark) after antigen retrieval in citrate buffer (pH 6.0) at 97°C for 20 minutes in a decloaking chamber (Biocare Medical; Eurobio-Abcys).
Sections were examined with a Zeiss Axio Observer Z1 M ApoTome microscope (Carl Zeiss S.A.S., Le Pecq, France) and processed with the AxioVision 4.6 computer program (Carl Zeiss S.A.S.). All images shown are individual sections of z-series stacks. RACK1 staining distribution was analyzed at the tissue and cellular levels. Distribution within the cytoplasm was graded 0 when homogeneous and 1 when heterogeneous. Samples were analyzed blindly and independently by 2 authors (CC, SL). The Ki-67 index was calculated as the percentage of labeled nuclei after counting 500 DAPI-positive nuclei in the areas of heaviest Ki-67 staining, avoiding areas of inflammation and necrosis. A Ki-67 index >15 is correlated with a low survival rate.3,15,29 The immunohistochemistry protocol used the same reagents as immunofluorescence except for the avidin-biotin peroxidase kit (Vector Labs) with a 3,3′-diaminobenzidine-nickel kit (Vector Labs) for MITF, which stains positive cells dark gray, and the 3-amino-9-ethylcarbazol kit (Vector Labs) for RACK1, which stains positive cells red. Sections were then counterstained with Gill’s hematoxylin. The incubation of MITF and RACK1 antibodies was sequential with MITF incubated for 30 minutes and RACK1 overnight or separately. Negative controls without the first antibody showed no background staining.
Analysis of Histological Features
Mitotic index
Four-micron sections were first bleached by immersion in 0.25% potassium permanganate solution for 1 hour followed by 5% oxalic acid for 5 minutes. Then sections were stained with hematoxylin-eosin-saffron (HES) according to standard histological procedures in a Leica ST 5020 multistainer (Leica Microsystems S.A.S., Nanterre, France). The mitotic index (MI) was the number of mitotic figures in 10 high-power fields (HPF), in areas with the highest mitotic activity. For cutaneous melanocytic tumors, an MI >3 is considered to indicate poor prognosis and hence is associated with melanoma diagnosis. 15 The equivalent value for oral melanoma is MI ≥4. 3
Elementary histomorphological features
Complementary histopathologic evaluations were carried out on routinely stained HES examined at 3 different magnifications (10×, 20×, and 40× objectives). Sections were observed with a Leica DMLB microscope (Leica Microsystems S.A.S.). Images were processed with the MetaVue Imaging System computer program (Molecular Devices, Sunnyvale, CA). Lesions were graded according to 11 histomorphometric criteria as previously defined in the classification of human and horse melanocytic tumors.5,33 The criteria analyzed were the ulceration, the upper scatter of intraepidermal melanocytes, the pigmentation of melanocytes, nest formation of intraepidermal melanocytes, the lateral circumscription, the contour of the epidermis, and the size and shape of cells, nuclei, and nucleoli.5,33
Briefly, the upper scatter of intraepidermal melanocytes was the proportion of intraepidermal melanocytes present above the basal layer, graded as absent (essentially all melanocytic cells situated at the dermoepidermal junction), slight (when up to 75% of melanocytes were at the dermoepidermal junction), medium (when melanocytic cells were equally distributed between the basal and upper epidermal layers), or prominent (when >50% of melanocytic cells localized to the upper layers of the epidermis). Lateral circumscription was assessed by examining the transition of the intraepidermal growth portion of the tumor to normal skin at the tumor periphery. The area with the most gradual transition was scored 0 for discontinuous areas of apparently uninvolved epidermis interspersed with tumor, 1 for a gradual but continuous decrease in the number of intraepidermal melanocytes, or 2 for an abrupt transition. The contour of the epidermis involved by the melanoma was compared with the adjacent normal epidermis and scored 0 for atrophic epidermis with effacement of the rete ridges, 1 for thinned epidermis with partial attenuation of the rete ridges, 2 for normal epidermis, 3 for up to 2-fold thickened epidermis by a moderate hyperplasia, or 4 for marked epidermal hyperplasia resulting in a greater than 2-fold increase in epidermal thickness.
Samples were graded blindly and independently by 2 authors (CC, FB) without reference to previous pathology reports. Tumor size and completeness of excision had not been clinically assessed.
Statistical Analysis
Statistical differences between means taken in pairs were evaluated by the Student’s t-test. The test was adapted for a number of samples below 30 as previously described.5 A P value less than .05 was considered statistically significant.
Results
MITF Is a Sensitive Immunofluorescence Marker to Identify Melanocytic Cells in Dogs
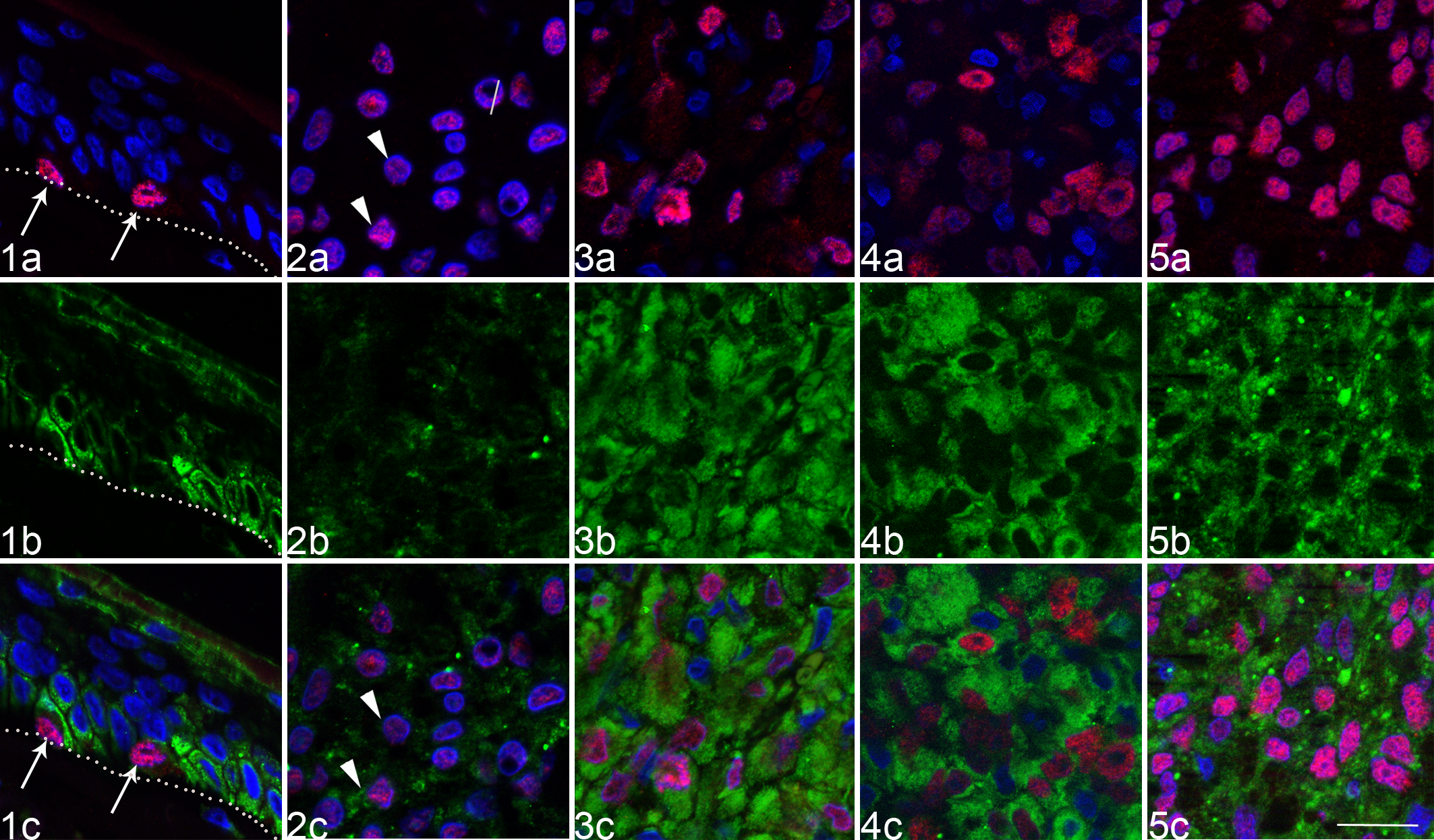
To identify melanocytic cells within tissue sections, we used an MITF in vitro diagnostic monoclonal antibody. 5 MITF-positive cells were identified at the basal layer of healthy skin and mucosa by a moderate to strong immunofluorescent nuclear signal (Fig. 1a). By immunofluorescence, tumoral cells were identified as MITF-positive cells in all melanocytic neoplasms regardless of the pigmentation of the lesion (Figs. 2a–5a). In contrast, by immunohistochemistry with a 30-minute incubation of the primary antibody, tumoral cells were labeled but not in every sample. Canine neoplasms considered in the differential diagnosis of melanocytic neoplasms, such as plasmocytomas, cutaneous histiocytomas, cutaneous nonepitheliotropic T-cell lymphoma, peripheral nerve sheath tumors, or low-grade fibrosarcoma, were all negative for MITF by immunofluorescence (not shown).
RACK1 Protein Distinguishes Melanoma From Melanocytoma in Dogs
In control healthy skins, RACK1 protein was cytoplasmic in keratinocytes (Fig. 1b,c). By contrast, MITF-positive melanocytes were negative for RACK1 (Fig. 1b,c). Triple immunofluorescence of RACK1-MITF together with cytokeratin 5 (KRT5), a marker of basal keratinocytes, showed that MITF-positive cells were RACK1-KRT5 negative, letting us exclude the RACK1 signal on melanocyte membranes of healthy skin (Suppl. Fig. S1).
In every melanocytic lesion examined, RACK1 was detected in all tumoral MITF-positive cells. The RACK1 signal appeared with 2 distinct distribution patterns. RACK1 was either heterogeneously (Fig. 2b) or homogeneously (Fig. 3b–5b) distributed over the lesion. The RACK1 heterogeneous pattern consisted of a granular cytoplasmic staining of low to moderate intensity. In sharp contrast, the RACK1 homogenous pattern was intense and diffuse, cytoplasmic, and perinuclear. RACK1 signal was detected by immunofluorescence regardless of the pigmentation load of the sample. Samples were arbitrarily graded 0 when homogenous and 1 when heterogeneous. When tested in samples of tumors considered in the melanocytic tumor differential diagnosis, RACK1 was detected in MITF-negative cells with variable intensity (not shown).
To correlate RACK1 distribution with prognosis markers of our tumoral samples, 2 authors independently estimated the mitotic index and Ki-67 index (CA, GE). Mitotic index values per 10 HPF were either below 1 or higher than 4, with few exceptions (Table 2, column 1). Applying the prognostic cutoff according to the recent literature, which distinguishes melanocytic neoplasms with cutaneous or oral origin,3,15 data were concordant with the pathology reports except for sample 22. The Ki-67 index, also known as the growth fraction, displayed values either below 7 or higher than 15 with few exceptions (Table 2, column 2). The accepted cutoff of 15% for canine melanocytic neoplasms3,15,28,29 supported pathology reports, with the exception of samples 16, 20, and 23, which displayed an otherwise high mitotic index. Conversely, sample 22 displayed a high Ki-67 index. Overall, these data confirmed pathology reports with statistical significance (P < .01, Student’s t-test).
Histomorphological Features, Ki-67 Index, and RACK1 Pattern in Canine Melanocytic Lesions.
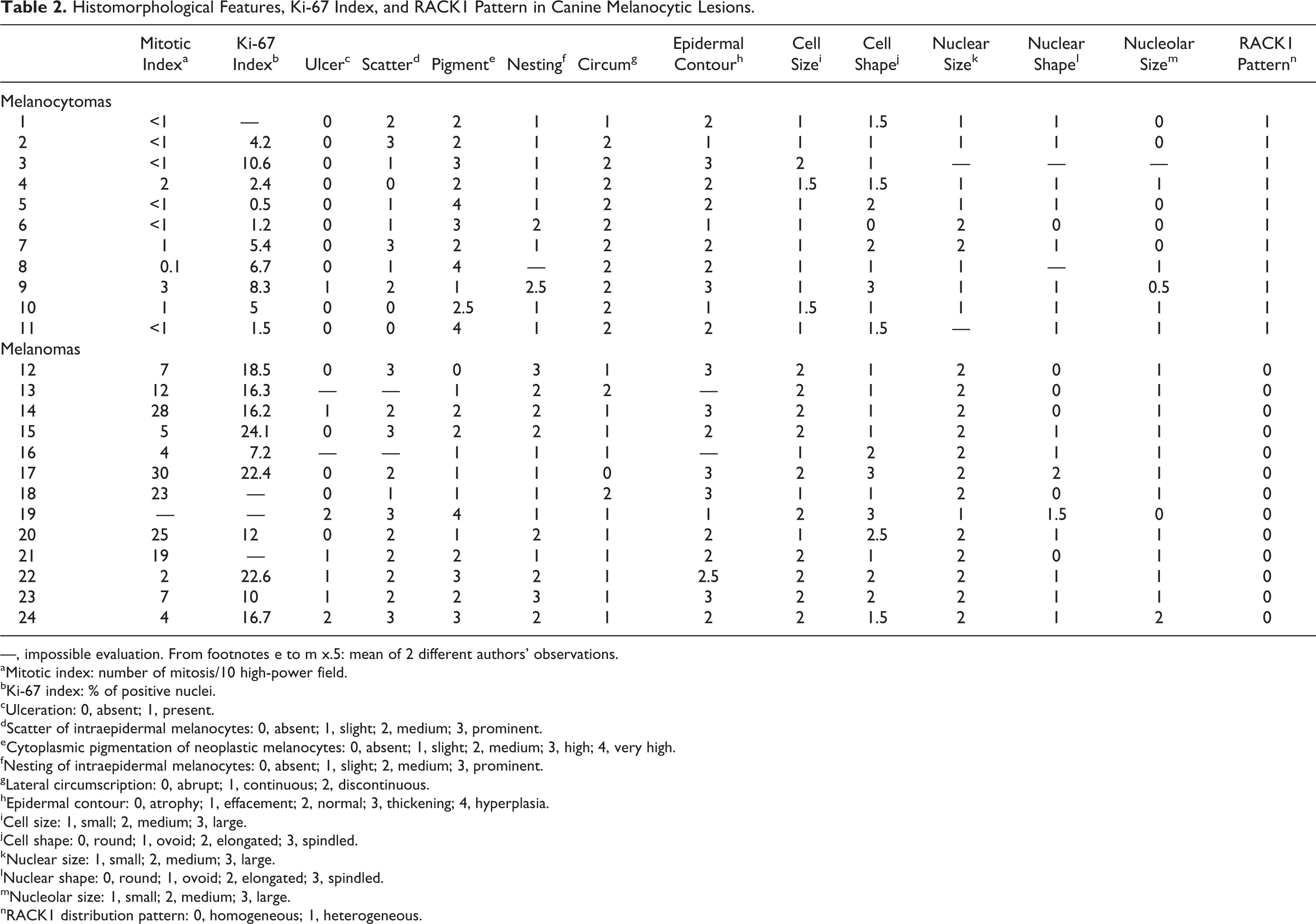
—, impossible evaluation. From footnotes e to m x.5: mean of 2 different authors’ observations.
aMitotic index: number of mitosis/10 high-power field.
bKi-67 index: % of positive nuclei.
cUlceration: 0, absent; 1, present.
dScatter of intraepidermal melanocytes: 0, absent; 1, slight; 2, medium; 3, prominent.
eCytoplasmic pigmentation of neoplastic melanocytes: 0, absent; 1, slight; 2, medium; 3, high; 4, very high.
fNesting of intraepidermal melanocytes: 0, absent; 1, slight; 2, medium; 3, prominent.
gLateral circumscription: 0, abrupt; 1, continuous; 2, discontinuous.
hEpidermal contour: 0, atrophy; 1, effacement; 2, normal; 3, thickening; 4, hyperplasia.
iCell size: 1, small; 2, medium; 3, large.
jCell shape: 0, round; 1, ovoid; 2, elongated; 3, spindled.
kNuclear size: 1, small; 2, medium; 3, large.
lNuclear shape: 0, round; 1, ovoid; 2, elongated; 3, spindled.
mNucleolar size: 1, small; 2, medium; 3, large.
nRACK1 distribution pattern: 0, homogeneous; 1, heterogeneous.
Hence, subsequently, melanocytic neoplasms were grouped according to the histopathologic reports (ie, melanocytomas vs melanomas). Comparison of RACK1 grading between both groups resulted in a statistically significant difference (P < .001, Student’s t-test) (Table 2, last column). All samples histologically classified as melanomas (n = 13) stained homogeneously for RACK1, with a strong diffuse expression, localized in perinuclear and cytoplasmic compartments. In sharp contrast, all samples classified as melanocytomas (n = 11) stained heterogeneously for RACK1, with a low to moderate cytoplasmic signal with a granular aspect. Thus, the cytoplasmic pattern of RACK1 labeling helped to distinguish melanomas from melanocytomas. Of note, with identical acquisition times, RACK1 staining intensity was higher in melanomas than in melanocytomas. This was also visible when the detection was performed by immunohistochemistry (Suppl. Figs. S2–S4), although for heavily pigmented sections, it was more difficult to assess than with immunofluorescence.
RACK1 Detection Is More Informative Than Individual Histomorphological Features
To extensively characterize our samples, we performed a detailed analysis based on morphological criteria for human melanomas 33 (Table 2). Compared with melanocytomas, we found that our samples of canine melanomas were frequently ulcerated (Table 2, column 3). At the cellular level, melanomas presented medium to prominent scatter of intraepidermal melanocytes (Table 2, column 4), lower pigmentation (Table 2, column 5), and higher nesting of intraepidermal melanocytes (Table 2, column 6). Moreover, the transition from the involved area to adjacent normal tissue, called lateral circumscription, tended to be continuous (Table 2, column 7), and the epidermal contour was thicker than in melanocytomas, indicating an epidermal hyperplasia (Table 2, column 8). These data correspond to described criteria. 11 However, none of these parameters reached statistical significance when compared with those of melanocytomas. Instead, a homogeneous RACK1 distribution pattern was correlated with 2 morphological variables: cell size (P < .005, Student's t-test) and nuclear size (P < .05, Student's t-test).
Discussion
In this immunofluorescence study, every cutaneous and oral sample presented a specific nuclear MITF labeling in melanocytic cells. Normal epidermal melanocytes as well as melanocytic cells from melanocytomas and melanomas were MITF positive. Amelanotic melanoma cells were also positively stained for MITF. By contrast, canine plasmocytomas, cutaneous histiocytomas, cutaneous nonepitheliotropic T-cell lymphoma, peripheral nerve sheath tumors, and low-grade fibrosarcoma were negative for MITF, highlighting its specificity when compared with these other neoplasms. Interestingly, MITF detection by immunohistochemistry was not positive in every sample, as reported. 28 The most likely explanation would be the increased sensitivity of immunofluorescence in the context of pigmentation, especially when acquired by a cooled CCD camera. We therefore propose MITF immunofluorescent labeling as a marker of melanocytic lineage in dogs as it labeled normal and neoplastic cells regardless of the malignancy status.
To extend previous observations made in human and horse melanomas,5,7 we assessed the value of RACK1 detection in canine melanoma diagnosis. We found RACK1 overexpression in melanocytic neoplasms compared with normal cutaneous melanocytes. RACK1 overexpression was visible in every lesion over the whole specimen. Using the mitotic index as a parameter with prognostic significance for canine melanocytic neoplasms,3,15,20,25 we were able to confirm the diagnosis of our sample collection. We showed here that the cellular distribution of RACK1 labeling distinguishes malignant from benign forms. Most of the routinely used immunostainings validate the melanocytic origin of the neoplasms without indication of malignancy.9,17,22,23,28 Recently, the matrix metallopeptidase 9 (MMP-9) distribution was reported to distinguish melanocytomas from melanoma in canine tissues. 6 Nevertheless, in contrast to RACK1 evaluation, the staining gradation for MMP-9 could occasionally show coincidence in benign and malignant tumors and thus requires trained pathologists to be assessed correctly. RACK1 detection is straightforward, with the heterogeneous, granular, often moderate signal being indicative of a melanocytoma and the intense cytoplasmic staining indicative of melanoma. Some canine cutaneous tumors considered in the melanocytic neoplasm differential diagnosis were also positive for RACK1, pointing out the necessity of a melanocytic labeling such as MITF to identify the origin of the tumor.
The Ki-67 index also was assessed to further evaluate the diagnosis. Although in 83% (20/24) of tumor samples, the mitotic index and the growth fraction were concordant, 4 melanocytic neoplasms classified histologically as melanomas presented discordant indexes. They were localized either at the lips or at the toes. Interestingly, 1 report shows that only 38% of feet and lip melanocytic neoplasms had exhibited malignant behavior despite the fact that 74% had been diagnosed as malignant by histological characteristics. 31 Parameters such as the mitotic index and nuclear atypia were highly correlated with malignancy, but the Ki-67 index has not been reported. In the present study, the discordant samples displayed other parameters correlating with malignancy, such as nuclear size or RACK1 pattern. These data could suggest that toe and lip melanocytic neoplasms form a group of tumors with a specific Ki-67 or mitotic index cutoff as proposed for oral and cutaneous melanocytic neoplasms. 31
Melanocytic neoplasms are essentially heterogeneous. Profiles of genetic changes within tumor cells offer the potential to detect the genetic alterations leading to malignant behavior without overt histological evidence of malignancy. The original work by Viros and colleagues 33 considered criteria that could possibly identify genetic subgroups of human melanomas through simple histomorphological features. To make a detailed morphological classification, we used these pathological criteria recently defined for human melanomas in canine melanocytomas and melanomas. Canine melanocytomas are benign melanocytic lesions that are not readily comparable to benign melanocytic lesions in humans but share some aspects of human melanomas in terms of clinical evolution, preferential metastatic sites, 11 and genetic alterations.13,14,18,24 We tested whether the criteria that had successfully identified melanomas with BRAF mutations 33 could help to reveal aspects of “mildness” or malignancy between heterogeneous groups of canine melanocytomas and melanomas. In the current study, we worked with archive material for which routine pathology reports were unequivocal. We checked if one of the histomorphological criteria would confirm the partitioning of the samples as benign or malignant. As previously reported, taken individually, most parameters were not informative.22,29
When comparing RACK1 distribution with each criterion, we identified a positive correlation between RACK1 distribution and cell size. The size of a cell plays an indirect role in cell proliferation, as a doubling in cell size is essential before cell division. Moreover, insulin-like growth factor 1 receptor (IGF1R), which controls about 50% of the cell size, 2 was shown to be expressed in canine melanomas. 32 Interestingly, RACK1 builds protein complexes with IGF1R in other cell types (reviewed in Adams et al 1 ). Besides, it is accepted that nuclear atypia and proliferative activity are significantly correlated with outcome in canine melanocytic neoplasms at any anatomical site. 3 , 28 , 29 , 31 Among the single histomorphometric criteria used in this study, nuclear volume seems to have accounted for nuclear atypia scored by others as a combination of increased nuclear volume and nucleolar size, position, and multiplicity. 3 , 31 Moreover, we found a correlation between RACK1 expression and Ki-67 index in melanocytic neoplasms. Similar results were reported for human oral squamous carcinoma. 34 Altogether, this suggests a correlation between RACK1 expression and melanocytic cell proliferation. Fittingly, in human melanoma cell lines, a role for RACK1, specific to melanoma, involving a crosstalk between the extracellular signal-regulated kinase and the c-Jun N-terminal kinase signaling pathways, was proposed. 16 In these models, activation of both pathways led to c-Jun phosphorylation and stabilization, which finally resulted in cyclin D1 (CCND1) transcription. CCND1, the regulatory subunit of cyclin-dependent kinases (CDK4 and CDK6) involved in cell cycle control, is often found overexpressed in human melanomas. 26 To date, no data are available on CCND1 expression in canine melanomas.
In the present study, cutaneous melanocytic neoplasms excised from different anatomical locations, together with oral melanomas, were analyzed. Although working with a limited collection of melanocytic neoplasms, our findings suggest that RACK1 distribution differs between benign and malignant canine melanocytic proliferations. A RACK1 homogeneous labeling pattern displays high concordance with other criteria such as histology, Ki-67, or mitotic index, as well as classic features of malignancy in a selected subset of tumors. RACK1 protein was detected with a homogenous, intense, and diffuse cytoplasmic staining in MITF-positive cells of canine melanomas. In contrast, it was heterogeneous, moderately intense, and granular in the cytoplasm of MITF-positive cells of canine melanocytomas. These findings, although suggestive, are not yet sufficient to demonstrate diagnostic utility of this marker. RACK1 diagnostic value should be further tested on a group of tumors with known outcome, which would include those with histological ambiguity. RACK1 could be involved in melanomagenesis, notably in uncontrolled cell proliferation. Functional studies are ongoing in our laboratory to establish the exact function of RACK1 in melanoma development.
Footnotes
Acknowledgements
We thank Inès Barthélémy and Stéphane Blot for providing control dog tissues and Kevin Cheesman for his contribution to the initial steps of the project. We are grateful to Agnès Champeix and Patricia Wattier for histologic techniques and to Uwe Maskos for careful review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by grants from Institut National de la Recherche Agronomique, Agence Nationale de la Recherche Emergence Bio (ANR-09-EBIO-006-01), and Association pour la Recherche contre le Cancer (ARC1002-0830F). C.C. received a Mitjaville scholarship from the Académie Nationale de Médecine (2008–2009) and a grant (Allocation de Recherche MENRT) from the French Ministry of Research (2009–2012).
