Abstract
Many studies have evaluated various prognostic markers for canine melanocytic neoplasms either as primary or secondary goals; however, design, methodology, and statistical validation vary widely across these studies. The goal of this article was to evaluate and compare published canine melanocytic neoplasm studies in relation to the principals established in the Recommended Guidelines for the Conduct and Evaluation of Prognostic Studies in Veterinary Oncology. Based on this evaluation, we determined which parameters currently have the most statistically supported validity for prognostic use in canine melanocytic neoplasia. This information can also be used as part of evidence-based prospective evaluations of treatment regimens. Additionally, we highlight areas in which the current data are incomplete and that warrant further evaluation. This article represents an initiative of the American College of Veterinary Pathologists' Oncology Committee and has been reviewed and endorsed by the World Small Animal Veterinary Association.
Melanocytic neoplasms are commonly diagnosed in dogs, and malignant melanoma is reportedly the most common canine oral malignancy. 3,13,15,16,36,44,47 Despite the prevalence of these neoplasms, one review states that “there is no single diagnostic technique capable of differentiating benign from malignant melanocytic neoplasms or of predicting survival time.” 44 Another study stated that the “behaviour of melanocytic neoplasms is a continuous spectrum ranging from strictly benign to highly malignant. Thus, the borderline between the 2 categories may be rather broad.” 37
The primary foundation literature that describes the morphology and biological behavior of melanocytic neoplasms in dogs and cats begins with veterinary publications of the late 1950s and early 1960s. In these early studies, some of which were written by medical or dental pathologists, 17,33 the morphology of neoplasms was often described as being malignant or benign without specifically defining the anatomic basis for that distinction. The behavior of these neoplasms was similarly determined anecdotally from necropsy reports, but there was often no correlation between specific histological criteria of malignancy and the metastatic lesions observed. 7
It has been an accepted observation that canine melanocytic neoplasms vary widely in biological behavior. Although many studies have evaluated various prognostic markers for canine melanocytic neoplasms, either as primary or secondary goals, few true prognostic studies exist. Additionally, there are no universally accepted criteria to prognosticate canine melanocytic neoplasms. It is therefore unfortunate that potentially invalid conclusions of both early and current studies regarding prognosis of melanocytic neoplasms have become widely cited in routine neoplasia pathology practice. Such studies are referenced regardless of whether the conclusions are adequately supported by well-designed methods with sufficient case numbers, an appropriate reference population, specific inclusion and classification criteria, uniform modalities of therapy, adequate statistical analysis, and complete clinical outcome assessment, including survival data. Furthermore, it is often difficult to compare prognostic studies because of different classification systems, different groupings of neoplasms such as by location or inclusion of both canine and feline neoplasms, and different methodologies for evaluating specific parameters such as mitotic index (MI) or growth fraction. Most published reports are retrospective studies, and few validate outcome and survival conclusions with postmortem findings. Without an accurate prognosis, appropriate recommendations regarding primary and/or adjunct therapy for dogs with melanocytic neoplasms cannot be made. 19
Prior to determining an accurate prognosis, an accurate diagnosis must be established. The lack of pigmentation and the tremendous cellular variability of some canine melanocytic neoplasms may pose significant challenges to the pathologist. Specific histological criteria such as junctional activity and/or finding nests of neoplastic cells within the overlying epithelium can help support a diagnosis of an amelanotic melanocytic neoplasm; 43,44 however, ultimate confirmation requires positive labeling with immunohistochemical markers that have been shown to have high specificity for these neoplasms, such as Melan-A, PNL-2, and tyrosine reactive proteins 1 and 2. 8,9,12,25,36,41,43,44,46 Some prognostic studies have included amelanotic melanocytic neoplasms within a case series without providing proper evidence that these neoplasms are truly of melanocytic origin. 25,32,37 The accidental inclusion of soft tissue sarcomas or other neoplasms into a case series of melanocytic neoplasms may substantially alter the prognostic significance of the parameters under investigation.
In this work, we conducted a detailed literature review of canine melanocytic neoplasia publications and evaluated the different postulated prognostic classification schemes according to the recently published Recommended Guidelines for the Conduct and Evaluation of Prognostic Studies in Veterinary Oncology. 49 These guidelines were initiated by the ACVP Oncology Committee and reflect the current consensus opinion of veterinary pathologists and oncologists on how best to assess the prognostic classification systems used to characterize canine neoplasms. Based on these criteria, we determined which prognostic parameters have been demonstrated to have statistical significance according to these standards (Table 1 ). Additionally, we have provided recommendations for the prognostication of canine cutaneous and digit melanocytic neoplasms and for oral and lip melanocytic neoplasms (Table 2 ). Ocular melanocytic neoplasms will not be discussed owing to insufficient material.
Significance of Prognostic Factors for Canine Melanocytic Neoplasms Based on Location
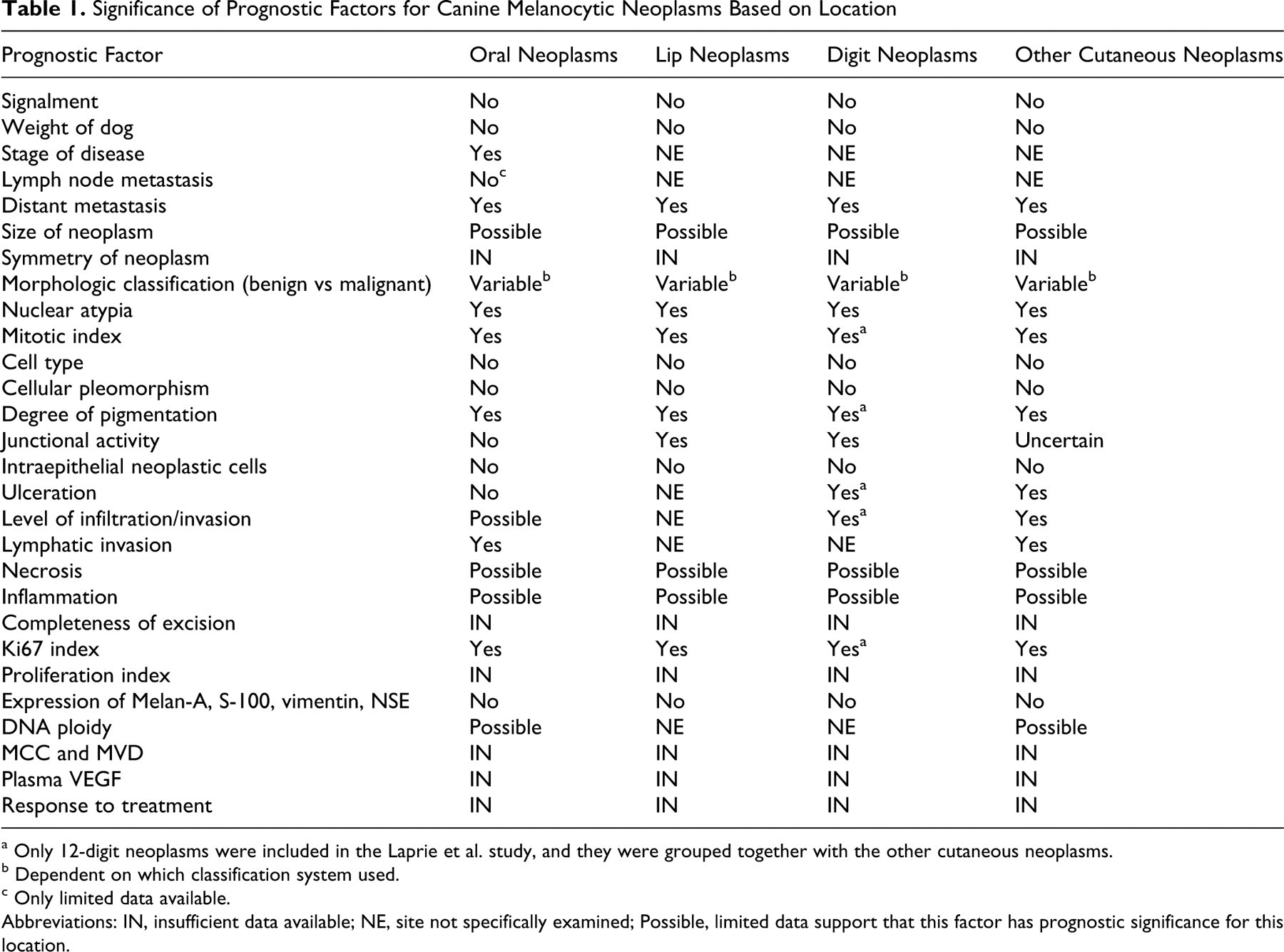
a Only 12-digit neoplasms were included in the Laprie et al. study, and they were grouped together with the other cutaneous neoplasms.
b Dependent on which classification system used.
c Only limited data available. Abbreviations: IN, insufficient data available; NE, site not specifically examined; Possible, limited data support that this factor has prognostic significance for this location.
Recommendations for Prognostication of Canine Melanocytic Neoplasms
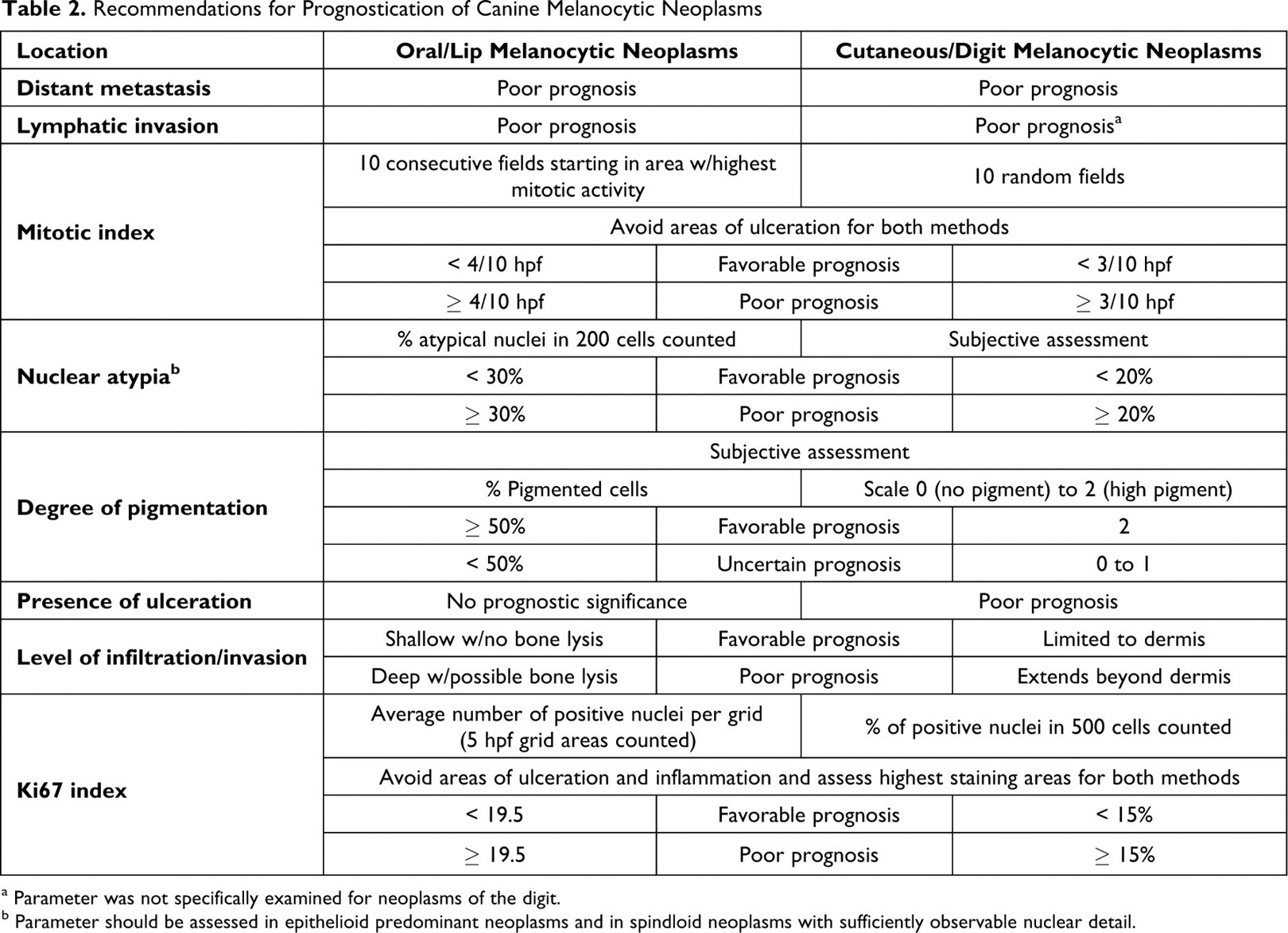
a Parameter was not specifically examined for neoplasms of the digit.
b Parameter should be assessed in epithelioid predominant neoplasms and in spindloid neoplasms with sufficiently observable nuclear detail.
The results and conclusions of each evaluated study are described under the relevant parameter headings, and the consensus significance of each parameter is summarized. The main categories of parameters include: signalment, clinical staging/response to treatment, neoplasm location, gross morphologic features, histological features, and molecular biologic attributes.
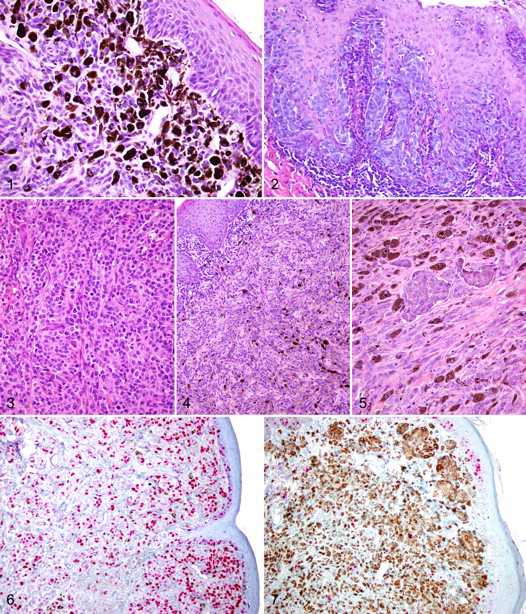
Throughout this article, the term “melanocytoma” will be used to refer to a benign neoplasm of melanocytic origin, whereas the term “malignant melanoma” will be used to refer to a malignant neoplasm of melanocytic origin according to the current World Health Organization (WHO) nomenclature, 15 regardless of the terminology employed in the study being discussed. The term “melanocytic neoplasms” will be used when it is unknown whether the lesions are benign or malignant and when it is known that both benign and malignant neoplasms are being referenced together. Junctional activity refers to proliferation of neoplastic cells at the dermo-epidermal junction (Fig. 1 ). 15,16 The term “junctional activity” has often been used incorrectly to describe the presence of intraepithelial neoplastic cells. Intraepithelial neoplastic cells are a distinct histological feature of many melanocytic neoplasms, and the term “compound neoplasm” is used to describe a neoplasm that has both an epidermal (Fig. 2) and dermal component. 15,16 Dermal neoplasms are confined to the dermis. 15,16
Signalment
Although many prognostic studies have recorded the breed of affected dogs, only one study has shown that breed may have some prognostic significance. This study reported that more than 75% of melanocytic neoplasms exhibited benign behavior in some breeds (Doberman Pinscher and Miniature Schnauzer), whereas in other breeds (Miniature Poodle), more than 85% of melanocytic neoplasms were malignant. 4 Both cutaneous and oral melanocytic neoplasms were included in that study. 4 Statistical analysis was not performed for this prognostic factor, however. In another report, Golden Retrievers, Labrador Retrievers, and Cocker Spaniels represented one-third of the dogs in the study, but there was no statistically significant difference in breed predisposition reported. 19
Sex has not been demonstrated to be a prognostic factor for dogs with melanocytic neoplasms of any site. Whereas earlier reports state that male dogs have a higher frequency of melanoma than female dogs, 44 several more recent studies have not reported any significant sex differences in terms of survival. 1,2,11,19,20,28,29,32,35,40,42,45
In general, it would appear that malignant melanocytic neoplasms are more common in older dogs. The mean age of dogs with benign melanocytic neoplasms was 8.1 (n = 86) years, whereas the mean age of dogs with malignant melanocytic neoplasms was 11.6 (n = 71) years in one study. 4 However, patient age does not have definitive prognostic significance. A few studies have reported that dogs with oral malignant melanomas are generally older than dogs with cutaneous malignant melanomas, 31,42 but age has not been shown to be correlated with survival for oral neoplasms in most studies. 2,19,29,31,35,42,45 One study stated that age negatively influenced survival of dogs with melanocytic neoplasms arising from the skin, feet, and lips. 45 Age is a difficult prognostic factor to evaluate, as older dogs are more likely to suffer intra- and postoperative complications and slower recovery from surgery. Older animals are also more likely to have additional life-limiting comorbid conditions. One report stated that age significantly affected survival of dogs with malignant oral neoplasms of various cell lines of origin, including malignant melanomas, that were treated by partial mandibulectomy (P < .001). 27
There is no evidence to suggest that the weight of the affected dog has any prognostic significance. 19,35
Clinical Staging and Response to Treatment
Stage of disease has been shown to be significantly associated with clinical and survival outcome for dogs with oral malignant melanoma. 19,20,29 In one study, the authors staged dogs with oral malignant melanomas according to the then-current WHO system for oral neoplasia and found no statistically significant difference for remission length or survival times using this method. 19 These authors proposed an alternative system for staging of canine oral malignant melanomas based on neoplasm size, location within the oral cavity, and mitotic index. 19 They reported statistically significant differences in remission length and survival time for different stages using this alternative system. 19
It seems intuitive that evidence of metastasis would indicate a poor prognosis. Perhaps this is the reason many studies have reported metastatic rates, but very few have adequately evaluated it as a prognostic factor. In one study, metastasis was a significant negative determinant of patient survival for all neoplasm locations. 45 Another study reported that for oral malignant melanomas, the absence of distant metastasis at the time of surgery was statistically significant for longer remission lengths and survival times; however, it was found to be of little consequence, as these substrata were overrepresented in their respective groups. 19 Only 2 studies specifically examined regional lymph node metastasis as a prognostic factor. 19,35 Unlike distant metastasis, regional lymph node metastasis had no prognostic value for remission length, “time to first event,” or survival time (P > .05) for dogs with oral “malignant” melanomas. 19,35 It should be noted that only neoplasms histologically designated as malignant were included in these latter 2 studies, thus there was no reference population.
Based on these data, it can be concluded that evidence of visceral metastasis is associated with a poor prognosis for canine melanocytic neoplasms of any site. Additional studies that include benign and malignant melanocytic neoplasms from various anatomic sites are needed in order to make definitive conclusions regarding lymph node metastasis as a prognostic factor.
It is difficult to draw sound conclusions regarding the effectiveness of various treatment modalities for canine melanocytic neoplasms for 2 main reasons. First, few clinical studies provide adequate description of the histological criteria used to diagnose a neoplasm as malignant. Neoplasm location alone should not be used to classify a melanocytic neoplasm as being malignant or benign. As discussed in the following, a subset of oral and lip melanocytic neoplasms has been identified that do not exhibit malignant behavior. 2,10,45 Thus, attempts to separate these neoplasms from true malignant melanomas must be performed before treatment strategies can accurately be evaluated. Without knowing the criteria used to categorize the neoplasms as malignant, it is not possible to accurately interpret the survival data. Second, many clinical studies have staged dogs based on the WHO recommended guidelines and compared historically published survival times for each stage with the survival times of the dogs under study in a given case series. Based on this system, stage I dogs have neoplasms that are < 2 cm in diameter, stage II dogs have neoplasms that are 2 to < 4 cm in diameter, stage III dogs have neoplasms that are ≥ 4 cm in diameter and/or there is evidence of lymph node metastasis, and stage IV dogs have evidence of distant metastasis. Extreme caution must be used when historically published survival times are used as comparators, because each study set of neoplasms can be extremely different, and, as stated above, many studies have not used strict histological criteria to select cases for inclusion. The location, histological features, and clinical parameters, in addition to the overall quality of the study, all need to be considered when choosing an appropriate reference set of melanocytic neoplasms for comparison. For example, in a study by Esplin, the mean survival time for dogs with oral or lip melanocytic neoplasms was 22.7 months, but this study included only histologically well-differentiated melanocytic neoplasms. 10 It would be erroneous to use this mean survival time for statistical analysis in a study set of neoplasms that included any melanocytic neoplasm other than histologically well-differentiated melanocytic neoplasms from the oral cavity or lip. Another concern with studies that use stage to classify cases is whether or not the staging classification system really has prognostic significance. As discussed above, Hahn et al. reported that there was no statistically significant difference for remission length or survival time using the WHO staging system for oral melanocytic neoplasms, but that an alternative staging system did have prognostic significance. 19 Additionally, the WHO staging system for oral melanocytic neoplasms is based largely on primary neoplasm size, which has not definitively been proven to have prognostic significance. Also, as discussed by Bergman (2007), the size of the neoplasm has not been standardized to the size of the patient and histological criteria are not incorporated into this system. 3 For these reasons, well-designed prospective studies with standardized clinical staging, histopathologic diagnoses, treatment protocols, follow-up, and statistical evaluation are greatly needed to accurately evaluate various treatment modalities for canine melanocytic neoplasms of various body sites. 47
Neoplasm Location
Perhaps more than any other parameter, neoplasm location has been historically relied on to predict the biological behavior of melanocytic neoplasia. Numerous studies have evaluated location in this context. 1,2,4,5,10,19,20,28,31,32,36,37,42,45 Historically, melanocytic neoplasms of the oral cavity and lip have been considered to have a poor prognosis and neoplasms of the skin have been considered to have an overall favorable prognosis. 3,4,5,19,36,44 The statement that all oral melanocytic neoplasms should be considered malignant is commonly found throughout the literature and has become dogma. 3,4,5,13,14,36,44 Although numerous studies have indeed reported that a high percentage of melanocytic neoplasms from the oral cavity exhibit malignant behavior, 4,5,31,32 generalized prognostic statements based on neoplasm location have been refuted by more recent studies that have identified a subset of oral and lip melanocytic neoplasms with a more favorable prognosis. 2,10,42,45
Melanocytic neoplasms of the oral cavity had the shortest median survival time (147 days) when compared with those of the feet and lips (676 days) and to those from the skin (725 days) in one study. 45 However, whereas 92% of the oral melanocytic neoplasms had been classified as malignant in the original biopsy report, metastasis or recurrence was observed in only 59% of the cases. 45 Case follow-up intervals for this study ranged from 12 months to 4 years. Independently, another study demonstrated that heavily pigmented oral and lip neoplasms composed of well-differentiated melanocytes with a low mitotic index of ≤ 3 per 10 high-power fields (hpf) had a favorable prognosis. 10 Ninety-five percent of dogs with neoplasms meeting these criteria either survived the duration of the study period (51 months) or died of other causes (23.4 months mean survival time). 10 The risk of neoplasm-associated death was the same for melanocytic neoplasms of the lip as compared with lesions at other locations within the oral cavity. 10 In a third study, approximately one-third of dogs with melanocytic neoplasia of the lip survived at least 1 year after surgical removal, despite the majority of neoplasms having a malignant histological appearance, which was not specifically defined. 42 Of dogs with “benign”-appearing melanocytic lip neoplasms, 9 of 10 (90%) survived at least 1 year in that study. 42 A major difficulty in comparing studies that have examined melanocytic neoplasms of the lip is that many authors do not differentiate between lip neoplasms that arise from the haired skin and those that arise from the mucosa. Differences in survival times may be a result of the type of lip neoplasms (haired skin vs mucosa) present in the various studies.
For cutaneous melanocytic neoplasms, digital neoplasms have been considered to have a worse prognosis than those from elsewhere on the skin. Neoplasms of the digit had a lower 2-year survival rate (56%) when compared with lesions of other cutaneous sites (83.8%) in one study; however, this difference was not significant because of the small number of cases evaluated. 28 Others have independently reported 1-year survival rates for digital neoplasms of 44% 21 and 42% 30 and 2-year survival rates of 11% 21 and 13%. 30 In another study, 74% of melanocytic neoplasms of the feet and lips had originally been reported to be malignant, but only 38% of those neoplasms actually demonstrated malignant behavior. 45 On the other hand, 59% of melanocytic neoplasms of the skin were originally reported as malignant, but only 12% actually exhibited malignant behavior. 45 There was also a subset of cutaneous melanocytic neoplasms that exhibited malignant behavior that would have been predicted to be benign based on current microscopic criteria for prognostication. 45 As a consequence of low mortality among dogs with cutaneous neoplasms, the positive predictive value of the mathematical model used by the authors was a “virtual coin toss (54.5%),” suggesting that additional prognostic factors should be sought when evaluating cutaneous melanocytic neoplasms. 45 Since amelanotic melanocytic neoplasms were excluded from the study, the relative percentages of benign melanocytic neoplasms at each of the anatomic sites may be higher in this particular study population than that of the general population. In contrast, one study found that the prognosis following histologic diagnosis of a malignant melanoma was considered poor, regardless of the primary neoplasm site, although only 20 dogs were included in this study. 37 In an additional study, 45.8% of dogs (11 of 24) with cutaneous and 42.9% of dogs (6 of 14) with nailbed melanocytic neoplasms survived at least 1 year after surgical removal of the neoplasm. 42 In another study, although a higher proportion of neoplasms on the feet were histologically malignant, the behavior of malignant neoplasms on the foot also did not vary greatly from that of lesions arising from other skin locations. 5 However, the exact location on the foot (eg, digit, nail bed, pad, dorsal surface) was not indicated in this report.
Taken as a whole, these results support that anatomic location is indeed an important and integral prognostic parameter for canine melanocytic neoplasia. Melanocytic neoplasms in and around the mouth are, more often than not, aggressive in their nature, although a subset of oral and lip melanocytic neoplasms exhibits benign behavior. In haired skin, excluding feet, the proportion of benign-to-malignant melanocytic neoplasms is reversed when compared to oral neoplasms. However, cutaneous neoplasms with malignant behavior are more difficult to distinguish histologically from benign neoplasms than are oral or lip neoplasms. The real and current challenge for pathologists examining canine melanocytic neoplasms is the accurate and consistent recognition of benign oral neoplasms and malignant cutaneous ones.
Gross Morphologic Features
There is conflicting information in the current literature regarding the prognostic significance of the size of melanocytic neoplasms. In one study that included canine melanocytic neoplasms from various anatomic sites, increasing neoplasm size or volume was a significant negative determinant of patient survival. 45 Two additional studies found a negative correlation between increasing neoplasm volume and survival time in dogs with oral malignant melanomas. 19,35 This parameter was significantly related to “time to first event,” development of pulmonary metastasis, and survival time in a set of 111 canine oral malignant melanomas based on univariate and multivariate Cox regression analysis. 35 Hahn et al. reported that dogs with oral malignant melanomas of less than 8 cm 3 had significantly longer remission lengths and survival times. 19 Again, neither of these 2 studies included a reference population. Several other studies have not identified a correlation between lesion size and clinical outcome. 5,20,27,31,37,42,48 Because of the conflicting reports, the practical value of this parameter as a prognostic indicator is still questionable. Further evaluation of this parameter is required, using a statistically determined threshold value for comparison to clinical outcome and survival data.
Symmetry has not been assessed as a prognostic factor for canine melanocytic neoplasms. Aronsohn et al. recorded the symmetry of the lesion for 14 melanocytomas of the distal extremities but did not assess this parameter for the 14 malignant melanomas reported in that study. 1 No conclusions were made with regard to prognosis.
Histological Features
The histological characteristics of benign and malignant melanocytic neoplasms in dogs were defined in detail in 1974 in the WHO International Histological Classification of Tumours of Domestic Animals. 50 Early reports were often descriptive and lacked correlation of histological features with clinical outcome. Consequently, there are conflicts in the literature when these descriptive criteria are used to predict outcome. Some reports show high correlation between histological appearance and clinical outcome, 4,28,37,47 whereas others state that histological appearance is of no prognostic value, 20 and a few studies have shown variable results. 5,42 A major complicating factor is that histological evaluation of atypia is somewhat subjective. Traditionally, the mitotic index is the only histological parameter to which a numerical value is assigned. Some argue that even mitotic index is not totally objective owing to variations in field selection, size and number of fields, number of counted cells, degree of pigmentation, or whether bleaching was performed. Another challenge is that histological features are sometimes combined with nonhistological features, such as neoplasm size or location, when assessing neoplasm behavior, which makes it difficult to determine the relative contribution of histological appearance to the predictive accuracy. Here we review the following histological parameters for their ability to predict prognosis of canine melanocytic neoplasms: morphologic classification (also referred to in the literature as histological diagnosis or cytologic features); nuclear atypia; predominant cell type (eg, spindloid, epithelioid, mixed); cellular pleomorphism; mitotic index; degree of pigmentation; junctional activity; intraepithelial neoplastic cells; ulceration; level of infiltration or invasion; necrosis; inflammation; and margin evaluation.
Morphologic Classification
Many studies use morphologic criteria to classify melanocytic neoplasms as benign or malignant. Although morphologic classification appears to be helpful in predicting prognosis, different classification schemes are often used in different studies, making comparisons difficult. Most studies have evaluated the prognostic significance of individual criteria, such as nuclear atypia or cellular pleomorphism, among other features, whereas some studies have evaluated these features under a broader assessment of “morphologic classification.” 4,5,20,28,37,42,47
An early report used multiple criteria described in a previous study to confirm malignancy in a set of 121 oral and pharyngeal melanocytic neoplasms. 7,47 These features were presence of pigment, degree of tissue infiltration, neoplasm-cell emboli in vessels, and metastatic deposits in regional lymph nodes. Nuclear pleomorphism and abundant mitoses, although the specific numbers were not given, were present in almost every case. 47 This study appeared to profile highly aggressive melanocytic neoplasms, as 52 of 54 necropsied dogs (96%) displayed evidence of metastasis or local recurrence. 47 Thus, it was not designed to evaluate the discriminatory power of these parameters between benign and malignant neoplasms. Additionally, details regarding classification criteria were not provided, the univariate significance of individual prognostic factors was not considered, and no statistical analysis was performed.
One study histologically classified melanocytic neoplasms as malignant or benign according to the WHO classification system available at that time. 5,50 This classification was based on criteria established in human medicine that had not yet been shown to be prognostically significant in dogs. Fifty-nine dogs with histologically benign neoplasms had a median survival time of 110 weeks, and within a 2-year follow-up period, 7 of these 59 (12%) dogs died. 5 Seventy-five dogs with histologically malignant neoplasms had a median survival time of 30 weeks, and in the same 2-year observation period, 53 of these 75 (71%) dogs died. 5 The study was unable to correctly classify a small number of neoplasms (about 10%) that appeared histologically benign but exhibited malignant behavior. 5 Furthermore, the median survival time of dogs with histologically malignant cutaneous melanomas was 70 weeks, with only 15 of 33 (45%) dogs dying from the neoplasm within a 2-year period. 5 This result raises a question about the validity of this classification to correctly identify malignant melanomas of the skin. When the same classification system was used in another study of 169 melanocytic neoplasms from various locations, the authors reported an overall accuracy rate of 89% to correctly identify the biological behavior based on the morphologic classification. 4 Nevertheless, 14 neoplasms were behaviorally benign but histologically malignant. Eight of these lesions arose in skin (4 from the digit), 4 were ocular, and 2 were of the oral cavity. These neoplasms reportedly exhibited at least 1 histological criterion of malignancy, which was usually cellular pleomorphism with or without a high mitotic index. 4
In another study, melanocytic neoplasms were classified as malignant or benign based on cell size and shape, nuclear size and shape, chromatin pattern, prominence of nucleoli, and lack of pigment. 42 These features were subjectively assessed. The study included 27 histologically malignant nail bed neoplasms, 58 histologically malignant skin neoplasms, and 80 lip neoplasms that were a mix of both histologically benign and malignant lesions. One-year outcomes were known for 84 cases (46/80 lip neoplasms, 14/27 nail bed neoplasms, and 24/58 haired-skin neoplasms). Thirty-two dogs had lip neoplasms that were histologically classified as malignant, yet 10 of these 32 (31%) dogs survived at least 1 year. Fourteen dogs had lip neoplasms that were histologically classified as benign. Of this group of dogs, 10 had lesions arising from the mucous membrane and 4 were from the haired skin. Twelve of these 14 dogs were neoplasm free for at least a year. Almost half of the dogs with histologically malignant skin or nail bed neoplasms survived over 1 year after neoplasm removal. The author concluded that traditional histological criteria of malignancy are useful, but a single feature or features correlating with outcome could not be identified. 42 Regression analysis was not performed in that study.
Regression analysis was performed in another study that classified neoplasms based on features of cellular pleomorphism and atypia, defined only as nuclear pleomorphism, nuclear hyperchromasia, mitotic figures, and microscopic infiltrative nature, which were used as cytologic features of malignancy. 37 The authors used an empirically determined cutoff point of more than 2 mitoses per 10 hpf to differentiate benign from malignant neoplasms. This histological classification method significantly correlated with survival (P < .001). 37 However, the study population was small (27 total neoplasms from various locations) and included both dog and cat cases that were not separated from one another for statistical assessment. Neoplasms were not separated by anatomic location, and the follow-up time to determine survival was only 6 months. Additionally, this study combined mitotic index and infiltrative nature in the histological classification, making it difficult to compare the results of this study to other publications.
Histological classification, based largely on mitotic index, correctly predicted the biological behavior in 63 of 68 cutaneous neoplasms (93%) in another study. 28 Dogs with histologically benign neoplasms had a statistically significant longer 2-year survival rate than dogs with malignant neoplasms (P < .0001). 28
Nuclear Atypia
Since classification by nuclear atypia relies on observer assessment and not a quantitative measure, highly specific and defined criteria for atypia are very important to maintain reproducibility and minimize inter-observer variation. When assessed by strict criteria, nuclear atypia was highly correlated with outcome for canine melanocytic neoplasms arising at any anatomic site in pigmented epithelioid-predominant neoplasms. 45 Furthermore, a specific threshold value could be statistically determined. 2,45 Well-differentiated neoplastic cells are defined as having a small nucleus with a single, centrally oriented nucleolus and minimal clumping of chromatin. 45 Condensed strands of nuclear chromatin commonly extend from the nucleolus to the nuclear membrane, with condensation of chromatin along the inner surface of the membrane. In sections of cells lacking a nucleolus, the chromatin is fine and evenly dispersed at the periphery of the nucleus. More undifferentiated neoplastic cells are characterized by larger nucleoli of less regular shape. 45 They are eccentrically located in the nucleus and often multiple. In some cases, multiple nucleoli haphazardly connect to the inner surface of the nuclear membrane by thin strands of chromatin and give the appearance of a coarsely vacuolated nucleus. 45 In the defining study, nuclear atypia was subjectively evaluated on a decile scale from 1 to 10, representing the estimated percentage of nuclei involved (ie, 0 = no nuclear atypia, 1 = 1-9% involved nuclei, 2 = 10-19% involved nuclei, 3 = 20–29% involved nuclei, and beyond, using this ordinal scale to a final score of 10.). 45 A variation on this method was used by Bergin et al., who used the same criteria semiquantitatively by assessing atypia in 200 counted cells within each oral and lip neoplasm. 2 This method was applied to all epithelioid and mixed neoplasms as well as to 4 of 5 spindloid variants that had sufficiently observable nuclear detail. 2 The 2 studies had comparable results but established slightly different threshold values for malignant behavior. One determined a threshold of ≥5 (≥ 40%) 45 and the other determined a threshold of ≥ 4 (≥30%) for oral melanocytic neoplasms. 2 This variation may be a result of remaining interobserver differences and slightly different statistical methods for establishing a threshold. When classified by nuclear atypia, 86.3% of the oral and lip melanocytic neoplasms were correctly classified with respect to outcome at 1 year. 2 Prognostic evaluation based on both nuclear atypia and MI had an overall correct behavioral classification of 81% for melanocytic neoplasms of the feet and lips. 45 A statistically predictive model using a nuclear atypia cutoff of >3 (> 20%) gave a sensitivity of 80%, a specificity of 94.4%, a positive predictive value of 54.5%, a negative predictive value of 98.2%, and an overall correct classification of 93.3% for cutaneous melanocytic neoplasms. 45
Despite these results, nuclear atypia scored in increments of 10 is time consuming to assess and still potentially susceptible to interobserver variation. Assessing the percentage of nuclei exhibiting atypia in 200 counted cells is somewhat more objective and less time consuming, but still perhaps too rigorous for routine diagnostic pathology. It remains to be seen whether the same criteria can be applied in a more subjective way (assessment as mild, moderate, severe) and still retain comparable sensitivity and specificity. An additional drawback is the inability to define nuclear atypia criteria in neoplasms with insufficient nuclear detail, such as those predominantly composed of spindle, whorled type, or signet-ring cells. 45
One study specifically evaluated anisokaryosis as a prognostic factor for cutaneous melanocytic neoplasms and found that it was associated with a significantly shorter survival time (P < .0001). 28 Anisokaryosis was determined subjectively on a scale of 1 to 4, 1 being low and 4 being marked, thereby making it difficult to reproduce the reported results. Three additional studies of oral and/or cutaneous neoplasms did not find a correlation between nuclear atypia and neoplasm behavior. 31,40,42 Possible reasons for this discrepancy include a low number of cases, a lack of neoplasms with a favorable outcome in the dataset, and less defined criteria for atypia.
Mitotic Index
MI is one of the factors that has been most commonly evaluated for prognostic utility in canine melanocytic neoplasia. 1,2,5,10,19,20,28,31,36,40,42,45 It has been shown to be a useful prognostic factor for melanocytic neoplasms of both the skin and oral cavity, including the lip, 2,5,19,31,40,45 although it does not appear to be as useful as nuclear atypia or growth fraction. 2,31,45 MI has been defined in many different ways, including: the average number of mitotic figures per hpf with variable numbers of fields counted, a range of numbers of mitotic figures per hpf (eg, 0-2 mitoses per hpf), and the number of mitotic figures per 10 hpf. MI has also been assessed in different ways, such as the number of mitotic figures counted in a certain number of random fields, or in a certain number of fields within an area of high mitotic activity. This variability in reporting criteria makes it difficult to compare results. When determining the mitotic index in 2 studies, areas underlying ulceration were avoided, 2,28 and in 3 other studies an MI of 0 was assigned to all areas where the nuclei were obscured by pigment. 5,10,19 Assigning an MI of 0 to heavily pigmented areas was supported by identification of no to rare mitotic figures in bleached samples of such areas in one study. 10 To avoid invalid comparisons, it is critical for the reader to pay close attention to how the MI is reported in each study and to refer to the original study.
In multiple studies, the MI was determined by counting the number of mitoses in 10 consecutive non-overlapping hpf with commencement in an area of high mitotic activity. 2,44,45 According to this method, oral and lip melanocytic neoplasms with ≥ 4 mitoses per 10 hpf have been associated with an increased risk of patient death within 1 year of diagnosis. 2 This threshold value had a sensitivity of 90% and a specificity of 84%. 2 To the best of our knowledge, this report provides the only statistically determined threshold value shown to have prognostic significance. This value is similar to the findings in a descriptive evaluation of 69 well-differentiated, highly pigmented lip and oral melanocytic neoplasms with a favorable outcome. In that study, none of these well-differentiated neoplasms had an MI > 3 per 10 hpf. 10 In another study, the MI was determined to be a significant negative determinant of patient survival for oral melanocytic neoplasms. 45 The cutoff value for MI in oral neoplasms in this study 45 (14 per 10 hpf) dramatically differed from the cutoff for oral neoplasms in the Bergin et al. study 2 (≥4 per 10 hpf), despite use of part of the same dataset. This finding may be a result of different methods of statistical evaluation and significantly larger numbers of well-differentiated neoplasms in one study. 2
For cutaneous melanocytic neoplasms, the MI was demonstrated to be a significant negative determinant of patient survival in 3 studies. 5,28,45 The MI (P ≤ .001) provided a strong indication of eventual clinical outcome for melanocytic neoplasms in one of these studies; however, a predictive model based on the MI could not be constructed. 45 In contrast, an MI ≥3 per 10 randomly selected hpf, with avoidance of areas underlying ulceration, was shown to be statistically correlated with a low 2-year survival rate in the other 2 studies. 5,28 In one of these studies, the histologic diagnosis of cutaneous melanocytic neoplasms was correlated with MI in 67 of 68 cases. 28 Based on multivariate analysis using a Cox regression model, increasing MI was highly correlated with poorer prognosis. 28 The empirically determined cutoff value of ≥ 3 per 10 hpf had a predictive value of 91% (62 of 68 cases were accurately predicted) for survival and was significantly correlated with the Ki67 index (r = 0.596; P < .0001). 28 In the second study, there was a significant difference (P < .01) in survival over a 2-year study period for dogs with a cutaneous melanocytic neoplasm in which the MI was < 3 per 10 hpf (median survival of 104 weeks) versus dogs with melanocytic neoplasms with a MI of ≥ 3 per 10 hpf (median survival of 30 weeks). 5
In 3 other studies, the MI was determined by the average number of mitoses per single hpf in various numbers of consecutive fields. 19,31,40 In one study, the MI was based on the average number of mitotic figures per hpf determined by counting the number of mitoses in 20 randomly selected 450 µm-diameter high-magnification fields in a set of oral malignant melanomas. 19 Neoplasms were grouped into 2 categories–those with an MI ≤ 3 and those with an MI > 3 mitoses per single hpf based on separation analysis. Twelve dogs had neoplasms with an MI ≤ 3 per hpf and had significantly longer remission lengths and survival times than did other dogs. 19 Twenty neoplasms in this study had an MI that was > 3 mitoses per hpf. 19 In another study, the MI was defined as the average number of mitoses per hpf (40×) in 3 fields counted. 31 The average MI for dogs that were still alive at 1 year of follow-up, with no evidence of recurrence or metastasis, was 2.6 mitoses per hpf. The average MI for dogs that were dead within 1 year was 13.4 mitoses per hpf (P = .001). 31 There was also a strong correlation between Ki67 index and mitotic counts (r = 0.706; P = .0001). 31 Neoplasms were not separated by anatomical location in this study, which included both oral and cutaneous neoplasms. 31 In the third study, the average number of mitotic figures per single hpf was determined, after counting mitoses in 10 hpf in each neoplasm. 40 The average MI was 3.33 mitoses per hpf in this small series of 10 oral malignant melanomas with osteocartilaginous differentiation. In this study, 4 neoplasms with the highest mitotic indices recurred 60 days after diagnosis, and 3 neoplasms with the lowest MI did not recur during the follow-up period. 40 Thus, in these 3 studies, 19,31,40 the reported MI for melanocytic neoplasms with a poor prognosis would be comparable to > 30, 134, or 33.3 mitoses per 10 hpf, respectively. These values are significantly higher than those reported by other investigators, 2,5,28,45 but only melanocytic neoplasms histologically designated as “malignant” were evaluated in the latter 3 studies. 19,31,40
A few reports, however, have not found a prognostic significance of the MI for melanocytic neoplasms at specific anatomic locations. 5,20,36,42 Three studies did not find a statistically significant correlation between MI and survival rates for dogs with oral melanocytic neoplasms, 5,20,36 although oral neoplasms tended to have a higher MI than those in the skin in one of these reports. 5 Kaplan-Meier analysis showed a trend toward differing survival times of dogs with neoplasms of different mitotic indices in another study; however, these results were not statistically significant. 36 A separate study, using an MI cutoff of < 3 mitoses per hpf or ≥ 3 mitoses per hpf, did not find a prognostic significance of MI for lip, skin, and nail bed melanocytic neoplasms that had been histologically classified as malignant. 42 The cutoff point in this study was selected based on prior reports, rather than statistically determined, at a similar high value as reported in 2 previous studies. 19,31 The difference in results may be a reflection of this high cutoff point, which is much higher than those used in most other studies. 2,5,28,45
Predominant Cell Type
Melanocytic neoplasms are most commonly composed of 1 of the following 3 cell types: (1) epithelioid or polygonal (Fig. 3); (2) spindloid or fibromatous (Fig. 4); and (3) mixed epithelioid and spindloid (Fig. 5). 5,19,28,31,32,36,37,45 Other less commonly described cell types include: whorled type; 5,31,45 cellular; 5 balloon cell; 45 signet ring; 45 clear cell; 36 and an adenomatous/papillary type mentioned in one report. 36 Several studies have examined the predominant cell type as a potential prognostic parameter for canine cutaneous and oral or lip melanocytic neoplasms, but none has found a statistically significant correlation with survival. 1,5,19,28,31,32,37,45 Only 3 studies have suggested that cell type may be related to biologic behavior, but no association with survival was demonstrated. 5,31,32
Cellular Pleomorphism
A few studies have evaluated cellular pleomorphism as a prognostic factor, but poor characterization of this term in the literature makes it impossible to draw meaningful conclusions about its prognostic significance. Cellular pleomorphism may encompass such characteristics as cell size, cell shape, pigmentation, and nuclear features, among other factors. One study found that cytologic features did not correlate with clinical outcome for the histologically malignant melanomas evaluated. 42 Another study, which did not specifically define cellular pleomorphism, did not find any prognostic value for this feature in terms of remission length or survival time (P > .05) for canine oral malignant melanomas. 19 A third study evaluated the presence of giant cells as a prognostic factor but did not find a statistically significant association with survival. 45
Degree of Pigmentation
As previously noted, it can be difficult to accurately diagnose an amelanotic melanocytic neoplasm. Furthermore, objectively measuring the degree of pigmentation has been shown to be equally difficult. Nevertheless, several studies have attempted to examine the degree of pigmentation as a potential prognostic marker for canine melanocytic neoplasms to varying degrees. 1,2,5,19,20,28,32,36,42 Thus far, all of these studies have only subjectively determined the amount of pigmentation in melanocytic neoplasms and have not been able to identify threshold levels that could be statistically evaluated, with the possible exception of “high” pigment, assessed as at least 50% pigmented cells in one study. 2
Although it would intuitively seem that well-differentiated neoplasms would be more highly pigmented than less differentiated neoplasms, with accordingly better prognoses, several studies have not found a significant correlation between degree of pigmentation of melanocytic neoplasms from various body sites with survival outcomes. 5,19,20,32,36,42 Conversely, in a descriptive (nonprognostic) study consisting of well-differentiated, highly pigmented oral and lip neoplasms, 95% of the dogs were either alive at the end of the study (mean survival 23 months) or died of unrelated causes. 10 This finding was statistically corroborated in a separate study in which oral and lip melanocytic neoplasms with high pigment (subjectively assessed as ≥ 50%) had significantly longer survival times compared to all other pigment categories (none, low, or moderate). 2 Despite the fact that increasing pigment generally correlated with better survival in this study, only high pigment could be used to classify neoplasms (high negative predictive value). 2 Low or no pigmentation did not reliably predict a poor outcome. 2 Similarly, another report showed that pigmentation was an independent prognostic factor (P = .0007) for cutaneous melanocytic neoplasms and heavy pigmentation had a positive influence on survival time (P <.0001). 28 Pigmentation was subjectively scored on a scale from 0 (no pigment) to 2 (highly pigmented). 28 Even though these studies support an association between a high degree of pigmentation and longer survival times, this parameter cannot be reliably assessed, as there are currently no objective measures of this parameter nor validated cutoff points for comparative evaluation. In addition, the prognosis of neoplasms with lesser degrees of pigmentation cannot be accurately determined based on this parameter alone.
Junctional Activity
Junctional activity is a common feature of melanocytic neoplasms and an important feature to support the diagnosis of an amelanotic melanocytic neoplasm. A limited number of studies have examined the prognostic significance of junctional activity. One study demonstrated junctional activity as an independent prognostic factor (P = .0239) for cutaneous melanocytic neoplasms, and its occurrence was associated with a longer survival time (P = .0046). 28 In contrast, junctional activity negatively influenced survival for feet and lip neoplasms but was not statistically significantly associated with survival for skin neoplasms in another study. 45 Furthermore, in 2 studies of canine oral melanocytic neoplasms, an association of junctional activity with survival time was not found. 19,45 However, the results of these studies may be skewed, because poorly differentiated amelanotic melanomas without junctional activity might not be classified as melanomas on routine histopathologic examination and would not be included in the study set. Thus, there is no evidence of prognostic significance of junctional activity for oral melanocytic neoplasms, but its significance for dogs with cutaneous neoplasms is uncertain.
Intraepithelial Neoplastic Cells
Only one study has examined the prognostic significance of intraepithelial neoplastic cells, and no statistically significant association with survival was found for melanocytic neoplasms arising from any anatomic location. 45 One study reported that 9 of 10 dogs with canine oral malignant melanomas with osteocartilaginous differentiation had evidence of intraepithelial neoplastic cells; however, no conclusions were made in regard to this parameter. 40 Thus, based on currently available information, the presence of intraepithelial neoplastic cells does not appear to be a useful prognostic factor.
Ulceration
Many oral neoplasms are ulcerated as a result of perpetual trauma in the oral cavity regardless of cell of origin. One would therefore intuit that this factor would be of little prognostic importance for oral melanocytic neoplasms. For canine oral malignant melanomas, one study found that ulceration was of no prognostic value for remission length or survival time (P >.05). 19 Another study reported that 9 of 10 dogs with osteocartilaginous differentiation of oral malignant melanoma had evidence of ulceration. 40 There is some support for this factor as a prognostic marker for cutaneous melanocytic neoplasms in one study, however. 28 In that study, ulceration of cutaneous melanocytic neoplasms was associated with a significantly shorter survival time (P = .0023) and was shown to be an independent prognostic factor (P =.0065). 28 In contrast, another study did not find a correlation between ulceration and clinical outcome for lip, cutaneous, or nail bed melanocytic neoplasms. 42 However, ulceration was examined only in histologically malignant neoplasms in that study; 42 thus, there was no reference population.
Level of Infiltration/Invasion
Several studies have examined the level of infiltration or invasion of surrounding tissues for utility as a prognostic factor. 1,19,28,31,32,35 –37 When only malignant melanomas (oral and cutaneous) are evaluated, the level of stromal infiltration does not appear to be related to survival time. 19,31,42 Two studies reported that radiographic evidence of bony lysis did not affect outcome in dogs with oral malignant melanomas. 27,48 Another study, however, did find that a lack of bone lysis observed on skull radiographs was statistically associated with longer “times to first event” and longer survival based on both univariate and multivariate analysis for dogs with oral “malignant melanomas.” 35 Also, when this factor was evaluated in a series of melanocytic neoplasms that contained both benign and malignant neoplasms, it appeared to be related to survival time. 28,37 For cutaneous melanocytic neoplasms, one study showed that lesions strictly limited to the dermis, and therefore of shallow depth, had a positive influence on survival time (P < .0001), and deep infiltration was shown to be an independent prognostic factor (P = .0012). 28 Another study showed that invasive growth was significantly negatively correlated with survival (P = .024) of dogs and cats with melanocytic neoplasms from various anatomic locations. 37 This study had a short follow-up time of only 6 months. 37 The definition of infiltration in some studies is ill defined, and thus it is difficult to draw meaningful conclusions regarding this factor. 32
Vascular invasion, whether of blood or lymphatic vessels, is generally regarded as the gold standard for designating a melanocytic neoplasm as being malignant, 15,44 and lymphatic vessel invasion was reported to have prognostic significance in one study that included both cutaneous and oral melanocytic neoplasms. 31 In that study, the authors reported statistically significant differences in the course of disease between neoplasms with lymphatic invasion and those without, as demonstrated by survival curves (P = .0144). 31
Necrosis
The presence of necrosis was negatively related to survival in a study set of 389 melanocytic neoplasms containing both benign and malignant lesions from various locations, 45 but it had no correlation with survival among a set of 38 malignant melanomas from various locations in another study. 31 In the larger study, the P values for intralesional necrosis varied among the different sites (mouth, feet and lip, skin), but were significantly negatively correlated with survival for all sites. 45 The smaller study reported that the degree (extensive, moderate, or absence) of necrosis was not significantly related to survival. 31 However, no reference population was included, as only primary “malignant” melanomas were examined. 31 Thus, based on the results of the larger study, it would appear that the presence of necrosis does have prognostic significance, but this feature is difficult to measure objectively, specific cutoff values have not been determined, and its presence may represent different biological processes such as surface necrosis owing to trauma and deeper necrosis owing to ischemia.
Inflammation
The presence of inflammation is another potential prognostic parameter that is difficult to objectively measure. Studies that have examined this parameter have reported conflicting results. 19,31,45 One could postulate the presence of neoplasm infiltrating lymphocytes as being a favorable prognostic feature, as these lesions are frequently the target of specific active immunotherapy approaches. One study recorded the degree of lymphocytic and mononuclear cell infiltration within primary malignant melanomas from various body sites and compared this feature to Ki67 expression and survival. 31 The authors found that the presence of inflammation was not significantly related to either growth fraction or patient outcome. 31 Another study confirmed these findings and showed that the degree of inflammation, specifically lymphocytic infiltration, did not have prognostic value for remission length or survival time (P > .05) for canine oral malignant melanomas. 19 However, a large study showed that P values for deep inflammation varied among different anatomical locations (mouth, feet and lips, skin) of neoplasms, but were negatively and significantly correlated with survival in all sites. 45 This study evaluated both benign and malignant melanocytic neoplasms, whereas the 2 previously described studies included only malignant melanocytic neoplasms. Although deep intratumoral inflammation significantly influenced survival in a negative manner in the large study, 45 the significance of this feature requires confirmation in a prospective trial, and prognostic cutoff values should be established.
Margin Evaluation
Complete surgical excision is recommended for all melanocytic neoplasms; however, there is little, if any, support for this recommendation in the published literature. 48 Only 2 studies have examined margin evaluation as a prognostic factor, and both studies included only malignant melanocytic neoplasms. 19,42 Treatment by radical surgical excision of oral malignant melanomas resulted in longer remissions and survival times than did conservative surgical excision, without resection of underlying bone, in one of these studies; 19 however, extension of neoplastic cells to the surgical margins, or lack of extension, did not predict outcome for the lip, oral, or cutaneous neoplasms in either study. 19,42 Surgical removal of the digit does appear worthwhile, however, with 6 of 14 dogs with histologically malignant digital melanoma living at least 12 months in one study. 42 Henry et al. also showed that surgical removal of digital neoplasms had a positive influence on survival. 21 This study included 64 digital neoplasms of various cell lines of origin, only 10 of which, however, were classified as malignant melanomas. 21 Authors of another study, which included various types of oral neoplasms, including malignant melanomas, stated that careful preoperative assessment and gross evidence of complete surgical removal do not consistently ensure a low prevalence of local recurrence; however, they did report that lack of extension of neoplastic cells to the surgical margins was an important prognostic factor. 48 Based on the current incomplete and contradictory information in the literature, further investigation of this parameter in a prospective manner is clearly needed. Nevertheless, complete and wide excision is desirable whenever possible, despite the current inability to use this parameter prognostically.
Molecular Prognostic Parameters
Various molecular parameters that are routinely used for prognostication of different neoplastic entities in human medicine, especially those assessed via immunohistochemical evaluation, 23 have been tested in canine neoplasms. In general, molecular tests have the advantage of being semiquantitative and more objective than traditional histological assessment. Thus, they are generally less affected by interobserver variation. The following molecular parameters have been examined to varying degrees in the recent veterinary literature for their ability to predict the prognosis of canine melanocytic neoplasms: growth fraction measured by Ki67 labeling; cell-cycle phase index (PI) measured by either bromodeoxyuridine (BrdU) or proliferating cell nuclear antigen (PCNA) labeling; expression of Melan A/MART-1, S-100, vimentin, and neuron-specific enolase (NSE); expression of p53, PTEN, Rb, p21 (waf-1), and p16 (ink-4a); DNA ploidy; mast cell count (MCC); microvessel density (MVD); and expression of vascular endothelial growth factor (VEGF). These parameters are reviewed below.
Growth Fraction
Tumor growth fraction, assessed by immunohistochemical labeling for Ki67, has been evaluated as a prognostic factor in several recent studies. 2,28,31,37,38,40 Some studies erroneously refer to the Ki67 index as the proliferation index rather than indicating growth fraction. 28,31,40 In order to be most accurate, the Ki67 index in this article will always be referred to as growth fraction, or simply as Ki67 index.
Although each of these retrospective studies used different methods to measure and report Ki67 index, they all have shown that this parameter has prognostic significance for canine melanocytic neoplasms. 2,28,31,37,40 The Ki67 index has been shown to be significantly different between benign and malignant melanocytic neoplasms, with increasing values negatively correlated with survival. 2,28,31,37 One study demonstrated that the mean Ki67 index was significantly higher in oral melanocytic neoplasms from dogs that died within 1 year of diagnosis (Fig. 6) than the mean for neoplasms from dogs that were still alive at 1 year (P < .0005). 2 This study reported a negative correlation with survival by Cox regression analysis and efficacy as a diagnostic test by ROC analysis (area under the curve = 0.887, 95% confidence interval [CI] = 0.807-0.968). 2 ROC analysis was also used to establish a threshold value for Ki67, and the classification capabilities of this value were tested by Kaplan-Meier survival analysis. 2 The Ki67 labeling index for each neoplasm was determined by manually counting the number of positively labeled neoplastic cell nuclei within the area of a 1 mm 2 optical grid reticle at 400× using a standard light microscope and a cell counter. Five grid areas within the areas of highest labeling were counted and averaged to determine the Ki67 labeling index. Areas under regions of ulceration were avoided. The threshold value was statistically determined to be 19.5 positive nuclei per grid reticle. The sensitivity and specificity of this threshold value as a prognostic marker in that study population was 87.1% and 85.7%, respectively. The positive predictive values, with respect to outcome at 1 year, were 86.3% when classified by the Ki67 threshold, 82.5% when classified by nuclear atypia, and 79% when classified by mitotic index. Kaplan-Meier survival analysis showed that the survival curves for dogs with a Ki67 index < 19.5 and dogs with a Ki67 index ≥ 19.5 were significantly different (P <.0001). Ki67 correlated with mitotic index, nuclear atypia, and pigment (inversely) with respect to survival, and the areas transcribed by the ROC curves (measure of test efficacy) were similar for Ki67, nuclear atypia, and mitotic index. Ki67 and nuclear atypia had somewhat better test performance than mitotic index, as indicated by tighter 95% CI for the AUC. 2
A Ki67 threshold value capable of predicting survival rates in cutaneous melanocytic neoplasms has also been determined and is expressed as a percentage of positive labeling cells. 28 This study evaluated labeling for Ki67 in 68 cutaneous neoplasms, 12 of which were from the digit. Only 3 of these 12 digital neoplasms involved the ungual epithelium. In neoplasms with a focal immunoreactivity pattern, nuclei with weak to strong labeling for Ki67 were counted in these aggregated areas; in neoplasms with a diffuse staining pattern, counting was performed in randomly selected fields. 28 Similar to the study by Bergin et al., positive nuclei were counted at 400× with the help of an eyepiece graticule, and areas under regions of ulceration were avoided. 28 Counting was performed without knowledge of neoplasm outcome. One observer calculated ratios for both 500-cell counts and 1,000-cell counts; the second observer calculated a ratio for 1,000-cell counts only. There was no statistically significant difference between the percentages for 500 cells and 1,000 cells, and there was little interobserver variation for the 1,000-cell counts. Thus, counting 500 cells was considered adequate. Empirically, it was noted that none of the biologically benign neoplasms had an index greater than 15% (Fig. 7), so this threshold level was evaluated in regard to survival using Kaplan-Meier survival curves. There was a statistically significant (P < .0001) lower survival rate for dogs with neoplasms with a Ki-67 index greater than 15%. MI and Ki67 index were also shown to be significantly correlated (r = 0.596; P < .0001). Although MI and histological criteria were shown to be significantly correlated with survival, the percentage correct classification of the Ki67 index (97%) was higher than that of MI (91%) and histological criteria (93%). 28 It should be remembered that predictive values and percent correct classification are population-dependent parameters as opposed to sensitivity and specificity or AUC from an ROC curve, which are relatively population-independent. The latter were not reported for this study.
The 2 studies described above are by far the most clinically useful studies for canine oral 2 and cutaneous 28 melanocytic neoplasms, respectively. Both studies used manual counting techniques to determine Ki67 index that are easily applied in a diagnostic setting. Even though one study reported the Ki67 index as the number of positive nuclei per grid reticle 2 and the other study reported it as a percentage, 28 the authors of the first study suggested that the threshold levels were similar. The authors stated that for the grid reticle method, each grid field typically contains between 100 and 150 cells, and counting 5 grids would be roughly comparable to counting 500 cells. 2 Thus, the threshold for oral or lip melanocytic neoplasms of 19.5 or more positively staining cells out of a mean 125 cells/grid would be equivalent to 15.6%, similar to the 15% cutoff in the cutaneous melanocytic neoplasia study. 2,28 This assertion necessitates subsequent direct comparison. Nevertheless, these 2 studies have similar qualitative findings that are supported by high case numbers, extensive statistical analysis, and survival data.
Three additional studies have examined Ki67 index less rigorously but reached similar conclusions. 31,37,40 One study evaluated Ki67 index as a percentage of positive labeling nuclei per 600 cells in each of the 10 canine oral melanomas with osteocartilaginous differentiation included in their study. 40 It is unclear how they selected the 600 cells (eg, randomly vs within areas of highest staining). A threshold value of 30% for Ki67 index was empirically determined but was not statistically evaluated with respect to survival, likely because of the small dataset and descriptive nature of the report. 40 Two studies have employed computerized quantitative image analysis systems to evaluate Ki67 labeling, which may be of limited use in routine diagnostic settings. 31,37 One of these did not determine a specific threshold value, but instead, separated canine melanocytic neoplasms from various body sites into quartiles based on their Ki67 index and assessed the difference in survival between dogs with values above and below the median. 31 Dogs with values above the median value had statistically lower survival times than those with values below the median. 31 The study concluded that Ki67 index showed good predictive value, as shown by survival curves. 31 Ki67 index strongly correlated with mitotic counts (r = 0.706; P = .0001) and was statistically higher in oral malignant melanomas than in cutaneous ones. 31 High Ki67 index displayed a borderline correlation with lymphatic vessel invasion. 31 There was no correlation between histologic cell types (eg, epithelioid, spindloid) of malignant melanomas and survival, but there were some differences in growth fraction among neoplasms with different cell types. Epithelioid, spindloid, and mixed malignant melanomas had a significantly higher growth fraction than melanocytomas, whereas whorled-type malignant melanomas had a Ki67 index that was not statistically different with respect to benign lesions, and thus dogs with whorled-type malignant melanomas had a better prognosis compared with dogs with malignant melanomas of other cell types. 31 Age, neoplasm thickness, stromal invasion, degree of atypia, and presence of inflammation or necrosis were not significantly related to Ki67 index and survival time. 31 The second quantitative image analysis study evaluated growth fraction (Ki67 index) and phase index, as measured by PCNA labeling in melanocytic neoplasms from various body sites of both dogs and cats. 37 An increasing Ki67 index was significantly correlated with decreasing survival time (P = .027) via Cox regression analysis, but a specific threshold value for neoplasm growth fraction was not determined. 37 There was a significant correlation between Ki67 index and PCNA labeling (r = 0.69, P < .001). 37 Additionally, Ki67 index significantly correlated with the macroscopic infiltrative aspect of the neoplasm at the time of surgical treatment (P = .01), but no correlations were found with cell type or neoplasm size. 37
Based on the above evidence, Ki67 index is a statistically valid prognostic marker for canine melanocytic neoplasms. In a diagnostic setting, the Ki67 index for cutaneous melanocytic neoplasms should be determined according to the method of Laprie et al. (percentage positive cells of 500 cells counted), 28 and the Ki67 index for oral and lip melanocytic neoplasms should be determined according to the method of Bergin et al. (average number of positively staining neoplastic cells per 1 mm 2 optical grid reticle at 400×), 2 as outlined above. This index is especially helpful for melanocytic neoplasms that exhibit both prognostically favorable and poor histological parameters, or so-called “gray zone” cases. Furthermore, some neoplasms that exhibit “histological criteria of malignancy,” but a low growth fraction have been identified and shown to have longer survival times than would have been expected based on histological features alone. 2,28 One study identified a subgroup of cutaneous neoplasms that were classified as benign based on the strict histological criteria described in that study, but that exhibited malignant behavior. 45 In another study, one subset of lip neoplasms with a histologically benign appearance exhibited malignant behavior, and a subset of lip neoplasms with a malignant histological appearance exhibited benign behavior. 42 Ki67 index is more objective and less subject to interobserver variation than histological atypia and mitotic index, although this characteristic should ideally be tested prospectively and with direct comparison of results from different observers.
Phase Index
Only 2 studies have examined cell cycle phase index (PI) as a prognostic indicator of canine melanocytic neoplasms and neither used methods that could easily be applied in a diagnostic setting. 37,52 One study evaluated the BrdU labeling index as a measure of PI in a set of 23 oral neoplasms of different types, including 1 malignant melanoma. 52 This n = 1 for melanocytic neoplasms does not allow for any conclusions to be drawn. Since evaluation of BrdU requires intravenous injection of BrdU to dogs prior to surgery for mass removal, this method is impractical for routine clinical use. Another study used immunohistochemical labeling for PCNA and quantitative image analysis to determine the PI in 20 random fields. 37 Because of its long half-life, PCNA can be detected in cells throughout the cell cycle, but it is produced only in late G1 and throughout the S phase of the cycle. 6,37 The authors found that the PI differed significantly between the histologically malignant and benign melanocytic neoplasms from various sites of both dogs and cats (P <.001). 37 The correlation between Ki67 and PCNA labeling was significant (r = 0.69, P < .001), but unlike Ki67, PCNA labeling was not statistically related to survival duration (P =.445). 37
PI appears to have some prognostic significance for melanocytic neoplasms; however, no specific conclusions or recommendations can be made from these limited studies. Additional investigation of this parameter using a larger number of canine neoplasms from both oral and cutaneous sites, with complete survival data, is needed to more fully evaluate the prognostic potential of PI. Also, this parameter should be measured using a method that can easily be applied in a diagnostic setting, such as determining immunohistochemical labeling for PCNA in a manner similar to the methods used for growth fraction.
Expression of Melan A/MART-1, S-100, Vimentin, and NSE
Immunohistochemical labeling for Melan-A/MART-1, S-100, vimentin, and NSE has been used in the diagnosis of melanocytic neoplasms, 8,9,12,25,36,43 but their value as prognostic markers is rather limited. Only one study has evaluated these antibodies for their potential use as prognostic markers for canine melanocytic neoplasms. 25 There did not appear to be any correlation between the labeling properties of vimentin, S-100, or NSE and the behavior of the melanocytic neoplasms in this study. The authors did, however, report that intensity of Melan A/MART-1 expression was significantly (P < .03) correlated with a favorable outcome. 25 This study used a small number of cases and did not perform survival analysis. In contrast, another study found that strong labeling for Melan-A in a primary neoplasm did not predict benign behavior of that neoplasm. 36 Based on our current knowledge, it seems unlikely that labeling for any of these markers can be used to predict the prognosis of canine melanocytic neoplasms. 44
Expression of p53, PTEN, Rb, p21 (waf-1), and p16 (ink-4a)
Two studies have examined the expression of p53, 26,39 and one also examined the expression of PTEN, Rb, p21, and p16 in relatively small numbers of canine melanocytic neoplasms from various sites. 26 Abnormalities in the expression or localization of these proteins were found in a proportion of the neoplasms, but there were no significant differences between benign and malignant neoplasms and there was no correlation with survival. 26,39
DNA Ploidy
Few studies have examined DNA ploidy for prognostic significance in canine melanocytic neoplasms. Differences in DNA ploidy and other morphometric variables have been shown between benign and malignant melanocytic neoplasms, but no correlation with survival time has been demonstrated. 4,38 The methodology is not easily applied for melanocytic neoplasms, since heavily pigmented neoplasms are difficult to evaluate and accurate interpretation of acceptable histograms is difficult. 4 Furthermore, assessment of DNA ploidy is labor intensive and not easily applied in a diagnostic setting. 4 Thus, use of this parameter for prognostication is not recommended at this time.
Mast Cell Count and Microvessel Density
Only one study has examined microvessel density (MVD) and the number of mast cells within the neoplasms (mast cell count, MCC) as prognostic parameters for canine melanocytic neoplasia. 32 The study concluded that MCC and MVD were significantly correlated and that high MCC and MVD in canine melanocytic neoplasms were associated with a poor prognosis. 32 Cutoff values of median MCC and MVD were used to divide dogs into high and low groups for both parameters and compared to survival for a portion of the study population. Statistically, MCC was found to be a better prognostic marker than MVD (p < .01); however, both MCC and MVD at the lesion periphery, as well as centrally, were shown to be independent prognostic factors. 32 The study was based on a small sample population, follow-up data were available for only 18 cases, and all 18 neoplasms exhibited malignant behavior. A reference population of benign neoplasms was not included in the study. Further evaluation of MCC and MVD, with regard to survival outcomes, must be performed using a larger study population that includes both benign and malignant neoplasms, further stratified by anatomic neoplasm location, before substantial conclusions can be made regarding their utility as prognostic parameters.
Vascular Endothelial Growth Factor
In human cancer patients, high plasma vascular endothelial growth factor (VEGF) levels have been correlated with a poor prognosis. 22,24 One study measured plasma VEGF in 70 dogs with various neoplasms, only 6 of which were of melanocytic origin. 51 The authors found a significant difference in plasma VEGF levels between the various types of neoplasms. 51 The lowest levels were found in dogs with epulides and the highest levels were found in dogs with oral melanocytic neoplasms. 51 There was no comparison of plasma VEGF to survival. No conclusions can be drawn regarding the prognostic significance of this marker for canine melanocytic neoplasms at this time, though evaluation of this parameter for its prognostic significance may be worthwhile.
Conclusions
Based on review of the current literature in terms of the Recommended Guidelines for the Conduct and Evaluation of Prognostic Studies in Veterinary Oncology, 49 we identified the prognostic parameters that have the most statistically demonstrable prognostic significance based on published survival times for canine cutaneous and digit melanocytic neoplasms and for oral and lip melanocytic neoplasms (Table 2) to date. In our recommendations, a favorable prognosis relates to expected survival times longer than 1 year and a poor prognosis relates to an expected death as a result of melanocytic neoplasia within less than 1 year post diagnosis for all melanocytic neoplasms. For specific survival times and clinical outcomes for each parameter, the reader should refer to the sections in this article specific to that parameter and the original study being cited. Only the most significant factors are discussed in the following.
When attempting to predict the biological behavior of a particular melanocytic neoplasm, accurate determination of melanocytic origin of the neoplasm is crucial prior to prognostication. Since amelanotic melanocytic neoplasms are difficult to distinguish from soft tissue sarcomas, which commonly exhibit an aggressive biological behavior, inaccurate differentiation will influence the validity of any prognostic study. According to the current literature, IHC labeling with a combination of Melan-A, PNL2, TRP-1, and TRP-2 as single antibodies, or as a cocktail, provides the highest sensitivity for detecting amelanotic melanomas while maintaining 100% specificity. 8,9,12,25,36,41,43,44,46 Other commonly used antibodies, such as S-100 or MiTF, are highly sensitive for detecting amelanotic melanocytic neoplasms, but they lack the specificity necessary to provide an accurate diagnosis. 9,18,34,41,43,44
In general, oral melanocytic neoplasms have a worse prognosis than cutaneous neoplasms, and those on the lip or digit have a worse prognosis than cutaneous melanocytic neoplasms at other sites. However, as several studies 2,10,28,42,45 have demonstrated, location alone cannot be used to predict prognosis for a given patient, as there are definitely exceptions to all generalizations. Some factors have prognostic significance for neoplasms of a particular site, but not for neoplasms arising at other locations (Table 1). Distant metastasis is indicative of a poor prognosis for all melanocytic neoplasms regardless of their location. It would also seem that lymph node metastasis would be associated with a poor prognosis; however, only 2 studies have compared this factor to prognosis, and both examined only oral “malignant” melanomas. 19,35 Neither study identified any association between regional lymph node metastasis and remission length, “time to first event,” or survival time (P > .05), but no reference population was included. 19,35 Thus, additional studies that include benign and malignant neoplasms from various anatomic locations are needed for definitive conclusions regarding lymph node metastasis as a prognostic factor.
Although there is some reported evidence that neoplasm size or volume has prognostic significance, because of conflicting results in the literature, recommendations regarding this parameter cannot be made at this time. Additional studies using a statistically determined threshold value with comparisons to complete survival data are needed before significant conclusions can be drawn.
Histological classification of canine melanocytic neoplasms as benign or malignant has generally been associated with clinical outcome and survival. However, “morphologic classification” as an individual prognostic parameter is difficult to evaluate in the current literature, as different classification criteria have been applied.
Histological evaluation of nuclear atypia has a high positive predictive value for epithelioid-predominant melanocytic neoplasms 2,45 and for spindloid neoplasms with sufficiently observable nuclear detail 2 in terms of prognosis. Significant nuclear atypia is associated with a poor prognosis, and minimal or no atypia is associated with a favorable prognosis; 2,45 however, assessment of this parameter is subject to interobserver variation, resulting in different specificities and sensitivities. Rigorous description and standardization of the criteria for nuclear atypia provide some measure of reproducibility. 2,45 Therefore, the degree of nuclear atypia should be evaluated for melanocytic neoplasms based on the criteria described by Spangler and Kass (2006), as follows:
Well-differentiated or typical melanocytic neoplastic cells
45
small nucleus single, centrally oriented nucleolus minimal clumping of chromatin may have condensed strands of nuclear chromatin extending from the nucleolus to the nuclear membrane condensation of chromatin along the inner surface of the membrane cells that lack a nucleolus have fine and evenly dispersed chromatin at the periphery of the nucleus
Poorly differentiated neoplastic cells (atypical)
45
larger nucleoli of less-regular shape that are eccentrically located in the nucleus often multiple nucleoli in some cases, multiple nucleoli are haphazardly connected to the inner surface of the nuclear membrane by thin strands of chromatin and give the appearance of a coarsely vacuolated nucleus.
Although the original evaluation was based on an incremental scale from 1 to 10, signifying the subjectively estimated percentage of nuclei involved,
45
such a rigorous grading scale is unlikely to be applied in a routine diagnostic setting. A statistically similar, but more practical method for oral and lip melanocytic neoplasms is to use a threshold value for semiquantification of nuclear atypia of ≥ 30%.
2
The MI should be determined by counting the number of mitotic figures in 10 consecutive hpf commencing in the area of highest mitotic activity for oral and lip neoplasms 2,45 and in random fields for cutaneous neoplasms. 28 Areas under ulceration should be avoided. MI should be evaluated in bleached tissue sections to ensure that the melanin within the cell does not obscure the mitoses, and mitotic figures need to be distinguished from small chromatic fibroblast nuclei within the interstitium. The MI should be reported as the number of mitoses per 10 hpf. 2,15,28,45 Dogs with cutaneous neoplasms that have an MI ≥ 3 per 10 hpf 5,28 and dogs with oral or lip neoplasms that have an MI ≥ 4 per 10 hpf 2 are expected to have shorter survival times. At least 50% of dogs with cutaneous melanocytic neoplasms with an MI ≥ 3 per 10 hpf were dead by 7 months in 2 studies. 5,28 Approximately 80% of dogs with oral or lip melanocytic neoplasms with an MI ≥ 4 per 10 hpf were dead by 1 year in another study. 2 Dogs with melanocytic neoplasms with an MI less than those values, for each respective site, are expected to have longer survival times. 2,5,28 Less than 10% of dogs with cutaneous melanocytic neoplasms with am MI < 3 per 10 hpf died within a 2-year follow-up period in 2 studies. 5,28 Less than 10% of dogs with oral or lip melanocytic neoplasms with an MI < 4 per 10 hpf died within a 1-year follow-up period in one study. 2
The presence of inflammation and/or necrosis should not be used to determine prognosis. Although both have been associated with a poor prognosis for melanocytic neoplasms at any site, 45 both are difficult to objectively evaluate, and no cutoff values have been determined.
Pigmentation should be evaluated but not used as a sole predicting factor. Although a high degree of pigmentation is associated with a favorable clinical outcome in both cutaneous and oral melanocytic neoplasms, 2,10,28 outcome is not predictable in oral neoplasms with moderate, low, or no pigmentation. 2 When paired with assessment of nuclear atypia, the degree of pigmentation has greater prognostic significance for oral and lip melanocytic neoplasms. 2 Dogs with highly pigmented, well-differentiated oral or lip neoplasms were found to have longer post diagnosis survival times that approached 2 years. 10
One study demonstrated lymphatic invasion as a negative prognostic factor for oral and cutaneous melanocytic neoplasms. 31 Even though lymphatic invasion was not specifically evaluated as a prognostic factor for melanocytic neoplasms of the lip or digit in that study, 31 vascular invasion is considered by some as the best indicator of malignancy. 15
The presence of ulceration has been associated with a poor prognosis in one study, which included cutaneous neoplasms from various sites including 12 digital neoplasms. 28 Deep infiltration/invasion has also been shown to be associated with a poor prognosis for cutaneous melanocytic neoplasms, 28 and possibly for oral melanocytic neoplasms. 35,37 Junctional activity has been associated with shorter survival times for melanocytic neoplasms arising on the feet or lips, 45 but its significance for cutaneous melanocytic neoplasms at other sites is uncertain because of conflicting reports. 28,45
Because of the inherent subjectivity in histological evaluation, we recommend that more than 1 parameter be considered to classify melanocytic neoplasms as benign or malignant. If these features conflict with one another, a neoplasm should be diagnosed as a melanocytic neoplasm and both the favorable and the poor prognostic factors should be discussed.
No criteria have yet been established to allow for completely objective assessment of histological parameters for prognostic purposes in a routine laboratory setting. In contrast, the Ki67 index, as a measure of the growth fraction of neoplastic cells, is a highly objective test that has a higher predictive value than histologic criteria. 2,28 Assessment of Ki67 index is recommended for all melanocytic canine neoplasms and is especially useful for cases with conflicting histological parameters, or so-called “grey zone” cases. Based on the current literature, Ki67 index has been statistically shown to have prognostic significance using 2 separate methods for oral and lip melanocytic neoplasms and for cutaneous and digit neoplasms. For oral and lip melanocytic neoplasms, the Ki67 index should be reported as the average number of positively labeled nuclei per 1 mm 2 grid reticle at 400× by counting the number of positive nuclei in 5 grid areas. 2 Neoplasms with a Ki67 index ≥ 19.5 are expected to have a poor prognosis (Fig. 6), and neoplasms with an index less than 19.5 are expected to have a favorable prognosis. 2 For cutaneous melanocytic neoplasms, the Ki67 index should be reported as a percentage of positive-staining nuclei per 500 cells. 28 Neoplasms with a Ki67 index ≥ 15% are expected to have a poor prognosis, and neoplasms with a Ki67 index <15% (Fig. 7) are expected to have a favorable prognosis. 28 For both methods, counting should commence in an area with high labeling, but areas of inflammation and areas underlying ulceration should be avoided. 2,28 Although Bergin et al. reported Ki67 index as the average number of positive nuclei per 1 mm 2 grid reticle, Laprie et al. also used a grid reticle for assistance in counting 500 cells. It would be ideal to be able to report Ki67 index in the same manner regardless of location; however, the current literature does not allow for this. Thus, unless additional prognostic studies are performed to determine statistically valid threshold values using one or both of these methods in melanocytic neoplasms from various sites, the Ki67 index should be determined as the average number of positive nuclei per 1 mm 2 grid reticle for oral and lip melanocytic neoplasms and as the percentage of positive nuclei in 500 cells for cutaneous and digit melanocytic neoplasms. The use of grid reticles is becoming more common in diagnostic pathology because it allows for standardization of field area between different microscopes. They are inexpensive and can easily be inserted into most microscopes. Thus, diagnostic laboratories should consider acquiring at least one optical with a grid reticle that can be shared between pathologists. Assessing the average number of positive cells per grid within 5 grid areas is less labor intensive and less prone to error compared to counting 500 neoplastic cells.
Although it is impractical to accurately predict, on an individual basis, the biological behavior of melanocytic neoplasms by applying elaborate numeric criteria to specific histological features, 45 evaluation of nuclear atypia and MI, in combination with Ki67 index and clinical features, will maximize the percentage of correctly classified neoplasms. An accurate prognosis is becoming more and more important in veterinary oncology, as various treatment options are now available for specific types of neoplasms and many more clients are pursuing these options. These treatments are: often costly; may involve radical surgeries or costly radiotherapy, chemotherapy, or immunotherapy; and may have significant or yet unknown side effects. Thus, it is important to identify which melanocytic neoplasms require additional therapies and which will likely be cured by excision alone.
None of the previously published studies met all the standards outlined by the Recommended Guidelines for the Conduct and Evaluation of Prognostic Studies in Veterinary Oncology. 49 We based our recommendations regarding prognostic factors for canine melanocytic neoplasms on the studies that came the closest to meeting these standards. Very few studies had adequate sample numbers, strict selection criteria, and detailed statistical analysis with comprehensive clinical outcome and survival data. A major difficulty in veterinary prognostic studies is to accurately evaluate survival data, as many pets are euthanized, rather than die, as a result of neoplastic disease. Often, it is even difficult to associate the cause of euthanasia with the progression of neoplastic disease. Some dogs with melanocytic neoplasms may be euthanized as a result of deterioration of health secondary to neoplastic disease, others may be euthanized because of management issues in patient care that the owner is reluctant or unable to manage, and others may be euthanized for completely unrelated medical comorbidities. It is also often difficult to determine a cause of death, even in dogs that die a natural death, as necropsy is rarely performed. Additionally, none of the published studies specifically stated the expected magnitude of each factor’s contribution or the expected prognosis of the population. Also, few studies specifically defined a reference population. In some cases, this information could be inferred by the reader. Another major pitfall is that many of the studies that have examined various treatments for dogs with melanocytic neoplasms have failed to use rigorous inclusion and exclusion criteria and well-defined end point evaluation. Thus, few conclusions can be drawn from those studies. All of the studies examined here were retrospective. Clearly, well-designed prospective studies are greatly needed to confirm the validity of the described prognostic parameters. Future canine melanocytic neoplasm prognostic studies should use the Recommended Guidelines for the Conduct and Evaluation of Prognostic Studies in Veterinary Oncology 49 to aid in study design. An effort should be made to design more rigorous prospective trials, especially to evaluate specific treatment protocols that would allow a more targeted therapy approach.
Footnotes
Acknowledgements
This study represents an initiative of the American College of Veterinary Pathologists' Oncology Committee. This manuscript has also been reviewed and endorsed by the World Small Animal Veterinary Association. We thank both organizations for their support and guidance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
