Abstract
Histologic diagnosis of less well-differentiated cases of canine extramedullary plasmacytomas (CEMPs) may require immunohistochemical confirmation to discriminate these tumors from other round cells tumors including lymphoma, cutaneous histiocytoma, and amelanotic melanomas. CEMPs are characterized by widespread immunoreactivity for multiple myeloma 1 (MUM1) antigen and λ light chains, while the melanocytic marker melan-A has been reported to yield negative results. Here, 33 randomly selected CEMPs, 20 melanocytomas, and 20 malignant melanomas were immunohistochemically tested for MUM1, melan-A, and PNL2. In addition, CEMPs were examined for PAX5, E-cadherin, CD3, CD18, CD20, S100, as well as λ and κ light chain immunoreactivity. All CEMPs were characterized by labeling for MUM1 and λ light chain, as well as variable immunopositivity for the remaining antibodies. Notably, 13 cases of CEMPs (39.4%) exhibited immunolabeling for melan-A. Melanocytic tumors immunolabeled for melan-A (40/40; 100%) and PNL2 (34/40; 85%). An unexpected cytoplasmic immunoreactivity for MUM1 was observed in 2 melanocytic tumors. Summarized, MUM1 or melan-A immunomarkers alone are not sufficient to differentiate between CEMPs and amelanotic melanomas and should be part of a larger immunopanel including λ light chain, CD20, and PNL2.
Canine extramedullary plasmacytomas (CEMPs) are nonencapsulated, nonpigmented round cell tumors. 36 CEMPs represent a clonal expansion of neoplastic plasma cells not arising from the bone marrow. They are most commonly found in the skin and less frequently in the oral cavity and the gastrointestinal tract.7,30,49 Histologically, CEMPs are characterized by the presence of neoplastic plasma cells with varying degrees of maturity and cell polymorphism, supported by small amounts of fibrovascular stroma and the inconsistent presence of primary amyloid.36,39 Frequently, bi- or multinucleated tumor cells are found. Moreover, different variants of CEMPs with similar clinical relevance have been described including mature, cleaved, hyaline, asynchronous, and polymorphous-blastic subtypes.7,30 Because a subset of these variants is less well-differentiated, immunohistochemical examinations may be needed for a final diagnosis to discriminate CEMPs from other nonpigmented round cell tumors like plasmablastic lymphoma, histiocytoma, histiocytic sarcoma, or amelanotic melanocytic tumors.
CEMPs are known to show widespread immunoreactivity for multiple myeloma 1/interferon regulatory factor 4 (MUM1/IRF-4) and additional labeling for λ (lambda) light chain.3,35,38 Both represent valuable markers to identify plasma cell tumors using immunohistochemistry. Furthermore, variable immunoreactivity for CD18, CD20, and CD79a has been reported.25,35 In contrast, CD3 and PAX5 for the detection of T- and B-lymphocytes, respectively, as well as melanoma antigen A (melan-A), a marker for cells of melanocytic origin, are reported to yield negative results in canine plasma cell tumors;28,33,48 thus, differentiating these tumors from blastoid lymphoma and nonpigmented melanoma. Moreover, canine melanomas are reportedly immunonegative for MUM1, 35 contrary to their human counterparts.26,45
In dogs, melanocytoma is frequently found in the skin, whereas malignant melanoma most often is present in the oral cavity and on the digits.27,42 Malignant melanomas and CEMPs exhibit highly different clinical behaviors. Oral malignant melanomas are characterized by a poor prognosis, 2 while oral CEMPs are usually associated with a good prognosis after complete surgical excision.31,49 Therefore, differentiation between these tumors is of great importance, and reliable markers are necessary in cases where further immunohistochemical examinations are required. In this study, we report the unexpected finding of variable immunoreactivity of CEMPs for melan-A, a marker routinely used to identify neuroectodermal neoplasms, especially melanocytic tumors, in dogs.33,34
Materials and Methods
Case Selection
All tumors included in this study represent surgical biopsies submitted to the Department of Pathology, University of Veterinary Medicine, Hannover, Germany, from 2021 to 2022. 33 CEMPs (cases 1-33) were selected randomly and manually from the archive of the Department of Pathology. The tumor location, age, sex, and further clinical data of the dogs are summarized in Table 1. Furthermore, 20 randomly and manually selected melanocytomas (cases 34-53; skin, n = 18 and eyelid, n = 2) and 20 randomly and manually selected malignant melanomas (cases 54-73; oral cavity, n = 13; lip, n = 3; skin, n = 2; digit, n = 1; and eyelid, n = 1) were included in the investigation for comparison.
Signalment and localization of investigated canine extramedullary plasmacytomas (CEMPs), melanocytomas, and malignant melanomas.
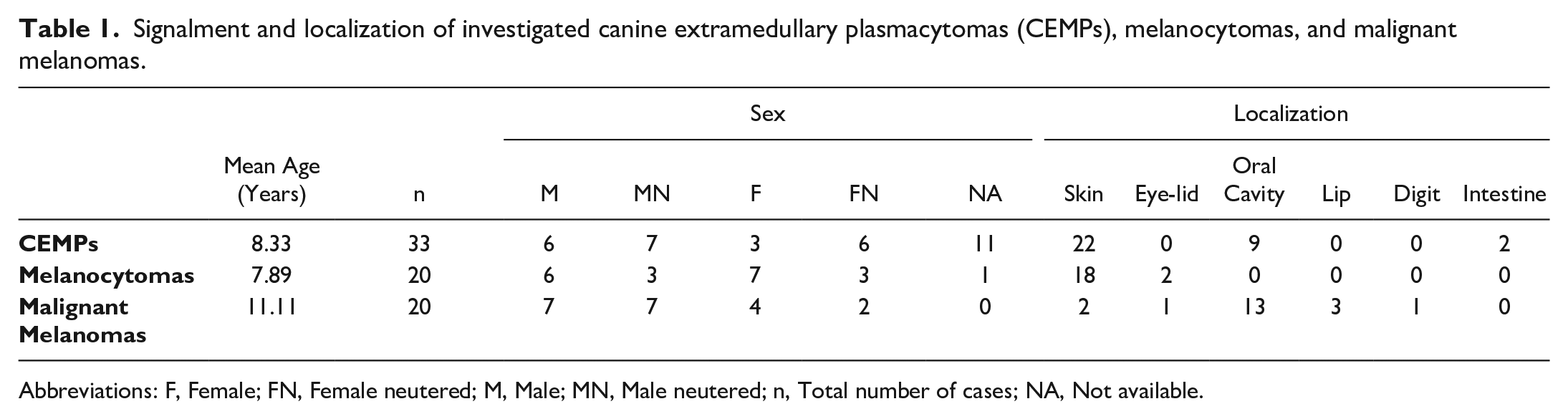
Abbreviations: F, Female; FN, Female neutered; M, Male; MN, Male neutered; n, Total number of cases; NA, Not available.
Histology, Immunohistochemistry, and Double-Labeling Immunofluorescence
Histologic and immunohistochemical examinations were performed by 1 board-certified pathologist and 3 board-registered residents using formalin-fixed, paraffin-embedded tissues. For histologic evaluation, 2- to 3-µm-thick paraffin sections were routinely stained with hematoxylin and eosin. Histologic classification of extramedullary plasmacytomas including mature, cleaved, hyaline, asynchronous, and polymorphous-blastic variants was performed as previously described.23,25,30,35 Mitotic count was evaluated in 2.37 mm2 (10 high-power fields [40×] using oculars with a field number of 22). The mitotic count was determined in areas of the tumor where the highest density of mitotic figures was present. There, high-power fields were randomly selected. Areas of necrosis or hemorrhage were not considered for the determination of the mitotic count. Cutaneous melanocytic tumors with > 3 mitoses in 2.37 mm2 were considered malignant, according to the World Health Organization classification of epithelial and melanocytic tumors of the skin in domestic animals. 12 Oral melanomas with > 4 mitoses in 2.37 mm2 were considered malignant, according to published literature. 41
Immunohistochemical examination of the CEMPs was carried out using monoclonal (directed against MUM1, melan-A, PNL2, canine CD18, and PAX5) and polyclonal antibodies (directed against CD3, CD20, S100, E-cadherin, and λ and κ [kappa] light chains; Table 2). For negative controls, primary antibodies were replaced by sera of the species from which the respective antibody was derived (mouse or rabbit). For each immunohistochemical assay, positive controls were included that were known to exhibit immunoreactivity for the respective marker. Biotinylated goat anti-mouse, goat anti-rabbit, and goat anti-rat secondary antibodies (1:200; Vector Laboratories, Burlingame, USA), the avidin-biotin-peroxidase-complex method (PK-6100; Vector Laboratories), and 3,3′-diaminobenzidine tetrahydrochloride as chromogen (DAB; Sigma-Aldrich, Chemie GmbH, Munich, Germany) were further applied to visualize the reaction. Immunolabeling was considered positive when the following immunoreactive distribution patterns were observed: nuclear and cytoplasmic for MUM1; nuclear for PAX5; membranous and cytoplasmic for melan-A; cytoplasmic for PNL2, S100, and λ and κ light chains; and membranous for E-cadherin, CD3, CD18, and CD20. In all tumors that were suspected to represent a CEMP based on hematoxylin and eosin staining, immunoreactivity for MUM-1 and λ light chain and absent immunolabeling for CD3 and PAX5 of neoplastic cells were mandatory for a final diagnosis and their inclusion in this study.
Antigenicity, clone, host species, pretreatment, dilution, supplier, and order number of primary antibodies used for immunohistochemistry.
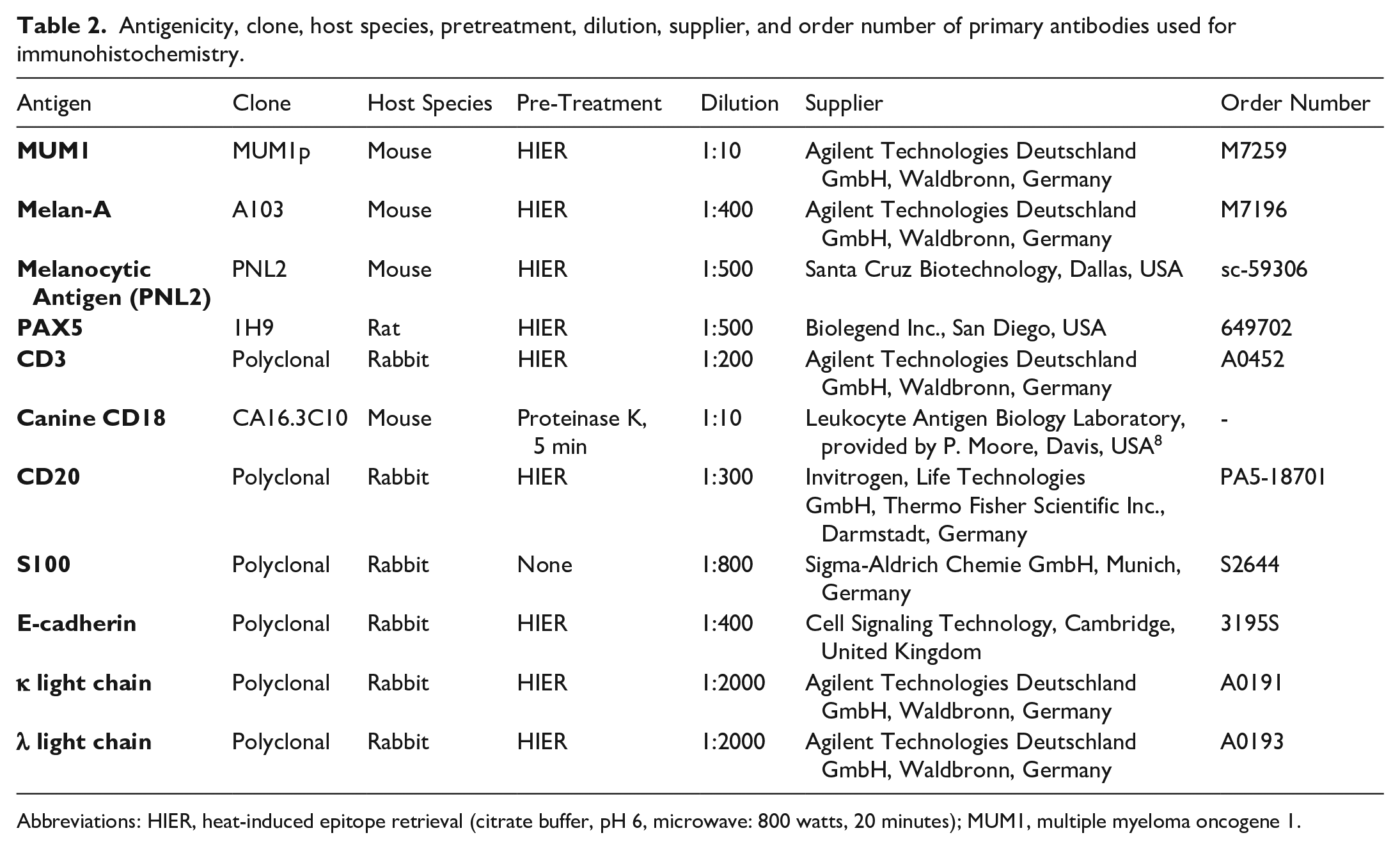
Abbreviations: HIER, heat-induced epitope retrieval (citrate buffer, pH 6, microwave: 800 watts, 20 minutes); MUM1, multiple myeloma oncogene 1.
To examine the co-labeling of MUM1 and melan-A antigen in neoplastic CEMP cells, double-labeling immunofluorescence was performed using the antibodies mentioned above (Table 2). Briefly, following the incubation with the first primary antibody (MUM1) and consecutive administration of cyanine 3-conjugated secondary antibody (Cy3), the second primary antibody (melan-A) and Alexa Fluor 488-conjugated secondary antibody (goat anti-mouse each [1:200]; Jackson ImmunoResearch Europe Ltd, Ely, UK) were applied. Sections were counterstained using bisbenzimide according to previously published protocols for formalin-fixed, paraffin-embedded tissue.16,24
For the examination of melanocytic tumors, immunohistochemistry was performed using primary antibodies directed against MUM1, melan-A, and PNL2. Due to endogenous pigment accumulation in these tumors, visualization of immunolabeling was achieved by applying HistoGreen (Linaris Biologische Produkte GmbH, Dossenheim, Germany) instead of DAB as chromogen. 20
The immunoreactivity of neoplastic cells in CEMPs was scored semi-quantitatively using 3 modified categories as previously published (mild, 6% to 29% positive cells; moderate, 30% to 59% positive cells; high, ≥60% positive cells; negative ≤5% positive cells). 35
Data Visualization
For data visualization, GraphPad Prism 9.0 (GraphPad Software, San Diego, CA, USA) was used. Generated and analyzed raw data of this study are summarized in Supplemental Table S1 for extramedullary plasmacytomas and Supplemental Table S2 for melanocytic tumors.
Results
Histology
In CEMPs, neoplastic cells were arranged in sheets and nests supported by small amounts of fibrovascular stroma. The majority of tumor cells were medium to large in size (approximately 12 to 20 µm in diameter), round, and had moderate amounts of eosinophilic cytoplasm with indistinct cell borders. They contained eccentric to paracentral, round, medium-sized to large nuclei with variably stippled chromatin, and 1 to 2 small nucleoli. Sometimes, a perinuclear halo was detectable. There was moderate anisocytosis, anisokaryosis, and variation in nucleolar size. Mitotic figures were rarely detected, ranging from 0 to 3 mitotic figures in 2.37 mm2. Furthermore, variable but, in general, few numbers of bi- or multinucleated tumor cells were present. Infrequently, the surrounding tissue was compressed by the neoplastic mass. CEMPs were classified as mature (12/33, 36%), cleaved (8/33, 24%), asynchronous (7/33, 21%), hyaline (1/33, 3%), and polymorphous-blastic (5/33, 15%).
Melanocytic tumors were nonencapsulated and consisted of highly cellular neoplastic infiltrates of varying cell shapes, ranging from round to polygonal to spindloid. Tumor cells were arranged in sheets, cords, whorls, and nests. The cells were medium-sized (approximately 12 to 18 µm in diameter) and consisted of moderate amounts of eosinophilic cytoplasm with variable amounts of brownish to black granular pigment and indistinct cell borders. They contained round to oval, paracentral, euchromatic nuclei and 1 to 2 small-to-medium-sized nucleoli. In melanocytomas, there was mild to moderate anisocytosis, anisokaryosis, and variation in nucleolar size, while in malignant melanomas, a higher degree of cellular atypia was present. In malignant cutaneous melanomas, the mitotic count was > 3 mitoses in 2.37 mm2 (ranging from 4 to 12 mitoses/2.37 mm2) whereas in oral melanomas, a mitotic count > 4 in 2.37 mm2 (ranging from 5 to 18 mitoses/2.37 mm2) was detected.
In 12 of the 40 melanocytic tumors (30%), only minimal or no pigment was present (Supplemental Table S2). In these cases, 10/12 (83%) tumors were malignant melanomas and 8/12 (67%) were detected in the oral cavity. Melanocytomas showed predominantly moderate-to-abundant pigmentation, with 2 amelanotic exceptions (cases 42 and 47).
Immunohistochemistry and Double-Labeling Immunofluorescence of CEMPs
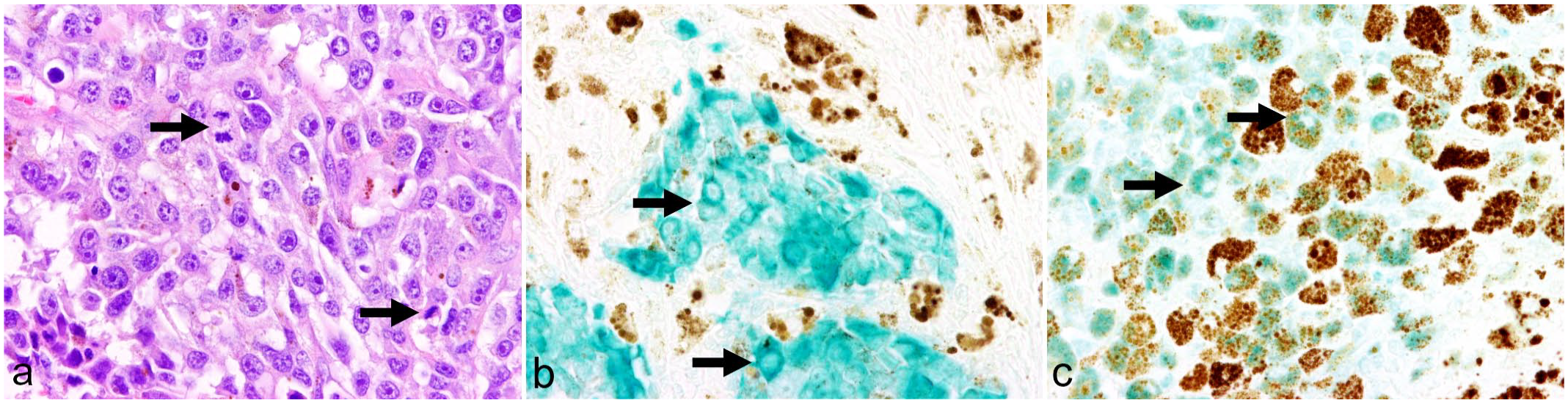
For all CEMPs, positive immunolabeling for MUM1 and λ light chain and no immunoreactivity for CD3 and PAX5 were observed in neoplastic cells. Thirteen out of 33 (39%) CEMPs showed variable immunoreactivity for melan-A. The immunohistochemical antigen labeling was membranous or cytoplasmic (Fig. 1a–c). Melan-A immunoreactivity of CEMPs was observed independently of the histologic subtype classification or tumor location (Supplemental Figures S1 and S2).
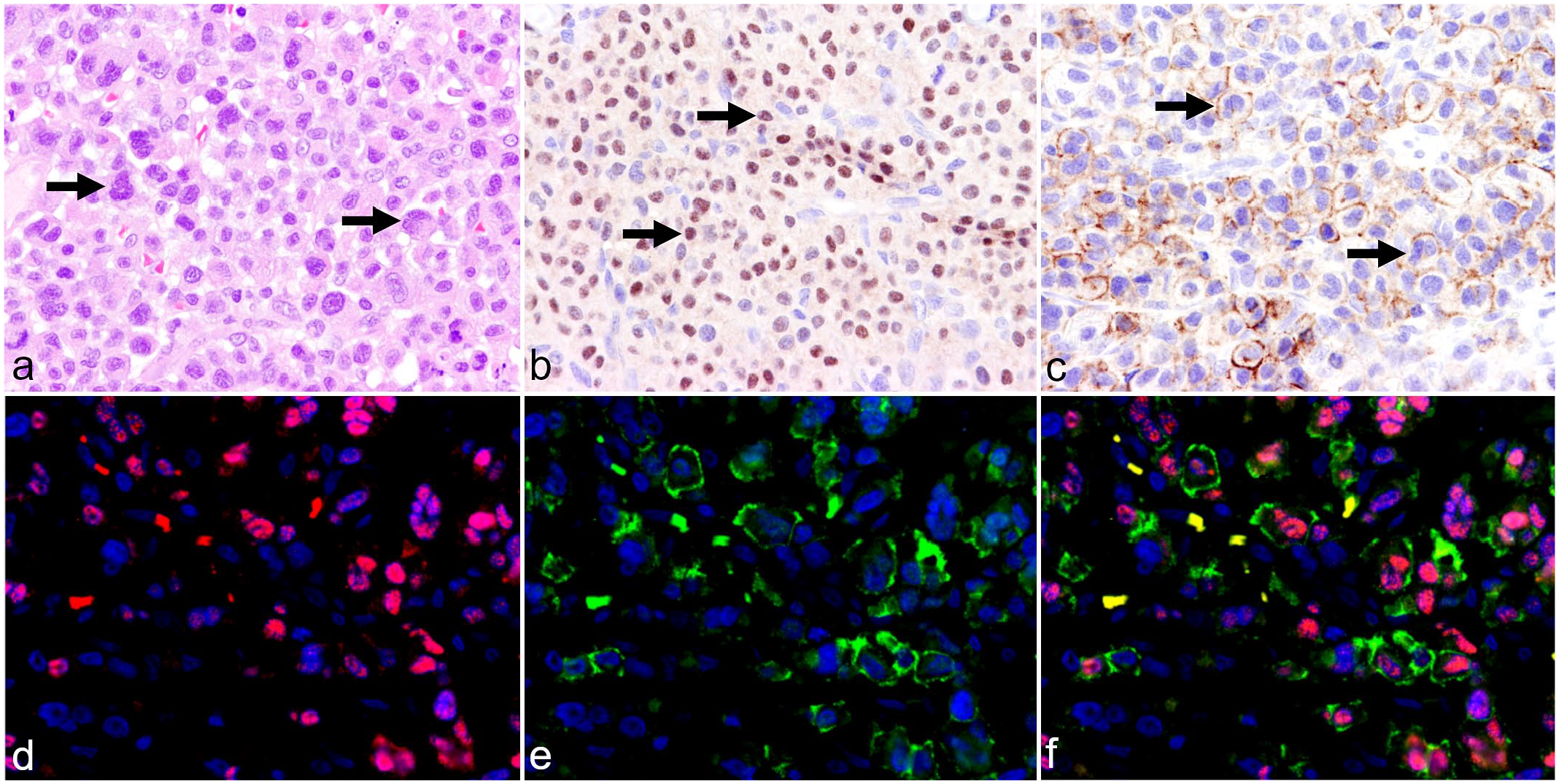
Plasma cell tumor, skin, dog (case 32, using step sections). a) Neoplastic plasma cells, arranged in sheets and nests with few bi- and multinucleated cells (arrows). Hematoxylin and eosin. b) Nuclear immunolabeling of neoplastic plasma cells for MUM1 (arrows). MUM1 immunohistochemistry (IHC). c) Concurrent membranous immunolabeling of neoplastic cells including binucleated cells (arrows) using melan-A. Melan-A IHC. d) Visualization of MUM1 antigen in nuclei of tumor cells (red). Double-labeling immunofluorescence (DLI). e) Visualization of melan-A in the cytoplasm of neoplastic cells (green). DLI. f) Overlaid signals of cytoplasmic melan-A (green), nuclear MUM1 (red), and nuclear bisbenzimide (blue) labeling in neoplastic cells. Erythrocytes show a yellowish discoloration in the merged image due to autofluorescence. DLI.
Double-labeling immunofluorescence verified the concurrent labeling of MUM1 and melan-A in neoplastic CEMP cells (Fig. 1d–f).
In all CEMPs, variable immunopositivity was seen for the remaining markers (Table 3). Semiquantitative evaluation in each plasma cell tumor revealed variable numbers of immunolabeled cells for these markers (Fig. 2).
Immunohistochemical results of 73 examined canine neoplasms (33 extramedullary plasmacytomas, 20 melanocytomas, and 20 malignant melanomas).
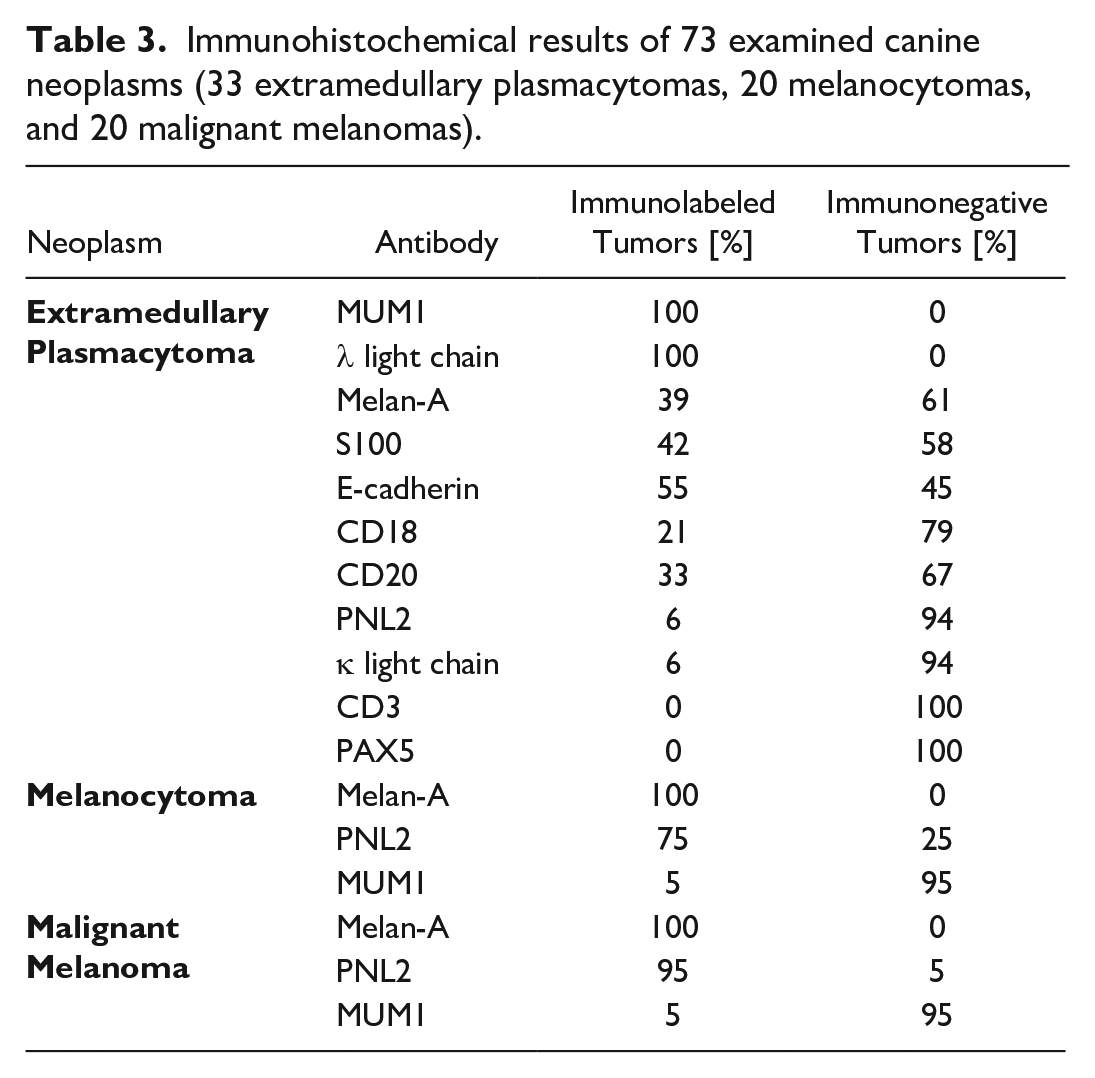
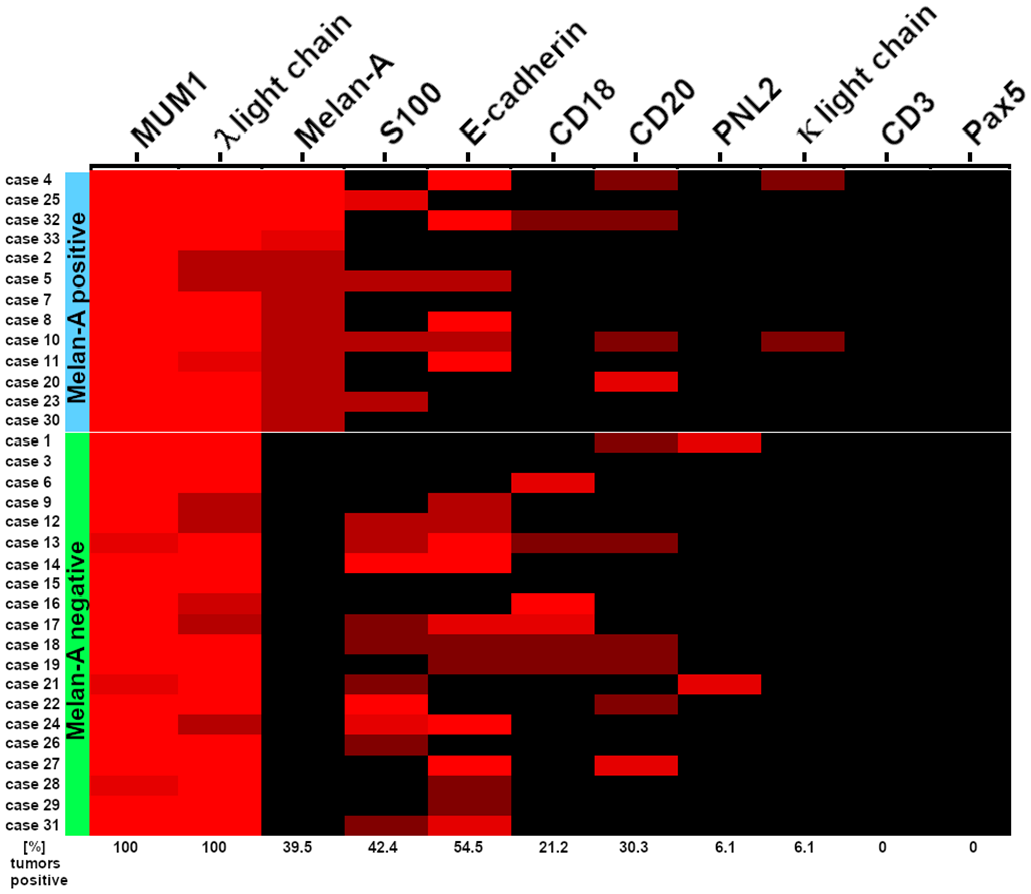
Immunohistochemical findings in 33 canine extramedullary plasmacytomas (CEMPs). In all CEMPs, neoplastic cells exhibited immunolabeling for MUM1 and λ light chain and lacked labeling for CD3 and PAX5. 13 CEMPs showed variable numbers of melan-A positive tumor cells. For the other applied antibodies, no differences regarding the immunolabeling between melan-A positive (blue cluster) and melan-A negative CEMPs (green cluster) were detected. The different shades of the red color indicate the semiquantitative reactivity of neoplastic cells for the respective marker (bright red: high, ≥60% positive cells; semi-dark red: moderate, 30% to 59% positive cells; dark red: mild, 6% to 29% positive cells; black: ≤5% immunolabeling).
In the 13 melan-A-positive CEMPs, mild immunolabeling for this marker (6%-29% immunolabeled neoplastic cells) was most frequently observed (n = 9), while immunolabeling of the 4 remaining cases was moderate (30%-59% immunolabeled cells, n = 1) and high (>60% immunolabeled cells, n = 3). Two CEMPs (cases 1 and 21) had moderate numbers of neoplastic cells immunolabeled by PNL2, but no cells were immunoreactive for melan-A. Furthermore, these 2 cases had high (case 1) and moderate (case 21) numbers of MUM1-immunolabeled cells. Notably, immunoreactivity for PNL2 consisted of a mild, diffuse, cytoplasmic labeling of approximately 50% of the neoplastic cells. Comparing melan-A-immunolabeled CEMPs and their melan-A-immunonegative counterparts, a similar immunoreactivity pattern for all other applied antibodies was observed (Fig. 2). Immunopositivity for κ light chain was restricted to 2 melan-A-positive CEMPs (case 4 and 10).
Immunohistochemistry of Canine Melanocytic Tumors
Independent of histologic features of malignancy, labeling for melan-A was seen in all melanocytic tumors (40/40; 100%). In addition, 19 malignant melanomas (95%) and 15 melanocytomas (75%) exhibited immunolabeling for PNL2. Immunoreactivity for MUM1 was seen in 1 melanocytoma (case 45, skin) and 1 malignant melanoma (case 65, eyelid), characterized by cytoplasmic labeling of small numbers of neoplastic cells (Fig. 3). The presence of an inflammatory process mimicking the labeling of neoplastic cells was ruled out, as no inflammatory cells were observed in hematoxylin and eosin-stained sections and immunohistochemistry. An overview of the immunohistochemical results in CEMPs and canine melanocytic tumors is summarized in Table 3. Detailed results are listed in Supplemental Tables S1 and S2.
Histologic and immunohistochemical findings in canine malignant melanoma, eyelid, dog (case 65, using following sections). a) Neoplastic cells are arranged in whorls and nests with mitotic figures (arrows) and occasional brown intracytoplasmic pigment. Hematoxylin and eosin. b) Neoplastic cells show green cytoplasmic immunolabeling for melan-A (arrows). Melan-A immunohistochemistry (IHC). c) Focal, green cytoplasmic immunolabeling for MUM1 (arrows) is present next to unlabeled cells. MUM1 IHC.
Discussion
Canine extramedullary plasmacytomas are usually associated with a good prognosis7,30,49 and have to be differentiated from other nonpigmented round cell tumors like lymphoma, histiocytic sarcoma, or amelanotic melanoma that may also arise in these locations but have a guarded to poor prognosis.2,9,10,13,33,36 Based on the present findings, immunoreactivity for melan-A alone is not sufficient to discriminate between CEMPs and amelanotic melanoma, as a subset of CEMPs (13/33, 39.4%) label for this marker. In contrast to melan-A, we observed immunoreactivity for PNL2 in only 2 CEMPs. In each of these cases, about half of the tumor cells were labeled. The examination of 20 melanocytomas and 20 malignant melanomas showed, as expected, widespread immunoreactivity for melan-A (100%) and PNL2 (95% in malignant melanoma and 75% in melanocytoma). Moreover, only 2 melanomas had cytoplasmic labeling by MUM1.
The histologic diagnosis of well-differentiated CEMPs is usually not a diagnostic challenge due to the characteristic histologic findings. In our study, diagnosis was based on the plasmacytic appearance of tumor cells in hematoxylin and eosin-staining, immunoreactivity for MUM1 and λ light chain, as well as absent immunolabeling of neoplastic cells for CD3 and PAX5.3,35 MUM1 affects and influences the immune system by regulating interferon signaling and is essential for the differentiation and function of lymphocytes.15,21 Moreover, this protein is crucial for the rearrangement of immunoglobulin light chains by influencing gene expression and inducing conventional light chain transcription. 22 Therefore, λ light chain labeling can be seen in the vast majority of CEMPs.3,30,43 The detection of MUM1 is considered an indicator of the transition from germinal center B cells to immunoblasts and plasma cells. Thus, MUM1 can be used as a marker for plasma cell maturation. 11 As previously shown, MUM1 has a higher sensitivity in canine tissues for the detection of CEMPs than CD20 or CD79a, 35 thus providing a good marker to confirm a suspected diagnosis of a plasma cell tumor.
In the examined collection of CEMPs, variable immunoreactivity of neoplastic cells was seen for CD20, a feature that has been previously described for CEMPs. 35 CD20 is a marker widely used to detect B-lymphocytes. 18 In contrast to PAX5, CD20 is not suitable to distinguish CEMPs from B-cell lymphomas. 17 PAX5 seems to be a more appropriate candidate for the differentiation between B-cell and plasma cell tumors1,46 and is used as a pre-B- and B-cell marker in veterinary medicine. 47
In 13 of the examined CEMPs, an unexpected immunoreactivity for melan-A was observed. To examine whether this immunoreactivity is associated with the histologic appearance of the tumor, CEMPs were classified into 5 subtypes. 30 However, no difference in the immunohistochemical reaction pattern was observed between these subtypes (Supplemental Figure S1), indicating that immunoreactivity for melan-A occurs independently of the morphologic differentiation. Melan-A is a product of MART1 (melanoma antigen recognized by T-cells 1), a surface protein that is detected by CD8+ T-lymphocytes, and is widely expressed in primary and metastatic melanomas.5,29 In both veterinary and human medicine, monoclonal antibodies directed against melan-A (clone A103) are commonly used to identify the melanocytic cell of origin or cell differentiation.4,19,32,34 Melan-A immunoreactivity has been described for various cell types, including cells of salivary carcinoma, transitional cell carcinoma, adrenal cortex and associated neoplasia, respiratory and alveolar epithelium in the lung, pancreatic and salivary duct epithelium, testicular interstitial cells and associated neoplasia, prostatic epithelium, and transitional epithelium of the urinary bladder in dogs.32,33 In humans, immunoreactivity for melan-A has been described in the adrenal cortex, testis, and ovary. 4 Since no MELAN-A transcripts have been detected by PCR in these tissues, it is assumed that the melan-A antibody has an aberrant immunoreactivity in the adrenal cortex and the gonads. 19 These reports indicate that melan-A represents a marker with high sensitivity but limited specificity for cells of melanocytic origin. This limited specificity may explain its immunoreactivity in CEMPs; however, the cause and underlying mechanisms for the observed immunoreaction of melan-A in a subset of these tumors remain unclear. Upregulation and labeling of melan-A due to genetic, metabolic, environmental, or other unidentified reasons may represent an explanation, analogous to previous considerations in reports of unexpected immunoreactions for other markers like MUM1 in canine cutaneous histiocytoma. 44 It is under current discussion that cytoplasmic immunoglobulins might react with secondary antibodies or other substrates during immunohistochemical reactions. The mechanism is still not fully understood as not all antibodies show cross-reactivity with plasma cells.35,39
Other markers used in this study showed variable immunoreactivity in CEMPs, independently of the immunoreactivity for melan-A, consistent with previous immunohistochemical findings in canine plasmacytic tumors (Supplemental Table S3). Notably, all CEMPs that exhibited immunoreactivity for melan-A lacked immunolabeling for PNL2. PNL2 is a monoclonal antibody directed against the C-terminal part of the subtype 2 of human somatostatin receptor. 37 Except for few exceptions, this marker is known to be highly specific for melanocytic tissues in humans. 6 Immunoreactivity of PNL2 is described for clear cell sarcoma, renal angiomyolipomas, granulocytes, lymphangioleiomyomatosis, and 1 case of melanotic schwannoma in human tissues, and for the granular cell layer in the cornified esophagus epithelium in mice. 37 In dogs, immunoreactivity of PNL2 is largely restricted to epidermal melanocytes and spermatids of seminiferous tubules, while no immunoreactivity has been observed in a range of nonmelanocytic canine tissues and neoplasia. 34 Thus, PNL2 is considered a highly specific marker for the identification and discrimination of melanocytic tumors from other tumors of various histogenesis in dogs. 34 As suggested by previous studies, PNL2 has a higher specificity for the detection of melanocytic tumors than melan-A and may be helpful to further discriminate poorly differentiated CEMPs from nonpigmented melanoma.34,40 Still, 2 CEMPs were positive for PNL2 in our investigations, raising further questions about the significance of this observation in single cases. The lower sensitivity of PNL2 in our study indicates that both antibodies, melan-A and PNL2, should be used in cases of suspected melanoma.
The melanocytic tumors investigated in this study show widespread immunoreactivity for melan-A and PNL2 in accordance with previous reports. 34 In addition, 2 melanocytic tumors exhibited cytoplasmic immunolabeling for MUM1. Surprisingly, MUM1-positive melanocytic tumors exhibited membranous or cytoplasmic immunoreactivity, whereas plasmacytomas showed nuclear immunolabeling. Therefore, the immunoreactivity for MUM1 in melanomas may represent aberrant labeling, which needs to be investigated in future studies. This finding, however, seems to be a rather exceptional observation as only 2 out of 40 cases showed this immunoreaction, independent of histologic features of malignancy of the tumor. Thus, our findings are mostly consistent with former studies describing the lack of MUM1 immunolabeling in canine melanomas. 35 Besides plasma cells and their tumors, immunoreactivity with MUM1 is reported in B-cell and anaplastic lymphomas, as well as canine cutaneous histiocytoma.35,44 However, for unknown reasons, MUM1 is variably expressed in human melanocytes and has been found in human malignant melanomas.8,14,26 In humans, the labeling pattern of MUM1 in malignant melanomas is nuclear to diffuse with unclear relevance regarding associations with the biological behavior of the tumor and clinical outcome compared to their MUM1-immunonegative counterparts.8,26 Further studies are required to shed light on the clinical significance of canine malignant melanomas that immunolabel for MUM1.
In summary, unexpected immunolabeling for melan-A was detected in a subset of canine extramedullary plasmacytomas. Our results indicate that reliable differentiation between CEMPs and nonpigmented melanocytic tumors in questionable cases should not be based on immunoreactivity for melan-A or MUM1 only. As in many other diagnostic challenges, an immunopanel is recommended to reach a final diagnosis. 39 In the case of suspected CEMPs or nonpigmented melanocytic tumors, this should include MUM1, λ light chain, CD20, melan-A, and PNL2. Moreover, further investigations, using tools such as PCR, are needed to understand the underlying mechanism that causes melan-A labeling in canine extramedullary plasmacytomas.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241246979 – Supplemental material for Melan-A immunolabeling in canine extramedullary plasmacytomas
Supplemental material, sj-pdf-1-vet-10.1177_03009858241246979 for Melan-A immunolabeling in canine extramedullary plasmacytomas by Lukas Schuwerk, Anastasiia Ulianytska, Wolfgang Baumgärtner and Wencke Reineking in Veterinary Pathology
Footnotes
Acknowledgements
The authors wish to thank Caroline Schütz, Julia Baskas, Petra Grünig, Jana-Svea Harre, and Kerstin Rohn for their excellent technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was in part supported by a scholarship (AU) of the University of Veterinary Medicine, Foundation, Hanover, Germany.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
