Abstract
Several members of the SRY-related HMG-box (SOX) protein family are implicated in tumorigenesis, metastasis, and regulation of the tumor microenvironment. SOX10, which is involved in neural crest cell migration and differentiation, has long been recognized a sensitive and specific immunohistochemical (IHC) marker in the diagnosis of melanoma in humans. However, expression of SOX10 in other tumor types has infrequently been evaluated in humans until recently and has not been thoroughly investigated in the dog. Our aim was to characterize the expression of SOX10 in canine neoplasms to objectively assess its value as a diagnostic IHC marker. Immunohistochemistry for SOX10 was performed on 437 archived, formalin-fixed paraffin-embedded tissues from representative canine neoplasms of ectodermal (15 tumor types), mesodermal (13 tumor types), endodermal (8 tumor types), and mixed/unknown (7 tumor types) embryologic origin. Oral and cutaneous tumors of melanocytic origin were used as positive controls. Intense SOX10 immunolabeling was observed in most tumors of ectodermal origin, including consistent expression in mammary carcinomas, and gliomas. Embryonal and hair follicle neoplasms inconsistently exhibited strong nuclear immunolabeling. Oral fibrosarcomas and undifferentiated oral sarcomas both inconsistently exhibited moderate to strong nuclear immunolabeling. Neoplasms of mesodermal and endodermal origin lacked immunolabeling. Salivary carcinomas, representing an unknown/mixed embryologic origin, were strongly labeled. SOX10 expression is not limited to melanomas, but is expressed by canine tumors of diverse tissues and embryologic derivation. Importantly, expression of SOX10 by a subset of oral sarcomas impairs its value as a marker for spindle cell oral melanomas.
The SRY-related HMG-box (SOX) protein family consists of 20 transcriptional regulators with a highly conserved high-mobility group (HMG) domain, subclassified into groups SOXA through SOXH. Several of these transcription factors are implicated in cancer progression, regulation of the tumor microenvironment, and metastasis in solid tumors. 8 Recently, the characterization of SOX2 expression in canine neoplasia as a marker of cancer stemness with potential prognostic indications, as well as extensive investigation of SOX9 expression in canine epithelial skin tumors, both suggest that SOX transcription factors represent valuable immunohistochemical (IHC) markers in the dog.7,19 SOX10, which is involved in neural crest cell migration and differentiation and belongs to the subgroup SOXE, is considered a sensitive and specific IHC marker in the diagnosis of melanoma in humans, even compared with the gold standard, S100.1,2,12 The diagnosis of melanoma can be challenging especially for scarcely pigmented cases; therefore, a number of IHC markers, including melanA, PNL-2, TRP-1, and TRP-2 are often used in conjunction to help confirm the diagnosis. 27 SOX10 has additionally been used without extensive validatory work as an IHC marker to diagnose canine melanocytic tumors and is reported to exhibit good diagnostic sensitivity based on the examination of small tumor cohorts.24,31
The potential use of SOX10 as a single, specific, confirmatory immunomarker would represent a considerable monetary, and potential time, gain in the veterinary diagnostic field. 28 Recently, a comprehensive study has suggested the usefulness and a high degree of sensitivity of SOX10 as a marker to differentiate canine oral melanomas and soft tissue sarcomas. 31
Contrary to the human literature, in which SOX10 expression has been evaluated in normal tissue as well as in a wide range of neoplasms, including breast carcinoma, nerve sheath tumor, basaloid and adnexal cutaneous neoplasms, salivary carcinoma, nasopharyngeal carcinoma, and prostate cancer, the current veterinary literature lacks adequate characterization of SOX10 in canine neoplasms.3,4,13,16 In healthy human tissue, apart from cells of melanocyte lineage, SOX10 expression is reported in salivary gland tissue, breast myoepithelial cells, and bronchial cells. 23 SOX10 is similarly expressed in canine melanocytes in various dermal and mucosal locations, and within basal cells of meibomian glands, dermal nerves, and myoepithelial cells of apocrine and mammary glands. 21 Furthermore, SOX10 plays a crucial role in the development of the peripheral nervous system in all vertebrates, where it is expressed in Schwann cells and autonomic ganglia, and in the development of oligodendrocytes where it persists in terminally differentiated cells.5,29 We thus hypothesized that SOX10 is expressed not only in canine melanocytic neoplasms, but also more broadly in tumors derived from ectoderm and neuroectoderm. To confirm this hypothesis, we assessed SOX10 IHC expression in canine tumors originating from the 3 primary germ layers.
Materials and Methods
Ten samples each from 42 different non-melanocytic neoplasms were retrieved from the Section of Anatomic Pathology archives at the New York State Animal Health Diagnostic Center, Cornell University College of Veterinary Medicine. Ectodermal tumors included the following: glioma, mammary carcinoma, meibomian gland epithelioma, oral fibrosarcoma, nasal adenocarcinoma, trichoblastoma, embryonal tumors of the central nervous system, apocrine gland anal sac adenocarcinoma (AGASACA), nerve sheath tumor, squamous cell carcinoma (nondigital), sebaceous adenoma, paraganglionoma, pheochromocytoma, and osteochondrosarcoma. Because craniofacial connective tissues, including gingival fibroblasts, originate from cranial neural crest cells, 2 mesenchymal-like tumor groups, osteochondrosarcoma (formerly known as multilobular tumor of bone) and oral fibrosarcoma, were classified in the ectodermal group. 32 For mesodermal tumors, the following were included: intestinal leiomyosarcoma, adrenocortical carcinoma, osteosarcoma, splenic hemangiosarcoma, gastrointestinal stroma tumor, renal carcinoma, cutaneous mast cell tumor, cutaneous histiocytoma, histiocytic sarcoma, Sertoli cell tumor, ovarian carcinoma, Leydig cell tumor, and granulosa cell tumor. Endodermal tumors included the following: cholangiocellular carcinoma, hepatocellular carcinoma, thyroid carcinoma, pancreatic adenocarcinoma, pulmonary adenocarcinoma, gastric carcinoma, colorectal carcinoma, and urothelial carcinoma. Seven tumor types (salivary carcinoma, undifferentiated oral sarcoma, soft tissue tumor, ovarian teratoma, carcinoids/neuroendocrine tumors, meningioma, and dysgerminoma), in which the germ cell origin is either unknown or mixed, were grouped under an unknown embryologic origin category. Only 2 dysgerminomas and 5 ovarian teratomas were available for inclusion. Overall, 407 nonmelanocytic tumors were thus included. In addition, 3 sets of 10 melanocytic neoplasms each were included. These were melanocytic neoplasms affected the oral cavity, cutaneous tissue, and digital/subungual area (total of 30 cases), for a total of 437 cases evaluated. In the melanocytic tumor group, 6 melanocytomas and 4 melanomas arising within the oral cavity or in the immediate surrounding soft tissues (i.e. lips) were selected, including 2 amelanotic melanomas. Five cutaneous melanocytomas and 5 cutaneous melanomas were evaluated. Finally, 2 digital/subungual melanocytomas and 8 digital/subungual melanomas were assessed, including 4 amelanotic melanomas. In total, the evaluated melanocytic tumors thus consisted of 13 melanocytomas and 17 melanomas, including 6 that were amelanotic.
Validation and expression of SOX10 was evaluated in normal canine tissues utilizing a tissue microarray, which included brain, liver, lung, kidney, urinary bladder, heart muscle, skeletal muscle, aorta, haired skin, oral mucosa, esophagus, stomach, small intestine, large intestine, prostate gland, testis, and lymph node. Validation included utilizing different epitope retrieval methods and incubation times to determine the optimal labeling. One canine melanoma expressing SOX10, melanA, and TRP-2 was used as the positive control. Formalin-fixed paraffin-embedded tissues were processed for immunohistochemistry with SOX10 (rabbit monoclonal anti-SOX10 antibody, Abcam, clone no. EPR4007-104, catalog no. ab180862) using the Leica Bond Max Automated Immunohistochemistry Staining System, according to the manufacturer’s instructions (Leica Microsystems, Buffalo Grove, IL). Before proceeding with immunohistochemistry, tissues were sectioned at 5.0 μm and deparaffinized with Bond Dewax Solution (Leica, catalog no. AR9222). Pretreatment with heat-induced epitope retrieval was performed for 30 minutes using Tris/ethylenediaminetetraacetic acid (EDTA) ph9 (Bond Epitope Retrieval Solution 2, Leica, catalog no. AR9640). Endogenous peroxidase activity was blocked with a 3% peroxide solution (Leica, catalog no. DS9800) for 5 minutes. SOX10 was diluted at 1:500 and applied to the slides for 60 minutes. Tissues were then incubated with the Leica Bond Polymer reagent (Leica, catalog no. DS9800) for 30 minutes. Subsequently, tissues were developed with 3,3-diaminobenzidine (DAB) chromogen (Leica, catalog no. DS9800) for 10 minutes. Finally, the slides were counterstained with Harris hematoxylin (Anatech Ltd., catalog no. 842) for 5 minutes, and successively dehydrated, cleared, and mounted. In the negative controls, antibody diluent replaced the primary antibody. In addition, immunohistochemistry for S100 (dilution 1:400, Dako, catalog no. Z0311), melanA (ready-to-use, Leica, catalog no. PA0233), and TRP-2 (dilution 1:100, ABCAM polyclonal rabbit, catalog no. ab74073) was performed on all oral melanocytic tumors, and on all undifferentiated oral sarcomas and fibrosarcomas that exhibited SOX10 immunolabeling. S100 and melanA were performed according to previously published protocols. 33 For TRP-2, slides were similarly deparaffinized with Bond Dewax Solution and heat epitope retrieval was performed with Bond Epitope Retrieval solution 1 (Leica, catalog no. AR9961) applied for 40 minutes. The antibody was diluted at 1:100 and applied to slides for 15 minutes. Then, PV-AP-Anti-Mouse immunoglobulin G (IgG) reagent (Leica, catalog no. PV6110) was applied to slides for 10 minutes. Tissues were then incubated with the Leica Bond Polymer Refine Red Detection reagent (Leica, catalog no. DS9390) for 15 minutes. Finally, slides were counterstained with hematoxylin (Leica, catalog no. DS9390) for 5 minutes and successively dehydrated, cleared, and mounted.
Results
Normal Tissues and Melanocytic Tumors
The tissue microarray and normal tissue within the tumor samples revealed strong nuclear SOX10 labeling in dermal melanocytes, ganglion cells of plexi, including enteric (Fig. 1a), oligodendrocytes, Schwann cells (Fig. 1b), dermal papillae of hair follicles (Fig. 1c), and cutaneous apocrine glands and associated myoepithelium (Fig. 1c). In 64/437 samples, regardless of the embryonic layer of origin, a background population of scattered intercalated cells with strong nuclear immunolabeling was noted within the tumors (Fig. 1d). This finding did not correlate with more malignant features. Because this is the first study in veterinary pathology to characterize immunostaining for SOX10 in a large number of canine tumor samples, labeling for SOX10 was considered a binary event (absence of immunolabeling: negative, presence of immunolabeling: positive). Most positive tumors, especially melanocytic tumors, had on average between 75% and 100% of strong nuclear immunolabeling throughout the whole examined section. Small numbers of tumors (e.g. nerve sheath tumors and AGASACAs) had a lower percentage of neoplastic cells with strong nuclear immunolabeling (15%–20%). Whether this represents areas of stemness or de-differentiation, or variable expression by neoplastic cells prompted by cellular and microenvironmental differences, remains unclear for these tumors with lower expression. Results were thus cautiously cataloged as positive to provide comprehensive literature records as a basis for future studies.
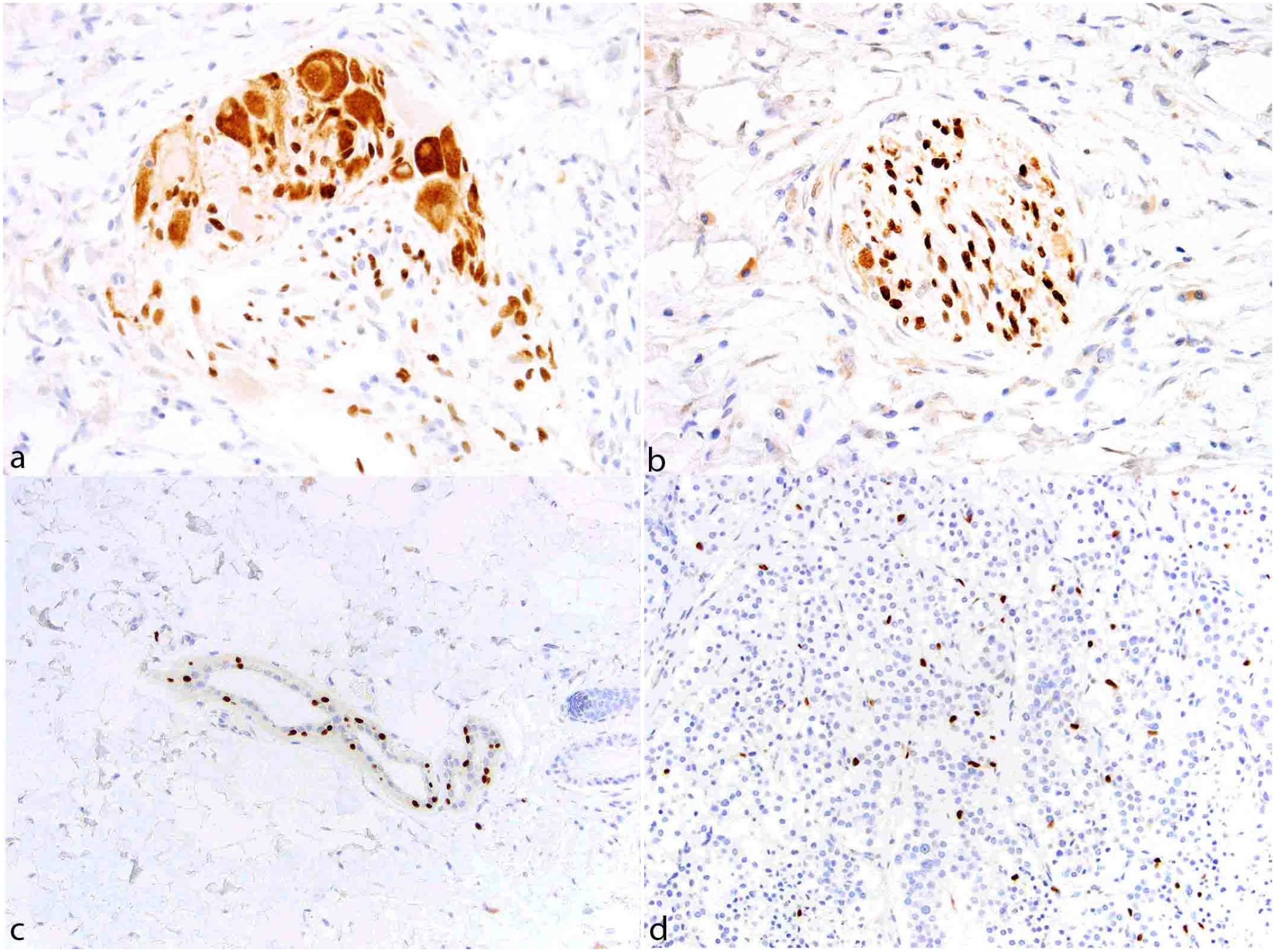
SOX10 immunohistochemistry, control canine tissue. (a) Ganglion cells within intestinal plexi have moderate, faintly granular immunolabeling. Satellite cells and Schwann cells have moderate to strong SOX10 nuclear and cytoplasmic immunolabeling. (b) Schwann cells within a nerve have strong nuclear immunolabeling. (c) Cutaneous apocrine glands epithelium and myoepithelium have multifocal strong nuclear immunolabeling. (d) Interstitial cells within a neuroendocrine carcinoma exhibit strong nuclear immunolabeling.
Melanomas had traditionally described histomorphologic features, including sheets of neoplastic round to polygonal to spindle cells with large nuclei, open chromatin patterns, a prominent nucleolus, and variable melanin pigment (Fig. 2a). All melanocytic tumors (30/30) had strong nuclear immunolabeling for SOX10 regardless of their anatomic location (Fig. 2b-d) and were thus used as a comparative reference of immunolabeling intensity. In highly pigmented melanocytic neoplasms with dense melanophage infiltrates, SOX10 immunolabeling additionally allowed the differentiation of neoplastic melanocytes, which had strong nuclear labeling, from melanophages, which lacked nuclear immunolabeling (Fig. 2c).
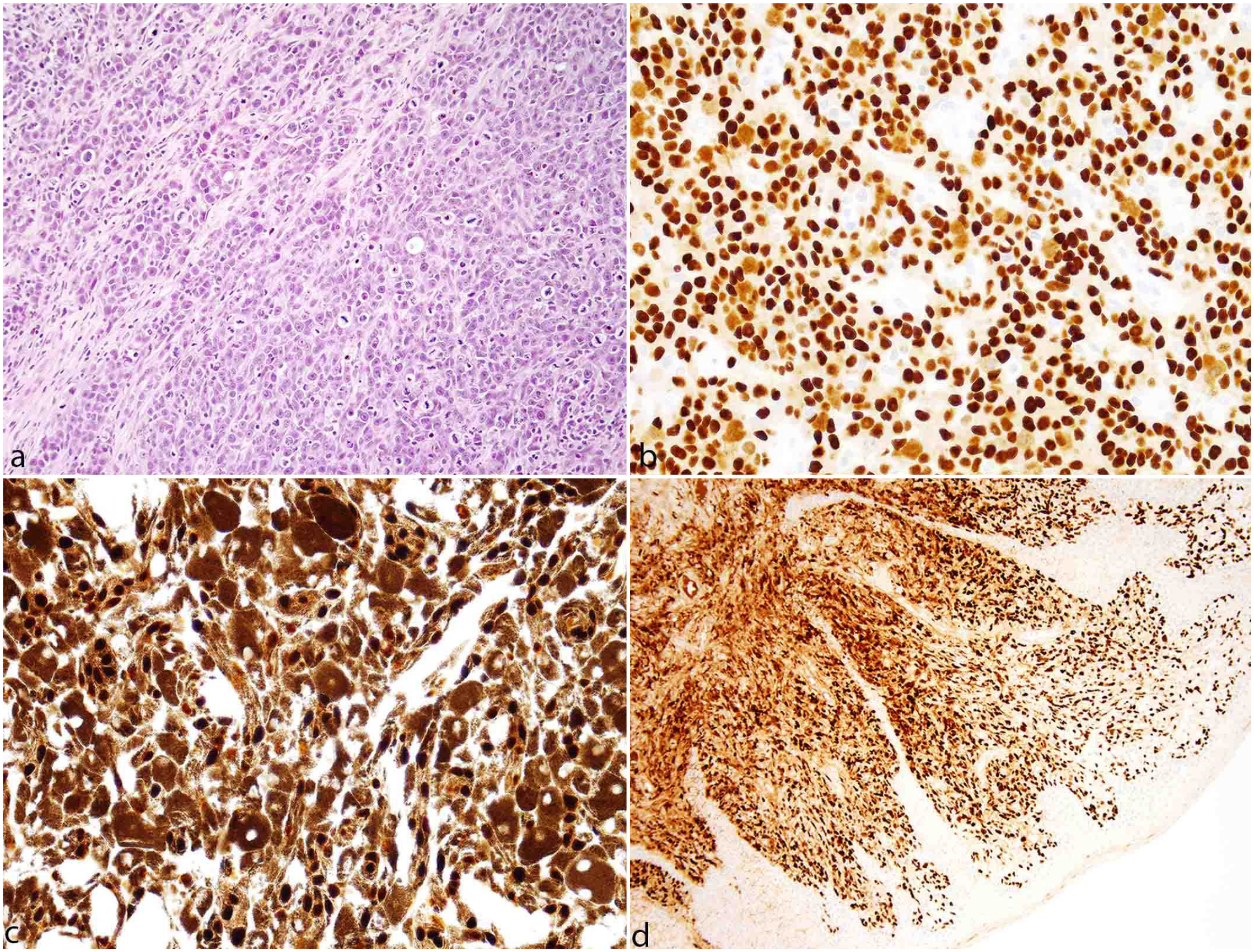
Melanoma, canine. (a) Sheets and streams of neoplastic melanocytes. Hematoxylin and eosin. (b) Dermal melanoma with strong nuclear immunolabeling in the neoplastic cells. SOX10 immunohistochemistry (IHC). (c) Heavily pigmented oral melanoma with strong nuclear immunolabeling differentiated from the cytoplasmic melanin pigment. SOX10 IHC. (d) Amelanotic oral melanoma with strong nuclear immunolabeling. SOX10 IHC.
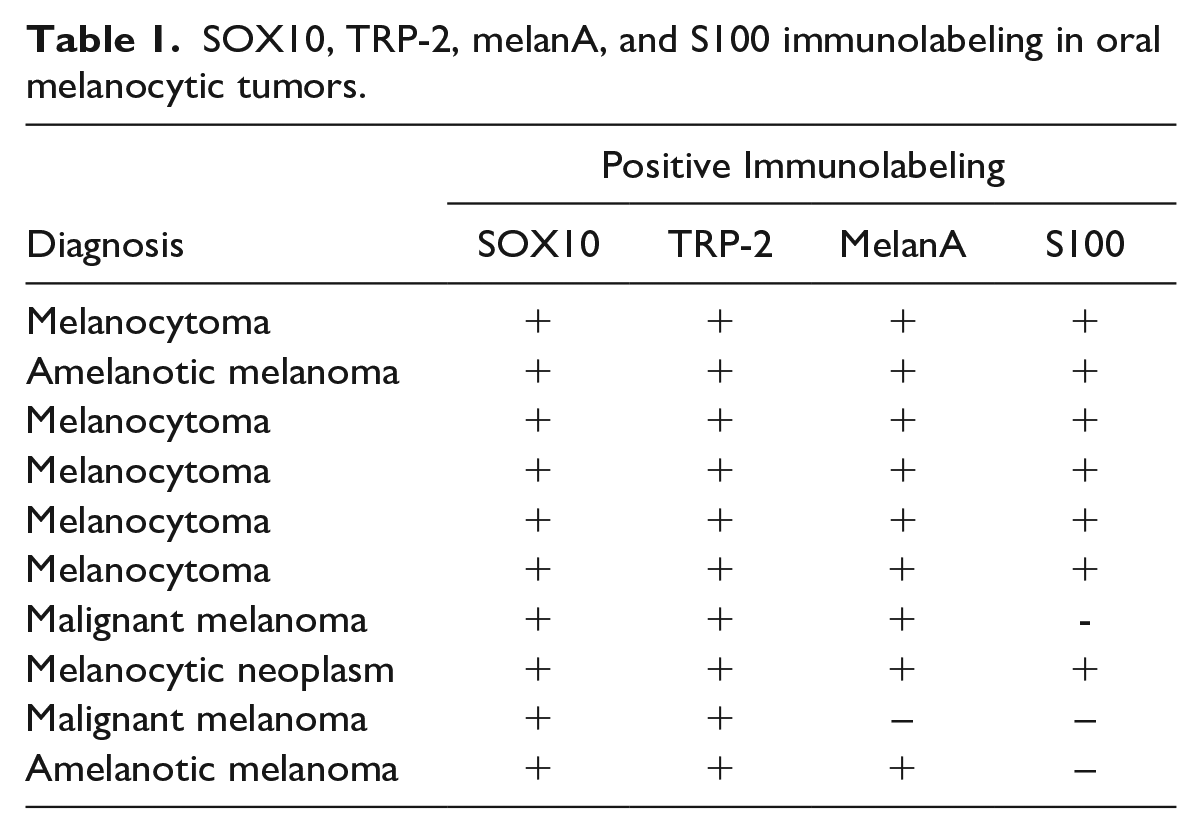
All oral melanocytic tumors consistently had strong nuclear immunolabeling for SOX10, TRP-2, melanA, and more inconsistent labeling for S100. Six oral fibrosarcomas and 5 undifferentiated oral sarcomas also had strong nuclear immunolabeling for SOX10, but they were variably immunopositive for TRP-2, melanA, and S100 (Tables 1 and 2).
SOX10, TRP-2, melanA, and S100 immunolabeling in oral melanocytic tumors.
TRP-2, melanA, and S100 immunolabeling in SOX10-positive oral fibrosarcomas and undifferentiated oral sarcomas.
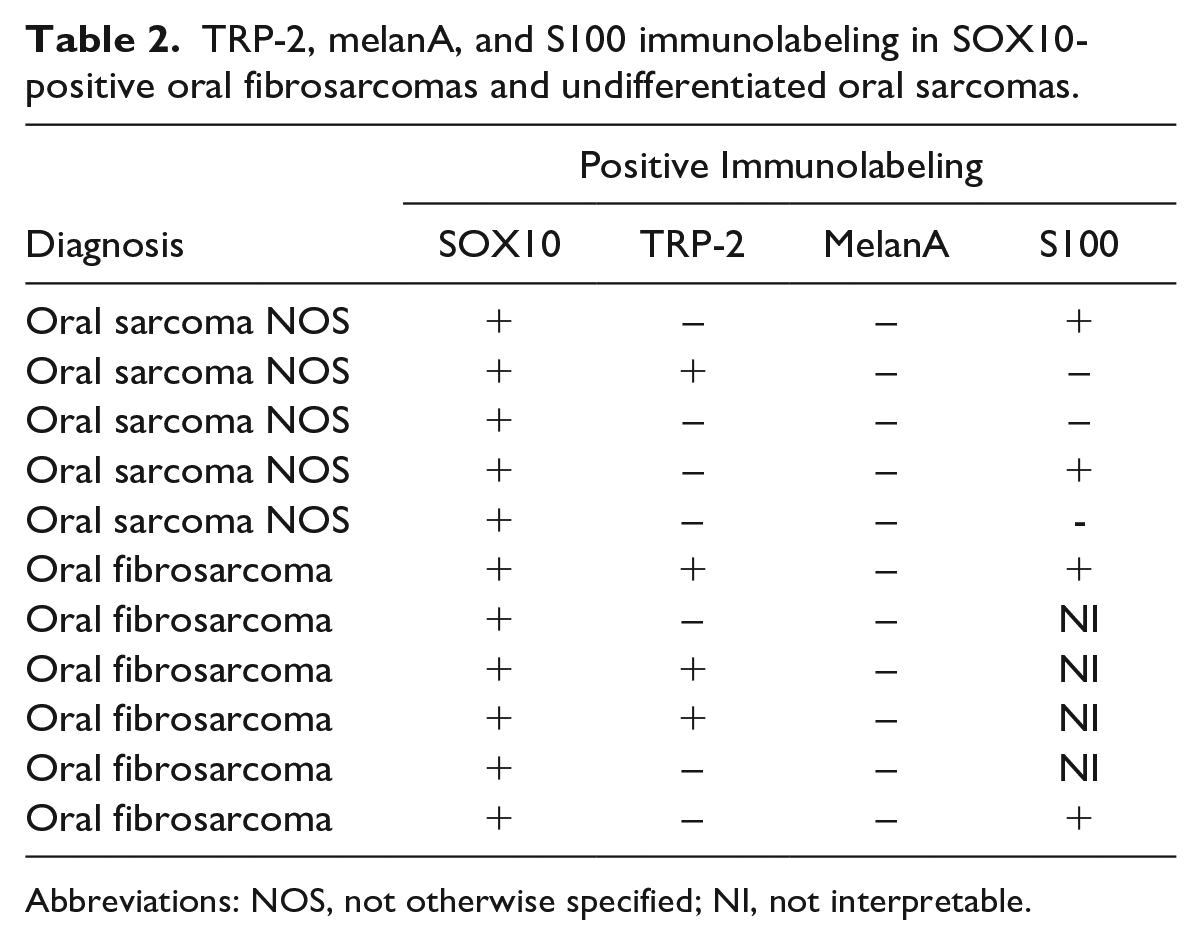
Abbreviations: NOS, not otherwise specified; NI, not interpretable.
Tumors of Ectodermal Origin
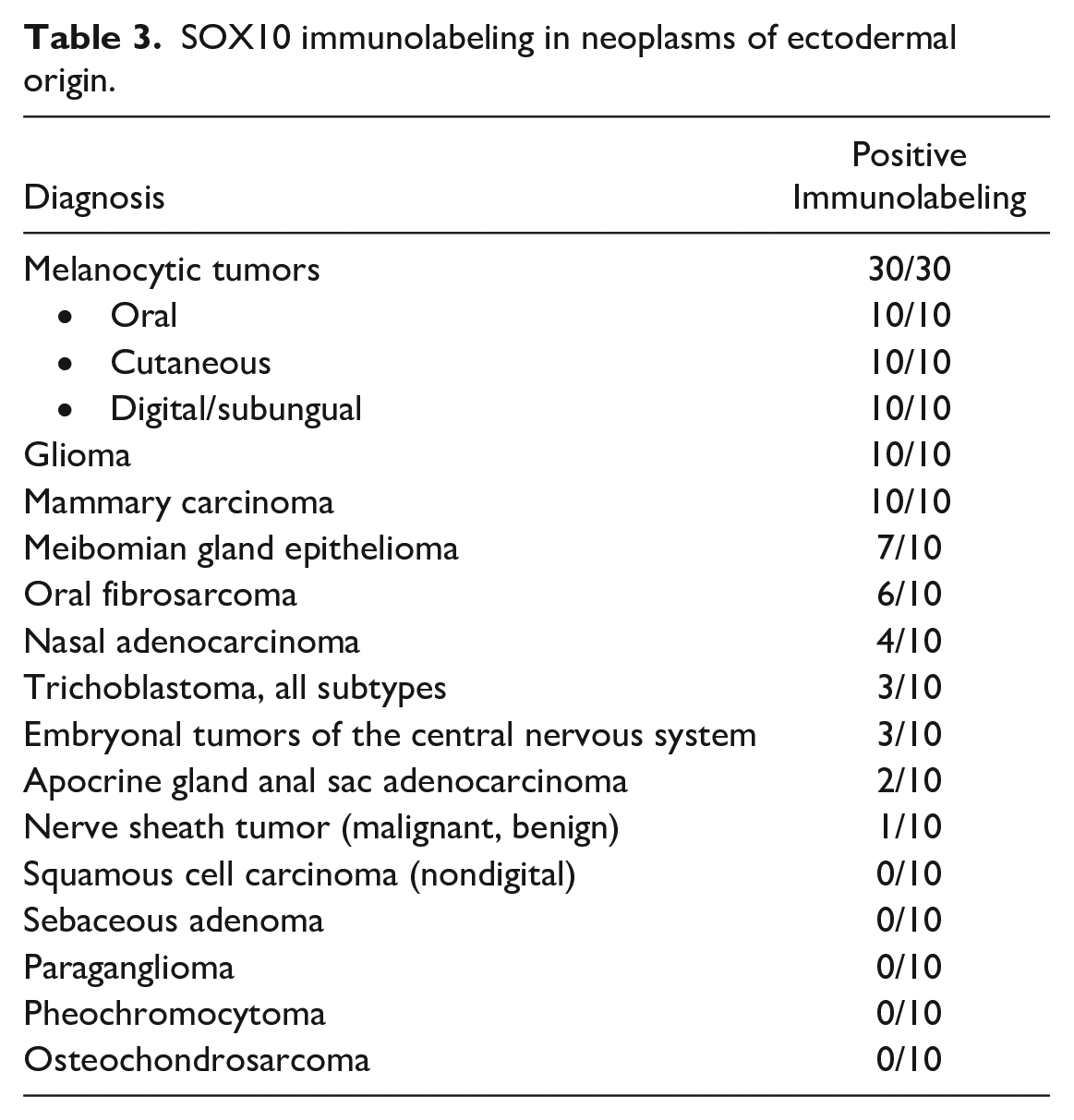
All mammary carcinomas (Fig. 3a) and gliomas (Fig. 3b) had strong nuclear immunolabeling. The immunolabeling pattern and intensity within mammary carcinomas did not differ across tumor grade (grade 1: 4/10, grade 2: 3/10, and grade 3: 3/10). Gliomas were consistently immunolabeled, including an astrocytoma, 3 undefined gliomas, and 6 oligodendrogliomas. Variable numbers of meibomian gland epitheliomas, oral fibrosarcomas (Fig. 3c), nasal adenocarcinomas, trichoblastomas (Fig. 3d), embryonal tumors, and nerve sheath tumors had strong nuclear immunolabeling (Fig. 3e; Table 3). Immunolabeling for SOX10 was also present in 6/10 neoplasms classified as oral fibrosarcoma (Table 3).
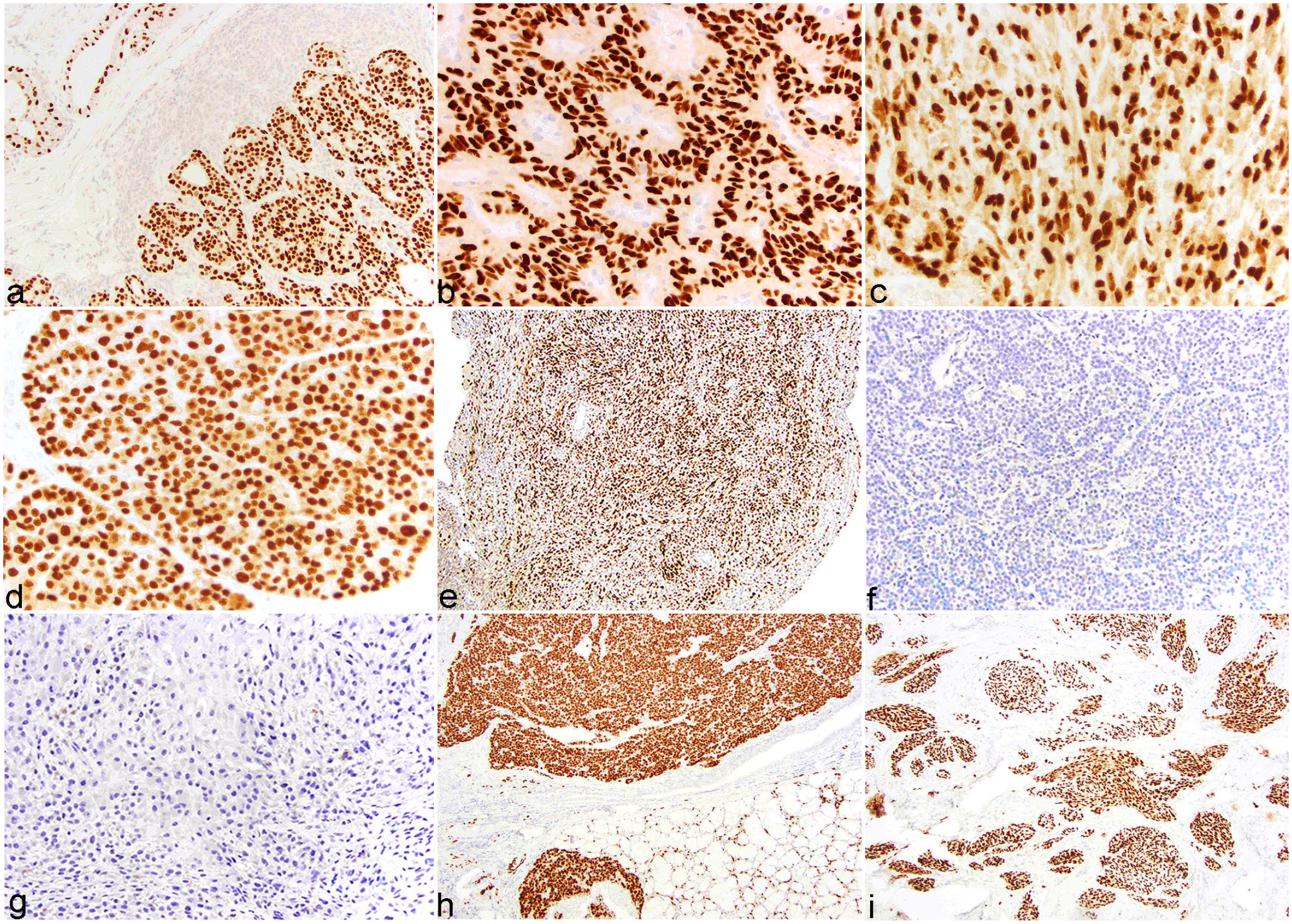
Canine tumors, SOX10 immunohistochemistry. (a) Mammary carcinoma with strong nuclear immunolabeling. Adjacent glandular epithelium and myoepithelium have multifocal strong nuclear immunolabeling. (b) Oligodendroglioma with strong, diffuse nuclear immunolabeling. (c) Oral fibrosarcoma with strong nuclear immunolabeling. (d) Solid-cystic trichoblastoma with strong nuclear immunolabeling. (e) Malignant nerve sheath neoplasm with strong nuclear immunolabeling. (f) Osteochondrosarcoma with diffuse absence of immunolabeling. (g) Hepatocellular carcinoma with diffuse absence of immunolabeling. (h) Salivary carcinoma with strong, diffuse nuclear immunolabeling. Normal glandular epithelium and myoepithelium have strong nuclear labeling. (i) Undifferentiated oral sarcoma with strong nuclear immunolabeling.
SOX10 immunolabeling in neoplasms of ectodermal origin.
One AGASACA had diffuse immunolabeling and was the only specimen with neuroendocrine patterning. Another AGASACA had focal expression of SOX10 near the border of the tumor, in a poorly differentiated and desmoplastic region, suggesting an association with more profound de-differentiation and/or a more invasive behavior.
Squamous cell carcinomas (nondigital), sebaceous adenomas, paragangliomas, pheochromocytomas, and osteochondrosarcomas (Fig. 3f) all lacked immunolabeling.
Tumors of Mesodermal Origin
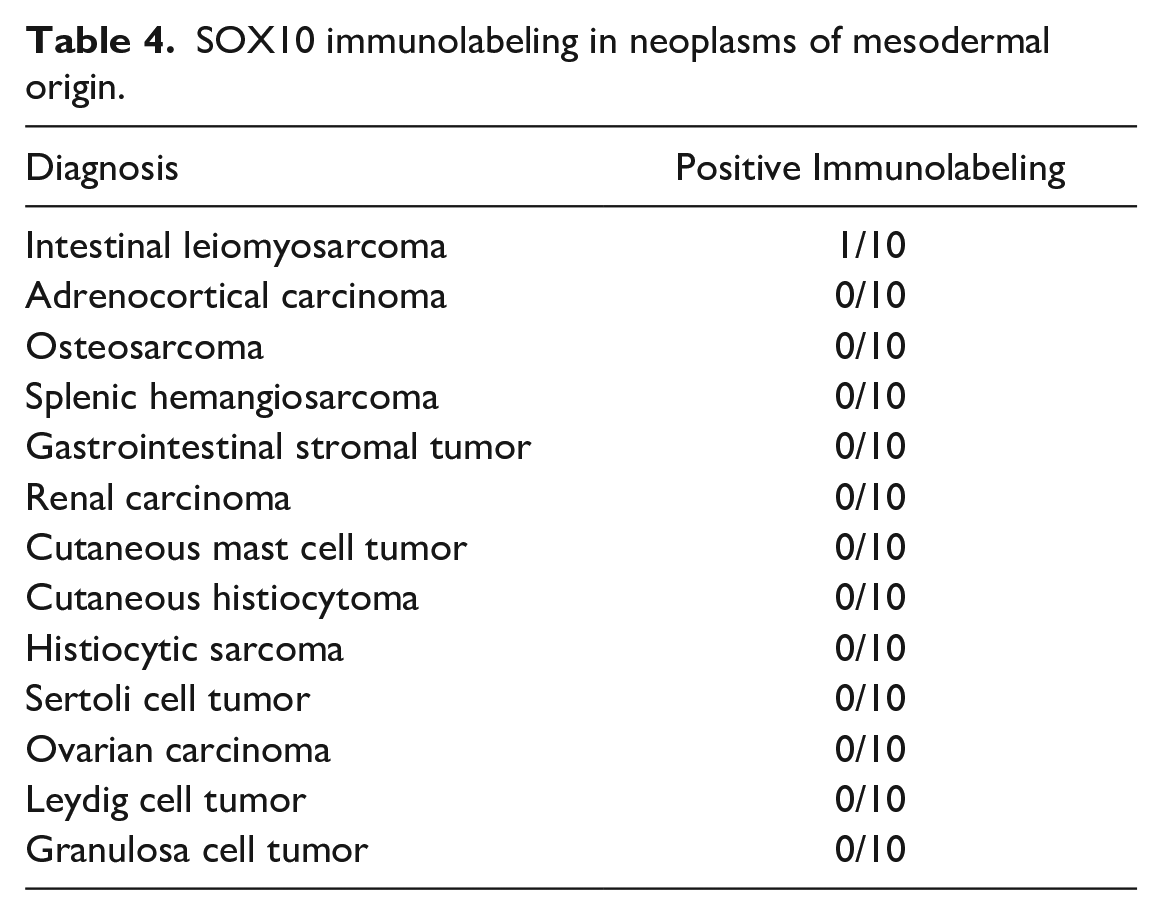
Almost all mesodermal tumors (adrenocortical carcinoma, osteosarcoma, splenic hemangiosarcoma, gastrointestinal stromal tumor, renal carcinoma, cutaneous mast cell tumor, cutaneous histiocytoma, histiocytic sarcoma, Sertoli cell tumor, ovarian carcinoma, Leydig cell tumor, and granulosa cell tumor) lacked immunolabeling (Table 4).
SOX10 immunolabeling in neoplasms of mesodermal origin.
Tumors of Endodermal Origin
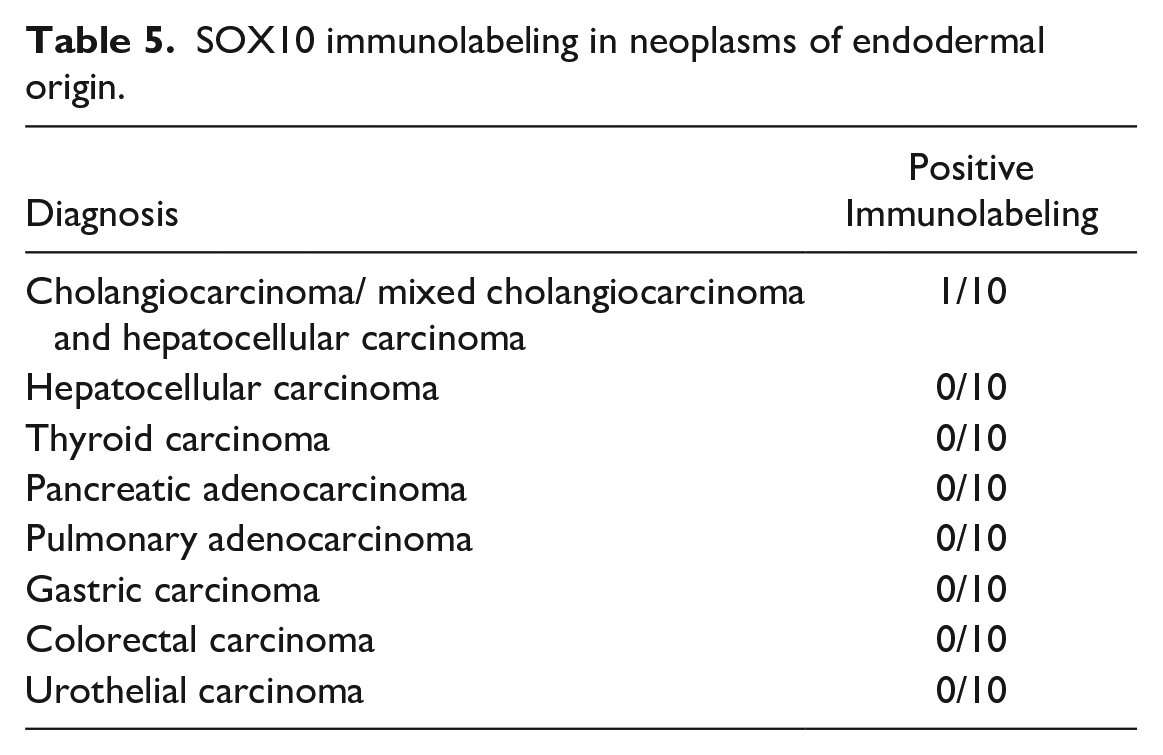
All endodermal tumors (cholangiocarcinoma, mixed hepatocellular, and cholangiocellular carcinoma, hepatocellular carcinoma, Fig. 3g, thyroid carcinoma, pancreatic adenocarcinoma, pulmonary adenocarcinoma, gastric carcinoma, colorectal carcinoma, and urothelial carcinoma) lacked immunolabeling, apart from 1 cholangiocarcinoma (1/10) (Table 5).
SOX10 immunolabeling in neoplasms of endodermal origin.
Tumors of Unknown Embryologic Origin
In the neoplasms subclassified as “others” or of mixed origin, salivary carcinomas consistently had strong nuclear immunolabeling (Fig. 3h). Half of the undifferentiated oral sarcomas had strong immunolabeling (Fig. 3i, Table 6). In ovarian teratomas, the nervous and cutaneous tissue had strong nuclear immunolabeling, echoing the results obtained in normal tissue. Carcinoids/neuroendocrine tumors, meningiomas, and dysgerminomas consistently lacked immunolabeling.
SOX10 immunolabeling in neoplasms of undetermined or mixed embryologic origin.

Abbreviation: NOS, not otherwise specified.
Discussion
The pattern of SOX10 expression reported herein expands on previous canine reports with labeling widely observed in unaltered ectodermal tissue, including cutaneous apocrine glands, myoepithelium, follicular papillae cells, nonneoplastic melanocytes, peripheral nerve cells, ganglion cells, and glial cells, correlating with the reported labeling patterns in human tissue. 18 The relevance of the ganglion cell immunolabeling is uncertain since it is unexpectedly cytoplasmic, though remarkably consistent throughout formalin-fixed paraffin-embedded samples obtained from a variety of canine patients and was present in the normal canine tissue microarray as well. In addition, in almost all tissues examined, a small population of intercalated cells was labeled for SOX10. The origin of these cells is unclear; however, they are unlikely to represent resident macrophages as macrophages were not labeled in any tissues, including lymphoid tissues. It is probable that these entrapped cells are of neural crest origin, like part of the diffuse neuroendocrine system, or represent an active population of dedifferentiated cells with retained SOX10 expression.
Our results suggest that SOX10 is generally a reliable IHC marker for the diagnosis of well-differentiated canine melanocytic neoplasms. However, the fact that SOX10 is expressed in various tumors of (neuro)ectodermal origin indicates that it should not be used as a sole diagnostic marker. Of particular interest among the outcomes of this study are the TRP-2, melanA, and S100 IHC results obtained for the SOX10 expressing poorly differentiated/undifferentiated oral sarcomas and oral fibrosarcomas. While all 6 SOX10-positive oral fibrosarcomas had conventional sarcomatous histomorphologic features, such as a prominent herringbone pattern consisting of tightly interlaced bundles of neoplastic spindle cells, 3 of these 6 tumors additionally expressed TRP-2. Since TRP-2 is a melanogenic enzyme physiologically expressed by melanocytes and found in melanoma, these results trumped the initial morphologic evaluation and prompted a diagnosis of melanoma for all 3 SOX10-positive, TRP-2-positive spindle cell tumors. 25 Such results, unfortunately, raise concerns about the misdiagnosis of poorly differentiated, amelanotic melanomas that lack biphasic distinction into polygonal cells and spindle cells. In addition, a single undifferentiated oral sarcoma expressed melanA, and another undifferentiated oral sarcoma expressed S100. S100 results were equivocal for 4 oral fibrosarcomas that broadly lacked nuclear labeling despite convincing cytoplasmic labeling. After eliminating potentially misdiagnosed melanomas based on positive IHC results combined with at least 1 positive well-recognized IHC marker for melanoma, a subset of oral spindle cell tumors solely expressing SOX10 remained. We were unable to determine whether these tumors represented melanomas with expression of SOX10 and concurrent loss of expression of all other routinely used markers (TRP-2, S100, and melanA), or spindle cell neoplasms that had gained SOX10 expression due to the neuroectodermal embryonic origin of craniofacial connective tissues.
Unfortunately, the current veterinary literature lacks systematic reports comparing the use of SOX10 to other IHC markers in the context of canine oral tumors. To this day, SOX10 has mostly been characterized in veterinary pathology as an IHC marker for oligodendrocyte precursor cells,14,35 Schwann cells, and perineurial cells, 15 and has been used to identify neoplastic melanocytes in select canine tumors, including angiomatoid melanomas, and melanocytic variants of nerve sheath tumors.24,33 SOX10 is also reported to be a poor melanocyte marker in teleosts compared with PNL-2 and melanA. 26 Recent research suggests that RNA expression levels rather than protein might more accurately differentiate oral spindle cell tumors from oral melanomas. 31 Furthermore, the results of the same study suggest that SOX10 is expressed by nonmelanocytic oral sarcomas, converging with the results described herein, and further confirming the disputable interest of using SOX10 IHC to differentiate oral melanomas from oral sarcomas. 31
In humans, several studies comparing the use of SOX10 to S100 immunolabeling in sentinel lymph nodes indicate an at least equivalent, if not superior, specificity of SOX10 compared with S100 in identifying metastases from melanoma.30,34 Human studies regrettably tend to exclude oral tumors, perhaps because of their relatively rare occurrence in humans, and thus do not address the problematic of the embryonic origin of the affected tissues. 9 A systematic study investigating the expression of SOX10 in more than 5000 human tumors found results similar to these of this study, although the authors divided their samples into neurogenic tumors, epithelial tumors, and mesenchymal tumors, rather than classifying them according to their embryonic origin. SOX10, however, was consistently expressed in neurogenic tumors and often in epithelial tumors and was consistently negative in mesenchymal tumors. 18
In our study, nerve sheath tumors almost consistently lacked labeling for SOX10, though a bias may have been introduced by the small number of tumors evaluated, and by the mixed malignant and nonmalignant neoplasms composing this group of tumors. This contrasts with the human literature, where relatively consistent expression of SOX10 has been described in malignant nerve sheath tumors (MNSTs). In 1 report evaluating the diagnostic use of SOX10 in MNSTs compared with other soft tissue tumors, MNSTs had variable S100 and SOX10 expression, with a significant percentage of cases (56%) lacking expression of both markers. One study suggested using SOX10 rather than S100 in the diagnosis of MNSTs, since it was less frequently expressed than S100. 12 Another study identified a higher percentage of SOX10 positive MNSTs (67%) in the context of evaluating the value of SOX10 to differentiate MNSTs from synovial sarcomas. 11 The authors compared histological features of malignancy to the absence or presence of SOX10 immunolabeling and failed to demonstrate a correlation between both. 11
The strong SOX10 immunolabeling in canine tumors of salivary origin correlates with similar results reported in human salivary tumors and unaffected salivary myofibroblasts. 10 The lack of correlation between expression of SOX10 and grade in mammary carcinomas is in opposition to the human literature, where expression of SOX10 has mostly been found in invasive mammary carcinomas.4, 22 Of note, osteochondrosarcoma, despite being classified in the ectodermal neoplasm group, consistently lacked SOX10 immunolabeling. 6
The results of this study indicate that SOX10 is a highly sensitive marker for the diagnosis of well-differentiated canine melanoma, but it lacks specificity and cannot consistently discriminate between oral melanoma and oral tumors with sarcomatous features, such as fibrosarcoma or undifferentiated spindle cell tumors. While these tumors all typically display an aggressive, locally invasive behavior, the more systemic pattern of metastasis for melanoma makes it crucial to obtain a proper diagnosis.17,20 SOX10 is also widely expressed in a number of other tumors, including glioma, mammary carcinoma, meibomian gland epithelioma, nasal adenocarcinoma, some adnexal tumors, and salivary carcinomas. As such, we would warrant against the sole use of SOX10 as a diagnostic marker for poorly differentiated oral spindle cell tumors. Speculatively, based on the consistent SOX10-negative results obtained from the very small number of soft tissue tumors evaluated in this study, SOX10 expression is not expected to be as much of a diagnostic conundrum in poorly differentiated cutaneous melanocytic tumors as it is in oral sarcomas. Further comprehensive studies comparing SOX10 expression in poorly differentiated cutaneous sarcomas and cutaneous melanocytic tumors are needed to better understand the labeling patterns seen in these neoplasms. SOX10 additionally appears to be a promising marker for neurogenic tumors and gliomas, though wider evaluation and validation remain to be undertaken.
Footnotes
Acknowledgements
We wholeheartedly thank the histopathology technicians of the New York AHDC for their assistance with case processing and handling of an unreasonably excessive number of blocks and slides, as well as Jenn Patterson for her help editing and creating the figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
