Abstract
To compare clinicopathologic findings of transmissible mink encephalopathy (TME) with other transmissible spongiform encephalopathies (TSE, prion diseases) that have been shown to be experimentally transmissible to cattle (sheep scrapie and chronic wasting disease [CWD]), two groups of calves (n = 4 each) were intracerebrally inoculated with TME agents from two different sources (mink with TME and a steer with TME). Two uninoculated calves served as controls. Within 15.3 months postinoculation, all animals from both inoculated groups developed clinical signs of central nervous system (CNS) abnormality; their CNS tissues had microscopic spongiform encephalopathy (SE); and abnormal prion protein (PrPres) as detected in their CNS tissues by immunohistochemistry (IHC) and Western blot (WB) techniques. These findings demonstrate that intracerebrally inoculated cattle not only amplify TME PrPres but also develop clinical CNS signs and extensive lesions of SE. The latter has not been shown with other TSE agents (scrapie and CWD) similarly inoculated into cattle. The findings also suggest that the diagnostic techniques currently used for confirmation of bovine spongiform encephalopathy (BSE) would detect TME in cattle should it occur naturally. However, it would be a diagnostic challenge to differentiate TME in cattle from BSE by clinical signs, neuropathology, or the presence of PrPres by IHC and WB.
Keywords
Transmissible mink encephalopathy (TME), a prion disease, is a neurodegenerative transmissible spongiform encephalopathy (TSE) that has been sporadically identified in ranch-raised mink. It was first documented in Wisconsin, USA, in 1947, 8 and the last reported outbreak in this country was in 1985. 19 TME is a food-borne disease that has been experimentally transmitted to a variety of animal species, including cattle, 18, 23 sheep and goats, 10 monkeys, 9, 20 hamsters, 20 mink, 1 American sable (pine marten) and beech marten, 17 skunks and ferrets, 8 and raccoons. 8, 14 The origin of TME is unknown, but is speculated to have come from sheep scrapie or from an unknown TSE in cattle. 19, 20
The primary objective of this study was to determine if second passage of the TME agent in cattle would result in similar clinicopathologic findings as seen in the first passage. Prior to this study, immunohistochemical (IHC) and Western blot (WB) findings of TME in cattle had not been described. Therefore, a secondary objective was to characterize the IHC and WB findings in TME-inoculated cattle, using methods similarly employed for bovine spongiform encephalopathy (BSE) surveillance in the USA.
Materials and Methods
Animals
Ten 4–6-month-old Jersey bull calves were purchased and assigned to two inoculated groups (each n = 4) and a control (n = 2) group. Inoculated calves were housed in a Biosafety Level 2 isolation barn (two animals per pen) at the National Animal Disease Center (NADC), Ames, Iowa. They were fed pelleted growth and maintenance rations that contained no ruminant protein, and clean water was available ad libitum. Control calves were housed together in an open shed and fed the pelleted growth ration (without ruminant protein) and alfalfa hay. Personnel wore protective clothing while in the isolation facility and showered before leaving the facility.
Inoculum
TME brain suspension for inoculation was obtained from two different source animals. One was a mink that had TME, which was biologically cloned in mink three times by limiting end-point titration. 3 The other was a Holstein steer (No. 2), which had been intracerebrally inoculated in 1990 with TME and had developed spongiform encephalopathy. 19 The brain material from the latter case was a 10% brain suspension (wt/vol) and had been stored at −70°C. In both cases, the original source was from the 1985 Stetsonville, Wisconsin, outbreak of TME in mink. The brain tissue was ground in a mechanical grinder, gentamicin was added at 100 µg/ml, and the final concentration of 10% (wt/vol) was made with phosphate buffered saline, a method previously used at this laboratory. 6, 7, 12– 15 Both inocula were positive for PrPres by WB.
Inoculation procedure
Calves were inoculated intracerebrally with 1 ml of TME from either bovine or mink inocula (No. A261 received only 0.6 ml of inoculum; Table 1) as described previously. 6, 7 Briefly, the calves were sedated with xylazine, a midline incision was made in the skin at the junction of the parietal and frontal bones, and a 1-mm hole was drilled through the calvarium. The inoculum was injected into the midbrain via a 22-gauge 9-cm-long disposable needle while withdrawing the needle from the brain. The skin incision was closed with a single suture. After each group of inoculations, the surgical instruments, including the drill bit, were discarded. Two calves (controls) were not inoculated.
Findings in cattle intracerebrally inoculated with the transmissible mink encephalopathy agent from mink and from cattle (second passage).∗
∗ TME = transmissible mink encephalopathy; PI = postinoculation; CNS = central nervous system; IHC = immunohistochemistry for PrPres; SE = spongiform encephalopathy; S/cord = spinal cord; NA = not applicable; WB = Western blot; + = lesions or antigen present; − = lesions or antigen absent.
Necropsy and samples
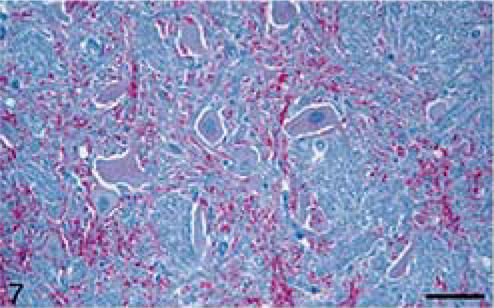
Animals were euthanatized with pentobarbital and a complete necropsy was conducted on each of the carcasses. Representative samples of nasal mucosa, lung, liver, kidney, spleen, salivary gland, thyroid gland, reticulum, rumen, omasum, abomasum, intestines (ileum, colon), adrenal gland, pancreas, urinary bladder, lymph nodes (retropharyngeal, prescapular, mesenteric, popliteal), tonsils (pharyngeal, palatine), striated muscles (heart, tongue, masseter, diaphragm, psoas major, biceps femoris, triceps), eye, sciatic nerve, trigeminal ganglion, pituitary gland, and spinal cord (cervical, thoracic, lumbar) were immersion fixed in 10% neutral buffered formalin. The brain was cut longitudinally and one half was fixed in formalin for not less than 3 weeks and the remainder of the brain was frozen (−20°C). The formalin-fixed brain was cut into 2–4-mm-wide coronal sections. Sections of various anatomic sites (a minimum of five brain sections per animal) of rostral cerebrum (frontal lobe), hippocampus, midbrain (at the level of superior colliculus) cerebellum, and brain stem (at the level of obex), and spinal cord (cervical, thoracic, and lumbar; two sections each) were processed (Fig. 1) for routine histopathology, embedded in paraffin wax, and sectioned at 5 µm. All sections were stained with hematoxylin and eosin (HE), and by an IHC method 21 using a monoclonal antibody, F99/97.6. 22 Detailed examination of various nuclei (cuneate, spinal vestibular, solitary tract, dorsal motor vagus, hypoglossal, and olivary) was done on HE-stained hemisection of brain stem for lesions of SE (Table 2).
Presence of spongiform encephalopathy in various nuclei of brain stem (hemisection) at the level of obex of cattle experimentally inoculated intracerebrally with transmissible mink encephalopathy (TME) agents from bovine and mink sources.∗

∗ NE = not examined; for spongiform encephalopathy, severity of vacuolation was recorded as minimal (+), mild (++), moderate (+++), severe (++++), or not present (−).
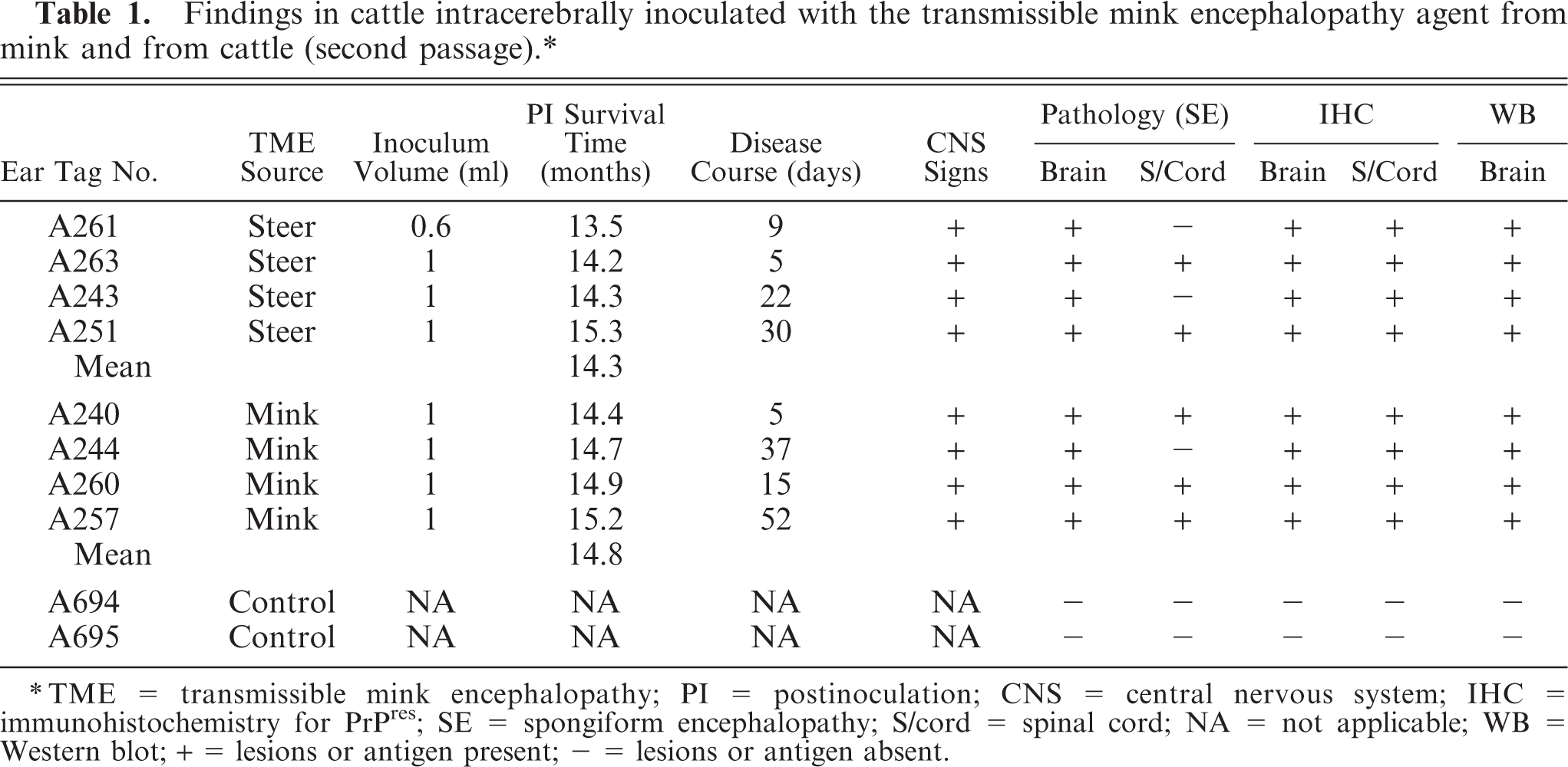
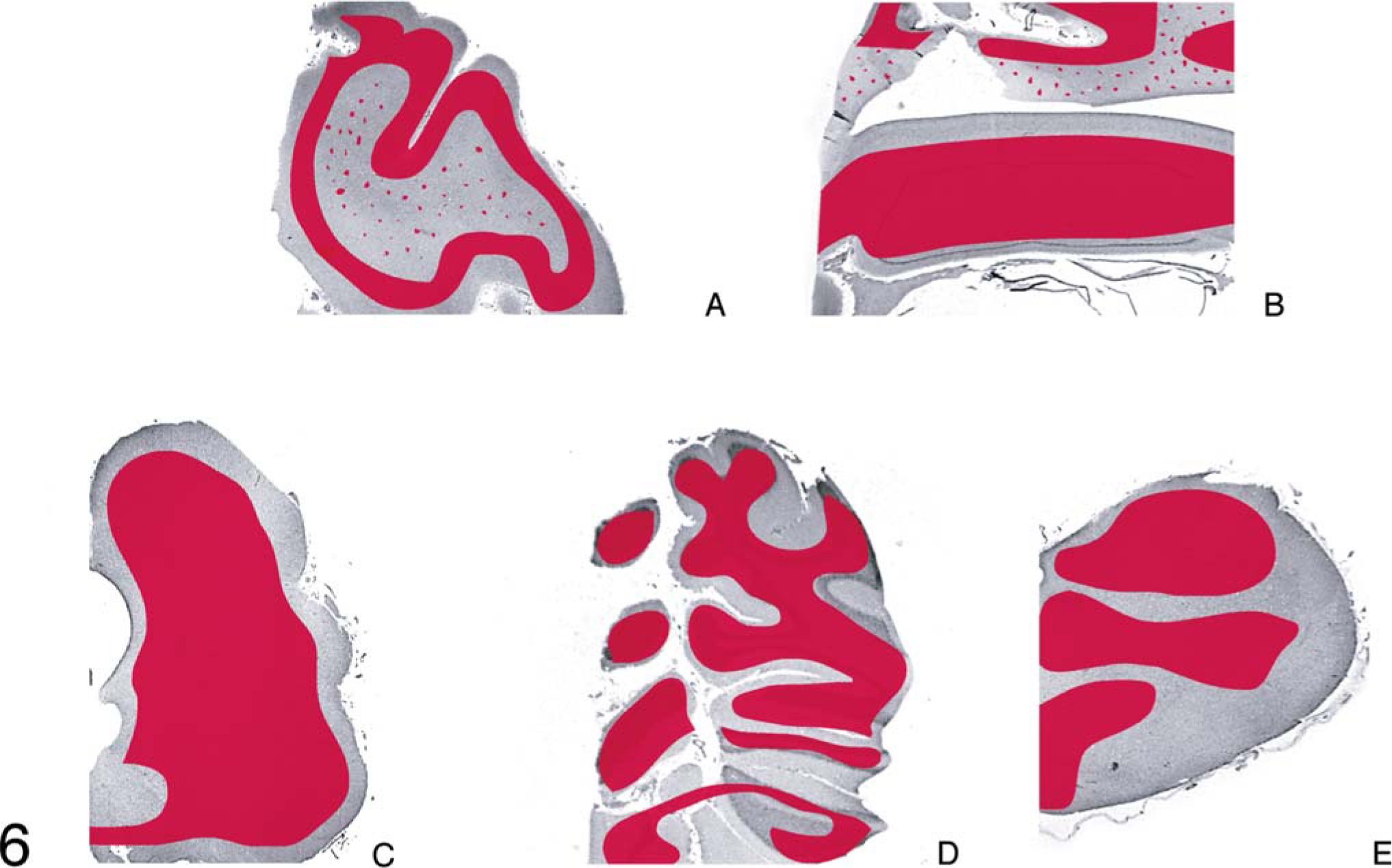
Diagrammatic representation of the distribution of lesions of spongiform encephalopathy (in blue) in five areas of the brain of animal A257 inoculated with TME from mink.
For immunodetection of PrPres, a WB method described previously was used on frozen brain (caudal medulla). 24 Animals were considered positive when the three isoforms of the pathological form of the prion protein, PrPres, were detected on the immunoblot after proteinase K enzyme treatment. PrPres-positive (from sheep scrapie) and negative control tissues were included from previous TSE studies.
Results
By 13 months postinoculation (PI), all inoculated cattle were showing clinical signs of central nervous system (CNS) abnormality (Table 1). Initially, these consisted of a variable degree of hyperexcitability, especially to something new or unusual in the barn; standing with head lowered; and occasional falling to the ground while the pen was being cleaned by animal handlers. Severe clinical signs of CNS abnormality were seen in four animals (A243, A244, A257, A261), and these consisted of aimless circling in the pen and aggressive behavior toward animal handlers. One animal (A261) charged at a window, which was approximately 6 ft above the ground, fell to the ground on its side, started paddling with its feet, and at this point was euthanatized. Within 15.3 months PI, all animals in both groups were euthanatized (Table 1). No significant difference was observed in survival times of animals between the two inoculated groups (14.3 and 14.8 months; Table 1).
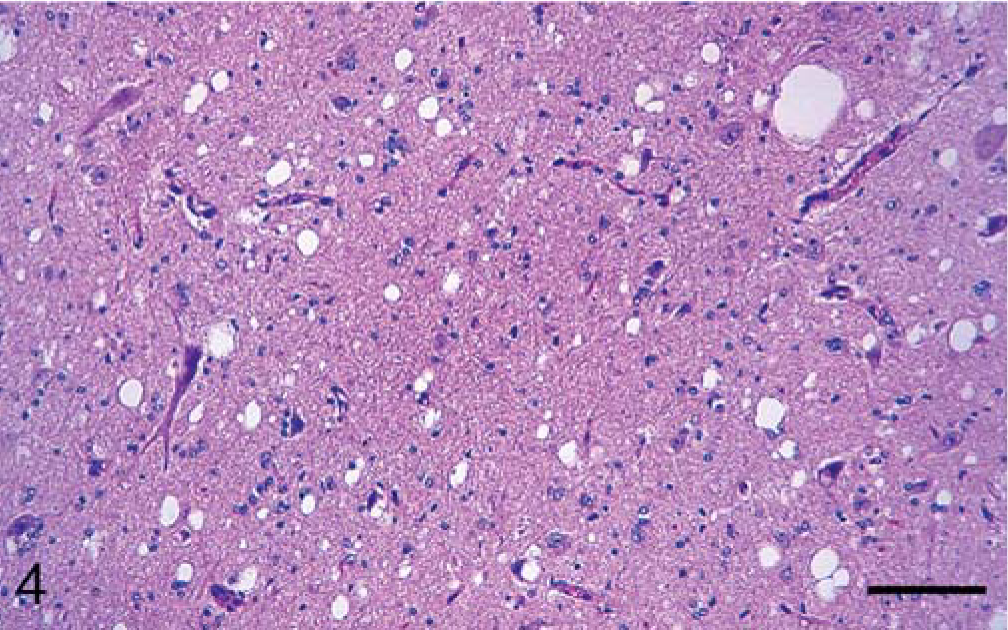
At necropsy, all animals were in fair body condition and significant gross lesions were not evident in any of the carcasses. Microscopically, however, severe lesions characteristic of SE (vacuolation of neuronal perikarya and neuropil) were seen in the brains (Figs. 1–5) and spinal cords of all inoculated cattle (Table 1). Morphologically, the severity and distribution of spongiform changes were generally similar in all (Table 3). At affected sites, there was a mild multifocal increase in glial cells, but neuronal degeneration was not a prominent feature in any of the sections.
Severity of spongiform encephalopathy in various areas of brain and spinal cord of cattle experimentally inoculated intracerebrally with transmissible mink encephalopathy (TME) agents from bovine and mink sources.∗

∗ NE = not examined; for spongiform encephalopathy, severity of vacuolation was recorded as minimal (+), mild (++), moderate (+++), severe (++++), or not present (−).
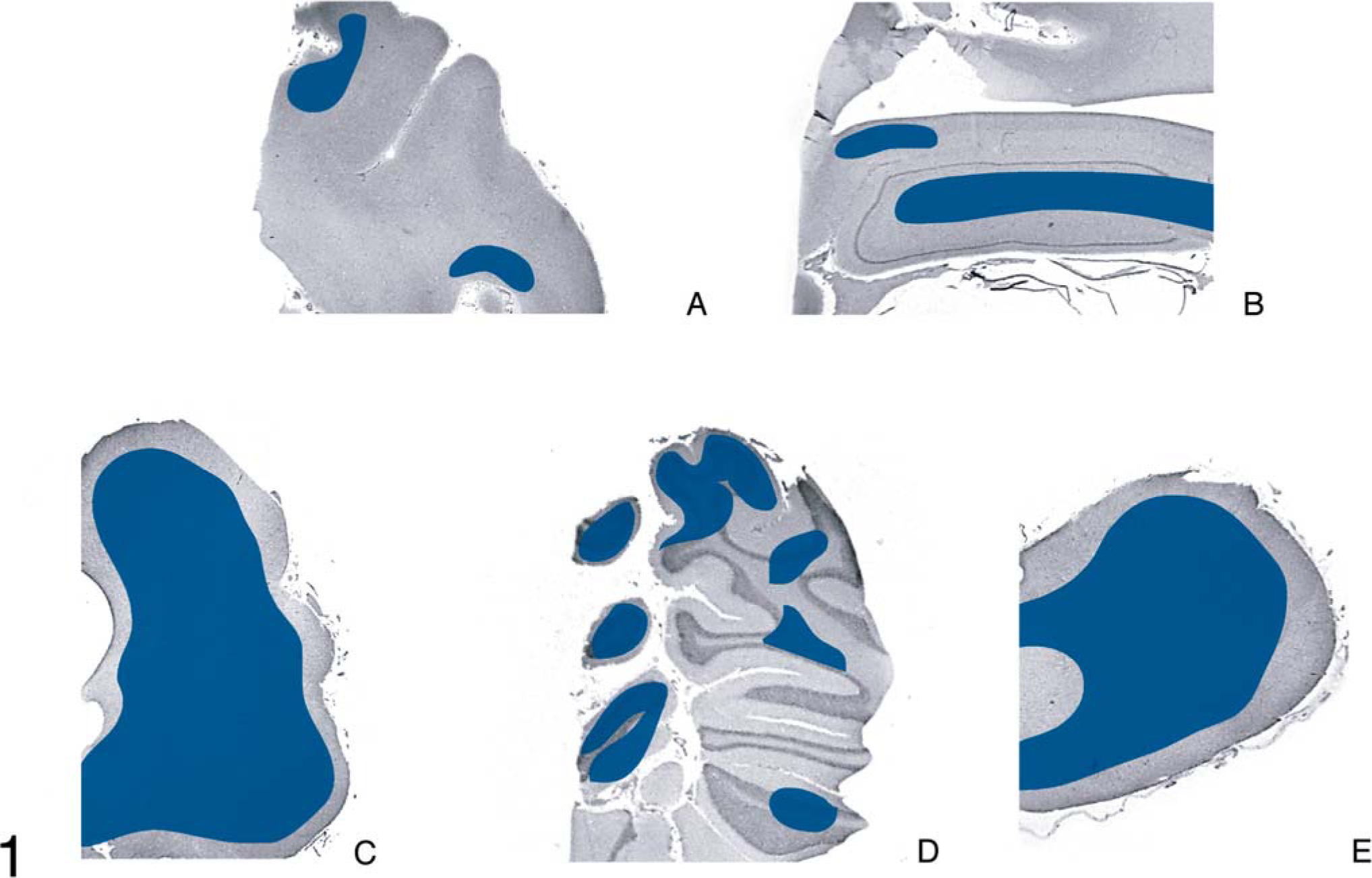
Calf; brain of animal A257 inoculated with TME from mink. HE. Bar = 140 µm. Medulla oblongata at the level of obex; Spinal vestibular tract nucleus. There is extensive vacuolation of the neuropil and presence of multiple vacuoles in neuronal perykaria (arrows).
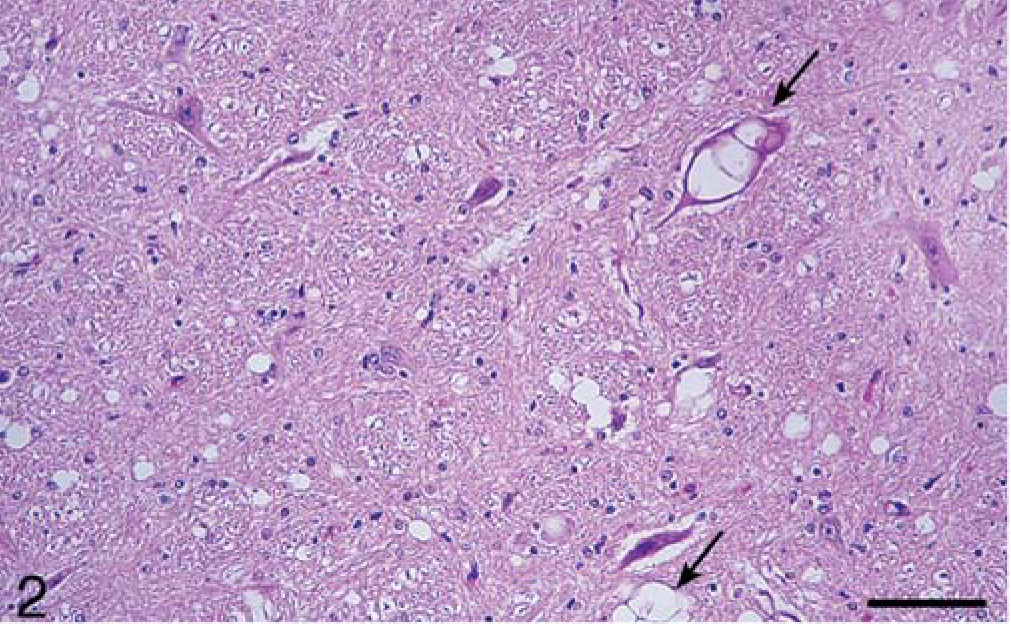
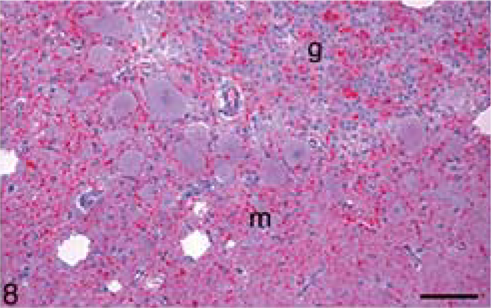
Calf; brain of animal A257 inoculated with TME from mink. HE. Bar = 140 µm. Cerebellum. Many large clear vacuoles are present in the molecular layer (m) and a few small vacuoles are in the granular layer (g).
Calf; brain of animal A257 inoculated with TME from mink. HE. Bar = 140 µm. Hippocampus. There is a focal area with scattered vacuoles in the neuropil.
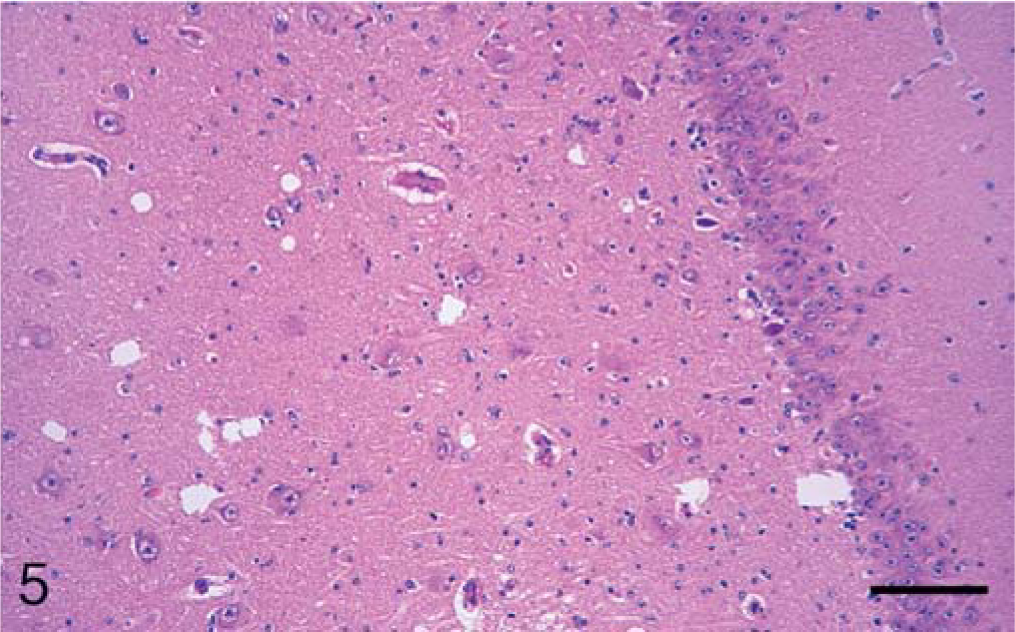
In the brain, SE lesions were variable in individual cases; however, in most animals, they were severe and diffuse in sections of colliculus and obex (Figs. 1, 2, and 4; Tables 2 and 3). At the former site, there were widespread, usually small (approximately 30–50 µm in diameter), clear vacuoles in neuropil (Fig. 4). In the obex, most of the dorsolateral and ventral areas were affected by similar vacuoles and were occasionally present in neuronal cytoplasm (Figs. 1 and 2). However, in all the inoculated cattle, the nucleus of the dorsal motor vagus was either not affected or showed minimal lesions of SE (Table 2). In the cerebellum, besides the presence of many larger (up to 80 µm in diameter) vacuoles in the neuropil of the molecular layer, there were similar but fewer vacuoles in the granular layer (Fig. 3). The Purkinje cell layer was generally unaffected. Except for a few sarcocysts in striated muscles, no significant lesions were present in the non-CNS tissues. Also, no significant abnormalities were present in any tissues of the two control animals.
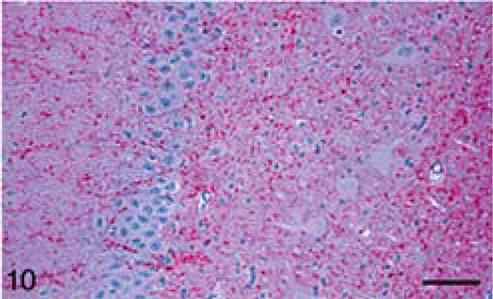
Calf; brain of animal A257 inoculated with TME from mink. HE. Bar = 140 µm. Midbrain at the level of superior calliculus. Many small and isolated large vacuoles are present throughout the tissue.
All sections of brain and spinal cord from inoculated animals revealed PrPres staining by IHC. The predominant pattern of staining in these tissues was diffuse, evenly distributed, punctate, and coarse granules that involved most areas of neuropil (Figs. 6–10). There were also some small aggregates of these particles but, unlike in the intracerebrally chronic wasting disease (CWD)-inoculated cattle, 11 large plaques of PrPres-stained foci were not seen. Perineuronal staining was present, but not common, and intracytoplasmic staining of neurons was quite rare. Perivascular staining was not observed. Anatomically, staining intensity was greatest in the midbrain (Fig. 9), but the medulla was also a frequent site of heavy and widespread immunoreactivity (Fig. 8). Although there typically was less staining of gray matter in hippocampus and rostral cerebrum, some of these tissues had a few large, individual, stained particles in white matter. In spinal cord, the staining intensity was greatest in dorsal horns. With respect to inoculum, no difference was noted between the two groups of cattle except that staining of cerebellum, both molecular and granular layers, was consistently observed only in cattle given TME of bovine origin (second passage). In contrast, three of the four cattle given TME from mink brain had no IHC reactivity in the cerebellum. Among all of the non-CNS tissues examined, including multiple lymphoid tissues, no staining was observed except in the plexiform layers of retina (seven of seven) and the pars nervosa of pituitary gland (three of eight). All positive pituitary glands were from cattle that had received the bovine TME inoculum.
Diagrammatic representation of the distribution of PrPres (in red) in 5 areas of the brain of animal A257 inoculated with TME from mink.
Calf; brain of animal A257 inoculated with TME from mink. Stained for PrPres (red) by IHC (immunoalkaline phosphatase) and counter-stained with hematoxylin. Bar = 140 µm. Medulla oblongata at the level of obex; dorsal nucleus of vagus. There is a presence of diffuse staining in the neuropil as well as some fine granular staining in the cytoplasm of neurons.
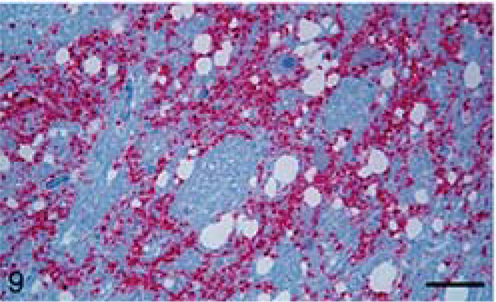
Calf; brain of animal A257 inoculated with TME from mink. Stained for PrPres (red) by IHC (immunoalkaline phosphatase) and counter-stained with hematoxylin. Bar = 140 µm. Cerebellum. PrPres is present in molecular (m) and granular (g) layers.
Calf; brain of animal A257 inoculated with TME from mink. Stained for PrPres (red) by IHC (immunoalkaline phosphatase) and counter-stained with hematoxylin. Bar = 140 µm. Midbrain at the level of calliculus. There is a presence of strong extensive staining throughout the section.
Calf; brain of animal A257 inoculated with TME from mink. Stained for PrPres (red) by IHC (immunoalkaline phosphatase) and counter-stained with hematoxylin. Bar = 140 µm. Hippocampus. Note the presence of diffuse staining in the neuropil but not in the neuronal cytoplasm.
PrPres was detected by WB analysis with monoclonal antibody 6H4 in all cattle with clinical CNS signs (Table 1, Fig. 11), whereas the two control animals were negative for PrPres (Table 1). The first and second passage of TME in cattle did not have obvious differences in molecular size of the respective PrPres isoforms (Fig. 11). The mink TME inoculum sample showed a lower molecular weight and the sheep scrapie sample a higher molecular weight of the unglycosylated isoform of the PrPres when compared with the cattle-passaged TME samples (Fig. 11). When monoclonal antibody P4 was used, only the positive sheep scrapie control reacted with this antibody (data not shown).
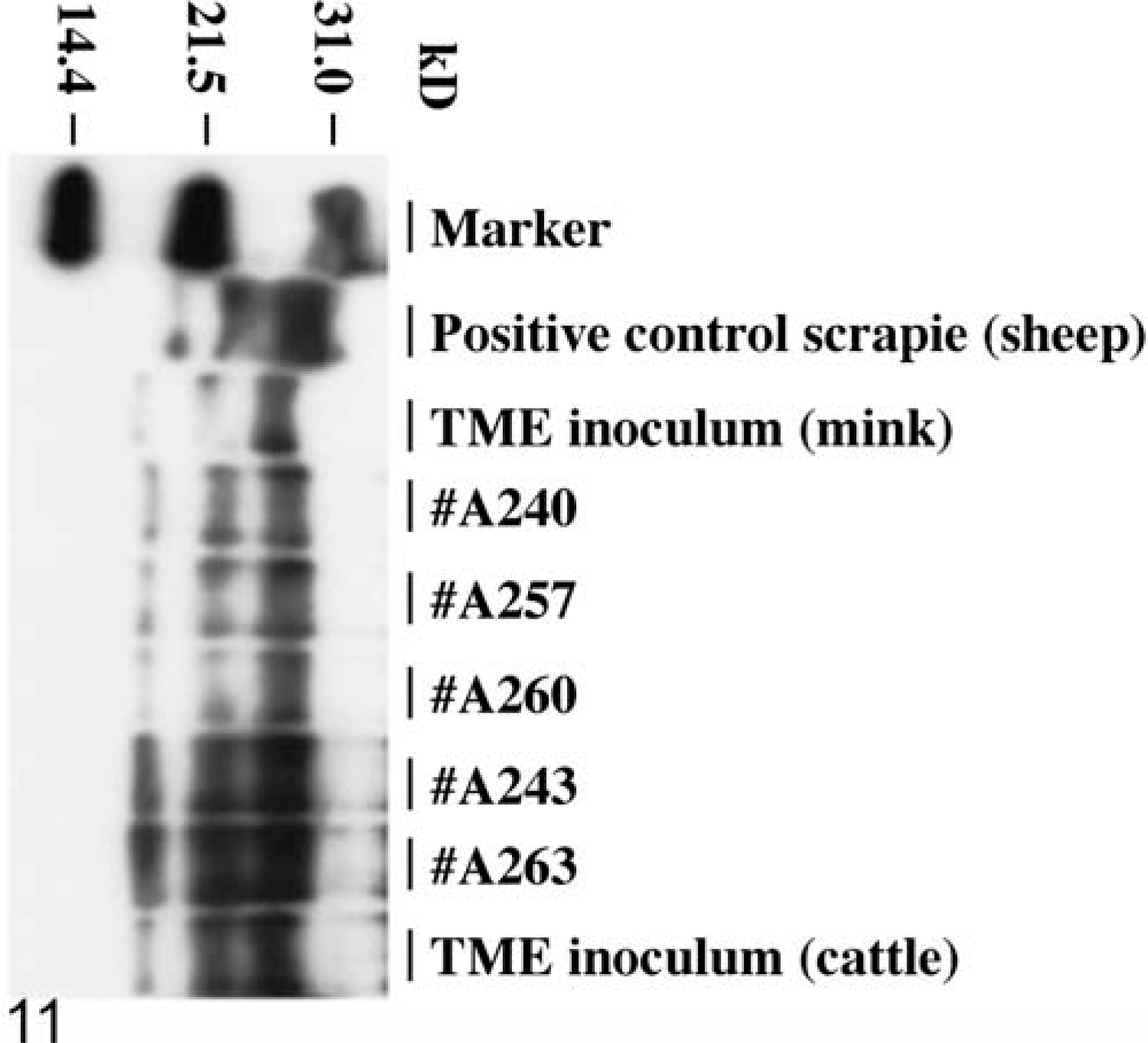
Hybrid WB analysis of brain homogenates. Analysis of brain homogenates with monoclonal antibody 6H4. Marker = protein weight marker; positive control scrapie (sheep) = brain homogenate of sheep with scrapie; TME inoculum (mink): mink TME used for inoculation of cattle; animals #A240, 257, 260: cattle infected with mink TME inoculum; animals #A243, 263: cattle infected with cattle-passaged TME inoculum; TME inoculum (cattle): cattle-passaged TME inoculum used for inoculation of cattle. All brain homogenates are positive showing the distinct profile of PrPres.
Discussion
TME is a food-borne disease of ranch-raised mink. Since 1947, there have been five outbreaks of the disease on 11 ranches in the United States. 20, 23 Three of these outbreaks were in Wisconsin and were attributed to the use of nonambulatory (downer) cattle carcasses in mink feed. 16, 19 TME, like BSE, was considered to have originated through the use of scrapie-infected sheep carcasses in mink feed. 19 However, experimental inoculation of mink with sheep scrapie failed to show clinicopathologic similarities to TME. 19 Also, intracerebral inoculation of sheep scrapie material into cattle produced neither the lesions of SE nor the IHC pattern of PrPres deposition characteristic of BSE. 5, 6 On the other hand, when TME was inoculated intracerebrally into cattle, microscopic lesions of SE were similar to those seen in BSE. 19, 23 At that time, the IHC findings of TME in cattle were mentioned but not described.
In the present investigation, there was no significant difference in incubation or survival periods between the bovine and mink inoculation groups. The bovine TME inoculum used in the present study was obtained from a steer (No. 2) that was intracerebrally inoculated with TME, developed spongiform encephalopathy, and was euthanatized at 19 months PI. 19 In a similar study, cattle inoculated intracerebrally with TME brain material succumbed to the disease and died or were euthanatized between 16.5 and 28.7 months PI. 23
The distribution of microscopic lesions of SE was variable in individual animals (Table 1). The lesions were not obviously different in the two inoculated groups of cattle (Table 3). They were generally more severe in the section of midbrain taken at the level of the superior colliculus, followed by brainstem (obex), and cerebellum. Sections of hippocampus and frontal cortex had multifocal areas with vacuoles in the neuropil (Table 3, Fig. 1).
Microscopic lesions of SE were present in the gray matter of brain and occasionally in spinal cord sections of all inoculated cattle (Table 1). The severity of lesions was not only variable between individual animals but also at various neuroanatomic sites (Table 3, Fig. 1). As indicated, spongiform change was most prominent in the area of the midbrain (Fig. 1). Although at most anatomic sites the majority of vacuoles were seen in the neuropil (Figs. 2–5), there were variable numbers of neurons at affected sites with either single or multiple vacuoles (Fig. 2). Generally, the vacuoles in the neuropil were round to oval and varied in size (30–50 µm in diameter). However, when present in the cerebellum, they were considerably larger in diameter—up to 80 µm in diameter (Fig. 3). There was minimal to mild gliosis in regions affected by spongiform change. Similar, but less severe, lesions were present in most nuclei of the brain stem and in gray matter of the spinal cord. However, in all animals, the dorsal vagal nuclei were either not affected or had minimal lesions. Also, neuronal degeneration (other than SE) was not a prominent feature in areas with spongiform change in the neuropil. Uninoculated control cattle did not have spongiform lesions in their CNS tissues.
The most prominent IHC staining pattern in TME-inoculated cattle (i.e., widespread, diffuse, particulate staining in gray matter neuropil; Figs. 7–10) was consistent with published descriptions of the IHC reaction in BSE cases. 27, 28 Although staining in neuronal cytoplasm is not unusual in BSE cases, 28 this type of IHC response was infrequently seen in TME-inoculated cattle. It has been shown that some IHC staining patterns, especially intraneuronal labeling, can be influenced by the primary antibody that is used. 18 It does not seem likely that the lack of neuronal staining in TME inoculates was an antibody phenomenon because the same monoclonal used in this study has been used to demonstrate specific intraneuronal staining in brains of cattle inoculated with scrapie. 6, 7 In comparing the two TME inocula used for this experiment, mink or bovine brain origin, the only difference noted was a somewhat broader tissue distribution of immunoreactivity in cattle that received the bovine-passaged TME. In all cases with second passage of TME in cattle, there was staining in cerebellum and also in three of four pituitary glands. In contrast, only one of four primary passages of TME in cattle (mink-origin inoculum) had cerebellar staining and none had a positive reaction in pituitary gland. These findings may reflect adaptation of the mink agent to a new species, although the small differences of incubation period between primary and second passage suggest that the mink–cattle species barrier for TME is very weak.
In the present experiment, there was extensive multifocal distribution of PrPres throughout the brain and also in the spinal cords of all inoculated animals. This indicates that there was de novo deposition of PrPres because, in experimental studies with sheep scrapie, it has been shown that intracerebrally inoculated brain material containing PrPres is present in large enough quantity to be detected only for the first few days PI. 15
WB analyses revealed that all cattle inoculated with either mink or cattle TME were positive for PrPres, showing a clear reaction with the three protease-resistant polypeptide isoforms (di-, mono-, and unglycosylated). The first and second passages of TME in cattle show similar molecular weights of the three PrPres isoforms. Interestingly, the molecular weight of cattle-passaged TME was lower than the respective PrPres isoforms of sheep scrapie. It has been shown previously that PrPres in brain stem homogenates from cattle infected with BSE had reproducible lower molecular masses for each of the PrPres isoforms than sheep infected with scrapie. 2 Our results suggest that cattle-passaged TME also has a lower molecular weight when compared with sheep scrapie. When using a hybrid WB analysis employing two different PrP-specific antibodies (6H4 and P4), cattle BSE could be easily distinguished from sheep scrapie 25 because it does not react with mAb P4. Similarly, cattle-passaged TME did not react with mAb P4 and therefore exhibits a BSE-like reaction pattern in the hybrid WB analysis. Additional studies are needed where cattle-passaged TME samples are analyzed in parallel with cattle BSE samples to directly compare their molecular features. Such analyses are planned in the near future.
Although intracerebral inoculation is an unnatural route for exposure of cattle to TME infection, this experiment shows that TME, like scrapie and CWD, 6, 7, 11 has potential for transmission to this species. TME isolates from other geographical locations may differ in their ability to amplify in cattle and, therefore, transmission studies using different TME isolates are needed.
It is likely that transmission of TME to cattle by natural exposure routes, such as per os, would be more difficult to accomplish than the intracerebral transmissions reported here. Although it may require a larger dose of inoculum and longer incubation time, it could be speculated that, given the 100% attack rate of TME in cattle by the intracerebral route, the disease may be able to develop in cattle when administered orally. In such an investigation, the clinicopathologic findings may be different from the present study. However, a previous study with BSE has shown that experimental routes of inoculation had little effect on the distribution of SE lesions. 29 Therefore, in the USA, the current testing for BSE would be expected to identify cattle with TME (should it occur in this country) as BSE cases. Given the similarity of clinical signs, the presence of severe microscopic evidence of SE, the pattern of IHC distribution of PrPres, and WB findings, it would be difficult to differentiate this disease from BSE.
TME agent is difficult to transmit to mice, 19, 26 whereas the BSE agent can be transmitted relatively easily to mice. 4 On the other hand, TME may be able to infect mice after passage in cattle. Such a possibility needs to be investigated under experimental conditions. Recently, the raccoon model has been shown to have potential for differentiating between TME and scrapie on the basis of incubation time and IHC localization. 12– 14 However, as yet, raccoons have not been inoculated with BSE to determine if there are any distinguishing features of BSE infection in that species. Such an experimental study is in the planning stage and will be initiated in the near future at this Center.
Addendum
Since writing the above article we have compared two BSE samples (from the 2003 and 2004 US BSE cases) and a cattle TME sample in Western blot analyses. Preliminary results indicate that the molecular phenotype of cattle TME appears to be different from both US BSE isolates, i.e., cattle TME exhibits a lower molecular weight of the unglycosylated form of the abnormal prion protein.
Footnotes
Acknowledgements
We thank Drs. Lawayne Nusz and Jean Laufer for clinical assistance with the experimental animals. Martha Church, Semakaleng Lebepe-Mazur, Dennis Orcutt, Jean Donald, Sharla Van Roekel, and animal handlers at NADC provided expert technical assistance.
This study was carried out under the guidelines of the institutional ACUC at NADC. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
