Abstract
Scrapie is a naturally occurring fatal neurodegenerative disease of sheep and goats. Susceptibility to the disease is partly dependent upon the genetic makeup of the host. In a recent study, it was shown that sheep intracerebrally inoculated with a US scrapie agent (No. 13–7) developed scrapie and survived for an average of 19 months post inoculation. In the present study, when this scrapie inoculum was further passaged for 3 successive generations, the survival time was reduced by approximately 8 months in scrapie-susceptible (QQ on prion protein gene [PRNP] at codon 171) Suffolk sheep. It is concluded that inoculum No. 13–7 appears to have been stabilized in susceptible (171 QQ) Suffolk sheep and may be considered a specific isolate of sheep scrapie agent in the USA and therefore that it can be used to evaluate other isolates of sheep scrapie in this country.
Keywords
Scrapie is a naturally occurring fatal neurodegenerative disease of sheep and goats. The susceptibility of sheep to scrapie is dependent on genetic variation of the host prion protein (PRNP) gene. 1, 4, 10 PRNP genotypes are defined by variations in the amino acids located at codons 136, 154, and 171. At least 5 variant alleles have been found, and they are depicted as ARQ, ARR, VRQ, AHQ, and ARH. The codes represent amino acids at codons 136, 154, and 171, that is, A-136, R-154, and Q-171 (ARQ), where A = alanine, R = arginine, Q = glutamine, H = histidine, and V = valine. The level of risk to contract scrapie varies depending on breed and the genotypes found within the flock; ARR are the most resistant, and VRQ are the most susceptible. 1, 4, 10 A study of Suffolk sheep in the United States found 61% of orally inoculated animals developed scrapie. 13 All were homozygous for glutamine (QQ) at allele 171 on PRNP gene. 13
In a recent study, it was shown that sheep intracerebrally inoculated with US scrapie agent (inoculum No. 13-7) developed disease within an average of 19 months post inoculation (MPI). 6 This inoculum was made with brain tissue of 13 sheep from 7 different source flocks. 6
This study documents incubation times, pathologic findings, and distribution of abnormal prion protein (PrPSc) by immunohistochemical (IHC) and Western blot (WB) techniques in tissues of genetically susceptible Suffolk sheep (QQ or QH at codon 171 of PRNP gene) that were intracerebrally inoculated with 3 consecutive passages of inoculum No. 13-7 (Table 1). The objectives of the study were to reduce the incubation (postinoculation) time of the scrapie inoculum and to stabilize the inoculum so that it can be used to evaluate other isolates of sheep scrapie in this country.
Postinoculation to terminal disease (survival) time after passage of scrapie inoculum (No. 13-7) by intracerebral inoculation to 4 generations of Suffolk sheep that were genetically susceptible (QQ or QH at PRNP codon 171) to scrapie.
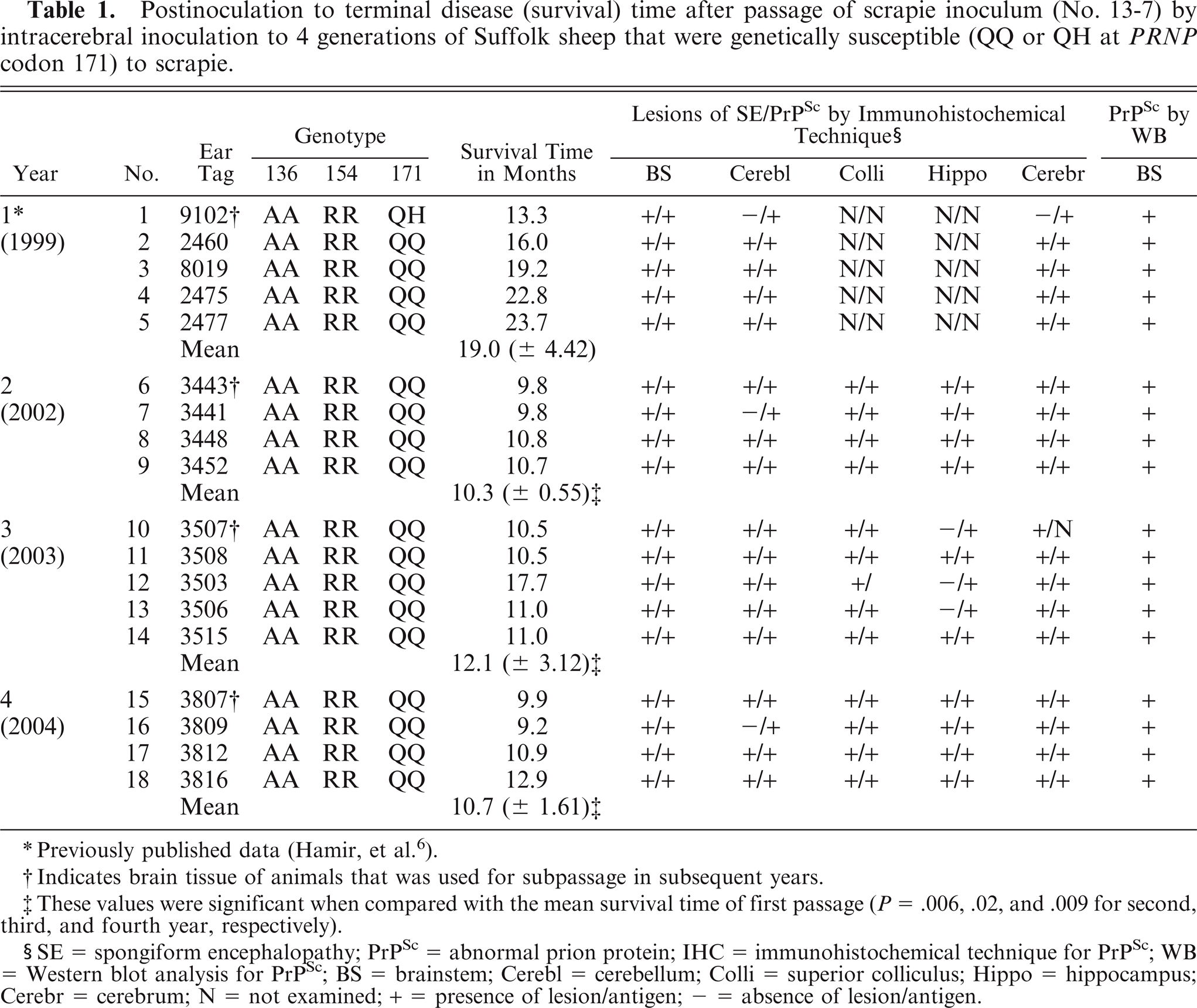
Previously published data (Hamir, et al.6).
Indicates brain tissue of animals that was used for subpassage in subsequent years.
These values were significant when compared with the mean survival time of first passage (P = .006, .02, and .009 for second, third, and fourth year, respectively).
SE = spongiform encephalopathy; PrPSc = abnormal prion protein; IHC = immunohistochemical technique for PrPSc; WB = Western blot analysis for PrPSc; BS = brainstem; Cerebl = cerebellum; Colli = superior colliculus; Hippo = hippocampus; Cerebr = cerebrum; N = not examined; + = presence of lesion/antigen; – = absence of lesion/antigen.
Materials and Methods
A total of 18 Suffolk lambs (females and castrated males) from National Animal Disease Center's (NADC) scrapie-free flock were obtained for this study and were inoculated with scrapie when they were approximately 4 months of age. All except 1 sheep (No. 9102, Table 1) were AA/RR/QQ at codons 136, 154, and 171, respectively (Table 1).
The scrapie inoculum (No. 13-7) was passaged in 4 generations of lambs (from 1999 to 2004) by intracerebral inoculations (Table 1). Originally this inoculum was prepared from a pool of 13 scrapie-affected sheep brains (all were positive by IHC technique) from 7 source flocks. 9 The inoculum was ground in a mechanical grinder, gentamicin was added at 100 μg/ml, and the final concentration of 10% (wt/vol) was made with phosphate-buffered saline. For subsequent passages, the scrapie-infected brain tissue was obtained from the animal with the shortest incubation to terminal disease (survival time) from the previous passage, and the inoculum was prepared as described (Table 1).
The procedure for intracerebral inoculation of lambs has been described previously. 9 Briefly, the animals were sedated with xylazine, a midline incision was made in the skin at the junction of the parietal and frontal bones, and a 1-mm hole was drilled through the calvarium. The inoculum (1 ml of 10% w/v brain suspension) was injected into the midbrain via a 22-gauge, 9-cm-long needle while withdrawing the needle from the brain. The skin incision was closed with a single suture.
Inoculated animals were initially housed in biosafety level–2 facilities for 2 weeks PI and then were transferred to outside pens. They were fed pelleted growth and maintenance rations that contained no ruminant protein, and clean water was available ad libitum.
The sheep were killed when they showed advanced clinical signs of scrapie. A detailed necropsy was carried out on each animal, and 2 sets of tissue samples were collected. The tissues were representative samples of nasal mucosa, liver, spleen, salivary gland, intestines (ileum), adrenal gland, pancreas, lymph nodes (retropharyngeal, mesenteric), tonsils (pharyngeal, palatine), striated muscles (heart, tongue, masseter, diaphragm, psoas major), eye, trigeminal ganglion, pituitary gland, and one half of brain, all of which were immersion fixed in 10% neutral-buffered formalin. The second set of tissues was frozen (−20°C). The formalin-fixed brain was cut into 2- to 4-mm-wide coronal sections. Sections of various anatomic sites (a minimum of 5 brain sections per animal) of rostral cerebrum, hippocampus, superior colliculus, cerebellum, and brainstem (at the level of obex) were cut for routine histopathology. Formalin-fixed tissues were embedded in paraffin wax, sectioned at 5 μm, and stained with hematoxylin and eosin for light microscopy. All paraffin-embedded tissues were also stained by an automated IHC method for detection of PrPSc, as described previously. 6 Briefly, after deparaffinization and rehydration, tissue sections were autoclaved for 30 minutes in an antigen retrieval solution (DAKO Target Retrieval Solution, DAKO Corp., Carpinteria, CA) and stained with an indirect avidin biotin system (Basic Alkaline Phosphatase Red Detection Kit, Ventana Medical Systems, Inc., Tucson, AZ) designed for an automated immunostainer (NexES IHC module, Ventana). The primary antibody was a cocktail of 2 monoclonal antibodies, F89/160.1.5 12 and F99/97.6.1, 11 each used at a concentration of 5 μg/ml, and incubation was carried out at 37°C for 32 minutes. The secondary antibody was biotinylated anti-mouse immunoglobulin G ([made in horse], Vector Laboratories, Burlingame, CA) diluted 1 : 200 and incubated for 8 minutes at 37°C.
The frozen brain was used for immunodetection of PrPSc by a commercial WB (Prionics Check Western blot, Prionics AG, Schlieren, Switzerland), as described previously. 6 Briefly, the modification consisted of the use of a secondary biotinylated anti-mouse immunoglobulin G antibody (dilution 1 : 10,000) (Amersham Biosciences, Piscataway, NJ) followed by a streptavidin horseradish-peroxidase conjugate (dilution 1 : 10,000) (Amersham). The blot was developed with an ECL Plus chemiluminescence solution (Amersham) before being exposed to film.
Results
All scrapie-inoculated sheep developed clinical disease and were killed between 9.2 and 23.7 MPI (Table 1). The individual and mean survival times of the inoculated sheep are given in Table 1.
Clinical signs of affected sheep consisted of a progressive decrease in appetite with associated weight loss. Later, the affected sheep developed fine head tremors, listlessness, progressive problems with locomotion, and terminal sternal recumbency. None of the affected sheep exhibited obvious pruritus or loss of wool from their fleece.
Apart from fair to poor nutritional condition of the carcasses, significant gross lesions were not seen in any of the examined animals. The presence of neurohistopathologic lesions (spongiform encephalopathy), and the results of PrPSc by IHC and WB assays are given in Table 1 and shown in Figs. 1 to 3. Severe spongiform lesions were consistently present in the sections of brainstem (obex), colliculi, cerebellum, and cerebrum. The lesions consisted of vacuolations in the neuropil and within neuronal perikarya (Fig. 1). They were most severe in the nucleus of the dorsal vagus. In the affected areas, there was a mild increase in the number of glial cells. However, neuronal degeneration was not seen in any of the sections. No significant differences were observed in the degree and the distribution of morphologic lesions in the various subpassaged sheep.
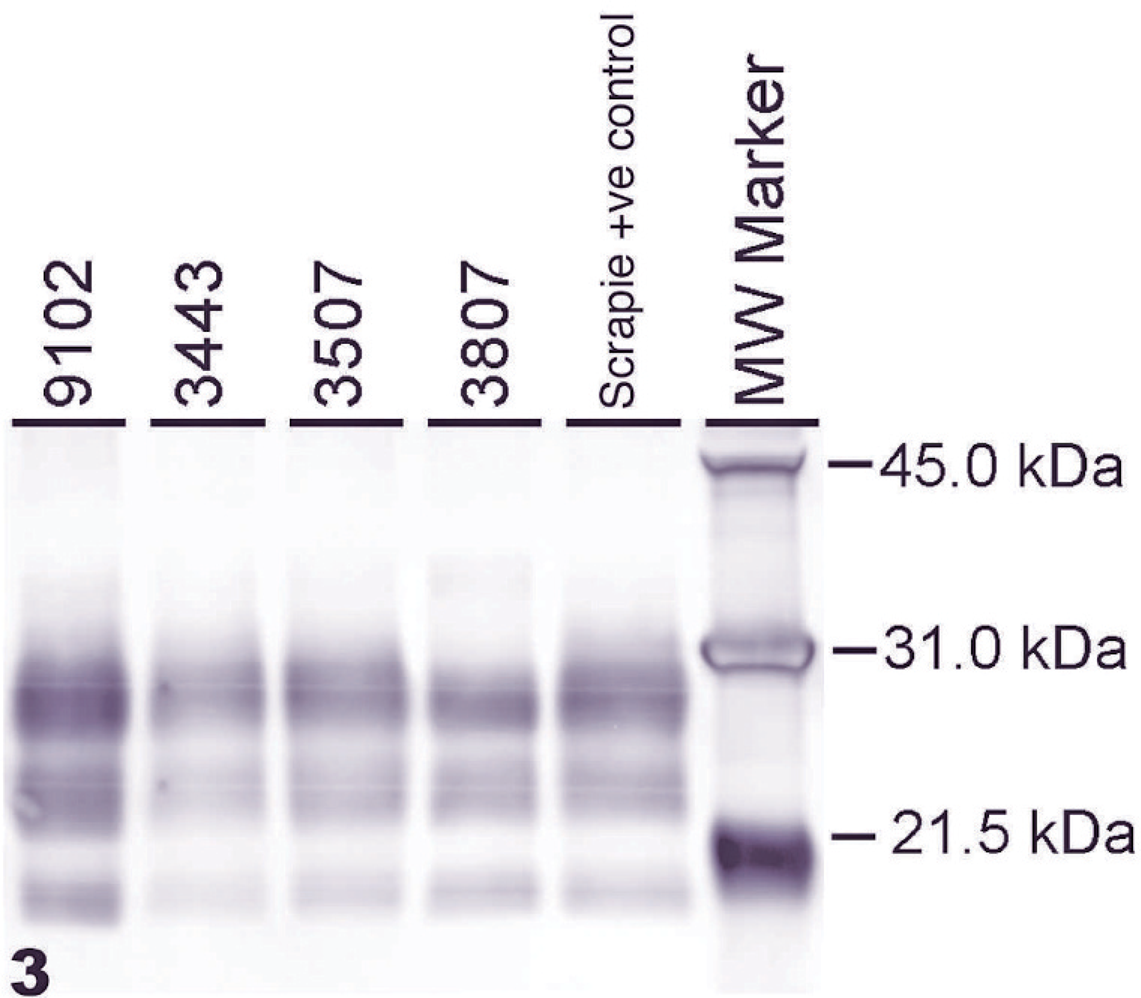
Medulla oblongata at the level of obex of sheep No. 11 (3508) with clinical scrapie. There is extensive vacuolation of the neuropil and presence of multiple vacuoles in perikarya of 1 neuron. HE stain. Bar = 50 μm.Fig. 2. Medulla oblongata at the level of obex of sheep No. 11 (3508) with clinical scrapie. There is extensive diffuse abnormal prion protein (PrPSc) labeling in the neuropil. Also, there is some PrPSc labeling within neurons. Stained for PrPSc labeling (red) by immunohistochemical technique (immunoalkaline phosphatase) and counterstained with hematoxylin. Bar = 50 μm.Fig. 3. Western blot analysis of representative brainstem samples from scrapie-infected sheep. Samples were probed with monoclonal antibody P4 at 0.1 μg/ml and loaded at 0.06 mg per lane, except 9102, which was loaded at 0.03 mg. Scrapie-positive control and molecular weight markers in kDa are indicated on the right.
Immunohistochemical labeling of PrPSc in central nervous system and lymphoid tissues was consistently seen in all scrapie-affected sheep (Fig. 2, Table 2). No differences in IHC labeling patterns or PrPSc tissue distribution were seen between successive subpassage groups. Multifocal punctate and coalescing PrPSc labeling of central nervous system neuropil with prominent perineuronal accumulations was typically seen. Labeling of neuronal perikarya was less intense on average and punctuate in character, and the only localization site of PrPSc was in Gasserian ganglia and myenteric plexi (2 of the peripheral nervous system sites examined). The plexiform layers of retina contained confluent accumulations, and receptor, nuclear, and ganglion cell layers had multifocal punctuate labeling of PrPSc. The majority of PrPSc in lymphoid tissues was present in germinal centers of lymphoid follicles, with lesser amounts in the interfollicular/paracortical areas. In the spleen, the marginal zones of periarteriolar lymphoid sheaths were often conspicuously labeled as well.
Results of examination of tissues by immumohistochemical technique for the presence/absence of abnormal prion protein labeling.

Numbers in parenthesis indicate numbers positive for abnormal prion protein/total numbers examined.
Western blot analyses using brainstem samples from all animals enrolled in the study were performed. When 0.5 mg brain equivalent was analyzed, all samples from scrapie-infected sheep were unambiguously positive, whereas the control samples were negative (Table 1). Positive samples show a WB pattern with the presence of all 3 isoforms (di-, mono-, and unglycosylated forms) of the protease-resistant PrPSc (Fig. 3). When representative samples from sheep inoculated with first, second, third, and fourth passage of the scrapie strain were compared side by side, no significant differences in the molecular weight of the 3 protease-resistant isoforms of the PrPSc were noted (Fig. 3). Likewise, a glycoform profile comparison of the first, second, third, and fourth passages of the scrapie material revealed a similar prevalence of the individual isoforms. The prevalence of the di-glycosylated isoform of scrapie passages 1 to 5 ranged from 61 to 69%, the prevalence of the mono-glycosylated isoform from 23 to 29%, and the prevalence of the unglycosylated isoform from 7 to 11%.
Discussion
In sheep, the PRNP gene determines relative resistance or susceptibility to scrapie. Suffolks with QQ or HQ at codon 171 are considered highly susceptible to scrapie. 1, 4, 10
The clinical signs of scrapie vary widely in individual sheep. In general, the early signs are subtle changes in behavior or temperament followed by obvious progressive neurologic signs, and in terminal cases there is often weight loss and debilitation. 4 In the presently described clinical cases, reduced dietary intake with subsequent weight loss and terminal debilitation and recumbency were the predominant signs.
In a previous study, when a pool of scrapie inoculum (No. 13-7) was injected directly into the brain of Suffolk sheep, a long incubation time was required and the mean survival time for these sheep was 19 MPI. 6 However, in the present study, when scrapie from individual animals was passaged, the survival time was reduced to between 10 and 12 MPI (Table 1). Because incubation time observed using standardized inoculum (1% brain homogenate) has been used as a reliable marker for distinguishing prion strains, 14 the results of the present study indicate that inoculum No. 13-7 had achieved stabilization by the second passage. Although up to 20 different scrapie strains have been identified in the UK, 2 no such systematic attempt has been made to identify strains in North America. For further characterization of inoculum 13-7, several strains of inbred mice have been inoculated, and this study is in progress at NADC.
The findings of IHC analyses were typical for sheep scrapie and were unchanged through successive subpassages, and the representative samples from sheep inoculated with first, second, third, and fourth passage of the scrapie strain revealed no obvious differences in the molecular weight and prevalence of the 3 protease-resistant isoforms of the PrPSc (Fig. 3). This might indicate that 1 prevalent scrapie strain in a mixture of strains present in the pooled 13-7 scrapie inoculum was selectively propagated in Suffolk QQ sheep. Whether this was the prevalent strain of inoculum 13-7 cannot be determined at this time. However, molecular comparison between inoculum 13-7 and passages 1 to 5 did not reveal significant differences in the molecular weight of the protease-resistant isoforms between the different isolates (data not shown).
Results of this investigation show that by serial passage of the inoculum, a considerable shortening of the survival times of sheep was achieved. Several mechanisms could be responsible for these shortened times. The most important would be that the serial passage of the inoculum could have selectively isolated a single virulent scrapie strain from a heterogeneous pool (original inoculum), thereby leading to a higher infectious dose of challenge by that strain. Other factors contributing would be the adaptation to the intracerebral route used; also, the year-to-year technical variation in the infectious protocol, clinical assessment, or endpoint determinations could have contributed to the shortened observed period.
Currently, the available methods for routine diagnosis of scrapie are demonstration of characteristic histologic changes and detection of PrPSc by IHC and WB analyses in the brains of affected animals. 8 In the present study, all 3 techniques were comparable for diagnosis of scrapie in animals with terminal disease.
There is a lack of information on experimental transmission of US scrapie agent in genetically diverse flocks of sheep. Our previous studies 5– 7 and the present investigation attempts to partially fill this void by documenting the pathogenesis and results of serial passage of inoculum No. 13-7 in Suffolk sheep. Recently, another scrapie inoculum (No. x124) has been identified, and it appears to have a considerably shorter incubation period (5.6 MPI) in Suffolk sheep with AV and QQ at codon 136 and 171, respectively. 3 In that study, sheep with VV at codon 136 and QQ at codon 171 had an even shorter survival period (4.3 MPI). Studies similar to the present investigation are needed for inoculum No. x124 to investigate the possibility of reducing the PI period to a couple of months by subpassaging in sheep.
It is concluded that inoculum No. 13-7 appears to have been stabilized in susceptible (171 QQ) Suffolk sheep. This isolate was originally derived from a pool of scrapie-affected sheep from several flocks and therefore was most likely a mixture of heterogeneous strains of scrapie. After several passages, the inoculum may now be considered a specific isolate of sheep scrapie agent in the USA, and therefore it can be used to evaluate other isolates of sheep scrapie in this country.
Footnotes
Acknowledgements
We thank Drs. L. Nusz and J. Laufer for clinical assistance; Dr. Marcus Kehrli Jr, for constructive comments and for his enthusiastic support, and James Fosse for the photomicrographs. Expert technical assistance was provided by Martha Church, Dennis Orcutt, Joseph Lesan, Trudy Tatum, Deb Clouser, Kevin Hassall, Ginny Montgomery, Micky Fenneman, and the transmissible spongiform encephalopathy animal caretakers. This study was carried out under the guidelines of the institutional ACUC committee at NADC. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
