Abstract
Prion diseases or transmissible spongiform encephalopathies (TSEs) of animals include scrapie of sheep and goats; transmissible mink encephalopathy (TME); chronic wasting disease (CWD) of deer, elk and moose; and bovine spongiform encephalopathy (BSE) of cattle. The emergence of BSE and its spread to human beings in the form of variant Creutzfeldt-Jakob disease (vCJD) resulted in interest in susceptibility of cattle to CWD, TME and scrapie. Experimental cross-species transmission of TSE agents provides valuable information for potential host ranges of known TSEs. Some interspecies transmission studies have been conducted by inoculating disease-causing prions intracerebrally (IC) rather than orally; the latter is generally effective in intraspecies transmission studies and is considered a natural route by which animals acquire TSEs. The “species barrier” concept for TSEs resulted from unsuccessful interspecies oral transmission attempts. Oral inoculation of prions mimics the natural disease pathogenesis route whereas IC inoculation is rather artificial; however, it is very efficient since it requires smaller dosage of inoculum, and typically results in higher attack rates and reduces incubation time compared to oral transmission. A species resistant to a TSE by IC inoculation would have negligible potential for successful oral transmission. To date, results indicate that cattle are susceptible to IC inoculation of scrapie, TME, and CWD but it is only when inoculated with TME do they develop spongiform lesions or clinical disease similar to BSE. Importantly, cattle are resistant to oral transmission of scrapie or CWD; susceptibility of cattle to oral transmission of TME is not yet determined.
Keywords
Introduction
Transmissible spongiform encephalopathies (TSEs) are fatal neurologic diseases that affect several mammalian species including human beings. Four animal TSE agents have been reported: scrapie of sheep and goats; chronic wasting disease (CWD) of deer, elk, and moose; transmissible mink encephalopathy (TME) and bovine spongiform encephalopathy (BSE). In comparison with contagious bacterial, viral, and parasitic infectious diseases, TSEs typically do not present with high morbidity or mortality in livestock, wildlife, or human populations. The TSEs, however, remain important because of public health and international or domestic trade issues involving movement of animals. In response to the discovery of BSE, governments around the world began investing in research to determine the origin of BSE and the host range of the recognized TSEs. The prevailing theory at the time of the BSE discovery was that it had resulted from transmission of scrapie from sheep to cattle. 113 Once the original interspecies transmission event had occurred it was then amplified by the subsequent feeding of meat and bone meal (MBM), a supplement that normally contains central nervous system (CNS) tissues, which inevitably became contaminated with CNS tissues from BSE affected cattle. Such practice precipitated more BSE cases, thus resulting in greater volumes of contaminated MBM supplement assisted by growing inventories of contaminated MBM prior to its discovery. To test the hypothesis that BSE arose from scrapie or another animal prion disease, several experimental interspecies transmission studies have now been completed and reported. Collectively, some interesting observations have emerged and the findings of the studies conducted to date are summarized herein.
Etiology and types of animal transmissible spongiform encephalopathies
The TSEs, a class of progressive and fatal neurodegenerative diseases, are caused by accumulation of abnormally folded disease-associated prion protein (PrPd). 82 Prion (proteinaceous infectious particles; pronounced pree-on) was the term given for this unique agent when it was defined as the cause of scrapie. 81 Infectious prions are composed largely of chromosomally encoded proteins that have undergone a conformational change such that the new altered conformation is capable of causing a conformational change in additional molecules of the normal prion protein. In all TSEs the normal cellular isoform of the prion protein (PrPC) undergoes a conversion to a relatively protease-resistant isoform (PrPRes). The disease-associated conformation of the protein is also referred to as PrPSc (denoting the association with scrapie) and PrPd (indicating the disease-associated form), as well as, PrPCWD, PrPBSE, and PrPTME denoting the host origin, or PrPTSE as a species neutral designation. 14 For the purposes of the current review, when referring to the disease-associated form of PrP for BSE, CWD, scrapie, and TME, the abbreviation PrPd will be used. Such abbreviation also eliminates the potential misconception that all PrPRes are capable of causing a prion disease 118 and that only PrPRes is causing disease. 57
It is critical to understand that not all forms of prions are associated with disease. The PrPC is present in all mammals as a host cellular protein whose normal physiological function is not completely understood but is purported to play roles in metal homeostasis, neuroprotective signaling, cellular responses to oxidative stress and as a mediator of amyloid β oligomer induced synaptic dysfunction. 65 The bovine prion protein (PRNP) gene encodes PrPC as an approximately 265 amino acid polypeptide with significant sequence homology among mammalian species. In its normal glycosylphosphatidylinositol (GPI)-anchored membrane bound form, bovine PrPC is processed by host cells at both the amino and carboxyl termini, resulting in an approximately 220 amino acid protein that is typically linked to cells by a GPI anchor that associates with detergent-resistant membranes or rafts. 4,21
The concept of an infectious protein contradicts much of what is typically understood about infectious diseases. According to the protein only hypothesis, PrPd can convert PrPC to the PrPd conformation. While controversy has surrounded the protein only hypothesis, recent results indicate that bacterially derived recombinant PrP may be folded to a disease-causing conformation in vitro, thus providing strong support for the concept of an infectious protein. 67 The PrPC to PrPd conversion process is believed to be purely a 3-dimensional structural alteration, where the largely unstructured regions of PrPC adopt the PrPd conformation, containing substantially higher β-sheet content. 82 Such structural change underlies the disease process and gives rise to enhanced resistance to proteolysis, an important aspect of most current diagnostic methods. The PrPd accumulates in specific tissues of an affected host, eventually leading to neurodegeneration and disease. The following is a brief description of the different naturally occurring animal TSEs in their natural hosts as a basis of comparison for the description of experimental transmission studies of naturally occurring TSEs into cattle.
Scrapie in sheep and goats
Scrapie of sheep and goats was the first spongiform encephalopathy for which transmissibility was demonstrated. The disease was initially reported in the 1700s and to date there is no evidence supporting the transmissibility of scrapie to human beings. 84,90 Clinical signs begin with impaired social behavior, restlessness, and nervousness. As the disease progresses, the condition of the animal deteriorates. The scratching behavior associated with the name of the disease may result in loss of wool in a small area or perhaps even an entire side of the body. Ultimately, ataxia may become pronounced, and the sheep become highly agitated by even minor stress. 84 In a clinically affected animal, common neuropathologic lesions in the brain include spongiform change (vacuoles in neurons and neuropil) and astrogliosis. 84
Following exposure, PrPd often accumulates in lymphoid tissues before spreading to the CNS. 32 In clinically diseased sheep, there is widespread PrPd accumulation within tingible body macrophages and in follicular dendritic cells of secondary lymphoid follicles. 60 Studies in scrapie-affected sheep have identified several types of retinal cells including Müller glia, 36 retinal bipolar neuronal cells, and a subset of retinal ganglion cells affected by PrPd accumulation. 96 Further, these cell type–specific changes are associated with abnormalities in retinal function in sheep with clinical disease. 95 In sheep, following oral exposure, PrPd crosses the intact intestinal barrier at the level of the enterocytes and passes rapidly into lymph and blood. These initial steps are identical in susceptible and resistant sheep; however, only in susceptible sheep has PrPd accumulation been shown to subsequently take place in lymphoid structures (particularly in association with follicular dendritic cells). 2,33,46–49,58,80
An atypical scrapie strain was first discovered in Norway 8 in 1998 and subsequently in many European countries with active scrapie surveillance programs and in the United States. The PrPd deposition in atypical scrapie cases occurs predominantly in the cerebellum rather than the medulla oblongata as seen in classical sheep scrapie, and has not yet been detected in lymphoid tissues. The PrPd molecule of atypical scrapie is relatively sensitive to proteinases, resulting in discrepant diagnostic test results depending on the test method used. 6 Atypical scrapie cases are sometimes identified in sheep with genotypes considered resistant to classical scrapie. 66 Because the disorder is typically detected in only one sheep in a flock and is usually found in older animals, a sporadic etiology (i.e., spontaneous occurrence or de novo pathogenesis) has been suggested. 35,50 Supporting the concept of a sporadic occurrence is the long held categorization of certain forms of CJD having a sporadic etiology and recent evidence that abnormally folded prions can arise de novo in experimental systems. 31
Chronic wasting disease of cervids
Naturally occurring CWD has been documented in mule deer (Odocoileus hemionus hemionus), black-tailed deer (Odocoileus hemionus columbianus), white-tailed deer (Odocoileus virginianus), Rocky Mountain elk (Cervus elaphus nelsoni) and moose (Alces alces shirasi). 3,115 The disease was first recognized in a captive population of mule deer at the Colorado Division of Wildlife Foothills Wildlife Research Facility in Fort Collins during 1967, although identification of the disease as a TSE did not occur until 1978. 84,115 Chronic wasting disease can be spread horizontally with an efficiency sufficient to sustain local persistence of CWD in natural populations 74 and no defined route of transmission has been determined to date, 75 although urine, feces, saliva, velvet, lymphoid tissues and blood of CWD-affected animals have been found to carry PrPd. Recent studies report experimental transmission via saliva and blood is possible. 73 Testing of hunter-harvested animals indicates that between approximately 1% and 20% of animals in free ranging deer populations in endemic areas may be affected. Natural transmission may be associated with prions shed in saliva followed by oral uptake during deer-to-deer social interactions or contamination of grazing areas. 76 The most prominent clinical sign of CWD is the basis for its name, namely progressive debilitation and eventual emaciation. Some animals may show hypersalivation and difficulty swallowing; elk, in particular, exhibit ataxia and tremors. 115 However, reports of carnivorous behavior of deer consuming cervid entrails left behind by hunter harvest of deer or natural causes merits further investigation as a potentially significant factor in horizontal transmission between cervids (Pete Squibb, Consultant Wildlife Solutions LLC, Potterville, Michigan, personal communication). The most widely accepted hypothesis at this time is that CWD may have originated from an interspecies transmission of scrapie. It is worth noting that experimental transmission of scrapie into elk via IC inoculation is clinically and neuropathologically indistinguishable from CWD with currently available experimental methods. 44
Bovine spongiform encephalopathy in cattle
Shortly after the first identification of what was ultimately termed BSE in British cattle in 1986, 108 it was recognized that a new neurologic disease of cattle had arisen with striking similarities to scrapie. 110 More than 180,000 positive BSE cases have been diagnosed in the United Kingdom to date. Moreover, different BSE phenotypes have been reported that include classical (C-type) BSE, H-type BSE, and L-type BSE, with the H- and L-type designations owing to the higher or lower apparent molecular mass profiles of the nonglycosylated PrPd band in a Western blot. 10,16,20 The C-type BSE was the first recognized and is an orally acquired, chronic degenerative prion disease of cattle affecting the CNS. Epidemiological studies suggest dietary protein supplements, in particular MBM containing CNS tissues with PrPd, as the source of the outbreak in Great Britain. 110 Evidence from studies in Britain has not detected vertical 112 or horizontal transmission of BSE between cattle (including an absence of evidence for transmission from purportedly “contaminated” pastures). 61 Affected animals display changes in temperament, abnormal posture, incoordination and difficulty in rising, decreased milk production, loss of body weight despite continued appetite and abnormal responses to environmental stimuli. 89,111 The average incubation period for C-type BSE is approximately 4–6 years, and all affected animals succumb to the disease. 62 Following the onset of clinical signs, the animal’s condition deteriorates until it either dies or is destroyed. Such clinical progression usually takes from 2 weeks to 6 months. Most cases in Great Britain occurred in dairy cows between 3 and 6 years of age with the highest susceptibility being in the first 6 months of life; adult cattle were at relatively lower risk of developing C-type BSE. 1
The brains from cattle clinically affected with C-type BSE have a consistent pattern of pathological tissue alterations, 92 which include vacuolation of neuronal perikarya and neurites, most noticeable in the medulla oblongata at the level of the obex. 56 Immunohistochemical detection of PrPd is predominantly recognized in the brainstem of C-type BSE cases with mainly granular and linear patterns of staining in both intraneural and perineuronal tissues of the hypoglossal nucleus, dorsal motor nucleus of the vagus, nucleus of the solitary tract, nucleus of the spinal tract of the trigeminal nerve, reticular formation and in the olivary nucleus (Chiara Porcario, et al., submitted to BMC Vet Res). In BSE-affected cattle, PrPd also accumulates in the retina, 13,89 the most rostral and accessible portion of the CNS. Tissues with apparent lower BSE infectivity levels have been reported for some peripheral nerves and distal ileum of experimentally diseased cattle, 102 and occasionally in the tonsil, nictitating membrane, and bone marrow. 106 Infectivity assays confirm these occasional findings and demonstrate PrPd in the enteric plexi of the distal ileum of experimental orally inoculated cattle. 109 Reports of PrPd in extraneural tissues need to be interpreted with caution since the possibility exists that the detected PrPd may actually be associated with nerve cells or innervation of that tissue 78 ; recent reports with transgenic mouse bioassays of bovine tissues reveal a restriction of infectivity to the nervous system in clinically diseased BSE cattle. 17 Although it has been tested by various detection methods, infectivity has not been demonstrated in milk or blood of cattle with natural or experimental BSE.
Most experimental transmission studies on BSE in cattle have been reported from Europe 17,105,106,109 and provide the basis for disease pathogenesis and tissue distribution of BSE infectivity using tissues and organs from cattle orally dosed with high amounts (approximately 100 g) of BSE-affected CNS material. Infectivity levels of various tissues obtained from such studies were then tested by bioassays in wild-type or bovinized transgenic mice or in cattle as recipient hosts. 17 Results obtained from these studies provide the current basis for the tissues with known BSE infectivity and therefore are included as specified risk materials to be removed at slaughter in many countries around the world.
Atypical bovine spongiform encephalopathy cases: H-type and L-type BSE
Bovine spongiform encephalopathies with molecular profiles different from that of C-type BSE have been reported since 2004 by investigators from several countries. To date, 2 molecular types of atypical BSE have been described, and a summary was published on the Internet in 2007 by the Spongiform Encephalopathy Advisory Committee (http://www.seac.gov.uk/statements/newforms-bse.htm). One molecular type is the L-type, which has been found in cattle in Italy, 20 Japan, 117 Germany, 16 Belgium, 27 and Canada. 28 Western blot analysis demonstrates the L-type form to have a lower molecular mass of the unglycosylated PrPd isoform when compared with C-type BSE. The second type of atypical BSE is the H-type, characterized by Western blot analysis to have a higher molecular mass of the unglycosylated isoform. To date, the H-type has been described in cattle from France, 11 Germany, 16 Japan, 101 the Netherlands, 55 Poland, 55 Switzerland, 103 the United Kingdom 99 and the United States. 86 The unusual molecular phenotype of the H-type BSE cases was characterized by 1) a higher molecular mass of the unglycosylated PrPd isoform, 2) a strong labeling of all 3 PrPd polypeptides (unglycosylated, monoglycosylated and diglycosylated isoforms) with the PrP-specific monoclonal antibodies 6H4 (amino acid epitope consisting of DYEDRYYRE) and P4 (amino acid epitope consisting of GGGWGQGGTHGQWNK), and 3) a glycoform profile with a less prominent diglycosylated PrPd isoform (French and U.S. cases). Some, but not all H-type BSE cases were positive by immunohistochemistry (IHC) because in some cases tissues were not available for immunohistochemical testing. In contrast, L-type cases were characterized by 1) a lower molecular mass of the unglycosylated PrPd isoform, 2) a strong labeling of all 3 PrPd polypeptides with the PrP-specific monoclonal antibody 6H4 but not P4, and 3) a glycoform profile with a monoglycosylated PrPd band at least equally as intense as the diglycosylated PrPd isoform. Epitope mapping with monoclonal antibodies as mentioned above is used as one tool to differentiate TSE strains by IHC 57,59 and Western blot. 100
Until these recent atypical BSE reports, BSE has been shown to be very consistent and uniform in appearance, even after transmission to other species. There are several hypotheses proposed to explain atypical BSE cases. 11 One theory proposes that there are variants of BSE with different molecular features in cattle; a second theory proposes that cattle may have been affected by another TSE (e.g., scrapie or CWD); a third theory proposes that a rare sporadic or genetic form of TSE disease could exist in cattle as described for human TSEs. Recently a new PRNP allele (E211K)85 was reported in a cow with H-type BSE indicating a possible genetic form of BSE that is heritable.77 Research on atypical BSE, first reported in 2004, 11 has investigated intra- and interspecies transmissibility, influence of host genotype, PrPd tissue distribution, and incidence rate of atypical BSE.* Both H- and L-type BSE cases have occurred in different breeds and PRNP genotypes. The majority of cases were in older cattle (>10 yrs of age) and very few of the animals had typical clinical signs of C-type BSE. Importantly, experimental transmission of selected H- and L-type BSE cases, into cattle, mice, and nonhuman primates has been reported. 7,16,18,23,63,68
Relatively less is known about the histopathological and immunohistochemical characteristics of atypical BSE. Microscopic examination of L-type BSE cases revealed prion deposition in the brain that differed in distribution from C-type BSE cases and included amyloid plaques and increased PrPd immunoreactivity in the olfactory bulbs, 19 although PrPRes immunoreactivity has been detected by Western blot in olfactory bulbs of cattle with C-type BSE. 104 The investigators designated this newly identified disease phenotype “bovine amyloidotic spongiform encephalopathy” or BASE. 20 The morphological PrPd deposition of BASE cases differed from that observed in C-type BSE cases: relatively few deposits were found in the obex region but much more occurred in the more rostral structures of the brain, namely in the thalamus and the olfactory bulb. The PrPd-positive deposits were predominantly in the form of amyloid-like plaques. 20 The latter has been reported for TSEs in human beings, but not for BSE in cattle. Less is known about the microscopic appearance of H-type BSE, but recent unpublished findings (Chiara Porcario, et al., submitted to BMC Vet Res) comparing the Italian and the U.S. IHC confirmatory methods for BSE differentiated the different phenotypes (C-, H-, and L-type BSE) as each appearing to be characterized by distinctive features of PrPd deposition. Granular and linear tract PrPd deposits were a distinct feature of C-type BSE cases, whereas intraglial and intraneuronal PrPd deposition appeared as the most representative trait of H-type BSE as reported previously, 16 and the presence of PrPd deposits organized as plaques was a distinguishing hallmark of L-type BSE (BASE) cases, also as previously reported with a preferential distribution in more rostral brain regions. 18,20
Transmissible mink encephalopathy
Transmissible mink encephalopathy has been sporadically identified in ranch-raised mink (Neovison vison). It was first documented in Wisconsin in 1947 29 and the last reported outbreak in the United States was in 1985. 70 Like BSE, TME is a food borne disease that has been experimentally transmitted to a variety of animal species, including cattle, sheep, goats, monkeys, hamsters, mink, American sable (pine marten), beech marten, skunks, ferrets, and raccoons. 29,30 Reported histopathological findings in mink with TME indicate detectable lesions limited to the CNS with microvacuolation of the gray matter, reactive astrocytosis in the cerebral cortex, and neuronal degeneration. 69 Microscopic lesions were reported as a scrapie-like spongiform encephalopathy, which preceded clinical disease by approximately 6 weeks. 69 Weeks before microscopic lesions were visible, ultrastructural alterations were recognized when assessed by electron microscopy and included loss of normal ultrastructure of nerve endings, larger dendritic segments, and variously shaped vesicles and vacuoles in the neuropil. 119 A review of published literature on TME found no descriptions of PrPd distribution patterns in mink as studies in mink were completed prior to development of current diagnostic methods including IHC and Western blotting. Moreover, the lack of natural cases of TME for the past several decades and the advent of the hamster model 83 made the mink a less desirable animal model for study. The origin of TME is unknown, but it is speculated to have been derived from sheep scrapie or from an unknown TSE in cattle. 70,71
Experimental interspecies transmission of transmissible spongiform encephalopathies into cattle
Experimental scrapie transmission to cattle
During the 1990s the possibility that U.S. strains of sheep scrapie might cause BSE following transmission to cattle was assessed experimentally through both IC and oral inoculations. Intracerebral inoculations resulted in a 100% transmission of a prion disease to cattle between 14–18 months following inoculation. 26 A separate study using multiple simultaneous routes of inoculation (including IC) found only 20–40% transmission depending on the source of inoculum and a longer incubation period of 24–48 months following inoculation. 22 Although the affected cattle exhibited anorexia, weight loss, leg and back stiffness, incoordination, and rear leg weakness eventually leading to severe lethargy and ataxia, they did not show signs of hyperactivity, one of the characteristic clinical signs of BSE. To differentiate scrapie in cattle from BSE, there was no microscopic evidence of spongiform changes in the scrapie-affected cattle. Neuropathological changes were not present in the CNS of scrapie-affected cattle whereas spongiform changes are usually observed in clinical BSE cases. 91 Immunoreactivity for PrPd in scrapie-affected cattle was observed predominately in neuronal cell bodies with relatively little accumulation in the neuropil, 25,26 in contrast to BSE where there is a diffuse distribution of PrPd in the CNS. 91 Following oral ingestion of the scrapie agent, cattle did not develop symptoms of neurological disease nor did they develop spongiform lesions nor PrPd deposits in the CNS after eight years post inoculation. 24 Such experiments demonstrate that IC inoculation of the U.S. scrapie agent into cattle results in a disease with clinicopathologic hallmarks that differ significantly from cattle with BSE.
Whereas oral BSE inoculation into cattle is a highly efficient means of transmission, 107 this is not the case for scrapie. 24 Despite the proposed linkage of the BSE epidemic initiation to scrapie, 114 scrapie isolates from U.S. sheep could only be transmitted to cattle by IC inoculation, and the pathology and clinical disease differed from both BSE in cattle and scrapie in sheep. 25,26 The results were later corroborated by inoculation of cattle with scrapie isolates from the United Kingdom. 64 Therefore, current experimental evidence from scrapie transmission studies into cattle does not support the hypothesis that the U.K. BSE epidemic originated from feeding of scrapie PrPd to cattle. However, no experimental transmission studies of atypical scrapie into cattle have been reported to date.
Experimental chronic wasting disease transmission to cattle
The recognition of CWD 116 in captive and free-ranging cervids in the United States raised questions about the possible transmissibility of such agent to other ruminant species that may contact affected cervids or their carcasses on pasturelands or farms. In 2001, preliminary findings of IC inoculation of cattle with the CWD agent from mule deer tissue were published. 37 Although brains of the animals showed no significant histopathologic changes, PrPd was detected by IHC and Western blot, indicating that amplification of the abnormal CWD prion had occurred. In cattle inoculated with CWD, the consistent and sentinel finding of localization of PrPd to multifocal and distinct aggregates confined to glial cells and associated neuropil clearly distinguished this IHC pattern from that seen in scrapie- and BSE-affected cattle, and for that matter, any other TSE. Another distinct feature of the distribution of immunoreactivity for PrPd in CWD of cattle was the infrequent finding of small (≤40 µm) plaques in the cerebrum. Although the characteristic pattern of distinct multifocal aggregates of PrPd predominated, in some white-tailed CWD inoculated cattle labeling in obex and midbrain appeared as coalescing foci. Unlike BSE- and TME-inoculated cattle, PrPd labeling of retina was not present. 38,45 On the other hand, in an ongoing study, none of the cattle given the same inoculum orally (50 g of pooled brain/animal) have shown any evidence of prion disease up to 9 years after inoculation. 115
In contrast to the current authors’ first study, 38 which demonstrated a low attack rate of mule deer CWD upon first passage, subsequent IC inoculation of mule deer CWD passed once in cattle (i.e., cattle-adapted mule deer CWD), showed clinicopathological findings (similar to first passage) in all inoculated cattle within 16.5 months postinoculation. 39 This increased attack rate with shorter incubation periods may indicate adaptation of the mule deer CWD agent to the new cattle host. However, it could also be argued that the inoculum used for the primary passage simply had a lower infectivity titer than that used for the second passage. 37–39 Recent findings of IC inoculation of CWD from white-tailed deer into cattle showed that the white-tailed deer inoculum had a higher attack rate (86%) in cattle than the mule deer CWD inoculum used previously; however, microscopic lesions typical of BSE were still not observed. 45 While cattle inoculated with CWD from white-tailed deer and mule deer CWD had similar Western blot molecular profile results, there was no change between first and second passage of mule deer CWD in cattle. 39
A recent study (Greenlee JJ, Nicholson EM, Kunkle RA, Hamir AN: 2009, Susceptibility of cattle to first-passage intracerebral inoculation with chronic wasting disease agent from elk. In: Proceedings of the American College of Veterinary Pathologists Annual Meeting, p. 1058) assessing transmissibility of CWD derived from elk to cattle also found a low rate of transmission. Clinical signs of poor appetite, weight loss, circling, and bruxism occurred in 2 out of 16 cattle at 16 and 17 months post-inoculation. No spongiform lesions were detected; however, in the 2 diseased cattle, PrPd was detected and confined to the CNS and was similar in distribution to cattle inoculated with CWD from mule deer with the most prominent immunoreactivity in midbrain, brainstem, and hippocampus with lesser immunoreactivity in the cervical spinal cord. The lack of spongiform lesions in any of the IC CWD-inoculated cattle (first or second passage of mule deer CWD) and no change in PrPd deposition patterns suggests the differences in attack rate between elk CWD, mule deer CWD, and white-tailed deer CWD upon first passage are likely a difference in interspecies transmission susceptibility (i.e., a species barrier), although differences in infectivity titer of each inoculum cannot be excluded. Additional studies are required to fully assess the potential for cattle to develop CWD through a more natural route of exposure, but the cumulative evidence, thus far, of the lack of spongiform lesions and the differences from BSE in PrPd distribution after IC inoculation, along with no evidence of transmission following oral exposure, suggests that risk of transmission through routes other than IC is low.
Experimental transmissible mink encephalopathy transmission to cattle
In 1995, 3 different sources of TME were tested in cattle and in all instances the animals developed clinical disease and severe spongiform encephalopathy. 88 The spongiform changes and astrocytic responses were considered more pronounced than those of natural BSE, but similar to the pathology observed after experimental IC BSE inoculations. This work confirmed an earlier report of TME transmission to cattle, 70 which lent strength to the proposal that TME outbreaks in the United States were caused by contaminations of feed with a TSE agent present in “downer” cows. This hypothesis was also partially supported by subsequent experiments that showed that the BSE agent produced spongiform encephalopathy in mink after oral exposure. 87 However, clinical signs and histopathologic lesions were reported to be distinguishable from natural TME. 87 Subsequent IC inoculations of cattle with first and second cattle-passaged TME confirmed the earlier findings and also described for the first time the immunohistochemical and Western blot characteristics (lower molecular weight of cattle-adapted TME vs. C-type BSE by Western blot) of the accumulated PrPd, which indicated further similarities between TME and BSE in cattle 40 and accentuated their dissimilarities from experimental scrapie and experimental CWD in cattle. A 2007 study lends further to the relationship between TME and L-type BSE, where in an ovinized transgenic mouse model, cattle-passaged TME presented with the same phenotypic characteristics as atypical L-type BSE. 5 With TME in cattle, the predominant pattern of immunohistochemical labeling was diffuse, evenly distributed, punctuate and coarse granules that involved most areas of the neuropil. Perineuronal labeling of PrPd was regularly noted, in contrast to its non-presence in scrapie- and CWD-inoculated cattle. However, to the authors’ knowledge, experimental studies investigating the oral route of transmission of TME to cattle have as yet not been conducted.
Host species exerts influenceover PrPd tissue distribution
A consistent finding from the experimental interspecies transmission studies of scrapie, CWD, and TME into cattle is the observation that the PrPd tissue distribution in cattle remains essentially restricted to the CNS and, aside from the distinctions noted above regarding PrPd immunoreactivity distribution within the CNS, it is no more extensive than naturally occurring BSE in cattle. 24–26,37,40 Importantly, cattle inoculated with TME, 40 scrapie, 24–26 and CWD 37,38,40 have no evidence of a lymphoid or blood phase of PrPd; which is a distinction from classical scrapie and CWD in their natural hosts. Although detectable PrPd has been reported in the distal ileum following experimental oral BSE challenge in cattle and occasionally in the tonsil, nictitating membrane, or bone marrow, 106,109 studies of naturally occurring clinical cases of BSE have found infectivity only in the CNS tissue using conventional mouse bioassays. 12 A recent experiment on bone marrow infectivity of cattle orally inoculated with BSE used IC inoculation of cattle as a bioassay with sternal bone marrow collected at 22, 26, 32, and 36 months after exposure and found no evidence of BSE in cattle 70–91 months post inoculation, suggesting that disease-causing BSE material in bone marrow is either a rare event or that it may be consistently present but at levels undetectable by what is perhaps considered the most sensitive bioassay (i.e., IC inoculation of cattle). 97 The consistent detectable tissue distribution of PrPd in cattle experimentally inoculated with BSE, TME, CWD, or scrapie is essentially restricted to the bovine nervous system, 24–26,37,38,40,42,94 as has been reported in naturally occurring cases of BSE. 17 Bovine tissue infectivity studies in transgenic mice that are highly sensitive to BSE have confirmed the essential restriction of infectivity to the nervous system in clinically diseased BSE cattle. 17 Collectively, these results indicate that the distribution of PrPd of BSE in cattle is fundamentally different from TSEs in sheep, cervids, or mice. 17 In contrast, sheep and cervids appear to have extensive lymphoid tissue involvement with PrPd deposition, regardless of the TSE with which they are inoculated. 38,41,44,59 The only exceptions to this paradigm have been studies where lymphoid involvement in elk or European red deer experimentally inoculated with scrapie or BSE, respectively, was not observed. 43,44,72 Up to 15% of elk with naturally occurring CWD show PrPd in the CNS and not in the lymphoid tissues. 98
It can be concluded that the animal host, especially cattle, exerts considerable influence over the pathogenesis of a prion disease in terms of what tissues are involved and what can be seen in one animal species does not always extrapolate to another. In particular, no evidence exists to suggest that infectivity can be found in the blood of cattle with BSE as tested by bioassay of spleen and/or blood in bovinized transgenic mice, 17,34 whereas lines of evidence exist that suggest that infectivity can be found in the blood of cervids with CWD, scrapie in sheep, and vCJD in human beings. Whole blood transfusion studies in sheep using donor sheep with experimental BSE or with natural scrapie have shown that infectivity resides in the blood of sheep. 51–54 Similarly, transmissibility by blood transfusion has been reported for deer with experimental CWD. 73
Differential diagnosis and conclusions
Over the past 20 years, several interspecies transmissibility studies of various endemic TSEs (scrapie, CWD, and TME) to various livestock hosts have now been completed. A limitation of the published research on experimental interspecies TSE transmissions to cattle is the possibility that IC inoculation results in the various clinical, histological, and diagnostic test differences observed between scrapie and CWD in cattle versus BSE. However, arguing against those findings being an artifact of the experimental design is the fact that oral challenge studies with both CWD and scrapie into cattle have failed to cause a TSE, and the differences in pathology, IHC, and Western blot that have been observed are in keeping with a species barrier for cattle against these two prion diseases. Moreover, the similarities of experimental BSE transmission to mink by oral or IC challenge support the IC route as a valid experimental approach. 87 A brief description of clinical, histopathological, and immunohistochemical findings, and molecular phenotype in cattle is summarized in Table 1. Figure 1 illustrates histological changes in the brain of cattle with the selected TSEs. Figure 2 illustrates the immunohistochemical immunoreactivity differences of cattle from these same studies and Figure 3 illustrates the Western blot molecular profile differences. Although the scrapie and CWD transmission to cattle studies failed to reproduce a prion disease exactly like BSE, they are important in that no reported bovine TSE cases to date appear similar to experimental CWD or scrapie in cattle, thus providing evidence that cattle seem naturally resistant to CWD and scrapie. In contrast to cattle-passaged scrapie and CWD, which are phenotypically distinct from BSE in the natural host, cattle-passaged TME shows intriguing phenotypic similarities with the L-type BSE. It is critical to note these findings give further scientific assurance that the confirmatory histological, immunohistochemical and Western blot tests employed in the current international TSEs surveillance programs are capable of detecting different prion strains in cattle and would implicate their origin, should such a cross-species transmission occur naturally in the future. Finally, these studies provide valuable confirmatory information regarding the range of tissues to include as specified risk material and have established an archive of tissues available to the greater scientific community for prion research.
Differential diagnostic features of naturally occurring transmissible spongiform encephalopathies in cattle versus experimental intracranial transmission of scrapie, chronic wasting disease, or transmissible mink encephalopathy to cattle.*
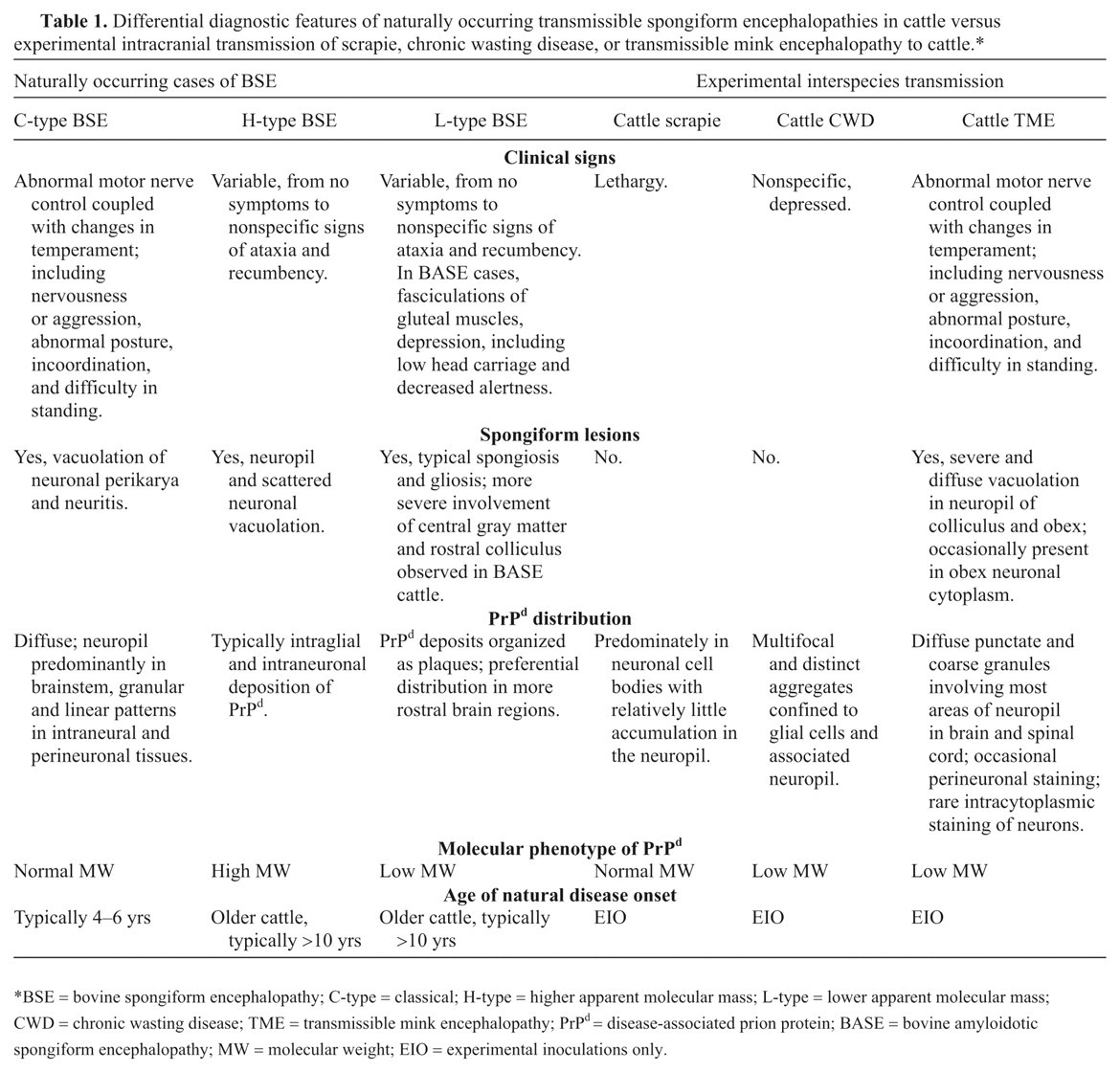
BSE = bovine spongiform encephalopathy; C-type = classical; H-type = higher apparent molecular mass; L-type = lower apparent molecular mass;CWD = chronic wasting disease; TME = transmissible mink encephalopathy; PrPd = disease-associated prion protein; BASE = bovine amyloidotic spongiform encephalopathy; MW = molecular weight; EIO = experimental inoculations only.
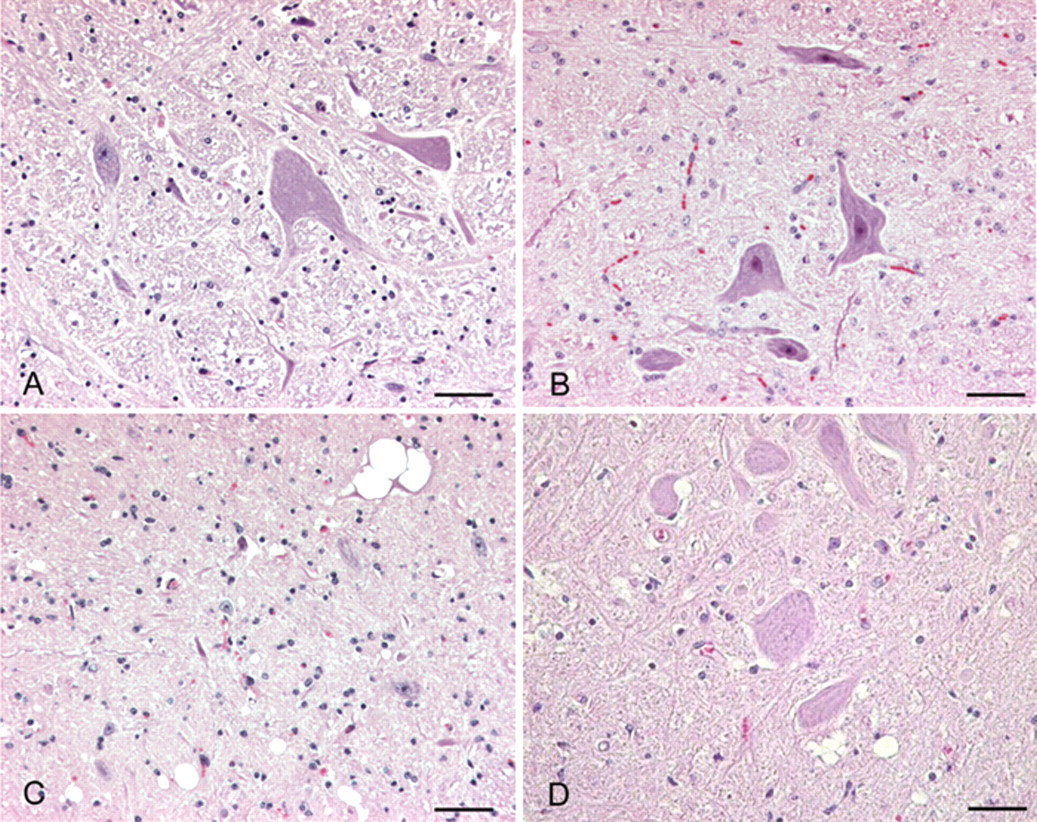
Brain; cattle. Sections of brainstem at the level of the obex from bovine affected with (
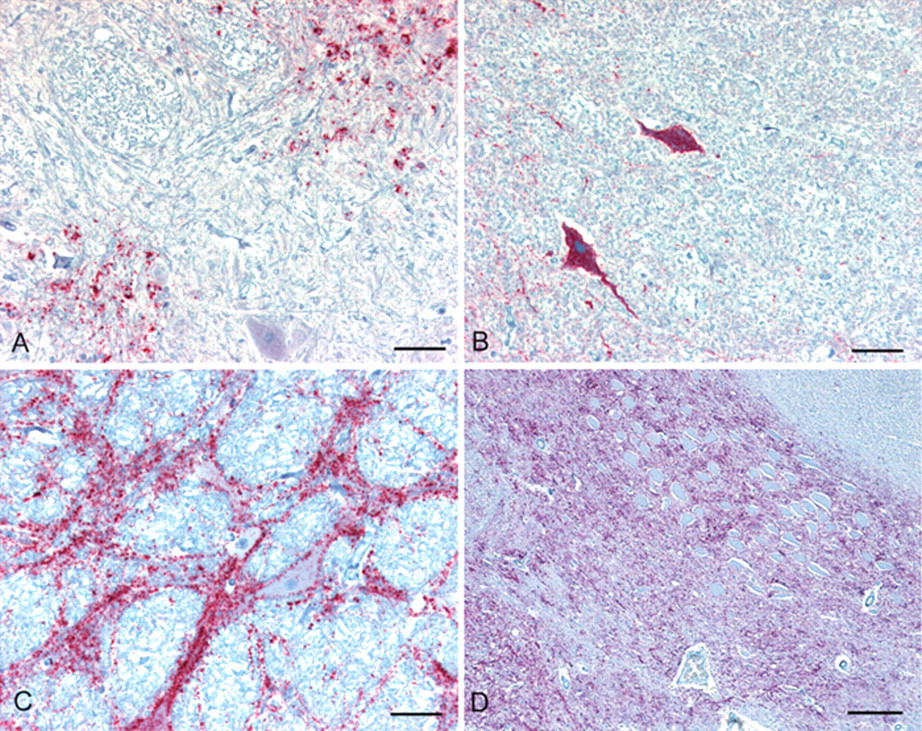
Brain; cattle. Immunohistochemical detection of PrPd in sections of brainstem at the level of the obex from bovine affected with (
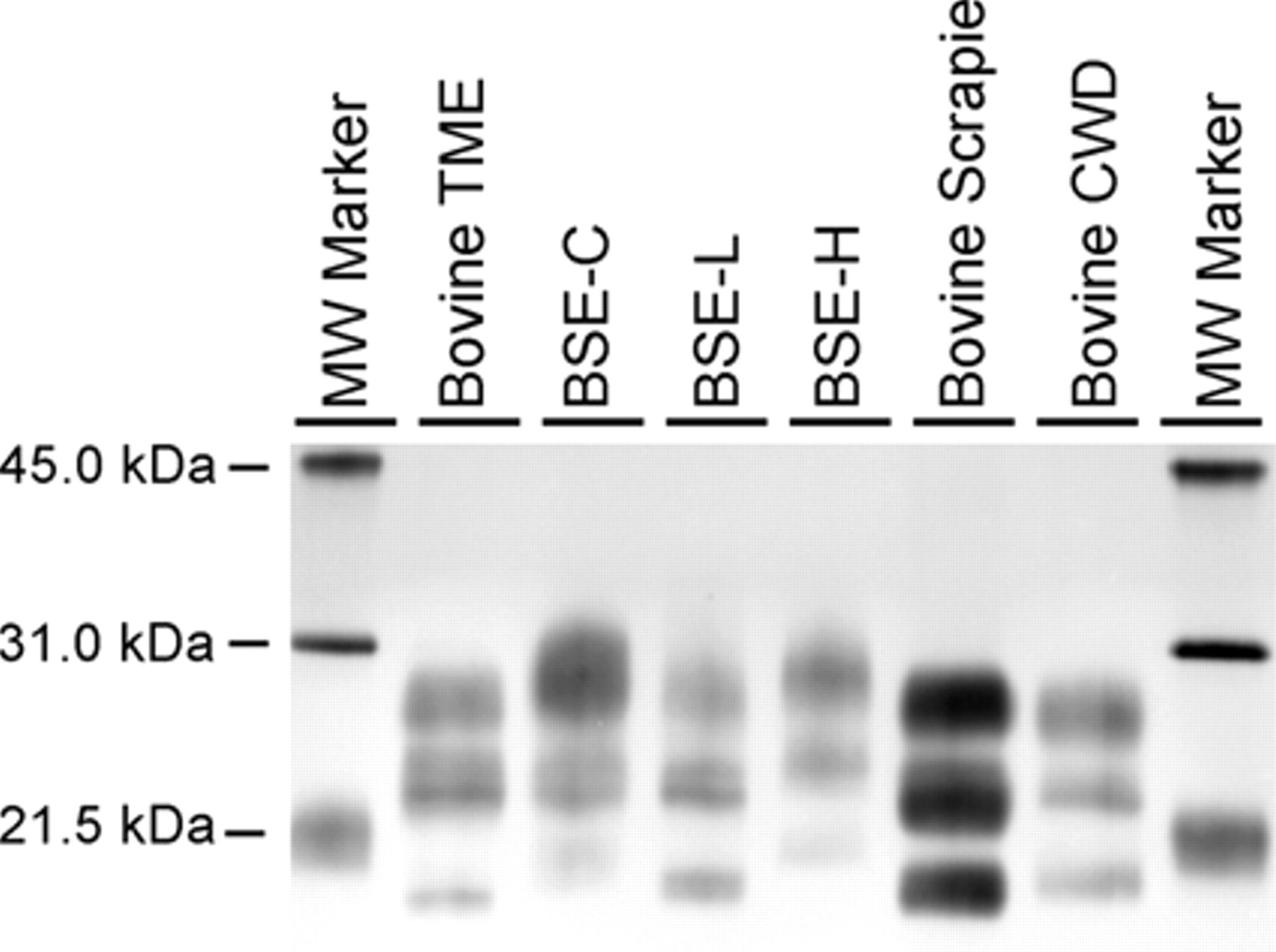
Western blot analysis of various transmissible spongiform encephalopathies (TSEs) in cattle using monoclonal antibody 6H4. Loading amount of brain homogenate is not constant to ensure sufficient signal intensity for each TSE. Bovine transmissible mink encephalopathy (TME), bovine scrapie, and bovine chronic wasting disease (CWD; mule deer) are all from experimental, intracranial inoculation cattle. The bovine spongiform encephalopathy (BSE)-C, BSE-L, and BSE-H are all naturally occurring BSE cases. The highest molecular weight band is generally referred to as the di-glycosylated band, the middle band as the monoglycosylated, and the lowest band as the unglycosylated band. BSE-H and BSE-C can be distinguished from each other and from all other TSEs in cattle by a combination of the molecular weight of the unglycosylated band (<21.5 kDa band) and the intensity of the diglycosylated band (approximately 31.0 kDa) with respect to the other 2 bands. Bovine TME and BSE-L are not distinguishable from each other by Western blot but can be distinguished from BSE-C, BSE-H, bovine scrapie, and bovine CWD. Similarly, bovine scrapie and bovine CWD cannot be distinguished by Western blot but are clearly different from bovine TME, BSE L, BSE-C, and BSE-H. Bovine CWD (mule deer) and bovine CWD (white-tailed deer) are indistinguishable by Western blot (data not shown).
Footnotes
Acknowledgements
The authors thank Dr. S. Mark Hall (Pathobiology Laboratory, National Veterinary Service Laboratory, USDA, Animal and Plant Health Inspection Agency, Ames, Iowa) for providing the images of BSE in Figures 1 and 2, and Kevin Hassal for Western blot technical support on Figure 3. Amir N. Hamir and Marcus E. Kehrli, Jr. contributed equally in the preparation of this manuscript. USDA is an equal opportunity provider and employer.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
