Abstract
A 6-year-old castrated German Shepherd Dog was presented with a 6-month history of progressive, nonpainful, left pelvic limb paresis. Magnetic resonance imaging revealed atrophy of left-sided epaxial and hypaxial muscles from L5–L7 and an enlarged L5 spinal nerve. Exploratory hemi-laminectomy revealed focally and cylindrically thickened L5 and L6 nerve roots. Histologic evaluation of a surgical biopsy specimen from the L6 dorsal nerve root, and the L5 nerve roots after later amputation revealed distended hypercellular fascicles. This distension was due to widely separated axons surrounded by concentric lamellations formed by neoplastic perineurial cells and their processes. These pseudo-onion bulbs were separated from each other by a basophilic myxoid stroma. The perineurioma cell processes were immunonegative for S-100 (a and b chains) and collagen IV, but were immunoreactive for laminin. The central axons were also immunoreactive for NF-200 and S-100. The proliferative index of the perineurioma cells, as determined by MIB-1 immunoreactivity, was about 3%. Ultrastructurally, the widely separated, interdigitating perineurioma cell processes were connected by desmosomal-like junctional complexes to form continuous circles. Their processes were covered by a discontinuous basal lamina. Each centrally placed axon was normally, thinly, or completely unmyelinated and was surrounded by a normal Schwann cell. These morphologic and immunologic features distinguish this lesion from hypertrophic neuropathy and were consistent with intraneural perineurioma.
The rare human intraneural perineurioma of perineurial cell origin is now considered to be a clinical and pathologically distinct neoplasm, and is further characterized cytogenetically by abnormalities of chromosome 22. 4, 10 The intraneural variant is typified by pseudo-onion bulb formation of perineurial cells, resulting in fusiform, segmental enlargement of spinal nerves of the extremities. Involvement of two adjacent spinal nerves has been reported. 4 The extraneural form develops in subcutaneous tissue of the limbs or trunk. Though clinical and gross features are different, both variants display immunocytochemical and ultrastructural features of perineurial cells. 1, 7, 9, 11 The intraneural form typically develops in adolescence or early adulthood without any sex predilection. 9 Histologically, the neoplastic perineurial cells form many layers of concentrically laminated cells around either myelinated or nonmyelinated nerve fibers. The cells are immunoreactive for epithelial membrane antigen (EMA), vimentin, collagen IV, and laminin, but are negative for S-100. 1, 9– 12 Ultrastructurally, the perineurial cells have long, thin processes bearing numerous pinocytotic vesicles, are closely opposed with junctional complexes, and are lined by a patchy basal lamina. 1, 4, 5, 9, 11, 12 Cytogenetically, most perineuriomas have a clonal loss of a portion of chromosome 22. 4 Results of long-term follow-up analysis indicate that human perineuriomas neither metastasize nor recur after surgical removal. We describe the clinical, light and electron microscopic, and immunocytochemical findings of an intraneural perineurioma in the left femoral and sciatic nerve roots of a dog.
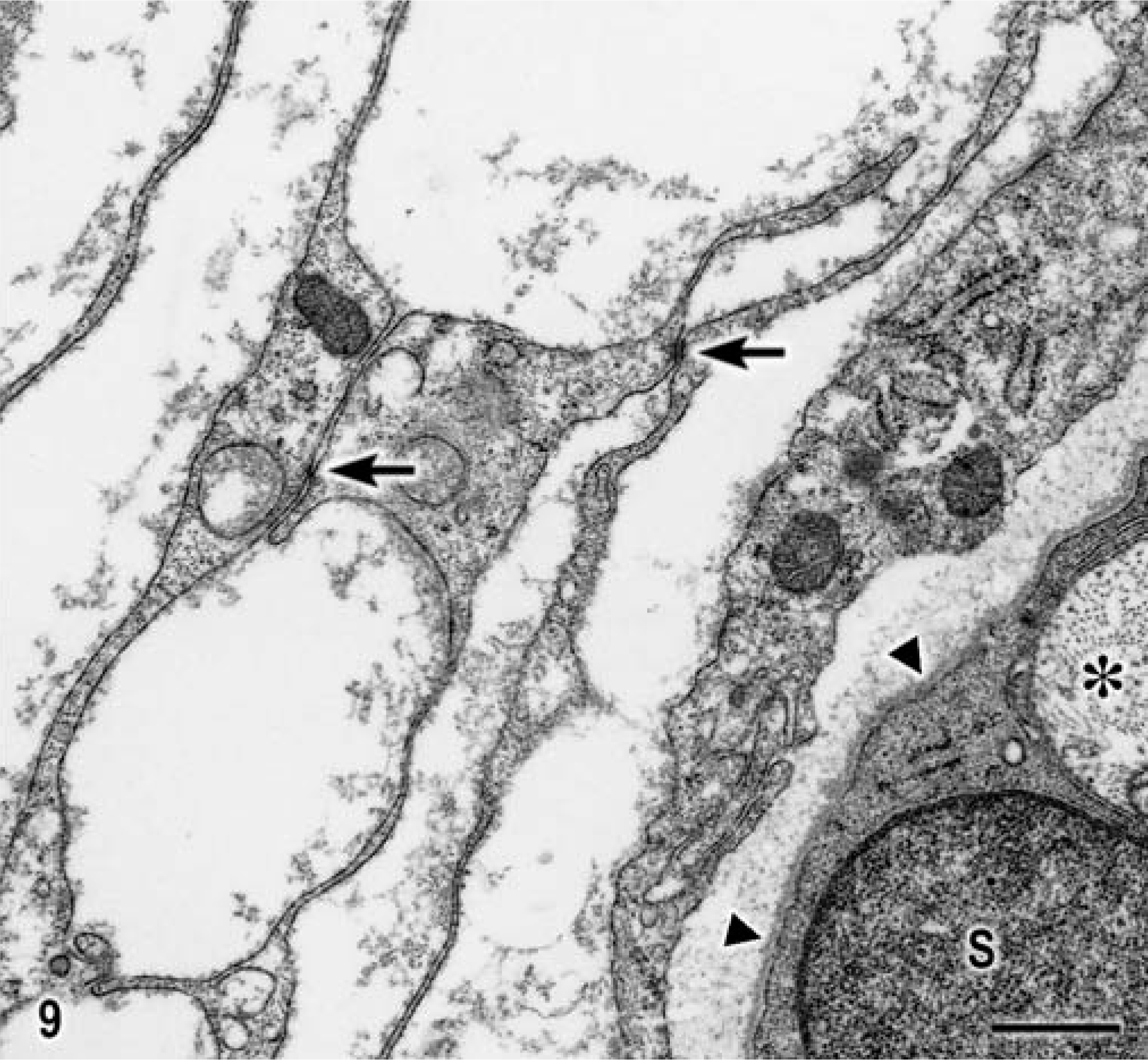
A 6-year-old, neutered male German Shepherd Dog was presented with a 6-month history of progressive, nonpainful, left pelvic limb paresis. Results of neurologic examination suggested left-sided L4–S2 myelopathy or neuropathy. Other than mild thrombocytopenia (data not presented), results of serum biochemical analysis, complete blood count, urinalysis, thoracic radiography, and abdominal ultrasonography were unremarkable. Electromyography revealed spontaneous activity consisting of fibrillation potentials and positive sharp waves restricted to muscles innervated by the femoral (L4–L6), and sciatic nerves (L6–S2) of the left pelvic limb. Both sensory and motor peroneal nerve conduction velocities were markedly reduced on the left side (s-49–54 m/s, m-35–47 m/s; normal values: s-65–77 m/s, m-69–80 m/s). Waveforms were dispersed and decreased in amplitude, and late waves and cord dorsum potentials were not elicited from the left side. Magnetic resonance (MR) images revealed marked atrophy of the epaxial and hypaxial muscles on the left side from L5–L7 and an enlarged left L5 spinal nerve (Fig. 1). Left-sided exploratory hemi-laminectomy was done. The dorsal and ventral nerve roots at L5 and L6 were focally and cylindrically thickened, up to about twice their normal diameter and about 1 cm long (Fig. 2). Nerve roots of adjacent L4 and L7 segments appeared grossly normal. The L6 dorsal nerve root was biopsied. The left pelvic limb was amputated 2 months later due to substantial loss of function. From this amputated limb, tissue specimens from the affected L5 and L6 ventral and dorsal nerve roots also were evaluated microscopically. The dog was euthanized 6 months later after rupture of the right cranial cruciate ligament, but at that time, the dog did not have other neurologic dysfunction.
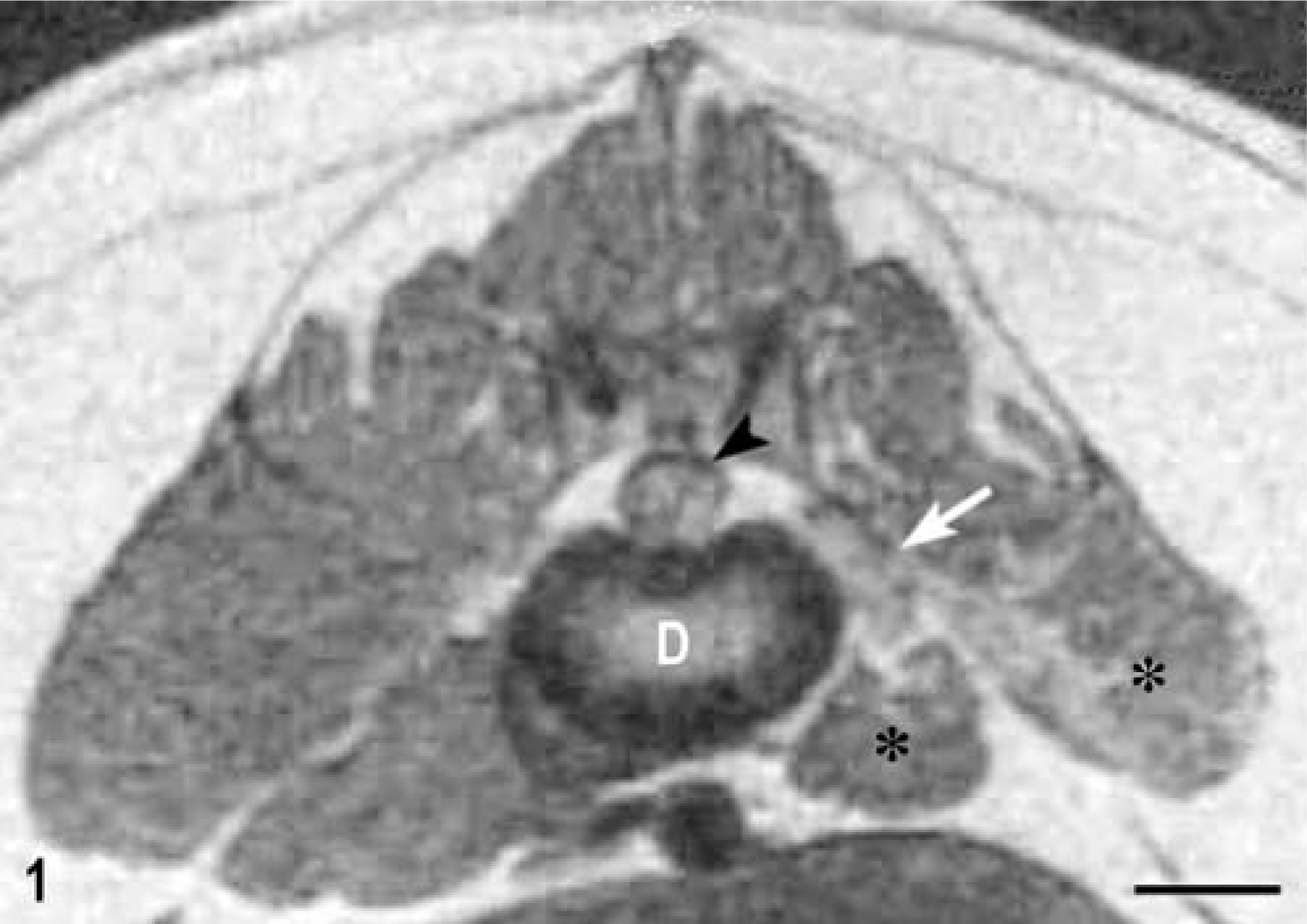
Spinal cord; dog. T1-weighted transverse MR image at the level of the L5–L6 intervertebral disc space (D) after contrast administration. A thickened L5 nerve root (white arrow) is seen on the left side. Notice marked atrophy of the left epaxial and hypaxial muscles (∗) at this level. Arrowhead indicates the spinal cord. Bar = 9 mm.
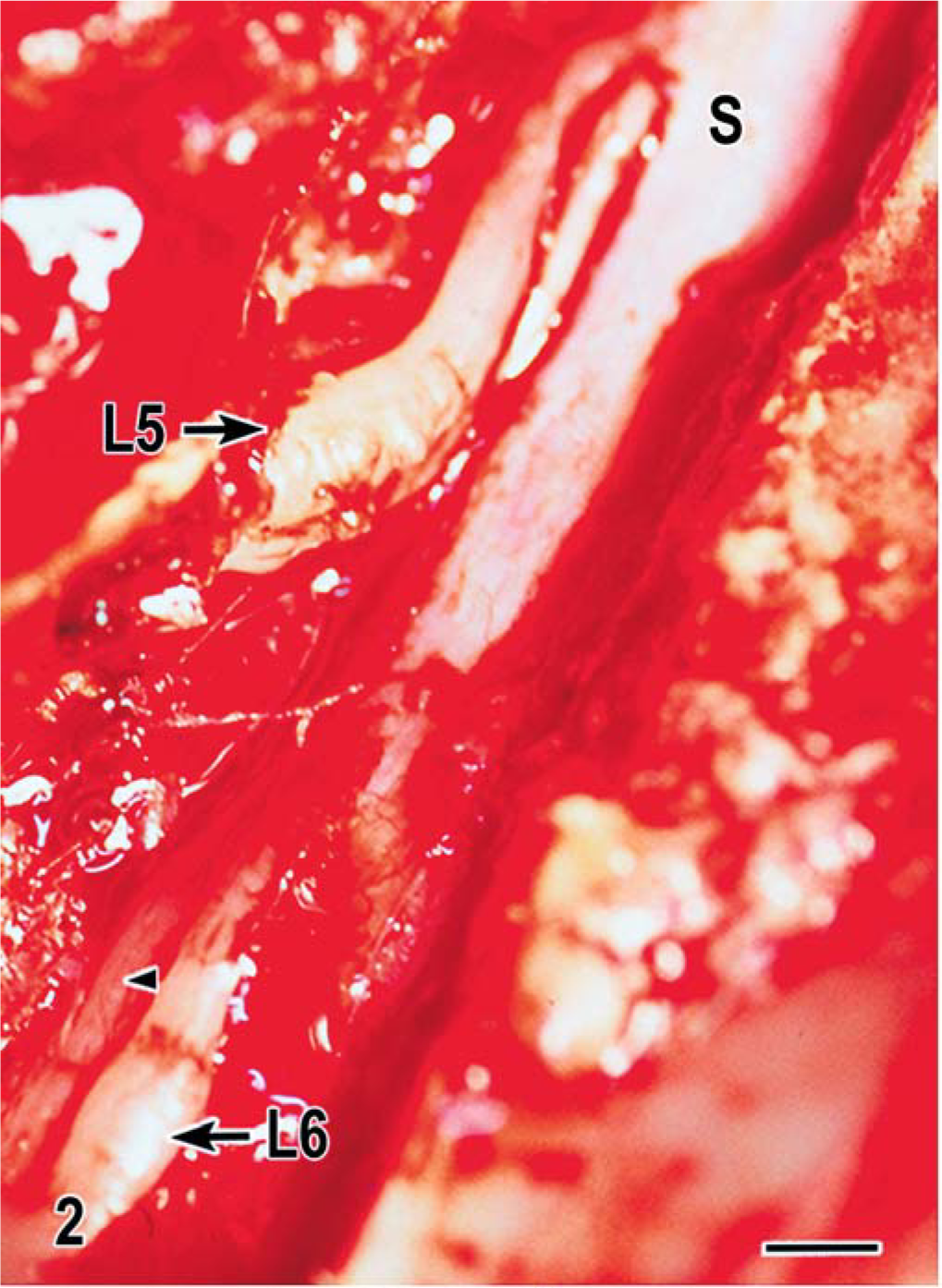
Spinal cord; dog. Intra-operative photograph taken during exploratory left-sided hemi-laminectomy at the level of the affected L5 and L6 nerve roots (arrows). Notice the spinal cord (S). The L6 ventral nerve root also is slightly thickened (arrowhead). Bar = 3 mm.
Histologic evaluation of formalin-fixed, paraffin-embedded hematoxylin and eosin (HE)-stained transverse sections from the lesion in the L6 dorsal nerve root biopsy specimen revealed prominent hypercellular and hypertrophic fascicles compartmentalized by an attenuated thickened perineurium. Fascicles were distended due to widely separated axons, which in turn, were surrounded by concentrically arranged multiple layers of perineurial cells. The thin spindle-shaped bipolar cytoplasmic processes of the cells were loosely arranged in a whorling laminar pattern, producing a distinctive pseudo-onion bulb appearance around each axon (Fig. 3a). Toward the edge of the fascicles, the perineurial cells tended to be closely compacted, but centrally, were widely separated from each other and from adjacent pseudo-onion bulbs by a loose lacy basophilic staining myxoid-like matrix. On longitudinal section, dense cellular cords of perineurial cells surrounding intact axons were separated from each other by the myxoid matrix (Fig. 3b). The inner layer of the perineurium was thickened by a laminar proliferation of perineurial cells. There was infiltration of the dorsal root ganglion by perineurial cells. Most cell nuclei were curved, with elongate tapered ends. Mitotic figures were rare.
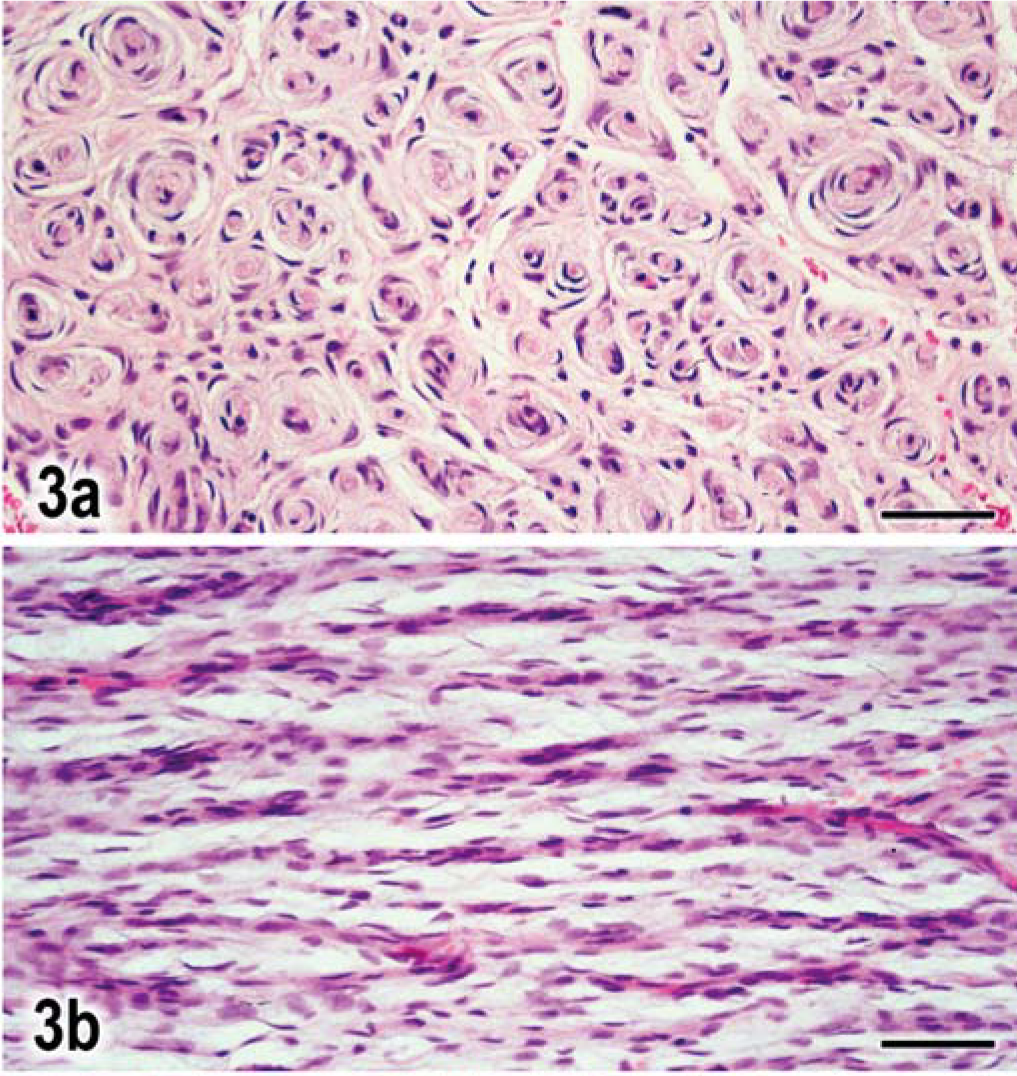
Nerve root L6; dog. Fig. 3a. Transverse section through L6 nerve root swelling, with concentric lamellations of cell processes around individual axons. HE. Bar = 40 µm. Fig. 3b. Longitudinal section of same nerve with hypercellularity along individual axons separated by spaces containing lightly staining myxoid material. HE. Bar = 35 µm.←
An immunocytochemical procedure using an unlabeled streptavidin-biotin-peroxidase technique was used as described. 6 There was positive S-100 (α and β chains) immunoreactivity restricted to the centrally placed axons (Fig. 4). The cell processes also had uniform, strongly positive immunoreactivity for vimentin and laminin (Fig. 5), but were immunonegative for collagen IV. The presence of axons at the center of most whorls was confirmed by their positive immunoreactivity for the high molecular weight neurofilament subunit (NF-200 kd; Fig. 6). Immunoreactiviety for MIB-1 indicated a proliferative index for the tumor cells of up to 3%. Examination of plastic-embedded toluidine blue-stained thick sections 6 indicated that most of the affected axons were thinly myelinated, whereas completely demyelinated axons were observed less often (Fig. 7).
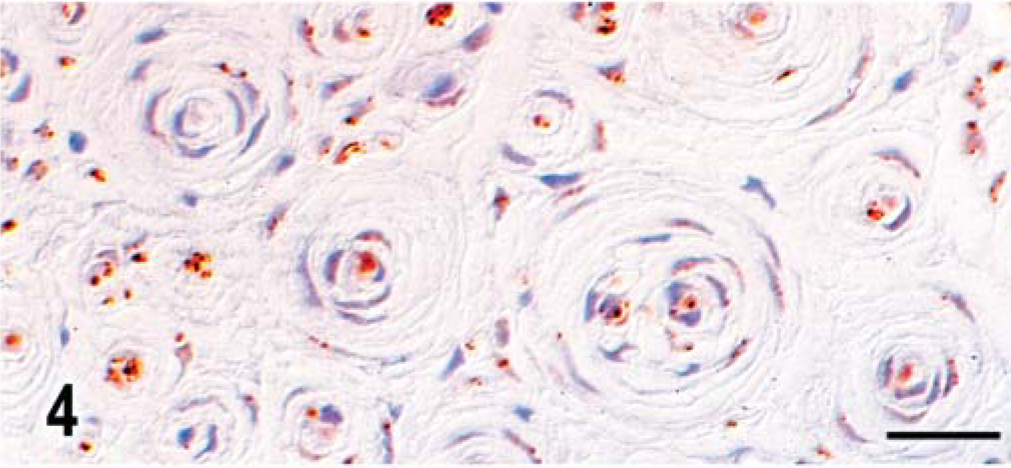
L6 nerve root; dog. Transverse section with central axons immunoreactive for S-100, whereas concentrically lamellated cell processes are uniformly negative. Unlabeled avidin-biotin peroxidase with hematoxylin counterstain. Bar = 25 µm.
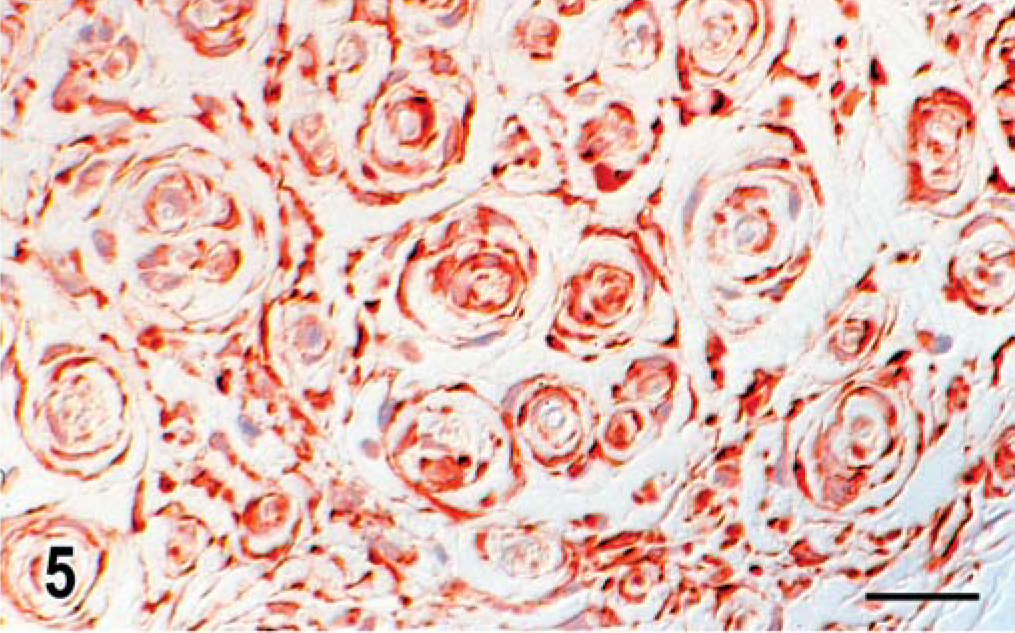
L5 nerve root; dog. Transverse section with lamellated cytoplasmic processes positively immunoreactive for laminin. Unlabeled avidin-biotin peroxidase with hematoxylin counterstain. Bar = 19 µm.
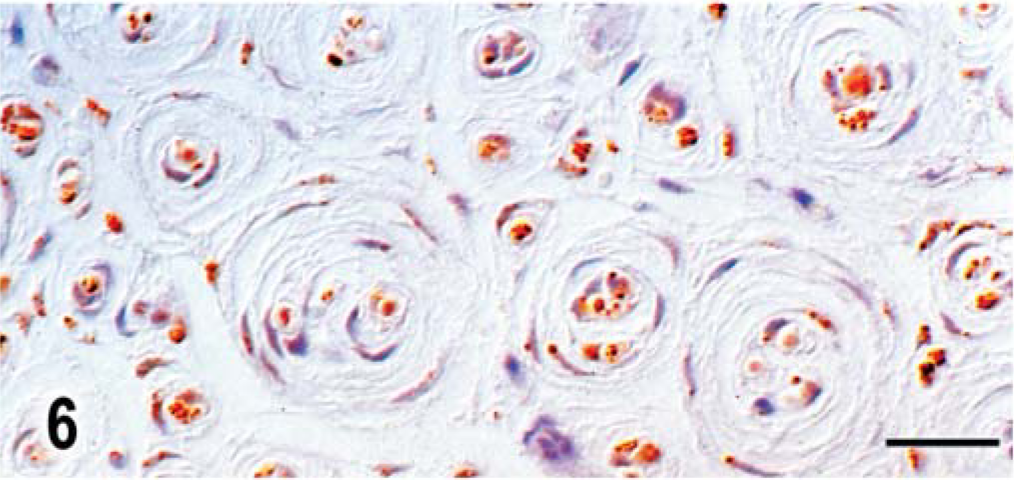
L6 nerve root; dog. Transverse section of nerve root with positive staining of central axons with NF-200, but all cell processes stain negatively. Unlabeled avidin-biotin peroxidase with hematoxylin counterstain. Bar = 17 µm.
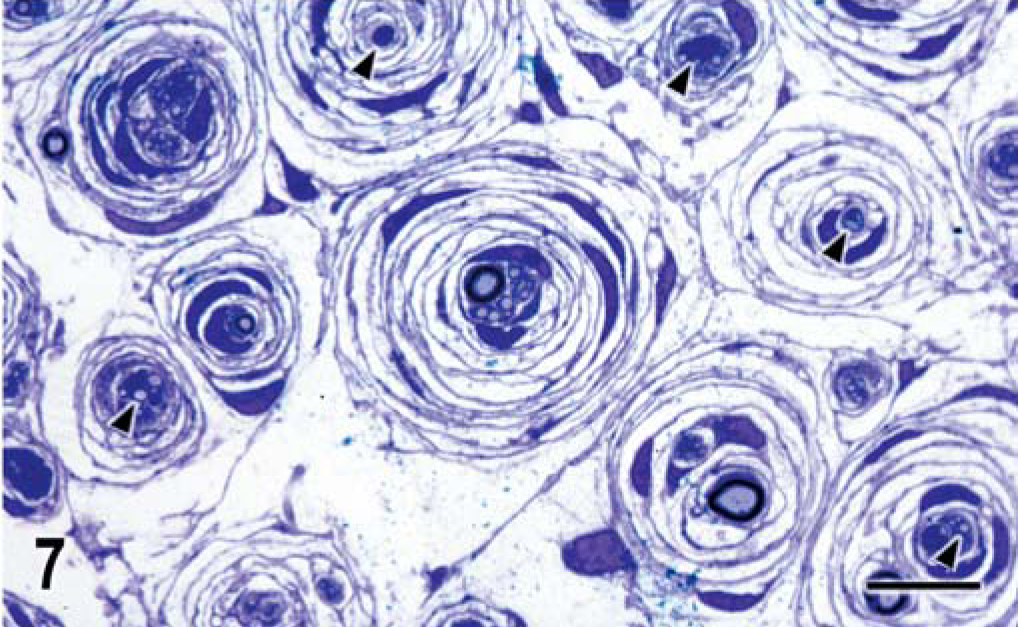
L6 nerve root; dog. Toluidine blue-stained, plastic embedded cross section illustrating the partially myelinated or unmyelinated axons (arrowheads) forming the core of the concentrically laminated cell processes of the perineurioma cells.
Small capillaries were prominent within the tumors. At the margins of the lesions was a gradual change from the characteristic microscopic appearance to normal individual nerve fibers and fascicular architecture. The earliest change appeared to be a laminar cellular thickening of the perineurium, with ingrowth into the fascicles. There was some progressive Wallerian-like degeneration of axons proximal and distal to the biopsy site. In distal branches of the femoral and sciatic nerves from the subsequently examined amputated limb, there was secondary axonal loss and replacement fibrosis.
Ultrastructural examination confirmed that the radial pattern of the pseudo-onion bulbs was formed by concentrically arranged perineurial cells with their thin, elongated nonbranching processes (Fig. 8). The elongated nuclei had a prominent nucleolus and evenly dispersed chromatin. The ends of the processes interdigitated or overlapped and frequently were connected to each other by desmosomal-like junctions to form continuous circles (Figs. 8, 9). The cells contained a few mitochondria, profiles of rough endoplasmic reticulum, few intermediate filaments, and some pinocytotic vesicles. The processes also were widely separated from each other by partially empty spaces containing some flocculent wispy aggregates. The processes were up to seven layers thick and occasionally displayed segments of a thickened discontinuous basal lamina (Figs. 8, 9). Perineurial cells of the outer layers often extended to participate in adjacent whorls (Fig. 7). At the core of these whorls was usually a single partially myelinated axon or a cluster of small nonmyelinated axons, or both. Axons were enveloped by normal Schwann cells, the cytoplasmic membrane of which was lined by a continuous basal lamina (Fig. 9).
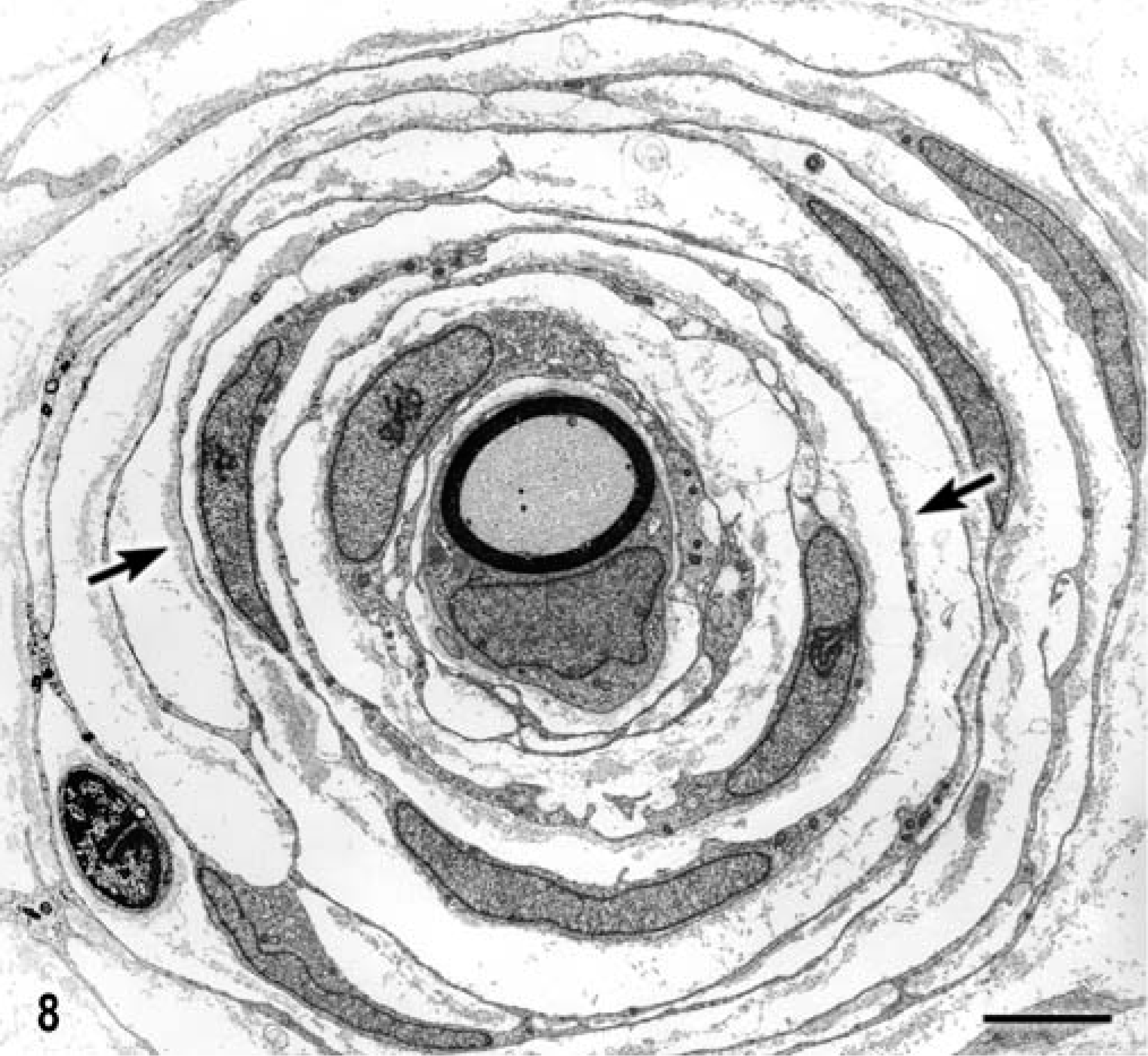
L6 nerve root; dog. Electron micrograph of a thinly myelinated axon and associated Schwann cell. There are at least seven concentrically organized layers of perineurioma cells and their overlapping processes wrapped around the axon. Notice the basal lamina on the outer surface of these processes (arrows). Bar = 3 µm.
L6 nerve root; dog. Electron micrograph of a naked axon (∗) enveloped by a Schwann cell (S), and overlapping processes of the perineurioma sometimes connected by desmosome-like junctional complexes (arrows) and a flocculent discontinuous basal lamina, compared with that of the Schwann cell (arrowheads). Bar = 1 µm.
Onion bulbs are concentric lamellated structures formed of Schwann cells that develop in focal or generalized diseases of peripheral nerves. 2, 8, 9 Onion bulb formation was originally described as pathognomonic of generalized hypertrophic sensory and motor neuropathies in people. It is now thought to be a nonspecific lesion resulting from Schwann cell proliferation after repeated demyelination and remyelination due to a wide variety of causes. 2, 9 This proliferation also is associated with other generalized diseases, such as inflammatory polyneuropathies, globoid cell leukodystrophy, diabetic neuropathy, neurofibromatosis type I, and chronic lead poisoning. A confusing and contentious nosology of entities, such as local hypertrophic neuropathy (LHN), hypertrophic interstitial neuritis, intraneural neurofibroma, and hypertrophic neurofibromatosis has described for the focal lesions. 2 Most of these are considered to result from hyperplastic Schwann cell proliferation with a lamellar concentric arrangement around intact axons. Recently, it was documented, in human perineuriomas, that neoplastic pseudo-onion bulb formation by perineurial cells is associated with heterozygosity or centromeric loss of chromosome 22, probably involving the NF2 gene. 4 This finding has provided clear distinction of LHN from proliferative Schwann cells due to “reactive” or traumatic etiology. 2 Human intraneural perineuriomas, therefore, can best be distinguished from hyperplastic Schwann cell proliferation by their positive and negative immunoreactivity to EMA and S-100 respectively. 2, 4, 9, 10, 12 Further, formation of junctional complexes between cell processes, detected ultrastructurally, also distinguishes them from Schwann cell processes. 2, 5, 7, 11, 12 Additional confirmation comes from cytogenetic studies of the clonal loss of part of chromosome 22. 4 In the dog of this report, EMA immunoreactivity of the perineurial cells was negative. However, we were unable to detect EMA expression in any normal canine tissue (unfixed frozen or formalin-fixed, paraffin-embedded) using currently available commercial antibodies immunoreactive in normal human tissue (RJH, personal communication). However, S-100 immunoreactivity was restricted to central axons, thus excluding Schwann cells as a component of the pseudo-onion bulb formation. Ultrastructurally, the perineurial cells abutted end to end with desmosomal-like intercellular junctions forming a continuous circle and had an incomplete basal lamina. 1, 10– 12 These findings were in contrast to LHN where Schwann cells have open spaces between their processes, do not have junctional complexes, and are coated with a thick continuous basal lamina. 2, 5, 8, 11, 12 Cytogenetic analysis of the canine tumor was not attempted.
In dogs, the differential diagnosis of a focal lesion in a peripheral nerve would have to include LHN. One case of chronic hypertrophic neuropathy, with involvement of the trigeminal ganglion and nerve and of most spinal roots and nerves in a dog, has been described. 3 However, interpretation of those light and electron microscopic morphologic studies concluded that the onion bulb formation was due to hyperplastic Schwann cell proliferation from a widespread recurrent demyelinating and remyelinating process. Positive immunostaining of those cells for S-100 would best have confirmed that diagnosis.
Findings in this dog indicated that this multi-focal but localized intraneural perineurioma shared many of the clinical and pathologic features of the human tumor counterpart. However a definitive diagnosis would depend on confirmation of equivalent canine cytogenetic abnormalities. Nevertheless, the light and electron microscopic findings, with the negative S-100 staining of the cell processes, together are strong circumstantial evidence to substantiate this diagnosis. 1, 4, 9, 11
