Abstract
An 8-y-old intact male Labrador Retriever dog developed cutaneous masses over the entire body. On histologic evaluation, the masses were composed of bundles of fusiform neoplastic cells arranged around adnexa, with mild atypia and no mitoses, consistent with peripheral nerve sheath tumors (PNSTs). Immunohistochemically, neoplastic cells were immunoreactive for vimentin, glial fibrillary acidic protein (GFAP), and S100, confirming their perineurial origin. The dog was euthanized because of deteriorating clinical signs. In addition to the cutaneous masses, a cardiac mass was identified at postmortem examination. The histopathologic and immunohistochemical features of the cardiac mass were similar to those of the cutaneous masses. To our knowledge, the combination of multiple cutaneous masses with features of PNSTs and a concurrent cardiac lesion has not been reported previously in a dog. We suggest “neurofibromatosis type 1–like” presentation for this unique combination of cutaneous and cardiac masses. Further studies are required to investigate the etiopathogenesis of this condition and explore its genetic background.
Peripheral nerve sheath tumors (PNSTs) are a heterogeneous group of neoplasms that originate from cells composing the nerve sheath (i.e., Schwann cells, fibroblasts, or perineurial cells).6,7 In humans, clinical and morphopathologic distinctions are recognized between schwannoma (World Health Organization [WHO] grade I), neurofibroma (WHO grade I), and malignant peripheral nerve sheath tumor (MPNST; WHO grade III and IV). Schwannoma in humans is considered a benign slowly growing neoplasm located anywhere in the peripheral nervous system (PNS), but its most fre-quent intracranial site is the vestibular segment of cranial nerve 8. Neurofibroma is usually indolent, with a favorable prognosis. Multiple neurofibromas in humans are the hallmark of neurofibromatosis type 1 (von Recklinghausen disease). MPNSTs are morphologically variable neoplasms with a distinct tendency toward divergent mesenchymal differentiation. In contrast, in the veterinary literature, those differentiation patterns are less recognized, and subclassifications as neurofibromas, neurofibrosarcomas, neurilemmomas, and schwannomas are arbitrary and based on extrapolation of human tumor studies.6,7 The WHO classification of tumors of the nervous system and mesenchymal tumors of skin and soft tissues in domestic animals combines malignant and benign forms of neurofibroma and schwannoma under peripheral nerve sheath tumor.5–7
In dogs, the incidence of PNSTs is difficult to assess given that they bear marked histologic similarity to other spindle cell tumors and most diagnoses are made without ancillary immunohistochemical testing. Furthermore, routine markers are often nonspecific or overlap in specificity.5,6,9 PNSTs demonstrate consistent S100 positivity 2 but variable positivity for glial fibrillary acidic protein (GFAP) on immunohistochemical staining. PNSTs may occur as solitary or multiple masses (plexiform variant), are generally 2–3 cm in diameter, with lesions most commonly located on the trunk and distal limbs. Histologically, PNSTs are composed of uniform, fusiform cells arranged in wavy bundles, streams, and concentric whorls. Occasionally there are areas of densely packed neoplastic spindle cells forming short interlacing streams and bundles (Antoni A arrangement). These are intermingled with degenerative, loose myxoid tissue, which is often poorly cellular (Antoni B arrangement). Other features include palisading nuclei, with the formation of Verocay bodies, and hyalinized micro vessels. Neoplastic cells may be pigmented, and tumors can contain areas of cartilaginous and osseous metaplasia.2,6 A myxoid variant has also been reported. 6 PNSTs have also been described in cats, 16 cattle, 12 horses, 15 donkeys, 13 pigs, 17 and a chicken. 15
We describe here a case of cutaneous neoplasms with perineurial features and concurrent cardiac involvement in a dog. An 8-y-old intact male Labrador Retriever was referred to Clinica Veterinaria Monte Rosa, Italy in August 2019 because of the presence of innumerable, rapidly growing, firm, off-white, well-demarcated cutaneous masses. An excisional biopsy of a mass from the occipital region was submitted to Idexx Laboratories Italia and was diagnosed as “multicentric/plexiform neural proliferation” based on clinical data and histopathologic findings.
On clinical examination, pleural effusion was identified by thoracic radiography, and hematologic evaluation revealed a mild normocytic hyperchromic regenerative anemia and mild leukocytosis reflecting mild mature neutrophilia. A month later the dog was euthanized because of deteriorating clinical signs. At postmortem examination, myriad cutaneous masses up to 4 cm diameter, occasionally arranged in a linear fashion and ulcerated, were evident all over the body (Fig. 1). These masses were grossly similar to the one removed previously, and had developed rapidly over a month. An off-white, firm mass, ~3 cm diameter was located within the myocardium at the apex of the left ventricle (Fig. 2). Other organs within both the thoracic and abdominal cavities were grossly unremarkable.
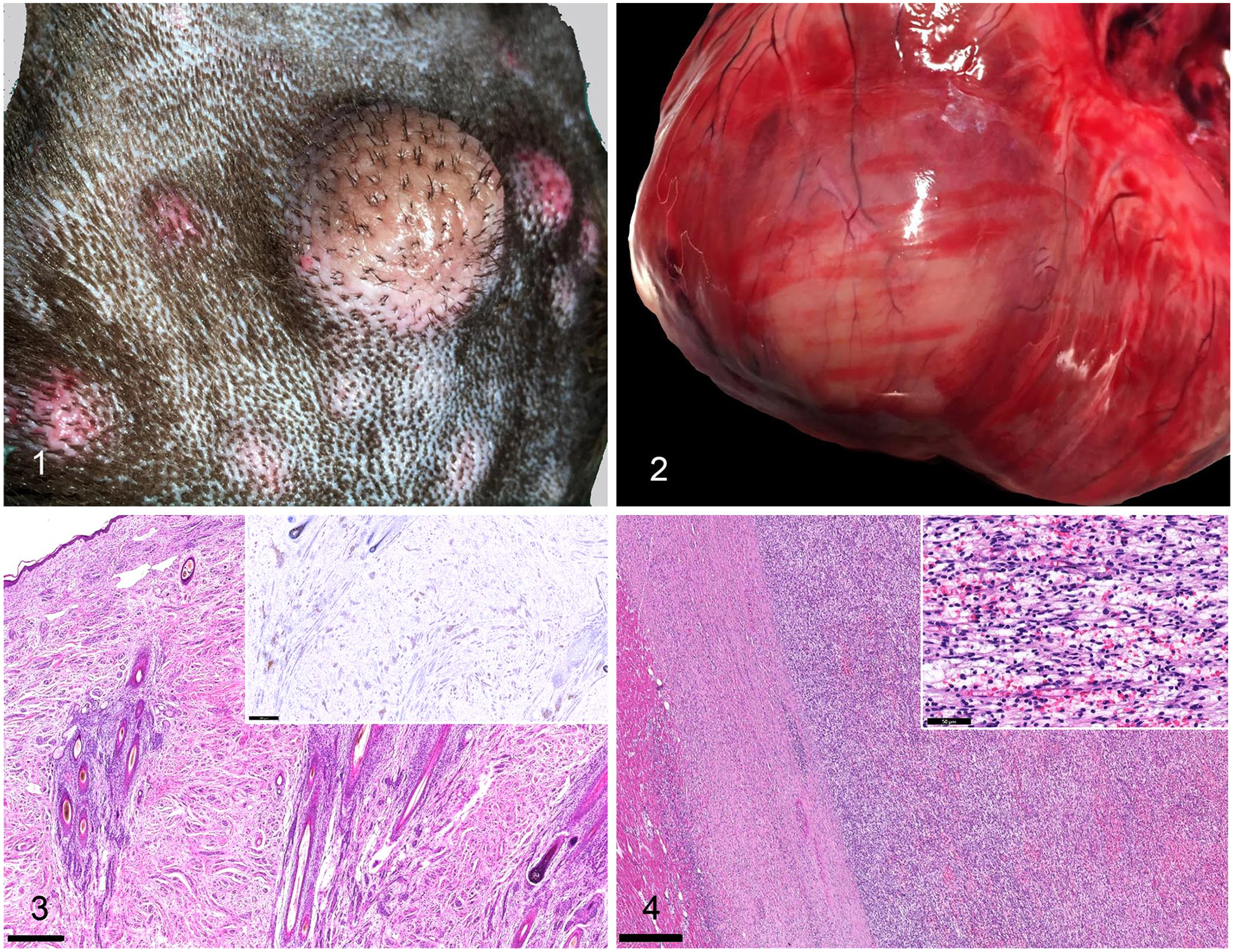
Variably sized cutaneous masses on the dorsum of an 8-y-old intact male Labrador Retriever dog.
Representative tissues were collected for histologic examination. Samples were fixed in 10% neutral-buffered formalin, processed routinely, and 4-µm sections stained with H&E. Analyses were performed using a panel of immunohistochemical stains (Table 1). Appropriate positive and negative controls were included.
Primary antibodies and conditions used for the immunohistochemical examination of cutaneous and cardiac neoplasms.

ABC = avidin-biotin complex (Vectastain Elite ABC kit PK-6100; Vector); GFAP = glial fibrillary acidic protein; HIER = heat-induced epitope retrieval, citrate buffer pH = 6.0; RT = room temperature.
Histologically, the cutaneous lesions were characterized by unencapsulated, densely cellular, infiltrative and multicentric neoplastic proliferations expanding the dermis and mainly arranged around adnexa. Cutaneous masses were composed of bundles of neoplastic cells supported by a moderate fibrovascular-to-myxoid stroma (Fig. 3). Neoplastic cells were fusiform, with scant eosinophilic fibrillar cytoplasm and paracentral, oval nuclei with coarsely clumped chromatin. Anisocytosis and anisokaryosis were mild. Mitoses were 0 in 2.37 mm2. There was no evidence of necrosis within examined sections. The myocardial mass contained a moderately well-demarcated densely cellular nodular neoplastic growth with the same histopathologic features as the cutaneous masses (Fig. 4). Immunohistochemically, neoplastic cells were diffusely positive for vimentin (data not shown) in both cutaneous and cardiac locations; up to ~90% of the neoplastic cells in the skin and up to 80% of those in the cardiac mass displayed patchy nuclear and cytoplasmic positivity for S100 (Fig. 3), and ~40% of the neoplastic cells in the skin and in the heart had positive cytoplasmic staining for GFAP (data not shown).
Overall, the histologic features and immunohistochemical staining profile were identical for both the cutaneous and cardiac masses, consistent with perineurial origin. It is unclear whether the cardiac lesion was a metastatic lesion originating from one of the cutaneous masses, or a concurrent manifestation of the multicentric and plexiform cutaneous neoplastic process. Given that no other visceral masses were identified at postmortem examination, as might be expected with metastatic disease, a de novo mass was considered most likely.
Interestingly, this case shares similarities with human neurofibromatosis type 1 (NF1; also known as von Recklinghausen disease), a relatively common inherited neurocutaneous disorder that results from a germline mutation in the NF1 tumor-suppressor gene. 8 Affected individuals are prone to develop neurofibromas, which often undergo malignant transformation and appear with additional neoplastic and non-neoplastic lesions in other organs, such as gliomas of the optic nerve, pigmented nodules of the iris (Lisch nodules), and cutaneous hyperpigmented macules (café-au-lait spots). 8 Furthermore, cardiac metastasis involving the conduction system of cutaneous MPNSTs that arose in the context of NF1 was reported in a 61-y-old man. 10 In our canine case, the spectrum seen in the human disease was lacking, and there was not sufficient evidence to prove that malignant transformation had occurred. Furthermore, no data were available regarding the siblings of the affected dog, and a possible genetic background for the condition cannot be proven. With these limitations, parallels with human type 1 neurofibromatosis remain speculative and therefore we consider this to be a “neurofibromatosis type 1–like” presentation.
The single cutaneous lesion initially submitted from our patient might have been classified as a cutaneous PNST to be included within the broad and heterogeneous group of canine cutaneous and subcutaneous soft tissue sarcomas (STSs), which are mesenchymal spindle cell neoplasms derived from connective tissue.7,11,14 Canine cutaneous and subcutaneous STSs are divided into 3 histologic grades based on cellular differentiation, mitotic count (MC) in 10 contiguous hpfs, and percentage of necrosis.1,4 However, given the cardiac involvement in our case, the classification and grading schemes proposed for cutaneous and subcutaneous STSs are not applicable, and our findings would warrant consideration of neurofibromatosis.
Furthermore, our case should be considered as malignant given its rapid clinical course, but it must be distinguished from MPNSTs. MPNSTs would have been characterized by infiltrative growth, increased cellularity, cellular pleomorphism, an increased mitotic rate, and areas of necrosis and/or evidence of metastases, which were features not detectable in our case. The neoplasms in our case were infiltrative in both cutaneous and cardiac locations but lacked other features of malignancy. In addition, the presence of cutaneous tumors and a similar size cardiac mass with no other evidence of metastasis would not be the expected behavior of a cutaneous or subcutaneous STS, which would be characterized by metastasis to the pulmonary parenchyma.
Our case shares some similarities with nodular dermatofibrosis, a genetic cancer syndrome observed predominantly in the German Shepherd dog breed and associated occasionally with mutation of the encoding folliculin gene (FLCN) and characterized by cutaneous lesions that might precede or coincide with unilateral or bilateral renal cysts, adenomas or carcinomas, and/or uterine leiomyomas. 3
To our knowledge, this presentation of concurrent cutaneous and cardiac involvement is a novelty in veterinary medicine, and we propose that it be considered a neurofibromatosis type 1–like lesion. Further investigation is required to develop our understanding of this neoplasm in dogs and to unveil possible underlying genetic alterations.
Footnotes
Acknowledgements
We thank the Histology Research Service, Diagnostic Services MVLS, School of Veterinary Medicine, University of Glasgow (UK).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
