Abstract
Multiple subcutaneous masses from two sibling bearded dragons were removed. Nodules were well demarcated, restricted to the subcutis, and soft, white to yellow, resembling adipose tissue. Histologically, the masses were composed of short interlacing streams and bundles of spindle cells, with regions of vague nuclear palisading. Two of the tumors contained a subpopulation of polygonal cells with abundant periodic acid-Schiff (PAS)-positive cytoplasmic granules. Neoplastic cells were immunohistochemically positive for S100 and neuron-specific enolase (NSE) but negative for desmin and smooth muscle actin. Electron microscopy and reticulin stains demonstrated a continuous basal lamina separating intertwining cells. Histologic, ultrastructural, and immunohistochemical features were consistent with a peripheral nerve sheath origin. At 1 year postexcision, local reoccurrence of a single incompletely excised mass from the left shoulder was noted.
Keywords
Peripheral nerve sheath tumors (PNSTs) are a heterogeneous group of neoplasms that originate from cells composing the nerve sheath (i.e., Schwann cells, fibroblasts, or peri-neurial cells). 7 Subclassifications as neurofibromas, neurofibrosarcomas, neurilemmomas, and schwannomas in animals are arbitrary and based on extrapolation of human tumor studies. 7 In dogs, cats, and cattle, benign tumors are generally presumed to be of Schwann cell origin and thus are called schwannomas. 7 Morphology, biological behavior, immunohistochemistry, and ultrastructural features are frequently necessary to differentiate benign peripheral nerve sheath tumors from other mesenchymal neoplasms. 7
Two adult bearded dragon siblings, a 6-year-old female (No. 1) and a 5-year-old male (No. 2), presented with multiple small subcutaneous masses in April and May 2002. Location of the masses on bearded dragon No. 1 included right shoulder and dorsal aspect of the tail, and on bearded dragon No. 2, left shoulder, left hindlimb, and the ventral aspect of the tail. The size of the nodules varied from 2 to 10 mm in diameter. All nodules were restricted to the subcutis, discrete, and easily excised (Fig. 1). Masses were soft, white to yellow, and resembled adipose tissue. Specimens were fixed in 10% neutral buffered formalin, routinely processed, sectioned at 5 μm, and stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS) reaction, and Manuel's reticulum. For immunohistochemistry, deparaffinized and rehydrated sections of tumor were immersed in hydrogen peroxide in methanol for 15 minutes and rinsed in Optimax buffer (BioGenex, San Ramon, California, USA). Microwave antigen retrieval with citrate buffer (pH 6) was used. Commercial primary antibodies were polyclonal rabbit anti-cow S100 antibody (Dako, Carpinteria, California), monoclonal mouse anti-human neuron-specific enolase (NSE) antibody (Dako), monoclonal mouse anti-human desmin antibody (Biogenex), and monoclonal anti–smooth muscle actin antibody (Biogenex). S100, desmin, and smooth muscle actin slides were incubated at room temperature for 30 minutes and NSE for 1 hour. A blocking reagent (Power Block, Biogenex) was applied for 10 minutes. A commercial streptavidin–biotin–peroxidase detection system (Super Sensitive Multilink, Biogenex) was used. Positive control slides consisted of mammalian adrenal gland (NSE), mammalian peripheral nerve (S100), and mammalian skeletal muscle and small intestine (desmin and smooth muscle actin). Slides containing the tumors and the tissues used for positive control were stained with buffer and served as negative control. For ultrastructural studies, formalin-fixed tissues were deparaffinized, hydrated, fixed in Karnovsky's fixative and 2% gluteraldehyde with 2.5% paraformaldehyde, postfixed in 1% osmium tetroxide, dehydrated, and embedded in epoxy resin. Sections were cut at 0.35 μm and stained with toluidine blue and basic fuchsin for preliminary microscopic examination. Samples were sectioned at 80–90 nm, stained with uranyl acetate and lead citrate, and examined with a Hitachi H600 transmission electron microscope.
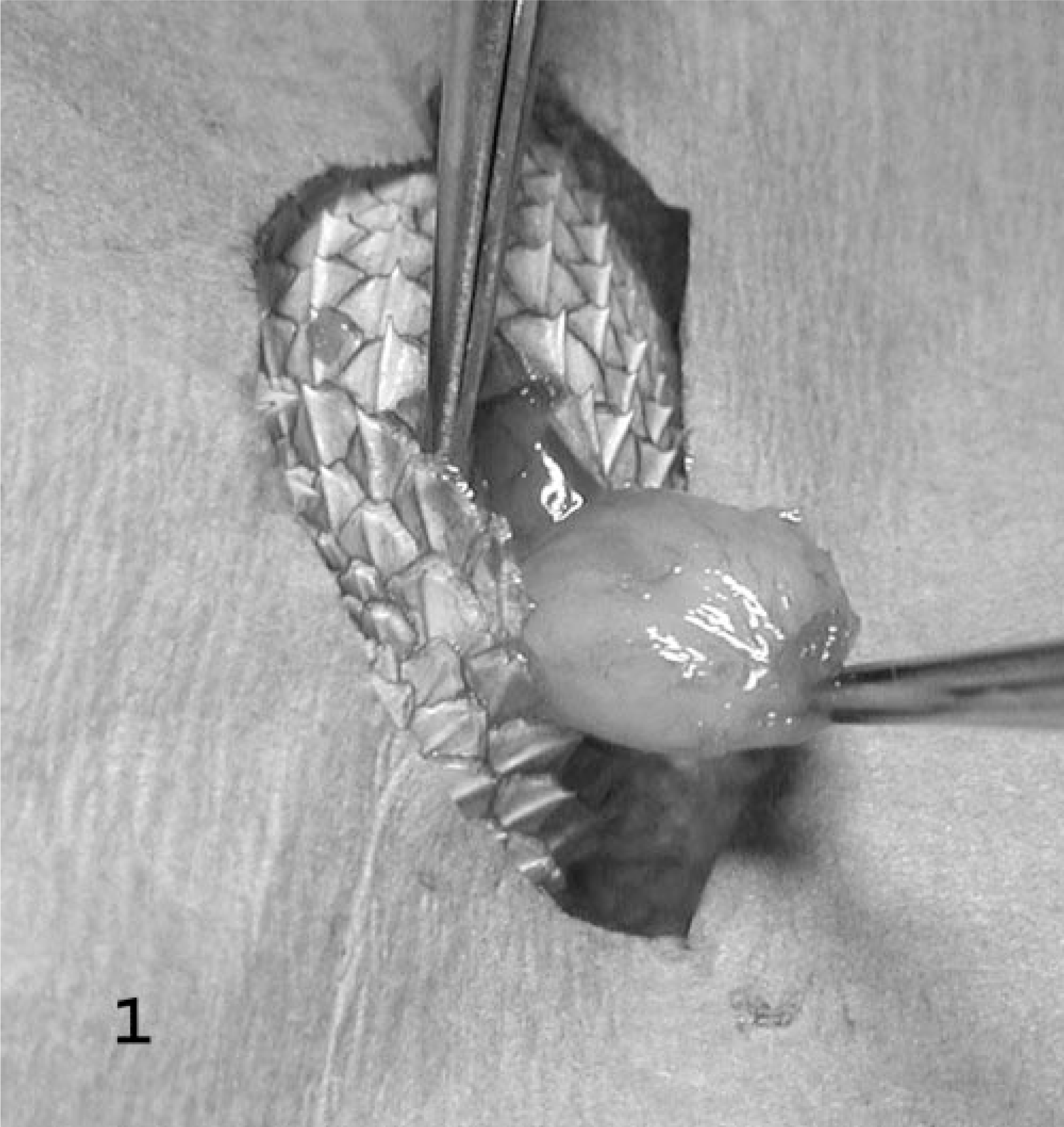
Left hindlimb; bearded dragon No. 2. Surgical excision of a subcutaneous mass.
Histologically, masses were well demarcated and partially encapsulated, reflecting their gross appearance. In general, they were composed of short interlacing streams and bundles of spindle cells supported by scant fibrovascular stroma (Fig. 2), and two masses from bearded dragon No. 2 had a sub-population of large polygonal cells with multiple small eosinophilic intracytoplasmic granules. There was occasional vague nuclear regimentation (Fig. 2). Patterns were nonspecific but in some regions appeared to have an Antoni type A and B arrangement, as described in schwannomas in humans, 1 characterized by densely cellular areas in which the cells are arranged in interwoven bundles, streams, and concentric whorls (Antoni type A) and cells in lower density, with small, dark nuclei, embedded in a loose fibrous stroma (Antoni type B). Individual cells contained moderate amounts of pale eosinophilic fibrillar to foamy cytoplasm. Nuclei were oval to fusiform, central, and hyperchromatic or had finely stippled chromatin and a small magenta nucleolus. Mitotic figures were rare. A PAS stain of the large polygonal cells revealed numerous PAS-positive cytoplasmic granules (Fig. 3). A reticulin stain demonstrated wrapping of individual neoplastic cells within a network of reticulum (Fig. 4), demonstrating that the cells had a continuous basal lamina, which was confirmed by transmission electron microscopy (TEM; Fig. 5). Transmission electron microscopy also revealed occasional intertwining cell processes (Fig. 5). An immunohistochemical stain to detect the presence of S100 demonstrated diffuse weak cytoplasmic and strong nuclear staining. A NSE stain demonstrated diffuse strong immunoreactivity in the neoplasms. Immunohistochemistry stains for desmin and smooth muscle actin were negative.
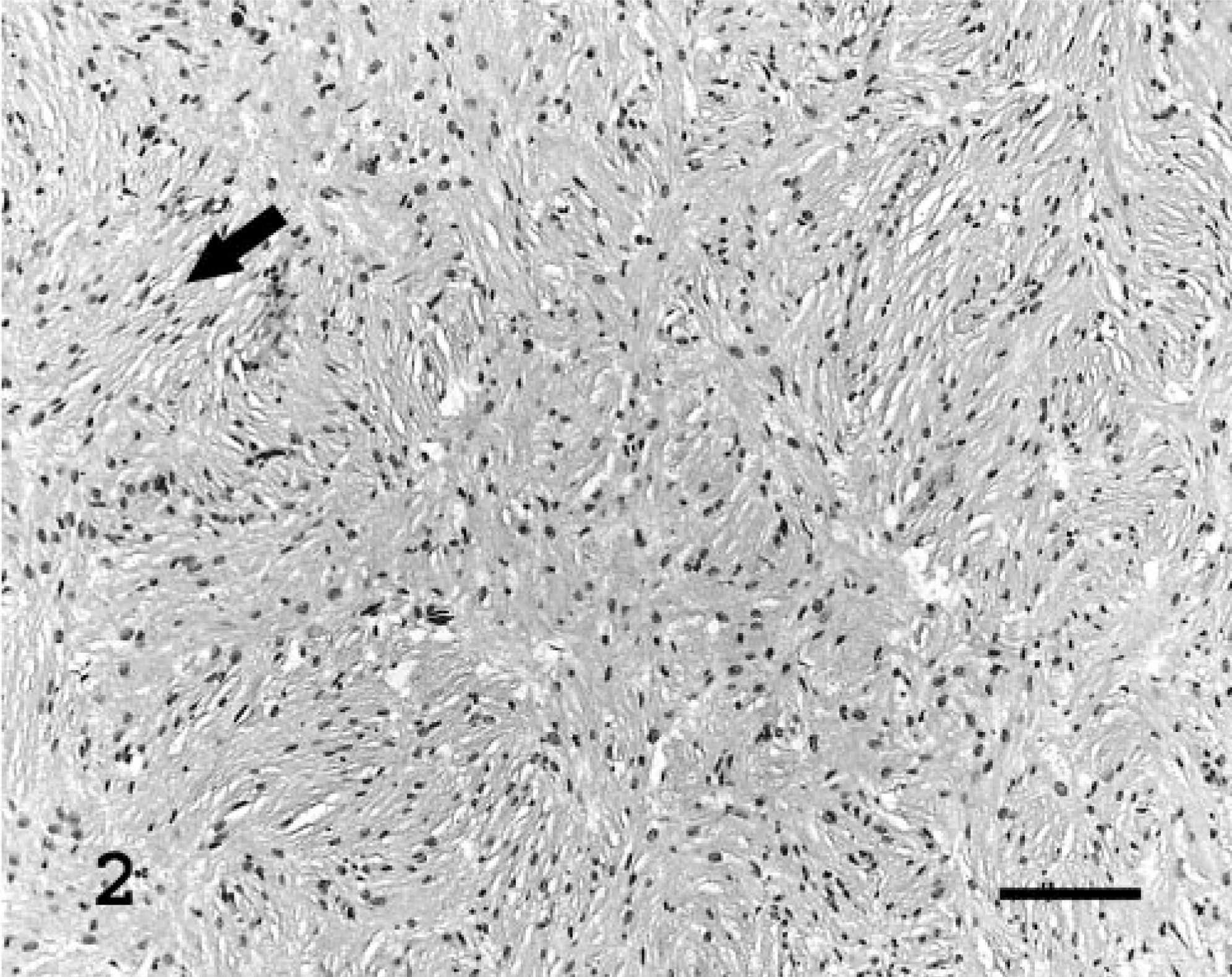
Subcutaneous mass, left shoulder; bearded dragon No. 2. Short interlacing streams and bundles of neoplastic spindle cells are supported by scant fibrovascular stroma. The arrow shows vague nuclear regimentation. Stain HE. Bar = 100 μm.
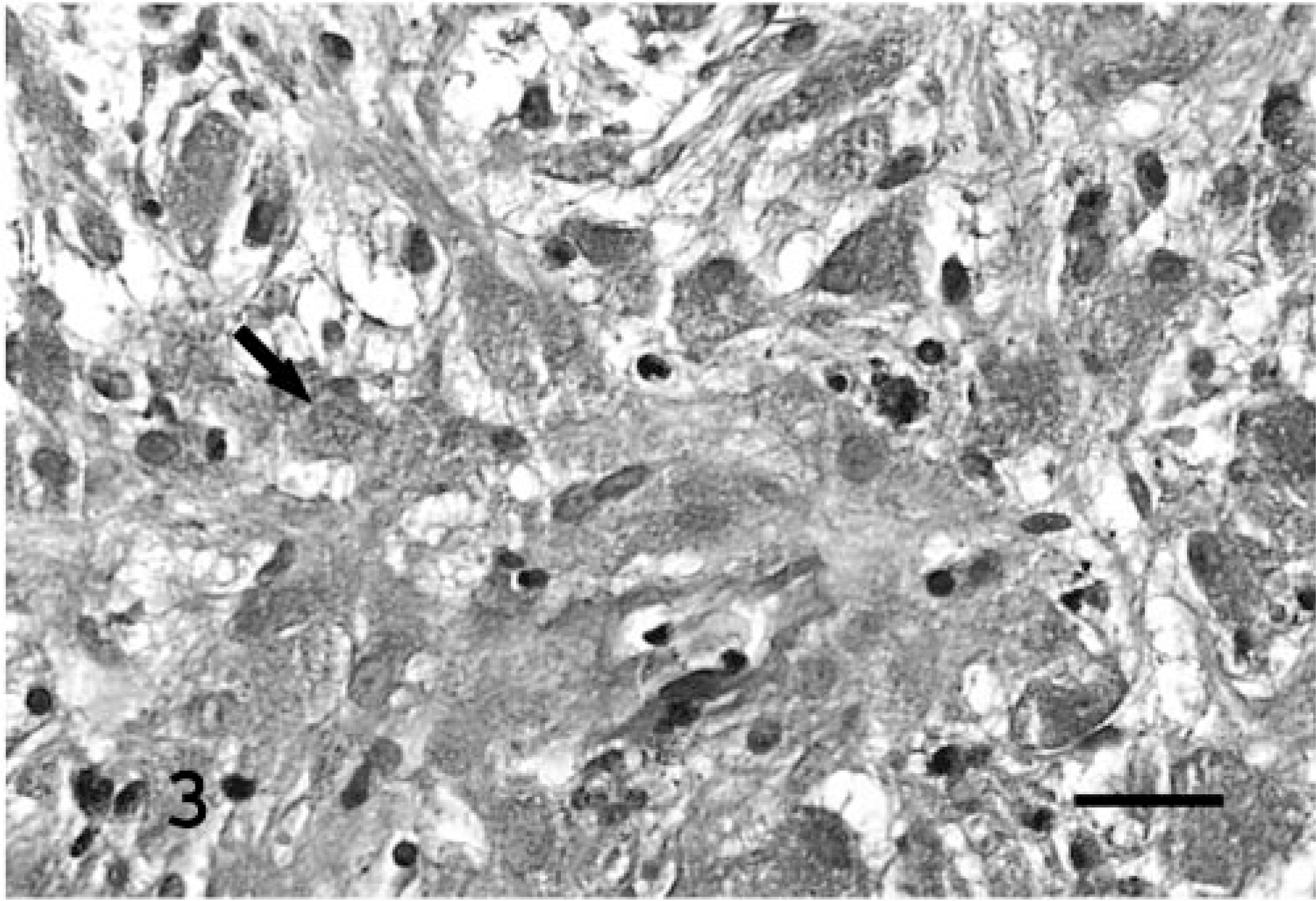
Subcutaneous mass, left shoulder; bearded dragon No. 2. The subpopulation of large polygonal cells contain numerous cytoplasmic granules (arrow). Stain periodic acid–Schiff; counterstain hematoxylin. Bar = 50 μm.
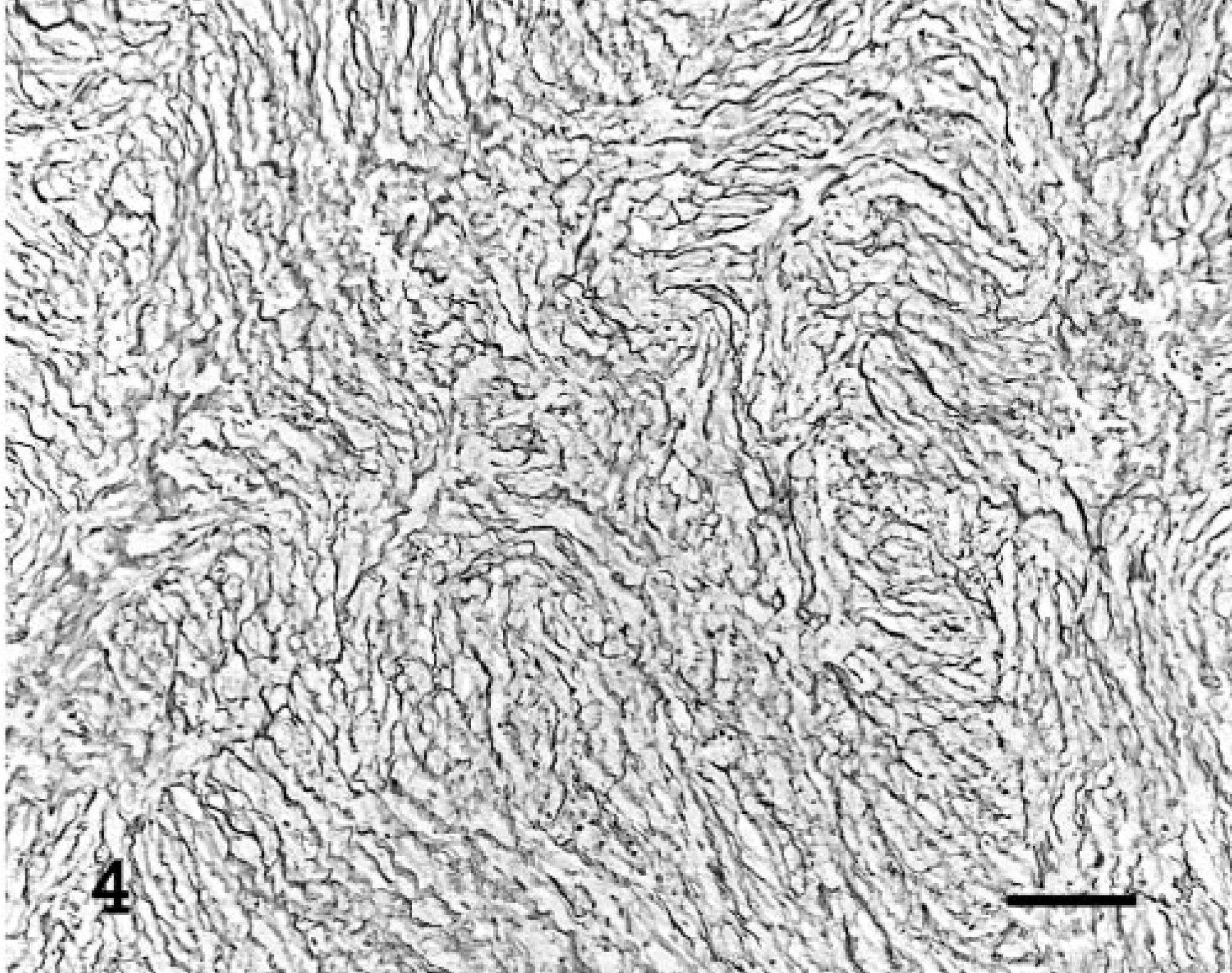
Subcutaneous mass, ventral aspect of the tail; bearded dragon No. 1. Individual neoplastic cells are wrapped within a network of reticulum. Stain reticulin; counterstain light green. Bar = 100 μm.
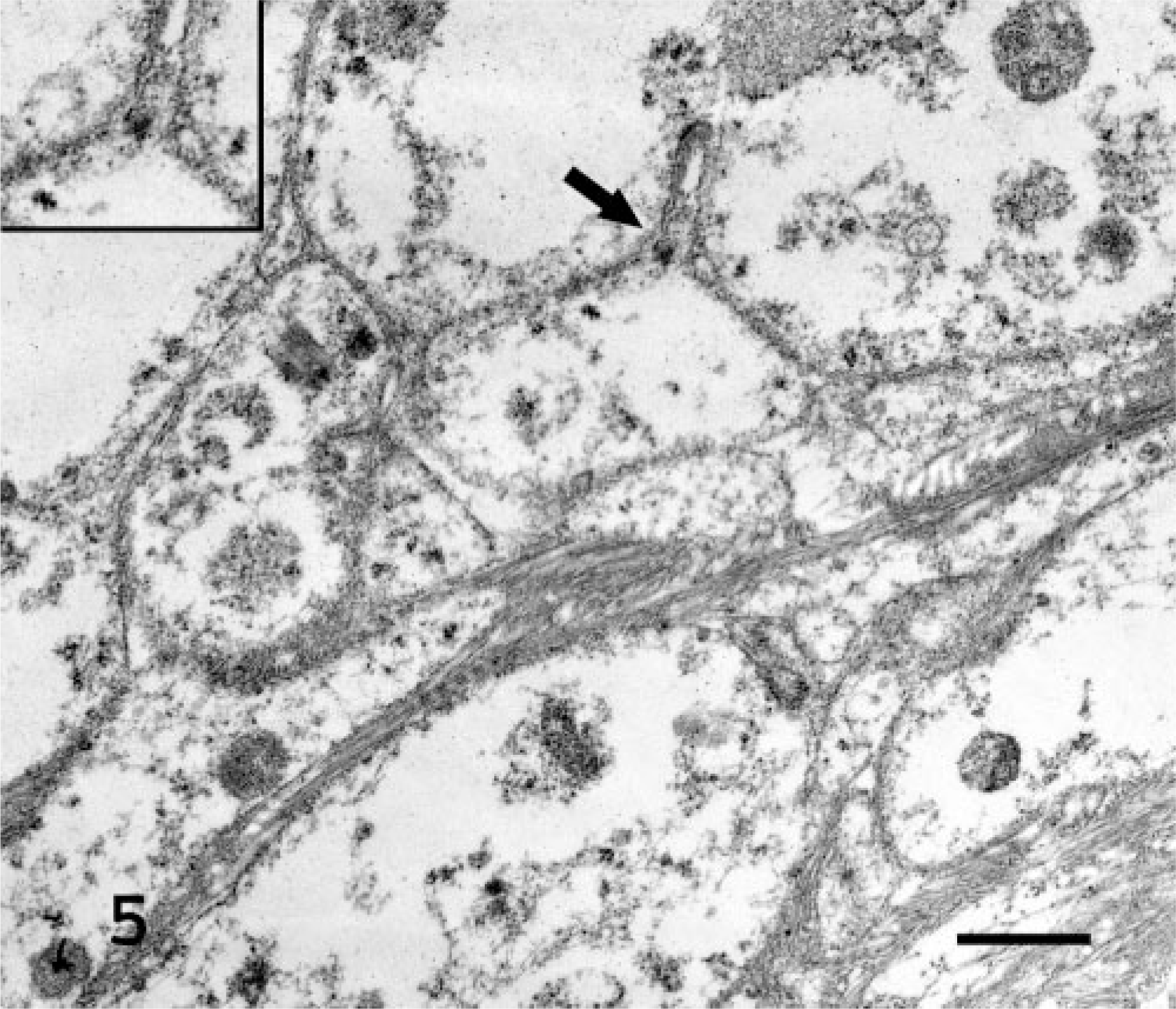
Subcutaneous mass, left shoulder; bearded dragon No. 2. Electron micrograph. Ultrastructural appearance of neoplastic cells which are separated by a continuous basal lamina (inset and arrow). Stain uranyl acetate and lead citrate. Bar = 10 μm.
At 18 months postexcision, bearded dragon No. 2 developed five more subcutaneous masses ranging from 2 to 10 mm in diameter, on which very slow growth had been observed. One of the masses located in the left shoulder likely represented local reoccurrence of a previously incompletely excised mass. Three masses were surgically excised on 27 January 2004 and were histologically similar to the previous neoplasms.
Histologic, ultrastructural, and immunohistochemical features were consistent with a peripheral nerve sheath origin. The subpopulation of PAS-positive granuleladen cells resembles cells of granular cell tumors (GCT), and the majority of GCTs involving soft tissue are believed to be derived from Schwann cells. 2 Granular cell tumors are rare in domestic animals and reported most commonly in dogs and horses. 2 S100 immunohistochemical staining is typically positive in schwannomas, 5 although staining might be inconsistent. 7 This stain has not yet been validated in reptiles; however, the peripheral nerves of another unrelated bearded dragon used as a control demonstrated strong immunoreactivity. NSE is an enzyme detected in cells that demonstrate neuronal and neuroendocrine differentiation, and schwan-nomas have occasional nonspecific staining for NSE. 3 Similar results were obtained with NSE applied to normal peripheral nerve from an unrelated bearded dragon. Immunohistochemical staining for smooth muscle actin was validated by application to sections of cloaca with distal intestine from another unrelated bearded dragon. Similar validation of desmin immunohistochemistry could not be obtained, and the negative staining of neoplastic cells was considered equivocal. The presence of a continuous basal lamina was supportive of a diagnosis of benign peripheral nerve sheath tumor, versus a malignant PNST or a neurofibroma, which are characterized by a discontinuous lamina. 4 The biological behavior further supported the diagnosis of benign peripheral nerve sheath tumor. 7 Histologic appearance, positive immunoreactivity of the neoplasms for S100, and presence of a continuous basal lamina also helped distinguish these tumors from sarcomas of undetermined origin. Undifferentiated sarcomas typically do not have a distinct histologic pattern and generally have a discontinuous lamina propria.
To the authors' knowledge, this is the first report of a benign peripheral nerve sheath tumor in a reptile. Malignant peripheral nerve sheath tumors have previously been reported in a bearded dragon 8 and a water moccasin 9 and presented as solitary, invasive, nonresectable masses with multicentric metastases. In dogs and cats, schwannomas are typically solitary masses. 7 Multicentric schwannomas have been reported in old cattle, 6, 7 and multicentric schwannomas and neurofibromas have been noted in goldfish. 10 Multicentric benign peripheral nerve sheath tumors have been described most extensively in humans. 11 Neurofibromatosis is a heritable condition characterized by multicentric cutaneous and visceral neurofibromas. This condition is subdivided into two types. Neurofibromatosis type 1, or Von Recklingshausen disease, is the most common form and is characterized by café au lait spots on the skin and neurofibromas in the skin and occasionally in the spinal cord. The hallmark lesion of neurofibromatosis type 2 is bilateral acoustic schwannomas. A syndrome called schwannomatosis has been described more recently and is characterized by cutaneous schwannomas and central nervous system tumors. It is debated whether schwannomatosis represents a clinical entity distinct from neurofibromatosis type 2. 11
Given the unusual simultaneous occurrence of multiple benign tumors in two related individuals, the question arises whether this is a heritable disease and whether similar syndromes as noted in humans could exist in lower vertebrates.
Footnotes
Acknowledgements
We thank Drs. Mike Kinsel and Marie Pinkerton for insightful comments; Drs. Kathryn Gamble and Claude Lacasse and the animal care staff of Lincoln Park Zoo for their assistance; and Lou-Ann Miller from the electron microscopy laboratory and Jane Chladny and the technicians of the histology laboratory of the University of Illinois Veterinary Diagnostic Laboratory for excellent technical support.
