Abstract
To determine the transmissibility and pathogenicity of sheep scrapie and transmissible mink encephalopathy (TME) agents derived from raccoons (first passage), raccoon kits were inoculated intracerebrally with either TME (one source) or scrapie (two sources—each in separate groups of raccoons). Two uninoculated raccoon kits served as controls. All animals in the TME-inoculated group developed clinical signs of neurologic dysfunction and were euthanatized between postinoculation month (PIM) 6 and 8. Raccoons in the two scrapie-inoculated groups manifested similar clinical signs of disease, but such signs were observed much later and the animals were euthanized between PIM 12 and 18. Necropsy revealed no gross lesions in any of the raccoons. Spongiform encephalopathy was observed by use of light microscopy, and the presence of protease-resistant prion protein (PrPres) was detected by use of immunohistochemical (IHC) and Western blot analytic techniques. Results of IHC analysis indicated a distinct pattern of anatomic distribution of PrPres in the TME- and scrapie-inoculated raccoons. These findings confirm that TME and sheep scrapie are experimentally transmissible to raccoons and that the incubation periods and IHC distribution for both agents are distinct. Therefore, it may be possible to use raccoons for differentiating unknown transmissible spongiform encephalopathy (TSE) agents. Further studies, with regard to the incubation period and the pattern of PrPres deposition by use of IHC analysis in bovine spongiform encephalopathy and for other isolates of scrapie, chronic wasting disease, and TME in raccoons are needed before the model can be further characterized for differentiation of TSE agents.
Keywords
Transmissible spongiform encephalopathies (TSEs) are fatal neurologic diseases. Three animal TSEs, namely, scrapie, transmissible mink encephalopathy (TME), and chronic wasting disease (CWD), have historically been present in the USA for some time. Recently, two cases of bovine spongiform encephalopathy (BSE) were diagnosed in cattle, one being an imported cow from Canada and the other an indigenous case. At present, the only reliable method for identification of TSE strains is use of the mouse model. This method is based on the duration of incubation period and the neurologic lesion profile in three to five strains of specially bred mice. 4 The inbred mouse model requires an incubation period of 1 to 2 years, and at present, is routinely done at only a few TSE laboratories. In addition to these disadvantages, TME has failed to propagate in mice, 12, 17 and only one reported incidence of infrequent TME transmission in this species has been reported (M. Bruce, personal communication).
Two recent reports indicated that TME and sheep scrapie can be transmitted to raccoons (Procyon lotor) within 6 months and 2 years, respectively. 10, 11 On the basis of these findings, raccoons have been suggested as a possible animal model for identification of indigenous TSEs in the USA. 10, 11 The advantage over the mouse model would be that raccoons are readily available in this country; they are susceptible to TME (whereas mice are not); and the results would be obtained quickly. However, before the raccoon can be recommended as a laboratory model for strain typing of indigenous TSEs in the USA, experimental studies using individual TSE isolates from naturally infected hosts and from experimentally infected raccoons with TSEs (i.e., additional passages in raccoons) need to be evaluated to verify that the various TSE isolates have well defined incubation periods. The study reported here was designed to obtain data for the second passage of two scrapie isolates and one TME isolate from earlier (first passage) experimental studies. 10, 11
Fifteen 8- to 10-week-old raccoon kits vaccinated against canine distemper were purchased from a commercial breeder with no prior history of TSE-like disease in his breeding animals. The raccoons were randomly assigned to two scrapie-inoculated groups (n = 4 each); one TME-inoculated group (n = 5); and an uninoculated control group (n = 2). The kits were group-housed in a biosafety level-2 containment facility at the National Animal Disease Center, Ames, Iowa (Table 1; Fig. 1). The raccoons were fed dry dog food (Premium formula, PMI Nutrition Inc., Brentwood, MO) twice a day, and clean water was available ad libitum. Personnel wore protective clothing while in the isolation facility and showered before leaving the facility.
Findings in raccoons intracerebrally inoculated with isolates of scrapie or transmissible mink encephalopathy obtained from raccoons with clinical transmissible spongiform encephalopathy(TSE).
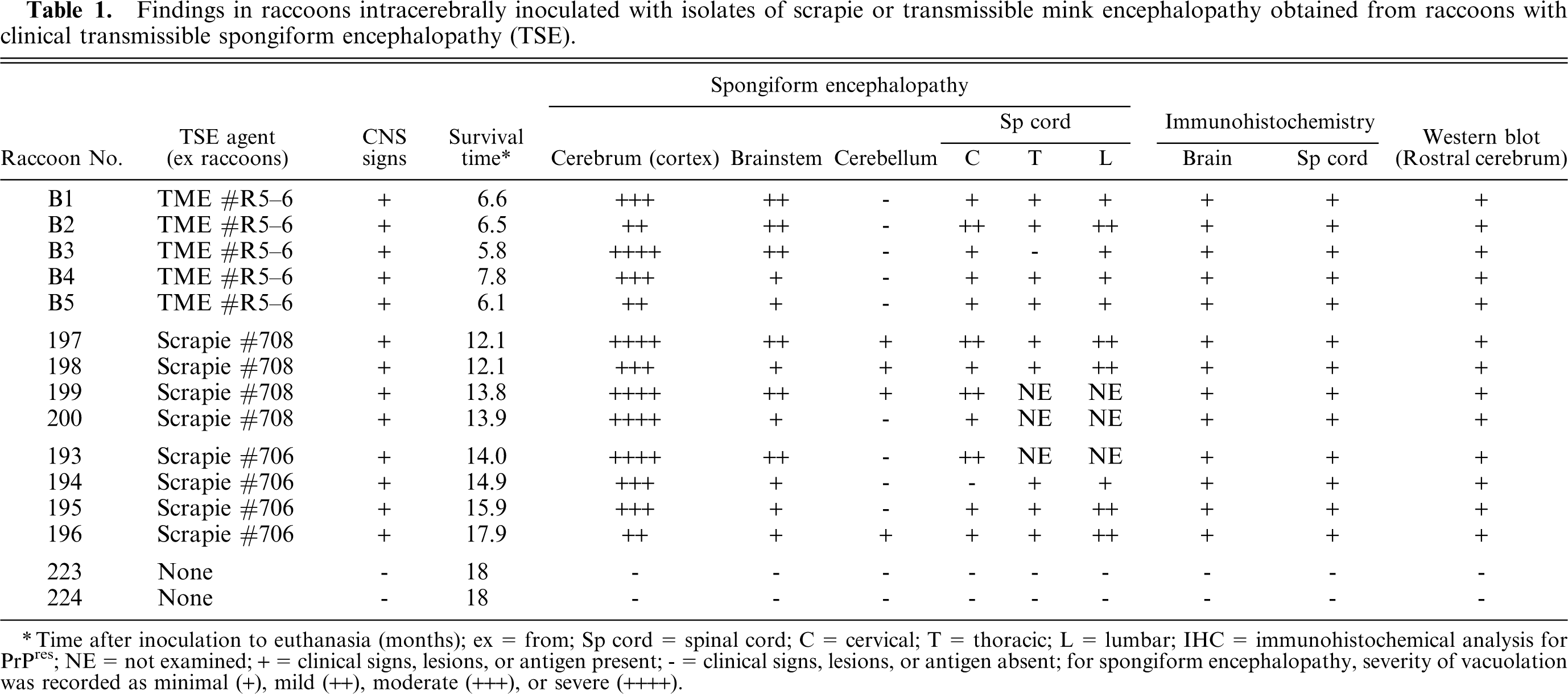
∗Time after inoculation to euthanasia (months); ex = from; Sp cord = spinal cord; C = cervical; T = thoracic; L = lumbar; IHC = immunohistochemical analysis for PrPres; NE = not examined; + = clinical signs, lesions, or antigen present; - = clinical signs, lesions, or antigen absent; for spongiform encephalopathy, severity of vacuolation was recorded as minimal (+), mild (++), moderate (+++), or severe (++++).
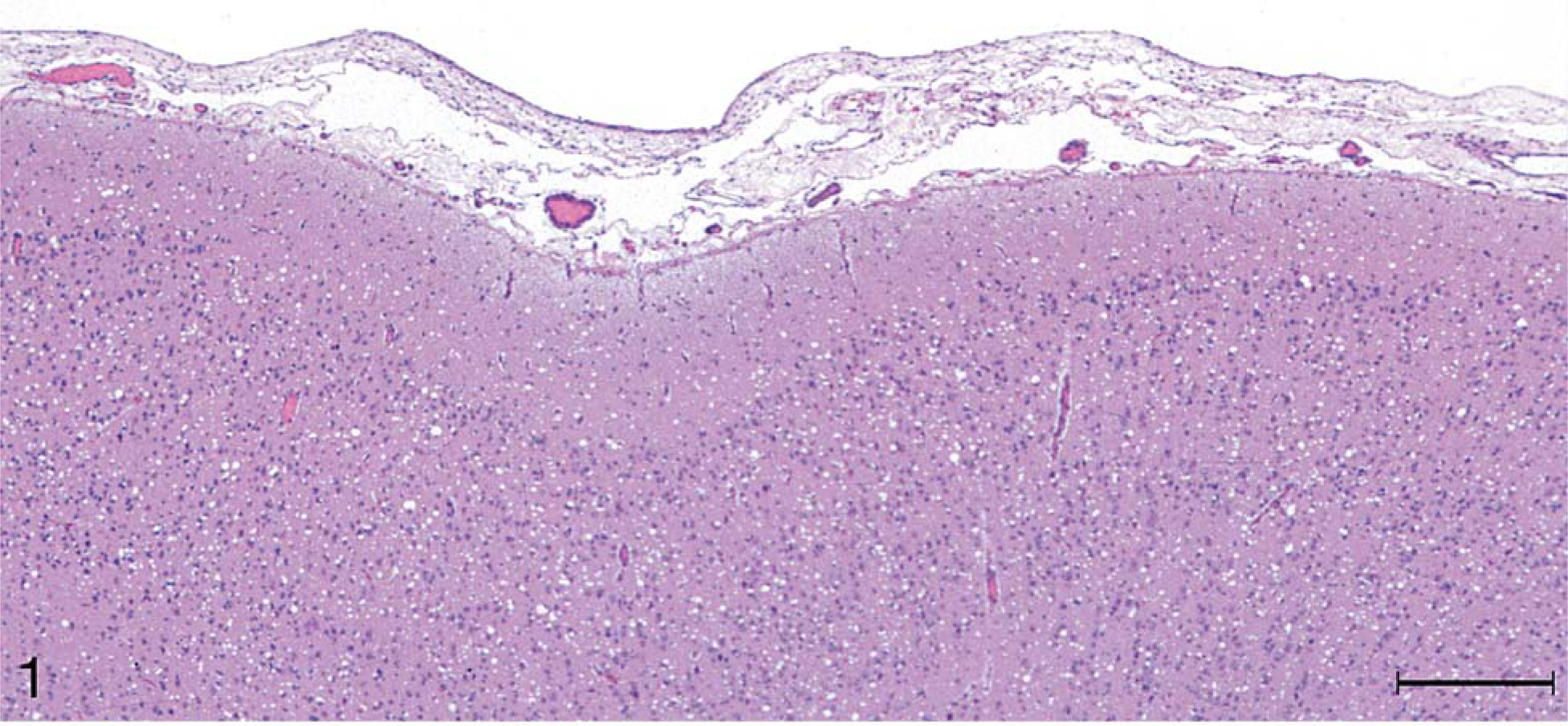
Brain, cerebrum; raccoon No. 199 inoculated with scrapie. Notice diffuse spongiform encephalopathy in the cortex. HE. Bar = 250 µm.
The scrapie inoculum was prepared from brains of two individual raccoons (isolate Nos. 708 and 706; Table 1) that had developed clinical TSE during a previous experiment. 10 The raccoon from which isolate No. 708 was obtained was euthanatized on PI day (PID) 554. Prior to euthanasia, this animal had begun to exhibit signs of self mutilation. Isolate No. 706 was obtained from a raccoon which had survived longer and had been euthanized on PID 692. At the time of euthanasia, this raccoon was lethargic, unaware of its surroundings, and was unresponsive to external stimuli. 10 These raccoons had been given a scrapie inoculum that had been prepared from a pool of 13 infected sheep brains from seven source flocks (inoculum No. 13-7). The TME inoculum was obtained from a raccoon (No. R5–6) that had developed clinical signs of TSE during a previous study. 11 The inoculum for that experiment was from the Stetsonville, Wisconsin, outbreak of TME in 1985, and had been biologically cloned in mink three times by use of limiting end-point titration. 3 Brain tissues of all three animals had microscopic lesions of spongiform encephalopathy and were positive for PrPres by use of immunohistochemical and Western blot analyses. The material for the inocula was ground in a hand-held tissue grinder, and the final concentration of 10% (wt/vol) was made with phosphate-buffered saline (PBS).
The three groups of experimental raccoons were inoculated intracerebrally with 0.1 ml of scrapie isolate No. 706 or No. 708, or with TME isolate No. R5–6 (Table 1). The procedure for inoculation has been described. 10, 11 Briefly, the raccoon kits were sedated with a mixture of ketamine and xylazine, a midline incision was made in the skin at the junction of the parietal and frontal bones, and a 2-mm hole was drilled through the calvarium. The inoculum was injected into the midbrain via a disposable 22-gauge, 1.5-inch-long needle while withdrawing the needle from the brain. The skin incision was closed by use of a single suture. After each group of inoculations, the surgical instruments including the drill bit were discarded. Two kits (controls) were not inoculated (Table 1).
Raccoons were euthanatized by administration of a pentobarbital overdose, and complete necropsy was conducted on each of the carcasses. Brain (except for a small portion of frontal cortex that was frozen) and spinal cord, together with representative samples of heart, diaphragm, tongue, masseter muscle, lung, liver, gall bladder, kidney, pancreas, skin, spleen, eye, mesenteric lymph node, stomach, intestines, tonsil, trachea, thyroid, esophagus, thoracic aorta, adrenal gland, salivary gland, and urinary bladder, were immersion fixed in neutral-buffered 10% formalin for histologic examination. The fixed brain was cut in serial 2- to 4-mm width coronal sections, and sections of the medulla, pons, cerebellum, superior colliculi, and cerebrum (parietal and frontal sections) were processed in routine manner for histologic examination. Three sections each of the cervical, thoracic, and lumbar parts of the spinal cord were selected for examination. All tissue sections were also stained using an immunohistochemical (IHC) analytic method for detection of PrPres. 11 A cocktail of two monoclonal primary antibodies was used. These antibodies would recognize PrP sequences conserved in most mammalian species in which natural TSEs have been reported. 13 A Western blot analytic method (Prionics-Check, Zurich, Switzerland) was used on frozen brain (rostral area of cerebrum) as described. 15
Frozen tissues (spleen) were used for characterization of the raccoon PrP gene polymorphism. The primers used for the amplification of PrP sequences spanning codons 23–243 of the open reading frame (ORF) were: forward primer 5′-CTG CAA GAA GCG GCC GAA GCC-3′ and reverse primer 5′-CAC GGG CGG CGG GGA GAA GAG GAT-3′. The RNA was isolated from 10% homogenates of frozen spleens in PBS using the QIAamp® Viral RNA mini kit according to the manufacturer's instructions (Qiagen, Valencia, CA). Reverse transcriptase-polymerase chain reaction (RT-PCR) analysis was performed in a 50-µl reaction volume using the OneStep RT-PCR kit (Qiagen, Valencia, CA) under the following conditions: 30 minutes at 50°C and 15 minutes at 95°C, followed by 30 cycles of denaturation, annealing, and extension at 94°C (30 seconds), 62°C (30 seconds), and 72°C (60 seconds), with a 72°C, 10-minute final extension. Eight microliters of RNA template, 400 µM dNTP, 2.5 mM MgCl2, 2.5 U of RNase inhibitor/reaction, and 20 pM each primer were used. Amplified DNA products were purified using a GENECLEAN® spin kit (BIO101®, Q-Biogene, Ervine, CA), and sequencing was performed using the ABI prism 3700 DNA sequencer with Big Dye Terminator chemistry and the forward and reverse primers.
Within PIM 6.5, all five raccoons of the TME-infected group developed signs of abnormal CNS function and were euthanatized. Raccoons of the two scrapie-infected groups developed similar signs of neurologic dysfunction at later times and were euthanatized between PIM 12.1 and 17.9, respectively (Table 1).
Signs of lethargy and lack of response to external physical stimuli developed gradually over a period of a few weeks and became progressively worse. Eventually, the raccoons were not able to climb up to or get down from their sleeping quarters and were generally unaware of their surroundings. None of them manifested a tendency for self mutilation. All were euthanatized before they became recumbent. The uninoculated control raccoons (Nos. 9, 10; Table 1) were euthanatized at termination (18 months after initiation) of the study. At necropsy, all carcasses were in good-to-fair nutritional state, with evidence of moderate fat deposition. Gross lesions were not seen in any animal.
Microscopic lesions of spongiform encephalopathy were observed in the gray matter of brain and spinal cord sections from all 13 inoculated raccoons (Table 1). The severity of the lesions was variable among individual animals and within the various neuroanatomic sites. In general, the spongiform change was most prominent in the cerebral cortex, manifested as severe, extensive, clear vacuolation of the neuropil (Figs. 1, 2). The vacuoles were round to oval and varied in size from 10 to 80 µm in diameter. In some regions affected by the spongiform change, there was mild to moderate gliosis. Similar, but generally less severe spongiform lesions were present in most nuclei of the brain stem and in gray matter of the spinal cord. Vacuolation of neuronal perikarya, although present in isolated neurons, was not seen in the cerebral cortex but was observed sporadically in brain stem and spinal cord sections. In both groups of scrapie-inoculated raccoons, vacuolation was seen in the molecular layer of cerebellum (Fig. 3; Table 1). Neuronal degeneration was not observed in any raccoon. Examination of the choroid plexus revealed a few isolated blood vessels with concentric areas of mineralization (psammoma bodies) in four raccoons (two inoculated and two controls).
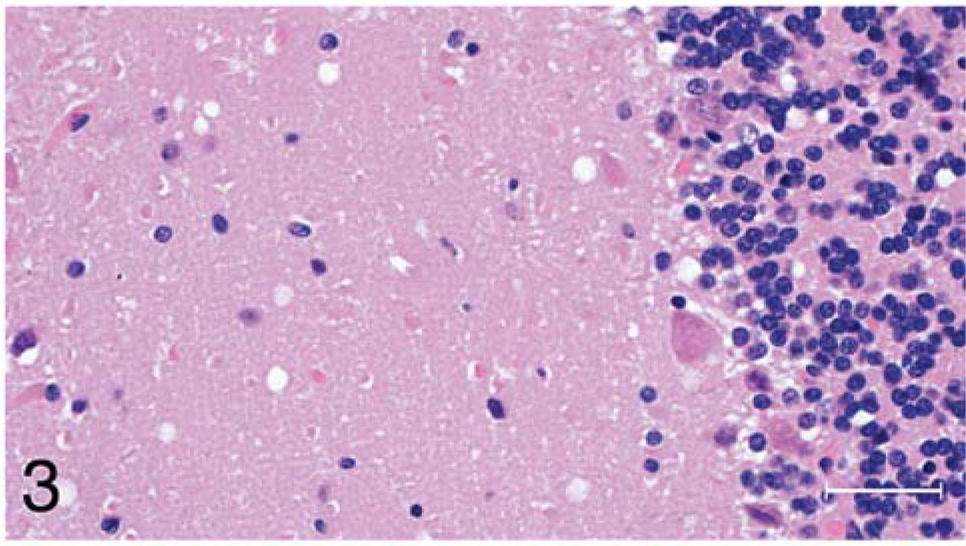
Brain, cerebellum; raccoon No. 199 inoculated with scrapie. Notice the presence of a few clear vacuoles in the molecular layer. HE. Bar = 50 µm.
Examination of IHC-stained sections for PrPres revealed moderate to heavy staining throughout the gray matter of the brain and spinal cord of all inoculated raccoons. The staining characteristics, however, were quite different between the group that was inoculated with TME and the two groups inoculated with scrapie isolates.
For TME inoculates, there was a diffuse and evenly distributed punctate staining pattern that was restricted to the neuropil, the neurons remaining generally unstained (Fig. 4). This type of staining also was observed for the scrapie inoculates, but a more prominent feature for those was a marked accumulation of coarse granular staining in cells, most notably within large neurons (Fig. 5). Clusters of stained granules were also present in or near other cells that appeared to be glial cells or, possibly, small neurons.
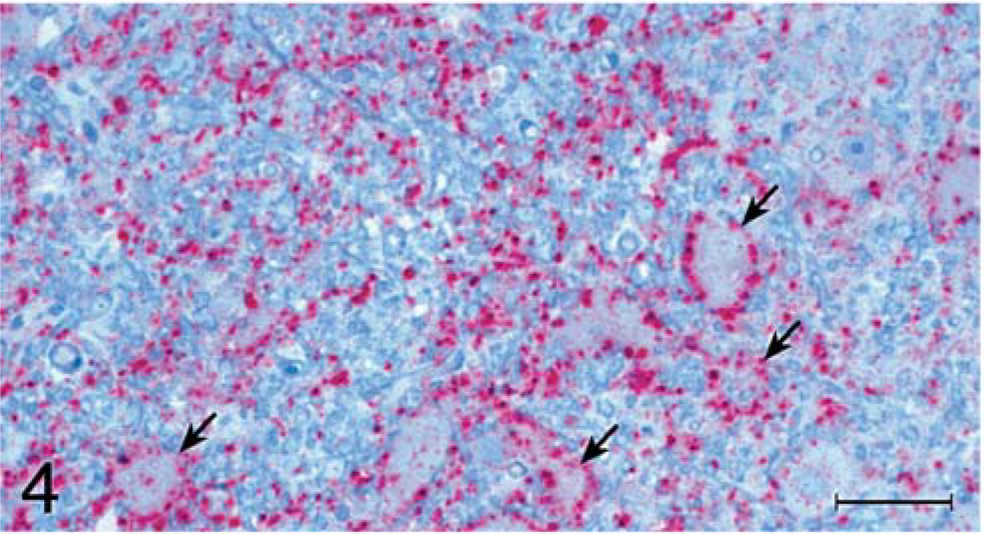
Brain, superior colliculus; raccoon No. B4 inoculated with TME. Accumulation of protease-resistant prion protein (PrPres [stained red]) is located predominantly in neuropil and around neurons (arrows). Stained for PrPres by use of immunohistochemical (IHC) analysis. Bar = 50 µm.
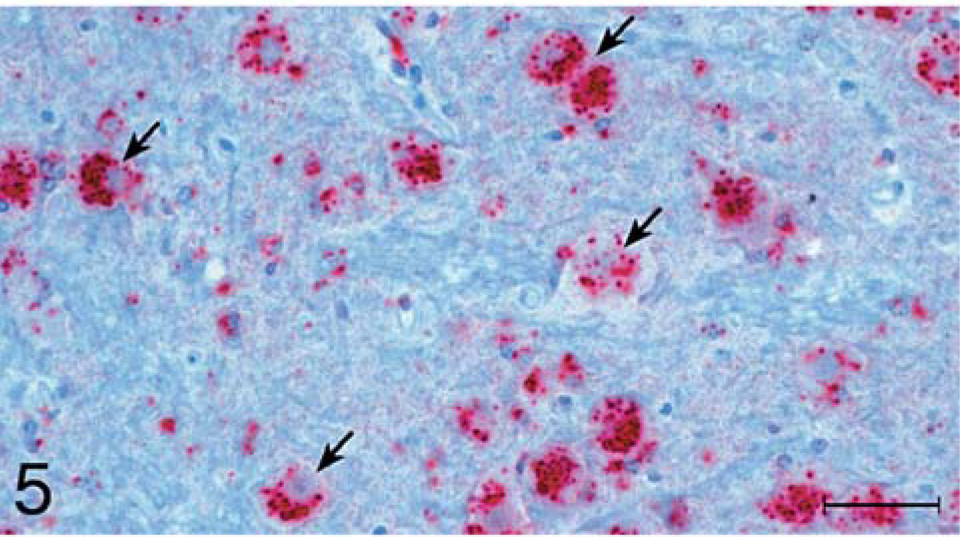
Brain, superior colliculus; raccoon No. 196 inoculated with scrapie. Accumulation of PrPres (stained red) is located predominantly within neuronal cytoplasm (arrows). Stained for PrPres by use of IHC analysis. Bar = 50 µm.
Another differentiation between the staining characteristics for TME and scrapie inoculates was the presence of staining in white matter. All but one (No. 198; Table 1) of the eight scrapie-inoculated raccoons had light, diffuse, punctuate staining in almost all white matter areas of the brain and spinal cord (Fig. 6), whereas staining of white matter was not associated with the five TME inoculates.
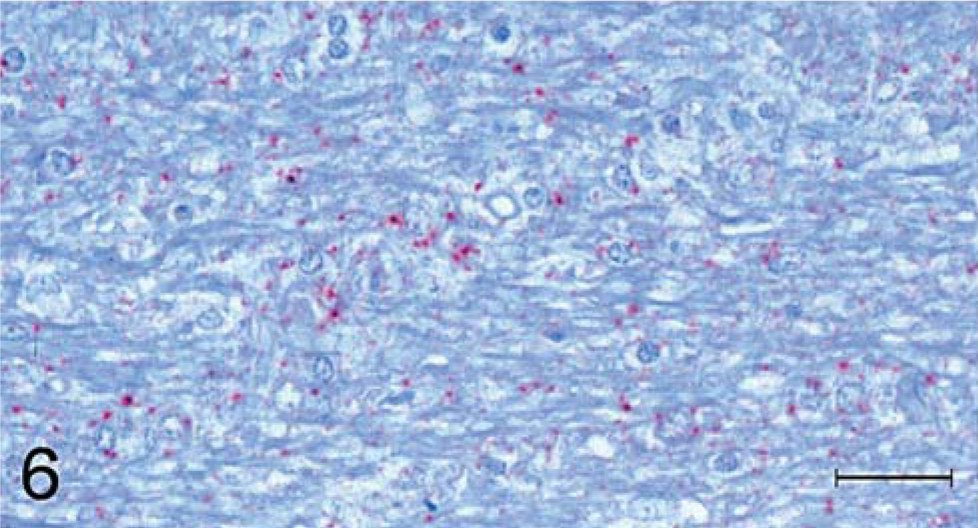
Brain; cerebrum; raccoon No. 196 inoculated with scrapie. Multifocal punctuate staining of PrPres (stained red) is seen in the white matter. Stained for PrPres by use of IHC analysis. Bar = 100 µm.
A third staining difference, which was much more subtle, was the degree of staining observed in the cerebellum. For all scrapie inoculates, there were punctuate and large granules of stain throughout the gray matter of the folia, with a heavier concentration in the granular, compared with the molecular layer (Fig. 7). Purkinje cells did not show positive staining in any of the inoculated raccoons. The staining pattern was similar for TME inoculates, but the amount of staining was generally less, and in two raccoons (Nos. B2, B5; Table 1), the molecular layers did not stain.
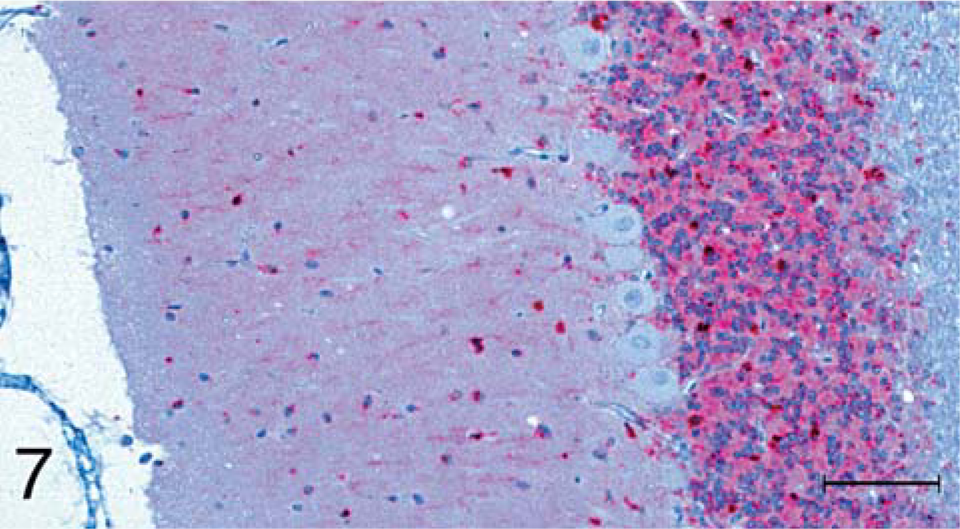
Brain; cerebellum; raccoon No. 196 inoculated with scrapie. PrPres (stained red) is present in both the molecular and granular layers. Notice absence of PrPres stain in Purkinje cells. Stained for PrPres by use of IHC analysis. Bar = 100 µm.
The pituitary gland was available from only five raccoons, but positive staining in the pars nervosa was observed for two of four TME inoculates. Although a large number of tissues were examined by use of IHC analysis, only a small number of tissues (other than brain and spinal cord) had positive staining reaction. The most commonly affected tissues were retina (inner and outer plexiform layers), for all of the TME inoculates and six of eight scrapie inoculates. In addition, the trigeminal nerve ganglion stained positively for one of five TME and four of five scrapie inoculates. The staining in this tissue was restricted to the cytoplasm of some ganglion cells.
PrPres was detected by use of Western blot analysis for the three protease-resistant polypeptide isoforms (di-, mono-, and unglycosylated) in all raccoons with clinical signs of CNS dysfunction (Table 1; Fig. 8A, B), whereas samples from the two control raccoons were negative for PrPres (Fig. 8A). When brain from raccoons infected with scrapie isolate No. 708 was compared with brain from raccoons infected with scrapie isolate No. 706, differences in molecular mass of the respective PrPres isoforms were not detected (Fig. 8A). However, a difference in molecular mass of PrPres was evident when the raccoon scrapie samples were compared with the sheep scrapie control sample. The sheep scrapie sample had higher molecular masses for all three PrPres isoforms, compared with that for the raccoon scrapie brain samples (Fig. 8A). In addition, when brain homogenates from raccoons infected with TME were compared by use of Western blot analysis, differences in molecular mass of the respective PrPres were not detected (Fig. 8B). Interestingly, PrPres isoforms in brain material from a steer infected with TME via intracerebral inoculation had similar molecular masses as that of PrPres isoforms in brain material from TME-infected raccoons.
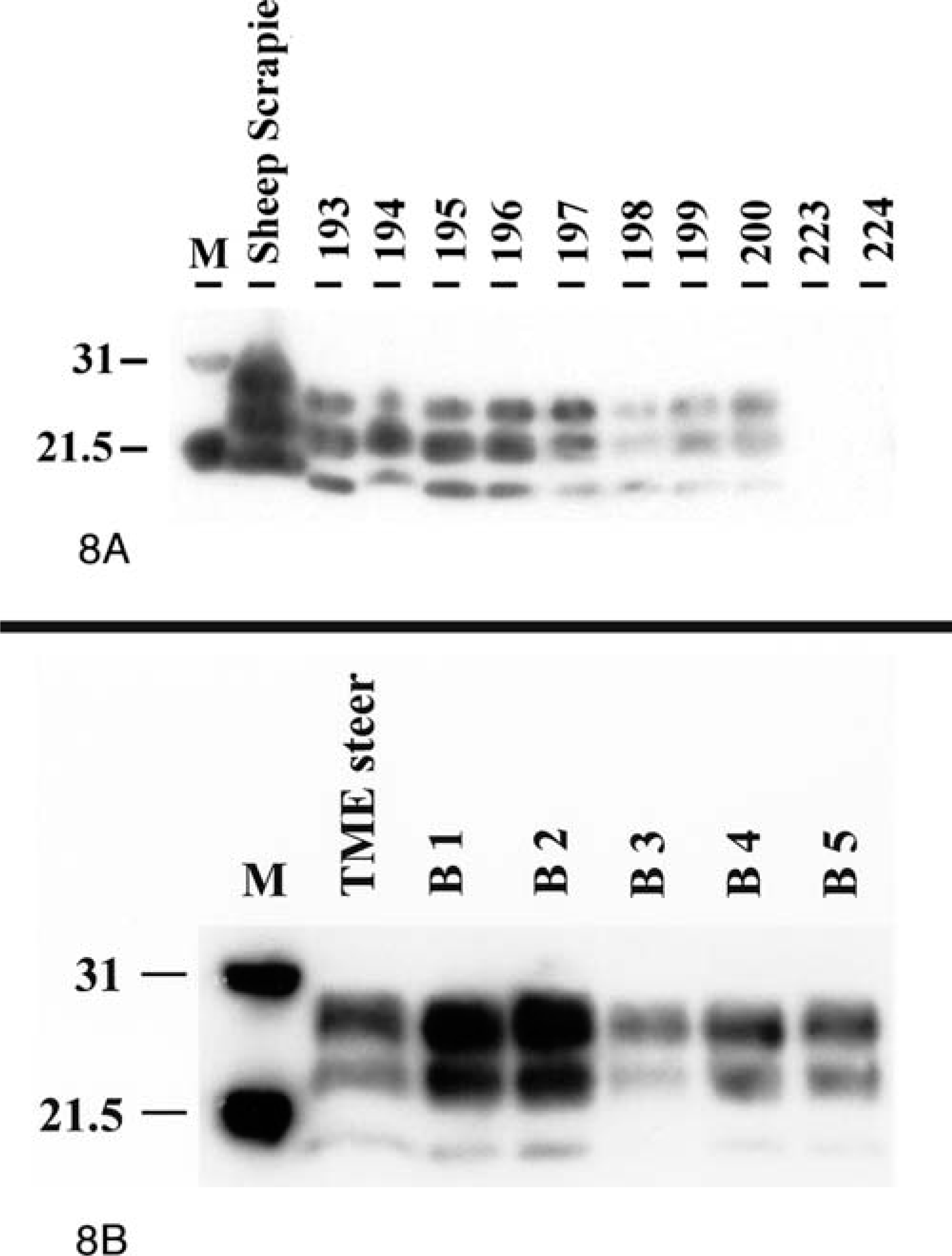
Western blot analysis of raccoon brain homogenates.
Fig 8A.
Analysis of scrapie-infected raccoon brain homogenates. M = protein weight marker (31.0 kd and 21.5 kd); animal Nos. 193–196: raccoons infected with scrapie isolate No. 706; animal Nos. 197–200: raccoons infected with scrapie isolate No. 708; sheep scrapie = brain homogenate from sheep with clinical scrapie.
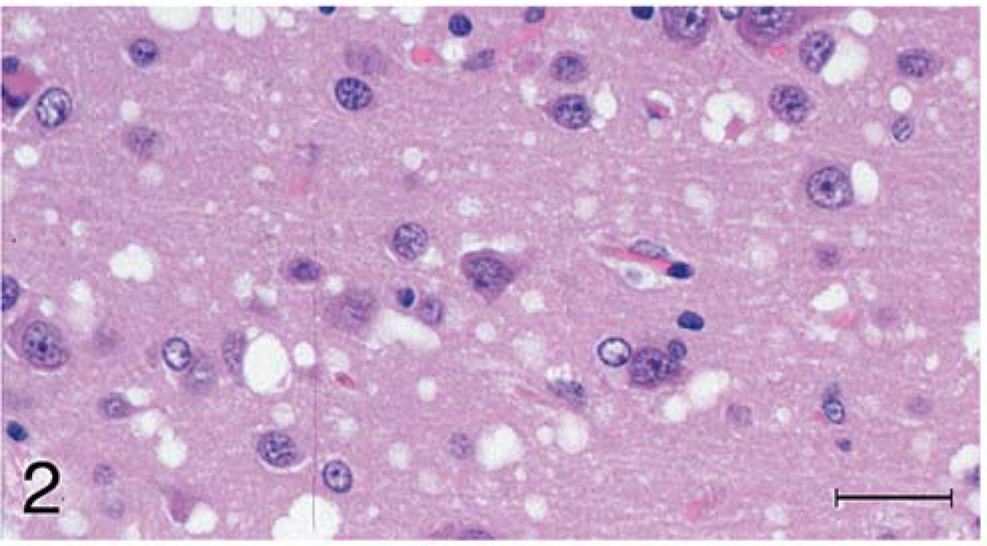
Brain, cerebrum; raccoon No. 199 inoculated with scrapie. Higher magnification of Fig. 1. Spongiform encephalopathy is characterized by variable-sized clear vacuoles in the cortex. Also present is mild gliosis. HE. Bar = 50 µm.
The ORF of the raccoon PrP gene contained two polymorphisms at codons 75 (Gly/Ser) and 107 (Asn/Ser). At codon 75, all animals were homozygous for Gly, except raccoon No. 200 that was heterozygous for Gly and Ser. At codon 107, most animals were homozygous for Ser, except for raccoon Nos. 195 and B2 that were heterozygous for Ser and Asn. Raccoons were previously reported to be homozygous for Asn in at codon 46. 11 All raccoons of this study were found to be homozygous for Ser at that position.
Raccoons are found throughout North America. Since they are omnivores and their diet may include carrion, it is likely that, in the wild, they could be exposed to carcasses of animals with TSEs. 7 Although to the authors' knowledge, naturally acquired TSEs in raccoons have not been described, experimental transmission of TME and sheep scrapie have been documented via intracerebral and oral inoculations. 7, 10, 11
Microscopic spongiform lesions in the CNS tissues were severe and diffuse in all clinically affected raccoons. These changes were most severe in the cerebral cortex where there was severe vacuolation of the neuropil and moderate gliosis without appreciable neuronal vacuolation or degeneration. Although these observations were similar to those described in raccoons from the initial passage experiments (TME and sheep scrapie 10, 11 ), the lesions appeared to be more severe in raccoons of the scrapie-inoculated groups in the study reported here. Also, in the present investigation, the lesions were more severe in the cerebral cortex of scrapie- than in that of TME-inoculated groups (Figs. 1, 2; Table 1). Presence of psammoma bodies in the choroid plexus of four raccoons was considered an incidental finding. 9
The TME- and scrapie-inoculated raccoons succumbed to TSE within PIM 6.5 and 17.9, respectively (Table 1). Although the transmission route used in this and in the initial passages does not mimic a natural route of infection, which in the wild would most likely be via ingestion of infective material, results of this study confirm that raccoons are susceptible to both TME and sheep scrapie. Also, in this study, when the agent was further passaged in the same species, the incubation time was shortened for raccoons of the two scrapie-inoculated groups (Nos. 708, 706; Table 1) from an average of 22.1 months to 12.9 and 15.6 months, respectively. The incubation period commonly is reduced following adaptation of a TSE agent to a new host species. In contrast, the incubation time was not appreciably altered in raccoons of the TME-inoculated group (from an average of 5.4 months 11 to 6.5 months). This difference of incubation times for scrapie and TME in raccoons indicates that raccoons could be used as a laboratory model to differentiate these TSEs (scrapie and TME). As for transmission of CWD of mule deer to raccoons, we have an ongoing study which, as yet (5 years after inoculation), has not documented successful transmission of the disease (first passage).
In addition to the differing incubation periods between TME and scrapie in raccoons, IHC analytic results also provided evidence of a clear biological difference between the two agents in this species. The cell-associated granular and aggregate staining of gray matter, especially in neurons, and the staining of white matter for scrapie inoculates was in marked contrast to the diffuse, punctuate neuropil staining and absence of white matter staining that characterized CNS tissues for the TME inoculates.
Results of Western blot analysis indicated that all raccoons inoculated with either scrapie or TME isolates were positive for PrPres, having a clear reaction with the three protease-resistant polypeptide isoforms (di-, mono-, and unglycosylated). Interestingly, the molecular mass of all three PrPres isoforms of raccoon-passaged scrapie was lower than that of the PrPres isoforms of the sheep-passaged scrapie. It has been documented that PrPres in brain stem homogenates from cattle infected with BSE had reproducible lower molecular mass for each of the PrPres isoforms than did those from sheep infected with scrapie. 2 When using a hybrid Western blot analysis and two different PrP-specific antibodies, cattle BSE could be distinguished from sheep scrapie. 16 Since raccoon-passaged scrapie also has a lower molecular mass for each of the PrPres isoforms, compared with that for sheep scrapie, it would be of interest to see whether raccoon-passaged scrapie has a BSE-like reaction pattern in the hybrid Western blot analysis. In this context, it should be noted that the raccoon-passaged TME isolates had molecular mass similar to that of cattle-passaged TME.
In this study, two polymorphic sites in the raccoon PRNP gene were identified. At least one other polymorphic site exists for codon 46. On the basis of a previous study reporting homozygosity for Ser, 11 and the present study reporting homozygosity for Asn at codon 46, raccoons have an Asn/Ser polymorphism at this site. Heterozygosity at positions 75 and 107 in the raccoon PRNP gene does not appear to have influence on survival time (Table 1), because all three heterozygous animals had survival time similar to that of their group mates (Table 1).
The classic TSE strain-typing procedure in mice includes evaluation of histologic lesion profiles (spongiform change) in the brain, but IHC starting pattern has not been published to our knowledge. In sheep, however, variations of IHC staining patterns have been described and the differences were suggested to be evidence for the existence of multiple scrapie strains in the population examined. 8 Such conclusion might be considered tenuous, when only material from individual field cases is available. In the present study, however, experimental serial passage of the TME and scrapie isolates in raccoons permitted clear resolution of different IHC staining patterns associated with these agents. This finding supports evidence from the incubation period data that indicates the raccoon model may be a useful biological assay for differentiating TME and scrapie, especially when these agents are suspected of crossing the species barrier. Of particular interest will be examination of tissues from cattle that have been experimentally inoculated with scrapie or TME, 5, 6, 12, 14 as well as tissues from cattle with BSE.
If the raccoon model can be developed successfully, it could prove to be a faster method for differentiation of the known TSEs in the USA. This would be particularly useful if an unknown TSE, such as that found in 2000 in milking sheep imported from Belgium to Vermont, were identified. 1 Further studies, however, are necessary before the raccoon can be recommended as a biological model for TSE strain differentiation. Studies such as experimental intracerebral inoculation of BSE should be evaluated in raccoons. Since the BSE agent appears to be more promiscuous than other TSE agents (i.e., easily transmitted to other species such as sheep, wild ungulates, cats, and primates), it may have a considerably shorter incubation period than does scrapie or TME in raccoons. Also, experimental inoculation, using individual scrapie isolates (and CWD isolates from other sources such as elk and white-tailed deer), would be useful. Data from these types of studies will also contribute to our knowledge of the behavior of various strains of TSEs in raccoons.
Footnotes
Acknowledgements
We thank Drs. L. Nusz and J. Laufer for clinical assistance and Dr. Marcus Kehrli Jr. for constructive comments and his enthusiastic support. Expert technical assistance was provided by Martha Church, Dennis Orcutt, Semakaleng Lebepe-Mazur, Jean Donald, and Sharla Van Roekel.
This study was carried out under the guidelines of the institutional ACUC committee at the National Animal Disease Center. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
