Abstract
Transmissible spongiform encephalopathies (TSEs) are fatal neurologic diseases. Infection by the causative agent, a prion, induces accumulations of an abnormal form of prion protein (PrPres) in tissues of nervous and lymphoid systems. Presence of characteristic histopathologic changes (spongiform encephalopathy) and detection of protease-resistant PrPres in neural and lymphoid tissues are the basis of currently available methods for diagnosis of TSEs. In this study, samples of striated muscle tissues (tongue, heart, diaphragm, and masseter muscle) from 20 animals (cattle, sheep, elk, and raccoons) were examined for PrPres by immunohistochemistry (IHC). All the animals had developed a TSE after experimental inoculation. PrPres was found by IHC in the brain but not in the muscle tissues of all the animals examined. These findings are contradictory to recently published reports of laboratory animals with TSEs, where these altered prion proteins were detected in tongue and other striated muscles. Further testing of muscle tissues is needed to confirm the findings of the present study.
Keywords
Transmissible spongiform encephalopathies (TSEs) are fatal neurologic diseases of animals, including man. Animal TSEs include scrapie of sheep and goats, chronic wasting disease (CWD) of cervids, transmissible mink encephalopathy (TME), and bovine spongiform encephalopathy (BSE). 14 As a result of consumption of infected bovine tissues, BSE-derived spongiform encephalopathies (SE) have been seen in humans, captive exotic ungulates, cats, numerous species of large, captive felines, 14 and nonhuman primates. 2,3 Infection by the causative agent, a prion, induces accumulations of an abnormal form of prion protein (PrPres) in tissues of nervous and lymphoid systems. Currently, diagnosis of a TSE relies on characteristic histopathologic changes and the use of specific antibodies to PrPres in immunohistochemical (IHC) and western blot (WB) techniques. 9 Three recent publications have indicated that PrPres is present in muscle tissues of TSE-infected laboratory animals. 1,4,13 The first study detected PrPres with WB in skeletal muscles of mice that had been inoculated with a laboratory-adapted strain of scrapie. 4 The second study demonstrated PrPres in the tongue of Syrian hamsters, using WB and IHC after inoculation with the hamster-adapted TME agent. 1 In this study, PrPres was also found within nerve fibers by both methods. 1 Syrian hamsters fed brain tissue from scrapie-infected hamsters were also used in the most recent study, and the tissues were examined by WB for the presence of PrPres. 13
The objective of this study was an attempt to detect PrPres by IHC in the tongues of cattle (Bos tarus), sheep (Ovis aries), elk (Cervus elaphus nelsoni), and raccoons (Procyon lotor) that had developed TSE as a result of experimental inoculations with scrapie, CWD, or TME. These experiments were conducted at the National Animal Disease Center (NADC) in Ames, Iowa. Details of some of these studies have been published previously. 5–8
A total of 20 TSE-inoculated animals from seven different experiments were available for this part of the study. Numbers and types of animals and TSE agents used in individual experiments are summarized in Table 1. All inoculated animals were housed in a biosafety level 2 containment facility at the NADC. Personnel wore protective clothing while in the isolation facility and showered when leaving the facility.
Immunohistochemical examination of striated muscles (heart, tongue, masseter, and diaphragm) of cattle, sheep, elk, and raccoons experimentally inoculated with agents of transmissible spongiform encephalopathy.
∗ Positive for TSEs.
† TSE = transmissible spongiform encephalopathy; CWD = chronic wasting disease; TME = transmissible mink encephalopathy; IC = intracerebral; UP = Hamir et al., unpublished.
Scrapie inoculum was from a pool of 13 scrapie-positive (by IHC) sheep brains from seven source flocks (inoculum No. 13-7), 7,8 CWD inoculum was prepared from a pool of 28 CWD-affected mule deer brains, 5 and the TME inoculum was from the Stetsonville, Wisconsin, outbreak of TME in 1985, which had been biologically cloned in mink three times by limiting endpoint titration. 6 All three inocula were positive for PrPres by either WB or IHC (or both). 5–8 The material for the inocula was ground in a mechanical grinder, 100 µg/ml of gentamicin was added, and the final concentration of 10% (wt/vol) was made with phosphate-buffered saline.
Most experimental animals were inoculated intracerebrally (experiment Nos. 1–6, Table 1) with 1 ml (sheep, cattle, and elk) or 0.1 ml (raccoons) of scrapie, CWD, or TME inoculum. Five sheep were inoculated orally (experiment No. 7, Table 1) with two doses (15 ml, 1 week apart) of 10% scrapie brain suspension.
TSE-inoculated animals were euthanatized (occasionally, animals died before euthanasia), and a complete necropsy was conducted on each of the carcasses. Representative samples of all major organs were immersion fixed in 10% neutral buffered formalin for histopathology. Sections were stained by hematoxylin and eosin and by an IHC method 10 for detection of PrPres. After deparaffinization and rehydration, tissue sections were autoclaved for 30 minutes in an antigen retrieval solution (DAKO™ Target Retrieval Solution, DAKO Corp., Carpinteria, CA) and stained with an indirect avidin–biotin system (Basic Alkaline Phosphatase Red Detection Kit, Ventana Medical Systems, Inc., Tucson, AZ) designed for an automated immunostainer (NexES IHC module, Ventana Medical Systems). The primary antibody was a cocktail of two monoclonal antibodies, F89/160.1.5 11 and F99/97.6.1, 12 each used at a concentration of 5 µg/ml, and incubation was carried out at 37 C for 32 minutes. The secondary antibody was biotinylated anti-mouse (biotinylated anti-mouse IgG [made in horse], Vector Laboratories, Burlingame, CA), diluted 1 : 200 and incubated for 8 minutes at 37 C.
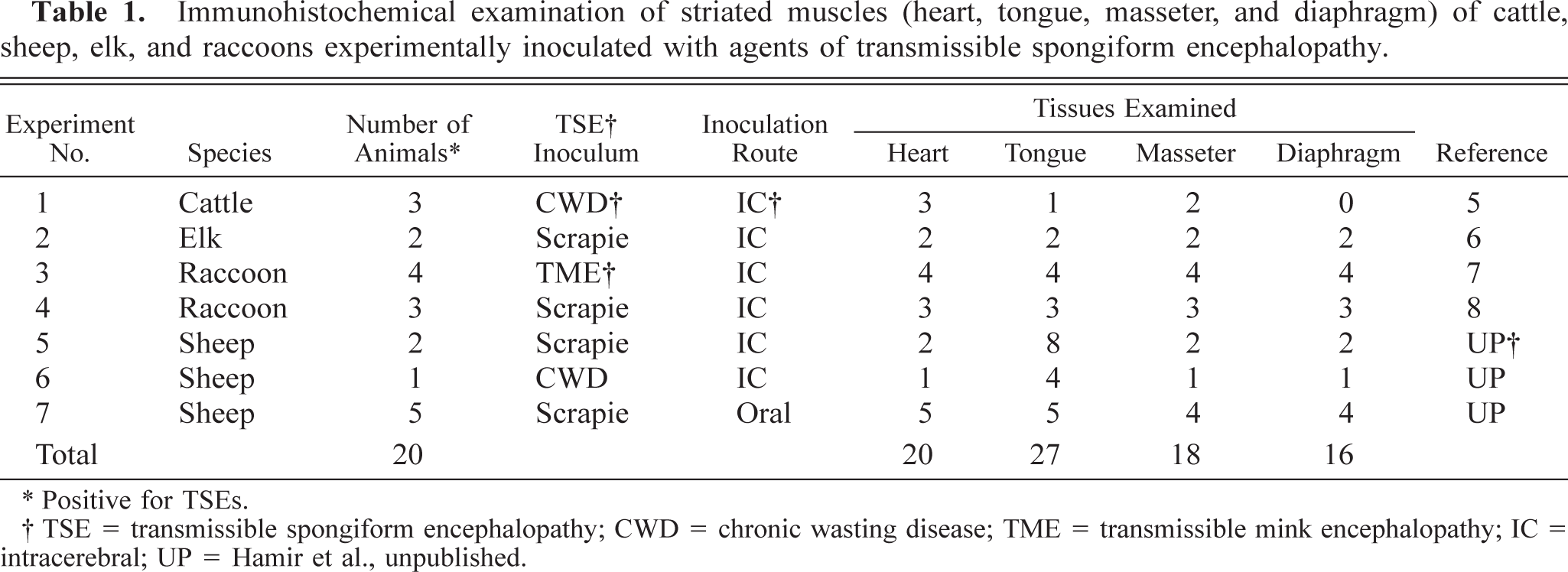
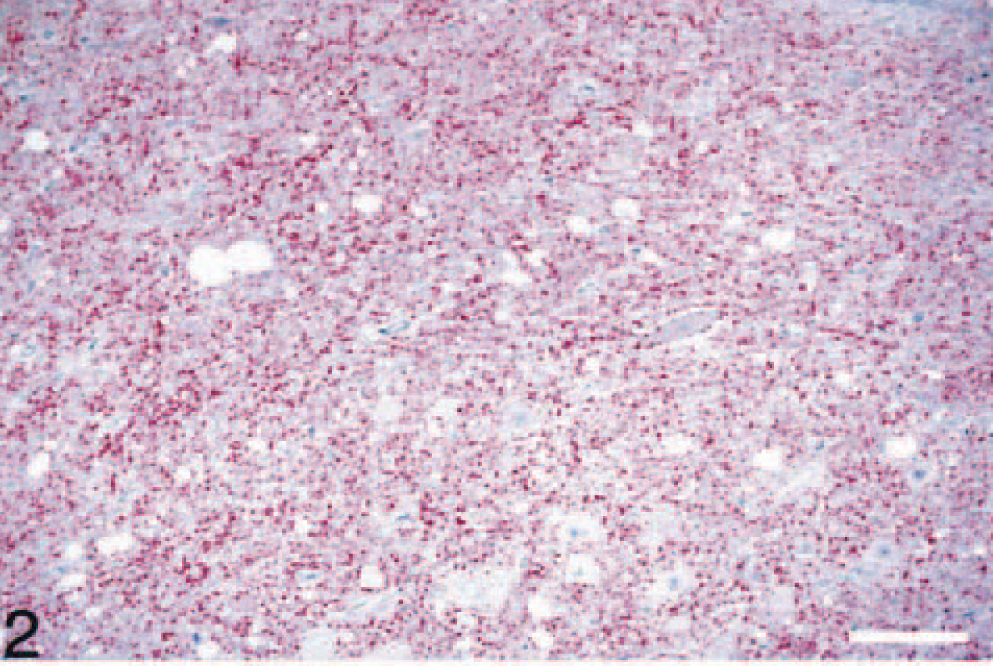
With the exception of three cattle inoculated with CWD (experiment No. 1, Table 1), all others inoculated with TSE agents developed severe neurologic signs and had extensive lesions of spongiform encephalopathy (Fig. 1). IHC (Fig. 2) and WB revealed the presence of PrPres in all the brains. For the purpose of this investigation, only results of striated muscles (tongue, heart, diaphragm, and masseter muscle) are given. In addition, tongues of sheep with experimental clinical scrapie (n = 2) and CWD (n = 1) with brain and lymph nodes positive for PrPres by WB and IHC were examined in more detail by histopathology and IHC (Table 1). For each of these cases, besides the heart, diaphragm, and masseter muscle, four anatomic sites of the tongue (two near the tip and one each from the middle dorsum and the root) were fixed in 10% formalin and examined using light microscopy and IHC.
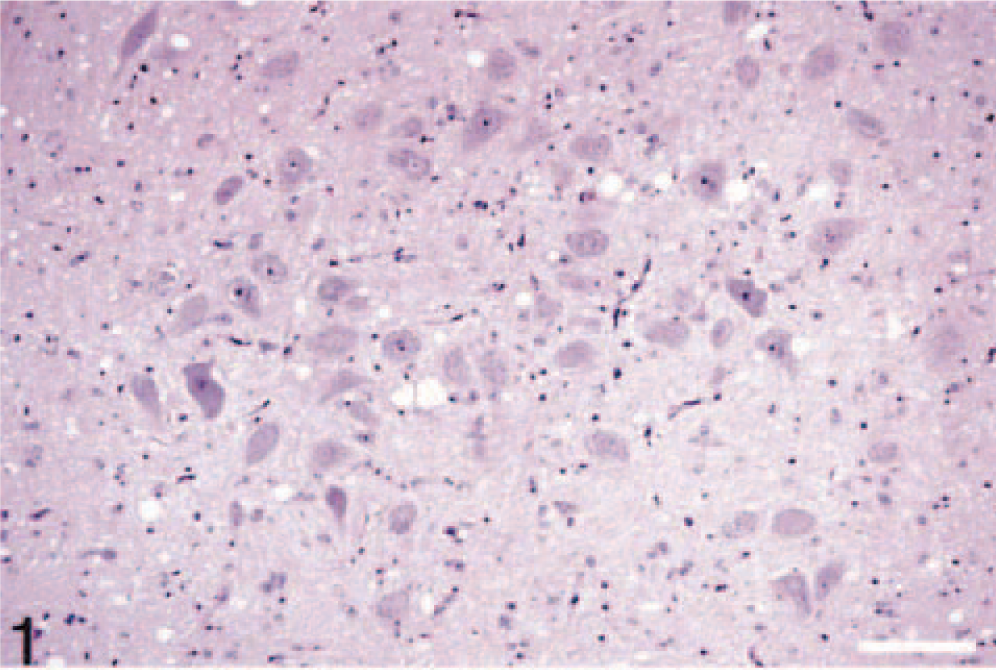
Tissues of sheep with clinical scrapie experimentally inoculated by oral route.
Brain, medulla oblongata, obex (dorsal vagal nucleus). There is extensive accumulation of PrPres (stained red) predominantly in the neuropil. Stained for PrPres by IHC. Bar = 70 µm.
Although striated muscle tissues were not available from all four organs for each animal, a total of 81 sections (16 diaphragms, 18 masseters, 20 hearts, and 27 tongues) were examined from 20 animals by using light microscopy and IHC (Table 1). In 78 of 81 sections of striated muscles, one or more cross-sections of nerve fibers were seen. However, in the tongue, numerous nerve fibers were observed, and in seven sections of this organ, there were variable numbers of ganglion cells (Fig. 3). Except for a few Sarcocystis spp. in tissue sections (n = 10), morphologic lesions were not seen in any of the HE-stained sections, and none of the IHC-stained sections revealed the presence of PrPres (Fig. 2).
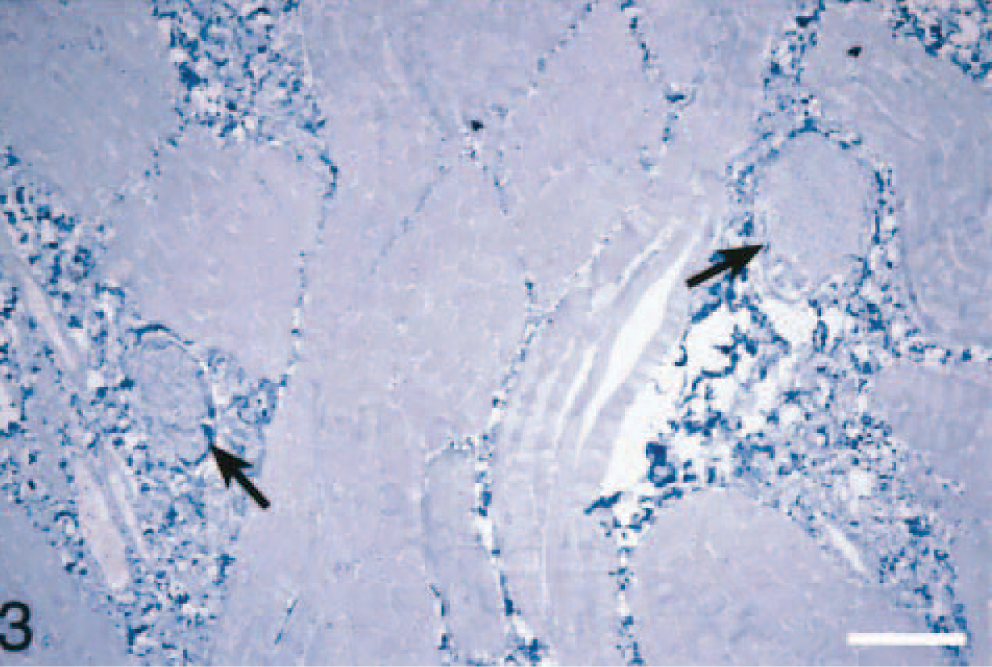
Tongue. Sections of peripheral nerves (arrows) are present among striated muscle fibers. No PrPres is present in any of the tissues. Stained for PrPres by IHC. Bar = 100 µm.
The presence of PrPres in the brains of all TSE-inoculated animals, but not in any of the striated muscle tissues, indicates that these tissues did not accumulate detectable PrPres. These results are contradictory to recent reports of TSE in laboratory animal models, where researchers found PrPres in skeletal muscles and the tongue. 1,4,13 However, in the most recent study, 13 the authors stated that their results may not mirror the findings in natural cases of TSEs because 1) the levels of infectivity were higher in their animal model than in naturally occurring TSEs, 2) heart tissue from variant Creutzfeldt-Jakob disease cases lacks detectable PrPres, and 3) infectivity was not detected in skeletal muscles and sciatic nerves from cattle with BSE by bioassays in mice and by titration in bovines.
Also, because all three studies that detected PrPres in skeletal muscles and tongue 1,4,13 had used laboratory-adapted TSE strains, it could be speculated that these strains of TSEs may have acquired the capacity to amplify in striated muscle tissues of laboratory animal hosts. On the other hand, it may be argued that the IHC test used in the present study to detect PrPres in tissue sections is not as sensitive as other tests such as the WB. However, in our experience, we have found the IHC test to have equal, if not slightly superior, sensitivity for detection of PrPres. 9
Further testing of striated muscle tissues from larger numbers of experimental and natural TSE cases, both clinical and subclinical, is needed to verify our finding that PrPres cannot be detected in muscles of affected animals. This is important not only from a scientific point of view but also from a public health perspective.
Footnotes
Acknowledgements
We thank Dr. Katherine O'Rourke, USDA Agricultural Research Service, Animal Diseases Research Unit, Pullman, Washington, for providing antibodies for IHC and Martha Church, Dennis Orcutt, Jean Donald, and Jennifer Slovak for expert technical assistance.
Experiments cited in this communication were carried out under the guidelines of the institutional ACUC committee at NADC. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
