Abstract
Sheep scrapie susceptibility or resistance is a function of genotype, with polymorphisms at codon 171 in the sheep prion gene playing a major role. Glutamine (Q) at codon 171 contributes to scrapie susceptibility, while arginine (R) is associated with resistance. In some breeds, lysine (K) occurs at codon 171, but its effect on scrapie resistance has not been determined. Charge and structural similarities between K and R suggest that they may contribute to prion disease susceptibility in a similar way, but studies have not been performed to confirm this. The purpose of the current study was to compare susceptibility and incubation times of AA136RR154QQ171 (where the letter denotes the amino acid and the number the position) with AA136RR154QK171 sheep after inoculation with scrapie. Barbado AA136RR154QQ171 and AA136RR154QK171 sheep were inoculated with scrapie intracerebrally to assess their susceptibility to scrapie. After inoculation, sheep were observed daily for clinical signs and were euthanized and necropsied after clinical signs were unequivocal. Tissues were collected at necropsy for immunohistochemistry and Western blot analyses. The QQ171 sheep had clinical signs approximately 12 months after inoculation, whereas QK171 animals had an average incubation time of 30 months to onset of clinical signs. The distribution of abnormal prion protein was similar in QQ171 and QK171 sheep. Results of the study indicate that sheep with a single K allele at codon 171 are susceptible to scrapie but with a prolonged incubation time. Work is currently underway to examine relative scrapie susceptibility or resistance of KK171 sheep.
Scrapie is a fatal neurodegenerative disease that affects sheep and goats and belongs to the family of transmissible spongiform encephalopathies, which includes bovine spongiform encephalopathy in cattle, chronic wasting disease (CWD) in deer and elk, and Creutzfeldt Jakob disease in human beings. Transmissible spongiform encephalopathies are caused by the accumulation of an infectious, misfolded, protease-resistant isoform of the prion protein (PrPSc). 23 PrPSc serves as a template to convert the normal protease-sensitive form of the prion protein (PrPC), a cell surface glycoprotein expressed in nervous 3 and other tissues, 13 to the abnormal form. PrPSc accumulates primarily in the nervous and lymphoreticular tissues in sheep with scrapie.10,25
Amino acid polymorphisms in the prion protein play a major role in transmissible spongiform encephalopathy susceptibility in sheep, 27 mice, 28 and human beings. 24 The codons of the prion protein gene (prnp) that are especially significant in determining susceptibility or resistance to scrapie include 136, 154, and 171. 5 Codon 171 appears to have the most discernible influence, where glutamine (Q) confers susceptibility while arginine (R) confers resistance.6,20
Another amino acid, lysine (K), occurs at codon 171 in some breeds of sheep, such as the Barbado Blackbelly, Dorper, and Suffolk crosses, 8 Italian Biellese, 2 Mongolian Khalkh, 7 Greek milking breed crosses, 4 Chinese Bayanbulak, 17 and Italian Bergamasca and Appenninica, 22 but the effect of K171 on susceptibility to scrapie has not been described. Similarities in charge and structure suggest that K may behave similar to R. Natural scrapie has been described in one K171-containing sheep, 1 but not enough information is available from natural cases to determine the effect of K171 on scrapie susceptibility. The purpose of the current study was to compare scrapie susceptibility of sheep with regard to K171 after experimental intracerebral inoculation.
Four Barbado lambs were obtained from a flock at the National Animal Disease Center (NADC; Ames, Iowa) that was continually monitored and remained negative for scrapie. Two lambs each that were either AA136RR154QQ171 or AA136RR154QK171 were used for the current study. The lambs were inoculated intracerebrally with scrapie at approximately 2 months of age. Animals were monitored daily for the duration of the experiment and euthanized when clinical signs of scrapie, such as ataxia, loss of balance, and recumbency, became severe. All animal experiments were conducted under the approval of the NADC Institutional Animal Care and Use Committee.
The inoculum (NADC 13-7) 10 was a 10% (w/v) scrapie-positive sheep brain homogenate in phosphate buffered saline with gentamicin at 100 µg/ml that had been passaged twice through susceptible sheep of the AA136RR154QQ171 genotype. 11 The procedure for intracerebral inoculation has been described. 10 Briefly, the animals were sedated with xylazine; the frontal area was clipped and scrubbed; a 1-cm midline incision was made in the skin slightly caudal to the junction of the parietal and frontal bones; and a 1-mm hole was drilled through the calvarium. A 22-guage spinal needle was advanced through the hole perpendicular to the frontal bones until the tip of the needle made contact with the opposite (bottom) side of the calvarium. The inoculum was slowly injected as the needle was withdrawn through the brain. The skin was closed with a single suture.
At necropsy, two sets of tissue samples were collected including representative sections of liver, kidney, spleen, skin, striated muscles (heart, tongue, diaphragm, masseter), thoracic aorta, thyroid gland, turbinates, trachea, lung, tonsils, esophagus, rumen, reticulum, omasum, abomasum, intestines (ileum), adrenal gland, urinary bladder, lymph nodes (retropharyngeal, prescapular, mesenteric, popliteal), nerves (sciatic, optic, trigeminal), pituitary gland, trigeminal ganglion, brain (cerebral cortex, cerebellum, midbrain including superior colliculus, brainstem including obex), spinal cord (cervical, thoracic, lumbar), and eye. The first set was collected into 10% buffered formalin (globes were fixed in Bouin fixative), embedded in paraffin wax, and sectioned at 5 µm for staining with hematoxylin and eosin (HE) and anti–prion protein antibodies. The second set of tissues was frozen.
All paraffin-embedded tissues were stained by an automated immunohistochemical method for detection of PrPSc as described previously 10 with a cocktail of 2 primary antibodies, F89/160.1.5 18 and F99/97.6.1, 26 each at 5 µg/ml. Frozen tissues were utilized for immunodetection of PrPSc by a Western blot technique described herein. Brain tissue from the brainstem at the level of the obex was homogenized at a final concentration of 10% (w/v) in 10 mM Tris and 5 mM MgCl2 (pH 7.5) using a tissue homogenizer a with disposable probe. Second, ultrasonic dismembranation b was performed for four 30-sec intervals in an ice bath with brief vortex mixing between sonication. Proteinase K c was added to a final concentration of 80 µg/ml; digestion was conducted at 37°C for 1 hr and stopped by the addition of Pefabloc d to a final concentration of 0.1 mg/ml. Samples were loaded on a 4–20% commercially prepared sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) gel at approximately 1 mg tissues equivalent in SDS-PAGE sample buffer and run according to the manufacturer’s instructions. The sample was then blotted to a polyvinylidene difluoride membrane e and blocked with 3% bovine serum albumin. Western blot detection was conducted using mouse anti-PrP monoclonal antibody P4, f which targets amino acids 89–104 of the ovine prion protein sequence, 12 at a 1:10,000 dilution (0.1 µg/ml) as the primary antibody. A biotinylated sheep anti-mouse secondary antibody g at 0.05 µg/ml and a streptavidin–horseradish peroxidase (HRP) conjugate h were used according to the manufacturer’s instructions in conjunction with a chemiluminescent detection system i that recognizes immobilized specific antigens conjugated to HRP-labeled antibodies and an imaging system capable of detecting luminescence. j Primary antibody incubations were conducted with the membrane at either room temperature for 1 hr or 4°C overnight (≥12 hr). Secondary antibody and streptavidin–HRP conjugate incubations were conducted at room temperature for 1 hr.
To determine whether recipient genotype has a treatment effect on the life span of sheep incubated with the NADC 13-7 scrapie isolate, previously published data 11 were used to allow for statistical analysis. Incubation times from 16 QQ171 sheep were compared to the QK171 sheep in the current study by means of a permutation test using the exact rank tests. k
Clinical signs of scrapie occurred in 3 sheep. One lamb (sheep 1) was found dead 4 days postinoculation (PI) due to complications associated with intracranial inoculation. The 3 remaining sheep developed clinical disease and were euthanized once signs became severe. Clinical signs included lethargy, weakness, loss of balance, ataxia, and lateral recumbency with pedaling of the legs. Sheep 2 (QQ171) was euthanized 12 months PI. Sheep 3 and 4 (QK171) were euthanized at 30 months PI (Table 1). A permutation test of the null hypothesis (no treatment effect) comparing incubation time of QQ171 and QK171 sheep inoculated with the isolate of scrapie (NADC 13-7) resulted in a P value of 0.0083333.
Findings in QQ171 and QK171 Barbado sheep inoculated with the scrapie agent from sheep.*
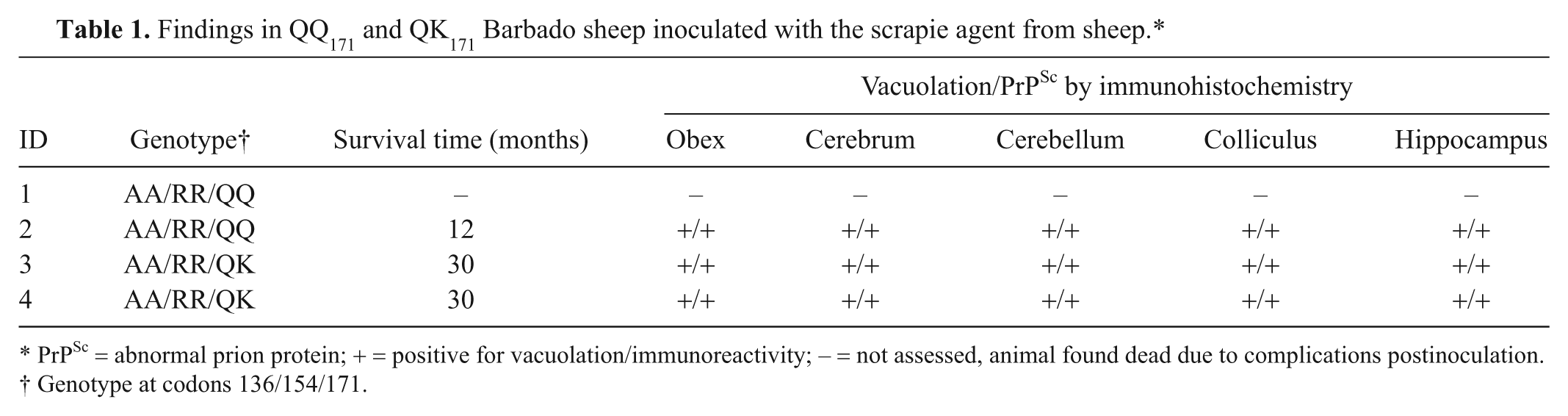
PrPSc = abnormal prion protein; + = positive for vacuolation/immunoreactivity; – = not assessed, animal found dead due to complications postinoculation.
Genotype at codons 136/154/171.
Microscopic examination revealed lesions in the brain consistent with scrapie in all animals that progressed to clinical disease. Spongiform change was observed in the rostral cerebrum, hippocampus, colliculus, cerebellum, and brainstem at the level of the obex (Fig. 1A, 1B). Spongiform change was most severe in obex, cerebellum, and colliculus and was similar in intensity regardless of genotype. Immunoreactivity for PrPSc was demonstrated by immunohistochemistry (IHC) in cerebrum, hippocampus, colliculus, cerebellum, obex (Fig. 1C, 1D), and trigeminal ganglia of each sheep. Immunoreactivity occurred as granular staining within the neuropil, in addition to intraneuronal, perineuronal, and linear patterns, and was strongest in the obex and colliculus. Lymphoreticular tissues that were immunoreactive for PrPSc by IHC included mesenteric lymph node, gut-associated lymphoid tissue associated with the ileum (Fig. 1E) and recto-anal junction, spleen, palatine tonsil, pharyngeal tonsil, and retropharyngeal lymph node (Fig. 1F). Immunoblots probed with monoclonal antibody P4 had a similar migration pattern regardless of genotype and demonstrated the characteristic 3-band pattern representing the di-, mono-, and unglycosylated isoforms of PrPSc (Fig. 2).
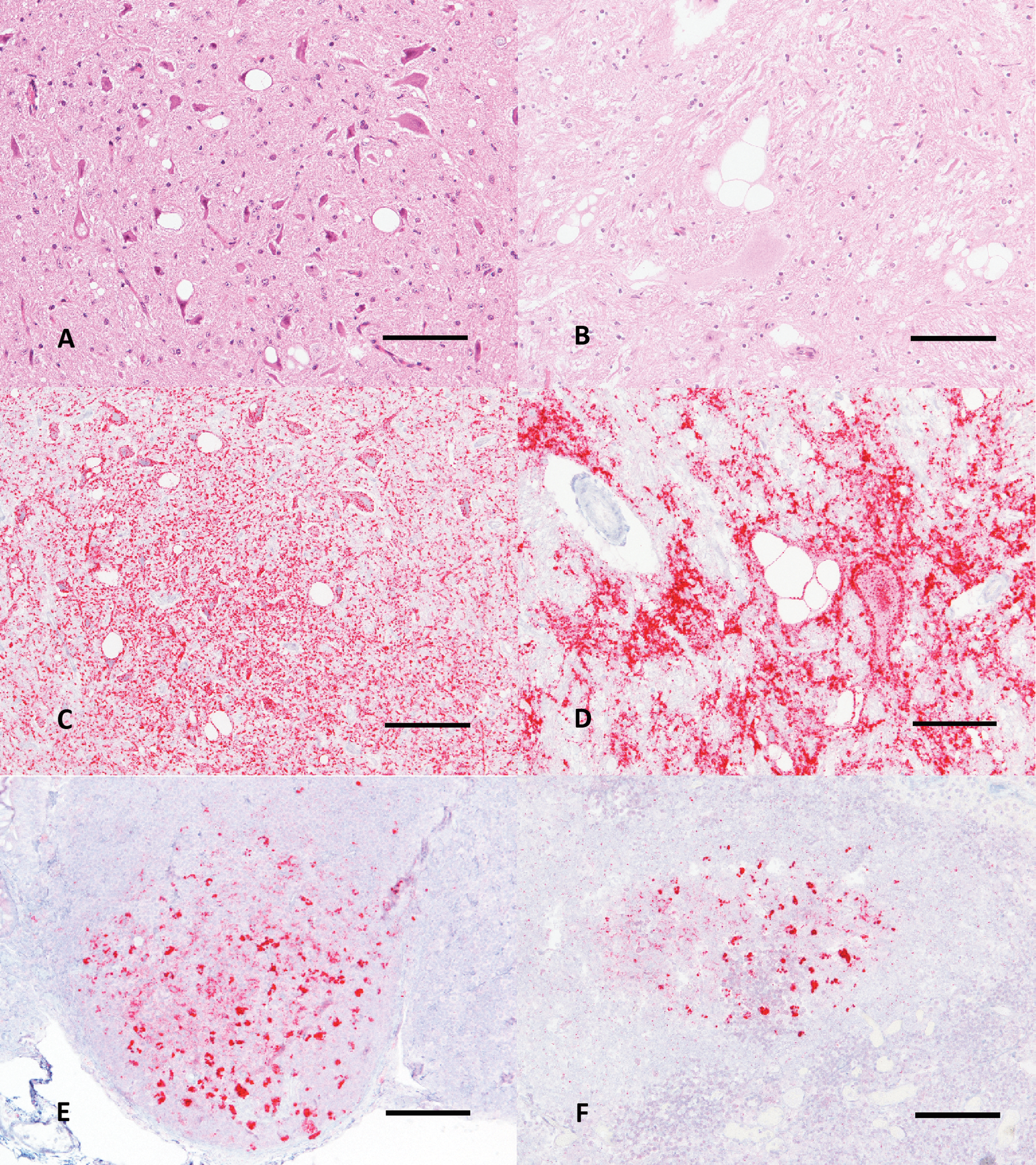
Microscopic features of the brain and lymphoid tissues of QQ171 (
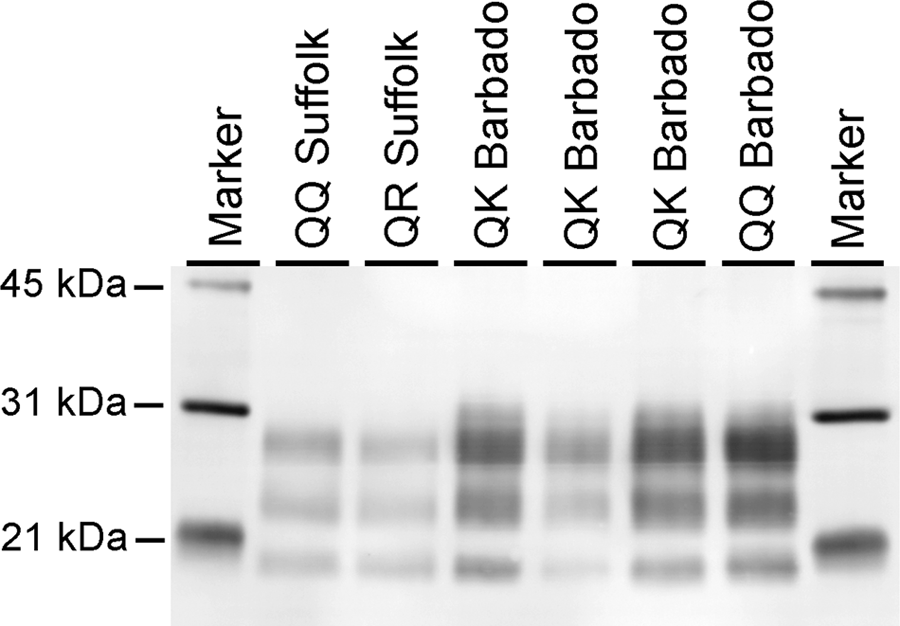
Western blot of QQ171and QK171 Barbado sheep in comparison to QQ171 and QR171 Suffolk sheep with monoclonal antibody P4. Regardless of genotype or breed, tissues from all sheep have similar apparent migrations patterns. The QQ Suffolk sample pictured is from the original NADC 13-7 inoculum.
The present study assessed the impact of heterozygosity for K at codon 171 of prnp on scrapie susceptibility. It was expected that K may behave similar to R due to congruency in charge and structure. While a single K allele did not confer resistance to scrapie, it did significantly increase the incubation period to the onset of clinical signs (P = 0.0083333). Sheep with the QQ171 genotype survived for 12 months PI, which was similar to Suffolk sheep of the same genotype.10,11 Sheep with the QK171 genotype survived for 30 months. This result was dissimilar from sheep with the QR171 genotype in that QR171 is associated with a much greater prolongation of incubation time to 60 months (Kunkle RA, Hamir AN, Richt J, et al.: 2009, Lack of prion accumulation in lymphoid tissues of scrapie-affected sheep with the AA136, QR171 prion protein genotype [abstract]. Proceedings of the American Association of Veterinary Laboratory Diagnosticians 52nd Annual Conference. Page 186). Despite significant differences in incubation time between sheep with either the QQ171 of QK171 genotype, there were no apparent differences between groups with respect to vacuolation, distribution of PrPSc by IHC, or migration patterns of PrPSc by Western blot.
Examples of amino acid polymorphisms in the prion protein that affect incubation times exist in other transmissible spongiform encephalopathies, most notably CWD. Polymorphisms at codon 132 of prnp are associated with CWD susceptibility and differences in incubation time in Rocky Mountain elk (Cervus elaphus). Elk that are of the MM132 genotype are predisposed to CWD following a natural challenge. 19 In experimental studies, elk that are of the ML132 genotype have a nearly doubled incubation time, 9 and elk that are LL132 have a tripled incubation time relative to those that are MM132. 21 In mule deer (Odocoileus hemionus) that are of the SS225 genotype, the incubation times are shorter (16 months PI) than those in deer that are SF225 (>25 months PI). 14 Finally, polymorphisms at codons 95 and 96 of prnp affect CWD susceptibility 15 and incubation in white-tailed deer (Odocoileus virginianus). White-tailed deer having the wild-type QQ95GG96 genotype have a much shorter incubation time (23 months PI) than those that had the QH95GS96 genotype (52 months PI). 16
More research must be conducted to fully understand what affect lysine at position 171 of the prion protein has on scrapie susceptibility and progression in sheep with that genotype. Such work will include determining susceptibility and incubation times of QK171 sheep after a more natural route of inoculation and assessing the scrapie susceptibility in sheep with the KK171 genotype. Unfortunately, R at position 171 has not been detected in the Barbado breed, so it is not possible to determine what affect R and K interactions at position 171 would have on scrapie susceptibility. It may be possible, however, to study these interactions in other breeds.
Footnotes
Acknowledgements
The authors thank Martha Church, Kevin Hassall, Joe Lesan, Leisa Mandell, and Dennis Orcutt for expert technical assistance, and Gary Hansen for providing excellent animal care. Disclaimer: Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity employer.
a.
PowerGen Model 125 Homogenizer, Fisher Scientific, Waltham, MA.
b.
Model 500 Ultrasonic Dismembrator, Fisher Scientific, Waltham, MA.
c.
Proteinase K (from Tritirachium album), USB Corp., Cleveland, OH.
d.
Pefabloc SC, Roche Diagnostics GmbH, Mannheim, Germany.
e.
Amersham Hybond-P PVDF membrane, GE Healthcare Technologies, Piscataway, NJ.
f.
Rida® mAb P4, R-Biopharm AG, Darmstadt, Germany.
g.
Mouse Ig (whole Ab), biotinylated, GE Healthcare Technologies, Piscataway, NJ.
h.
Streptavidin horseradish peroxidase conjugate, GE Healthcare Technologies, Piscataway, NJ.
i.
Amersham ECL Plus™ Western Blotting System, GE Healthcare Technologies, Piscataway, NJ.
j.
Kodak Image Station In-Vivo F, Eastman Kodak Co., New Haven, CT.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in its entirety by the U.S. Department of Agriculture, Agriculture Research Service.
