Abstract
Muscle samples were obtained at necropsy from 225 horses and ponies 1 year of age or older. Samples were processed in routine manner and were stained with hematoxylin and eosin and with periodic acid-Schiff for glycogen. Sections were examined for abnormal glycogen content and amylase- resistant complex polysaccharide and for chronic myopathic change (excessive fiber size variation, increase in number of internal nuclei). A total of 101 horses and ponies with lesions of polysaccharide storage myopathy were identified. Age of affected horses ranged from one to 30 years, with a mean of 14.7 years. Mean age of nonaffected horses was 12 years. Incidence of polysaccharide storage myopathy varied depending on breed; Thoroughbreds had the lowest (27%) and draft-related horses had the highest (86%) incidence. Chronic myopathic changes were more severe in polysaccharide storage myopathy-affected horses than in nonaffected horses. Results of this study indicate that polysaccharide storage myopathy is a common disorder of many breeds of horses and ponies.
Equine polysaccharide storage myopathy (EPSSM) is characterized by abnormal accumulation of glycogen and glycogen-related polysaccharide in skeletal muscle fibers. 3–12 This disorder is believed to involve abnormal carbohydrate metabolism, but to the authors' knowledge, a specific defect has not been identified. 7, 10 A range of clinical signs of disease related to muscle dysfunction, including exertional rhabdomyolysis, muscular atrophy, abnormal gait, back pain, and postanesthetic myopathy have been described. 3–9, 11 Subclinical or unrecognized disease also is common, 11, 12 This disorder is thought to be inherited, 3, 7 and is most often reported in Quarter horse-related breeds, warmbloods, and draft-related breeds. 3, 4, 6–11 Other breeds in which EPSSM has been reported include Thoroughbred crossbreds, Andalusian, Arabian, Morgan, Standardbred, and Welsh pony crossbreds. 4, 5, 12 In a necropsy study of 37 draft-related horses and one draft mule, the incidence of pathologic changes indicative of EPSSM was 66%. 11 To our knowledge, the incidence of this disorder in other breeds has not been reported. The purpose of the study reported here was to evaluate muscle samples from horses examined at necropsy to determine the incidence of EPSSM in various breeds.
A sample of semimembranosus or semitendinosus muscle was obtained from all horses and ponies 1 year of age or older that were examined after death at the Veterinary Diagnostic Laboratory at Oregon State University over a 2.5-year period. The sample obtained was a longitudinal strip of muscle approximately 5 cm long and 1 cm in diameter, similar to that obtained by biopsy. 10 The samples were placed on a portion of a wooden tongue depressor and were immersed in neutral-buffered 10% formalin. Five to eight sections, one longitudinal section from the periphery of the sample, and the rest full-thickness transverse sections, were embedded in paraffin, sectioned at 4-µm thickness, and stained with hematoxylin and eosin (HE) and periodic acid-Schiff (PAS), with and without amylase digestion. Sections were examined in blinded manner by one of the authors (B. A. Valentine), and were evaluated for abnormal polysaccharide storage (subsarcolemmal aggregates of glycogen, central cytoplasmic bodies containing glycogen, inclusions of amylase-resistant material) and chronic myopathic change (excessive fiber size variation and increase in number of internal nuclei). Subsarcolemmal glycogen aggregates were graded as 0 (average, fewer than 1/10x field), 1+ (average, 1–3/10x; field), 2+ (average, 4–6/10x field), or 3+ (average, more than 6/10x field). Fiber size variation was subjectively judged to be absent (0), mild (1+), moderate (2+), or severe (3+). Internal nuclei were judged to be 0 (absent), 1+ (present, but average of fewer than 1/10x field), 2+ (average, 1–2/10x field or presence of fibers with two or more internal nuclei), or 3+ (average of more than 3/10x field). Central cytoplasmic bodies and amylase-resistant inclusions were recorded as present or absent.
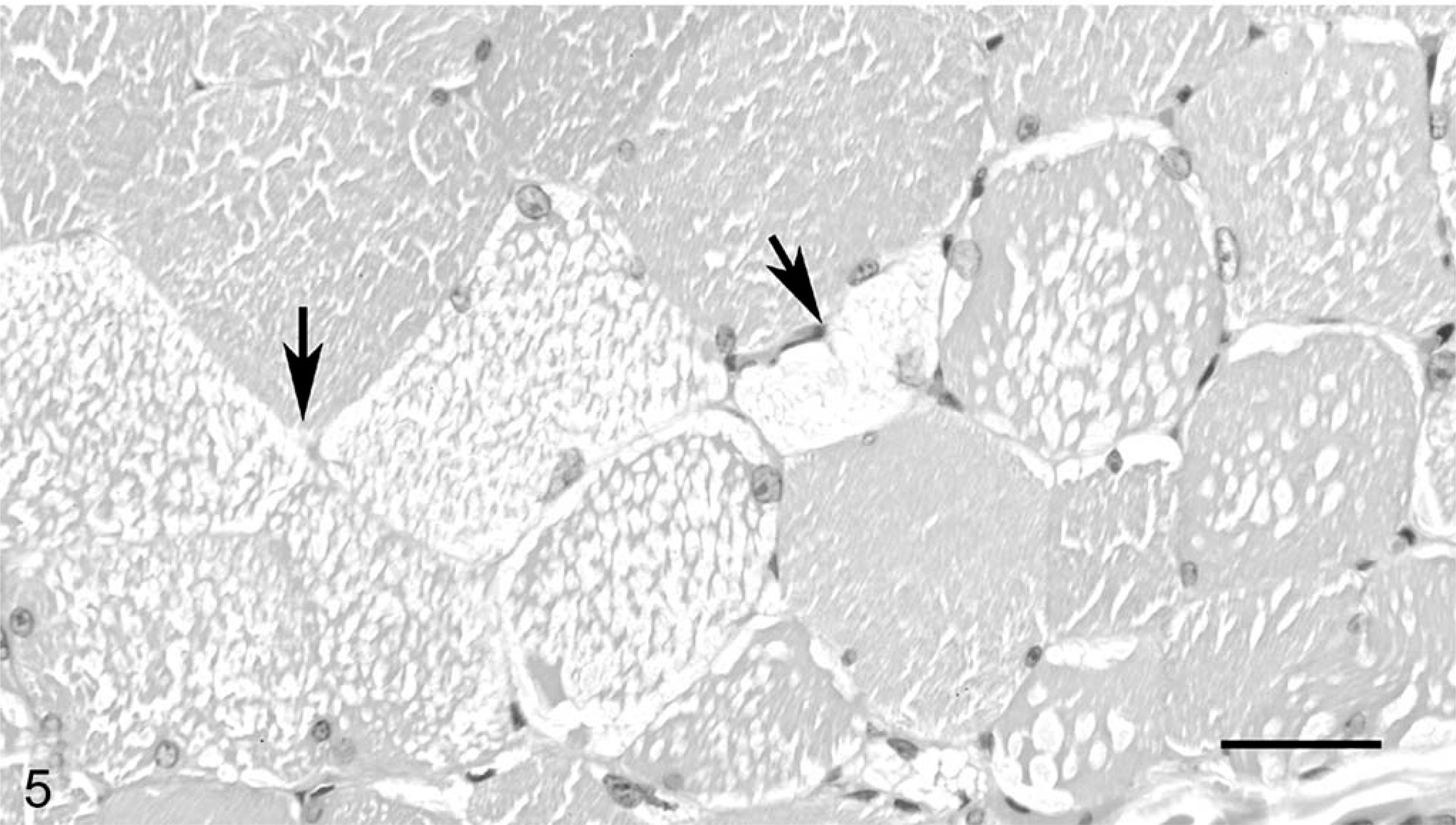
A diagnosis of EPSSM was made for samples containing amylase-resistant inclusions (Fig. 1), 3+ subsarcolemmal aggregates of glycogen (Fig. 2), central cytoplasmic bodies containing glycogen (Fig. 3), or any combination of these findings. These criteria were based on findings of previous studies. 3–12 Findings in PAS-stained sections of EPSSM-affected horses differed substantially from the glycogen staining pattern in muscle samples from horses considered normal (Fig. 4). Glycogen staining of all samples was extremely variable, and postmortem artifactual depletion of glycogen was common. Glycogen was often depleted in all but peripheral fibers within transverse sections (due to poor penetration of formalin into the interior of samples), and these areas were not included when evaluating samples for abnormal glycogen storage. Amylase-resistant inclusions persisted despite severe overall glycogen depletion, and the number present varied from one to many. In cases with numerous amylase-resistant inclusions, these could be seen as pale pink to blue-gray inclusions on HE-stained sections (Fig. 5). Subsarcolemmal vacuoles that were either clear or that contained pale pink hyaline material were also common in HE-stained sections from EPSSM-positive horses. Characteristic findings in many cases, however, were detected only after examination of PAS-stained sections. Myofiber necrosis or regeneration was uncommon. Chronic myopathic changes were best visualized on PAS-stained sections with amylase digestion.
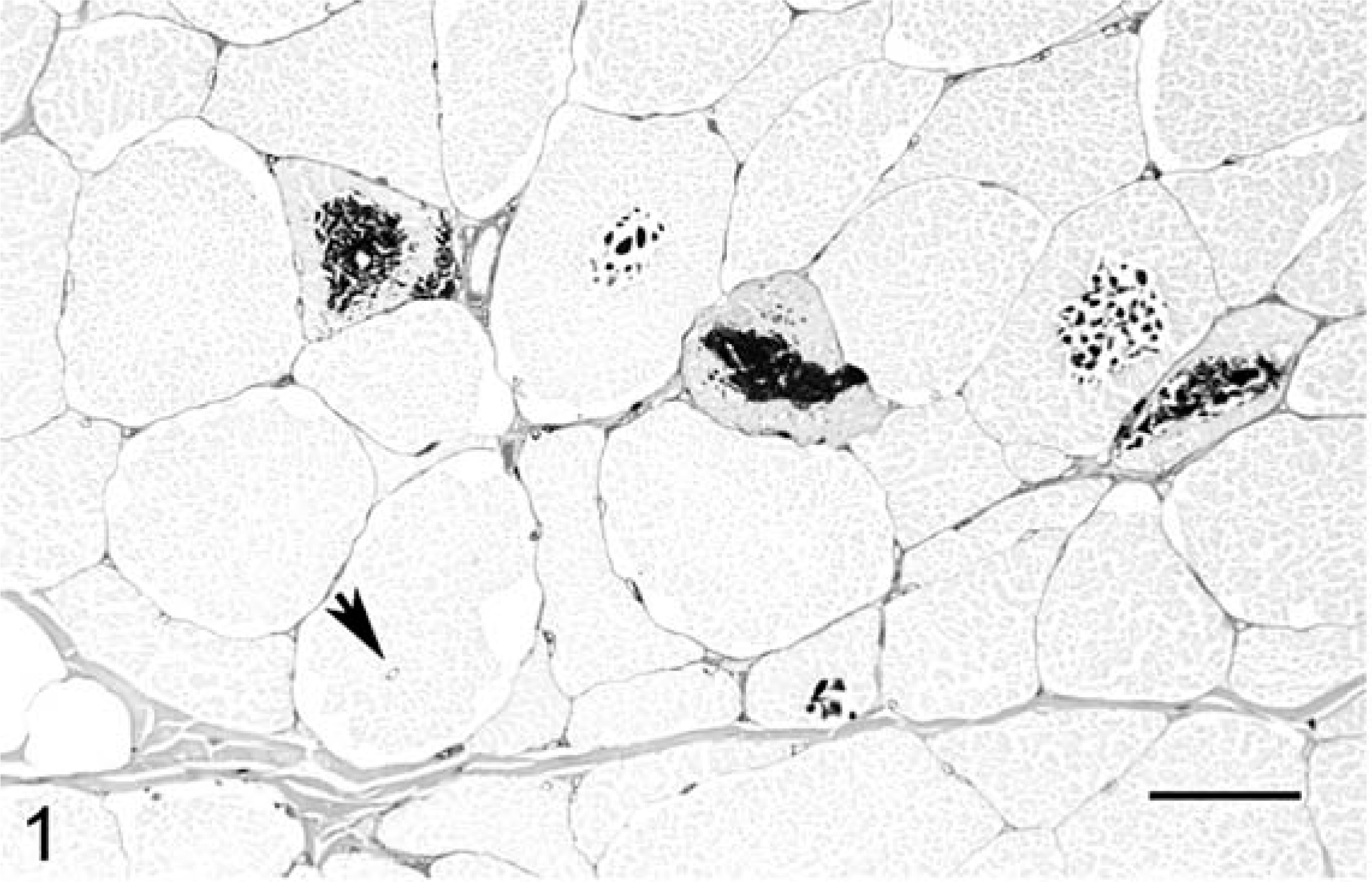
Skeletal muscle; 18-year-old draft mare with EPSSM. Multiple fibers contain central or subsarcolemmal darkly stained, PAS-positive, amylase-resistant inclusions. Adjacent fibers often exhibit clear subsarcolemmal vacuoles, many of which contained amylase-sensitive glycogen. There is a marked increase in fiber size variation, and a fiber with an internal nucleus is present (arrow). PAS with amylase digestion. Bar = 55 µm.
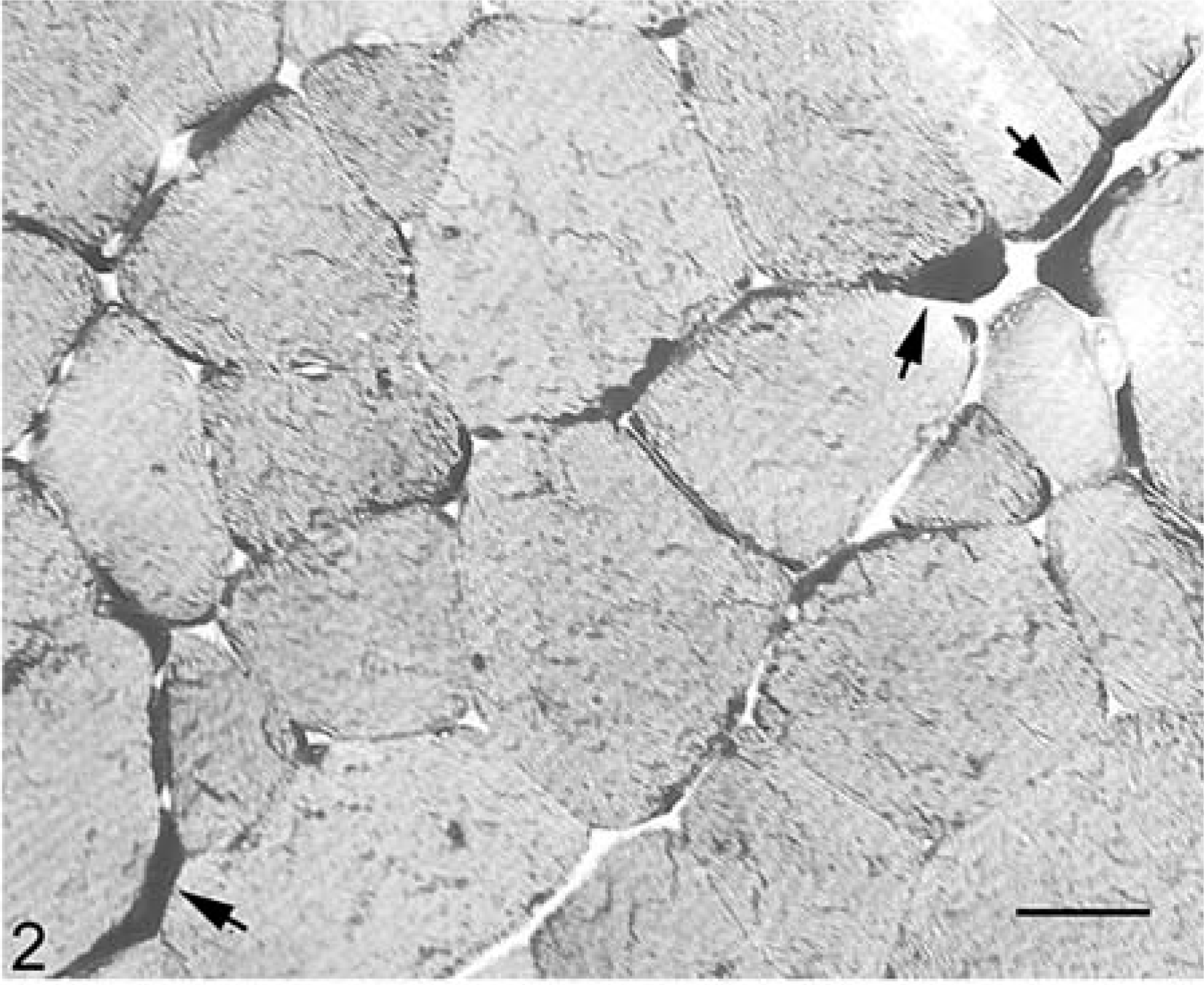
Skeletal muscle; 25-year-old Quarter horse gelding with EPSSM. Notice numerous prominent subsarcolemmal aggregates of PAS-positive glycogen (arrows). There is a mild increase in fiber size variation. PAS. Bar = 27 µm.

Skeletal muscle; 27-year-old Arabian mare with EPSSM. Two fibers containing central round to ovoid cytoplasmic bodies containing glycogen (arrows). PAS. Bar = 27 µm.
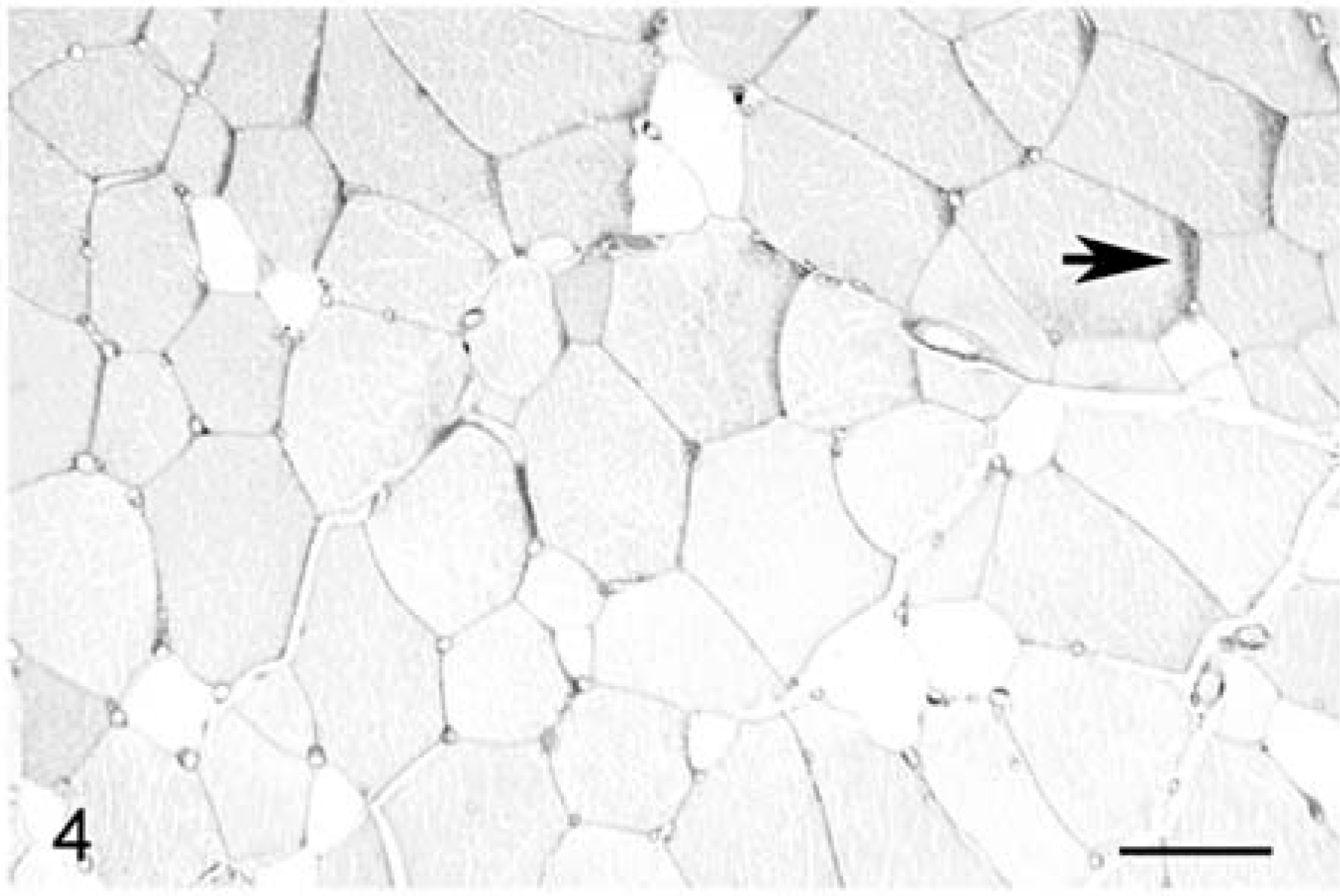
Skeletal muscle; 4-year-old Quarter horse mare with a normal PAS staining pattern and no evidence of EPSSM. The fiber size distribution is normal, and there are no internal nuclei. There is overall depletion of glycogen due to postmortem degradation. Larger-diameter, slightly darker fibers contain higher glycogen content than do the smaller-diameter, unstained fibers, corresponding to high glycolytic and high oxidative fibers, respectively. Narrow dark zones at the periphery of some fibers (arrow) are a common artifact indicative of slightly oblique orientation. HE. Bar = 55 µm.
Skeletal muscle; 30-year-old Mustang gelding with EPSSM. Multiple fibers contain multiple pale inclusions of abnormal polysaccharide, often observed in small clusters (left arrow). One fiber is filled with similar material (right arrow). HE. Bar = 55 µm.
A total of 225 horses were included in the study. Ten additional horses were excluded due to failure to obtain a muscle sample or to inadequate available medical records data. A total of 101 horses with lesions of EPSSM were identified; 124 horses did not have lesions of EPSSM. Amylase-resistant inclusions were present in 51 affected horses, and central cytoplasmic bodies were found in 17 horses. Diagnosis of EPSSM relied on the presence of central cytoplasmic bodies in six horses; in 11 horses, central cytoplasmic bodies were observed in conjunction with amylase-resistant inclusions and/or 3+ subsarcolemmal glycogen aggregates.
Medical records were obtained for all horses. The data were analyzed to determine the incidence of lesions of EPSSM in the major breeds represented. Crossbreds of Arabian, Morgan, draft, warmblood, Quarter horse, Paint, and Appaloosa were included in those breed headings. Other categories included only purebreds. The scores for fiber size variation and for internal nuclei were added to obtain a chronic myopathic score for each horse. The mean chronic myopathic score (± SE), mean score for subsarcolemmal glycogen aggregates (± SE), and mean age (± SE) were calculated for all EPSSM-positive horses and for all EPSSM-negative horses. Mean ages, mean glycogen scores, and mean chronic myopathic scores of EPSSM-positive and EPSSM-negative horses were analyzed for significant differences (Student's
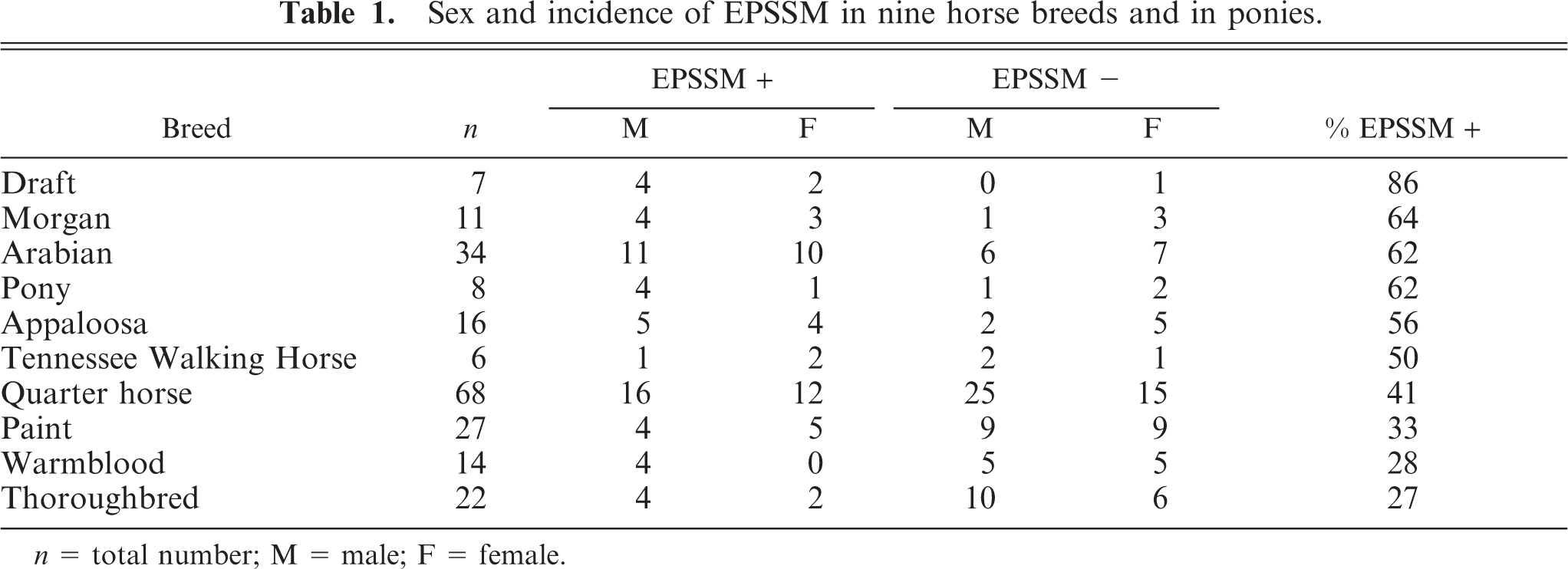
Data from breeds with six or more horses are presented in Table 1. Data from breeds with fewer than six horses are presented in Table 2. Pathologic findings characteristic of EPSSM were detected in horses and ponies of many breeds (Tables 1, 2), with incidence ranging from 27% to 86% (Table 1). A sex predisposition was not apparent (no analysis performed). The mean chronic myopathic score for EPSSM-positive horses (2.66 ± 0.14) was significantly higher than that for EPSSM-negative horses (1.33 ± 0.14;
Sex and incidence of EPSSM in nine horse breeds and in ponies.
Incidence of EPSSM in other horse breeds.
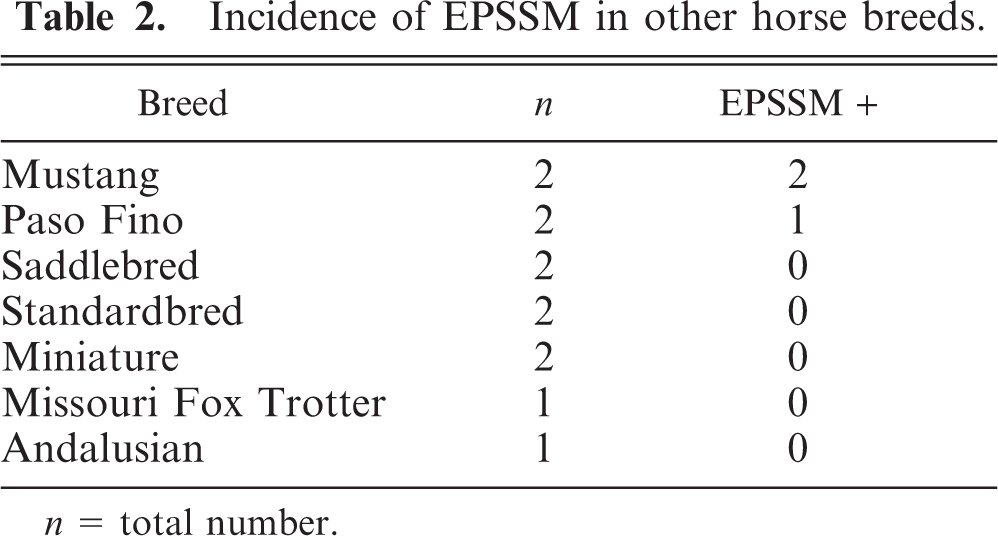
These findings indicate that pathologic evidence of EPSSM is common in many horse breeds. The highest incidence (86%) was found in draft-related horses and was higher than that previously reported. 11 The data from this study may be skewed due to the small number of draft-related horses studied and the fact that five of six affected draft horses were Percheron-related, a breed previously recognized to have a higher incidence of EPSSM than that of other draft breeds studied. 11 The high incidence found in ponies may reflect the fact that four of eight (three EPSSM-positive and one EPSSM-negative) animals studied were Pony of the Americas, a breed originally derived from Appaloosa horses. To the authors' knowledge, polysaccharide storage myopathy has not been reported in this pony breed. Other previously unreported breeds recognized to have lesions of EPSSM, including amylase-resistant inclusions, are Thoroughbred, Tennessee Walking Horse, Mustang, and Paso Fino.
To date and to our knowledge, an immunohistochemical procedure has not been developed for diagnosis of EPSSM. Although severe cases contained obvious inclusions on HE-stained sections (Fig. 5), detection of EPSSM often required examination of PAS-stained sections. Cases in which amylase-resistant inclusions and internal glycogen inclusions involved few fibers indicate that examination of small samples or of only one or two sections from a sample may not be sufficient to detect these changes. When present, chronic myopathic change was a diffuse lesion that was readily detected in all sections. Formation of subsarcolemmal glycogen aggregates also was a widespread lesion, but detection required adequate preservation of glycogen; severe postmortem glycogen depletion will make it difficult or impossible to diagnose EPSSM in such cases.
Central glycogen-associated cytoplasmic bodies were considered to be a finding related to EPSSM, and have been associated with this myopathy. 9 Similar structures have been reported in clinically normal horses as well as horses with neuromuscular disease, leading to the suggestion that they are a “non-pathological degenerative structure.” 1 Most of the horses of this study with central cytoplasmic bodies also had other findings indicative of EPSSM, suggesting that these structures are a feature of this myopathy. Given that muscle spasm and abnormal muscle tension are frequent clinical findings in EPSSM-positive horses, 3, 5, 9, 10 it is interesting that similar myofiber inclusions have developed in rat muscle following induction of localized tetanus. 2 Formation of cytoplasmic bodies may be secondary to increased muscle tension. Further studies of cytoplasmic bodies in equine muscle are clearly needed.
Chronic myopathic change is a feature of many neuromuscular diseases. 10 The finding of a significantly higher chronic myopathic change score in horses of this study that were diagnosed with EPSSM is evidence that the pathologic criteria used for diagnosis of this disorder are valid. It is also possible that increased degree of myopathic change is age related, as the mean age of EPSSM-positive horses was slightly but significantly higher than the mean age of nonaffected horses. However, the difference in mean age between EPSSM-affected and nonaffected horses does not appear to be sufficiently large to account for the highly significant difference in myopathic score. The characteristic pathologic changes in EPSSM are progressive 3 ; therefore, it is not surprising that the mean age of affected horses in this study was slightly higher than that of unaffected horses.
Most horses studied did not have a recent history of clinical signs of muscular dysfunction, nor had they been evaluated for evidence of muscle disease. Therefore, the clinical relevance of finding evidence of EPSSM in these horses cannot be determined. As an attempt was not made to determine whether clinical signs of muscular dysfunction had been evident prior to death or euthanasia, it is possible that such clinical signs had been recognized but were not recorded in the medical history. However, the presence of this underlying myopathy could result in clinical signs of disease (e.g., weakness, pain, muscle atrophy, difficult recovery from general anesthesia) and clinicopathologic findings (i.e., increased serum activity of creatine kinase and/or aspartate aminotransferase) in affected horses that develop other disorders.
Footnotes
Acknowledgements
We thank Dr. Jerry Heidel, Dr. Christiane Löhr, Dr. Rob Bildfell, Mehmet Kupeli, and Jermey Becker for collection of samples, and Patricia DeVaughn for assistance with medical record data collection.
This study was funded by a donation from Tom and Mindy Wolfe.
