Abstract
Several immunohistochemical markers have been used to define the differentiation pattern of urothelial cell tumors of the urinary bladder. We investigated the expression of the recently characterized uroplakin (UP) IIIb, an urothelium-specific and differentiation-dependent protein, in 39 urothelial tumors of the urinary bladder in cows that had suffered from chronic enzootic hematuria for several years. Bovine papillomavirus type 2 DNA was amplified and UP IIIb protein was detected in all these tumors. In papillomas and papillary carcinomas, UP IIIb expression was mostly seen as superficial staining; luminal and peripheral patterns were also observed. In nonpapillary carcinomas, UP IIIb appeared to define clearly the cell membrane lining intercellular and intracellular lumina as well as the cell borders in deeper cell layers. In benign and malignant lesions, an intracytoplasmic immunoreactivity was also detected. Coarse intracytoplasmic UP IIIb-positive material close to the nucleus occurred in some malignant cells. Focally strong membraneous immunostaining that marked single cells with complete ringlike peripheral pattern was seen. Although UP IIIb expression does not seem to correlate with the biological behavior of urothelial tumors, it appears to be a highly sensitive marker for bovine urothelial tumors.
Urinary bladder tumors are very rare in cattle, accounting for 0.01% of all bovine malignancies. 18 These tumors are commonly encountered in cows that have grazed on pastures rich in bracken fern (Pteridium spp.); the fern contains toxic principles, and its prolonged ingestion is responsible for a clinical syndrome known as chronic enzootic hematuria. It is believed that bovine papillomavirus type 2 (BPV-2) also plays an important role in the bladder carcinogenesis associated with this syndrome, 4, 6 although the precise synergism between the toxic principles of bracken fern and BPV-2 is still unclear.
Mammalian urothelium is composed of basal, intermediate, and superficial cells. The superficial (umbrella) cells are covered by highly specialized plaques whose luminal leaflet is twice as thick as their cytoplasmic leaflet. Urothelial plaques, also known as asymmetric unit membranes (AUMs), consist of two-dimensional crystalline arrays of 16-nm protein particles arranged hexagonally. 5, 11, 13, 24, 25
Characterization of the AUMs from bovine urothelium has led to the identification of several integral membrane proteins called uroplakins (UPs). Four uroplakins, i.e., UP Ia (27 kDa), UP Ib (28 kDa), UP II (15 kDa), and UP III (47 kDa), have been shown to be highly conserved during mammalian evolution in terms of their ultrastructure, protein composition, and immunological properties. 26 These four uroplakins interact, forming two pairs, both of which are required for normal urothelial plaque formation. 15 Recently, Deng et al. 8 described a novel 35-kDa uroplakin, p35, whose amino acid sequence is highly homologous to UP III. Because of the many similarities between these two proteins, it has been proposed to rename UP III and p35 as uroplakins IIIa and IIIb, respectively. Like UP IIIa, UP IIIb is associated with the differentiated urothelial cells, 8 and dimerizes with uroplakin Ib as an early step of urothelial plaque assembly.
It has been suggested that the ability of urothelium to terminally differentiate is progressively lost during neoplastic transformation. UPs may therefore be a useful biomarker for assessing the degree of differentation in bladder carcinogenesis. 28 The aim of the present paper is to evaluate UP IIIb immunoreactivity as a novel urothelial cell tumor marker in 39 cases of BPV 2-associated spontaneous urothelial tumors of the urinary bladder in cows grazing on bracken-infested lands in southern Italy. This is, to our knowledge, the first assessment of UP IIIb immunoexpression in urothelial cells of wild and domestic animals.
Materials and Methods
One hundred and fifteen samples of neoplastic urothelium were collected at public slaughterhouses from 4- to 24-year-old cows that had been suffering from chronic enzootic hematuria for several years. All the animals were known to have grazed on bracken-infested pasture as they were raised in mountain cattle households in the south of Italy, as reported elsewhere. 3 Urothelium, uterus, and vagina from five 4- to 14-year-old healthy cows were also examined as control. The control animals were from lowlands in which no bracken was present.
Virus analysis and selection of cases
For viral analysis, a small fragment of frozen tissue of 61 urothelial tumors was digested by Proteinase-K in the lysis buffer (50 mM KCl, 10 mM Tris HCl, pH 8.3, 2.5 mM MgCl2, 100 µg/ml gelatin, 0.45% NP-40, and 0.45% Tween-20) to recover DNA. Ten microliters of samples were amplified by PCR utilizing one unit of Taq polymerase (Finenzymes, Espoo, Finland) in the buffer provided by the manufacturer with 1.5 mM MgCl2. Amplification reaction was carried out in a Thermal Cycler (Perkin Elmer, Wellesley, MA) for 30 cycles with an annealing temperature of 50°C. Primers detecting the BPV-2 sequences were upstream primer 5′-TTGCTGCAATGCAACTGCTG-3′ (base pairs [bp] 3919–3938), downstream primer 3′-TCATAGGCACTGGCACGTT-5′ (bp 4207–4225). In each experiment, a blank sample consisting of reaction mixture without DNA and a positive sample consisting of BPV-2 cloned DNA (kindly provided by Dr. M. S. Campo) were included. A band corresponding to the amplified sequences of BPV-2 was detected in 39 cancer samples. 4 To validate the PCR results, the amplified products were sequenced.
Tissues, antibodies, and immunohistochemical procedure
The tissues were fixed in 10% buffered formalin and processed routinely for paraffin embedding. Histologic diagnosis was assessed on 5-µm-thick hematoxylin-eosin (HE)–stained sections.
For immunohistochemical analysis, the avidin-biotin-peroxidase method was used. Briefly, 5-µm-thick sections were blocked for endogenous peroxidase in 0.3% H2O2 in methanol for 20 minutes. Antigen enhancement was performed by pretreating with microwave heating (twice for 5 minutes each at 750 W) in citrate buffer, pH 6.00.
Anti-p35 antibody reacting with UP IIIb (generously provided by Drs. Fangming Deng, Fengxia Liang, and Tung-Tien Sun, Epithelial Biology Unit, Ronald Perelman Department of Dermatology, Department of Pharmacology and NYU Cancer Institute, New York University School of Medicine) was made against highly purified bovine UP IIIb. Briefly, the rabbit antibody to UP IIIb was generated against synthetic peptides of UP IIIb. The antibody was shown to be monospecific for UP IIIb when used to immunoblot total cellular proteins of bovine urothelium. 8 By immunofluorescent staining, the antibody was shown to stain preferentially the superficial umbrella cells of normal bladder urothelium. 8 It was applied at room temperature for 2 hours at a 1:200 dilution. Sections were washed three times, then incubated with secondary antibody (biotinylated goat anti-rabbit IgG, Dako, Glostrup, Denmark) for 30 minutes, followed by incubation with streptavidin-conjugated horseradish peroxidase (Dako, Glostrup, Denmark). Color development was obtained with 5–20 minutes of diaminobenzidine treatment. Sections were counterstained with hematoxylin. The primary antibody was omitted and replaced by PBS in the corresponding negative control section.
Confocal laser scanning investigation and immunofluorescence procedure
For immunofluorescence staining, the same primary anti-p35 antibody was applied at room temperature for 2 hours at a 1:50 dilution. Slides were washed three times, then incubated with FITC conjugate goat anti-rabbit (Jacksonimmuno, Milan, Italy) for 2 hours at 1:100 dilution. Slides were rinsed three times with PBS and incubated for nuclear staining for 30 minutes in 10 µg/mL propidium iodide (Sigma, Milan, Italy). After washing, the slides were mounted with an aqueous medium (Sigma, Milan, Italy). For scanning and photography, a laser scanning microscope (LSM 510, Zeiss, Göttingen, Germany) was used. FITC was irradiated at 488 nm and detected via a 505–560 nm band pass filter. Propidium iodide was irradiated at 488 nm and detected via a 560 nm long pass filter. The different frames were scanned separately, with appropriate installation of the optic path for excitation and emission of each scan according to the manufacturer's instructions.
The primary antibody was omitted and replaced by PBS in the corresponding negative control section.
Results
The primers allowed amplification of a 307 bp DNA amplicon encompassing the BPV E5 sequence. To determine the papillomavirus genotype, PCR amplicons were sequenced and proven to correspond to the expected region of BPV-2 (Fig. 1).
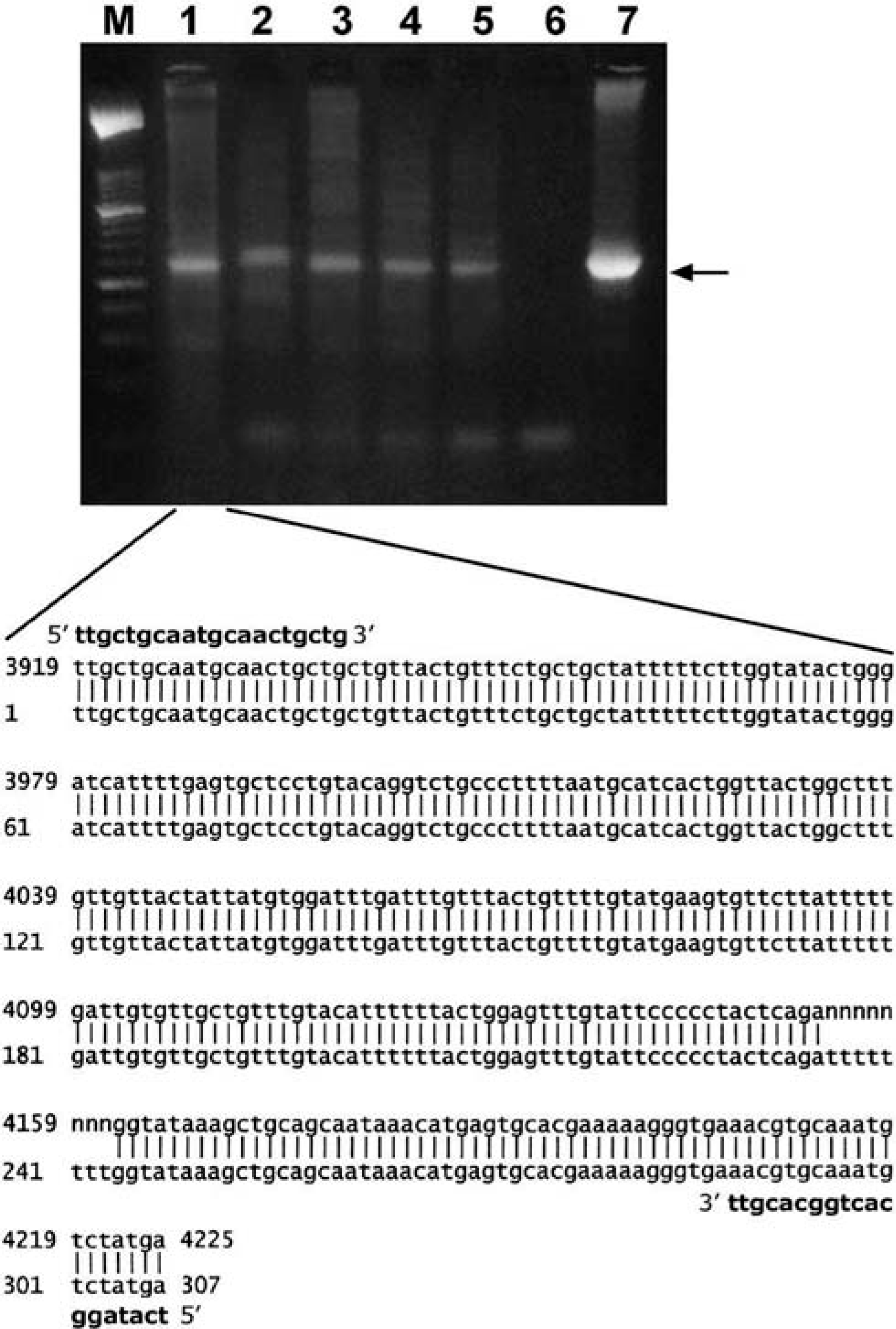
PCR amplification of urinary bladder samples. Lanes: M, molecular mass marker type VI (Roche Diagnostics); 1–5, tumor samples; 6, negative control with no DNA added; 7 positive control of BPV-2 plasmid. The arrow indicates the position of the 307-bp BPV-2 PCR product. The lower part of the figure shows similarity between the sequence of the amplicon in lane 1 (top line) and the sequence of the prototype BPV-2 (GenBank NC-001521; bottom line). Primer sequences are in bold. N = not properly identified bases.
UP IIIb was not immunohistochemically detected in uterus as well as vagina samples used as control (Fig. 2).
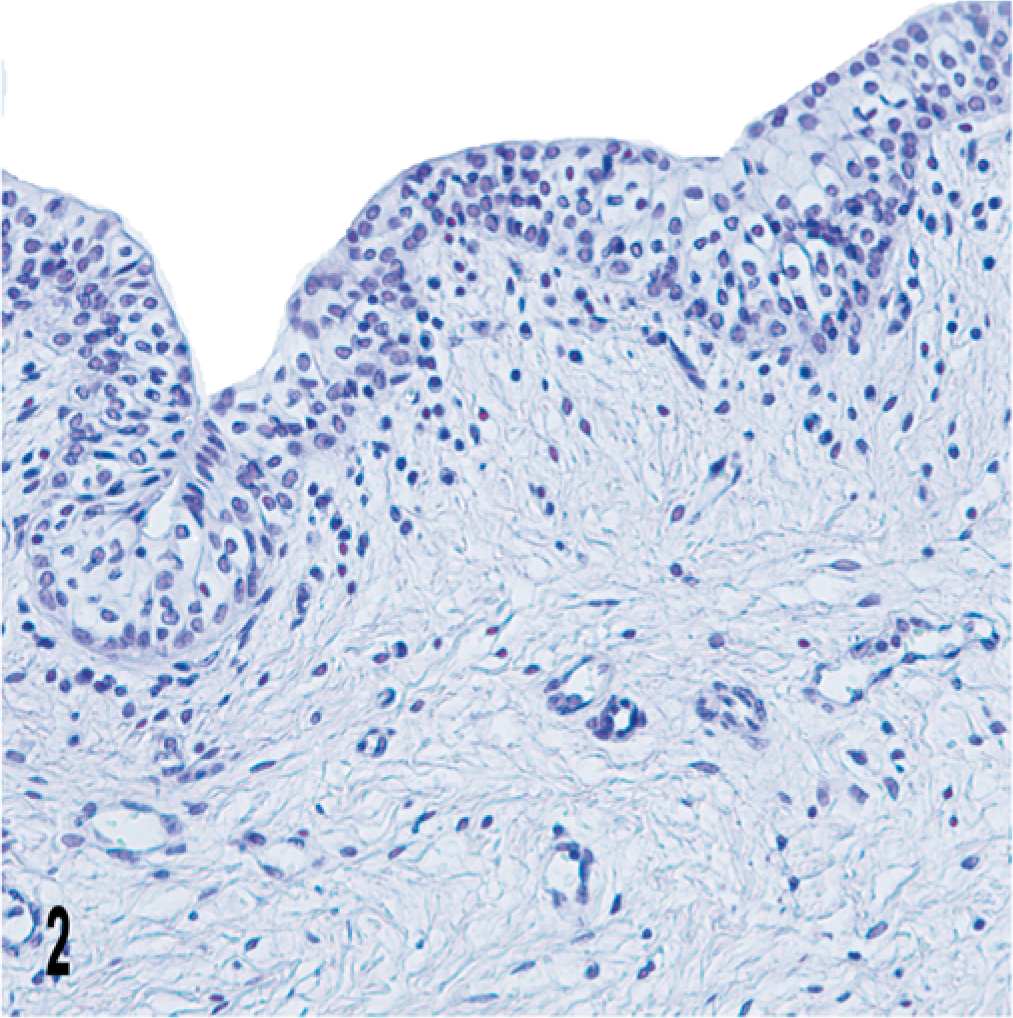
Vagina; cow. No immunohistochemical expression of UP IIIb is manifest in the vaginal epithelium. Streptavidin-biotin-peroxidase, Mayer's hematoxylin counterstain.
In normal bovine urothelium, UP IIIb was clearly manifest as a continuous line covering the entire apical surface of the umbrella cells. The cytoplasm of superficial and intermediate cells contained strongly stained cytoplasmic granules located mainly above the nucleus thus giving a half moon-like appearance (Fig. 3).
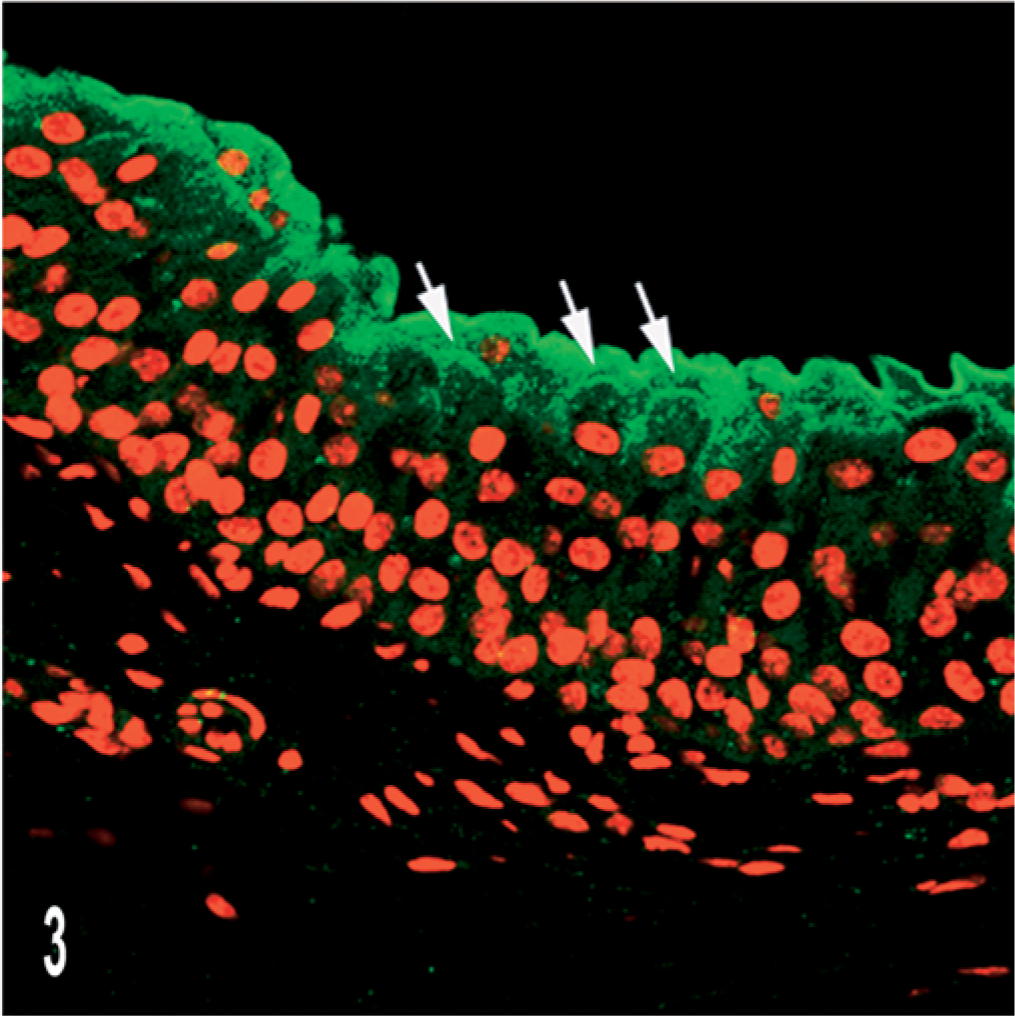
Normal urothelium; cow. UP IIIb expression is documented by scanning laser confocal microscopy. Notice continuous green line covering the umbrella cell as well as immunofluorescence in the adjacent intermediate cells (arrows). Indirect immunofluorescence. Propidium iodide red nuclear counterstain.
The results of immunohistochemical patterns of the 39 urinary bladder tumors for UP IIIb are summarized in Table 1.
Uroplakin IIIb in bovine urothelial carcinomas.

The bovine urothelial tumor exhibited three main staining patterns: “superficial,” “luminal,” and “peripheral.” In all three papillomas examined, UP IIIb expression was evident as a linear superficial membrane staining, i.e., the superficial pattern. Discontinuity of the apical membrane immunostaining of the superficial cells was rarely detected. Occasionally, intermediate cells showed an intense and diffuse cytoplasmic reactivity for UP IIIb (Fig. 4). The superficial pattern was evident in all papillary tumors, infiltrating or not. Some of these tumors also exhibited the luminal (including microluminal) pattern, which was characterized by the presence, within the carcinomatous proliferations, of numerous lumina and microlumina exhibiting membranous immunostaining located near the cell surface (Fig. 5).
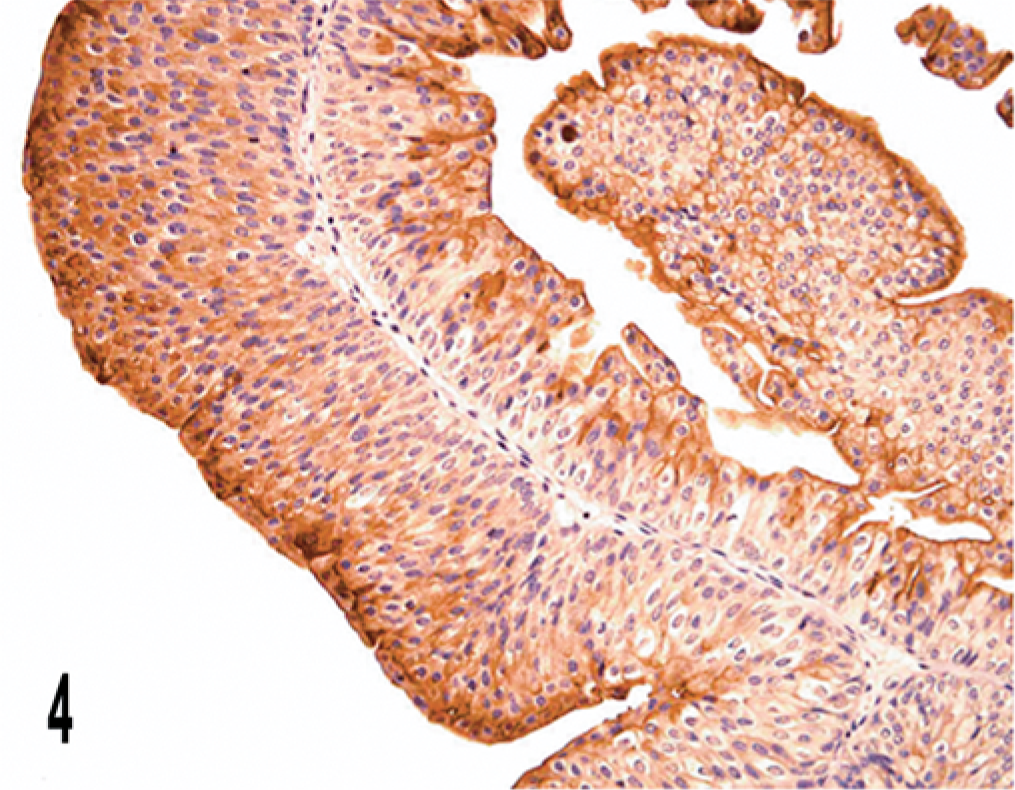
Papilloma; cow. Superficial pattern is documented; intercellular and intracellular immunostaining is also present. Streptavidin-biotin-peroxidase, Mayer's hematoxylin counterstain.
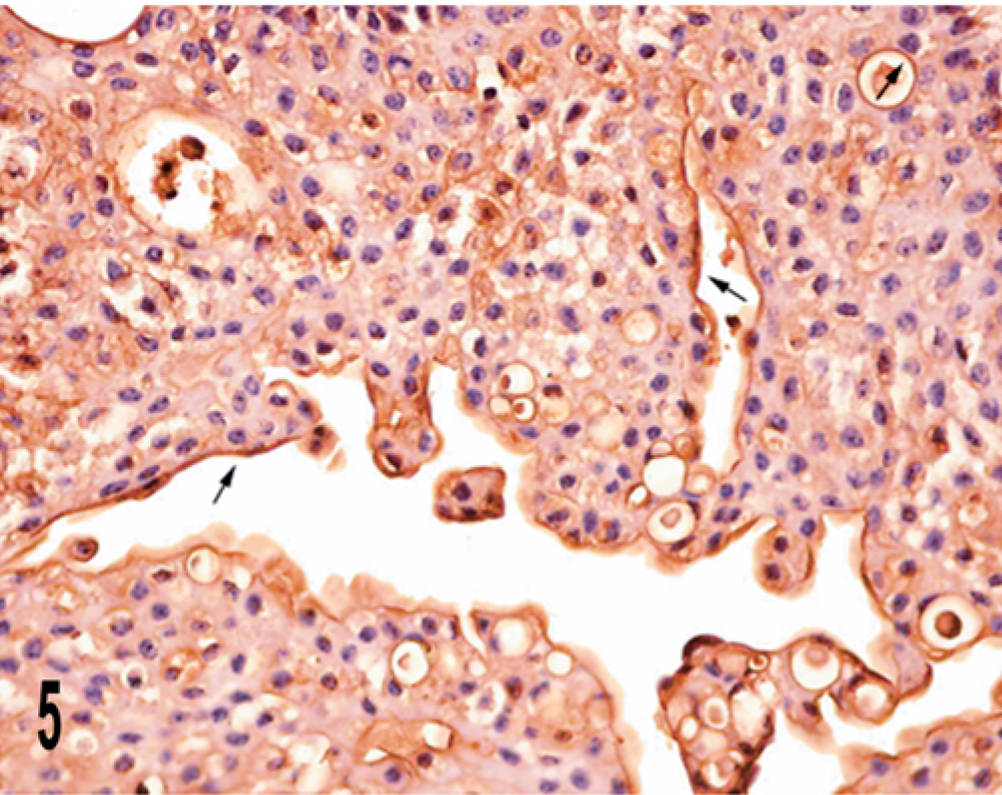
Papillary carcinoma; cow. Luminal and microluminal patterns are documented (arrows). Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain.
The irregular distribution of UP IIIb-positive cells was, in some cases, quite prominent (Fig. 6).
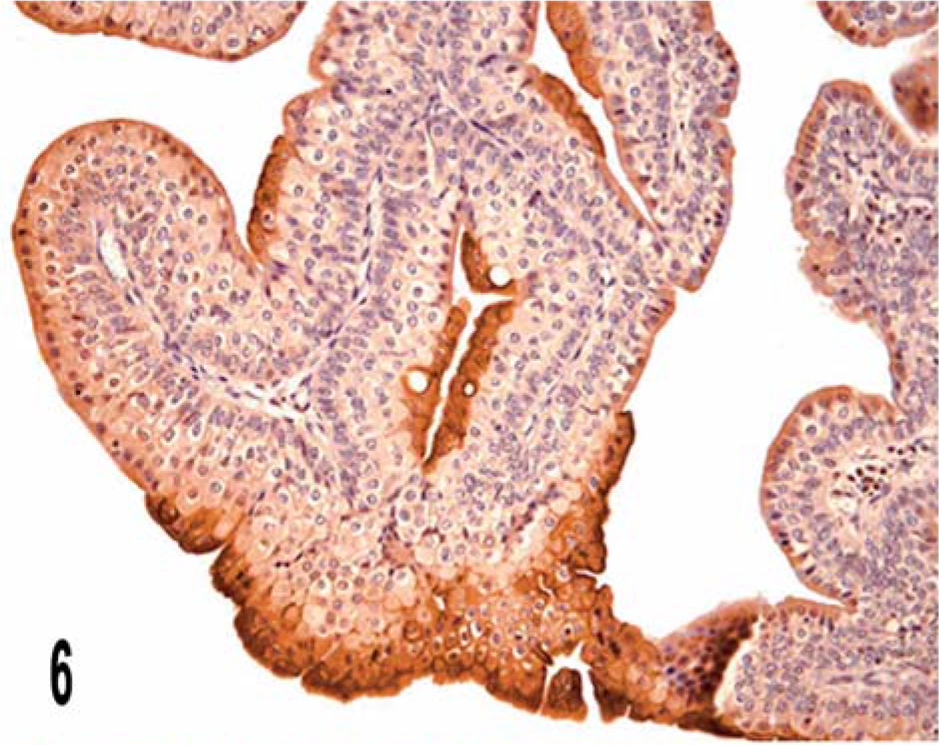
Papillary carcinoma; cow. Irregular distribution of UP IIIb in superficial and luminal cells is manifest. Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain.
The “peripheral pattern” was characterized by focal linear staining of the peripheral cell membrane of neoplastic cells. In many cases, a clearly membranous immunoreactivity marked and delineated single cells, and a complete ringlike peripheral staining was evident. Coarse cytoplasmic UP IIIb-positive material above the nucleus could be seen in some of these cells (Fig. 7a). A strong cytoplasmic immunoreactivity was detected in some intermediate cells of the urothelium adjacent to neoplastic proliferation.
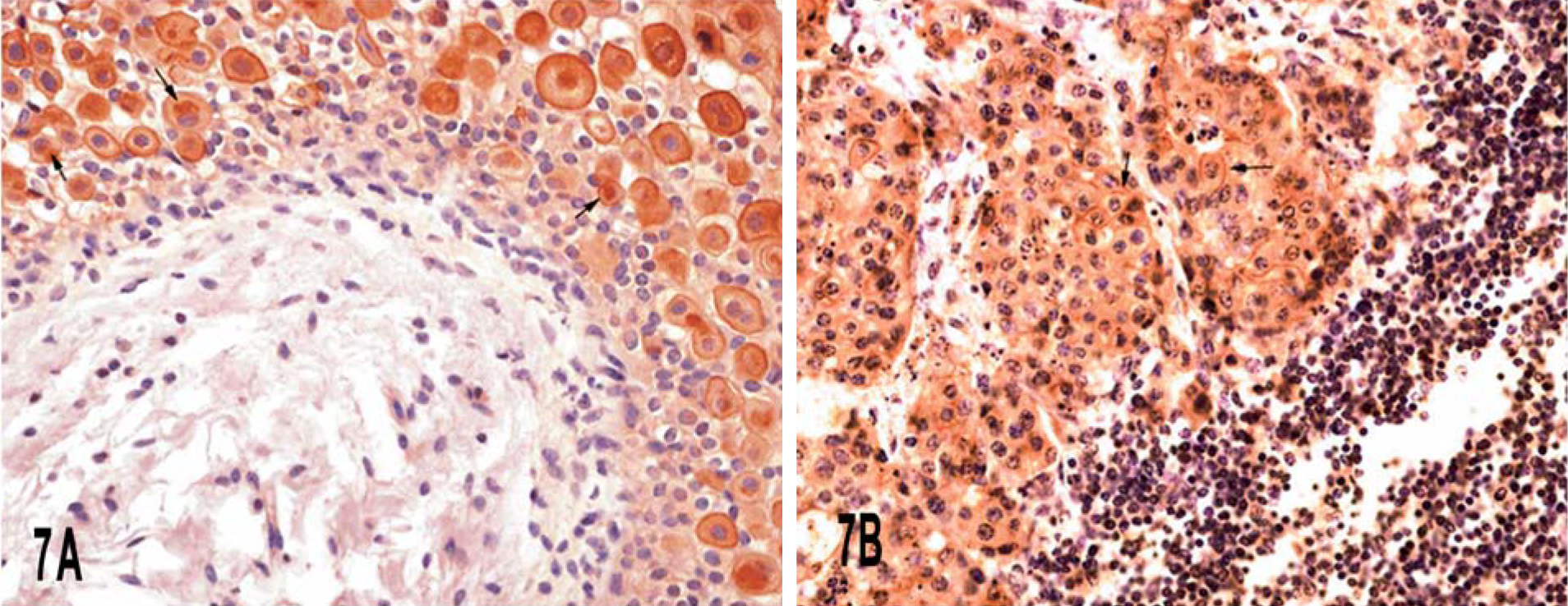
In invasive nonpapillary and infiltrating carcinomas, both luminal and peripheral patterns were detected, although the peripheral pattern was more clearly evident in two lomboaortic lymph node metastases (Fig. 7b). Cell of the urothelium covering infiltrating tumors had an intense, diffuse, cytoplasmic reactivity of UP IIIb; the discontinuous staining of the apical portion of the cell was also seen very frequently (Fig. 8).
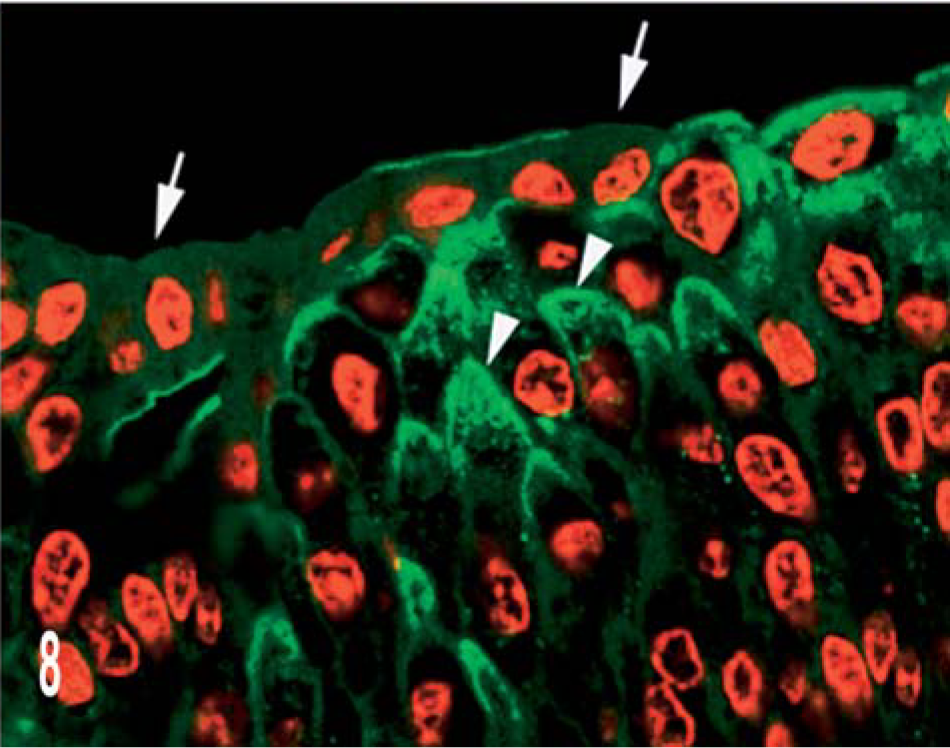
Invasive nonpapillary carcinoma; cow. Expression of UP IIIb in the urothelium covering infiltrating tumors is documented by confocal laser scanning microscopy. Note the discontinuous staining of the apical cell surface (arrows), and the accumulation of strong UP IIIb granules in intermediate cells, mostly in the apical portion of the cell (arrowheads). Indirect immunofluorescence. Propidium iodide red nuclear counterstain
Discussion
Uroplakin expression can be perturbed in spontaneous as well as experimentally induced urothelial tumors. 17, 20 For example, Ambrosio et al. 1 showed altered UP expression in bovine urothelial tumors.
UP IIIa is an integral and indispensable subunit of urothelial plaques, which are required for maintaining the morphologic and functional integrity of the urothelium. 12 UP IIIb is a novel, 35-kD isoform of UP IIIa. Their amino acid sequences are 34% identical, and they share an extracellular juxtamembrane stretch of 19 amino acid residues. 8 Both UP IIIa and UP IIIb possess a large cytoplasmic domain that may play a role in anchoring the AUM to the cytoskeleton. 8, 27 Although UP IIIb is present only as a minor component of urothelial plaques (10% of UP IIIa), it may be associated with a subpopulation of urothelial plaques 8 and may play an important role in regulated urothelial plaques assembly.
UP III has been shown to be a highly specific and moderately sensitive immunohistochemical marker for primary and metastatic human urothelial carcinomas, 14 as well as for canine urothelial tumors. 21 We investigated the expression pattern of UP IIIb in 39 cases of BPV-2–associated bovine bladder tumors. We observed mainly three UP IIIb expression patterns: superficial, luminal (including microluminal), and peripheral. The superficial pattern was expressed in all the papillomas and papillary carcinomas we examined, whereas the other two patterns were more rarely seen in both benign and malignant papillary and invasive tumors. Our results indicate that UP IIIb is a novel, highly sensitive immunohistochemical marker for the positive identification of bovine urothelial carcinomas. The sensitivity of UP IIIb in detecting bovine urothelial tumors seems higher than that of UP III when used to detect human and canine urothelial carcinomas. 14, 21 This may be explained by the fact that the primary antibodies used in these surveys were raised against bovine uroplakin sequences.
Irregular discontinuity of immunoreactivity in the superficial cells reflects alterations in the differentiation process. Similarly to what has been reported reported in experimental mouse bladder carcinogenesis 20, 19 and in urothelial tumors of humans, 14, 17 uroplakins are present in the surface membrane, but are usually not assembled into urothelial plaques, indicating that terminal differentiation has been lost in bovine bladder tumors. This is consistent also with the observed luminal and microluminal patterns. It has been demonstrated that AUMs are no longer detectable in membranous lining of the intercellular and intracellular lumina seen in papillary urothelial carcinomas. 7, 9 Although immunoreactivity for UPs has been observed in such lumina in urothelial tumors in both human and veterinary medicine, it is conceivable that UPs are integrated into luminal membranes without the development of highly ordered AUM structures. It is worthwhile to remember that UP expression and AUM formation are not strictly coupled. 22
The remarkable increase in the number of intermediate cells showing a very strong cytoplasmic immunoreactivity supports several interpretations. It might be related to changes in the migration of the differentiating cells toward the surface, an elevated synthesis of UP in these cells, or a combination of both.
It has been suggested that the dimerization of IIIb with UP Ib is a prerequisite allowing IIIb to exit from the endoplasmic reticulum and to reach the cell surface. In this context, abnormal protein synthesis can hinder oligomerization processes; consequently, unassembled UP Ib/IIIb heterodimer cannot exit from this compartment. Alternatively, because of the alterations of cytoplasm compartments, uroplakin-containing vesicles can be accumulated in the cytoplasm. It is worth noting that ultrastructural defects of endomembrane compartments resulting in an impaired transport and condensation of secretory products or in an abnormal secretory material are known to occur commonly in some malignant cells. 10
As far as bovine papillomavirus E5 oncoprotein is concerned, in vitro studies have recently demonstrated that BPV E5 is located in the endoplasmic reticulum (ER) and Golgi apparatus (GA) of the host cell. Furthermore, transformed bovine fibroblasts expressing BPV E5 are highly vacuolated and contain a swollen and misshapen GA. It has been suggested that BPV E5 prevents the transport of proteins to the cell surface and sequesters them within the cell, particularly in the Golgi cisternae, because it causes alkalinization of the endomembrane compartments. 2, 16 Immunofluorescence investigation performed with a scanning confocal laser microscope allows us to detect BPV E5 protein in the cytoplasm of urothelial neoplastic cells. 4 It is reasonable to assume that the intracytoplasmic immunoreactivity in benign and malignant tumors is caused by BPV E5 producing changes in the endomembrane compartments, thus disrupting transport of UP IIIb. It is worthwhile to note that Tu et al. 23 have recently shown the crucial role of the ER and Golgi cisternae in the transport of UP Ib/UP III complex.
Our data indicate that uroplakins are distributed in metastatic bovine bladder tumors, as in their human counterparts, in a peripheral staining pattern. It has been suggested that this staining pattern can be useful for identifying carcinoma metastases of uncertain origin; 17 this may also be true for bovine metastatic bladder tumors.
Finally, our data suggest that UP IIIb can be a marker of choice for bovine urothelial tumors. It is of limited value, however, for assessing the biological behavior of bovine urothelial tumors, because UP IIIb is detected in all urothelial tumors regardless of their grade and stage. Despite recent progress in studying the status and function of uroplakins, further studies are needed to better understand the role of these proteins in urothelial differentiation, and how their expression is perturbed in tumors.
Footnotes
Acknowledgements
We thank Drs Tung-Tien Sun, Fangming Deng and Fengxia Liang of NYU Medical School for their generous gifts of antibodies to bovine UP IIIb; Drs G. Di Domenico and G. Marino from the Azienda Sanitaria Locale SA3 for their collaboration. This study was supported in part by grants from the Ministero dell'Istruzione, dell'Università e della Ricerca (MIUR), Rome.
