Abstract
Expression of uroplakins (UPs) was investigated in 20 bladder tumors from cows that had been suffering from chronic enzootic hematuria for several years. In dysplastic urothelium and papillomatous proliferations, UP expression was evident both as luminal and intercellular staining. UPs appeared to clearly define the plasma membrane of luminal cells and the borders of cells placed in deeper layers, whether or not these intermediate cells were adjacent to superficial ones. Occasionally, some intermediate cells showed a remarkable cytoplasmic immunoreactivity. The pattern of UPs in grade I tumors was characterized by an evident discontinuity of luminal staining and by the presence of numerous intermediate cells showing a diffuse intracytoplasmic positivity for UPs. In grade II tumors, there was a decrease of luminal and intermediate cells showing UP expression and an apparent increase of clusters of intermediate cells with intracytoplasmic reactivity for UPs. In grade III tumors, immunoreactivity was heterogeneously distributed and a severe loss of UP-positive luminal and intermediate cells could be seen. Focally, superficial and deeper cells showed strong membraneous immunoreactivity that marked and delimited single cells, with complete circumferential peripheral staining clearly evident. UP expression in bladder tumors of cows reported in this study is similar to the UP pattern of some urothelial tumors in humans. Although UP expression is remarkably changed in bladder carcinogenesis of cattle, the UP gene(s) remains expressed during cell transformation and tumor progression.
Chronic enzootic hematuria is a clinical syndrome of cattle caused by prolonged ingestion of some toxic substances from bracken fern (Pteridium spp.). Epithelial and mesenchymal tumors of the urinary bladder are frequently associated with this syndrome. Uroplakins (UPs) are major specific urothelial differentiation products of mammalian urothelium. 5,10 They are mainly produced in cells of the superficial layer of transitional cell epithelium, and they consist of four components: the 27-kd UP Ia, the 28-kd UP Ib, the 15-kd UP II, and the 47-kd UP III. 12,13 UPs are conserved through a broad range of mammalian species and are the major protein components of the asymmetric unit membrane (AUM). 11 The AUM forms the apical plaques of mammalian urothelium 1 and may act as a permeability barrier; it also may act as a means of stabilizing the apical bladder surface. 9 It has been suggested that the ability of the urothelium to terminally differentiate is gradually lost in neoplastic processes, so UPs may be a useful morphologic biomarker of differentiation in bladder carcinogenesis. 14
The aim of the present study was to investigate UP expression immunohistochemically in spontaneous tumors of the urothelium in cows grazing on bracken fern–infested land in southern Italy.
Materials and Methods
Samples of neoplastic urothelium were obtained at public slaughterhouses from twenty 4–16-year-old cows that had been suffering from chronic enzootic hematuria for several years. All the animals were known to have grazed on bracken-infested pasture because they were raised in mountain areas. Urothelium, uterus, vagina, fallopian tube, ovary, and mammary gland from five 4–14-year-old healthy cows were also examined as controls. These control animals came from lowlands, where bracken fern is virtually absent. Tissues were fixed in 10% buffered formalin and processed routinely for paraffin embedment. The avidin–biotin–peroxidase complex method was used for immunohistochemical analysis of UP. Immunofluorescence staining for confocal laser scanning microscopic investigations was also performed. Five-micrometer-thick sections were blocked for endogenous peroxidase in 0.3% H2O2 in methanol for 20 minutes. Antigen was enhanced by pretreating with microwave heating (twice for 5 minutes each at 750 W). Anti-AUM antibody reacting with all four UPs (Dr. T. T. Sun, Epithelial Biology Unit, Ronald Perelman Department of Dermatology. Department of Pharmacology and Kaplan Comprehensive Cancer Center. New York University School of Medicine, New York, NY) was made against highly purified bovine AUM and applied overnight at 4 C at a 1:2,000 dilution. Slides were washed three times, incubated with secondary antibody (biotinylated goat anti-rabbit IgG, Dako, Denmark) for 30 minutes, and then incubated with streptavidin-conjugated horseradish peroxidase (Dako). Color development was obtained with 5–20 minutes of diaminobenzidine treatment. Sections were counterstained with hematoxylin. For immunofluorescence staining, the same primary anti-AUM antibody was applied overnight at 4 C at a 1:300 dilution. Slides were washed three times and then incubated with TRITC-conjugated goat anti-rabbit antibody (Chemicon, Germany) for 2 hours at 1:100 dilution. Slides were then rinsed three times with phosphate-buffered saline (PBS) and incubated for 30 minutes in 20 µg/ml Hoechst 33342 (Sigma) for nuclear staining. The slides were mounted and analyzed with the inverted Zeiss LSM-510 confocal laser scanning microscope. The primary antibody was omitted and replaced with PBS in the corresponding negative control section. Based on architectural disorder and nuclear atypia, neoplasms were graded on a scale of I–III as recently suggested for human tumors. 6
Results
In control animals, the so-called umbrella cells were the only urothelial cells showing strong membranous immunoreactivity, with a continuous line clearly evident in the luminal aspect of superficial cells. In general, no UP expression was detectable in the cytoplasm of urothelial cells (Fig. 1).
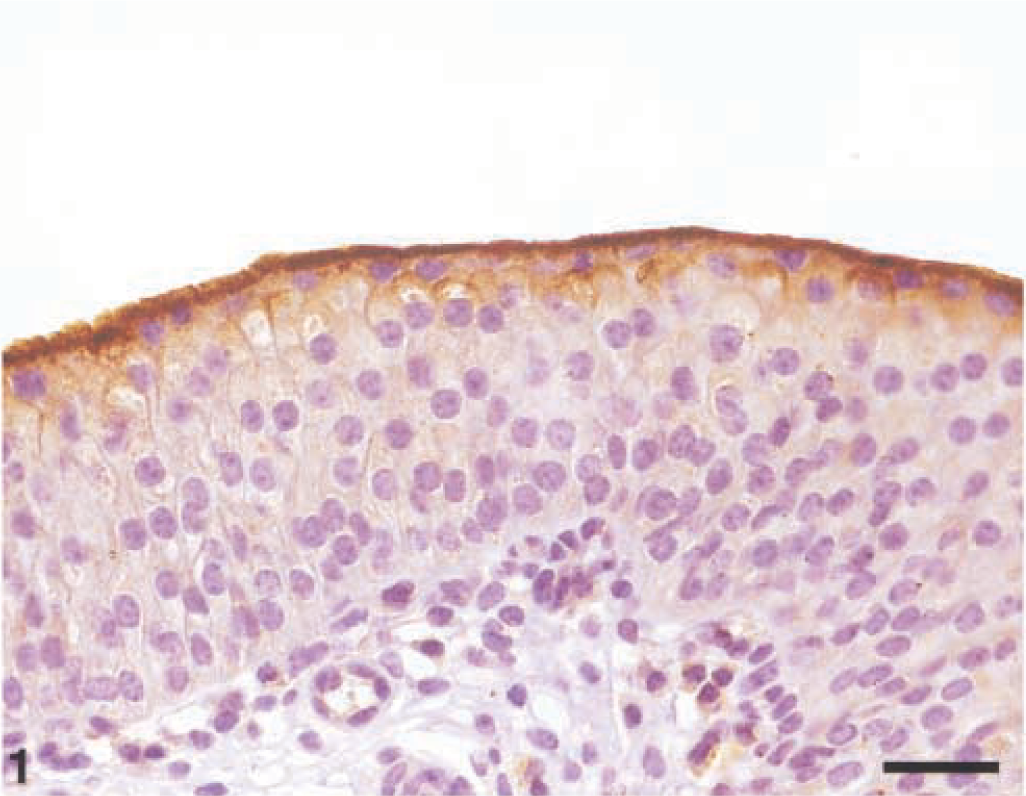
Normal urothelium; Cow. UP expression is clearly evident in the luminal cells only. Streptavidin–biotin–peroxidase. Mayer's hematoxylin counterstain. Bar = 50 µm.
In dysplastic urothelium and in papillomatous proliferations, UP expression was evident both as intercellular and luminal staining signals. UPs appeared to clearly define the borders of cells in deeper layers, whether or not these intermediate cells were adjacent to superficial ones. Occasionally, some intermediate cells showed an intense, diffuse intracytoplasmic reactivity for UPs (Fig. 2).
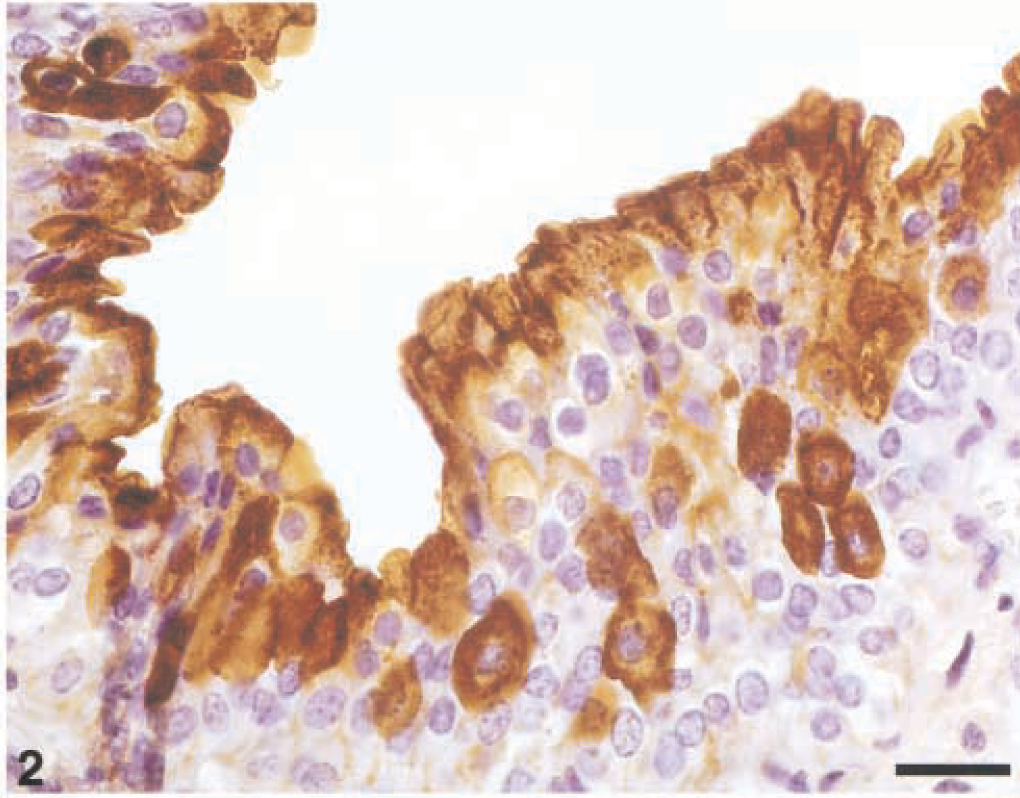
Dysplastic urothelium; Cow. UP expression is evident both as intercellular and luminal staining. A strong intracytoplasmic immunoreactivity is also manifested in some intermediate cells. Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain. Bar = 50 µm.
In transitional cell carcinomas, several patterns of UP expression were observed. In low-grade tumors (grade I). UPs occurred discontinuously in the luminal cells and, more markedly, in most of the cells underlying the superficial ones. In addition, UP expression was diffusely detected in the cytoplasm of some intermediate cells (Figs. 3, 4).
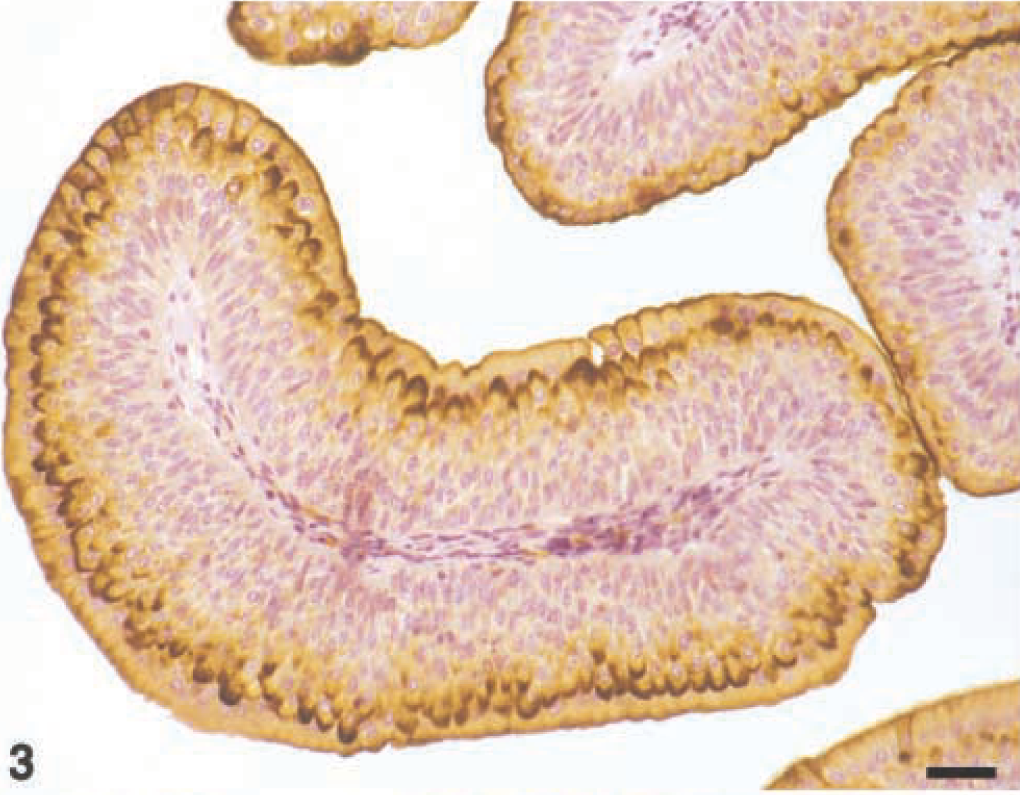
Grade I carcinoma; Cow. UP expression is manifested in luminal and intermediate cells. Some of the intermediate cells show a diffuse intracytoplasmic positivity. Streptavidin–biotin–peroxidase. Mayer's hematoxylin counterstain. Bar = 50 µm.
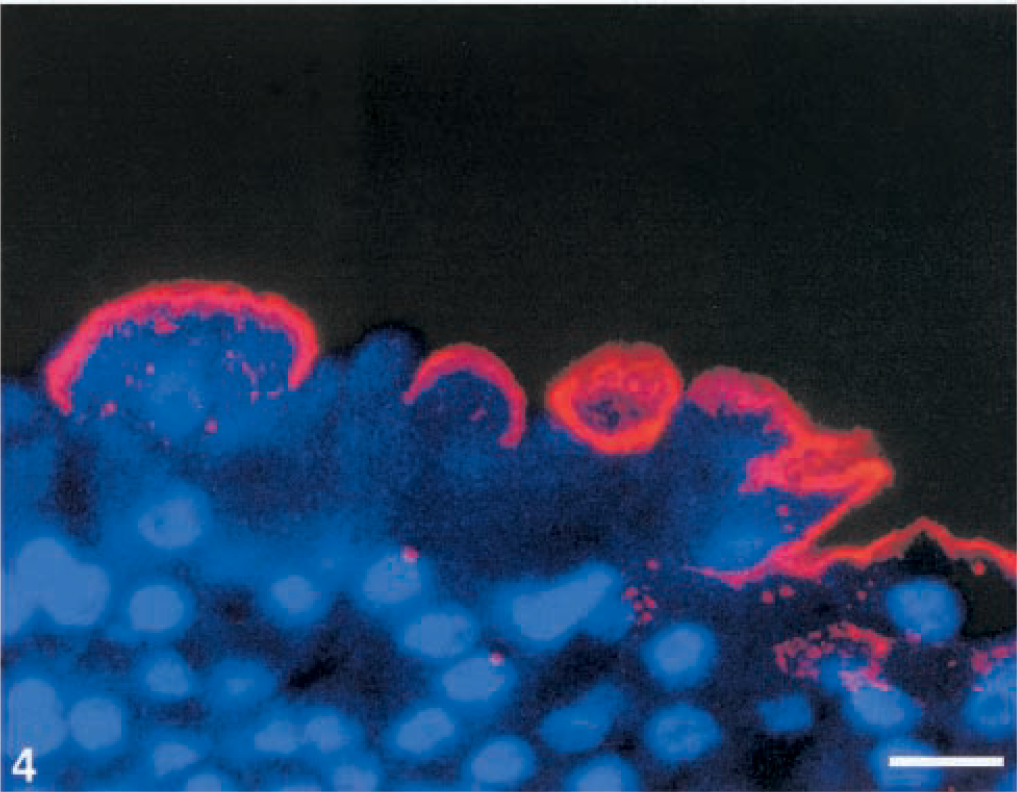
Grade I carcinoma; Cow. Discontinuity of luminal pattern is shown by confocal laser scanning microscopy. Indirect immunoflorescence, TRITC goat anti-rabbit, Hoechst 33342 counterstain. Bar = 50 µm.
In tumors exhibiting an intermediate degree of abnormality (grade II), the pattern for UPs was characterized by a remarkable decrease of luminal and intermediate cells showing UP expression. Furthermore, most intermediate cells focally expressed UPs on the membrane, so AUM appeared to occur as discontinuous plaques. Compared with low-grade carcinomas, intermediate cells showing intracytoplasmic positivity for UPs were irregularly clustered within areas of neoplastic proliferation (Fig. 5).
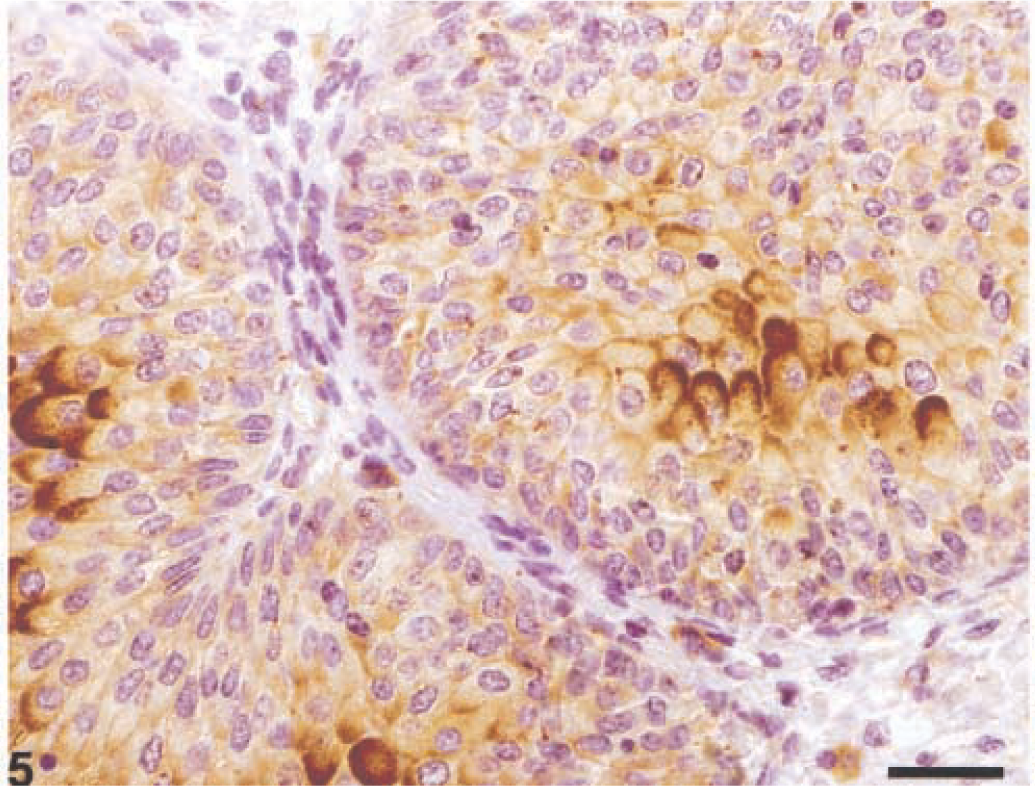
Grade II carcinoma; Cow. Irregular distribution of UPs. UP-positive cells are irregularly clustered within neoplastic proliferations. Streptavidin–biotin–peroxidase. Mayer's hematoxylin counterstain. Bar = 50 µm.
In high-grade carcinomas (grade III), immunoreactivity was distributed heterogeneously. There was severe loss of UP-positive luminal and intermediate cells. Focally, upper and lower cells showed strong membraneous immunoreactivity that marked and delimited single cells, with complete circumferential peripheral staining clearly evident. Both a diffuse and patchy cytoplasmic reactivity for UPs was also evident in some neoplastic cells (Fig. 6).
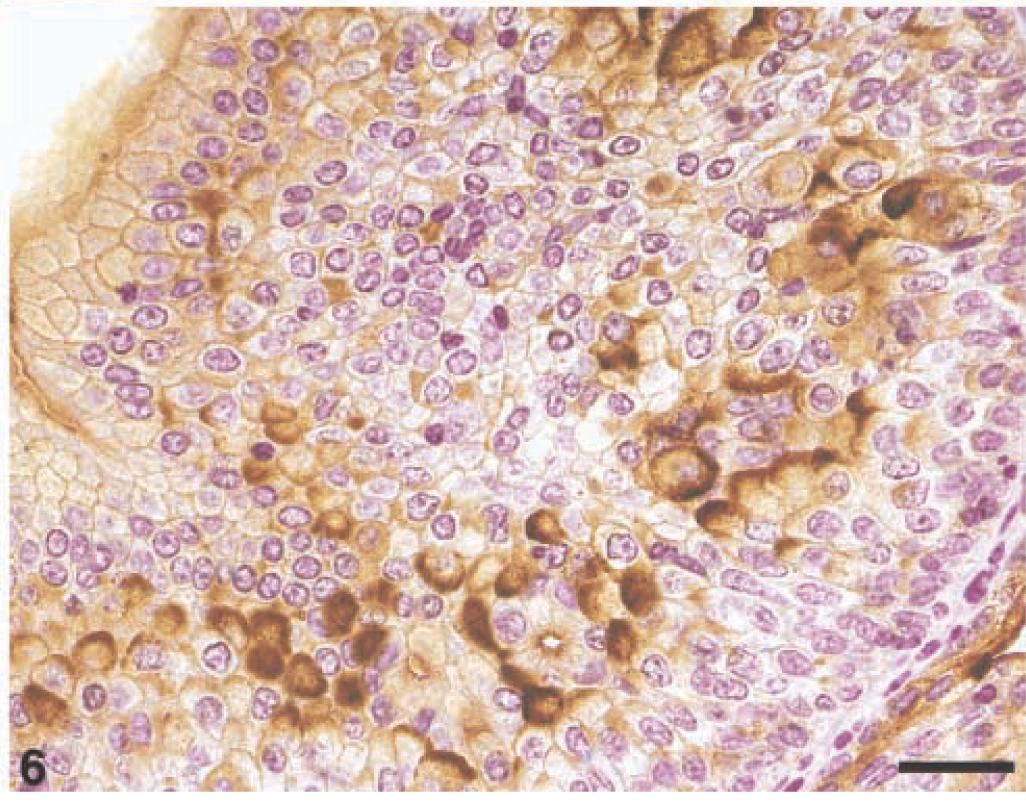
Grade III carcinoma, Cow. UP-positive cells are evident in deeper layers of the neoplastic urothelium. A diffuse and/or patchy intracytoplasmic immunoreactivity is also evident. Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain. Bar = 50 µm.
A negative reaction for UPs was recorded for all other bovine tissues, such as uterus, vagina, fallopian tube, ovary, and mammary gland.
Discussion
Recent investigations have demonstrated that UPs are excellent markers for detecting urothelial tumors 4 because UP expression is urothelium specific and differentiation dependent. 10 Although studies concerning the morphologic pattern of UPs in bladder carcinogenesis are becoming more numerous, many questions regarding the perturbation of urothelial differentiation in which UPs might be involved are still to be answered.
We investigated UP expression in urinary bladder tumors associated with chronic enzootic hematuria of cows grazing on pastures infested by bracken fern. This is the first study performed on spontaneous tumors of domestic animals. Some changes in UP distribution were characterized by an irregular discontinuity of immunoreactivity in the cells lining the lumen and by the presence of clustered intermediate cells with irregular membraneous staining and diffuse or patchy intracytoplasmic positivity. These changes may represent alterations in differentiation occurring during bladder carcinogenesis of cows. Similar alterations have been detected in chemically induced bladder cancers in rodents 7,8 and in urothelial tumors of humans. 3,5 Some atypical morphologic features, such as abnormal or bizarre microvilli, are known to occur in urothelial cells that are not fully differentiated, and these features are considered typical of the transformed luminal urothelial cells. 2 Discontinuity of UP expression at the luminal surface observed in this study can be another morphologic feature reflecting inability of luminal umbrella cells to terminally differentiate in these bovine tumors. Alterations of differentiation may be responsible for the presence of UP positive intermediate cells. Moreover, positivity of intermediate cells may be related to changes in the migration of the differentiating cells toward the surface. 8
Low-grade carcinomas show superficial staining of plasma membrane of luminal cells for UPs. A simultaneous peripheral pattern is characterized by a strong membrane staining. As reported in human tumors, 5 UPs abnormally expressed in the membrane of luminal and adjacent cells in bovine neoplasms probably do not form well-defined AUM plaques. However, no information is so far available on AUM structures in urothelial tumors of cows.
UP expression in bladder tumors of cows reported in this study is similar to UP expression patterns recently described in urothelial carcinomas in humans. 3,5 Membraneous positivity and intercellular, peripheral, and luminal staining signals have been reported to characterize UP staining patterns in the majority of urothelial carcinomas in both species. However, we did not investigate the relationship between various staining patterns and the potential clinical significance of such a relationship. UPs are not likely to be of value in assessing the biologic behavior or prognosis for a tumor because UPs have been detected in most of the urothelial carcinomas of cows regardless of their grade. Additional studies on the expression of UPs in urothelial carcinomas of animals are needed. UP expression is of interest for investigating differentation and/or dedifferentation of transitional epithelial cells in various species and for detecting urothelial cells in bovine metastases (G. Borzacchiello et al., unpublished). Cattle ingesting bracken fern might be a useful animal model for studying altered urothelial cell differentiation in neoplasms.
Footnotes
Acknowledgements
This study was supported in part by grants from the Consiglio Nazionale delle Ricerche (CNR) and the Italian Ministry of University and Scientific Research (MURST), Rome.
