Abstract
A case of urothelial carcinoma in situ of urinary bladder is reported in a 10-year-old cow naturally grazing on bracken-infested land. The cow suffered from enzootic hematuria for more than 5 years. The presence of bovine papillomavirus type 2 (BPV-2) DNA sequences was detected by polymerase chain reaction. The carcinoma in situ was characterized by the presence of anaplastic cells with amphophilic cytoplasm and pleomorphic nuclei containing granular, irregularly dispersed chromatin. Focal areas within the tumor contained large isolated and/or clustered cells. These cells had pale acidophilic cytoplasm, large nuclei with single or multiple nucleoli, and well-defined borders resembling Paget's cells. Immunohistochemically, all malignant cells were negative for vimentin and S-100 and positive for cytokeratins. In addition, normal and neoplastic cells expressed fragile histidine triad (FHIT) protein; surprisingly, some pagetoid cells did not. FHIT, the tumor suppressor gene at 3p14.2, encodes a protein of 147 amino acids (16.8 kd) with diadenosine triphosphate hydrolase activity and is a common target of deletions in human cancers of epithelial origin. Antibody to laminin detected a continuous epithelial basement membrane, thus clearly showing that neoplastic changes were limited to urothelial cells without invading stromal tissue. To our knowledge, this is the first report of an unusual pattern of spread of urothelial carcinoma in situ in a cow.
Enzootic hematuria of cattle is a severe syndrome caused by prolonged ingestion of toxic principles of bracken fern (Pteridium spp.). Urinary bladder tumors associated with this syndrome are common in 4- to 12-year-old cattle. 13 Recently, Ozkul and Aydin 11 detected 815 bladder tumors (14.6%) among 5,567 cattle from 14 provinces of the Black Sea region of Turkey in which enzootic hematuria of ruminants occurs endemically. Recent studies demonstrate a strong relationship between papillomaviruses and some clastogenic, mutagenic, and/or carcinogenic principles of bracken fern. DNA of bovine papillomavirus type 2 (BPV-2) was found in 69% of experimental and in 46% of naturally occurring bladder cancers. This high degree of association between bladder cancers and BPV-2 suggests that this virus plays a role in bladder oncogenesis. 6 It is worthwhile remembering that extracts of bovine cutaneous warts have been found to be responsible for cancer when injected into the urinary bladder of cows. BPV-2 is the virus inducing cutaneous warts of the head, neck, and shoulder of cattle. 7
Furthermore, epidemiological studies revealed a higher risk of esophageal as well as gastric cancers in people who consume milk of cows feeding on bracken, perhaps owing to the presence of ptaquiloside, the major bracken carcinogen component in milk. 1 14 We are investigating epidemiologic and etiopathogenic aspects of urinary bladder cancers associated with enzootic hematuria in cattle from the South of Italy, in which bracken fern is widely distributed. In this report, we describe a case of the pagetoid variant of urothelial carcinoma in situ (CIS) of the urinary bladder in a cow grazing on bracken-infested land; the cancer was associated with bovine papillomavirus 2 (BPV-2). Histologically, CIS may exist in the wide range of variant morphologies. The pagetoid variant of CIS is distinctive in that its histologic pattern is characterized by the presence of large cells with round to oval nuclei, prominent nucleoli, relatively low nuclear cytoplasmic ratios, fairly well-defined borders, and slightly acidophilic cytoplasm. 8 These cells resemble the Paget's neoplastic cells found in both mammary and extramammary Paget's disease. Pagetoid changes at extramammary sites are rare, and the most common location of extramammary Paget's disease (EPD) is the vulva, with the perianal region as the second most common site. EPD was also reported in other sites, including the perineum, groin, pubic area, thigh, axillae, buttocks, eyelids, external ear canal, scrotum, and penis. 8 Interestingly, almost all patients suffering from EPD of penis have had previous high-grade transitional cell carcinoma and CIS of the urinary bladder. 9
Carcinoma in situ is rarely found in cattle, 12 and to our knowledge, the pagetoid variant of urothelial carcinoma of the urinary bladder has not been previously reported.
A 10-year-old cow with clinical signs of severe hematuria was slaughtered at a public slaughterhouse. Clinical signs of hematuria were present since the animal was 4–5 years old. Macroscopically, multifocal erythematous areas and hemorrhagic foci were scattered over a thickened and uneven mucosa of bladder, the lumen of which contained sero-hemorrhagic fluid. Pedunculate intraluminal growths were also observed. Regional lymph nodes were enlarged.
Several samples of erythematous vesical mucosa and lymph nodes were fixed in 10% buffered formalin and embedded in paraffin; additional erythematous vesical samples were immediately frozen at −80 C. It is worthwhile noting that in an ongoing study about urinary bladder tumors in cattle, samples from all cases have routinely been processed for histologic as well as molecular investigations. Five-micron-thick sections were stained with hematoxylin and eosin. Silver methenamine and Movat's pentachrome stains were performed for epithelial basement membrane as well as periodic acid–Schiff (PAS) and mucicarmine for neutral and acid mucopolysaccharides.
For immunohistochemical studies, we used commercial polyclonal rabbit antibodies to cytokeratins (clone MNF 116, Dako, Denmark), vimentin (clone V9, Dako, Denmark), laminin (Sigma, USA), and S-100 protein (Biomeda, USA). Deparaffinized sections were blocked for endogenous peroxidase in 0.3% H2O2 in methanol for 20 minutes. Sections for laminin were pretreated with 1% acetic acid in distilled water containing 0.04% pepsin. In the remaining slides, antigen enhancement was performed by pretreating with microwave heating (twice for 5 minutes each at 750 W). Slides were washed three times, then incubated with secondary antibody (biotinylated goat anti-rabbit IgG, Dako, Denmark) for 30 minutes followed by incubation with streptavidin-conjugated horseradish peroxidase (Dako, Denmark). Color development was obtained with 5–20 minutes of diaminobenzidine treatment. Sections were counterstained with hematoxylin. The primary antibody was omitted and replaced by phosphate-buffered saline (PBS) in the corresponding negative control sections.
The expression of protein encoded by the fragile histidine triad (FHIT) gene was detected immunohistochemically and by immunoblotting analysis using a primary rabbit polyclonal anti–Glutathione S-Transferase-FHIT antiserum (generously provided by Dr. Kay Huebner, Kimmel Cancer Center, Jefferson Medical College, Philadelphia, PA). The cross-reactivity of this antiserum was previously demonstrated by western Blot analysis carried out on Madin Darby bovine kidney (MDBK) cell lysates. Briefly, MDBK cells and frozen neoplastic tissues were homogenized in lysis buffer (10 mM Tris HCl, pH 7.4; 150 mM NaCl; 1% NP-40; 1 mM EDTA; 1 mM DTT; 0.5 mM PMSF; 10 µg/ml aprotinin; 5 µg/ml leupeptin) and clarified by centrifugation. Proteins were quantified by Bradford assay, fractionated in 15% SDS-PAGE gel, and blotted onto PVDF membrane (Biorad, Italy). The blots were incubated with primary rabbit polyclonal anti–GST-FHIT antiserum at 1:2,000 dilution. A clear band at 17 kd was detected in MDBK cell lysates and tumor protein extract. Furthermore, a small fragment of frozen neoplastic tissue was digested by Proteinase-K in the lysis buffer (50 mM KCl; 10 mM Tris HCl, pH 8.3; 2.5 mM MgCl2; 100 µg/ml gelatin; 0.45% NP-40; and 0.45% Tween-20) to recover DNA. Ten microlitres of samples were amplified by PCR using one unit of Taq polymerase (Finenzymes, Espoo, Finland) in the manufacturer buffer with 1.5 mM MgCl2. Amplification reaction was carried out in a Thermal Cycler (Perkin Elmer, USA) for 30 cycles with an annealing temperature of 50 C. Primers detecting the BPV-2 sequences were used according to Otten et al. 10 : upstream primer 5′-TTGCTGCAATGCAACTGCTG-3′ (bp 3,919–4,225), downstream primer 5′-TCATAGGCACTGGCACGTT-3′ (bp 4,207–4,225). In each experiment, a blank sample consisting of reaction mixture without DNA and a positive sample consisting of BPV-2–cloned DNA (kindly provided by Dr. M. S. Campo) were included. A band corresponding to the amplified sequences of BPV-2 was detected in the cancer sample (Fig. 1). To validate the PCR result, the amplified products were sequenced. Our molecular investigation is consistent with the statement of Campo and co-workers 6 that first suggested the strong association of BPV-2 and bracken fern with bladder cancer in cattle.
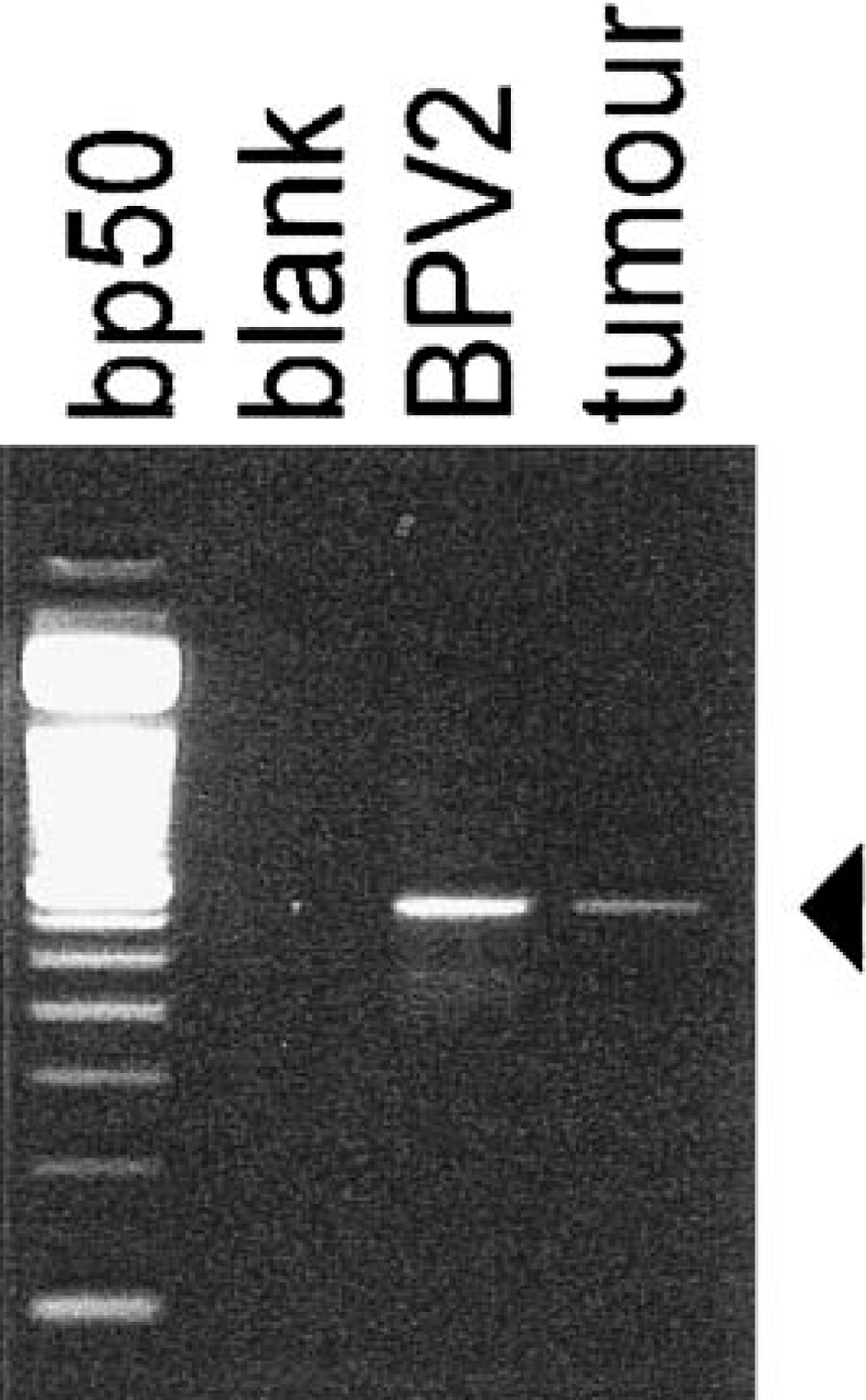
Detection of BPV-2 DNA sequences; cow. The arrowhead indicates the position of the 306-bp amplified products. The bp50 lane is a DNA ladder consisting of 16 blunt-ended fragments ranging in length from 50 to 800 bp (Life-Technology, Italy).
The in situ cancer was characterized by the presence of nests of round cancer cells intermingled with normal epithelium (Fig. 2). Anaplastic cells, present in all layers, contained amphophilic cytoplasm and pleomorphic nuclei with granular, irregularly dispersed chromatin and many large nucleoli. Cell loss from superficial layers made tumor cells more evident. Numerous mitotic figures were also seen. Some neoplastic cells were PAS-positive. Focal neoplastic areas showed large isolated and/or clustered cells with pale acidophilic cytoplasm, large nuclei with single or multiple nucleoli, and well-defined borders resembling Paget's cells (Figs. 3, 4). Immunohistochemically, all neoplastic cells were negative for vimentin and S-100 protein and positive for cytokeratins. Neoplastic cells, including pagetoid cells, expressed FHIT protein less intensely than normal epithelium. The superficial “umbrella” cells of the normal urothelium, and in neoplastic samples as well as some pagetoid cells, did not express FHIT protein. The latter were also PAS- and mucicarmine-negative (Table 1). Tumor changes were limited to the epithelial lining without stromal invasion. Histologic (Movat's pentachrome and silver methenamine) stains as well as immunohistochemical stain for laminin showed the epithelial basement membrane to be continuous (Fig. 5). A marked hyperplastic lymphadenitis was also observed.
Histochemical and immunohistochemical features of pagetoid cells and neoplastic urothelium.∗

CK = cytokeratin; Vim = vimentin; FHIT = fragile histidine triad; PAS = periodic acid-Schiff; + = positive; w = weak; f = focal; - = negative.
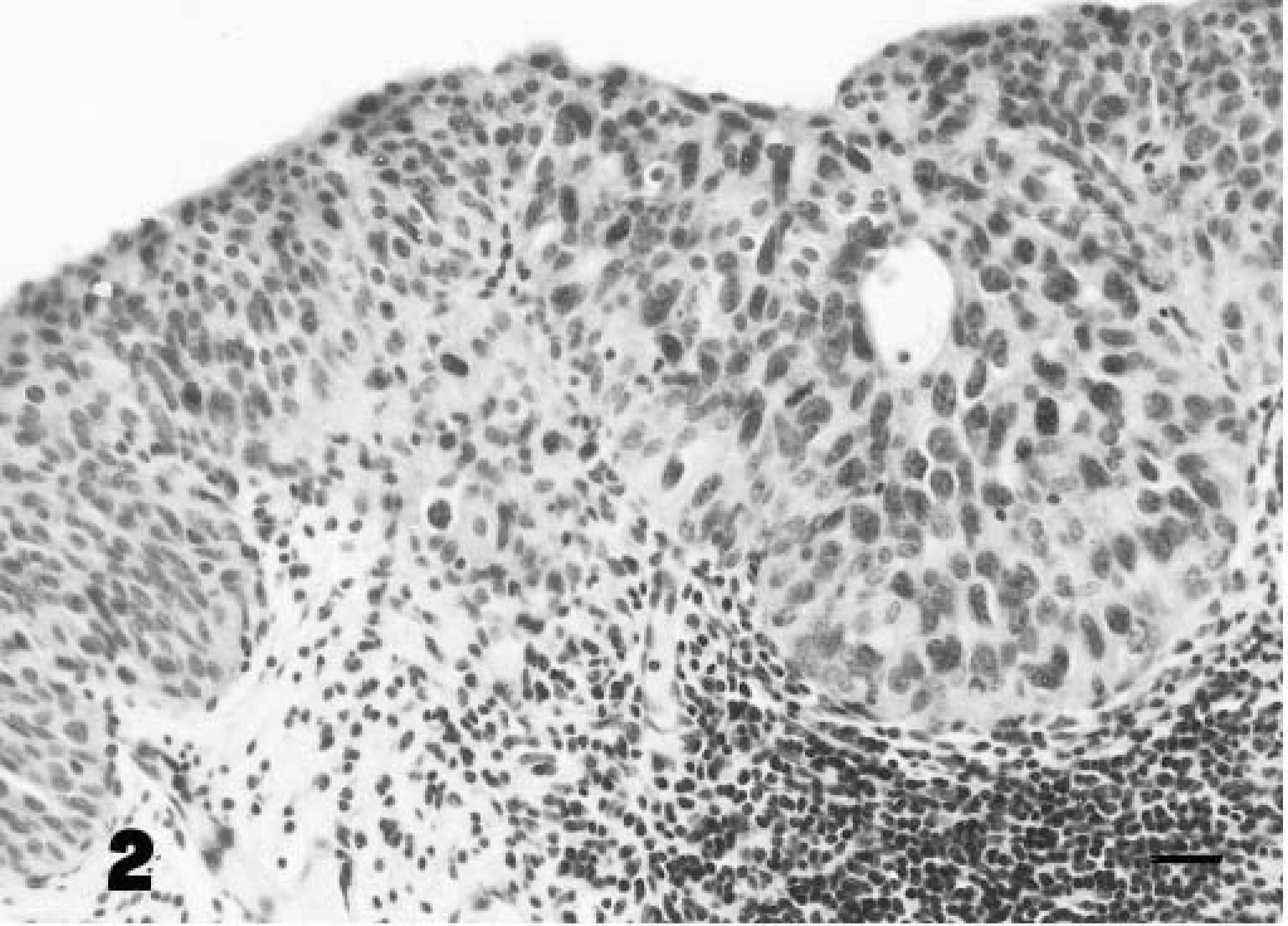
A nest of urothelial neoplastic cells overlying an area of marked inflammation of the lamina propria. Hematoxylin and eosin. Bar = 25 µm.
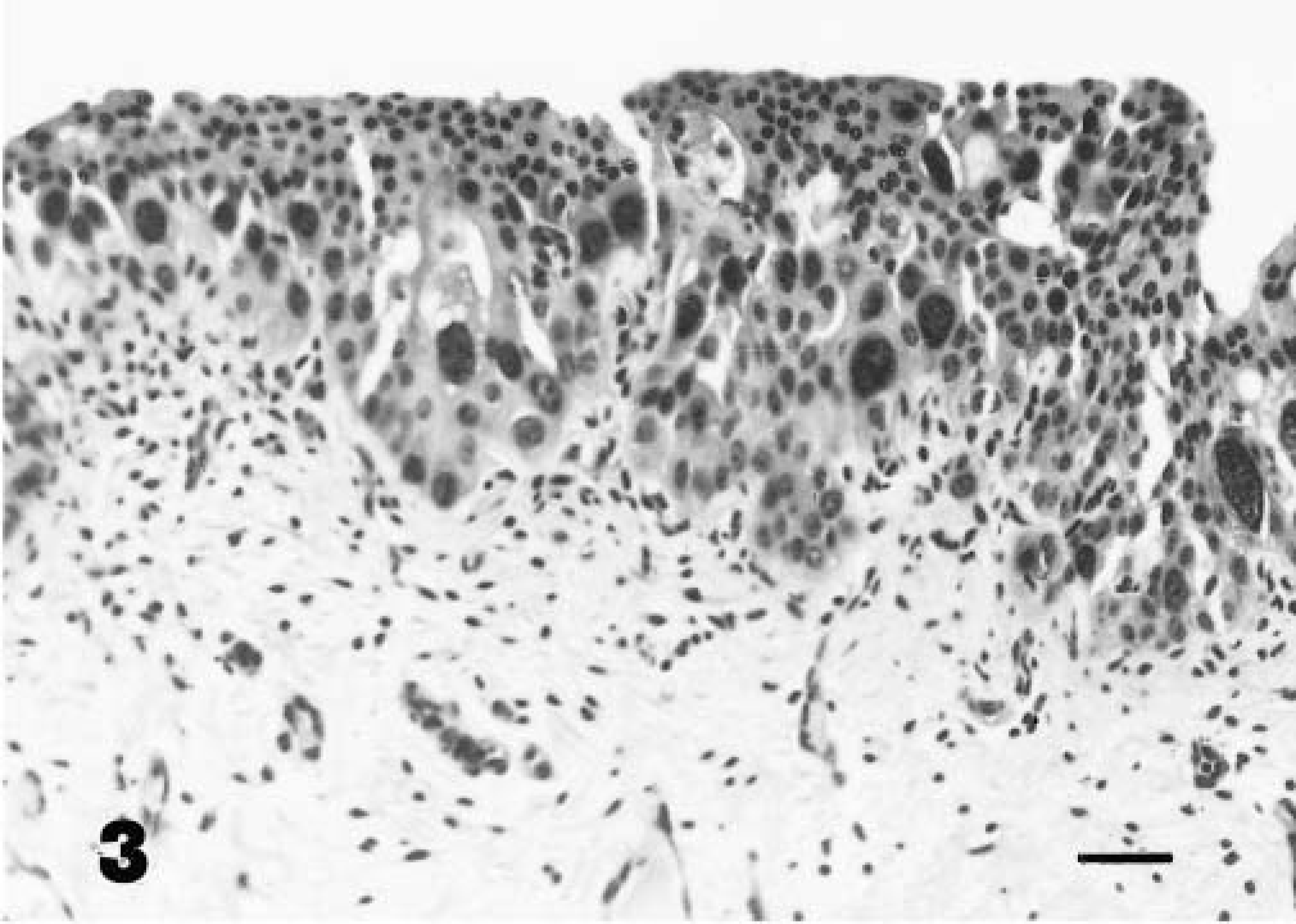
Focal area of neoplastic cells with bizarre, large nuclei. Hematoxylin and eosin. Bar = 25 µm.
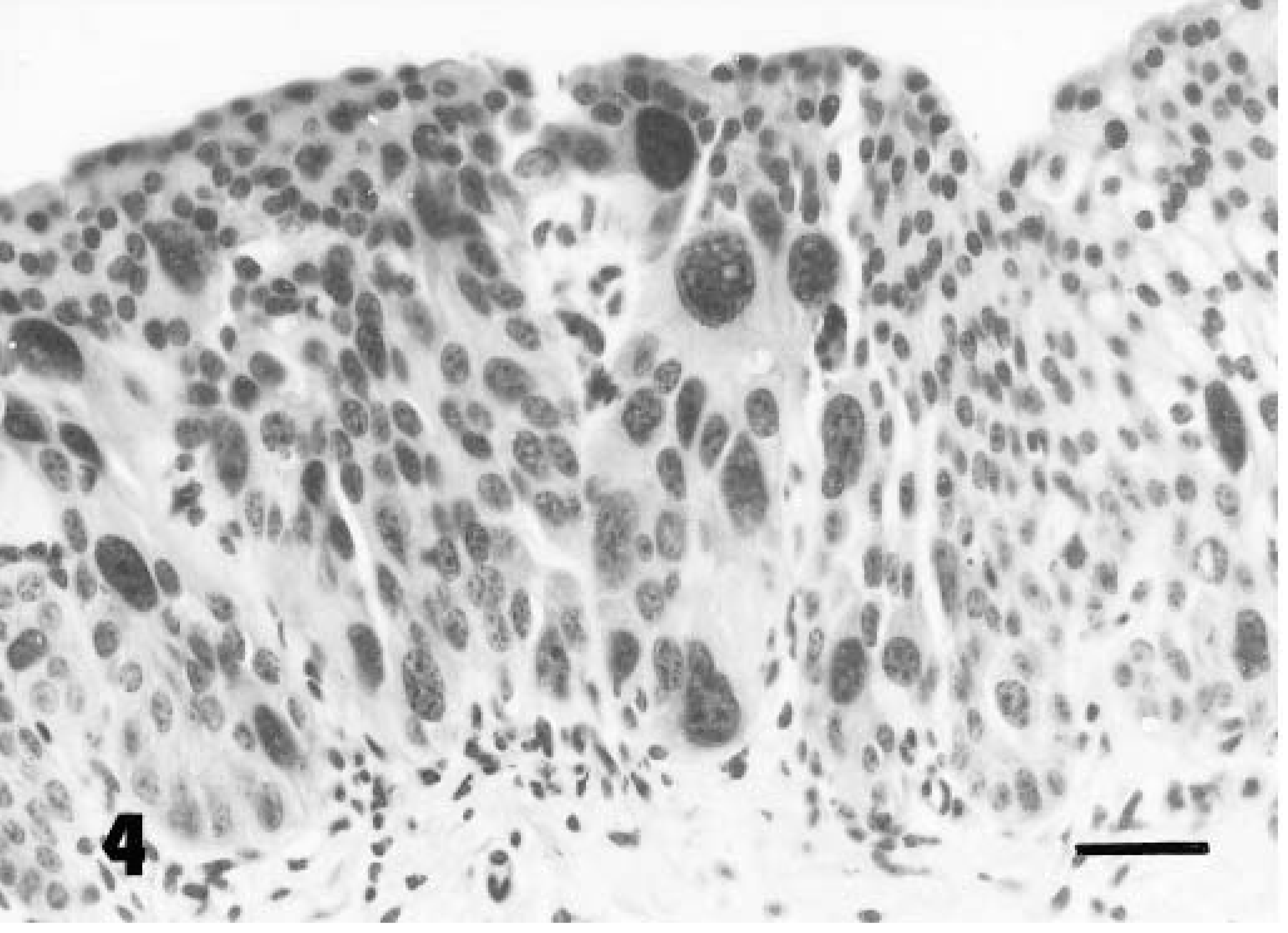
Neoplastic cells with pagetoid features at higher magnification. Hematoxylin and eosin. Bar = 25 µm.
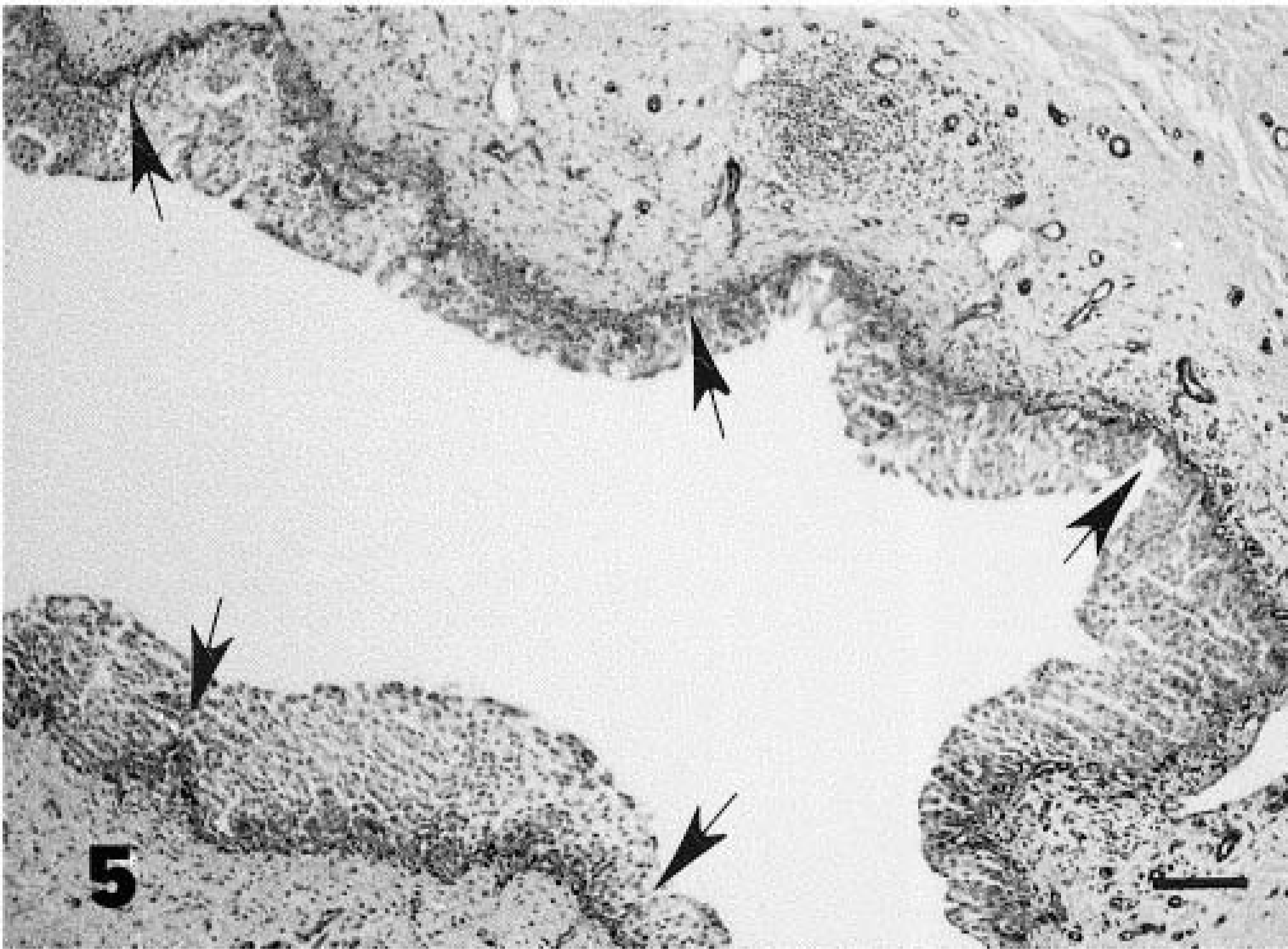
Note the integrity of the epithelial basement membrane (arrows). Streptavidin–biotin stain. Bar = 50 µm.
In humans, pagetoid CIS is very rare, accounting for about 0.6% of all carcinomas of the urinary bladder. 2 The histogenesis of pagetoid changes is unknown but may be due to a nonspecific local reaction of neoplastic cells to injury or loss of E-caderin expression. 2 9 In our case, some of the pagetoid cells did not express FHIT protein. The tumor suppressor gene FHIT belongs to the histidine triad superfamily. In man, this gene is located at chromosomal region 3p14.2 and encodes a protein of 147 amino acids (16.8 kd) with diadenosine triphosphate hydrolase activity, whose tumor-suppressor activity may be associated with involvement of the FHIT/Ap3A protein complex in cytokine signaling pathway(s) controlling cell proliferation. 4 The FHIT gene, a highly conserved gene homologous to a group of genes identified in eukaryocytes, encompasses the FRA3B fragile site and is a common target of deletions in human epithelial cancers. 15 It is known that the gene is inactivated by deletions in many primary tumors of man. 5 It has been suggested that loss of FHIT expression may be as important in the development of bladder cancer as it is for other neoplasms caused by environmental carcinogens. As matter of fact, it has recently been shown for the first time that FHIT is inactivated in the majority of transitional cell carcinomas of the human urinary bladder, and FHIT inactivation is a late event in neoplastic progression. 3
To our knowledge, there have been no previous reports about FHIT protein expression in pagetoid CIS. If our data are corroborated by further studies, it could be suggested that the cells with pagetoid features can be considered as more undifferentiated neoplastic cells, and this phenotype might be typical of aggressive cells. It has been suggested that the more undifferentiated neoplastic cells are, the greater the reduction and/or loss of FHIT protein expression. 15 In addition, the phenotype of neoplastic cells not expressing FHIT protein may characterize the morphologic pattern of tumors, the biological behavior of which shows a remarkable aggressiveness. 16
Footnotes
Acknowledgements
This study was supported in part by grants of Consiglio Nazionale delle Ricerche and the Italian Ministry of University and Scientific Research (MURST), Rome (Italy).
