Abstract
Bovine urinary bladder tumors occur frequently in animals suffering from chronic enzootic hematuria because of prolonged ingestion of bracken fern (Pteridium spp.). Cyclooxygenase (COX) genes (COX-1 and COX-2) are known to be involved in the carcinogenesis of human and some animal urothelial tumors. The aim of the present study was to investigate COX-1 and COX-2 expression by immunohistochemical methods in 20 bovine urothelial carcinomas collected at public slaughterhouses from cows that had been suffering from chronic enzootic hematuria. COX-1 immunostaining was identified intracytoplasmically in normal urothelium and in 15 of 20 neoplastic specimens. COX-1 immunosignal in the tumor cells was either absent or weak. COX-2 was also expressed intracytoplasmically in 17 of 20 urothelial carcinomas. Moderate to intense COX-2 labeling was detected in both noninvasive and invasive urothelial carcinomas. Coexpression of both enzyme isoforms was also revealed by confocal laser scanning microscopic investigations. This study indicates that COX-2 is overexpressed in naturally occurring urothelial carcinomas of cows.
Keywords
Urinary bladder tumors are known to occur rarely in animals. However, cows grazing on bracken fern–infested lands and suffering from chronic enzootic hematuria frequently develop urinary bladder tumors. Bracken fern (Pteridium spp.) has been proved to cause cancer naturally in animals. 2 , 16 The pathogenesis of these neoplasms lies in the mutagenic, clastogenic, and carcinogenic principles contained in bracken fern. Among these, ptaquiloside (PT), a norsesquiterpenoid glycoside, is considered the major carcinogen. 17
Cyclooxygenase (COX) is a rate-limiting enzyme for prostaglandin (PG) synthesis from arachidonic acid. 23 Two COX genes have been cloned (COX-1 and COX-2) that share > 60% identity at the amino acid level and have similar enzymatic activities. 19 , 20 COX-1 is considered to be involved in the production of PGs that modulate normal physiologic functions in several organs, whereas COX-2 is not detectable in normal tissues but is induced by cytokines, growth factors, oncogenes, and tumor promoters. 4 , 18 COX-2 overexpression is reported in many epithelial neoplasms, including human, rat, and canine urothelial neoplasms. 1 , 5 , 6 , 8
The purpose of this study was to investigate COX expression in spontaneous bovine urothelial carcinomas by immunohistochemical and immunofluorescence methods.
Materials and Methods
Animals and tumor samples
Samples of neoplastic urothelium were obtained at public slaughterhouses from twenty 4- to 18-year-old cows. All the animals were known to have come from mountain areas where bracken fern is widely distributed (Fig. 1). Local veterinarians referred to us that the examined cows had clinically been suffering from chronic enzootic hematuria for several years.
Map of Italy. Circles represent bracken fern–covered areas in southern Italy. Points denote the mountain districts where the cows came from
Urothelium from five 4- to 15-year-old apparently healthy cows was also examined. These control animals came from lowlands where bracken is virtually absent. Samples were fixed in 10% neutral buffered formalin, mounted in Killik frozen section medium (Bioptica, Milan, Italy), and quickly frozen in isopentane cooled to its freezing point by liquid nitrogen.
Formalin-fixed specimens were processed routinely, paraffin embedded, cut to 4-µm-thick sections, and stained with hematoxylin and eosin (HE).
Histologic diagnosis was based on the WHO Histological Typing of Urinary Bladder Tumours. 10
Immunohistochemical studies
Specimens were sectioned at 4 µm, deparaffinized, and blocked for endogenous peroxidase in 0.3% H2O2 in methanol for 20 minutes. Antigen enhancement was performed by pretreating with microwave heating in a citrate buffer, pH 6.00 (twice for 5 minutes each at 750 W). Immunostaining was performed with monoclonal mouse anti–COX-1 or polyclonal rabbit anti–COX-2 antibodies (Cayman Chemical Co., Ann Arbor, MI, USA) applied overnight at room temperature at 1 : 500 and 1 : 200 dilutions, respectively.
Sections were washed three times with phosphate-buffered saline (PBS) and incubated for 30 minutes with secondary antibody composed of a mixture of biotinylated anti-mouse, anti-rabbit, and anti-goat immunoglobulins (labeled streptavidin–biotin [LSAB] Kit; DakoCytomation, Glostrop Denmark). Sections were washed three times with PBS and then incubated with streptavidin conjugated to horseradish peroxidase (DakoCytomation).
Color development was obtained by treatment with diaminobenzidine (DakoCytomation) for 5–20 minutes. Sections were counterstained with Mayer's hematoxylin.
In the corresponding negative control section, the monoclonal or polyclonal antibody (primary antibody) was either omitted or replaced with normal mouse serum or normal rabbit serum, respectively.
Immunofluorescence, two-color immunofluorescence, and confocal laser scanning microscopy
Frozen sections (10 µm) were cut on a cryostat (Microm HM 505 E; Zeiss, Göttingen, Germany) and fixed with acetone at 4 C for 10 minutes.
Sections were washed in PBS, and the two primary antibodies for COX-1 and COX-2 (see above) were applied overnight at room temperature at a dilution of 1 : 10. Sections were washed three times with PBS. COX-2 antibody-labeled slides were incubated overnight at room temperature with a fluorescein isothiocyanate (FITC)–conjugated secondary antibody (Chemicon, Hofheim, Germany) at 1 : 100 dilution, and COX-1 slides were incubated with a tetramethylrhoadamineisothiocyanate (TRITC)-conjugated secondary antibody (Chemicon) at 1 : 100 dilution. The slides were washed again with PBS and then mounted with an aqueous medium (Sigma, Milan, Italy). A confocal laser scanning microscope (LSM 510, Zeiss) was used to evaluate the slides. COX-2 antibody bound to FITC was irradiated at 488 nm and detected with a 505- to 560-nm band pass filter. COX-1 antibody bound to TRITC was irradiated at 543 nm and detected with a 560-nm long pass filter.
Two-channel frame-by-frame multitracking was used for detection to avoid “cross-talk” signals. The different frames were scanned separately, with appropriate installation of the optical path for excitation and emission of each scan according to the manufacturer's instructions.
Scoring of immunoreactivity
The intensity of COX-1 and COX-2 immunosignal in tumors was determined in a blind study by two observers.
The immunosignal in each specimen was scored on a four-tiered scale of 0 to +++ (Table 1) as follows: 0 = absent or very weak immunosignal, + = weak immunosignal, ++ = moderate immunosignal, and +++ = strong immunosignal.
COX-1 and COX-2 expression in bovine urinary bladder tumors.∗
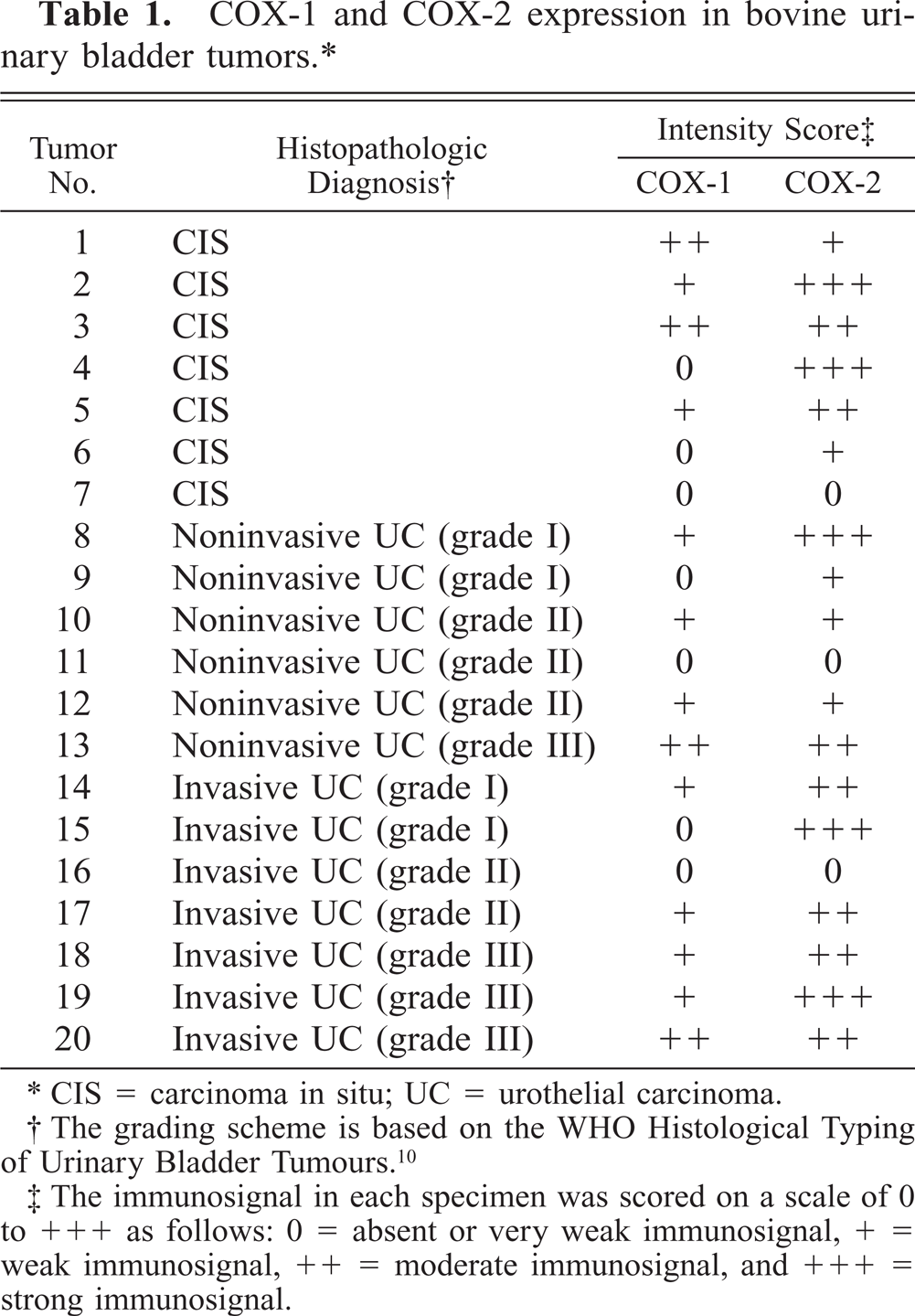
∗ CIS = carcinoma in situ; UC = urothelial carcinoma.
† The grading scheme is based on the WHO Histological Typing of Urinary Bladder Tumours.10
‡ The immunosignal in each specimen was scored on a scale of 0 to +++ as follows: 0 = absent or very weak immunosignal, + = weak immunosignal, ++ = moderate immunosignal, and +++ = strong immunosignal.
Results
COX-1 expression and cytoplasmic immunosignal was strong in normal urothelium and in smooth muscle cells of the bladder wall (Fig. 2). Fifteen of 20 (75%) urothelial carcinomas showed COX-1 immunostaining signal (Table 1), but most showed only a faint cytoplasmic immunosignal.
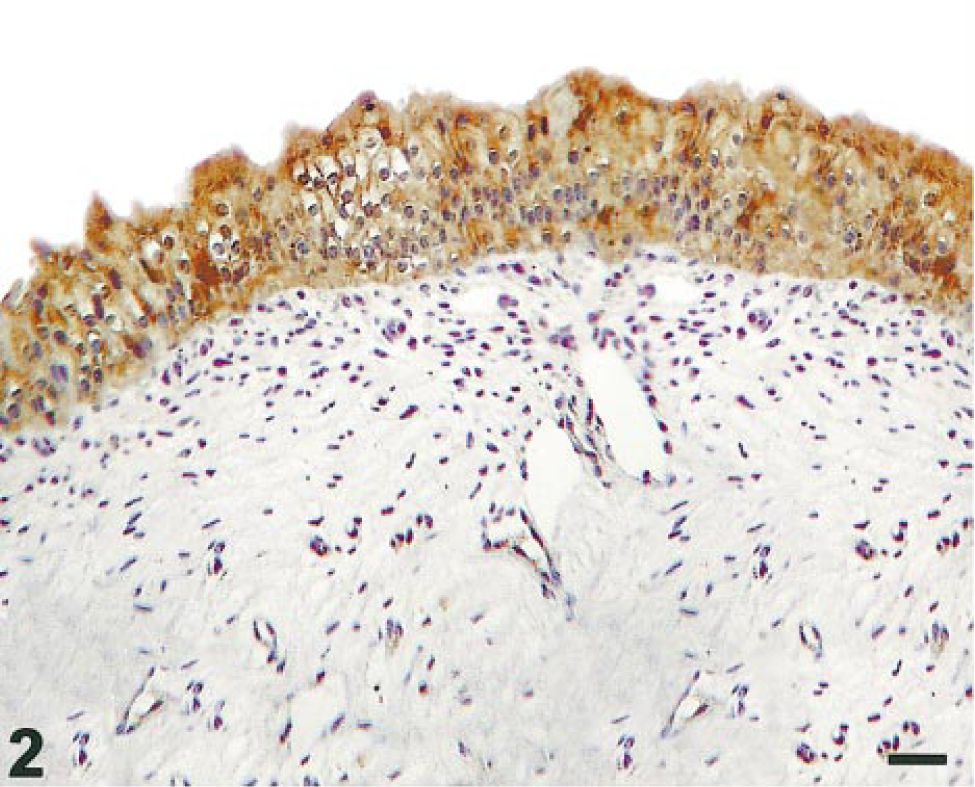
Normal urothelium; cow. Strong cytoplasmic COX-1 staining is present throughout the mucosa. Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain. Bar = 10 µm.
Seventeen of 20 neoplastic specimens (85%) revealed diffuse COX-2 immunostaining. Carcinoma in situ samples showed strong COX-2 immunoreactivity with diffuse granular cytoplasmic staining (Fig. 3). COX-2 staining intensity varied in both papillary and infiltrating urothelial carcinomas. In six of seven invasive urothelial tumors, clusters of infiltrating cells expressed a moderate to strong immunosignal (Fig. 4). COX-2 immunostaining was also detected in some mesenchymal cells such as those of the endothelium. A very low-intensity COX-2 immunosignal was also seen in samples of normal urothelium.
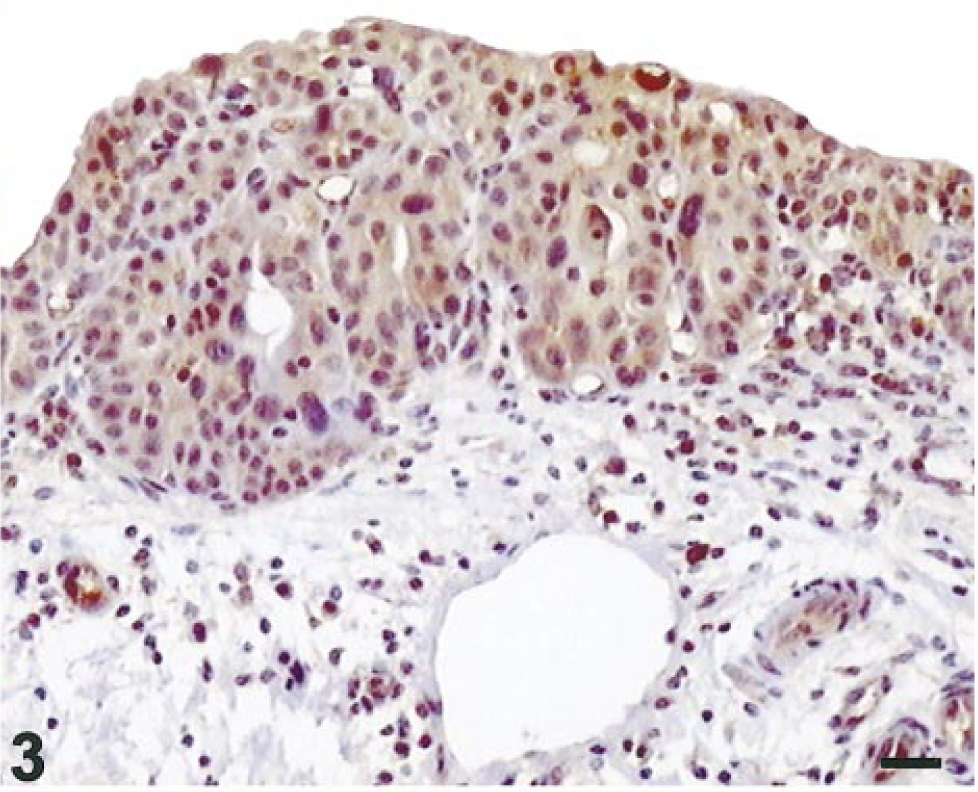
Urothelial carcinoma in situ; cow. Strong homogeneous cytoplasmic COX-2 staining in all neoplastic urothelial cells. Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain. Bar = 10 µm.
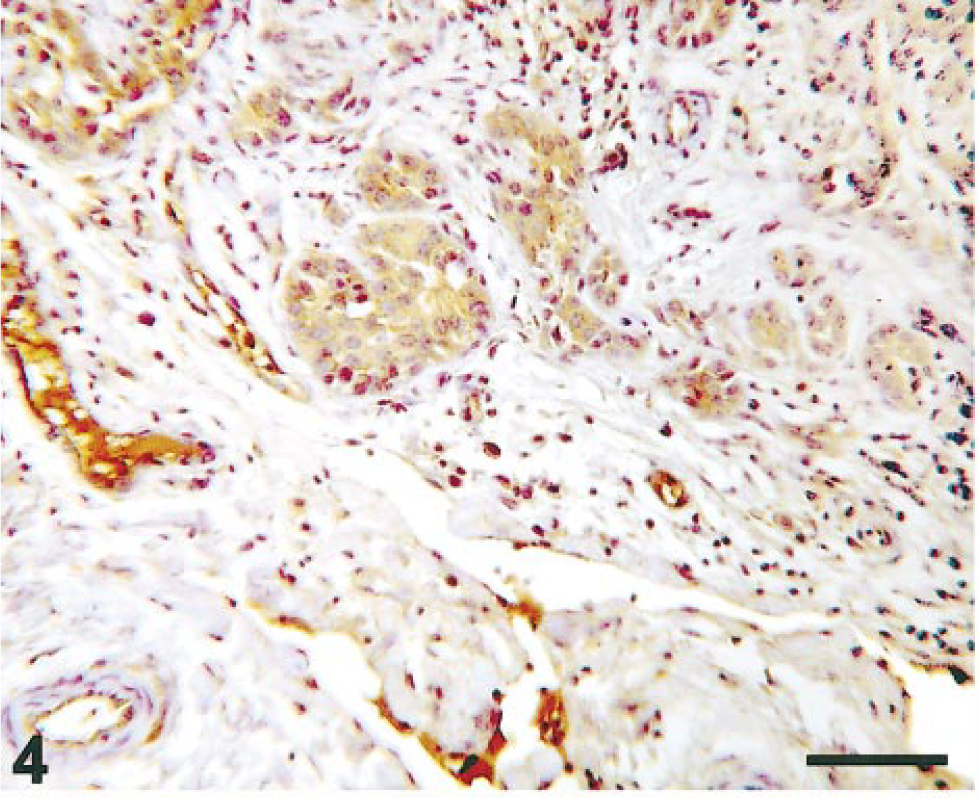
Invasive urothelial carcinoma; cow. COX-2–immunostained invasive epithelial cells scattered in the muscular layer of the urinary bladder. Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain. Bar = 10 µm.
Immunofluorescence showed a staining pattern superimposable on the immunohistochemical pattern. COX-1 and COX-2 were both expressed intracytoplasmically in neoplastic urothelial cells. In the same neoplastic cells, simultaneous immunosignals for COX-1 (red) and COX-2 (green) produced a yellow color, which indicates the coexpression of both enzyme isoforms (Fig. 5).
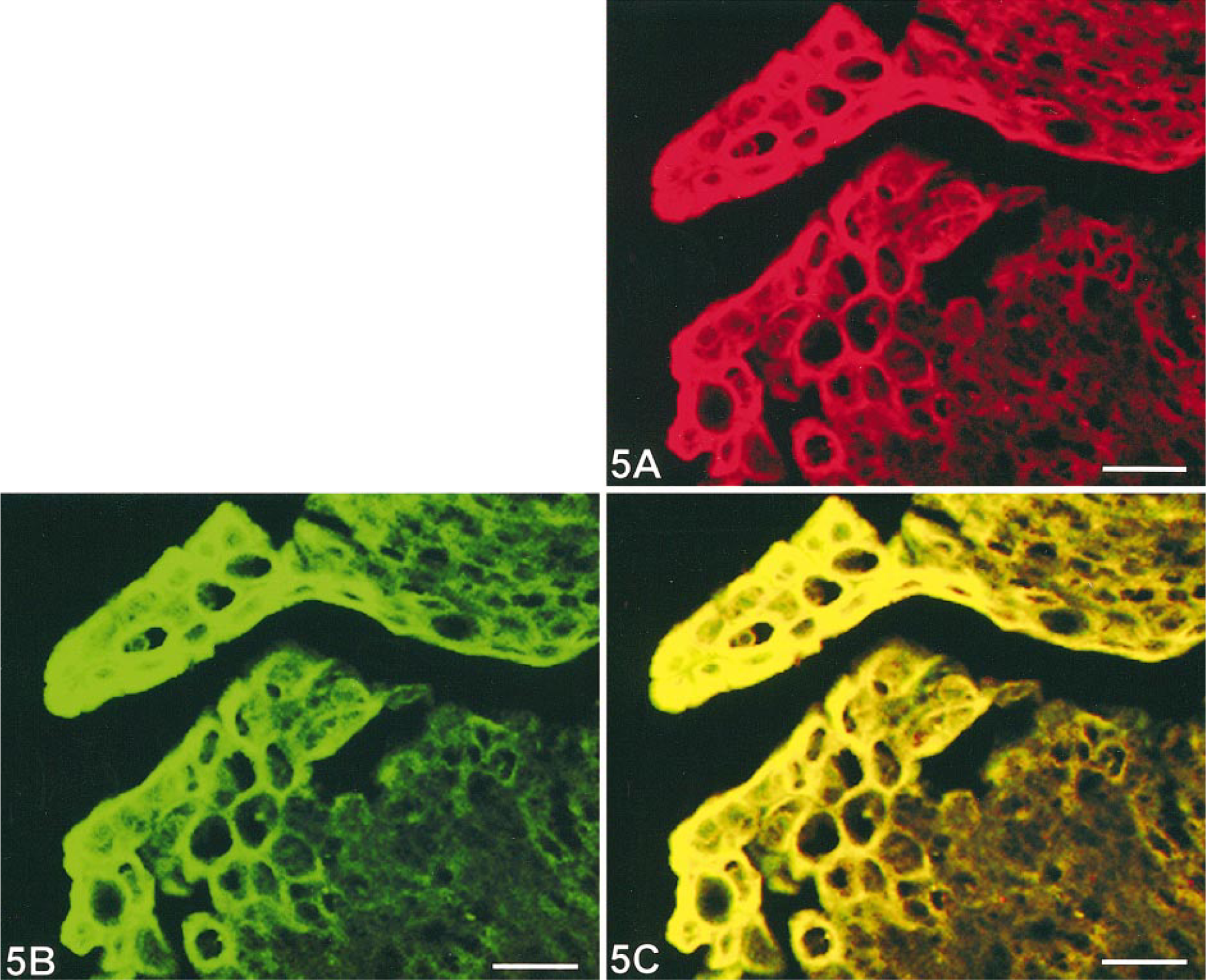
Papillary urothelial carcinoma; cow. Two-color immunofluorescence, cryostat section. Fig. 5A. COX-1 cytoplasmic immunostaining corresponds to red TRITC immunofluorescence. Fig. 5B. COX-2 cytoplasmic immunostaining corresponds to green FITC immunofluorescence. Fig. 5C. Yellow represents the cytoplasmic coexpression of both COX-1 and COX-2 in neoplastic urothelial cells. Bar = 10 µm
Discussion
Recent reports in human tumors indicate that urothelial cancer expresses both COX enzyme isoforms. 1 , 9 In this regard, our confocal laser scanning microscopic examination allowed us to detect coexpression of COX-1 and COX-2 in bovine urothelial neoplastic cells. Immunohistochemical patterns observed here are similar to those recently described in spontaneous human and canine urothelial tumors and chemically induced rodent urinary bladder carcinomas, 5 , 6 suggesting that COX expression is independent of the cause of carcinogenesis.
Furthermore, it has been demonstrated that COX-2 is overexpressed in human, rat, and canine urinary bladder carcinomas. 1 , 5 , 6 , 8 Our immunohistochemical study has shown that COX-2 is also overexpressed in bovine urothelial carcinomas of the urinary bladder. The role of COX-2 expression in the development of bovine cancer is still an open question.
Previous studies have demonstrated that H-ras–transformed cancer cells overexpress COX-2. 3 , 14 It is worthwhile noting that PT, a major carcinogen in bracken fern, alkylates DNA after activation and forms PT–DNA adducts. It has been shown that bracken fern–fed calves harbored PT–DNA adducts, which resulted in H-ras activation through mutation in codon 61, thus suggesting that activation of H-ras is an early event in PT-induced carcinogenesis. 12 , 13
We have been able to immunohistochemically detect H-ras in bovine urothelial carcinoma (not shown); H-ras activation might explain COX-2 overexpression in urothelial carcinoma in cows that grazed on bracken fern–infested lands in southern Italy.
Although recent studies have shown that COX-2 inhibitors might be chemopreventive agents of urinary bladder carcinogenesis in rodents and pet dogs, 7 , 11 the function of COX-2 during carcinogenesis is still uncertain. COX-2–derived PGE2 synthesis may contribute to tumor cell resistance to apoptosis and hence to tumor development and progression. 21 Further, COX-2 could increase tumor invasiveness, with activation of the matrix metalloproteinase MMP-2. 22
In the present study, COX-2 expression was found in both urothelial carcinoma in situ and invasive carcinoma, in agreement with comparable human studies, 15 suggesting a possible role of COX-2 in the early development of bovine urinary bladder carcinoma.
The present study suggests that COX-2 plays a role in spontaneous bovine urothelial carcinogenesis; however, further studies are needed to better understand COX-2 function in animal carcinogenesis.
In summary, animal models of bladder cancer are needed to evaluate the functional role of COX-2 inhibitors. 9 As far as COX-2 expression in urothelial carcinomas is concerned, our study shows that bovine naturally occurring bladder cancer shares similarities with the human counterpart, thus suggesting that it could serve as an animal model for further investigation of the role of COX-2 in bladder cancer and the mechanisms by which COX-2 inhibitors may exert antitumoral effects.
Footnotes
Acknowledgements
This study was supported in part by grants from the Italian Ministry of University and Scientific Research, Rome, Italy. We thank Drs. E. Massari and G. Toma from the Azienda Unità Sanitaria Locale no. 2 for their collaboration.
