Abstract
We report a case of multiple glomus tumors associated with bovine papillomavirus type 2 (BPV-2) infection in the urinary bladder of a 13-year-old cow suffering from severe chronic enzootic hematuria. Macroscopically, multiple submucosal reddish nodules were seen swelling the vesical mucosa. Histologically, neoplastic proliferation was characterized by the presence of numerous blood vessels. These were lined by normal endothelial cells surrounded by round epithelioid cells with central nuclei, prominent nucleoli, acidophilic cytoplasm, and well-defined cytoplasmic borders. Tumor cells were distributed around open vascular lumina and in perivascular spaces. They were immunohistochemically positive for actin and vimentin and negative for cytokeratins, desmin, and factor VIII-related antigen. On the basis of these findings, this tumor was diagnosed as glomus tumor, a neoplasm not previously reported in cattle and exceedingly rare in animals. BPV-2 DNA was amplified from the formalin-fixed, paraffin-processed tissue specimens obtained by laser capture microdissection. This report widens the spectrum of mesenchymal tumors of the bovine urinary bladder. Finally, the microscopic pattern of tumor described here shares striking morphologic and immunohistochemical similarities with the angiomatous form of glomus tumor known to occur in man.
Glomus tumor, also known as glomangioma, is a mesenchymal neoplasm that originates in the glomus body. 12 This is a neuromyoarterial body that functions as a specialized form of arteriovenous shunt composed of an afferent arteriole and efferent venule with multiple communications. Glomus cells have properties similar to smooth muscle cells, accounting for their contractility, and are thought to be related to thermoregulatory function. Glomus bodies can be found throughout the body but are most highly concentrated in the extremities. 15 Glomus tumor is very rare, accounting for less than 2% of the soft tissue tumours in man. 8 In veterinary medicine, glomus tumors have very rarely been reported. Few cases are known to occur in dogs. 7,9–11,14 Recently, a glomus tumor has also been described in a cat. 17 Digits seem to be the preferential anatomic site of the tumor both in dogs and cats. In humans, the classic location of the glomus tumor is the subungual region, but the tumor can occur elsewhere; 11 however, its occurrence in visceral organs is very rare and particularly so in the urinary tract. 1,16 The only case of glomus tumor arising in the urinary bladder has been recently described in human medicine. 13 We document a case of multiple glomus tumor of the urinary bladder in a cow associated with bovine papillomavirus type 2 (BPV-2) infection. To our knowledge, glomus tumor has not been reported in cattle previously.
A 13-year-old cow, which had been grazing on bracken-infested lands and showed clinical signs of severe hematuria, was slaughtered at a public slaughterhouse and, after anatomopathologic examination, was found fit for human consumption. Hematuria had been observed since the animal was 7 to 8 years old. Macroscopically, multiple submucosal reddish nodules were seen bulging from the vesical mucosa. The mandatory postmortem examination failed to detect any neoplastic proliferation in the regional lymph nodes as well as in distant organs. Several samples of these nodules were harvested and fixed in 10% buffered formalin and embedded in paraffin. Five-micron-thick sections were prepared; some of them were stained with hematoxylin and eosin and others utilized for immunohistochemical examination using a streptavidin-biotin-peroxidase system. Histologically, the tumor consisted of variable-size blood vessels lined by a single layer of normal endothelial cells. A solid proliferation of round epithelioid cells was present focally around the open vascular lumina and in the perivascular space as well. The proliferation was located to subepithelial connective tissue, expanding to the more superficial layers of the muscularis propria. Tumor cells showed central nuclei, prominent nucleoli, acidophilic cytoplasm, and well-defined cell borders (Fig. 1). A dense infiltrate composed mainly of lymphocytes and plasma cells was evident in the lamina propria; histiocytes and some occasional polymorphonuclear cells were also seen; mast cells were common. Atypical cells or mitoses were not seen. For immunohistochemistry, the following antibodies were used: commercial polyclonal rabbit antibodies against cytokeratins (dilution 1 : 50. Dako, Denmark) and vimentin (dilution 1 : 50. Dako), monoclonal mouse antibodies against actin (dilution 1 : 50. Santa Cruz Biotechnology, Santa Cruz, CA, USA), desmin (dilution 1 : 50. Novocastra Laboratories Ltd., Newcastle, UK), and factor VIII–related antigen (dilution 1 : 100. Dako). Briefly, deparaffinized sections were blocked for endogenous peroxidase in 0.3% H2O2 in methanol for 20 minutes. Antigen enhancement was performed by preheating in a microwave (twice for 5 minutes each at 750 W). A commercial kit (Dako, LSABK680, Dako Cytomation, Burlingame, CA, USA) containing peroxidase-conjugated streptavidin and a mixture of biotinylated antirabbit–antimouse immunoglobulins was used. Color development was obtained with 5–20 minutes of diaminobenzidine treatment. Sections were counterstained with hematoxylin. Control sections were treated with phosphate-buffered saline instead of primary antibodies. Tumor cells showed reactivity for actin and vimentin (Fig. 2) and negative staining for cytokeratins, desmin, and factor VIII–related antigen. The tumor cell morphology and distribution as well as the immunohistochemical features were consistent with the diagnosis of a benign form of glomus tumor (glomangioma). It has been suggested that a peculiar key to the diagnosis of this kind of tumor is the presence of glomus cells around and in between the blood vessels. 13 Additionally, immunohistochemical studies of glomus tumors at different anatomic sites have shown consistent immunoreactivity for actin. 1
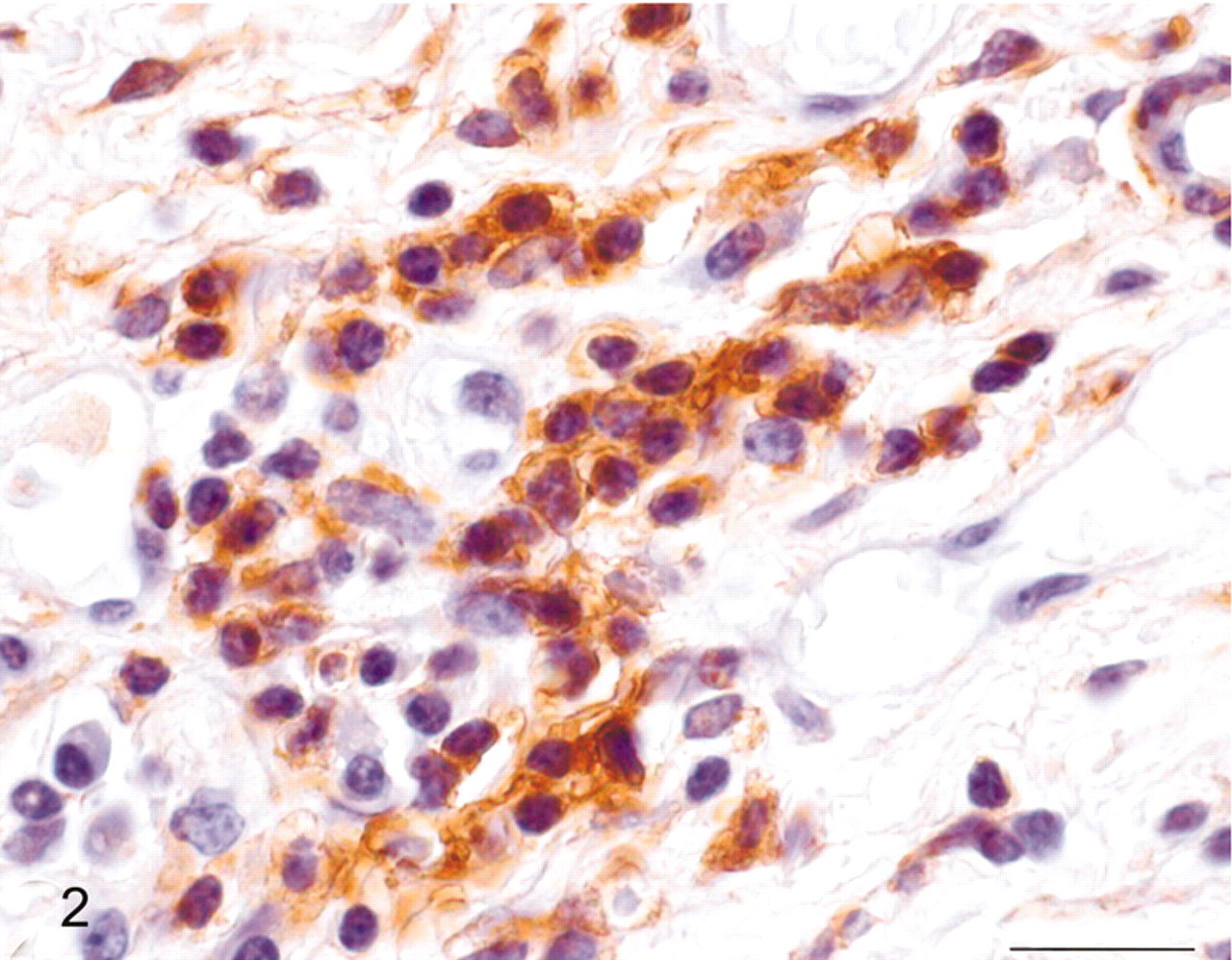
Urinary bladder; cow No. 10515. Positive immunohistochemical staining for actin of the round tumor cells. Streptavidin/biotin/peroxidase complex system, hematoxylin counterstain. Bar = 20 μm.
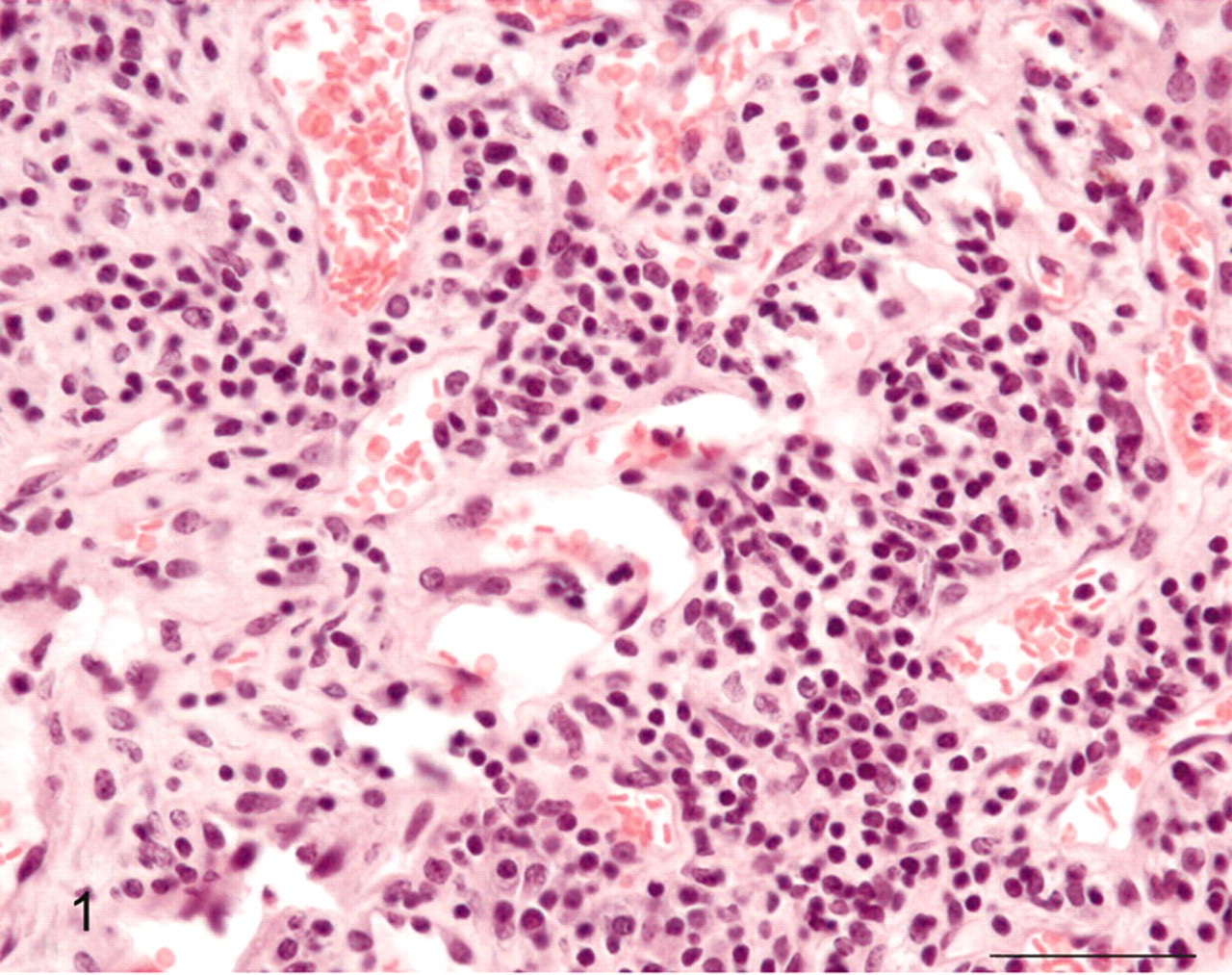
Urinary bladder; cow No. 10515. Glomus tumor consisting of blood vessels lined by normal endothelial cells and surrounded by round cells. HE. Bar = 25 μm.
Viral analysis was performed, because infection of the bladder mucosa by BPV-2 is very frequent, and BPV-2 is known to be casually involved in bladder cancer. 2,5 Tumor cells were microdissected from sections prepared from paraffin-processed tissue specimens using laser-capture microdissection (LMD laser microdissection, Leica Microsystems, Wetziar, Germany). DNA was extracted using the DNeasy Tissue Kit (Qiagen, Valencia, CA, USA) according to the manufacturer's protocol. The sample was lysed using proteinase K. Lysate was loaded onto DNeasy spin columns. After 2 washings pure DNA was eluted in low salt buffer. For the detection of BPV-2 DNA from the microdissected samples, specific primers for the E5 region were designed by Beacon Designer 2.0 software (Premier Biosoft International, Palo Alto, CA, USA). The forward primer BPV-2NS (5′-TACTGTTTCTGCTGCTATTT-3′) and the reverse primer BPV-2NAS (5′-ACAAATCAAATCCACATAATAGTA-3′) are known to amplify a fragment of 125 bp from 3943 to 4067 of the BPV-2 genome. In the experiment a blank sample consisting of reaction mixture without DNA and a positive sample consisting of cloned BPV-2 DNA were included. BPV-2 DNA was amplified from the tumor samples (Fig. 3). This single case of glomus tumor does not allow any ultimate conclusion about the oncogenic role of BPV-2 in this tumor type. However, BPV-2 might play a role also in glomus tumor. We have recently shown that E5 and E7 oncoproteins of BPV-2 are also expressed in mesenchymal tumors of urinary bladder in cattle grazing on pastures containing a large amount of bracken fern. 4
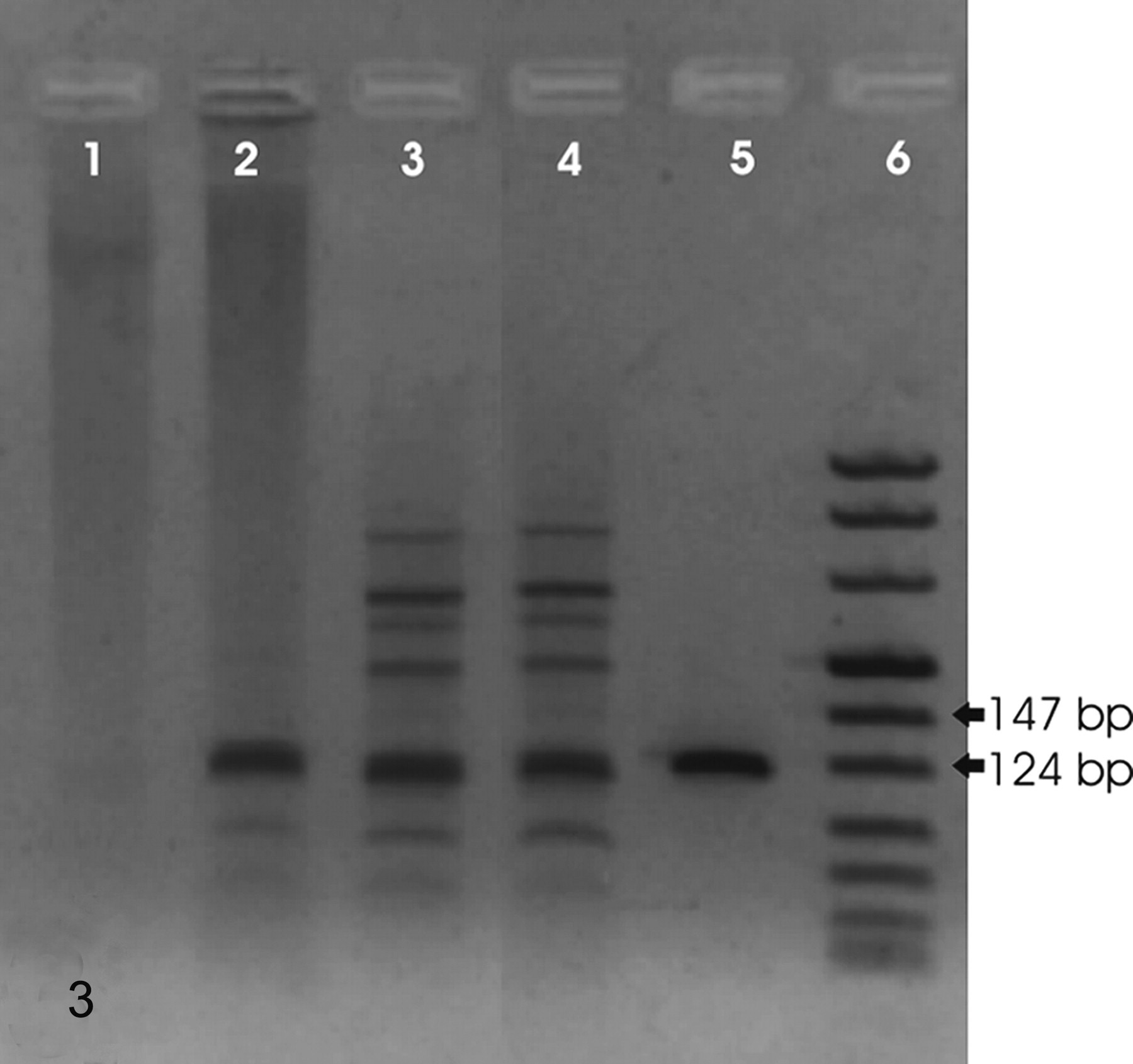
Urinary bladder; cow No. 10515. Detection of BPV-2 DNA sequences. Amplicons of 125 bp are evident in 3 tumor samples. Lane 1: negative control; lanes 2–4: samples from pathologic bladder; lane 5: positive control; lane 6: ladder.
The glomus tumor is composed of a mixture of glomus cells, blood vessels, and smooth muscle cells. Depending on the prevalent component, 3 microscopic patterns of the tumor have been described in man: solid, angiomatous, and myxoid. 12 The angiomatous variant is formed by groups or clusters of glomus cells around and between the normal blood vessels. The microscopic pattern of tumor described here shares striking morphologic and immunohistochemical similarities with the angiomatous form of glomus tumor known to occur in man. Hemangiomas could be considered in the differential diagnosis of glomus tumor, especially when the latter is characterized by numerous variable-size blood vessels. However, hemangiomas are composed of blood vessels, the endothelial cells of which are known to be factor VIII–related antigen positive, and their microscopic pattern is never characterized by predominant perivascular proliferation of smooth muscle cells. 1 For these reasons, hemangiomas should not be confused with glomus tumors.
The glomus tumor appears to be another histotype of tumor associated with bovine papillomavirus infection. This is further evidence that the wide spectrum of histologic neoplastic lesions of the urinary bladder observed in man can be found also in cattle and that human and bovine bladder tumors share striking morphologic features.
In agreement with Campo, 6 we suggest that cattle with papillomavirus infection may provide an in vivo model for human papillomavirus infection, inflammation, and carcinogenesis. Human papillomaviruses have emerged as one of the most important groups of direct and indirect infectious carcinogens. The cattle model may allow us to gain new insights in poorly understood aspect of human and animal carcinogenesis. 3
Footnotes
Acknowledgements
This work was supported in part by grants from the Italian Ministry of University and Scientific Research, the Assessorato alla Sanità of Regione Basilicata and Regione Campania, and the Assessorato all'Agricoltura, Foreste, Forestazione, Caccia e Pesca of Regione Calabria. We wish to thank Drs. G. Marino and G. Di Domenico of Azienda Sanitaria Locale SA/3 for their technical help.
