Abstract
The granulomatous lesions in bovine paratuberculosis have been classified into two types, i.e., the lepromatous type and the tuberculoid type. To clarify the immunopathologic mechanisms at the site of infection, we compared inflammatory cytokine gene expression between the two types of lesions. Samples were obtained from noninfected control cows (n =5) and naturally infected cows (n =7) that were diagnosed by enzyme-linked immunosorbent assay (ELISA) and fecal culture test. Although none of the infected cows showed clinical signs, tuberculoid lesions were observed in five cows (tuberculoid group) and lepromatous lesions in two cows (lepromatous group). Among the cytokines examined by reverse transcription-polymerase chain reaction (RT-PCR), Th2-type cytokines interleukin-4 (IL-4) and IL-10, and Th1-type cytokine IL-2 were expressed more significantly in the lepromatous group than in the tuberculoid (P < 0.01) and noninfected groups (P < 0.05). No statistical differences were observed in the expression of interferon-gamma, IL-1 beta, TNF-alpha, and GM-CSF among lepromatous, tuberculoid, and noninfected groups. Expression of proinflammatory cytokine IL-12 mRNA, however, did not differ among the three groups; IL-18 was expressed at lower levels in the lepromatous group than in the tuberculoid group and the noninfected group (P < 0.0001). Moreover, the number of cells in which IL-18 mRNAs were detected by in situ hybridization was markedly decreased in the lepromatous group. These results indicate that the formation of lepromatous-type lesions or tuberculoid-type lesions may be influenced by alterations in Th1/Th2-type cytokine production and that IL-18 may play an important role in a Th1-to-Th2 switch in paratuberculosis.
Paratuberculosis (Johne's disease) is characterized clinically by chronic diarrhea and progressive emaciation and with lesions of granulomatous enteritis and lymphadenitis caused by infection with Mycobacterium avium subsp. paratuberculosis (MAP). 9, 11 Granulomatous lesions observed in the disease have been classified into two types, the tuberculoid type and the lepromatous type, similar to hyperreactive form and anergic form in human leprosy. 5, 12 Tuberculoid-type lesions, which occur in the early stages of paratuberculosis, consist of small numbers of epithelioid cells and many lymphocytes, plasma cells, eosinophils, and macrophages, with none or only scant numbers of MAP (paucibacillary). It has been suggested that tuberculoid-type lesions are associated with strong cell-mediated immune responses on which resistance to paratuberculosis is dependent. 6, 8 Control of the disease in the early stages, therefore, is associated with CD4+ T-helper cell type 1 (Th1) response via the release of Th1-type inflammatory cytokines, interleukin-2 (IL-2), tumor necrosis factor (TNF), interferon-γ (IFN-γ), and IFN-β, which direct cell-mediated immunity. 21 Among them, IFN-γ plays a pivotal role in activation of macrophages and their subsequent ability to kill MAP. 19, 27, 30 In contrast, lepromatous-type lesions, which occur most often in the terminal stage of the disease, are composed mainly of macrophages and epithelioid cells bearing large numbers of mycobacteria (pluribacillary). 5, 6, 12 Lepromatous-type lesions are associated with strong humoral immune responses in conjunction with weak cell-mediated immunity resulting in progression of the disease, 6, 20 with a reduction in Th1 response and the induction of CD4+ T-helper cell-type 2 (Th2) induced humoral immunity by the anti-inflammatory cytokines IL-4, IL-5, IL-6, and IL-10. 22 Recently, mounting evidence has suggested that two proinflammatory cytokines, IL-12 and IL-18, play an important role in the induction of IFN-γ production in T cells, 23, 25, 26, 40 B cells, 40 NK cells, 3, 25 and dendritic cells. 15 Interestingly, it has been reported that mycobacterial infection of macrophages results in progressive suppression of IL-12 production in vitro and in vivo. 33, 34 In humans, IL-18 production in response to mycobacterial antigens correlates strongly with IFN-γ production and protective immunity. 31, 32 However, the relationship between the secretion of proinflammatory cytokines and types of granulomatous lesions, as well as the mechanism of a Th1-to-Th2 switch, remain to be elucidated during asymptomatic stages of bovine paratuberculosis. Work in this field has previously focused on the use of a mouse model and on in vitro experiments. In the present study, we compared immunoregulatory cytokine mRNA expression between the two types of granulomatous lesions in cows naturally infected with MAP to clarify the regulatory mechanisms of Th1/Th2 balance at the site of MAP infection.
Materials and Methods
Animals
Seven naturally infected Holstein and Japanese Black cows (cow Nos. 1–7) and five noninfected Hol-stein control cows (noninfected group: cow Nos. 8–12) were used in this study. All cows were diagnosed by a combination of an absorbed ELISA method using a commercial kit (Johnelisa, Kyoritsu Seiyaku Co., Tokyo, Japan) and fecal culture test modified by Yokomizo et al. 38, 39 All of the infected cows were obtained from the local Livestock Hygiene Service Center, and noninfected cows were raised at our institute with no history either of paratuberculosis or vaccination against the disease.
Sample collection and preparation
Samples of ileal lymph nodes were collected at the time of slaughter, immediately frozen in liquid nitrogen, and stored until RNA extraction was performed. Additional samples of ileal lymph nodes and ileum were fixed in either 10% buffered formalin at room temperature for 48 hours or 10% neutral formalin in absolute ethanol at 4°C for 24 hours. For histopathologic examination, 20 blocks of the ileum and the ileal lymph node from each animal were collected and were fixed within either 10% buffered formalin at room temperature for 48 hours or 10% neutral formalin in absolute ethanol at 4°C for 24 hours and processed for paraffin-wax embedding. One pair of sections from each block fixed with buffered formalin was stained with hematoxylin and eosin (HE) and by the Ziehl–Neelsen (ZN) method. To differentiate the type of lesion, the type and the number of inflammatory cells and the number of acid-fast bacteria found within all sections were assessed. The samples fixed with formalin-alcohol were submitted for in situ hybridization to detect interleukin-18 (IL-18) mRNAs.
RT-PCR
Total cellular RNA was extracted by the acid guanidinium thiocyanate-phenolchloroform method. 10 From 1 μg of the extracted total RNA from each sample, cDNA was reverse transcribed using random hexameric primers and subjected to PCR amplification using primer pairs listed in Table 1. A standard PCR mix was used with 2.5 units of Taq polymerase and supplied buffer (Invitrogen Co., Carlsbad, CA), with the following cycles: 40 cycles of 95°C for 30 seconds, 55°C for 30 seconds, 72°C for 1 minute, and 5 minutes in the final cycle. Amplification products were resolved by electrophoresis in 2% agarose gels and were confirmed by dideoxy sequencing. Densitometry was used for relative semiquantification of the RT-PCR product. Cytokine mRNA expressions were evaluated by the housekeeping gene β-actin, as determined by densitometric analysis to insure that an equal amount of RNA was used in each reaction from each sample. Beta-actin was chosen as the endogenous internal standard because it is among those recommended for this purpose and is frequently used in studies of this nature. 17 Complementary DNA (cDNA) derived from healthy bovine peripheral blood mononuclear cells stimulated with 100 ng of lipopolysaccharide (LPS) isolated from Escherichia coli 0111:B4 (List Biological Laboratories Inc., Campbell, CA) for 6 hours were used as positive controls for each cytokine PCR product.
Primers used for semiquantitative RT-PCR.

In situ hybridization
Bovine sequence-specific sense and antisense RNA probes for IL-18 were synthesized using the amplified cDNA fragment, cloned with the TA Cloning Kit (In-vitrogen Co.) and labeled with digoxigenin by the DIG RNA Labeling Kit (Roche Molecular Biochemicals, Mannheim, Germany). Ten percent formalin-alcohol–fixed paraffin-embedded tissue sections were deparaf-finized and treated with 10 μg/ml proteinase K (Roche) for 20 minutes at 37°C, following additional fixation in 4% paraformaldehyde in phosphate buffered saline (PBS) for 10 minutes. Slides were treated with 0.2 M HCl for 10 minutes and with 0.25% acetic anhydride added to 0.1 M triethanolamine-HCl (pH 8.0) for 10 minutes. The antisense RNA probe (50 μg of RNA/ml) in hybridization solution containing 50% formamide, 600 mM NaCl, 10 mM Tris-HCl (pH 7.6), 1 mM EDTA, 1× Denhart's solution, 10% dextran sulfate, 200 μg/ml tRNA, and 1.38% sodium dodecyl sulfate (SDS) was applied onto a sample slide and covered with EasiSeal (Hybaid Limited, UK). Slides were preheated at 90°C for 10 minutes and hybridized for 16 hours in the OmniSlide System (Hybaid Limited) at 42°C. After washing slides with 5× sodium chloride/sodium citrate (SSC) for 5 minutes and 50% formamide/2× SSC for 30 minutes at 42°C, slides were incubated for 5 minutes at 37°C with RNase A (6 μg/ml, Roche), then washed sequentially in 2× SSC and 0.2× SSC for 40 minutes each at 42°C. Slides were then incubated with sheep anti-DIG antibody labeled with alkaline phosphatase (Roche) and developed in nitro blue tetrazolium chloride/5-Bromo-4-chloro-3-in-dolyl phosphate (NBT/BCIP) solution (Roche) in the dark for 3 hours at room temperature. After the in situ hybridization to visualize IL-18 mRNA, immunohistochemical assessment of the detection for MAP antigens was continuously performed in the same sections. The primary antibody was rabbit anti-Mycobacterium bovis (DAKO), which has confirmed cross-reactivity against MAP antigens. 35 The sections were then labeled using a HISTOFINE SAB-PO kit (Nichirei Co., Tokyo) containing a biotinylated secondary antibody for anti-rabbit IgG and streptavidin-conjugated peroxidase. Localized peroxidase conjugates were visualized with 3,3′-diaminobenzidine. Negative controls were prepared by using the sense RNA probe for IL-18 for in situ hybridization and a normal rabbit immunoglobulin fraction (DAKO) instead of the primary antibody for immunohistochemistry. All sections were lightly counterstained with veronal acetate-buffered 1% methyl green solution for 15 minutes.
Statistical analysis
Quantitative data were expressed as mean ± SD of three experiments per group. Statistical significance of the data was determined by analysis of variance followed by multiple comparisons of means with the Bonferroni/Dunn method; statistical significance was set at P < 0.05.
Results
Clinical and histological findings
Relevant clinical data are shown in Table 2. In naturally infected cows, positive results of ELISA tests were obtained in five cows and negative results in two cows. The positive result of the ELISA test was diagnosed when sample ELISA optical density (OD) value was above 0.4 according to the manufacturer's manual. Particularly high ELISA antibody levels were observed in cow Nos. 1 and 2. Cow Nos. 4 and 5 had positive results in the fecal culture test but negative results in the ELISA test. In the noninfected group, negative results of the ELISA and fecal culture test were obtained. In ileum and ileal lymph nodes from cow Nos. 1–2, lepromatous-type granulomatous lesions were observed pathologically (lepromatous group). The multifocal granulomata were found in the lamina propria, submucosa, and the lymphoid follicles of Peyer's patches in ileum as well as in the cortex and paracortex of the ileal lymph nodes and were composed predominantly of macrophages and nodular epithelioid cells with a few lymphocytes. These epithelioid granulomata frequently contained many acid-fast bacteria. In contrast, tuberculoid type lesions were observed in cow Nos. 3–7 (tuberculoid group). The granulomatous lesions were present in the ileal lamina propria as well as the subcapsular sinus and paracortical zone of the lymph nodes, and were composed primarily of macrophages with many lymphocytes and occasionally Langhans-type multinucleate giant cells with small numbers of epithelioid cells. Among that tuberculoid group, relatively mild lesions were found in the ileal tissues of cow Nos. 4 and 5. None or low numbers of acid-fast bacteria were detected in tuberculoid-type lesions. Although, mixed-type granulomas were observed only in boundary between normal tissue and the lepromatous-type granulomas in cow Nos. 1–2 but never in the tuberculoid group. In noninfected cow Nos. 8–12, no lesions were observed in the ileum and the ileal lymph nodes.
Relevant clinical data for MAP-infected cows and noninfected cows.

∗ ++ = sample ELISA OD value < 1.0; + = 0.4 ≤ sample ELISA OD value ≤ 1.0; − = sample ELISA OD value < 0.4.
† The results were determined after a 2-month culture period.
‡ Lepromatous-type lesion.
§ Tuberculoid-type lesion.
RT-PCR for inflammatory cytokine-related mRNAs
Th2 type anti-inflammatory cytokines IL-4 and IL-10 were expressed significantly more in the ileal lymph nodes of the lepromatous group than in those of the tuberculoid group (P < 0.01) and the noninfected group (P < 0.05) (Fig. 1). Among Th1-type cytokines examined, IL-2 mRNA was expressed more in the lepromatous group than in the tuberculoid and noninfected groups (P < 0.001). Moreover, significantly greater expression of the IL-2 receptor (IL-2R) mRNA was detected in the lepromatous group than in the tuberculoid group (P < 0.05) (Fig. 2). Although high interferon-γ (IFN-γ) mRNA expression was observed in cow Nos. 4 and 5 in the tuberculoid group, no statistical differences between the lepromatous, tuberculoid, and noninfected group were found (Fig. 2), as was also the case with mRNA expressions of IL-1β, TNF-α, and GM-CSF (Fig. 3). Messenger RNA expression of preinflammatory cytokines IL-12p35 and IL-12p40, however, did not differ among the three groups, with only IL-18 mRNA being expressed at lower levels in the lepromatous group than in the tuberculoid group and the noninfected group (P < 0.0001) (Figs. 4, 5).
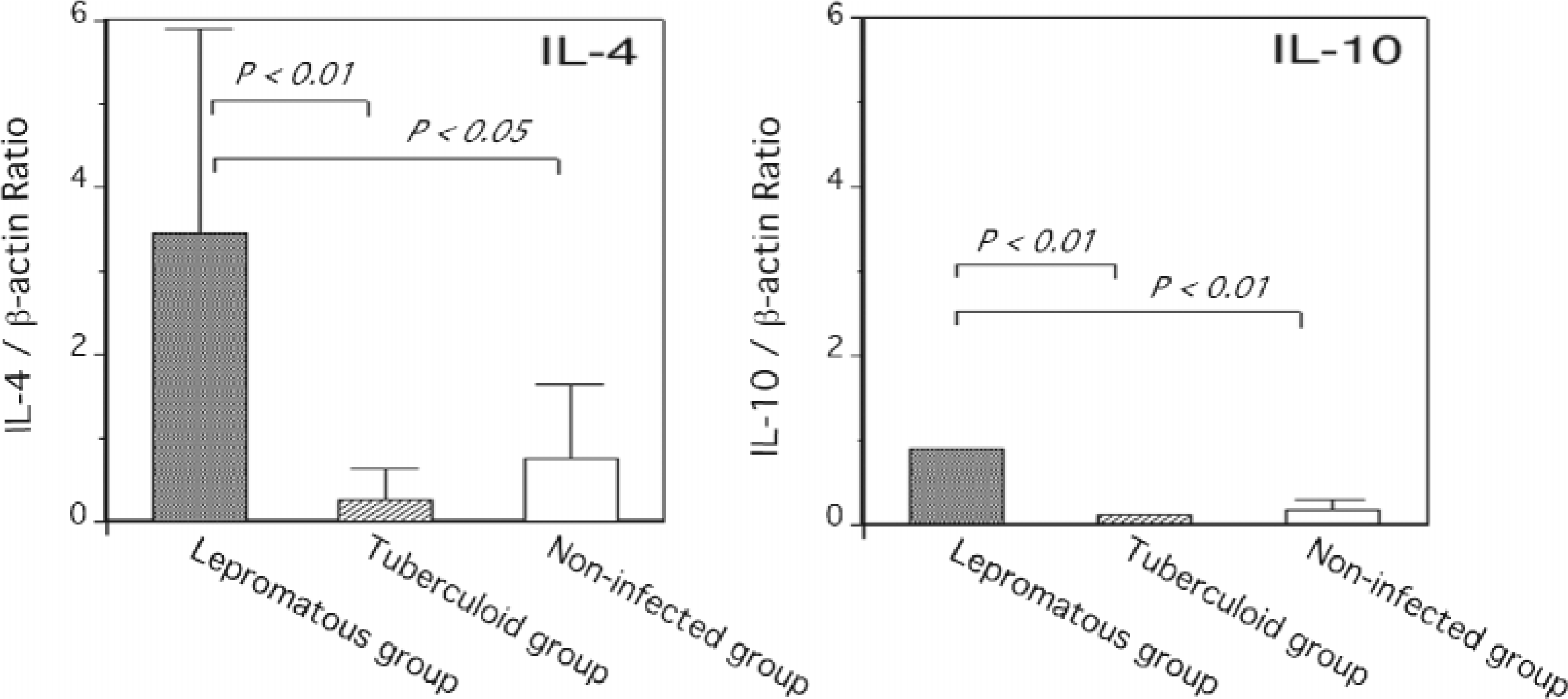
Differences in IL-4 and IL-10 gene expression in the ileal lymph node from cows in lepromatous group (n = 2), tuberculoid group (n = 5), and noninfected group (n = 5). The data are expressed as mean ± SEM of three experiments per group.
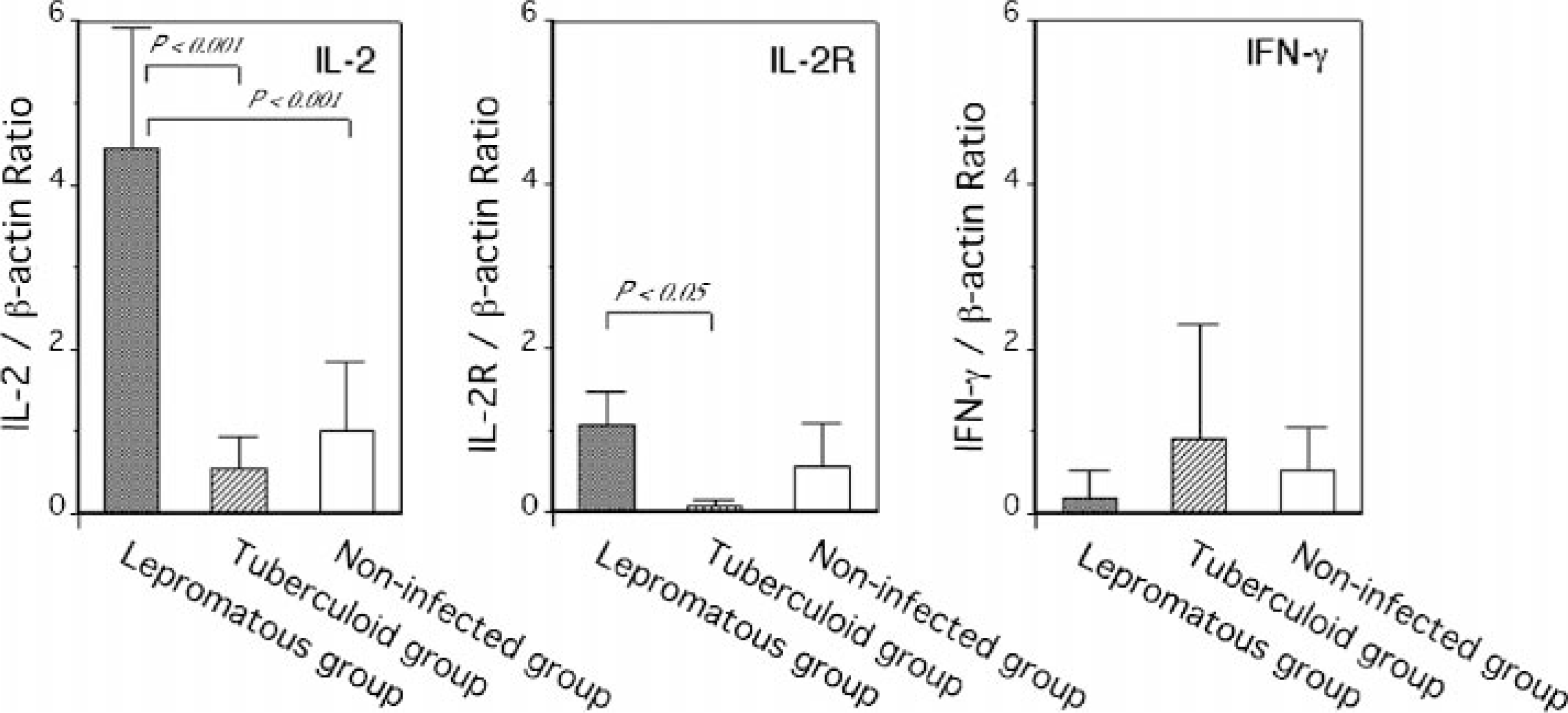
Differences in IL-2, IL-2 receptor (IL-2R), and IFN-γ gene expression in the ileal lymph node from cows in lepromatous group (n = 2), tuberculoid group (n = 5), and noninfected group (n = 5). The data are expressed as mean ± SEM of three experiments per group.
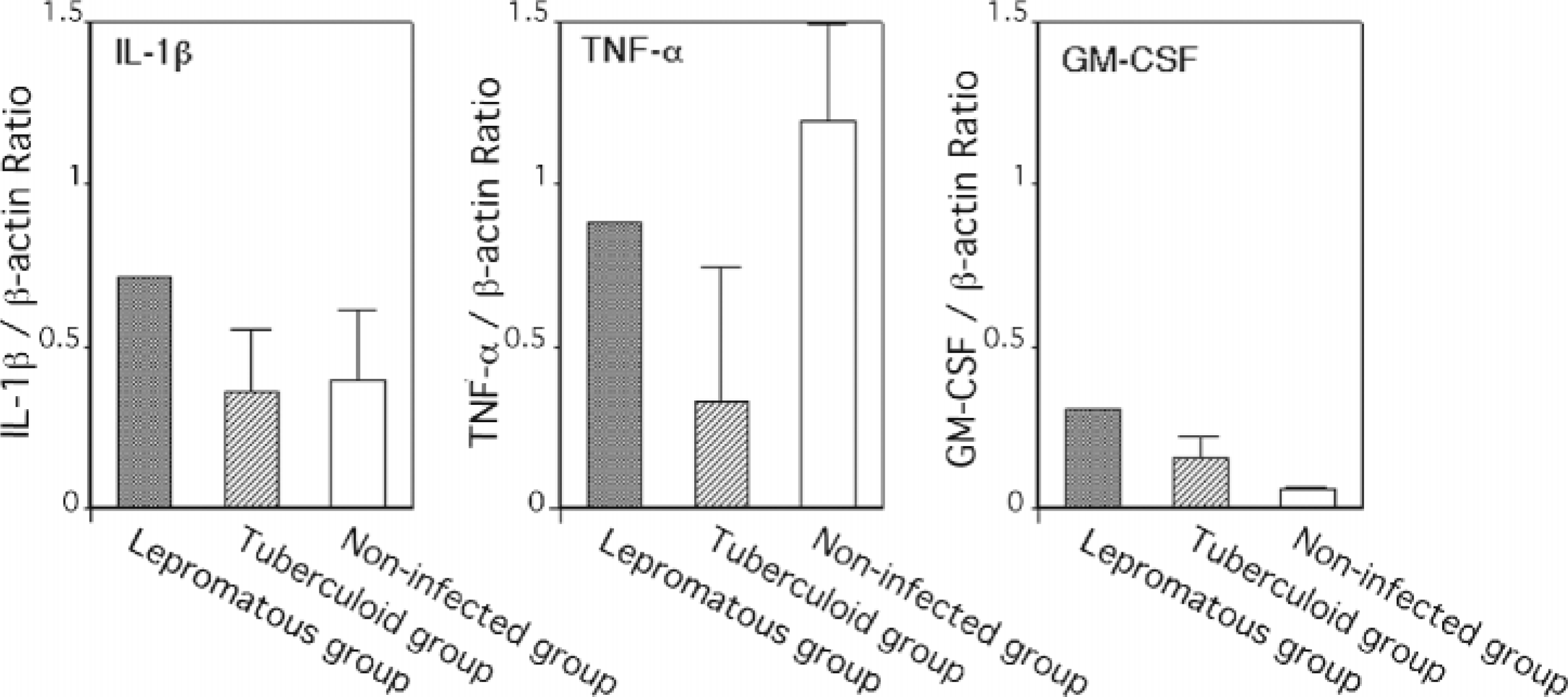
Differences in IL-1β, TNF-α, and GM-CSF gene expression in the ileal lymph node from cows in lepromatous group (n = 2), tuberculoid group (n = 5), and noninfected group (n = 5). The data are expressed as mean ± SEM of three experiments per group.
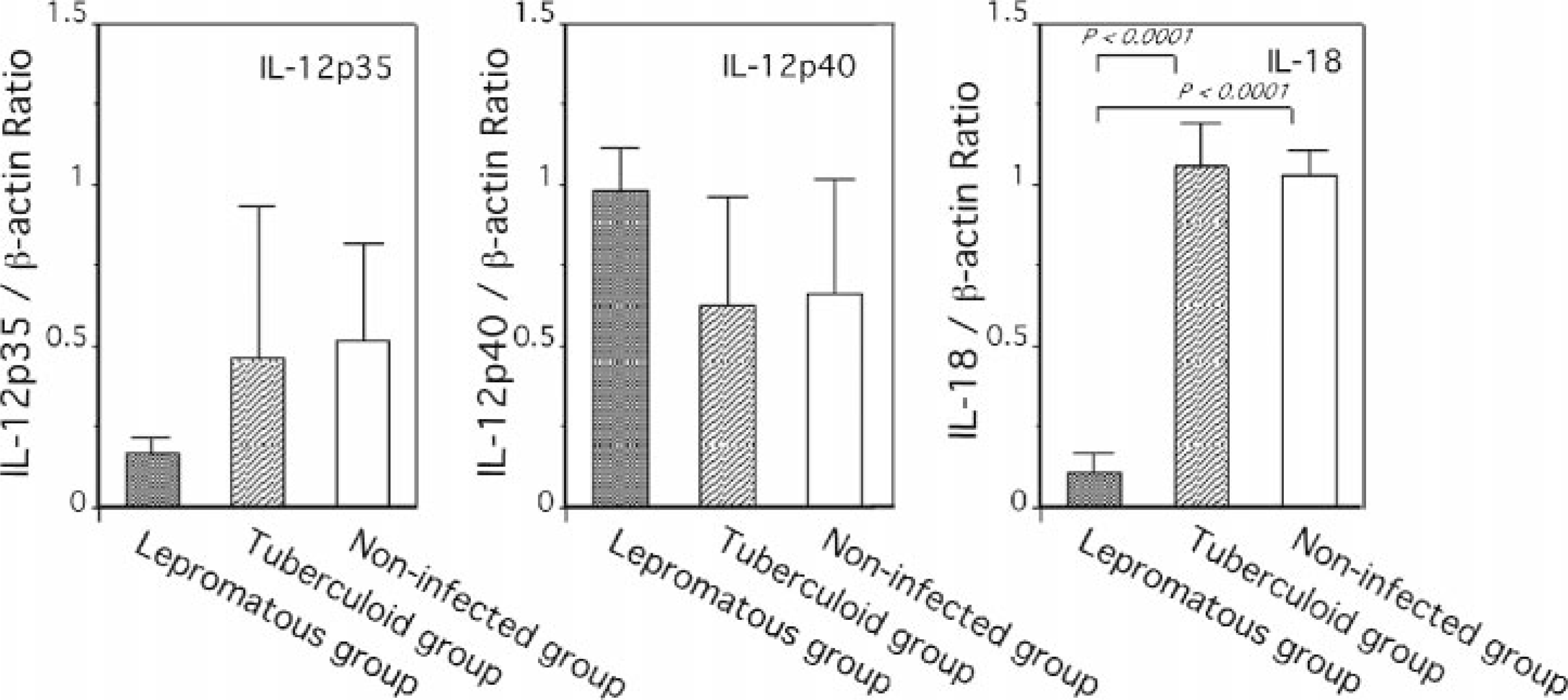
Differences in IL-12p35, IL-12p40, and IL-18 gene expression in the ileal lymph node from cows in lepromatous group (n = 2), tuberculoid group (n = 5), and noninfected group (n = 5). The data are expressed as mean ± SEM of three experiments per group.
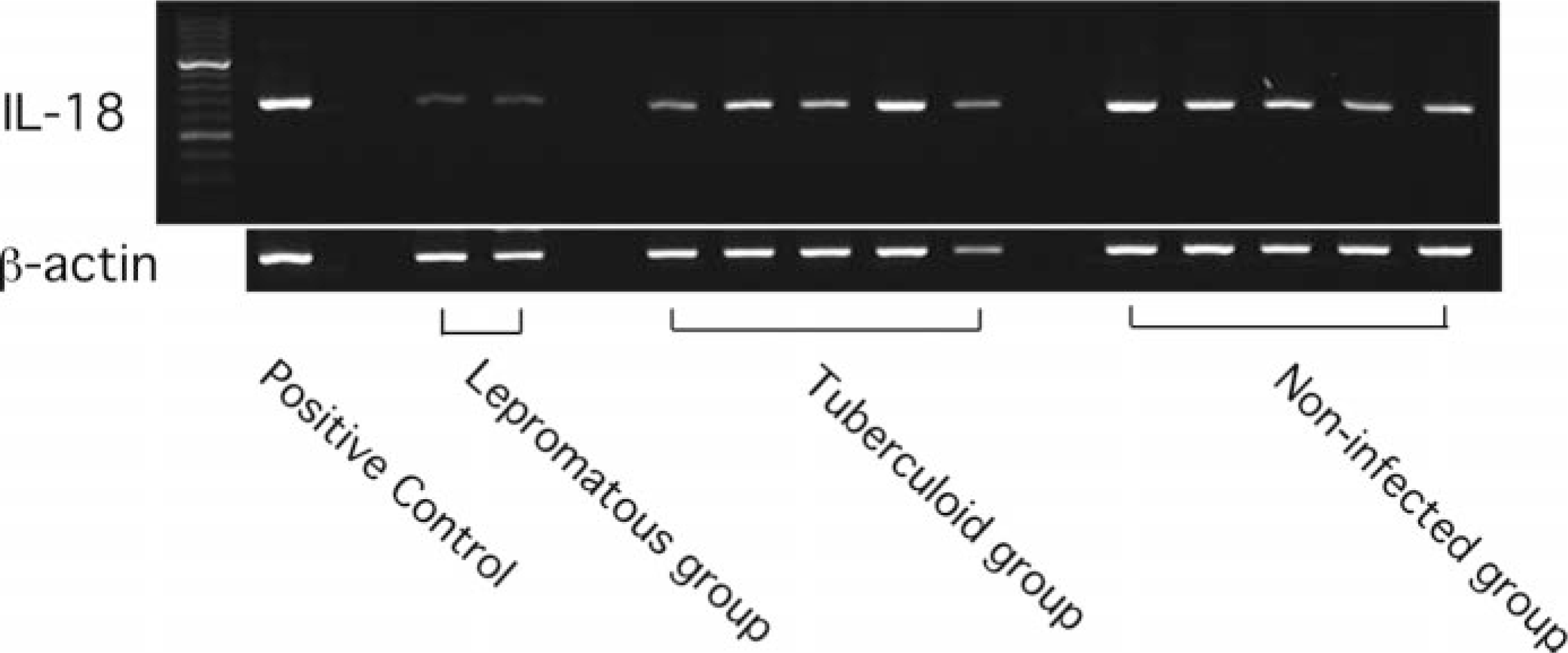
Expression of mRNA for IL-18 gene in the ileal lymph node from cows in lepromatous group (n = 2), tuberculoid group (n = 5), and noninfected group (n = 5) was examined by RT-PCR. The bottom lanes correspond to the amplification with β-actin mRNA-specific primers as control. As a positive control, cDNA derived from bovine peripheral mononuclear cells stimulated with 100 ng of lipopolysaccharide were used. The predicted sizes for the amplification products were 342 bp for IL-18 and 270 bp for β-actin.
In situ hybridization for IL-18 mRNA
In the ileum of the tuberculoid group, preinflammatory cytokine IL-18 mRNA signals were strongly detected in large numbers of macrophages infiltrating the lamina propria, submucosa, and lymphoid follicles of Peyer's patches in the sections performed in situ hybridization alone. However, in the sections that achieved the combination of in situ hybridization and immunohistochemistry, the cytokine signals were slight in epithelial cells and Langhans-type multinucleated giant cells in which no MAP antigens were observed (Fig. 6). Frequently, macrophages bearing small amounts of MAP without IL-18 signals were surrounded by infiltrating macrophages with strong signals on the same sections (Fig. 7). In contrast, in the ileum of the lepromatous group, no IL-18 mRNA signals were detected in the majority of nodular epithelioid cells and infiltrating macrophages containing large numbers of MAP, and only slight signals were observed in ileal epithelial cells (Fig. 8A). Nonspecific reactions were not observed in any negative controls using the sense RNA probe for in situ hybridization or the commercial normal rabbit immunoglobulin fraction for immunohistochemistry (Fig. 8B).
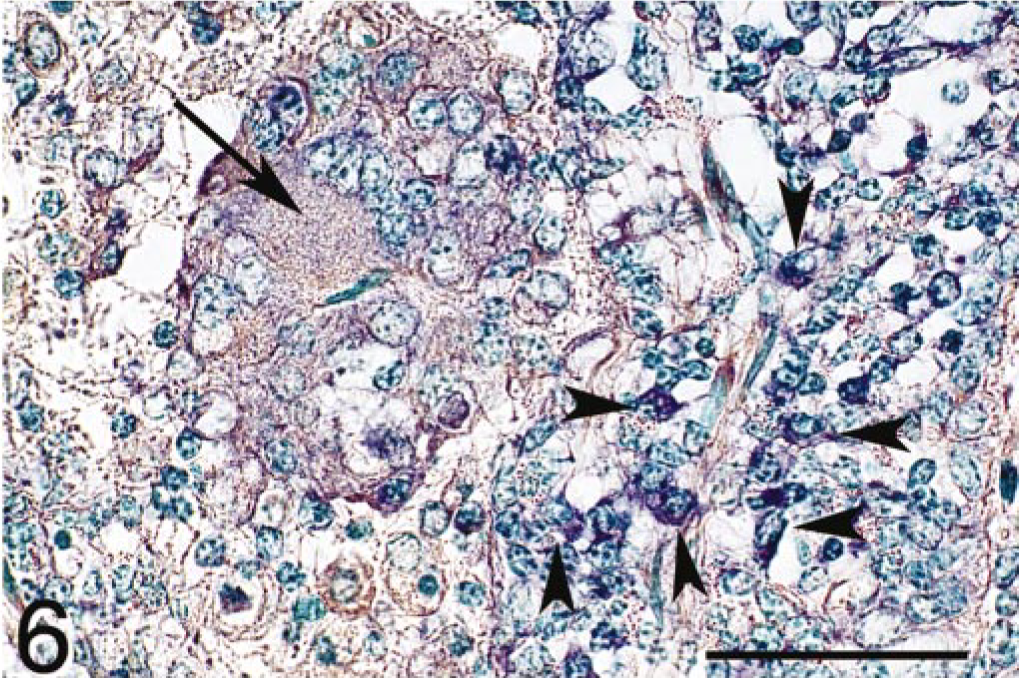
Ileum; cow; tuberculoid-type lesion. Signals for IL-18 mRNA (purple color) were detected in infiltrating macrophages (arrow heads) and in a multinucleated giant cell (arrow). MAP antigens were not found. Combination of in situ hybridization for IL-18 mRNA and immunohistochemistry for M. avium subsp. paratuberculosis (MAP) antigens. Methyl green counterstain. Bar = 50 μm.
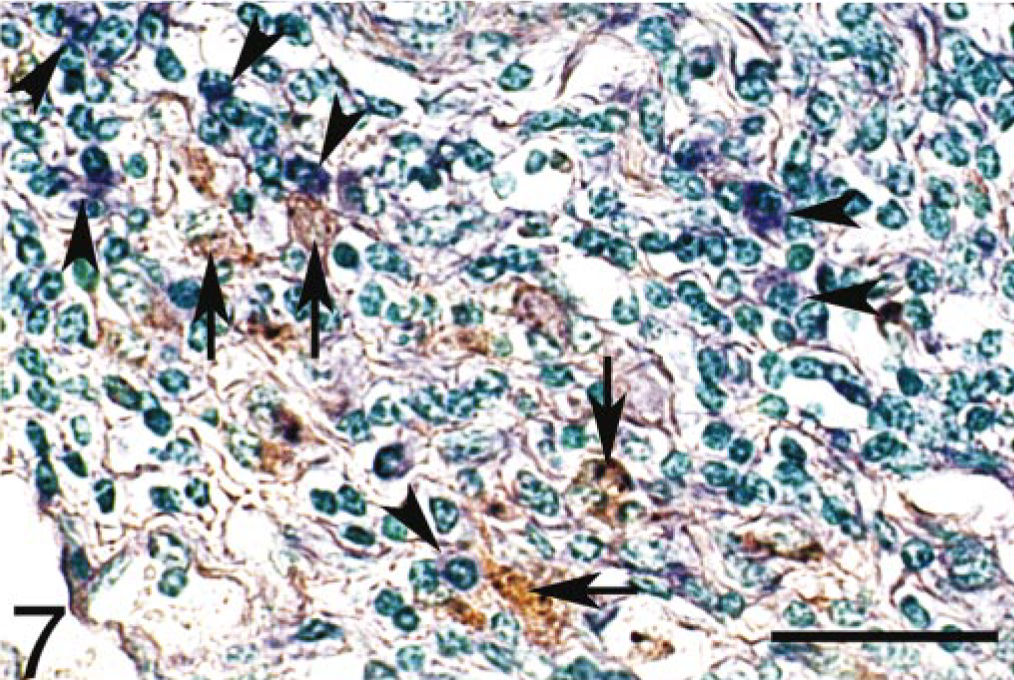
Ileum; cow; tuberculoid-type lesion. MAP-infected macrophages (arrows) without IL-18 signals were surrounded by infiltrated macrophages (arrow heads) with IL-18 signals (purple color). Combination of in situ hybridization for IL-18 mRNA and immunohistochemistry for M. avium subsp. paratuberculosis (MAP) antigens. Methyl green counterstain. Bar = 50 μm.
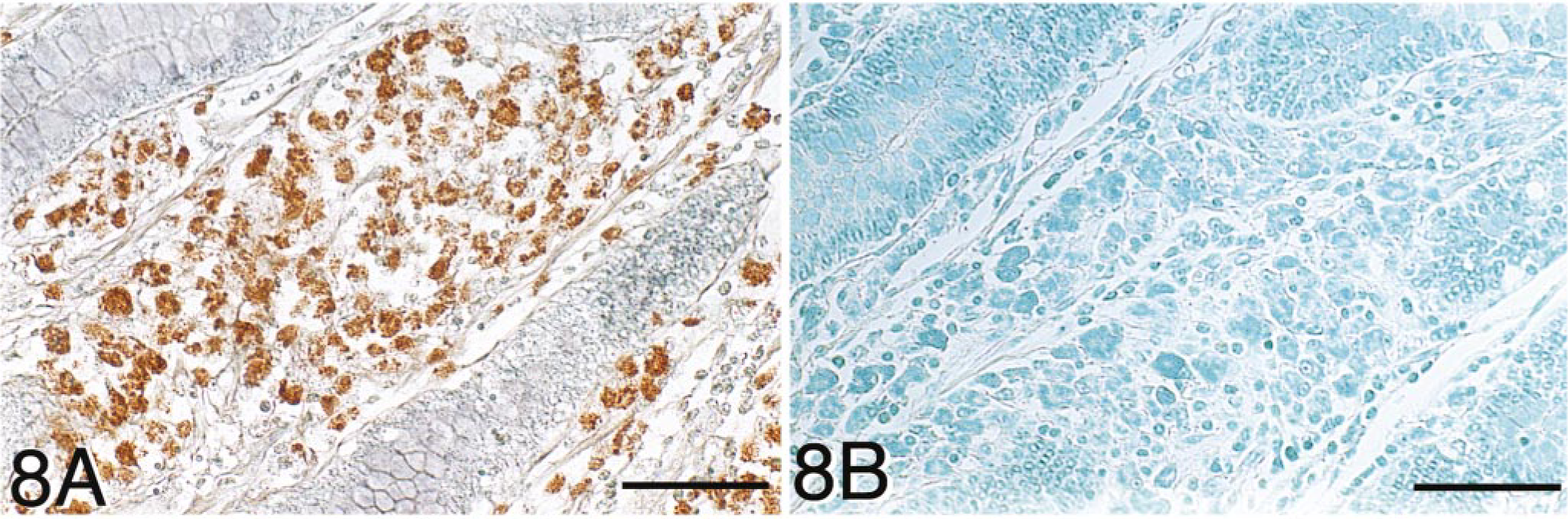
Ileum; cow; lepromatous-type lesion. Combination of in situ hybridization for IL-18 mRNA and immunohistochemistry for M. avium subsp. paratuberculosis (MAP) antigens. Methyl green counterstain.
In the ileal lymph nodes, strong IL-18 signals visualized by in situ hybridization techniques were detected in large numbers of macrophages and multinucleate giant cells (Fig. 9) located in the subcapsular sinus, lymphoid follicles, paracortex zone, medullary cord, and medullary sinus in the tuberculoid group. However, in the lepromatous group, most of the nodular epithelioid cells and macrophages containing numerous MAP had negligible IL-18 signals detected by the combination of in situ hybridization and immunohistochemical assessment (Fig. 10). Nevertheless, small numbers of macrophages located in the medullary sinus without MAP had strong IL-18 signals in the lepromatous group as well as in the noninfected group.
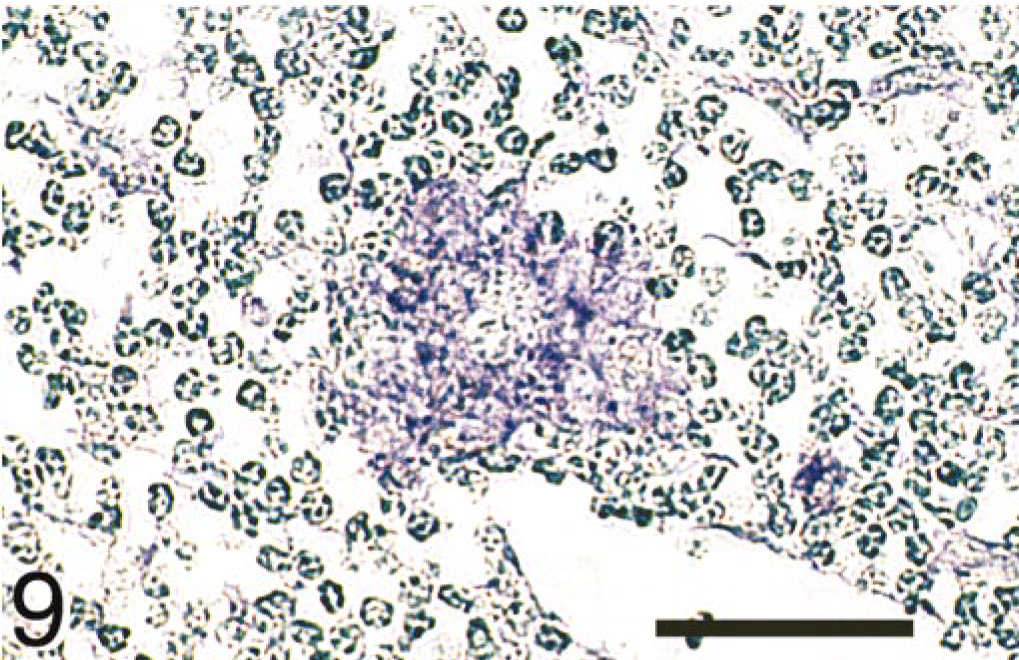
Mesenteric lymph nodes; cow; tuberculoid-type lesion. Strong signals for IL-18 mRNA (purple color) were detected in a multinucleate giant cell located in the cortex. MAP antigens were not found. Combination of in situ hybridization for IL-18 mRNA and immunohistochemistry for M. avium subsp. paratuberculosis (MAP) antigens. Methyl green counterstain. Bar = 100 μm.
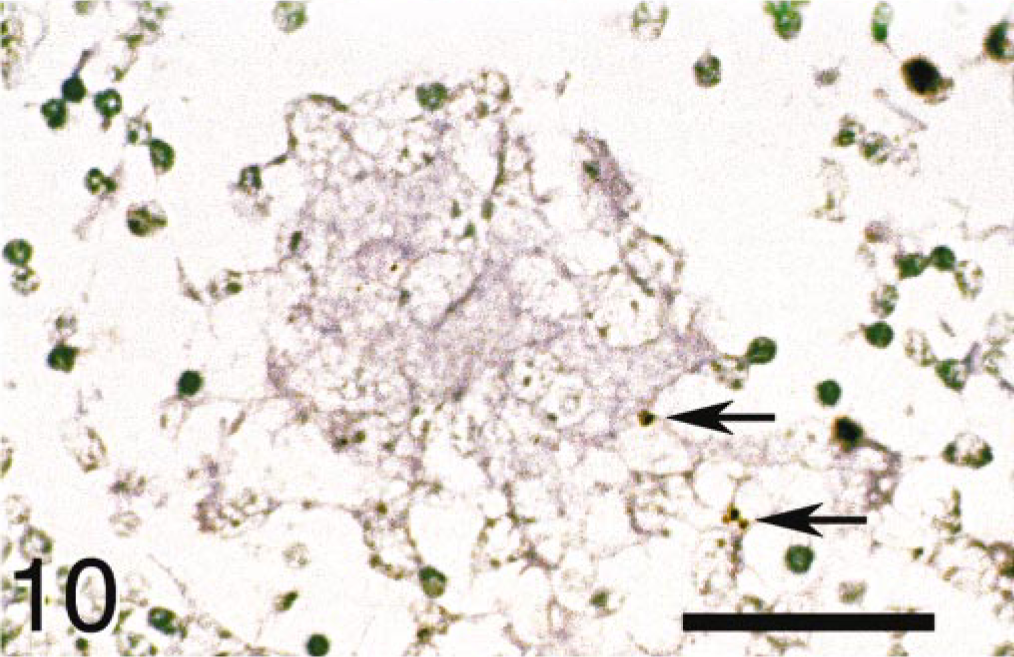
Mesenteric lymph nodes; cow; lepromatous-type lesion. No signals for IL-18 mRNA were detected with MAP antigens (arrows) in an epithelioid cell tubercle. Combination of in situ hybridization for IL-18 mRNA and immunohisto-chemistry for M. avium subsp. paratuberculosis (MAP) antigens. Methyl green counterstain. Bar = 100 μm.
Discussion
In the present study, we compared immunoregulatory cytokine mRNA expressions between tuberculoid and lepromatous lesions and gained information of Th1/Th2 balance at the site of the lesions during asymptomatic stages of bovine paratuberculosis. Although there was no significant difference in IFN-γ mRNA expression between the lepromatous group and the tuberculoid group, higher IFN-γ mRNA expression was observed in the two cows in the tuberculoid group, for which positive results were obtained by a fecal culture test and negative results by an ELISA test with relatively mild tuberculoid lesions. Interferon-γ probably plays an important role in acquired resistance against MAP infection 18, 27, 28, 30, 41 by means of the induction of reactive nitrogen intermediates, which are necessary for the killing of the intracellular mycobacteria. 4 As such, IFN-γ-mediated protection for MAP infection will be efficient only in early stages of the disease. In contrast, the expression of IL-4, IL-10, IL-2, and its receptor IL-2R were significantly higher in the lepromatous group than the tuberculoid group. The prominent IL-2 and IL-2R gene expressions in the lepromatous group are involved in Th1-type responses, 36 yet the IL-2/IL-2R interaction also efficiently promotes Th2 cell growth in the presence of IL-1 and IL-4. 13, 37 Interleukin-4 and IL-10 are known as anti-inflammatory cytokines and are involved in the ascendant differentiation of Th2 cells and the restraining generation of Th1 responses, 7, 34 as well as suppression of anti-mycobacterial macrophage functions involving IFN-γ-induced production of TNF-α and reactive nitrogen intermediates. 14 In the lepromatous group, we found low levels of IFN-γ mRNA expression with high ELISA antibody levels. Thus, the combination of high IL-4 and IL-10 mRNA expressions may result in shifting from the Th1 state to the Th2-dominant state in the lepromatous group and therefore in progressive infection.
Current evidence suggests that the combination of IL-12 and IL-18 induces naive T cells that proliferate and produce IFN-γ in a synergistic manner. 1, 2, 24 This synergistic effect also enhances NK cell cytotoxicity and Th1 cell differentiation. 4 Therefore, the deficient expression of IL-12 and IL-18 may result in diminished levels of cell-mediated immune response against the infection and uncontrolled replication of mycobacteria in susceptible hosts. In vitro and in vivo experiments have revealed progressive suppression of IL-12 production in macrophages infected with Mycobacterium avium 33 or MAP. 34 This suppression seems to be a result of the oversecretion of IL-10 in bovine macrophages infected with MAP. 34 In humans, IL-18 production in response to mycobacterial antigens correlates strongly with IFN-γ production and with protective immunity to mycobacterial infection. 29, 32 Moreover, in human leprosy, in vivo IL-18 mRNA levels are higher in lesions of tuberculoid patients than in those of lepromatous patients. 16 However, no significant differences in IL-12p35 and IL-12p40 gene expression were found among the tuberculoid group, lepromatous group, and noninfected group in this study, with only IL-18 mRNA expression in the lepromatous group being significantly lower than that in the tuberculoid and noninfected groups. For the first time, this inhibition of IL-18 mRNA expression detected by RT-PCR in the lepromatous group was also evidenced by the combination of in situ hybridization for the detection of IL-18 mRNA and immunohistochemical assessment for the finding of MAP antigens performed on the same sections. The inhibition of IL-18 mRNA expression prior to the suppression of IL-12 mRNA expression in the lepromatous group may be relevant to the negative regulation of IFN-γ 13 and induction of the Th2 predominant state. These results suggest that the formation of different granulomatous types in para-tuberculosis is closely linked to differences in the cytokine profile within the lesions. Expression of high levels of Th2 cytokines IL-4 and IL-10 in lepromatous-type lesions may induce a progressive weakening of cell-mediated immunity, resulting in the progression of bacterial growth and sideration of paratuberculosis. In addition, inhibition of proinflammatory cytokine IL-18 gene expression in lepromatous-type lesions indicates that IL-18 may play an important role in resistance to MAP infection in association with the shift from the Th1-dominant state to Th2-predominant state in paratuberculosis.
Footnotes
Acknowledgements
This work was supported in part by a grant (Recombinant Cytokine Project No. 2350) from the Ministry of Agriculture, Forestry, and Fisheries of Japan.
