Abstract
West Nile fever caused fatal disease in humans, horses, and birds in the northeastern United States during 1999. We studied birds from two wildlife facilities in New York City, New York, that died or were euthanatized and were suspected to have West Nile virus infections. Using standard histologic and ultrastructural methods, virus isolation, immunohistochemistry, in situ hybridization and reverse-transcriptase polymerase chain reaction, we identified West Nile virus as the cause of clinical disease, severe pathologic changes, and death in 27 birds representing eight orders and 14 species. Virus was detected in 23/26 brains (88%), 24/25 hearts (96%), 15/18 spleens (83%), 14/20 livers (70%), 20/20 kidneys (100%), 10/13 adrenals (77%), 13/14 intestines (93%), 10/12 pancreata (83%), 5/12 lungs (42%), and 4/8 ovaries (50%) by one or more methods. Cellular targets included neurons and glial cells in the brain, spinal cord, and peripheral ganglia; myocardial fibers; macrophages and blood monocytes; renal tubular epithelium; adrenal cortical cells; pancreatic acinar cells and islet cells; intestinal crypt epithelium; oocytes; and fibroblasts and smooth muscle cells. Purkinje cells were especially targeted, except in crows and magpies. Gross hemorrhage of the brain, splenomegaly, menin-goencephalitis, and myocarditis were the most prominent lesions. Immunohistochemistry was an efficient and reliable method for identifying infected cases, but the polyclonal antibody cross-reacted with St. Louis encephalitis virus and other flaviviruses. In contrast, the in situ hybridization probe pWNV-E (WN-USAMRIID99) reacted only with West Nile virus. These methods should aid diagnosticians faced with the emergence of West Nile virus in the United States.
Keywords
During the late summer and early fall of 1999, an outbreak of viral encephalitis occurred in the northeastern United States, with most cases occurring in and around New York City, New York. The outbreak resulted in fatal neurologic disease in humans and a variety of native and exotic birds, 3–5 as well as some horses. 17 The incidence of disease was particularly high among crows. Initially, the human cases were attributed to St. Louis encephalitis (SLE) virus, 3 a mosquito-borne flavivirus that is endemic to the United States. SLE virus typically does not cause clinical disease in birds. Later, West Nile virus (WNV), a related flavivirus never before identified in the western hemisphere, was shown to be the cause of disease in several birds that died following neurologic illness. 5 , 12 WNV was subsequently identified as the cause of infection in both the human 5 and equine 17 cases as well. Phylogenetic analysis of the virus associated with the US outbreak, WN-New York 1999, demonstrated it is closely related to a strain of WNV isolated from a sick goose in Israel in 1998. 12 Confirmed human and equine cases of WNV infection have been limited to the state of New York; however, infected birds and mosquitoes were also identified in Connecticut and New Jersey during the height of the outbreak. 1 , 5 The possible recurrence and spread of WNV in the United States represents a serious potential public health concern.
WNV is a member of the Japanese encephalitis virus antigenic complex of arthropod-borne flaviviruses. This complex also includes SLE virus, Kunjin virus, and Murray Valley encephalitis virus. 2 , 14 WNV, like other members of the Japanese encephalitis complex, typically circulates in nature between Culex mosquitoes and reservoir hosts in sylvatic transmission cycles. 9 , 10 Humans and horses, considered incidental hosts, become infected during urban transmission cycles. 9 , 19 , 21
Unique and unexplained aspects of the US outbreak of WNV are that birds from a large variety of species exhibited fatal neurologic disease associated with confirmed WNV infection. WNV has been shown to infect a variety of birds. 6 , 8 , 9 , 15 , 20 In one experimental study, 24 infected hooded crows (Corvus corone) and house sparrows (Passer domesticus) developed moderate to high level viremia and died from 3 to 7 days after infection. However, reports of birds showing clinical disease during epizootics are rare. 9 In this report, we describe the viral tropism and pathological aspects of the WNV outbreak that produced fatal disease in native crows and several exotic bird species in New York City during the late summer and early fall of 1999. The methods used during the course of this study should provide valuable tools for the diagnosis of additional cases of WNV, should they occur.
Materials and Methods
Case histories
Between 10 August 1999 and 23 September 1999, 27 wild or exotic birds (24 birds at the Bronx Zoo/Wildlife Conservation Park, Bronx, NY, and 3 birds at the Queens Wildlife Center, Queens, NY) died or were humanely euthanatized because of severe illness. Eight orders and 14 species of birds were involved. These included eight Passeriformes (five common crows [Corvus brachyrhynchos; Nos. 1, 15–18]; one fish crow [Corvus ossifragus; No. 2]; two black-billed magpies [Pica pica; Nos. 3, 19]); six Ciconiiformes (five Chilean flamingos [Phoenicopterus chilensis; Nos. 4, 20–23]; one black-crowned night heron [Nycticorax nycticorax nycticorax; No. 5]); three Pelecaniformes (three guanay cormorants [Phalacrocorax bougainvillei; Nos. 7, 8, 26]); two Charadriiformes (laughing gulls [Larus atricilla; Nos. 10, 27]); three Anseriformes (two bronze-winged ducks [Anas specularis; Nos. 12, 13]; one mallard duck [Anas platyrhynchos; No. 11]); three Galliformes (two Himalayan Impeyan pheasants [Lophophorus impeyanus; Nos. 9, 24]; one Blyth's tragopan [Tragopan blythi; No. 25]); one Falconiformes (northern bald eagle [Haliaeetus leucocephalus alascanus; No. 6]); and one Strigiformes (snowy owl [Nyctea scandiaca; No. 14]). Captive birds were housed either individually in outdoor cages (eagle and owl), or maintained in flocks on ponds (flamingos, cormorants, and ducks) or aviaries (gulls, pheasants, and magpies). All crows were free-ranging wild birds. All affected birds were native western hemisphere species with the exception of the Galliformes, which are native to the eastern hemisphere; however, all three Galliformes birds were captive-born in North America and had been in the collection since 1998 (tragopan) and 1995 (pheasants). With the exception of the ducks (aged 6 months or less), all captive birds were considered adult or aged (range, 3 to 34 years).
Postmortem examination and collection of tissue specimens
Complete necropsies were performed on all 27 birds. Tissues were fixed in 10% neutral phosphate buffered formalin (NBF), routinely processed and stained with hematoxylin and eosin for histopathologic examination. Tissues from major organs were also frozen and stored at −86 C. Impression smears of selected tissues were prepared from fresh and thawed tissues and were fixed in NBF for 24 hours. Selected tissues were immersion fixed in 4% paraformaldehyde and 1% glutaraldehyde in 0.1 M Millonig's phosphate buffer (pH 7.4) for electron microscopy.
Virology
Selected tissues from birds were assessed for levels of infectious virus and viral RNA. Virus isolation and quantitation from tissue samples were accomplished by plaque assays in Vero cell monolayers. Briefly, approximately 0.5 cm3 of frozen tissue was homogenized with alum in a Ten Broeck homogenizer containing 2 ml of a buffer composed of M-199 salts, 1% bovine serum albumin (BSA), 0.35 g/liter sodium bicarbonate, 100 U/ml penicillin, 0.1 g/liter streptomycin, and 1 mg/liter Fungizone in 0.05 M Tris, pH 7.6. After clarification, 0.1 ml of the supernatant was added to duplicate monolayers of Vero cells (American Type Culture Collection, Rockville, MD) in a six-well cell culture plate (Costar), incubated 1 hour at 37 C in 5% CO2, then overlaid with 3 ml 0.5% agarose prepared in M-199 supplemented with 100 U/ml penicillin, 100 mg/liter streptomycin, and 1 mg/liter Fungizone, and returned to the incubator. After 2 days of incubation, an additional 3 ml of agarose overlay, supplemented with 26.6 mg/liter neutral red for viewing of virus plaques was added. Plaques were recorded between days 3 and 5 of incubation. Reverse transcriptase–polymerase chain reaction (RT-PCR) was performed from the same tissue homogenates used to inoculate cells in the virus isolation assays. For RT-PCR testing, total RNA was extracted from the clarified tissue homogenate/supernatant using the Qiagen viral RNA kit (Lanciotti et al., personal communication).
Immunohistochemistry and in situ hybridization
Immunohistochemistry (IHC) was used to identify birds infected with West Nile virus (WNV) and to determine the tissue and cellular tropism of the virus. In situ hybridization was developed and used to confirm the IHC results in selected cases. In a related portion of the study, the specificities of the IHC and in situ methods were analyzed using tissues from animals experimentally infected with known flaviviruses.
Paraffin blocks of bird tissues were sectioned at 5 µm, and the sections were mounted on positively charged glass slides (Superfrost Plus; Fisher, Pittsburgh, PA). All cases were analyzed by IHC for WNV using an immunoperoxidase method (Envision System; DAKO Corporation, Carpinteria, CA), performed according to the manufacturer's instructions. Tissue impression smears were also available from a few bird cases and were also analyzed by IHC. The primary antibody was a rabbit hyperimmune polyclonal antiserum prepared against WNV (provided by Cindy Rossi, USAMRIID). For negative controls, replicate paraffin sections or tissue smears were incubated with nonimmune rabbit serum in place of the primary antibody. Prior to immunostaining, the tissue sections and smears were incubated with proteinase K (DAKO) at room temperature for 6 minutes, followed by peroxidase blocking in 0.03% H2O2 at room temperature for 5 minutes. The primary antibody was diluted 1:500. The primary antibody and the peroxidase-conjugated secondary antibody (Envision System reagent) were incubated with the tissue sections at room temperature for 30 minutes each. Color was developed with 3,3′-diaminobenzidine solution containing H2O2 at room temperature for 8 minutes. All immunostained samples were counterstained with hematoxylin. Heart sections of infected birds identified early in the course of the study were used as positive controls.
Before the WNV IHC studies, several bird cases were initially immunostained for alphaviruses using a polyclonal antiserum prepared against eastern equine encephalitis virus, Venezuelan equine encephalitis virus, western equine encephalitis virus, and Sindbis virus. Additionally, immunostaining was performed for the flavivirus St. Louis encephalitis (SLE) virus using a rabbit polyclonal antibody against SLE virus (provided by Cindy Rossi, USAMRIID).
The WNV in situ hybridization probe, pWNV-E, was constructed by inserting an amplified DNA fragment containing the envelope (E) gene of the WN-USAMRIID99 strain into the TA cloning vector, pCR2.1 (Invitrogen, Carlsbad, CA). This virus was originally isolated in Vero cells from a sick crow collected on the Bronx Zoo grounds. 12 RNA was extracted from an aliquot of the virus stock using TRIzol LS (Gibco-BRL, Gaithersburg, MD) as described by the manufacturer. Briefly, 0.25 ml of virus was combined with 0.75 ml TRIzol LS, mixed well, and incubated at room temperature for 5 minutes. To the mixture, 0.2 ml of chloroform was added, mixed well, and incubated at room temperature for 10 minutes. The aqueous and organic phases were separated by centrifugation at 12,000 × g for 15 minutes at 4 C. RNA was precipitated from the aqueous phase by the addition of 0.5 ml isopropanol and incubation at room temperature for 10 minutes, and was collected by centrifugation. The pellet was washed with 75% ethanol, air dried, and resuspended in 50 µl of nuclease-free water.
The first strand complementary DNA (cDNA) synthesis was performed using the SuperScript preamplification system (Gibco-BRL) and random hexamers for priming. The WNV RNA (5 µl) was combined with 250 ng of random hexamers, heated to 70 C for 10 minutes, and placed on ice for 1 minute. The cDNA synthesis reaction was prepared by adding the remaining reagents to the final concentrations of 20 mM Tris-HCl (pH 8.4), 50 mM KCl, 2.5 mM MgCl2, 10 mM dithiothreitol (DTT), and 0.5 mM deoxynucleotide triphosphates (dNTPs), then incubated at room temperature for 5 minutes. To the reaction mix, 200 U of SuperScript II RT was added and incubated at room temperature for 10 minutes, then transferred to 42 C for an additional 50 minutes. The reaction was terminated by heating to 70 C for 15 minutes.
The E gene was amplified by PCR using the cDNA as a template and primers that annealed to sequences flanking the WNV E gene. The primers used for amplification were WNV811F, 5′-AACACCATGCAGAGAGTTG-3′ (bases 811 to 829) and WNV2580R, 5′-TGATGTCTATGGCACACCC-3′ (bases 2598 to 2580). The PCR mix consisted of 10 mM Tris-HCl (pH 8.3); 50 mM KCl; 1.5 mM MgCl2; 0.001% (w/v) gelatin; 200 µM dNTPs; 200 nM of each primer; 1.25 units of AmpliTaq DNA polymerase (Perkin Elmer, Norwalk, CT); 1 µl WNV cDNA; and water to a final volume of 50 µl. Thermocycling was performed with 30 cycles of denaturing at 94 C for 30 seconds, annealing at 53 C for 30 seconds, and then extension at 72 C for 1.5 minutes, followed by extension at 72 C for 7 minutes and cooling to 4 C. The resulting 1,769-bp product was gel purified using the GeneClean Spin kit (Bio101, Vista, CA) as described by the manufacturer. The E gene fragment was ligated into the pCR2.1 TA cloning vector and TOP 10F′ competent bacterial cells were transformed. An isolated colony was replicated and the plasmid DNA extracted using the Qiafilter Maxiprep system (Qiagen, Chatsworth, CA). The nucleic acid probe was prepared by nick-translating purified DNA to incorporate digoxigenin(DIG)-11-dUTP as described by the manufacturer (Boehringer Mannheim, Indianapolis, IN).
In situ hybridization was performed as previously described. 11 Briefly, tissue sections were deparaffinized and rehydrated, then incubated in 2× standard saline citrate (SSC) for 10 minutes at 70 C. Tissues were permeabilized by digestion with 20 µg/ml proteinase K (Boehringer Mannheim) in Tris-buffered saline at 37 C for 30 minutes in a humid chamber. Slides were rinsed in nuclease-free water and air dried, and the hybridization mixture was added. The hybridization mixture for each slide contained 45% (v/v) deionized formamide, 10% dextran sulphate, 4× SSC, 2 mg/ml BSA, 25 ng of denatured DIG-labeled, nick-translated plasmid probe, and nuclease-free water to a volume of 50 µl. Coverslips were applied to each tissue section, and the slides were incubated overnight at 37 C in a humid chamber.
After hybridization, the slides were washed with 1× SSC and then incubated in Tris-buffered saline with 0.1% BSA and 0.1% Triton X-100. The DIG probe was detected with an alkaline phosphatase–labeled anti-DIG antibody (Boehringer Mannheim) and addition of BCIP (5-bromo-4-chloro-3-indolyl phosphate) as the substrate and NBT (4-nitro blue tetrazolium chloride) as the chromogen. Endogenous alkaline phosphatase activity was blocked by adding 1 mM levamisole (Vector Laboratories, Burlingame, CA) to the enzyme substrate solution. Washing the slides with deionized water stopped color development. The slides were counterstained with nuclear fast red (Vector Laboratories) and dehydrated, and coverslips were applied with Permount.
For specificity testing of the IHC and in situ hybridization methods, formalin-fixed, paraffin-embedded tissues infected with WNV or other encephalitic flaviviruses were used. Tissues taken from suckling mice (2–3 days old) inoculated intracranially with WN-USAMRIID99 or St. Louis encephalitis (SLE) virus strains TBH-28 and MSI-7 were tested as representatives of the Japanese encephalitis group (provided by Dr. George Ludwig, USAMRIID). Other flaviviruses of the tick-borne encephalitis (TBE) group were obtained from the pathology archives at USAMRIID. Mouse brains infected with Central European encephalitis virus or Russian spring–summer encephalitis virus were tested, as were monkey brains infected with Langat virus. Mouse brains infected with Venezuelan equine encephalitis virus (Togaviridae, alphavirus genus) were tested as an unrelated negative control.
Electron microscopy
Portions of tissues from several birds which had been immersion-fixed in 10% NBF or 4% paraformaldehyde and 1% glutaraldehyde in 0.1 M Millonig's phosphate buffer (pH 7.4) were processed for transmission electron microscopy according to conventional methods. Briefly, tissues were postfixed in 1% osmium tetroxide, rinsed, treated with 0.5% uranyl acetate in ethanol, dehydrated in ethanol and propylene oxide, and embedded in Poly/Bed 812 resin (Polysciences, Warrington, PA). Ultrathin sections were cut, placed on 200-mesh copper transmission electron microscope grids, stained with uranyl acetate and lead citrate, and examined with a JEOL 1200-EX transmission electron microscope (JEOL, Peabody, MA).
Postembedment immunoelectron microscopy was performed on portions of formalin-fixed cerebrum and cerebellum from a Chilean flamingo (No. 21) and on Vero cells infected with the WN-USAMRIID99 virus. 12 Monolayers of Vero 76 cells (American Type Culture Collection) in 25 cm2 flasks were adsorbed with stock virus at a multiplicity of infection of five for 1 hour at 37 C. After adsorption, unbound virus was removed, and 4.5 ml of culture medium (Eagle minimal essential medium plus 10% fetal bovine serum) was added, and flasks were incubated at 37 C for up to 72 hours. Inoculated and control flasks were harvested at 18, 48, and 72 hours. The in vitro specimens and the brain samples from the Chilean flamingo were further processed as previously described. 7 Briefly, ultrathin sections on nickel grids were first floated on drops of 4% normal goat serum in 0.1% BSA plus 0.05% Tween 20 in 0.02 M Tris (BTT), then incubated with either anti-WNV mouse ascitic fluid (provided by Cindy Rossi, USAMRIID) or nonimmune mouse ascitic fluid (diluted 1:1000 in BTT). Finally, the sections were incubated with goat anti-mouse immunoglobulin G conjugated to 10 nm colloidal gold (diluted 1:25 in BTT). Grids were then stained with uranyl acetate and lead citrate and examined with the transmission electron microscope.
Results
Clinical findings
Seventeen of the 27 birds were observed clinically from several hours to 6 days before death or euthanasia. The chief clinical presentation in most birds was neurological with varying degrees of severity. Specific neurologic symptoms consisted of ataxia (3/17), tremors (6/17), abnormal head posture (3/17), circling (1/17), or convulsions (1/17). In one of the flamingos, the head was held low with the neck held in an S-shaped conformation. The two laughing gulls were unable to hold their heads normally at rest or upon a forced change in posture. One gull had a weak drop-flap response. Several of the flamingos were uncoordinated, with a wobbly gait and a progressive inability to stand; they were easily approached and caught. Two flamingos presented with anisocoria or impaired vision. One cormorant was noted to be swimming in circles shortly before death. Other birds presented with more nonspecific signs including weakness (12/17) or sternal recumbency (10/17).
Gross findings
Birds presented in good (20/27) to fair (7/27) postmortem condition. The body condition of most birds ranged from slightly thin (8/27) to moderately thin (12/27) (Table 1). The calvaria, meninges, brain, heart, spleen, intestine, pancreas, lung, and kidney had gross lesions of varying severity.
Common lesions in birds infected with West Nile virus. ∗
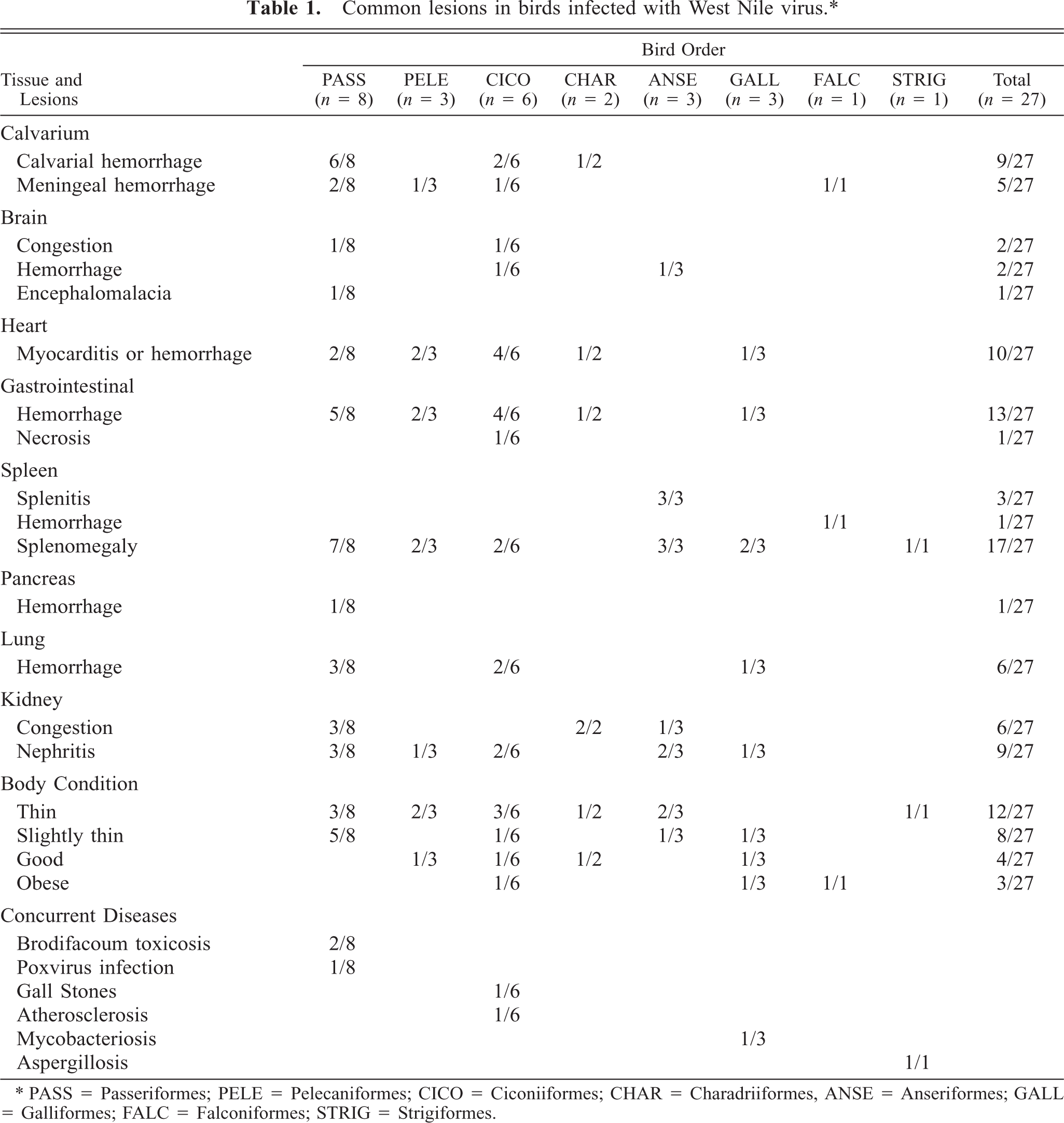
∗ PASS = Passeriformes; PELE = Pelecaniformes; CICO = Ciconiiformes; CHAR = Charadriiformes, ANSE = Anseriformes; GALL = Galliformes; FALC = Falconiformes; STRIG = Strigiformes.
Intraosseous calvarial hemorrhage was prominent in 5 crows (Nos. 1, 2, 15, 17, 18), one magpie (No. 19), and one flamingo (No. 22). Hemorrhage was typically located in the supraoccipital region of the skull, but two birds had diffuse calvarial hemorrhage. Meningeal hemorrhage was present in two flamingos (Nos. 20, 21), one cormorant (No. 26) and the bald eagle (No. 6). The bald eagle had diffuse, severe meningeal hemorrhage. In the other birds, subdural hematomas were localized to the area overlying the cerebellum. The meningeal vasculature was engorged in several cases. Cerebral congestion was severe in the mallard (No. 11) and flamingos (Nos. 4, 23), all of which had diffusely dark pink to deep reddish-purple brains. Hemorrhage was present within the neuropil in a flamingo (No. 21; Fig. 1), a cormorant (No. 26), and a mallard (No. 11).
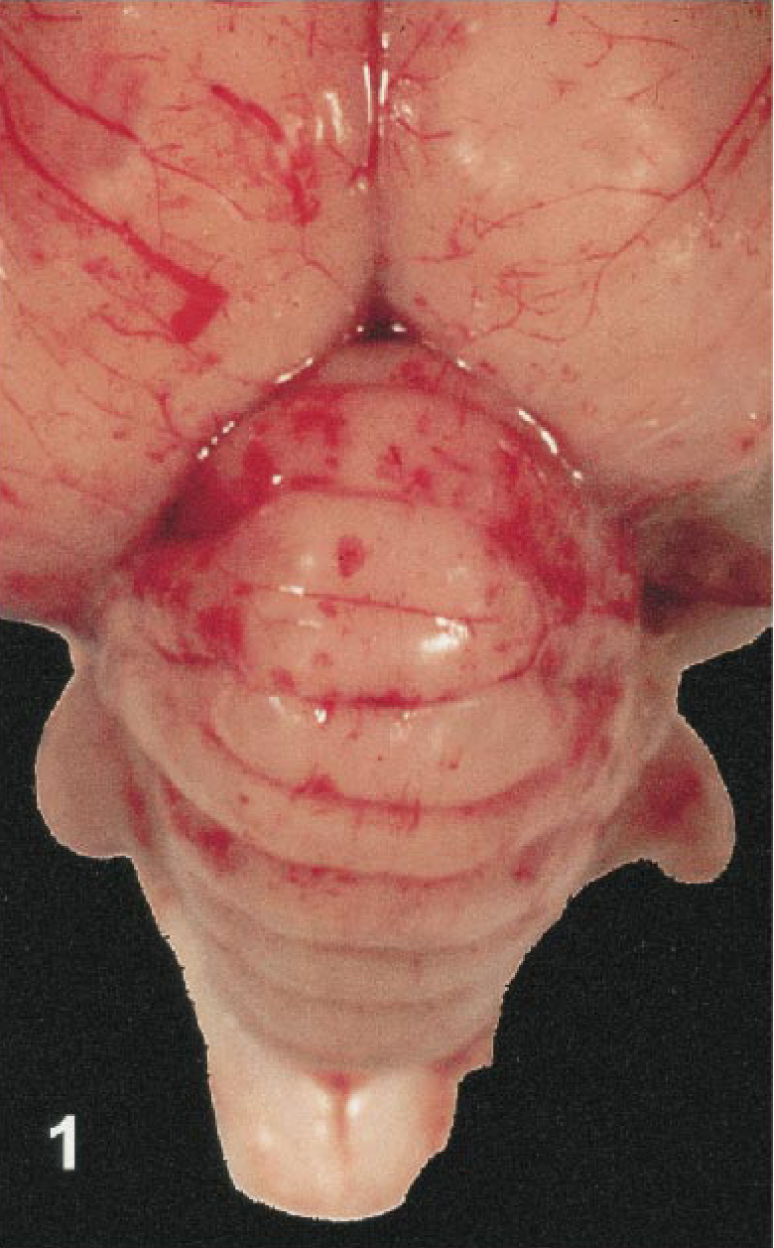
Brain; Chilean flamingo, bird No. 21. Multifocal hemorrhage is present in the cerebellum and both cerebral hemispheres.
Eleven birds had myocardial lesions. In four cases (bird Nos. 4, 10, 11, 17), the myocardium contained multiple pale foci measuring up to 0.2 to 0.3 cm in diameter. In others (bird Nos. 16, 20–22), large portions of the ventricular walls and interventricular septum had white streaks that produced a mottled to striped appearance (Fig. 2). The hearts of some birds were diffusely pale or had myocardial hemorrhage.
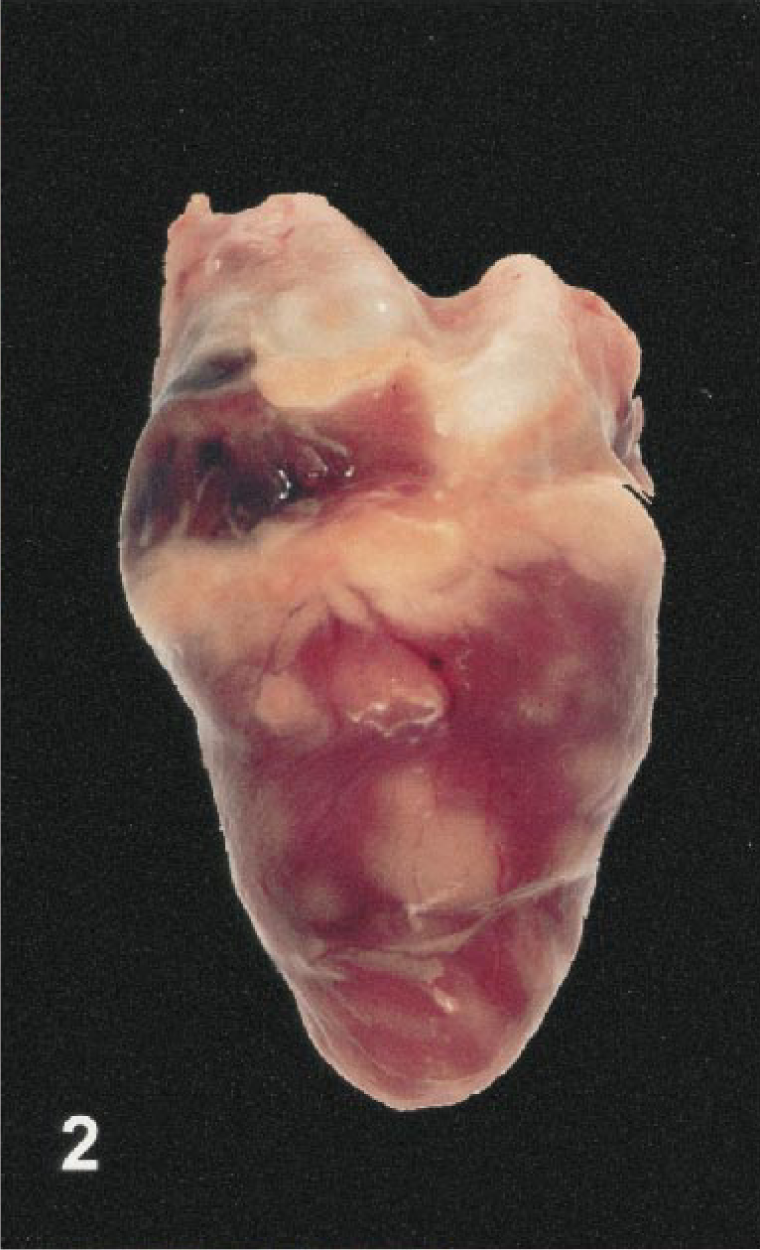
Heart; Chilean flamingo, bird No. 22. Multiple pale foci represent areas of myocardial necrosis and inflammation.
Seventeen birds had splenomegaly, ranging from mild to severe (Fig. 3). Two birds (Nos. 12, 13) had severe mottling and swelling of the spleen consistent with splenitis. Intestinal lesions were present in 18 birds. The most severe of these lesions, characterized by mucosal or lumenal hemorrhage, were found in the duodenum (bird Nos. 5, 7, 11, 15, 17, 18) and jejunum (bird Nos. 4, 5, 7, 11, 17). In the jejunum of one flamingo (No. 4), the mucosa was replaced by a diphtheritic membrane (Fig. 4). Smaller foci of hemorrhage were present in the esophageal mucosa, proventriculus, proximal ventriculus and cloaca. One crow (No. 1) had focal pancreatic hemorrhage.
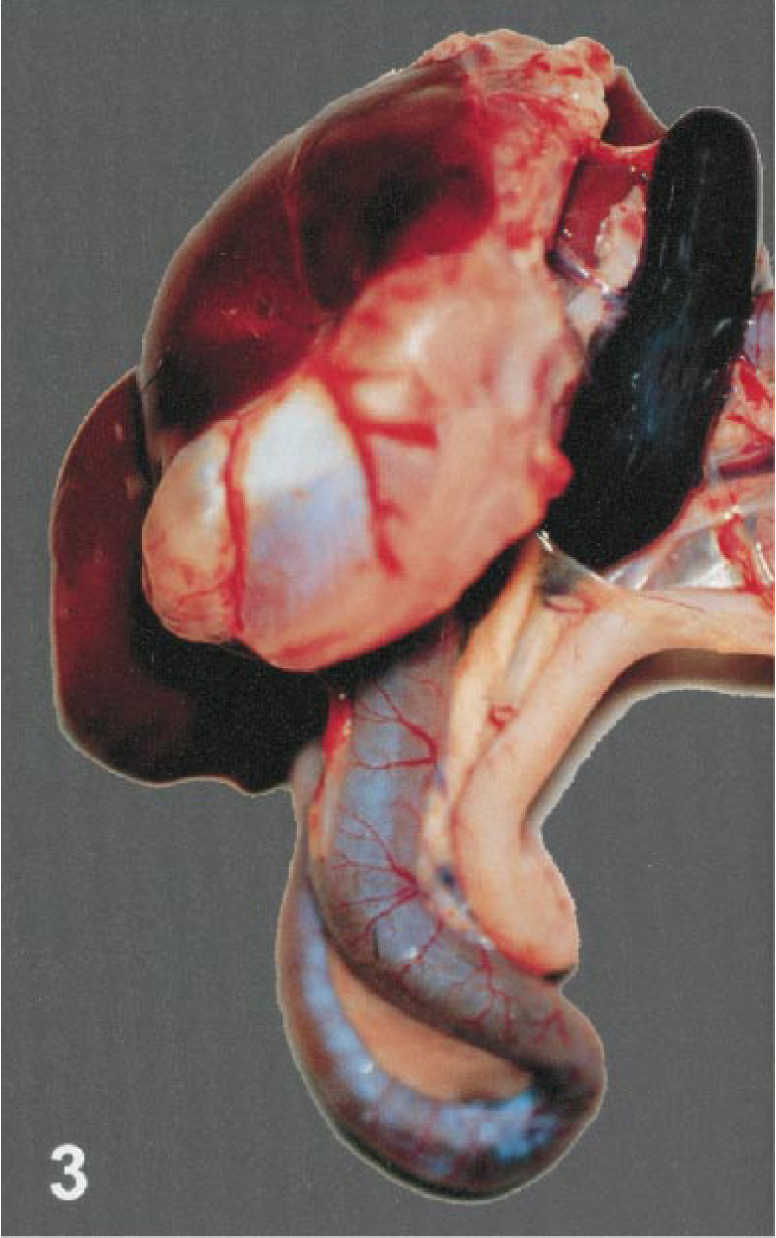
Viscera; crow, bird No. 17. The spleen is swollen and the duodenum is hyperemic.
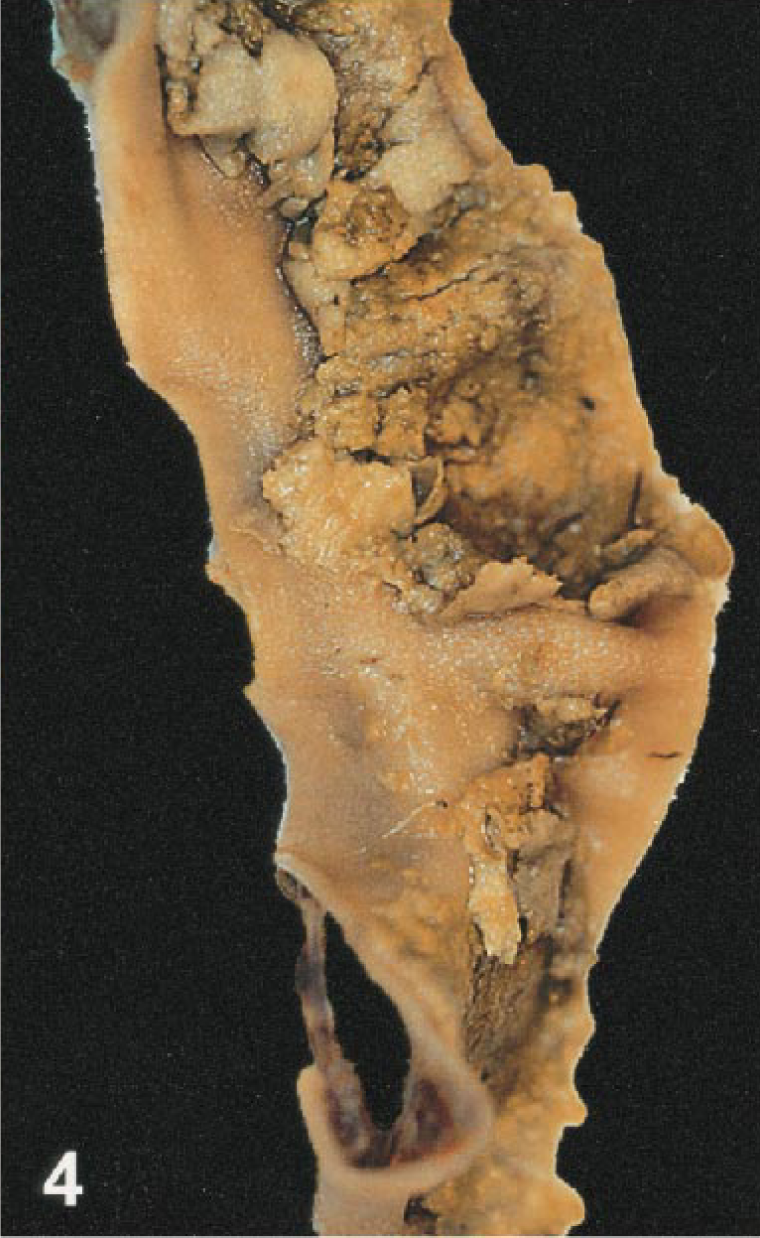
Intestine; Chilean flamingo, bird No. 4. A diffuse diphtheritic membrane covers the mucosal surface.
Gross lesions were present in the kidneys of 15 birds. Of these, nine had mild to moderate swelling, congestion, accentuation of the lobular pattern, and multifocal, pinpoint white foci consistent with nephritis. The remaining six animals had renal congestion alone. Thirteen birds had pulmonary lesions that ranged from mild to moderate congestion and edema to focal or multifocal hemorrhage.
Histologic findings
Almost all of the birds (25/27) had lesions in the brain. Multifocal, acute hemorrhage was a feature in nine cases. Hemorrhage was most severe in the cerebellar folia (Fig. 5) but was also present in the cerebral hemispheres, thalamus, brain stem, and cervical spinal cord. Ten cases had mild to severe meningitis (Figs. 6, 7). Most meningeal infiltrates were composed predominantly of lymphocytes and plasma cells, although one cormorant (No. 8) and the eagle (No. 6) had moderate numbers of heterophils admixed as well.
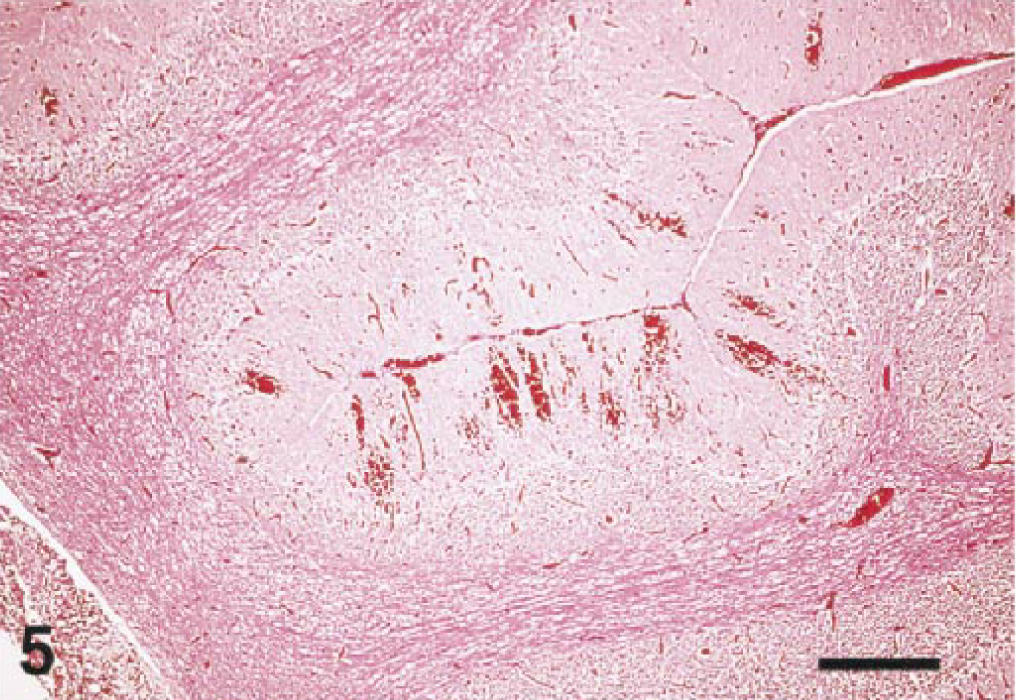
Cerebellum; Guanay cormorant, bird No. 26. Subgross image demonstrates extensive hemorrhage in the cerebellar folia. HE. Bar = 150 µm.
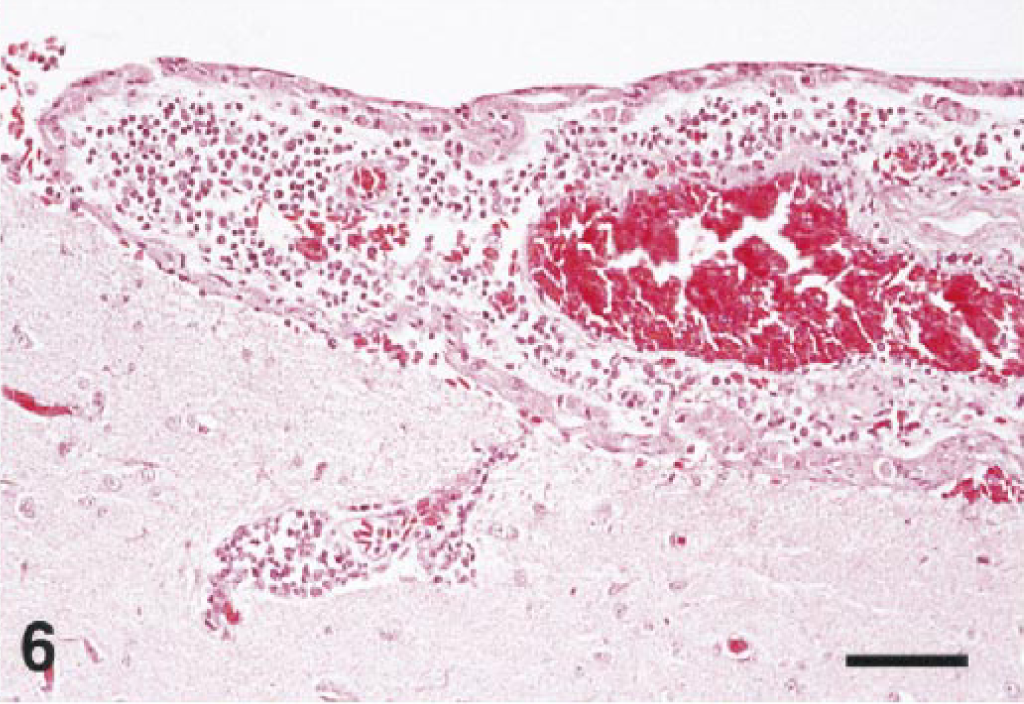
Cerebrum; northern bald eagle, bird No. 6. Inflammatory infiltrates thicken the meninx and form a perivascular cuff in the outer cortex. HE. Bar = 50 µm.
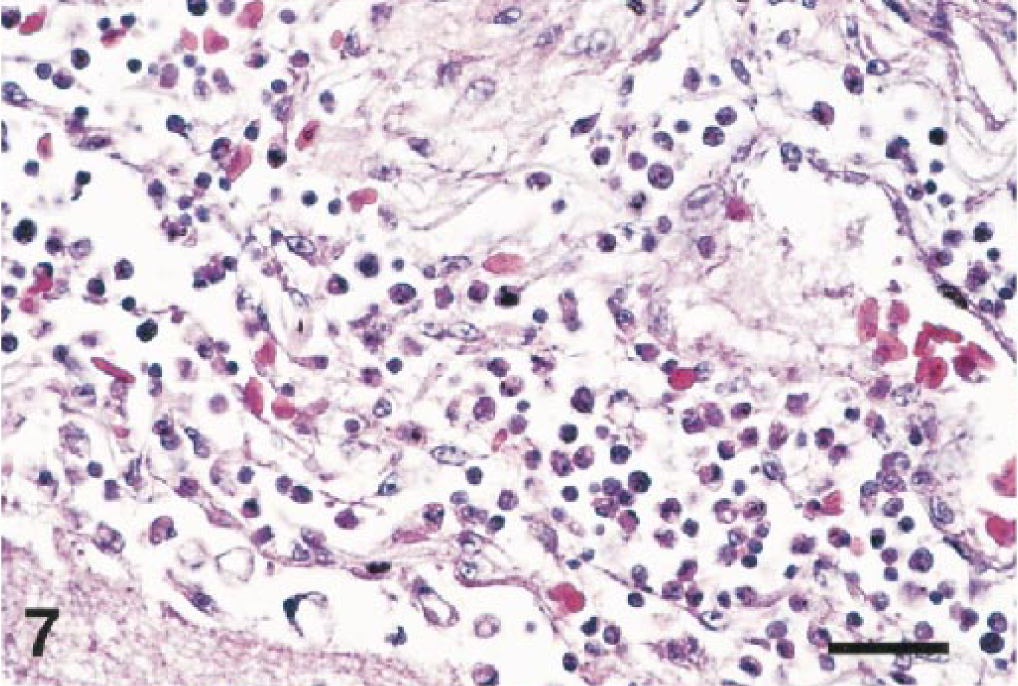
Brain; Chilean flamingo, bird No. 21. The thickened meninx is edematous and infiltrated by a mixture of inflammatory cells. HE. Bar = 35 µm.
Several birds (Nos. 6, 10, 13, 14, 16, 18, 21, 22, 25, 26) had perivascular cuffing, gliosis, or glial nodules. Perivascular cuffs were composed primarily of lymphocytes and plasma cells and ranged from two to five cell layers in thickness. Mild lesions were limited to the cerebellum and brain stem. In severely affected birds, lesions were present in the cerebrum, thalamus, optic lobe, cerebellum, medulla, and cervical spinal cord. Lesions in the brain were most common in the molecular layer of the cerebellum, whereas those in the spinal cord were usually within the gray matter.
In addition to inflammatory lesions, damage to the Purkinje cells of the cerebellum and neurons of the brain stem and cervical spinal cord was also present. Many birds had degeneration or necrosis of Purkinje cells. Affected cells were shrunken, hyperchromatic, and lacked nuclear detail. Multifocally, there appeared to be dropout of Purkinje cells. Variably sized cytoplasmic vacuoles were seen in the perikaryon of neurons in the eagle (No. 6), cormorant (No. 8), gull (No. 27), crow (No. 17), and tragopan (No. 25).
Myocardial lesions were present in 13/27 birds from 6/8 orders (excluding Strigiformes and Anseriformes). Lesions ranged from subtle to severe and involved all portions of the heart. Most affected birds had moderate to severe inflammatory infiltrates in the myocardium, epicardium, or endocardium. In most cases, lymphocytes, plasma cells, and histiocytes were the predominant cell types involved. Several birds had acute noninflammatory changes of disseminated myocytolysis (Fig. 8), hemorrhage, and mineralization.
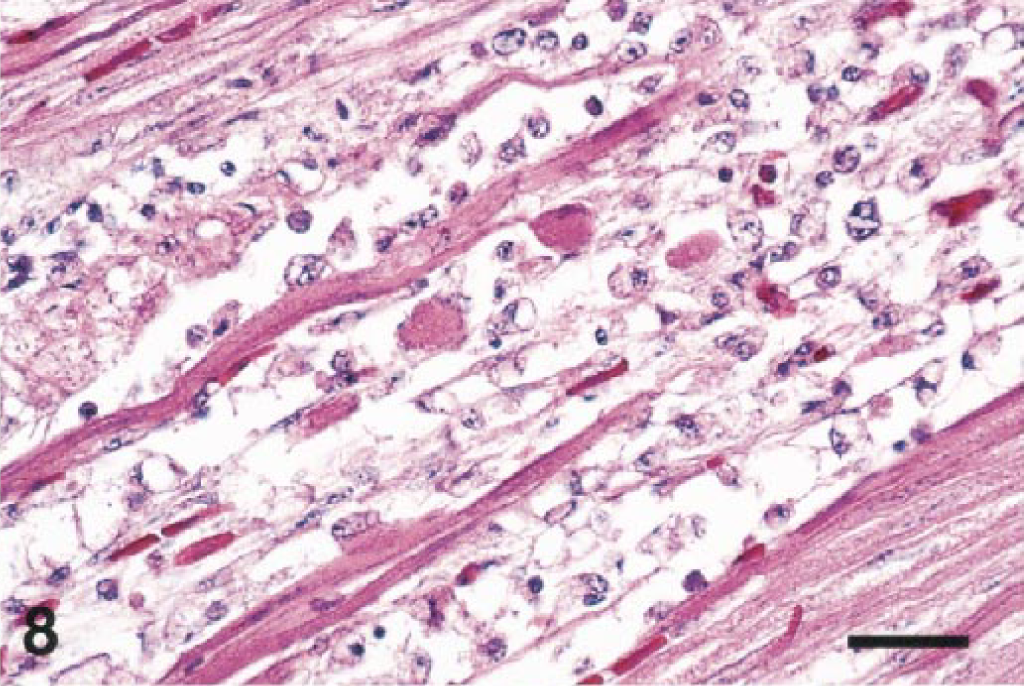
Heart; Chilean flamingo, bird No. 20. The myocardium is necrotic and contains an infiltrate of lymphocytes and macrophages. HE. Bar = 35 µm.
There was some degree of nonspecific chronic lymphoplasmacytic enterocolitis in 22 birds. However, nine birds also had more acute lesions attributed to WNV infection. These consisted of severe dilation and congestion of serosal and mucosal vasculature as well as that of the tunica muscularis. Several birds had moderate to severe infiltrates of heterophils admixed with fewer lymphocytes, plasma cells, and macrophages that markedly expanded the lamina propria. These birds (Nos. 2, 4, 5, 8) also had multifocal dilation and abscessation of crypts. Focal to multifocal acute hemorrhage was present in the proventricular glands of a crow (No. 17) and a duck (No. 12) and in the ventriculus of a flamingo (No. 4).
Severe lymphoid depletion was a prominent feature in the spleens of seven birds. The heron (No. 5) and a cormorant (No. 8; Fig. 9) had necrotizing splenitis with acute coagulative necrosis, fibrin deposition, and hemorrhage. All three ducks (Nos. 11–13) had massive splenic necrosis with vascular thrombosis and infarction. However, in two of these cases (Nos. 12, 13), bacterial colonies were found in association with the lesions.
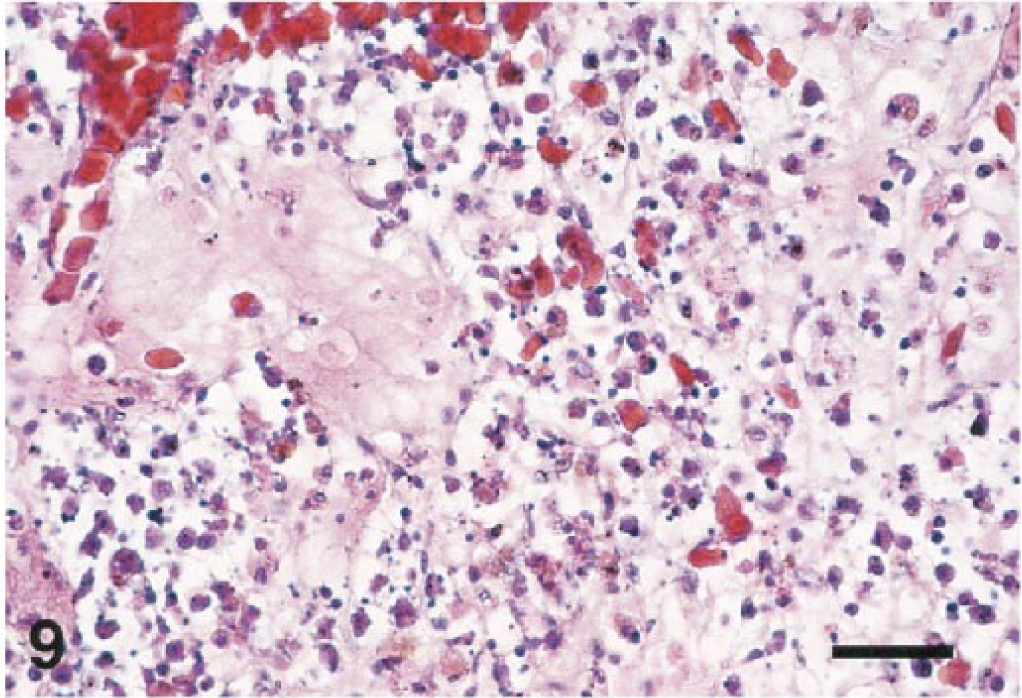
Spleen; Guanay cormorant, bird No. 8. Splenic necrosis is extensive, and there is edema and deposition of fibrin. HE. Bar = 50 µm.
Hepatic lesions occurred in 18 birds. Four birds (Nos. 8, 14, 16, 21) had acute multifocal coagulative necrosis of hepatocytes, and several birds had moderate to severe hepatic congestion. Mild to severe, periportal to random, chronic hepatitis was the most common liver lesion. Biliary hyperplasia was present in association with the hepatitis in two cases (Nos. 4, 6). Hemosiderin deposition in Kupffer cells and hepatocytes of varying severity was also a common finding.
Seven birds had pancreatitis (Fig. 10) of varying severity. The inflammatory infiltrates ranged from acute to chronic or chronic-active. In a few cases, the infiltrates focally obscured or effaced normal pancreatic architecture (Nos. 6, 20), or involved occasional islets (No. 7) as well as acinar cells. Pulmonary hemorrhage was present in 12 cases. It was usually multifocal and acute, and within parabronchial lumens.
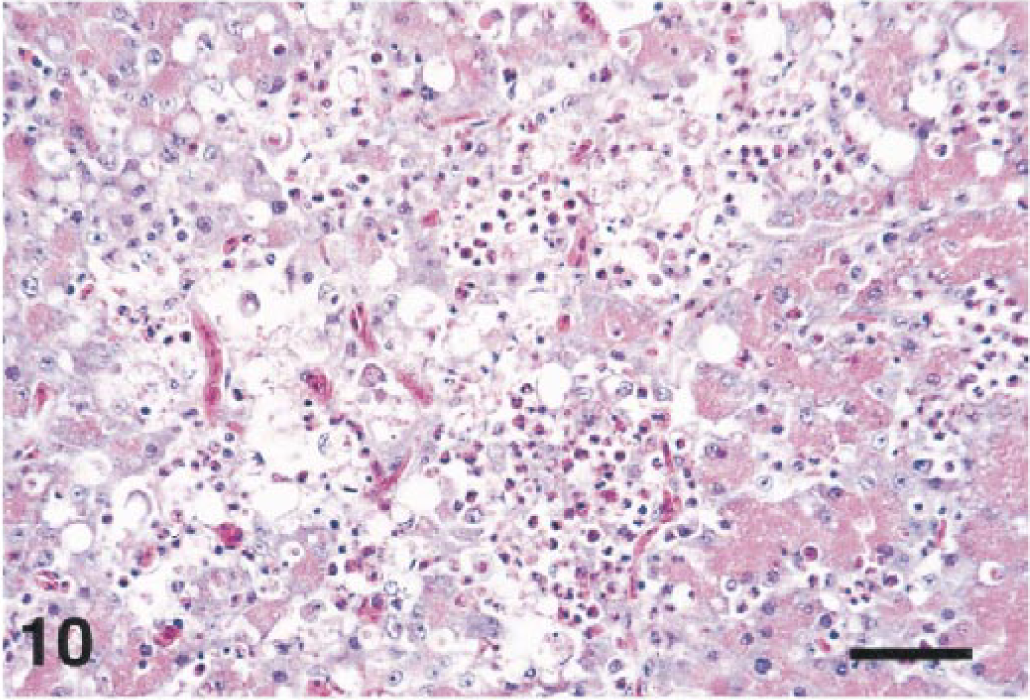
Pancreas; Chilean flamingo, bird No. 20. The pancreatic acini are necrotic, and a mixed inflammatory infiltrate is present. HE. Bar = 50 µm.
Seven birds had subacute to chronic inflammation of the adrenal gland, the pericapsular adrenal ganglion, or both. Four of these were crows (Nos. 1, 2, 15, 18). The amount of inflammation was generally mild and invariably associated with the ganglion and adjacent tissue. Cytoplasmic vacuoles were found in a ganglionic cell body in one crow (No. 15). Fifteen birds had lesions in the kidneys. These consisted of mild to moderate, subacute to lymphoplasmacytic, interstitial nephritis. There was little or no involvement of the tubules or glomeruli.
Virology
A variety of tissues from selected cases were assayed for infectious virus particles and WNV RNA (Table 2) with positive results. In particular, kidney and heart consistently produced infectious virus and positive RT-PCR results. In contrast, the liver failed to produce positive results in more than half of the specimens.
Immunohistochemical and virological analysis for West Nile virus in birds from the New York City outbreak, 1999. ∗
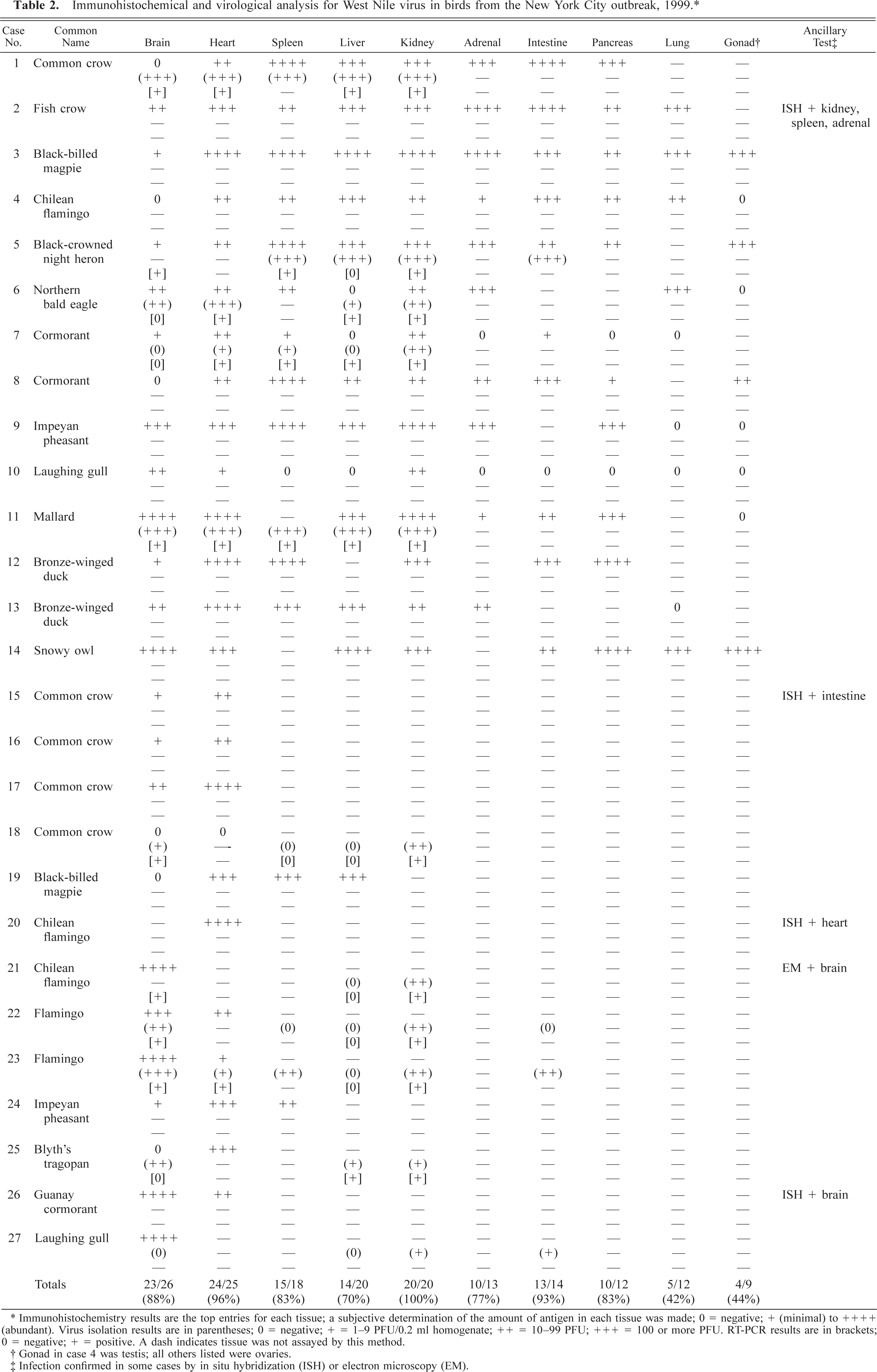
∗ Immunohistochemistry results are the top entries for each tissue; a subjective determination of the amount of antigen in each tissue was made; 0 = negative; + (minimal) to ++++ (abundant). Virus isolation results are in parentheses; 0 = negative; + = 1–9 PFU/0.2 ml homogenate; ++ = 10–99 PFU; +++ = 100 or more PFU. RT-PCR results are in brackets; 0 = negative; + = positive. A dash indicates tissue was not assayed by this method.
† Gonad in case 4 was testis; all others listed were ovaries.
‡ Infection confirmed in some cases by in situ hybridization (ISH) or electron microscopy (EM).
Immunohistochemistry and in situ hybridization
The alphavirus immunostains were uniformly negative. Immunostains of tissues from several birds (Nos. 4, 7–10, 15, 17, 20, 21, 26) were positive with the SLE virus antiserum (data not shown). Immunohistochemistry with the WNV antiserum demonstrated viral antigen in many tissues of these and additional birds (Table 2).
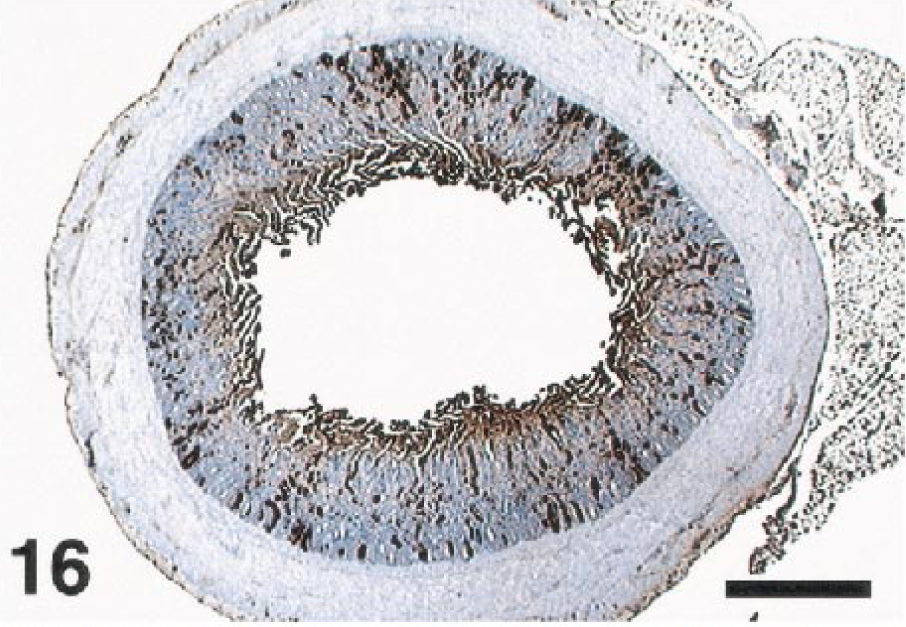
The brains of most birds were infected with WNV. Viral antigen was most common in the Purkinje cell and molecular layers of the cerebellum (Fig. 11). Antigen was also commonly present in the brain stem, primarily in neurons, but was scarce in cerebral neurons. Both Purkinje cell bodies and dendrites (Fig. 12) were often labeled, but antigen was seldom present in the axons. Basket cells, granule cells, glial cells of the molecular and white matter layers, and occasionally neurons of the cerebellar nuclei also contained antigen. The meninges of a few birds contained antigen, either in fibroblasts or macrophages. Viral infection of the brains of the crows and magpies was limited. When present, viral antigen in these birds was usually in neurons or glial cells of the brainstem, it but was extremely rare in the Purkinje cells.
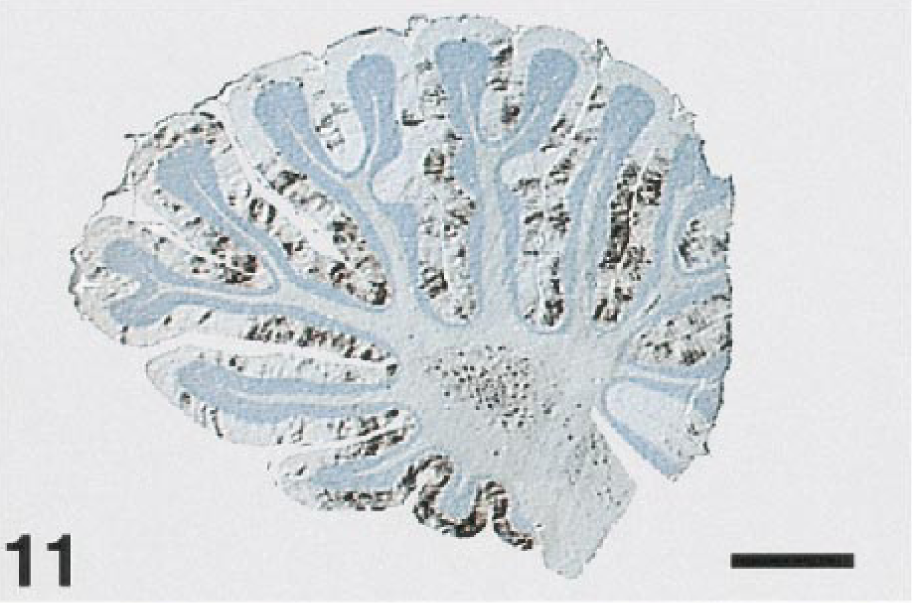
Cerebellum; laughing gull, bird No. 27. Abundant WNV antigen decorates the Purkinje cell and molecular layers of the cerebellar folia. Immunoperoxidase labeling, hematoxylin counterstain. Bar = 1400 µm.
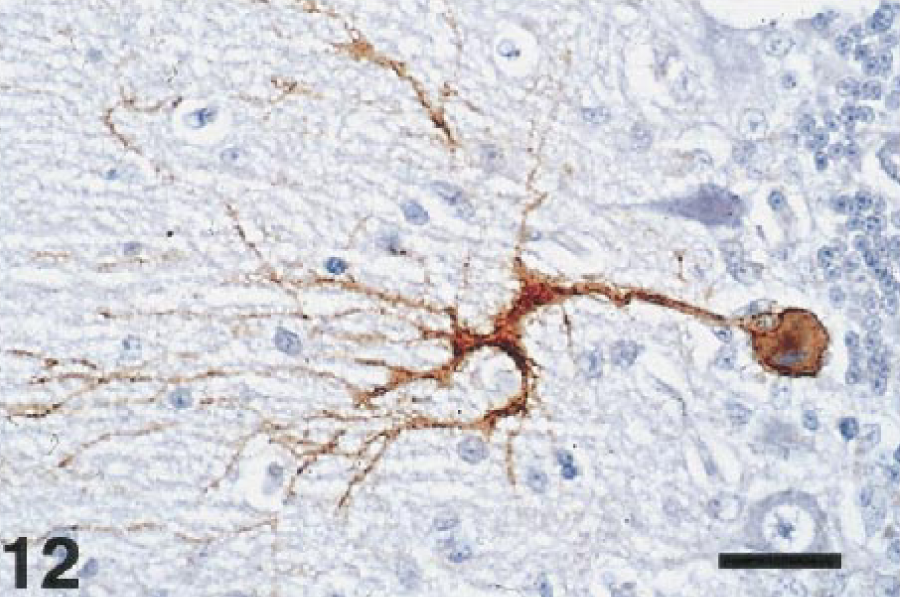
Cerebellum; laughing gull, bird No. 10. WNV antigen is present in the cell body of a Purkinje cell and the arborizing dendrites. Immunoperoxidase labeling, hematoxylin counterstain. Bar = 38 µm.
The spinal cord and the peripheral nervous system also contained WNV antigen. In the spinal cord, antigen was present mainly in the gray matter neurons. In the peripheral nervous system, the myenteric plexus and peripheral ganglia of several birds was immunolabeled. Both peripheral neurons and Schwann cells contained antigen.
Viral antigen was common in the heart and was abundant in the hearts of several birds. Myofibers were usually labeled (Fig. 13). Macrophages in foci of necrosis and inflammation, as well as interstitial fibroblasts, were commonly labeled as well. Smooth muscle in the walls of the gut or occasional blood vessels contained antigen in a few cases.
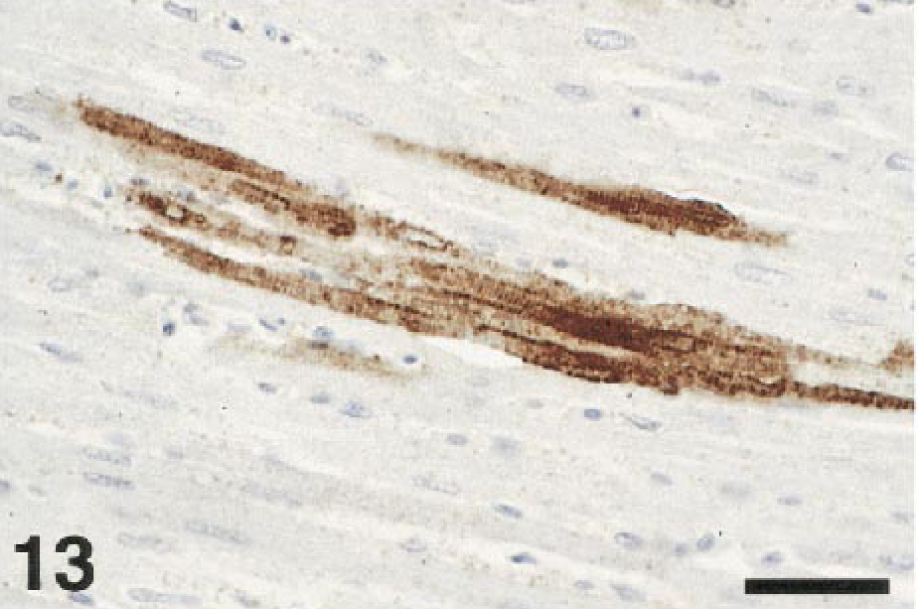
Heart; crow, bird No. 15. Immunolabeling indicates viral infection of the myocardial fibers. Immunoperoxidase labeling, hematoxylin counterstain. Bar = 38 µm.
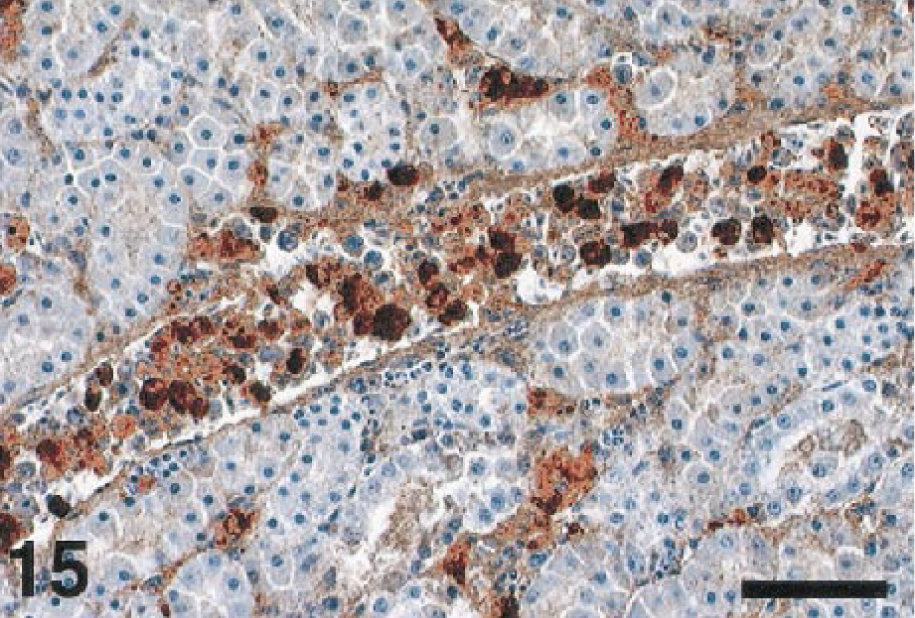
Tissue macrophages and blood monocytes appeared to be significant targets of the virus. Splenic macrophages (Fig. 14), Kupffer cells, blood monocytes (Fig. 15), and macrophages in various interstitial connective tissues, as well as macrophages in a variety of inflammatory lesions, often contained WNV antigen. Macrophages and blood monocytes were strongly immunolabeled in the tissues of the crows and magpies in particular.
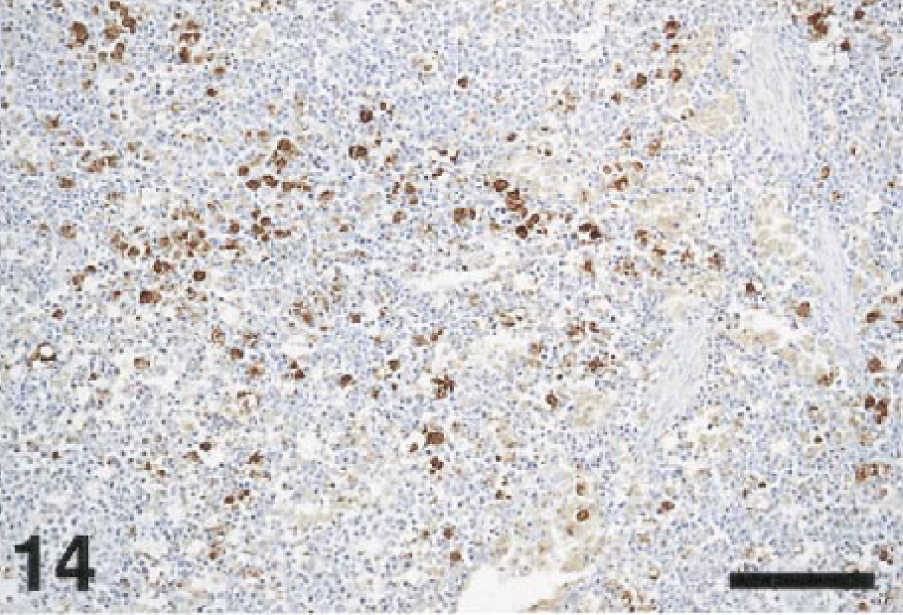
Spleen; cormorant, bird No. 8. Macrophages containing WNV antigen are evenly scattered throughout the spleen. Immunoperoxidase labeling, hematoxylin counterstain. Bar = 50 µm.
Kidney; crow, bird No. 1. A renal vessel contains numerous, WNV antigen–laden monocytes. Immunoperoxidase labeling, hematoxylin counterstain. Bar = 60 µm.
In the intestine, the crypt epithelial cells (Fig. 16) were common targets for WNV infection. Macrophages, fibroblasts, and the myenteric plexus also contained antigen. In the kidney, antigen was present in the epithelium of renal tubules and collecting ducts, in interstitial fibroblasts and macrophages, and in glomeruli. In some birds, WNV antigen in the kidney appeared to predominate in either the renal tubules or the interstitium, whereas in other birds, the pattern of antigen localization was mixed.
Intestine; fish crow, bird No. 2. Immunolabeling indicates viral infection of the epithelium of numerous intestinal crypts. Immunoperoxidase labeling, hematoxylin counterstain. Bar = 650 µm.
The pancreas of several birds contained WNV antigen. Pancreatic acinar cells were usually immunolabeled, along with a few pancreatic islets. In several cases, the majority of antigen in the pancreas was within necrotic foci, so identification of infected cell types was not possible. Epithelial cells lining the air capillaries of the lungs contained antigen in some birds. WNV antigen in the adrenal gland was usually present in cells that contained abundant cytoplasmic vacuoles, indicating they were steroid-producing cells. In the ovary, oocytes, the surrounding follicular cells, thecal cells, and stromal cells variably contained antigen. Although macrophages in the liver and spleen were often heavily laden with WNV antigen, immunolabeled hepatocytes were uncommon, and positive splenic lymphocytes were not evident. We did not specifically identify endothelial cells as a target of infection. However, the extensive antigen present in some tissues and intravascular cells precluded ruling out infection of endothelial cells.
Immunostaining also identified antigen-containing cells in the impression smears from a few birds (data not shown). These included smears of spleen from two crows (Nos. 1, 2) that had positive splenic tissue immunostains, coelomic fluid from a Chilean flamingo (No. 4) with multiple positive tissues, and spleen and liver from a crow and a blue jay, respectively, for which tissue sections were not processed. In all cases, most of the positive cells appeared to be histiocytic. Smears from a few other birds had equivocally positive cells. Four birds (Nos. 9, 12, 13, 24) yielded splenic smears in which no immunolabeled cells were identified, even though the tissue sections of their spleens were positive.
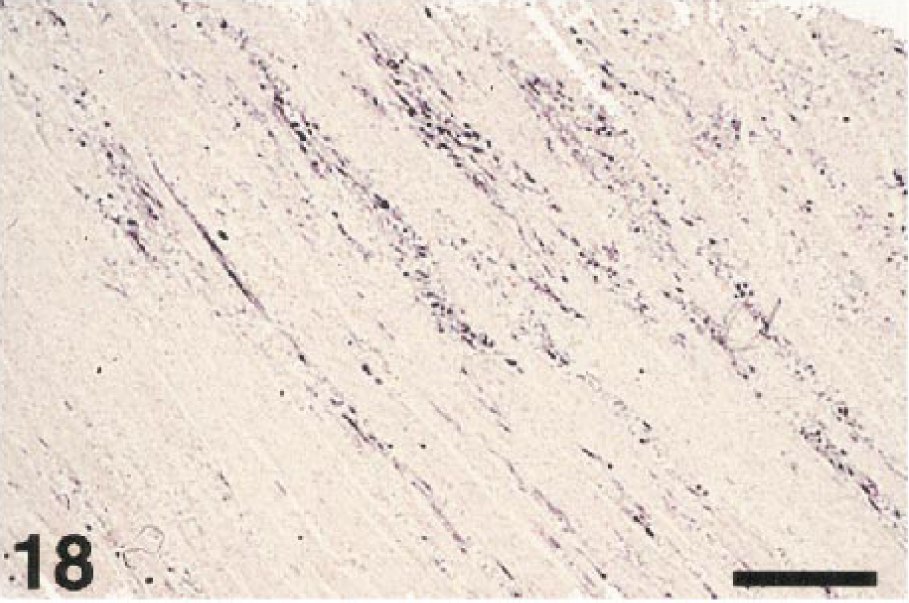
Specificity testing demonstrated that the rabbit antibody to WNV cross-reacted with other flaviviruses (data not shown). Relatively strong immunolabeling was present in various tissues of the suckling mice infected with SLE strains TBH-28 (mouse No. 2) and MSI-7 (mouse No. 3), as well as the mouse infected with WNV (mouse No. 1). The sections of brain from animals infected with TBE viruses had weak to light immunolabeling. The in situ hybridization assay was significantly more specific. The pWNV-E probe strongly labeled tissues of the mouse infected with WNV (Fig. 17). It did not label any tissues in the mice infected with the two SLE strains or tissues infected with the TBE viruses or Venezuelan equine encephalitis (VEE) virus. Analysis of selected bird tissues by in situ hybridization (Table 2) confirmed the immunostaining results (Fig. 18).
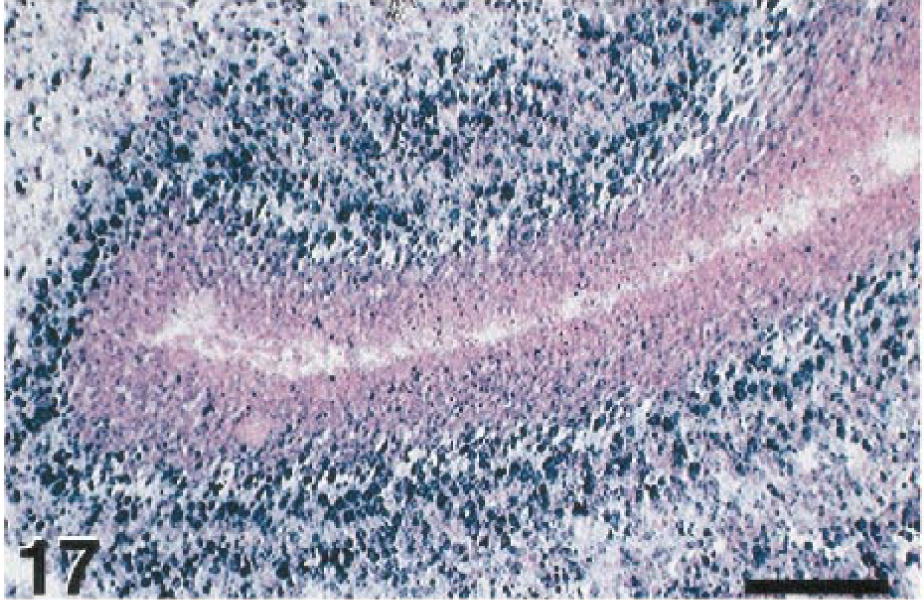
Cerebellum; suckling mouse, mouse No. 1. Using the WNV E gene probe to detect WN-USAMRIID99, extensive hybridization confirms experimental infection of this mouse. In situ hybridization, NBT-BCIP chromogen, nuclear fast red counterstain. Bar = 100 µm.
Heart; Chilean flamingo, bird No. 20. WNV is demonstrated in multiple myocardial foci. In situ hybridization, NBT-BCIP chromogen, nuclear fast red counterstain. Bar = 400 µm.
Electron microscopy
Flavivirus-like particles were frequently seen in the cerebrum and cerebellum of a Chilean flamingo (No. 21, Fig. 19) and were infrequently observed in the heart of this bird. The particles measured 35–45 nm in diameter and showed typical flaviviral morphology, that is, a dense core surrounded by a thin, diffuse outer layer. The particles were usually present in cytoplasmic vacuoles in the perikaryon and in neuritic processes. Virions were less frequent in dilated endoplasmic reticulum (rER) of the perikaryon and extracellular spaces in the neuropil. Some virions were present in vacuoles of stromal cells in the connective tissue septa of the heart. In addition to virions, membranous vesicles measuring about 100 nm in diameter were in dilated rER and vacuoles (Fig. 19). These vesicles were identical to the smooth membrane structures described by other investigators in the rER cisternae of flavivirus-infected cells. 13 , 22 , 23 Nonstructural proteins of Kunjin virus, a WNV subtype, 12 were recently associated with the smooth membrane structures. 23 Aggregates of electron-dense granules associated with dense membrane vesicle structures (MVS) and convoluted membranes (CM), consistent in morphology with flavivirus-induced structures described in Japanese encephalitis virus-infected neurons 22 and Kunjin virus-infected Vero cells, 23 were present in the perikaryon of a few neurons. These dense MVS were also infrequently seen in oligodendrocytes of the Impeyan pheasant (No. 9). Features associated with cells containing flavivirus-like particles or flavivirus-induced structures included disorganization of the rER and Golgi apparatus, and marked vesiculation and vacuolation of the cytoplasm. Some sections of brain from the Chilean flamingo exhibited vacuolar breakdown of myelin sheaths. Flavivirus-like particles or intracellular inclusion bodies were not evident in the examined specimens of the other birds.
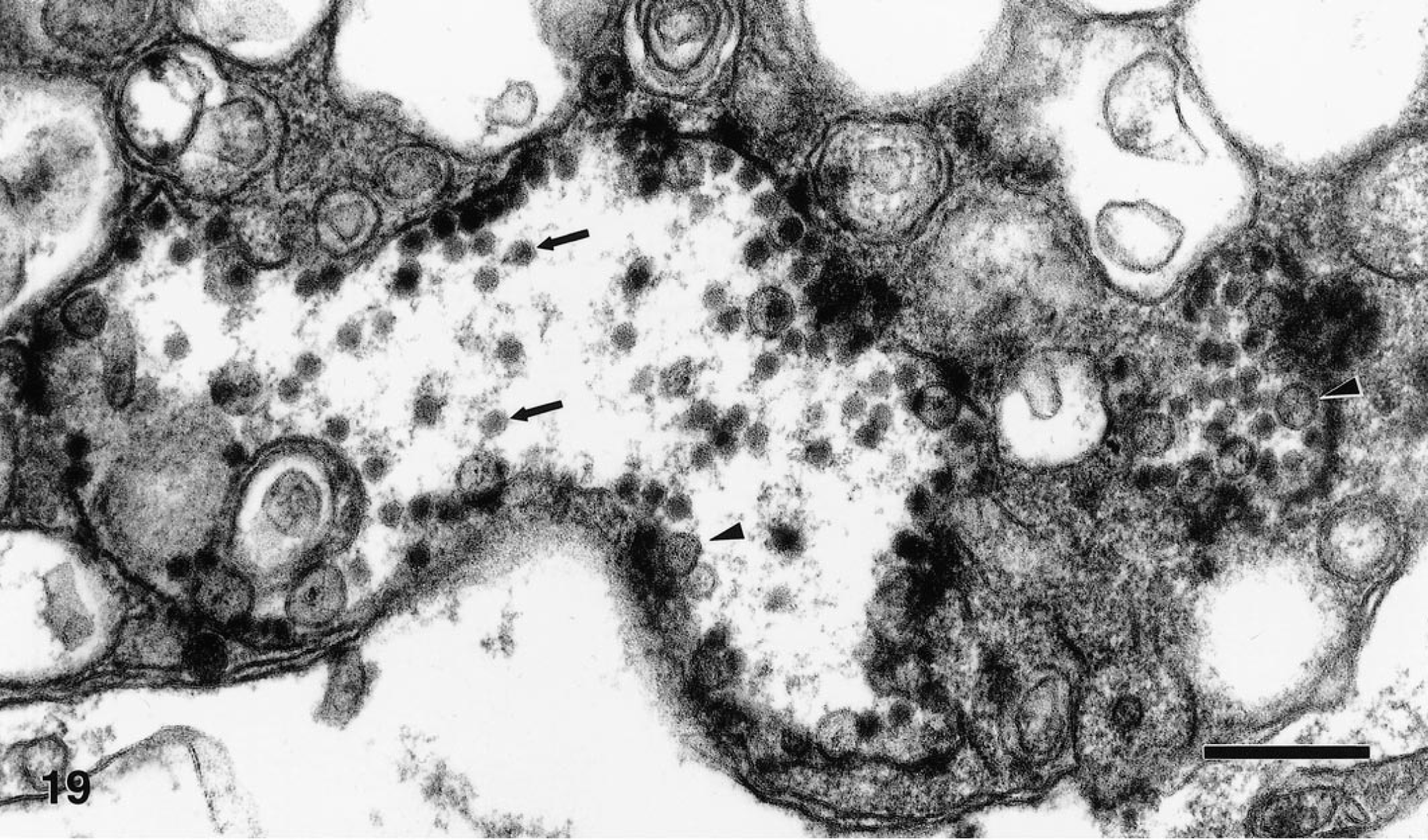
Ultrathin section of a neuron in the cerebellum of a Chilean flamingo. Vacuoles contain numerous virions (arrows) and characteristic smooth membrane structures (arrowheads). Bar = 250 nm.
Postembedment immunoelectron microscopy of cerebrum and cerebellum from the Chilean flamingo showed diffuse gold-sphere labeling of some neuronal processes and intense labeling of amorphous electron-dense material and associated membranes in the perikaryon and neuronal processes. It was impossible to associate the diffuse labeling in the neuronal processes with distinct structures. However, labeling was specific for WNV as many processes in the same section were unlabeled, and no labeling was observed when sections were incubated with nonimmune mouse ascitic fluid. Virions were considerably less intensely labeled than the presumed flaviviral-induced dense MVS and CM. These findings were consistent with immunoelectron microscopy of WNV-infected Vero cells. In the Vero cells, intracellular dense MVS and associated CM were heavily decorated with gold spheres (Fig. 20), whereas labeling of virions was sparse.
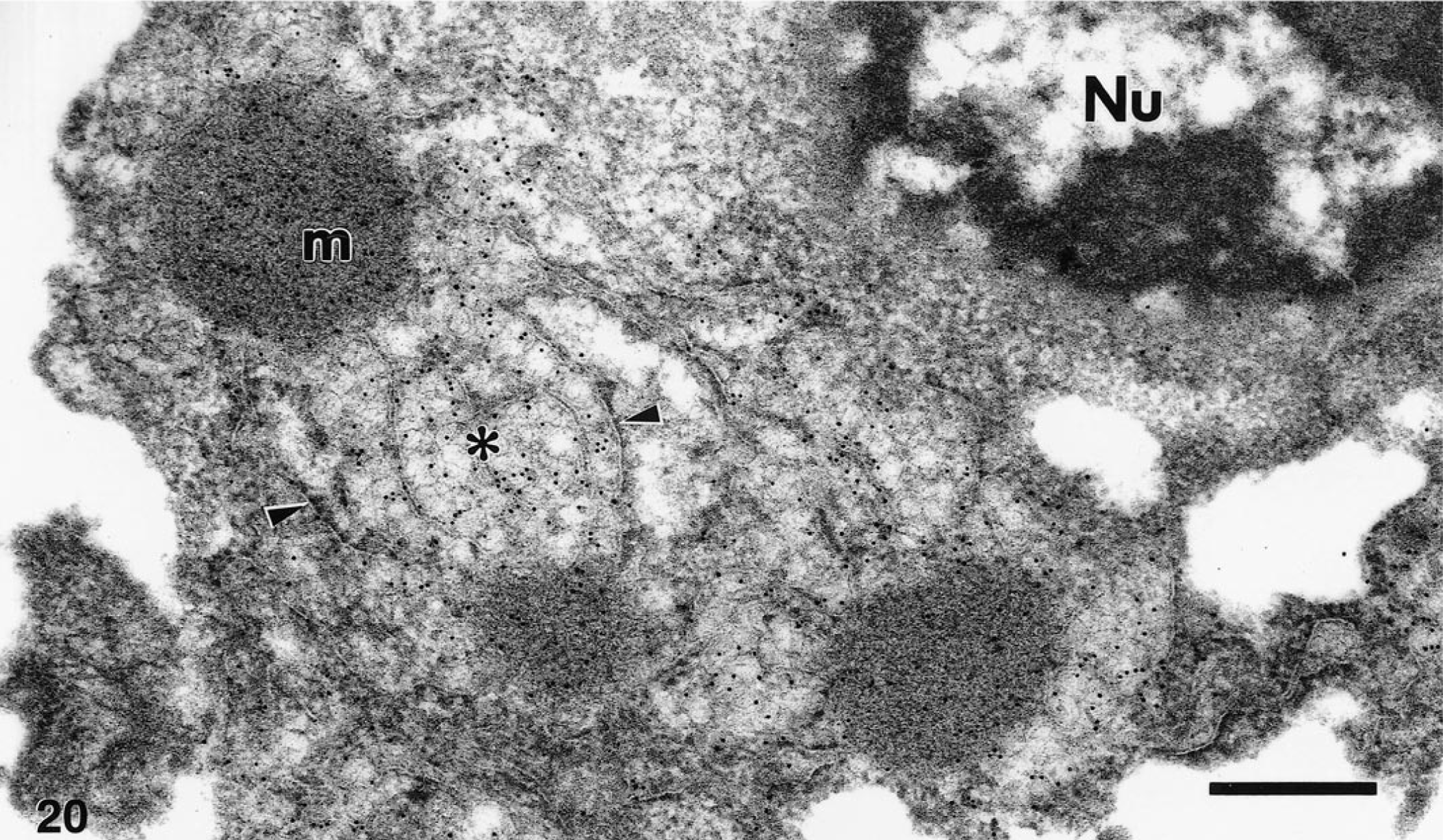
Postembedment immunoelectron microscopy of WNV-infected Vero cell. Gold spheres were strongly associated with dense membrane vesicle structures (m) and convoluted membranes (∗), and were occasionally localized with membranes of the endoplasmic reticulum (arrowheads) surrounding these structures. Nu, nucleus. Bar = 370 nm.
Discussion
The sudden emergence of West Nile fever in the northeastern United States in 1999, the first time this disease has ever been seen in the western hemisphere, was a dramatic and unexpected event. That numerous birds succumbed to disease was a surprising characteristic of this outbreak. Although crows and house sparrows die following experimental infection, 24 free-ranging birds have not been previously reported to be significantly affected by WNV. In this report, we document that the 1999 WNV outbreak caused disease, severe pathologic changes, and death in a variety of birds. We also show that the virus that caused this outbreak demonstrated a nonrestricted tropism in affected birds, infecting essentially all major organ systems and a wide variety of individual cell types. Finally, we present diagnostic methods that may be useful to pathologists and others who will play important roles in the public health response should WNV reemerge in 2000 or subsequent years.
Specific pathologic changes and WNV infection of multiple tissues occurred in one or more birds of eight orders representing 14 species. The virus exhibited tropism for the central and peripheral nervous systems, the myocardium, cells of the mononuclear phagocyte system (MPS), multiple epithelial cell types, fibrous connective tissues, and oocytes. The unprecedented pathogenicity observed in birds during this outbreak is surprising, given that WNV rarely appears to cause avian disease. The reason for this is unclear. Presumably, birds along the eastern seaboard of the United States represented a population with little or no coevolutionary adaptation to WNV. Perhaps a more likely explanation is that the strain of WNV involved in this outbreak is a recently evolved genetic variant with greater pathogenicity for a variety of birds. This possibility is supported by the recent finding that the New York 99 strain of WNV (WN-NY99) is similar to a strain of WNV isolated from a goose in Israel in 1998 (WN-Israel 1998) that has demonstrated significant pathogenicity for birds. 12 The continued epornitic of WNV in Israel during 1999 due to this novel strain 16 suggests that WNV could continue to infect and cause disease in birds in the United States as well.
The brain was a significant target of infection in many of the birds in this study. In particular, the cerebellum was specifically targeted. Viral antigen was most heavily and consistently present in the cell bodies and dendrites of Purkinje cells. Gross and histologic lesions were common in the cerebellum of many birds as well, including hemorrhage in the molecular and granular layers, Purkinje cell necrosis and dropout, gliosis, and inflammatory infiltrates. Similar changes also occurred in other portions of the brain but were usually less severe than in the cerebellum. As a group, the crows and magpies did not exhibit the extensive infection of the brain seen in other birds. These birds had relatively limited viral antigen in the brain and relatively mild histologic changes. Interestingly, the virus appeared to spare the cerebellum of the crows and magpies, although limited infection of the brain stem was observed. The reason for the apparent decreased neurovirulence in these closely related corvids is unclear.
The neurologic signs seen in those birds for which clinical signs were recorded appear somewhat inconsistent with our finding that the cerebellum was significantly affected. Since Purkinje cells function to modulate or dampen motor responses, ataxia is a cardinal sign of cerebellar dysfunction. Yet only 3 of 17 birds exhibited ataxia. The most common clinical signs were weakness and recumbency, ones not typical of cerebellar disease. However, since there was abundant virus and often severe lesions in most major organs, signs of cerebellar disease may have been masked by the effects of extraneural infection.
Gross and histologic lesions were seen in many extraneural locations where viral antigen occurred. Although several of the birds had nonspecific lesions in a variety of tissues consistent with their advanced age, lesions considered to be specifically virus-induced were present in the heart, spleen, pancreas, and adrenal glands. Lesions in these tissues were characteristically necrotizing and contained inflammatory infiltrates ranging from mixed to lymphoplasmacytic. Similar inflammatory changes which were less necrotizing were seen in the liver, kidney, and gastrointestinal tract, as well as peripheral ganglia.
Extensive changes to the heart, pancreas, kidney, adrenal gland, and liver might result in a variety of physiologic alterations possibly leading to recumbency and weakness, the main clinical signs noted in these birds. Ultimately, a combination of neural and extraneural alterations probably contributed to the demise of many of the birds.
Meningoencephalitis and necrotizing myocarditis, in particular, should alert pathologists to the possible presence of WNV in birds. The differential diagnosis for encephalitis and myocarditis in birds should include eastern equine encephalitis virus. Eastern equine encephalitis was ruled out early in the course of this study by immunostaining tissues from affected birds. Avian influenza and exotic Newcastle's disease should also be considered in the differential diagnosis. The SLE virus, which frequently infects birds, is not expected to cause overt illness.
The targeting of cells of the MPS by WNV may have played a significant pathogenetic role in the affected birds. Replication of the virus within these cells and dissemination throughout the body via mobile MPS cells could be of primary importance in the wide variety of tissues infected. In addition, macrophage dysfunction and infection of connective tissues, as was present in many birds, may have contributed to the hemorrhagic manifestations by triggering mediators of the coagulation system. Although endothelial cell damage cannot be ruled out, we did not observe virus targeting or specific damage of endothelial cells. Virus infection of macrophages may have had other negative consequences as well, such as inducing the elaboration of deleterious inflammatory mediators or disruption of the host immune response.
IHC provided a valuable asset in this study in that many tissues could be efficiently analyzed. In 25 instances where replicate samples of the same tissue were analyzed by IHC, virus isolation (VI) and RT-PCR, virus was demonstrated by all three methods in 17 instances (68 %). Of the eight instances where the three methods did not agree, five represented samples of brain. Since IHC demonstrated that antigen was often localized in particular regions of the brain in specific birds, these apparent discrepancies may be due to different portions of the brain being analyzed by different methods. Differences in the sensitivities of the three methods could also explain these discrepancies. Nonetheless, our results show that IHC is a reliable and efficient means of demonstrating the presence of WNV in formalin-fixed, routinely processed tissues.
A potential weakness of the IHC method we employed is its relative lack of specificity. The rabbit polyclonal antibody we used cross-reacted with other flaviviruses, including significant crossreactivity with SLE virus and weak to mild crossreactivity with three members of the TBE group of flaviviruses. Crossreactivity was not considered a shortcoming in the present study since the purpose of immunostaining was to demonstrate the presence of WNV antigen in tissues, whereas other means were relied upon for specificity. Nonetheless, specificity might be important in other situations, such as when only formalin-fixed, paraffin-embedded tissues are available and methods such as RT-PCR or VI are not feasible. The use of WNV-specific monoclonal antibodies could answer this concern, but we are not aware of any monoclonal antibodies that are reported to detect antigen in formalin-fixed tissues. Our in situ hybridization method, however, provided much greater specificity than the IHC method. The pWNV-E probe reacted with the WNV strain responsible for the 1999 outbreak (WN-USAMRIID99), from which it was developed, but did not react with two different strains of SLE virus or any of the TBE viruses. We have yet to test the in situ probe with other strains of WNV.
Another potential of IHC which requires further study is the possibility that tissue smears might make a very rapid screening test that could be performed at the time of necropsy. Although we detected antigen-positive cells in a few smears, the numbers are not yet sufficient to adequately determine the usefulness of this approach. Our results indicate, however, that heart, kidney, or spleen would be the best candidates for testing tissue smears. These tissues are easily obtained at necropsy. Also, when we tested replicate samples by tissue section IHC, VI, and RT-PCR, the heart, kidney, and spleen were consistently positive by all three methods.
Should WNV reappear in the United States and possibly spread from the region affected during the 1999 outbreak, veterinary and other medical diagnosticians will be challenged to provide an early, accurate diagnosis. Birds exhibiting unexplained neurologic signs or evidence of meningoencephalitis and myocarditis should be tested for WNV by available means. Caution is urged in handling suspect cases, however, since WNV is a known human pathogen. It is currently listed as a biosafety level 3 agent, and cases of laboratory-acquired infections have been known to occur. 18
Footnotes
Acknowledgements
The authors thank Dr. T. Wilson for assistance with necropsies, Dr. G. Ludwig for providing suckling mice, C. Rossi for providing diagnostic antisera, J. Hauer (Mayor's Office of Emergency Management, the City of New York), J. Anelli, D. Wilson (Emergency Programs of the USDA), and NVSL for their continuing support, and A. Ngbokoli, A. Goldman, J. Tonry, J. Brubaker, R. Moxley, A. Castillo, A. Thomas, D. Sledge, and L. Ostby for expert technical assistance.
In conducting research using animals, the investigators adhered to the Guide for the Care and Use of Laboratory Animals (Committee on Care and Use of Laboratory Animals of the Institute of Laboratory Animal Resources, National Research Council, NIH Publication No. 86-23, revised 1996).
