Abstract
Mycobacterium bovis is a serious zoonotic pathogen and the cause of tuberculosis in many mammalian species, most notably, cattle. The hallmark lesion of tuberculosis is the granuloma. It is within the developing granuloma where host and pathogen interact; therefore, it is critical to understand host-pathogen interactions at the granuloma level. Cytokines and chemokines drive cell recruitment, activity, and function and ultimately determine the success or failure of the host to control infection. In calves, early lesions (ie, 15 and 30 days) after experimental aerosol infection were examined microscopically using in situ hybridization and immunohistochemistry to demonstrate early infiltrates of CD68+ macrophages within alveoli and alveolar interstitium, as well as the presence of CD4, CD8, and γδ T cells. Unlike lesions at 15 days, lesions at 30 days after infection contained small foci of necrosis among infiltrates of macrophages, lymphocytes, neutrophils, and multinucleated giant cells and extracellular acid-fast bacilli within necrotic areas. At both time points, there was abundant expression of the chemokines CXCL9, MCP-1/CCL2, and the cytokine transforming growth factor (TGF)–β. The proinflammatory cytokines tumor necrosis factor (TNF)–α and interleukin (IL)–1β, as well as the anti-inflammatory cytokine IL-10, were expressed at moderate levels at both time points, while expression of IFN-γ was limited. These findings document the early pulmonary lesions after M. bovis infection in calves and are in general agreement with the proposed pathogenesis of tuberculosis described in laboratory animal and nonhuman primate models of tuberculosis.
Bacteria of the genus Mycobacterium are nonmotile, nonsporulating, slow-growing, Gram-positive, acid-fast bacilli (AFB) that include major human and animal pathogens. 48 Tuberculosis in humans is primarily caused by Mycobacterium tuberculosis; however, indistinguishable disease can be caused by the zoonotic agent Mycobacterium bovis, 30 the most common cause of tuberculosis in animals, most notably cattle. Worldwide, it is estimated that 50 million cattle are infected with M. bovis, with agricultural losses of $3 to $4 billion annually. 52 The pathogenesis of bovine tuberculosis parallels many aspects of human tuberculosis, and cattle have been a useful model of human tuberculosis. 60 Although both diseases have been known for centuries, their pathogeneses are not completely understood but are known to involve lungs and regional lymph nodes in a coordinated exchange of cells under the influence of cytokines, chemokines, and other mediators.
M. bovis and M. tuberculosis are members of the M. tuberculosis complex, which also includes Mycobacterium africanum, Mycobacterium canetti, Mycobacterium microti, Mycobacterium caprae, Mycobacterium pinnipedii, and Mycobacterium mungi. 44 The hallmark lesion of tuberculosis, regardless of host or tissue, is the granuloma. A granuloma resulting from infection with a member of the M. tuberculosis complex may also be referred to as a tubercle or tuberculoid granuloma. 35 The tuberculoid granuloma is a morphologically distinctive lesion in which the predominant cell type is a modified epithelial-like (epithelioid) macrophage. Other prominent cell types found within granulomas are lymphocytes and multinucleated giant cells, thought to be formed through the fusion of multiple macrophages, although recent findings suggest multinucleated giant cells may form through a distinct macrophage differentiation pathway induced by persistent inflammation. 9,24 The prototypical tuberculoid granuloma has a central region of caseous necrosis, surrounded by epithelioid macrophages and multinucleated giant cells, with an outermost zone containing increasing numbers of lymphocytes and occasional plasma cells. The entire mass of necrotic tissue and inflammatory cell infiltrate is often encased within a variably thick fibrous capsule.
The tuberculoid granuloma is dynamic, both growing and shrinking over time. 23 It is where key interactions between host and pathogen determine disease containment or dissemination, bacterial replication, killing, or latency. 15,38 The factors that lead to disease containment or progression are not well understood. The granuloma can function both as a niche for pathogen persistence as well as a hostile mycobactericidal focus of infection control; consequently, granulomas are heterogenous. Individual granulomas within the same host and tissue can have different fates, suggesting that local rather than systemic responses determine outcome. Therefore, to understand tuberculosis pathogenesis, it is imperative to understand granuloma-level immune responses. 5 –7,19,22
Cattle studies describing granuloma-level responses have demonstrated the presence of various cytokines, chemokines, and enzymes using immunohistochemistry (IHC), in situ hybridization (ISH), and quantitative real-time polymerase chain reaction (qPCR) combined with laser capture microscopy (LCM).* Previous studies in cattle have examined tuberculoid granulomas in later stages of disease from 13 to 29 weeks after infection, 2,26,45,47,59 with the exception of Cassidy et al, 8 who evaluated lung lesions as early as 7 days following intranasal inoculation with 1 × 107 colony-forming units (CFU).
In cattle, as in humans, it is believed cell-mediated immunity, in contrast to humoral immunity, is central to the response to tuberculous mycobacteria. Indeed, a measurable cell-mediated response to mycobacterial antigens is the principle underlying both the tuberculin skin test and interferon γ (IFN) release assays, used in both human and bovine tuberculosis diagnosis. 46,56 Important in the cell-mediated antimycobacterial response of cattle are T cells, specifically, CD4, CD8, and γδ T cells. 25 These cell types can all be producers of IFN-γ, production of which is a key feature of the peripheral response to M. bovis infection in cattle. 41 Together with IFN-γ, tumor necrosis factor–α (TNF-α) is crucial for control of mycobacterial infections and critical to the formation of organized granulomas. 20 In contrast, interleukin (IL)–10 is seen to downregulate IFN-γ production. 4
In cattle, the events involving these critical cellular and biochemical responses at the site of infection during the first 30 days following aerosol infection with M. bovis remain largely unknown. Understanding these early host-pathogen interactions may be key in development of improved diagnostic assays or possible vaccines. The aim of the present study was to evaluate pulmonary lesions of cattle 15 and 30 days after aerosol inoculation with virulent M. bovis, including gross and microscopic findings, cellular composition, and expression of cytokines and chemokines of known or implied relevance to bovine tuberculosis.
Materials and Methods
Mycobacterium bovis Aerosol Challenge
M. bovis strain 10-7428 was used in this experiment. This field strain was of low passage (<3) and had been shown previously to be virulent in the calf aerosol model. 61 Inoculum was prepared using standard techniques 31 in Middlebrook’s 7H9 liquid media (Becton Dickinson, Franklin Lakes, NJ) supplemented with 10% oleic acid-albumin-dextrose complex (OADC; Difco, Detroit, MI) plus 0.05% Tween 80 (Sigma Chemical Co., St. Louis, MO). Mid-log-phase growth bacilli were pelleted by centrifugation at 750 × g, washed twice with phosphate-buffered saline (PBS) (0.01 M, pH 7.2), and stored at –80°C until used. Frozen stock was warmed to room temperature and diluted to the appropriate cell density in 2 ml PBS. Bacilli were enumerated by serial dilution plate counting on Middlebrook’s 7H11 selective media (Becton Dickinson). A single dose was determined to be 1.12 × 104 colony-forming units (CFU) per calf.
Aerosol infection of calves with virulent M. bovis has been described in detail previously. 36,39,61 Briefly, 10 Holstein calves (9 months of age, castrated males) were infected with a single dose of virulent M. bovis strain 10-7428 by nebulization of inoculum into a mask (Equine AeroMask; Trudell Medical International, London, ON, Canada) covering the nostrils and mouth. Six additional age-matched calves were used as noninfected controls. All experimental animal procedures were conducted in accordance with recommendations in the Care and Use of Laboratory Animals of the National Institutes of Health and the Guide for the Care and Use of Agricultural Animals in Research and Teaching. 16,21 Procedures were also approved by the US Department of Agriculture (USDA)–National Animal Disease Center Animal Care and Use Committee.
Sample Collection
Five calves each were euthanized at 15 and 30 days after infection; 3 noninfected control calves were also euthanized at each time point. Calves were humanely euthanized by intravenous administration of sodium pentobarbital. Tissues were examined for gross lesions and processed for microscopic analysis as described previously. 61 Tissues collected included lung, liver, and medial retropharyngeal, mediastinal, tracheobronchial, hepatic, and mesenteric lymph nodes. Lymph nodes were sectioned at 0.5-cm intervals and examined. Each lung lobe was examined separately and sectioned at 0.5- to 1.0-cm intervals. Lesions suspected to be early granulomas were dissected out and processed as individual samples. A maximum of 5 samples each were collected from the right and left caudal lung lobes. Tissue samples were collected (≤0.5 cm in width) and fixed by immersion in 10% neutral buffered formalin (≥20 volumes fixative to 1 volume tissue) for approximately 24 hours and transferred to 70% ethanol, followed by standard paraffin embedding techniques. Paraffin-embedded samples were cut in 4-μm-thick sections, transferred to Superfrost Plus charged microscope slides (Thermo Fisher Scientific, Pittsburg, PA), and stained with hematoxylin and eosin (HE). Adjacent sections were stained by the Ziehl-Neelsen technique for visualization of acid-fast bacilli (AFB). Numerous near-adjacent unstained sections were used for IHC and ISH cytokine expression analysis.
Microscopic Examination
In cattle, pulmonary lesions as early as these have not been staged or categorized previously; as such, previously used methods for categorization of granuloma types (ie, stages I to IV) 36,37,59 in cattle were insufficient. Rather, the descriptive nomenclature used for early lung lesions in rhesus macaques (Macaca mulatta) experimentally infected with M. tuberculosis was more appropriate. 42 Type 1 lesions were unorganized, lacking defined boundaries or peripheral lymphocyte-rich zones and were composed of small single or multiple foci of macrophages and few lymphocytes that expanded alveolar septa and extended into the alveoli. Type 2 lesions were composed of similar inflammatory cells as type 1 lesions, still lacking a peripheral lymphocyte-rich zone, but were more circumscribed and roughly circular with variably demarcated borders compared to type 1 lesions. Type 3 lesions were similar to type 2 lesions but contained small areas of necrosis, characterized by loss of cellular detail, nuclear pyknosis, and karyorrhexis. Type 4 lesions were organized, well-circumscribed granulomas consisting primarily of macrophages admixed with lesser numbers of granulocytes and variable numbers of peripheral lymphocytes (nonnecrotizing granulomas). Type 5 lesions were similar to type 4 lesions but exhibited central necrotic foci (necrotizing granulomas).
Chromogenic In Situ Hybridization
RNAscope ZZ probe technology (Advanced Cell Diagnostics, Newark, CA) was used to perform messenger RNA (mRNA) ISH in formalin-fixed, paraffin-embedded (FFPE) tissue sections using the RNAscope 2.5 HD Reagents–RED kit (Advanced Cell Diagnostics). Proprietary ZZ probes complementary to mRNA sequences of interest were used for visualization of mRNA transcripts for the following cytokines (Suppl. Table S1): IFN-γ, TNF-α, IL-10, transforming growth factor–β (TGF-β), macrophage chemoattractant protein-1/chemokine (C-C motif) ligand 2 (MCP-1/CCL2), interleukin-1β (IL-1β), chemokine (C-C-C motif) ligand 9 (CXCL9), and cell surface markers: CD4, CD8A, and T-cell receptor delta (TRD). A positive control probe targeted the Bos taurus–specific cyclophilin B (PPIB) housekeeping gene, while a probe targeting dapB of Bacillus subtilis was used as a negative control. The RNAscope labeling technique has been shown to be capable of single mRNA molecule detection. 57
FFPE tissue pretreatment was performed with manual antigen retrieval according to the manufacturer’s instructions. Slides were baked in a dry oven for 1 hour at 60°C to promote tissue-to-slide adherence, deparaffinized and rehydrated in fresh xylenes and 100% ethanol, and air dried. RNAscope hydrogen peroxide (Advanced Cell Diagnostics) was next applied to each tissue section for 10 minutes at room temperature (RT) to block endogenous peroxidase activity, followed by rinsing with fresh distilled water (dH2O). Disruption of formalin cross-linking and unmasking of antigenic epitopes was achieved by submerging slides in a boiling 1× RNAscope target retrieval solution (Advanced Cell Diagnostics) for 15 minutes, followed by rinsing with fresh dH2O and 100% ethanol. Once slides had completely air dried, a hydrophobic barrier was drawn around each tissue using an ImmEdge pen (Vector Laboratories, Burlingame, CA), and slides were stored at RT overnight with desiccants. The following day, RNAscope Protease Plus was applied to each tissue section and incubated in a humidifying tray at 40°C in a HybEZ Hybridization System oven (Advanced Cell Diagnostics) for 30 minutes. Slides were then rinsed with fresh dH2O before proceeding to probe hybridization.
Probe hybridization, amplification, and detection were performed according to the manufacturer’s instructions. All incubations were carried out in a humidifying tray either at RT or in a HybEZ oven at 40°C. Between each incubation step, slides were washed with fresh 1× Wash Buffer (Advanced Cell Diagnostics). To allow binding of the ZZ probe to target mRNA, a customized probe, prewarmed to 40°C, was applied to each tissue section and incubated at 40°C for 2 hours. Branched amplification and detection of the probe with Fast Red chromogen (Advanced Cell Diagnostics) was achieved by incubating slides with kit reagents (Advanced Cell Diagnostics) as follows: AMP 1 (30 minutes), AMP 2 (15 minutes), AMP 3 (30 minuts), and AMP 4 (15 minutes) at 40°C; AMP 5 (30 minutes) and AMP 6 (15 minutes) at RT; and a 60:1 solution of RED-A: RED-B (Advanced Cell Diagnostics) at RT for 10 minutes.
Following RED detection, slides were rinsed with fresh dH2O before being transferred to a 1:1 Gill’s hematoxylin I/dH2O (American MasterTech, Lodi, CA) counterstain. Slides were submerged in hematoxylin solution for 2 minutes, rinsed with fresh dH2O thrice, submerged in 0.02% ammonia water for bluing, and dry baked at 40°C for 20 minutes or until completely dry. Tissue dehydration was not completed due to the alcohol-sensitive nature of the Fast Red chromogen. To mount the tissue samples, slides were dipped in fresh xylenes, 1 to 2 drops of aqueous EcoMount mounting medium (Biocare Medical, Pacheco, CA) were applied to each tissue section, and a #1 thickness cover slip was applied over top of the tissue section. Slides were dried at RT in the dark overnight before microscopic examination.
Immunohistochemistry
FFPE sections of lung from each animal were processed for IHC as described 1,38 using mouse anti–human CD68 (clone EBM11, Dako Cytomation, Glostrup, Denmark) at a 1:50 dilution in combination with reagents from the KPL HistoMark Biotin Streptavidin-HRP Systems Goat Anti-Mouse IgG (H+L) kit (KPL, Gaithersburg, MD). Incubations took place at RT, and washes were performed with fresh 0.05 M Tris (2 × 3 minutes) followed by fresh 0.02 M Tris with NaCl and Tween 20 (2 × 3 minutes), unless specified otherwise. Tissue sections were baked in a dry oven at 60°C for 20 minutes to promote tissue-to-slide adherence, then deparaffinized and rehydrated using fresh xylenes and a graded ethanol series. Slides were then placed in 0.05 M Tris buffer for 20 minutes, incubated with 0.3% freshly prepared hydrogen peroxide in methanol for 30 minutes to block endogenous peroxidase activity, and washed. Background Buster (Innovex Biosciences, Richmond, CA) was applied as a blocking agent for 20 minutes to reduce background staining. Without washing, anti-CD68 primary antibody was applied to tissue sections overnight at 4°C. The following day, slides were washed, biotinylated antibody (KPL) was applied for 30 minutes, slides were washed, streptavidin-peroxidase (KPL) was applied for 30 minutes, slides were washed, and freshly prepared DAB chromogen (Vector Laboratories) was applied for 5 minutes. Next, slides were rinsed well with fresh dH2O, counterstained, and blued as described in the ISH methods. Slides were dehydrated through a graded ethanol series, placed into fresh xylenes, and mounted with Refrax mounting media (Anatech, Battle Creek, MI) and #1 thickness coverslips. Slides were dried at RT in the dark overnight before microscopic examination. A negative control, absent the primary antibody, was also used.
Results
Fifteen Days Postinfection
Three of 5 calves had gross lesions, which were limited to the lungs, involving 1 to 3 lung lobes per calf and between 1 and 5 lesions per calf. Lesions were characterized by small (up to 1 mm) dark red circular foci on the pleural surface. Gross lesions were not present in other organs. Microscopically, organized granulomas were not present; rather, type 1 lesions were evident, characterized by focally extensive, expanded, congested interstitium containing increased numbers of lymphocytes, CD68+ macrophages, and low numbers of granulocytes. Alveoli were variably filled with homogeneous eosinophilic to finely fibrillar material consistent with edema fluid and fibrin, respectively (Fig. 1a), and variable numbers of CD68+ alveolar macrophages (Fig. 1b) with abundant foamy amphophilic to basophilic cytoplasm. Variable numbers of AFB were seen within cells morphologically consistent with macrophages (Fig. 1a, inset).
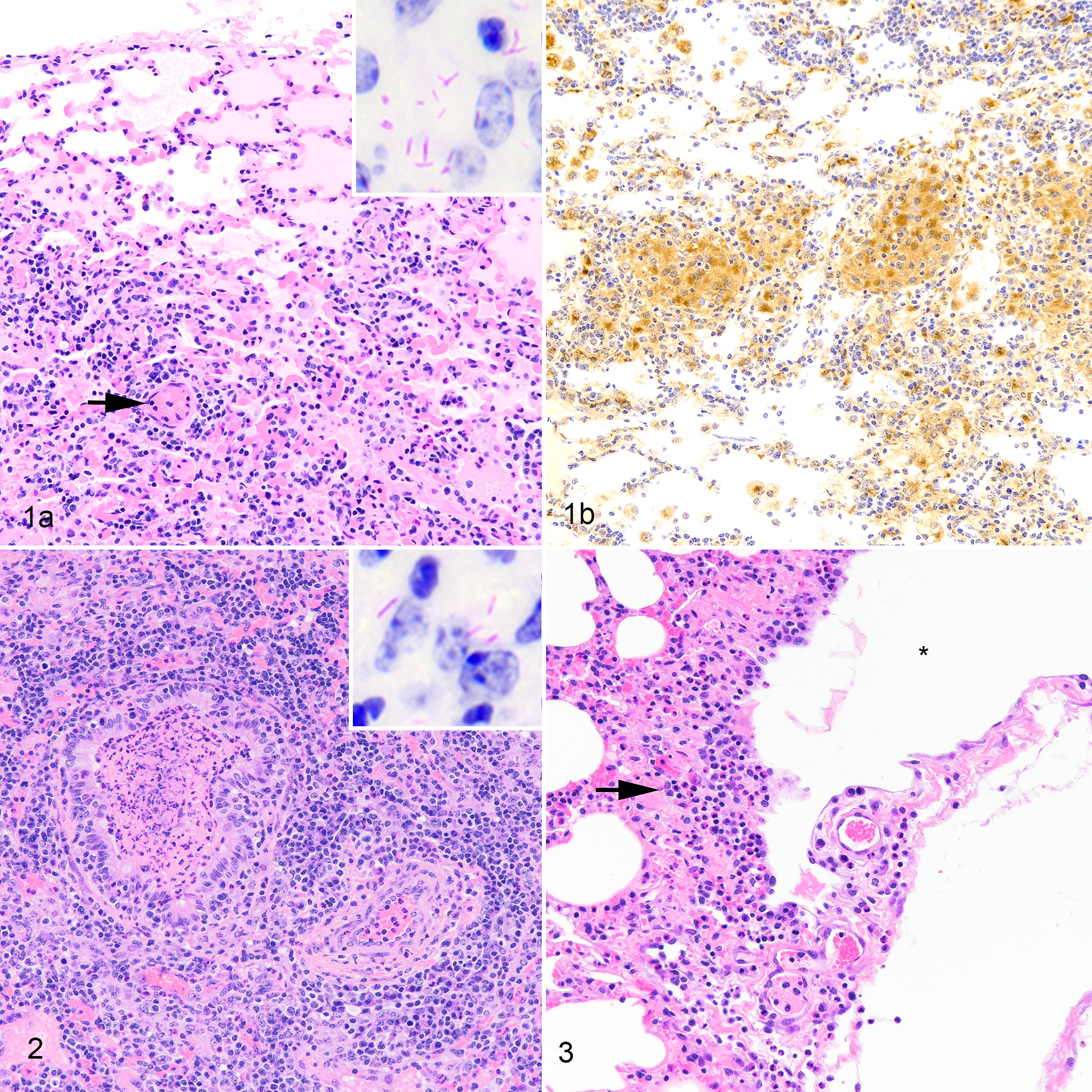
Mycobacterium bovis infection, lung, bovine, 15 days postchallenge.
In 2 of the 3 calves with gross lesions, multiple bronchioles contained intraluminal fibrin, granulocytes, necrotic cell debris, and variable numbers of AFB (Fig. 2, inset). Multifocally, blood vessels and lymphatics were incompletely surrounded by low to moderate numbers of lymphocytes, CD68+ macrophages, and granulocytes (Fig. 2). Within mildly edematous interlobular septa were lymphatic and blood vessels surrounded by low to moderate numbers of neutrophils, lymphocytes, and macrophages, resulting in perilymphangitis and perivasculitis, respectively (Fig. 3).
Cells morphologically consistent with lymphocytes and expressing mRNA for the CD4, CD8, or γδ T cell surface markers were present in low to moderate numbers in type 1 lung lesions (Fig. 4). While CD4- or CD8-expressing cells were infrequently present in nonlesioned, normal-appearing lung, γδ T cells were scattered in moderate numbers throughout the interstitium of less affected, normal-appearing lung.
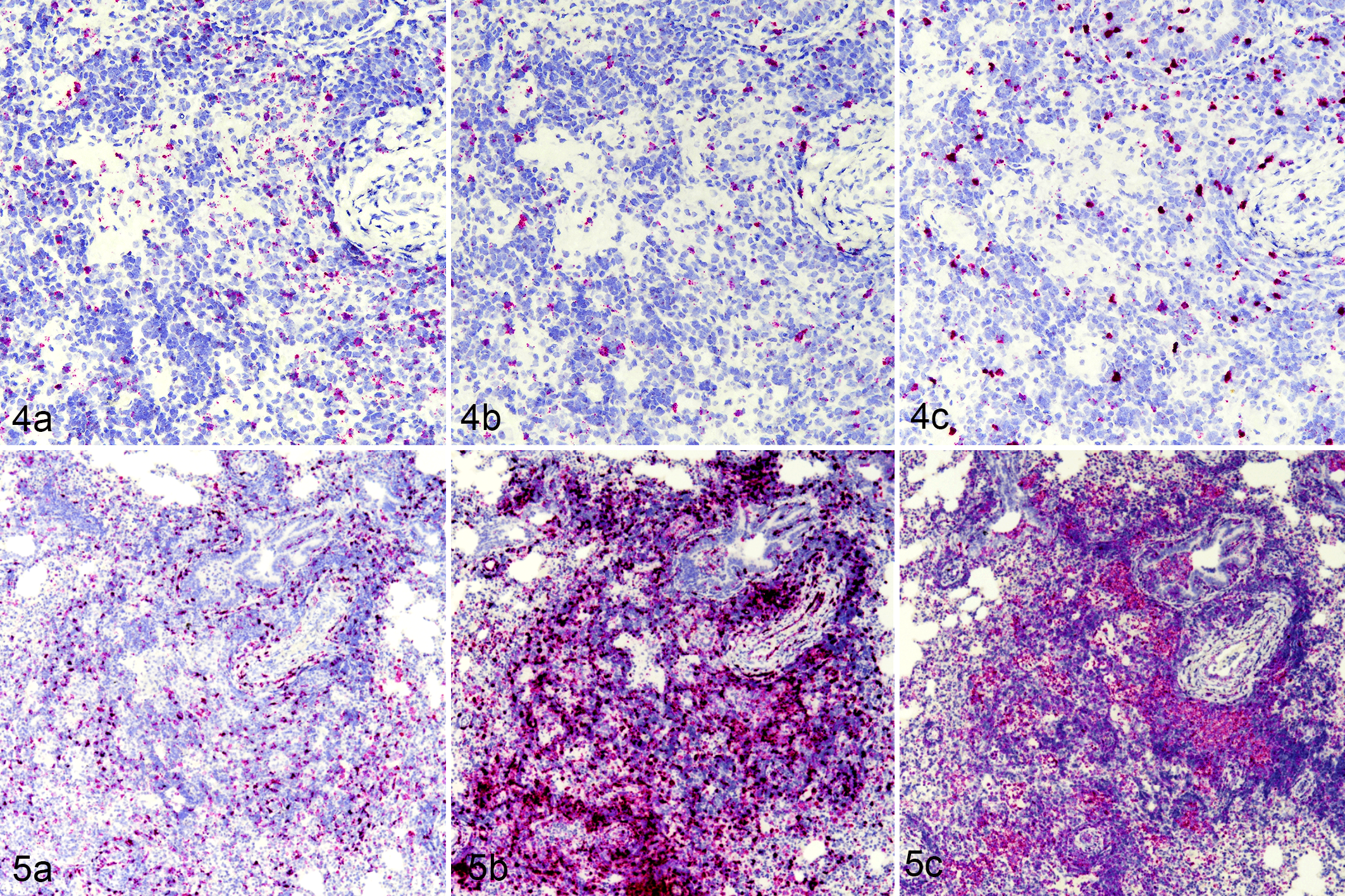
Mycobacterium bovis infection, lung, bovine, 15 days postchallenge. In situ hybridization. The early lesion is characterized by infiltrates of T cells expressing (4a) CD4, (4b) CD8, and (4c) γδ messenger RNA (mRNA) and by cells expressing (5a) MCP-1/CCL2, (5b) CXCL9, and (5c) transforming growth factor–β mRNA.
In situ hybridization also revealed numerous cells within type 1 lesions expressing large amounts of mRNA for the chemokines MCP-1/CCL2 and CXCL9 and for TGF-β (Fig. 5). Expression of these 3 cytokines was also noted around bronchioles and vasculature in both affected areas and less affected, normal-appearing areas of the lung; moreover, numerous vessels demonstrated endothelial cell expression of CXCL9 (Fig. 5b). Moderate to large numbers of cells expressing IL-1β and TNF-α were seen in both type 1 lesions (Fig. 6a,b) and in less affected, normal-appearing areas of lung. There were low numbers of cells expressing mRNA for IFN-γ within affected areas (Fig. 6c) and even fewer present in normal-appearing areas of the lung. IL-10 mRNA-expressing cells were seen in low numbers in affected areas (Fig. 6d) but in more moderate numbers throughout the interstitium of less affected, normal-appearing areas of lung. No gross or microscopic lesions were present in tissues examined from noninfected calves.
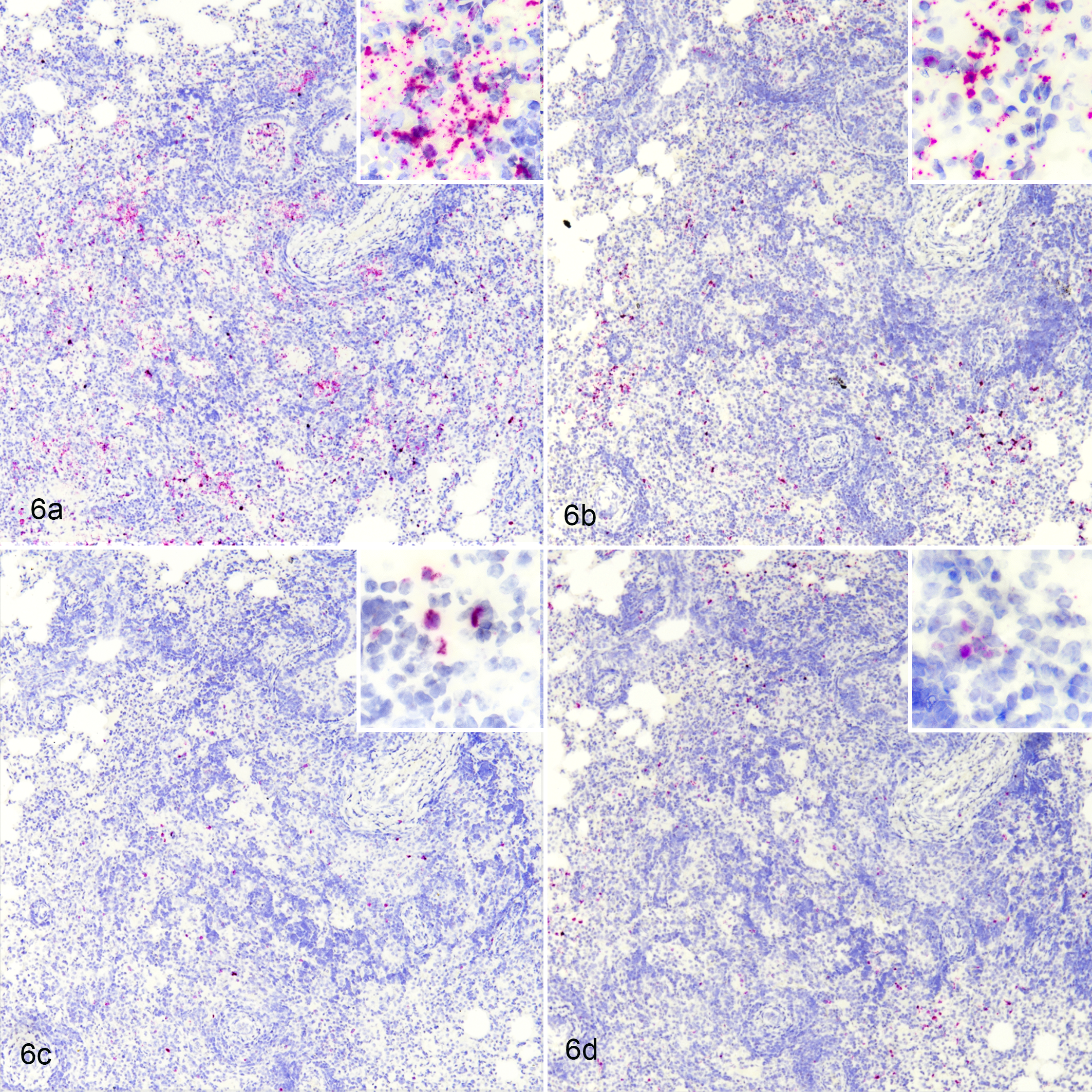
Mycobacterium bovis infection, lung, bovine, 15 days postchallenge. In situ hybridization. The early lesion is characterized by cells expressing (a) interleukin (IL)–1β, (b) tumor necrosis factor–α, (c) interferon-γ, and (d) IL-10 messenger RNA. In situ hybridization. Insets: higher magnification.
Thirty Days Postinfection
All 5 calves had gross lesions in all 5 lung lobes. Lesions were 1 to 15 mm, pale to dark red, round to irregular on the pleural surface. In some cases, dark red regions surrounded smaller pale foci. All 5 calves had at least 5 lesions in both right and left caudal lung lobes. Four of 5 calves also had mild enlargement of tracheobronchial and mediastinal lymph nodes. On cut surface, lymph nodes contained irregular coalescing cortical regions of pallor. Lesions were not present in other organs.
In the lung, microscopically, type 1 lesions similar to those at 15 days postinfection were present. Also present were type 2 lesions characterized by diffuse to near-diffuse alveolar filling with eosinophilic edema fluid, fibrin, and infiltrates of moderate numbers of CD68+ alveolar macrophages, lymphocytes, CD68+ multinucleated giant cells, and low numbers of neutrophils variably filling alveolar spaces and surrounded by moderate numbers of lymphocytes forming small, roughly circular lesions. Some lesions contained small regions of necrosis (Fig. 7) composed of pyknotic and karyorrhectic nuclear debris and a mild increase in neutrophils (type 3 lesions). This mixture of small nonnecrotizing and necrotizing lesions was evident in all 5 calves examined 30 days after infection. Intracellular AFB were present in both nonnecrotizing lesions and necrotizing lesions but were also present extracellularly within regions of necrosis (Fig. 7, inset).
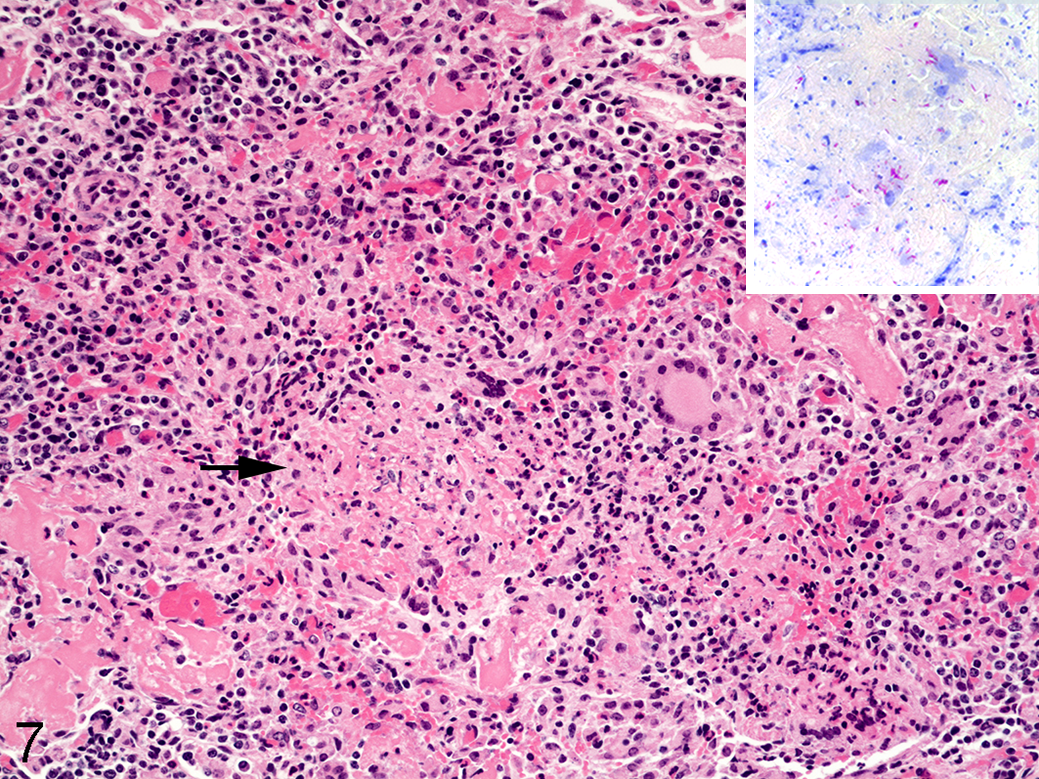
Mycobacterium bovis infection, lung, bovine, 30 days postchallenge. Ziehl-Neelsen. There are infiltrates of macrophages, lymphocytes, and multinucleated giant cells with an area of necrosis (arrow). Acid-fast bacilli are present within the necrotic caseum (inset).
All 5 calves also had multiple bronchioles with regions of disrupted or absent epithelium with intraluminal and peribronchiolar granulomatous infiltrates composed of epithelioid macrophages, lymphocytes, and multinucleated giant cells. No lesions were present in samples collected from the 3 noninfected calves.
Using ISH, the majority of lymphocytes expanding the lung interstitium and in the surrounding lesions expressed mRNA for the CD4 cell surface marker, while fewer expressed mRNA for the CD8 or γδ T-cell surface markers. Cells within the lymphocyte-rich zones of lesions expressed IFN-γ and MCP-1/CCL2, while expression of IL-1β was pronounced in the macrophage-rich zones. As with lesions at 15 days postinfection, there was pronounced expression of CXCL9 and MCP-1/CCL2. There was also pronounced expression of TGF-β, IL-1β, and TNF-α; the level of IL-10 expression was similar to that seen in 15-day lesions.
Microscopically, lymph node lesions were characterized by cortical granulomas composed of epithelioid macrophages, multinucleated giant cells, and lymphocytes, arranged in variably sized, roughly circular patterns, a minority of which had small central foci of caseous necrosis (caseating granulomas). Features often associated with tuberculoid granulomas such as dystrophic mineralization of the central caseum or peripheral collagen deposition were not present.
Discussion
In both human and bovine tuberculosis, inhaled bacilli enter terminal bronchioles and alveolar spaces where they are engulfed by resident alveolar macrophages. Alveolar macrophages, through innate microbicidal mechanisms, attempt to control infection and limit bacterial replication through phagolysosome fusion, phagolysosome acidification, lysosomal proteolytic enzymes, and production of antimicrobial reactive oxygen and nitrogen species. If innate defenses fail, surviving bacilli establish intracellular residence within phagolysosomes. There, virulent mycobacteria use proteins such as early secreted antigenic target (ESAT)–6 to lyse phagolysosomal membranes 49 and gain entry to the cytosol. In the cytosol, mycobacteria multiply, eventually causing macrophage cell death that induces the release of chemokines and cytokines, thus recruiting other cells to the site of infection. As part of the adaptive immune response, dendritic cells containing bacilli exit the primary site of infection and migrate to local lymph nodes where, through antigen presentation and cytokine production, naive T cells recognize their cognate antigens, proliferate, and gain effector functions. Primed, antigen-specific T cells migrate to the site of infection and there activate monocytes and macrophages; the recruitment of these cells contributes to the developing granuloma.
Consistent with the above pathogenesis, 15-day and 30-day lesions in the present study were characterized by clusters of CD68+ macrophages admixed with CD4, CD8, and γ/δ T lymphocytes within the interstitium and alveoli. Acid-fast bacilli were seen in cells morphologically consistent with macrophages. Expansion of the alveolar interstitium occurs as these migrating cells extravasate from blood and lymphatic vessels, developing small septal and alveolar lesions. Accompanying congestion and increased microvascular permeability result in leakage of protein-rich fluid and fibrin within alveolar spaces as seen in the present study. These vasculature changes are not specific for tuberculosis but rather the likely result of early release of mediators such as histamine, bradykinin, leukotrienes, substance P, and cytokines such as TNF-α and IFN-γ. 35
A previous study examined lung lesions as early as 7 and 14 days after intranasal inoculation of 2 × 107 CFU of virulent M. bovis. They observed gross lesions in the lung and tracheobronchial lymph nodes as early as 14 days and microscopic lesions as early as 7 days after infection. 8 Lung lesions seen at 7 days were characterized by infiltrates of macrophages and multinucleated giant cells, degenerate neutrophils, and intralesional AFB. By 14 days after infection, there were multifocal aggregates of neutrophils surrounded by a layer of epithelioid macrophages and numerous intralesional AFB. 8 In the present study, features such as multinucleated giant cells and neutrophilic infiltrates were not found in lesions 15 days after infection but were more characteristic of lesions 30 days after infection. These differences could be due to different routes of inoculation (aerosol vs intranasal), inoculum dosage (104 vs 107), and the finding of intercurrent pulmonary disease characterized by cranioventral plum-colored consolidation and lymphocytic perivascular cuffing in the 1998 study.
In cattle, the kinetics of antigen-presenting cell migration from the site of infection to the draining lymph nodes and priming of antigen-specific T cells that then migrate back to the lung as effector cells remains unclear. In the present study, a primary difference between 15-day and 30-day lesions was the presence of necrosis and extracellular AFB in 30-day lesions. This 30-day period is consistent with previous studies in cattle demonstrating evidence of a type IV hypersensitivity response and a robust IFN-γ response to complex (M. bovis PPD) and specific (ESAT-6) antigens 3 to 4 weeks after infection. 12,43,53 Such responses to intracellular pathogens often involve necrosis. 50 As necrosis proceeds, bacilli are released from their intracellular niche and are often in high numbers within areas of necrosis associated with bovine tuberculoid granulomas. 2,38,45 The presence of neutrophils in 30-day lesions coincident with the development of necrosis is consistent with observations in the zebrafish model of tuberculosis using Mycobacterium marinum. 62 The optical transparency of zebrafish larvae has allowed investigators to visualize the recruitment of neutrophils not to initial sites of inflammation but rather to identifiable granulomas through signals triggered by dying macrophages. 62 Neutrophils have been described previously in early stage bovine tuberculoid granulomas with and without necrosis. 2,38,45,59
Strong gene expression of chemokines such as MCP-1/CCL2 (monocyte chemoattractant) and CXCL9 (T-cell chemoattractant) and the cytokine TGF-β was present at both 15 and 30 days after infection. MCP-1/CCL2 is primarily secreted by monocytes, macrophages, and dendritic cells to recruit monocytes, memory T cells, and dendritic cells to sites of inflammation. CXCL9, also known as monokine-induced by gamma interferon (MIG), is a T-cell chemoattractant induced by IFN-γ and is often associated with T helper 1 (Th1) responses. Using ISH, intense expression of CXCL9 mRNA has also been observed in early granulomas of macaques experimentally infected with M. tuberculosis. 33 Endothelial cell expression of CXCL9, as seen in the present study, is consistent with observations in other infectious disease models. 54 These models further suggest that IFN-γ or a combination of IFN-γ and TNF-α may be necessary to elicit CXCL9 expression. 54 It is, therefore, reasonable to expect strong expression of these chemokines at the time points examined in the present study. In contrast, TGF-β may play an anti-inflammatory role, downregulating Th1 immune responses, possibly to limit host tissue damage. 17 TGF-β has been demonstrated previously by IHC and ISH in epithelioid macrophages and multinucleated giant cells of bovine tuberculoid granulomas, although the earliest time point previously examined was 13 weeks after infection and is not representative of the lesions in the current study. 2,3,45,58,59 In addition to an anti-inflammatory role, TGF-β has been shown to be involved in cell growth, differentiation, and apoptosis, and in macaques, it is a chief cytokine in driving fibrogenesis and tissue repair as tuberculoid granulomas mature. 13 Regulation of TGF-β is complex, and the details of an in vivo role in granuloma formation or disease protection in cattle remain unclear.
Proinflammatory cytokines such as TNF-α and IL-1β were moderately expressed at both 15-day and 30-day time points. TNF-α has been described as essential for the formation of organized granulomas and would be expected in early developing granulomas such as those observed in the present study. An important mediator of inflammation, low levels of TNF-α are associated with fatal disease progression, 20 while excessive expression induces a hyperinflammatory state, promoting tissue damage and destruction. 14 There are likely multiple mechanisms by which TNF-α promotes effective granuloma formation, maintenance, and function that remain to be elucidated. 17 The cytokine IL-1β is also a pyrogen produced by activated macrophages as a proprotein, proteolytically cleaved to its active form by caspase 1. IL-1β is involved in cell proliferation, differentiation, and apoptosis.
Expressed at a low level in the current study was IFN-γ, a cytokine deemed critical for granuloma maintenance and control of mycobacterial infection. IFN-γ is a proinflammatory cytokine expressed during M. bovis and M. tuberculosis infection, and it has been described as pivotal in immunity to infection with M. tuberculosis complex pathogens. 29 Murine studies using M. tuberculosis have shown that even before the appearance of antigen-specific T cells, noncognate IFN-γ production by antigen-independent T cells can occur. This may be consistent with the present findings of IFN-γ production at 15 days postinfection. 28 A major role of IFN-γ is to stimulate macrophages to produce other important proinflammatory cytokines such as TNF-α and IL-1β; moreover, mice deficient in IFN-γ succumb to fatal tuberculosis. 10,18,43 As a dominant and critical cytokine, higher levels of IFN-γ expression might have been expected in the developing granulomas of the present study. However, in experimentally infected macaques, low to undetectable IFN-γ mRNA and protein expression was seen in some individual pulmonary granulomas, as well as in stage I bovine tuberculoid granulomas in experimentally infected cattle. 2,22 Indeed, it has been shown that only 8% of T cells within granulomas express any Th1-type cytokines (IFN-γ, TNF-α, or IL-2). 6,22
IL-10 is generally considered an anti-inflammatory mediator and is produced primarily by macrophages and T cells. IL-10 dampens macrophage activation, resulting in decreased production of reactive nitrogen and oxygen species and diminished Th1 responses. 4 The presence of roughly equal expression of IL-10 within developing granulomas, as well as in less-affected and normal-appearing areas of lung in 15-day lesions, is interesting. IL-10 directly inhibits CD4 T-cell responses and downregulates IFN-γ production. Moderate amounts of IL-10 in 15- and 30-day samples of the current study may be partially responsible for the observed low expression of IFN-γ. In a mouse model, elimination of IL-10 activity during the first 21 days after infection with M. tuberculosis corresponded with increased CD4 and CD8 T-cell numbers and their expression of IFN-γ. 11
As in the current study, CD4, CD8, and γδ T cells have all been noted in bovine tuberculoid pulmonary granulomas, yet their presence has not been documented at very early time points. 32 Through their interactions with mycobacteria-infected, antigen-presenting cells, CD4 T cells are known to be central to immunity against mycobacterial infections and are believed to be the predominant source of IFN-γ. 51,55 γδ T cells were prominent in 15-day lesions and represent a unique subset of CD3+ cells that reportedly play a role in bridging the innate and adaptive immune responses to M. bovis. 34 In agreement with the current study, other examinations of γδ T cells in M. bovis infection have noted their increased presence in early bovine tuberculoid granuloma formation. 8,38
The host response induced by infection with M. tuberculosis or M. bovis is complex and must be carefully balanced to provide sufficient activation to provide protection, while at the same time limiting inflammation and host tissue destruction. The present study documents the in vivo early phases of cytokine and chemokine expression, as well as of immune cell recruitment to sites of M. bovis infection in cattle, and is in general agreement with previously described events drawn from studies of M. tuberculosis infection in laboratory animal and nonhuman primate models of human tuberculosis.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819833454 - Early Pulmonary Lesions in Cattle Infected via Aerosolized Mycobacterium bovis
Supplemental Material, DS1_VET_10.1177_0300985819833454 for Early Pulmonary Lesions in Cattle Infected via Aerosolized Mycobacterium bovis by Mitchell V. Palmer, Jayne Wiarda, Carly Kanipe and Tyler C. Thacker in Veterinary Pathology
Footnotes
Acknowledgements
We thank clinical veterinarian, Dr. Rebecca Cox and animal caretakers Lisa Ashburn, Hannah Shroeder, Robin Zeisneiss, and David Lubbers for excellent animal care and Shelly Zimmerman, Allison Lasley, and Hayley Fisher for excellent technical assistance. Mention of tradenames or commercial products is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research funds were provided by the US Department of Agriculture, Agricultural Research Service Project (CRIS# 5030-32000-222).
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
