Abstract
Thirty-two Large White X Landrace pigs, 4 months old, were inoculated with the classical swine fever (CSF) or hog cholera virus strain “Alfort” in order to identify the mechanism responsible for the lymphopenia and thrombocytopenia observed in the spleen during the experimental induction of disease, by immunohistochemical and ultrastructural techniques. Results showed a progressive depletion of splenic lymphoid structures and evidence of platelet aggregation processes. Lymphoid depletion was due to lymphocyte apoptosis, which could not be ascribed to the direct action of the virus on these cells; direct virus action could play only a secondary role in the death of these cells. Absence of severe tissue and endothelial damage, together with moderate procoagulant cytokine levels in the serum, suggest that these pathologies can be ruled out as the cause of platelet aggregation and thrombocytopenia in CSF. Monocyte/macrophages were the main target cells for the CSF virus, and they exhibited phagocytic and secretory activation leading to the synthesis and release of tumor necrosis factor α, which proved to be the chief mediator, followed by IL-6, IL-1α, and C1q complement component. In view of their characteristics, TNF-α and, to a lesser extent, IL-1α and IL-6 appear to be the major cytokines involved in the pathogenesis of lymphocytopenia and thrombocytopenia; a clear spatial and temporal relationship was observed between these two phenomena.
Cytokines are a heterogeneous group of regulatory proteins that often share biologic activities; attention has been drawn to their role in inflammatory and hemorrhagic processes, as well as to the role they play in the immune response. 20, 51 They are produced by various cell types, chief among which are monocyte/macrophage (m-M⊘s) cells. The major cytokines secreted by these cells include tumor necrosis factor α (TNF-α), which causes tissue necrosis and prompts death by apoptosis, 1, 55 and IL-1, which displays chemotactic activity 54 and is also involved in the induction of apoptosis. 38, 40 These two cytokines trigger production of the major pyrogenic cytokine IL-6, 43 which is involved both in the hepatocyte production of acute-phase proteins and in the regulation and differentiation of several cell types. 2 These cytokines appear to be associated with increased vascular permeability, and reports suggest that they are partly responsible for the hemorrhaging characteristic of human viral diseases. 8 Attention is also drawn to the procoagulant activity of both TNF-α and IL-1 and, to a lesser extent, IL-6, 16, 46 as well as to the involvement of TNF-α, together with tissue damage, in the activation of the extrinsic coagulation pathway. 52
Numerous studies have shown that the m-M⊘ is the prime target cell of classical swine fever (CSF) or hog cholera virus, 21, 41, 42, 47 although lymphocyte infection is also reported in later stages of the disease. 21, 47, 49 The pathogenic mechanisms involved in the thrombocytopenia and lymphoid depletion observed in CSF remain unclear. A number of studies have sought to ascribe these processes to the direct action of the virus either on lymphocytes 6, 31, 32 or platelets; 56 other authors, however, attribute cell destruction to a series of indirect mechanisms. 19, 47 Recent research suggests that changes in the function of m-M⊘s, prompted by infection, alter the secretory properties of these cells; this alteration, coupled with their mobility, may be a decisive factor in the pathogenesis of the disease. 21, 41, 42
CSF thus affords a useful animal model for biomedical research into the pathogenesis of human virus infections prompting immunosuppression and hemorrhage. Moreover, given its anatomic and physiologic characteristics, the pig is seen as a likely source of organs for xenotransplantation; therefore, more detailed knowledge is required regarding its immune system and the potential consequences of the transmission of pathogens to humans treated with living pig tissues. 14
The spleen is an ideal organ for studying the pathogenesis of CSF, not only because of its particular histologic characteristics and the range of immunocompetent cell populations (m-M⊘s, T and B lymphocytes, and dendritic cells), but also in view of the significant vascular alterations displayed during infection. The goal of the present study was to ascertain the mechanisms responsible for lymphoid depletion and thrombocytopenia; spleen samples were taken to evaluate, in situ, the distribution of virus antigen and to examine both the quantitative alterations taking place and their possible relationship with the cytokine expression in m-M⊘s.
Materials and Methods
Animals, virus, and experimental design
Thirty-six Large White × Landrace pigs of either sex, 4 months old and serologically negative for CSF, African swine fever, porcine reproductive respiratory syndrome, and Aujeszky disease, were used; the live weight at the start of the study was approximately 30 kg. All animals were housed in the Centro de Investigación en Sanidad Animal in Valdeolmos, Madrid, Spain. Four pigs were used as uninfected controls, while the other 32 animals each received an intramuscular inoculation with 50% tissue cultures of 105 infective doses of the CSF virus isolate “Alfort 187,” classified as virulent. 57 Control pigs received only phosphate-buffered saline (PBS), pH 7.2. After virus inoculation, clinical signs and rectal temperatures were monitored daily. This experiment was carried out in accordance with the Code of Practice for Housing and Care of Animals used in Scientific Procedures, approved by the European Economic Community Union in 1986 (86/609/EEC).
Preinoculation blood samples were taken from all pigs to obtain baseline values. Blood samples were taken from the anterior vena cava of inoculated animals for leukocyte and platelet counts with a hemocytometer. Blood samples were taken under sterile conditions with plastic syringes and disposable needles.
Processing of specimens for light and transmission electron microscopy
Pigs that were inoculated were sedated with azaperone (Stresnil, Janssen Animal Health) and anesthetized with thiopental-sodium (Thiovet, Vet Limited); finally, they were killed by overdosing with thiopental-sodium in batches of four at 2, 3, 4, 7, 9, 11, 14, and 15 postinoculation days (pid); The remaining four pigs (controls) were killed at the end of the experiment. Samples of spleen were fixed in 10% buffered formalin solution (pH 7.2) and Bouin solution for structural and immunohistochemical study.
Samples were dehydrated through a graded series of alcohols, washed with xylol, and embedded in paraffin wax by routine techniques in preparation for light microscopy. Wax-embedded sections (4 μm) were cut and stained with hematoxylin and eosin (HE).
For transmission electron microscopy (TEM), samples were postfixed in 2% osmium tetroxide, dehydrated in acetone, and embedded in Epon 812 (Fluka Chemie AG). Epon 812–embedded sections (1 μm) were stained with toluidine blue (1% aqueous solution). Further sections (50 nm) of spleen for ultrastructural examination were counterstained with uranyl acetate and lead citrate and viewed through a Philips CM-10 transmission electron microscope.
Apoptosis detection and immunohistochemistry
For the detection of DNA fragmentation, the paraffin-wax sections of spleen, previously fixed in 10% buffered formalin solution (pH 7.2), were stained by the terminal deoxynu-cleotidyl transferase–mediated dUTP nick end labeling (TUNEL) method, with a kit designated In Situ Cell Death Detection, POD (Boehringer Mannheim), according to the manufacturer's instructions.
The previously described avidin–biotin–peroxidase complex (ABC) method 25 was used. To demonstrate CSF virus glycoprotein E2 (gp55), the monoclonal antibody WH303 in Bouin solution–fixed samples was used. The identification of m-M⊘s and cells expressing different chemical mediators was carried out on samples fixed in Bouin solution and 10% buffered formalin solution with monoclonal and polyclonal antibodies, as previously described. 39 Details of the primary antibodies used in this study, including dilutions and pre-treatments, are summarized in Table 1. Ten percent goat non-immune serum diluted in PBS was used in place of specific primary antibodies as negative controls. Samples from the four uninoculated animals were also used as controls.
Details of the immunolabeling reagents.

∗The avidin–biotin complex (ABC) method (Vector Laboratories, Burlingame, CA, USA) was used with 3′3 diaminobenzidine tetrahydrochloride (Sigma-Aldrich Chemie, Steinheim, Germany) as chromogen.
† Veterinary Laboratories Agency, Addlestone, UK.
‡ TC = trisodium citrate dihydrate (Merck, F.R., Germany), 0.01 mol/liter (pH 3.2), microwave 10 minutes.
§ BioVet-UCO, Córdoba, Spain.
| Dako, Glostrup, Denmark.
# Protease type XIV (Sigma-Aldrich Chemie, Steinheim, Germany) 0.1%, 10 minutes.
¶ Endogen, Woburn, MA, USA.
∗∗ Tween® 20 (Merck, München, Germany) 0.1%, 10 minutes.
†† R&D Systems, Minneapolis, MN, USA.
‡‡ Biosource, Nivelles, Belgium.
§§ TNF-α = tumor necrosis factor alpha.
‖ Institut für Pathologie, Tierärztlichen Hochschule, Hannover, Germany.
Cell counting and statistic analysis
To calculate the number of immunolabeled cells that were present and correlated with the results obtained from the different antibodies used, two paraffin-wax blocks from the spleen of each animal were selected, and serial sections were used for the different immunohistochemical studies. The cells immunolabeled against viral antigen, the TUNEL-positive cells, and the m-M⊘s labeled using specific antibodies and expressing different chemical mediators were counted. Cell counts were made in 25 consecutive areas of 0.20 mm2 (splenic cords) and in 25 structures that included lymphoid follicles, periarterial lymphoid sheaths, and the marginal zone of lymphoid follicles. The areas and structures were chosen randomly in four diagonally positioned squares. The distribution was expressed as cells per 0.20 mm2 or as cells per structure. Cellular identification was based on morphologic features, location, and size of cells. Data on immuno-positive cells were assessed to calculate means and standard deviations by Microsoft Excel 97, and differences between the values in uninoculated and inoculated animals were tested for significance (P ≤ 0.05) by the Student's t-test.
Results
Clinical signs
Control animals remained healthy. The clinical signs observed in infected animals were characteristic of CSF. From 2 pid, inoculated animals showed nonspecific symptoms with persistent pyrexia (40.5–41.5°C), varying degrees of anorexia, and constipation. Ocular discharges associated with conjunctivitis were present from 4 to 5 pid. From 6 pid onward, deterioration was rapid; pigs developed body temperatures of greater than 42°C and showed markedly decreased activity. From 9 pid onward, all inoculated animals presented semiliquid yellowish-gray diarrhea and tenesmus when rectal temperatures were taken.
Leukocyte and platelet counts
Total leukocytes in blood (normal values in uninoculated animals = 13 × 106 cells/ml), especially lymphocytes, showed a significant decrease from 4 pid to the end of the experiment (2 × 106 leukocytes/ml). Lymphopenia was accompanied of neutropenia, although there was a significant increase in the number of band neutrophils from 4 pid. The number of monocytes, eosinophils, and basophils did not show significant changes.
Infected animals showed a decrease in platelets from 450 × 103 cells/μl to 310 × 103 cells/μl at 4 pid, which became more severe during the next few days.
Gross lesions
Gross lesions were characteristic of CSF. From 2 pid, hyperemia and petechiae were observed in sub-mandibular lymph nodes and tonsils. From 4 pid, petechial and ecchymotic hemorrhages in the lung and necrosis in the tonsils appeared. Some animals displayed petechiae in the kidney and gall bladder, and there was evidence of congestion, diarrhea, and melena at the ileum level. Reduction in the size of the thymus was observed from 7 pid onward, together with splenic infarcts characteristic of CSF, which appeared rarely. The first petechiae were observed in the ileum, ileocecal valve, cecum, and colon at 9 pid, while 2 days later, small necrotic lesions were apparent in the distal portion of the ileum and in the ileocecal valve.
Morphologic study of spleen
From 2 pid onward, there was greater evidence of pyknosis and karyorrhexis, both possibly attributable to apoptosis; splenic lymphoid structures displayed increasing cell depletion, particularly in the lymphoid follicles (Fig. 1a, b), which retained their normal size. These findings were more marked by 4 pid, when large cells with abundant cytoplasm and vesicular nuclei were also visible; these cells, morphologically identified as macrophages, were interspersed among lymphoid follicle lymphocytes and contained intracytoplasmic engulfed cell debris (tingible bodies). Lymphoid follicle cell depletion was more intense from 7 pid; a decrease in follicle size and the disappearance of germinal centers continued to the end of the study. The intense lymphoid depletion gave rise from 11 pid to a mesh of reticular cells, containing abundant cell debris and tingible body macrophages; by 15 pid, these had become scarce.
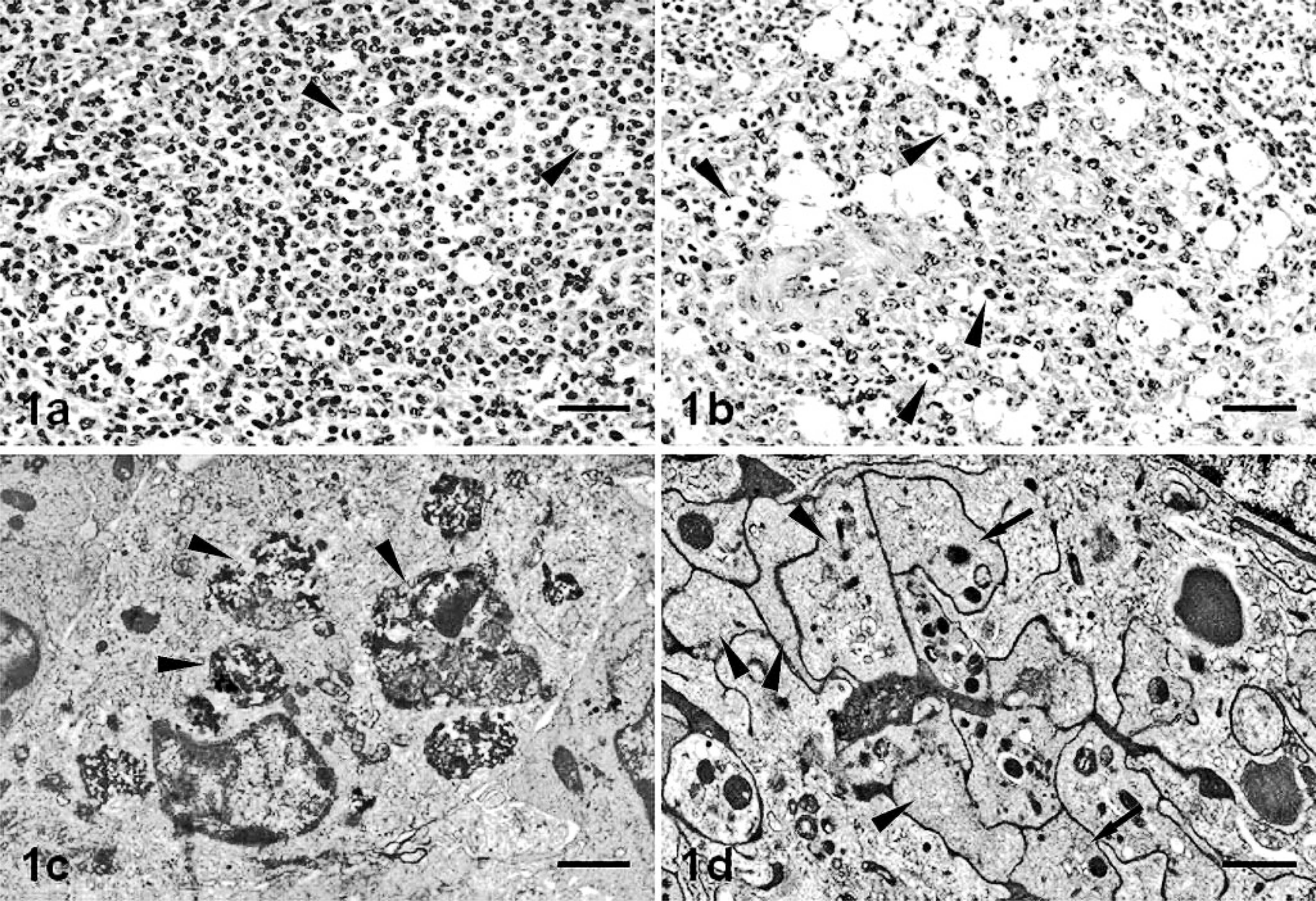
Spleen; pig.
Evidence of pyknosis and karyorrhexis was also apparent from 4 pid, both in the marginal zone and in splenic red pulp cords and sinuses, together with enlarged macrophages containing phagocytized cell debris and occasional neutrophils, plasma cells, and erythrocytes; all these findings subsequently became more common.
These alterations were confirmed at ultrastructural examination (Fig. 1c), which showed phagocyte activation of m-M⊘s, evident in the enlarged cell size, the abundant engulfed cell debris, the loss of filopodia, and the increase in lysosome numbers.
Splenic cords and the marginal zone showed ultrastructural alterations suggestive of platelet activation from 2 pid (Fig. 1d); platelets were enlarged (2–5 μm) and deformed, exhibiting one or more pseudopodia, complete or partial degranulation, and dilation of the open canicular system. Platelet clusters retained their characteristic morphology, and membrane fusion was evident as a visible, highly electron-dense line; clusters, though varying in size, generally comprised more than 10 platelets. On occasion, membranes disappeared completely, giving rise to a finely granular structure of low electron density, containing vestiges of platelet organelles and surrounded by a membrane layer. During the middle phase of the study (7 pid), fibrin deposition was occasionally observed in splenic cord lumina.
Detection of viral antigen in the spleen
Control cells stained negatively for CSF virus glycoprotein E2 (gp55). In inoculated animals, virus antigen was detected from 2 pid onward, mainly in the splenic cord m-M⊘s, in the marginal zone of follicles, and in the periarterial lymphoid sheaths, sometimes appearing within these structures (Fig. 2a).
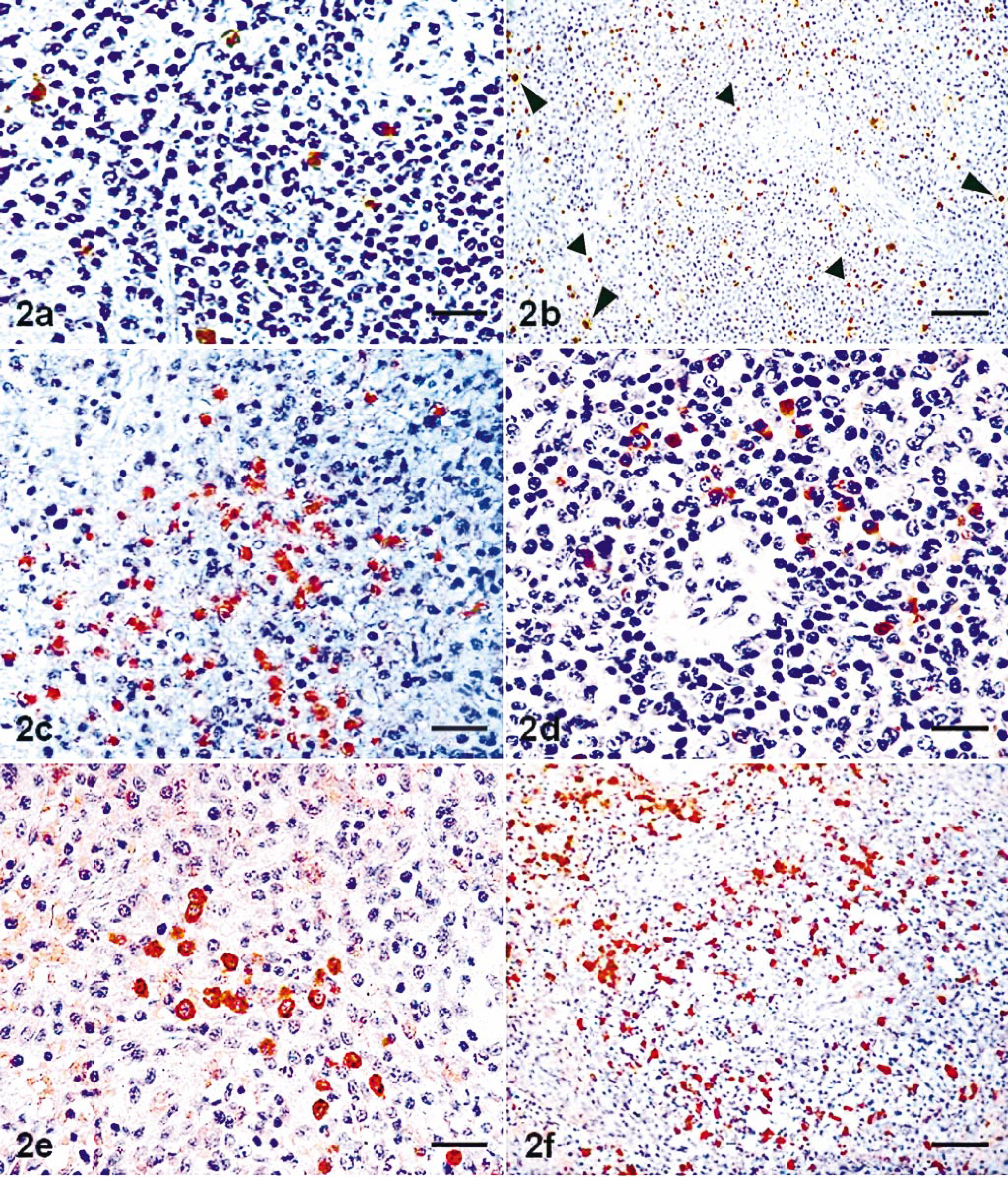
Spleen; pig.
Star-shaped immunopositive cells, tentatively identified as reticular cells, were also observed from 3 pid inside follicle structures; by 4 pid, these were more abundant and were accompanied by occasional positive-staining lymphocytes. From 7 pid (Fig. 3), there was a marked increase in the number of infected cells, mainly m-M⊘s, in all splenic compartments, but particularly within the lymphoid follicles. Virus antigen-positive apoptotic bodies were also visible, both free and engulfed by m-M⊘s, together with polimorphonuclear neutrophils and endothelial cells showing signs of infection. The increase in the number of infected cells in all splenic structures peaked at 11 pid, declining thereafter. The presence of the virus and its replicative characteristics was confirmed by ultrastructural examination.

Count of cells immunolabeled for E2 (gp55) (—▴—) and number (mean ± SD) of m-M⊘s labeled for SWC3 ( ) and MAC387 (□) per area (0.20 mm2) of splenic cords [A] and per individual lymphoid structure (marginal zone of lymphoid follicles [B], follicles [C], and periarterial lymphoid sheaths [D]), in uninoculated (UI) pigs (n = 4) and pigs inoculated with the CSF virus (n = 4 pigs per time point). ∗ = significantly different (P ≤ 0.05) from controls.
) and MAC387 (□) per area (0.20 mm2) of splenic cords [A] and per individual lymphoid structure (marginal zone of lymphoid follicles [B], follicles [C], and periarterial lymphoid sheaths [D]), in uninoculated (UI) pigs (n = 4) and pigs inoculated with the CSF virus (n = 4 pigs per time point). ∗ = significantly different (P ≤ 0.05) from controls.
Distribution of apoptotic cells in the spleen
TUNEL-positive cells were mainly composed of lymphocytes and a smaller number of m-M⊘s (Fig. 2b). Positive reactions were also observed in apoptotic bodies deriving from nuclear and cytoplasmic fragmentation, visible either in isolation or engulfed by macrophages found within and around lymphoid structures as well as within splenic cords.
The number of apoptotic cells (Fig. 4) increased significantly with respect to controls from 2 pid onward in the lymphoid follicles, marginal zone, and periarterial lymphoid sheaths and from 4 pid in the splenic cords. Follicles showed the greatest abundance of positive cells throughout the experiment, peaking at 7 pid and declining thereafter until the end of the study; a similar decrease was observed in the size of lymphoid structures. No positive apoptotic cells were observed from day 11 onward in the periarterial lymphoid sheaths, whereas the number of apoptotic cells in the marginal zone and splenic cords diminished gradually until the end of the experiment.

Count (mean ± SD) of TUNEL-positive cells per area (0.20 mm2) of splenic cords (—▴—) and per individual lymphoid structure (marginal zone of lymphoid follicles [▪], follicles [□], and periarterial lymphoid sheaths [ ]), in uninoculated (UI) pigs (n = 4) and pigs inoculated with the CSF virus (n = 4 pigs per time point). ∗ = significantly different (P ≤ 0.05) from controls.
]), in uninoculated (UI) pigs (n = 4) and pigs inoculated with the CSF virus (n = 4 pigs per time point). ∗ = significantly different (P ≤ 0.05) from controls.
From 2 pid, ultrastructural examination showed changes consistent with lymphocyte apoptosis observed histologically and by the TUNEL technique. Apoptotic cells showed condensation and margination of chromatin and fragmentation of lymphocyte nuclei and cytoplasm; highly electron-dense membrane-bound cell fragments (apoptotic bodies) were visible, either free or engulfed by macrophages. Similar alterations were observed in some macrophages and neutrophils.
Quantitative changes in the m-M⊘ population of spleen
Myeloid cells, mainly m-M⊘s and some neutrophils, were immunolabeled against anti-SWC3 and MAC387 antibodies in both the control and infected animals. Neutrophils had a variable distribution in the splenic compartments, especially in the red pulp. Quantitative changes in macrophage populations (Fig. 3) in the various spleen compartments—identified by morphologic characteristics—were evident from 2 pid, mainly in the splenic cords and the marginal zone. Macrophage numbers in the various compartments peaked between 4 and 7 pid, thereafter remaining relatively stable until the end of the experiment, except in the marginal zone, despite considerable depletion and decrease in the size of lymphoid structures. The greatest increase in m-M⊘ numbers was recorded in follicles showing evident signs of lymphoid depletion; in follicles that retained their normal structure, the increases were less marked.
Although marginal zone m-M⊘s mostly reacted to MAC387, the main m-M⊘ marker in the other splenic structures was SWC3; this antibody additionally enabled the immunostaining of sheathed capillaries, which failed to react to MAC387.
Qualitative changes and expression of chemical mediators by m-M⊘ populations in the spleen
In addition to quantitative changes, m-M⊘s exhibited—from 2 pid onward—subcellular alterations indicative of secretory activity in areas where lymphocyte apoptosis was taking place. These changes included a decrease in heterochromatin, together with a proliferation and dilation of rough endoplasmic reticulum cisternae and a Golgi complex proliferation.
Analysis of cytokine expression showed that TNF-α was the major chemical mediator expressed by m-M⊘s in all splenic compartments, followed by IL-6 and IL-1α; cytokines were expressed, in addition, by some lymphocytes and neutrophils sited mainly in the splenic cord and marginal zone of lymphoid follicles. Few m-M⊘s stained positive for IL-1β (Fig. 5).

Count (mean ± SD) of immunolabeled m-M⊘s expressing TNF-α ( ), IL-6 (
), IL-6 ( ), IL-1α (□), and IL-1β (▪) per area (0.20 mm2) of splenic cords [A] and per individual lymphoid structure (marginal zone of lymphoid follicles [B], follicles [C], and periarterial lymphoid sheaths [D]), in uninoculated (UI) pigs (n = 4) and pigs inoculated with CSF virus (n = 4 pigs per time point). ∗ = significantly different (P ≤ 0.05) from controls.
), IL-1α (□), and IL-1β (▪) per area (0.20 mm2) of splenic cords [A] and per individual lymphoid structure (marginal zone of lymphoid follicles [B], follicles [C], and periarterial lymphoid sheaths [D]), in uninoculated (UI) pigs (n = 4) and pigs inoculated with CSF virus (n = 4 pigs per time point). ∗ = significantly different (P ≤ 0.05) from controls.
From 2 pid onward, there was a significant increase in the number of m-M⊘s staining positive for TNF-α and IL-6 (Fig. 2c, d) in the lymphoid follicles, marginal zone, periarterial lymphoid sheaths, and splenic cords. Peak values (Fig. 5) were reached at different times in different areas of the spleen: the number of cells expressing these cytokines peaked at 2 pid in the marginal zone and in the periarterial lymphoid sheaths, whereas in the splenic cords and lymphoid follicles, the maximum number of m-M⊘s staining positive to TNF-α and IL-6 was recorded at 11 and 14 pid, respectively. Expression of IL-1α (Fig. 2e), though less marked, increased from the start of the experiment, reaching significant values in the splenic cords and marginal zone between 2 and 7 pid, and peaking at 11 pid in follicles. Few cells stained positive to IL-1β; the change in numbers was barely significant, and no positive cells were found in any spleen compartment after 7 pid.
The number of m-M⊘s staining positive to the complement C1q activation factor (Fig. 2f) increased significantly from 2 pid onward in the follicles, periarterial lymphoid sheaths, and splenic cords, peaking at 4 pid in the lymphoid sheaths and at 7 pid in the follicles and splenic cords; thereafter, their numbers declined to levels that were even below those recorded in the uninoculated controls. In the marginal zone, variations were not significant, although the numbers declined from 4 pid.
Discussion
The splenic m-M⊘s of pigs inoculated with the CSF virus strain Alfort showed an increase from the onset of the disease; many showed signs of virus infection as well as phagocytic and secretory activation that coincided, in both time and space, with the lymphocyte apoptosis and platelet aggregation observed in the spleen during CSF.
Various authors have reported a similar increase in m-M⊘s during the course of CSF, mainly in the splenic cords and marginal zone of lymphoid structures; 4, 21, 31, 32 however, these studies failed to chart changes in m-M⊘ numbers within the lymphoid structures. The values obtained in this study were higher than those recorded in earlier studies, 21 perhaps because of the greater species specificity of the swine anti-SWC3 antibody used, which additionally enabled the immunostaining of sheathed capillary macrophages, not, to our knowledge, hitherto reported in the literature. The different functions played by the splenic macrophages, according to their location and activation level, could determine the expression of different amounts of molecular structures, such us SWC3 surface antigen, recognized by anti-SWC3 antibody, or cytoplasmic L-1 antigen (calprotectin), the target molecule for the MAC387 antibody, explaining the differences we have found in the number of macrophages labeled with the two different macrophage markers in the different splenic compartments. The large number of infected m-M⊘s located in the splenic cords and marginal zone from the beginning of the disease suggests that these are the main antigen-presenting cells in the pig, just as they are in humans. 7, 26
The synthesis and release of proinflammatory cytokines by m-M⊘s observed in this study represent a major change in the biologic behavior of these cells during the course of CSF, highlighting the involvement of different cytokines in the pathogenesis of the disease, in which TNF-α may be one of the most important chemical mediators. Secretion in the spleen should be induced by the continued presence of viral antigen, because the maximum expression of TNF-α coincided with the largest number of cells showing signs of viral infection. A similar parallel has been reported during the course of porcine viral infections. 17, 40
The greater abundance of IL-1α–positive cells when compared with IL-1β–positive cells may be because IL-1β is a cell secretion, whereas IL-1α remains bound to the cell membrane, 12 which facilitates its detection on tissue samples. Tissue levels of IL-1α and IL-1β (agonist cytokines with similar biologic functions) around and inside lymphoid follicles, together with the observation of m-M⊘s expressing TNF-α in these areas, point to the involvement of these mediators in the pathogenic mechanisms responsible for lymphocyte apoptosis in the spleen. An autocrine effect of these chemical mediators could be prompting a more intense effect. 53 Because TNF-α induces the release of IL-1 by m-M⊘s, endothelial cells, and other mononuclear cells, 34 IL-1–stimulated cells may initiate additional autocrine secretion of IL-1, TNF-α, and other cytokines, giving rise to the amplification and potentiation of the biologic signal. 30 One of the other cytokines secreted is IL-6, a major regulator of the immune response, 43 which plays an important role during the course of other viral diseases. 24, 40 The expression of IL-6 in the spleen was increased significantly during CSF and recorded an increase that was similar to that observed for IL-1α. The synergy described above may therefore occur in CSF and could be involved in the regulation of pathogenic mechanisms.
Moreover, these cytokines, particularly TNF-α and IL-1, are important regulators of inflammatory processes, acting as chemotactic agents and contributing to the appearance of a massive mononuclear-cell infiltration in several pathologic processes; 35, 54 also, they may well be responsible for the sustained increase in m-M⊘s numbers in the spleen from the onset of CSF.
Spleen lymphoid structures showed a depletion and decrease in size from the onset of the disease due to lymphocyte apoptosis; these alterations worsened over time. Although other studies attribute lymphoid depletion to necrosis, 31, 32 recent research has shown that lymphocyte death in CSF is due to apoptosis. 6, 41, 42, 47 The results obtained in this study suggest that the direct virus action on lymphocytes, described in previous studies, 6, 32 is not responsible per se for the massive lymphoid depletion in the spleen, but rather, that it plays a secondary role; infection of lymphocytes is certainly observed, but it occurs following the death of these cells. The small number of infected lymphocytes does not tally with the considerable lymphoid depletion exhibited by affected animals, but this depletion does coincide with the numeric increase and secretory activation in m-M⊘s.
TNF-α and, to a lesser extent, IL-1 and IL-6 have been shown to induce apoptosis in a number of cell populations. 23, 38 This mechanism prompts lymphoid depletion and immunosuppression in several viral diseases. 33, 37 The activation of TNF receptors by TNF-α may trigger a chain of events leading to apoptosis through the activation of initiatory and executory caspases acting in cytoplasm and nuclei, a mechanism that has been previously reported. 10 In the present study, the appearance of cytokines coincided with the onset and propagation of lymphocyte apoptosis; this spatial and temporal relationship would suggest that TNF, IL-1, and IL-6 are involved and behave like mediators of this mechanism. Lymphocyte apoptosis in the spleen during CSF may also be enhanced by increased expression of the complement component C1q, which regulates antigen presentation 15 and induces the production and secretion of cytokines by m-M⊘s. 45 In this regard, other cytokines not studied in this report may also participate in the pathogenesis of CSF; however, further studies will be required to confirm this hypothesis.
The interaction of ligands expressed by T lymphocytes with B cells is a decisive signal, influencing B-cell proliferation and differentiation and, at the same time, preventing apoptosis of these cells. 50 Considerable functional alterations of T lymphocytes have been demonstrated in CSF. 36, 47, 48 B cells are therefore deprived of the protection they would otherwise enjoy during the course of the disease, which may aggravate the apoptosis observed in B cells in the spleen during CSF.
Finally, the occasional observation in spleen lymphoid follicles of phagocytized apoptotic bodies showing signs of viral infection bears out the hypothesis advanced previously, 21 i.e., that apoptosis may play a major role in the organic spread of the virus. A number of other studies report that viruses may inhibit or induce apoptosis, possibly to facilitate the excretion and dissemination of viral progeny from infected host cells. 13, 22 This mechanism, which also enables the virus to evade the immune response, as occurs in other RNA virus infections, 28, 44 may be responsible for the absence of a specific immune response in the early stages of CSF. 11, 27
The hemorrhages that occur during the course of CSF are prompted by increased vascular permeability and intense thrombocytopenia, rather than by endothelial damage. 20 The platelet aggregation giving rise to thrombocytopenia was not accompanied by fibrin deposition; therefore, both endothelial damage (which would activate the intrinsic coagulation pathway) and tissue damage (activating the extrinsic pathway) can be ruled out as the cause of platelet aggregation in the spleen. The subcellular alterations that are observed in platelets during their activation could be induced by a platelet-activating factor, 3, 29 a powerful mediator of platelet aggregation that is released mainly by macrophages, 9, 29 and this could be another area for future research. These changes, together with the qualitative and quantitative alterations observed in the splenic cord m-M⊘s, support the idea that these cells could play a role in the pathogenetic mechanism responsible for platelet aggregation in the spleen and could be related to the thrombocytopenia characteristic of CSF.
However, it should be noted that, despite the increase in cytokines with proven procoagulant activity, such as TNF-α and IL-1, 16, 46 there was no evidence of the disseminated intravascular coagulation reported in other viral hemorrhagic diseases. 5, 18, 58 This may be partly because of the absence of endothelial and tissue damage and partly, too, because the TNF-α and IL-1 levels detected in this study—though higher than in the controls—could be considerably lower than those reported in other viral diseases in which disseminated intravascular coagulation was observed. 18, 40
Footnotes
Acknowledgements
This work was supported by grants from DGESIC-MEC (PB98-1033) and Junta de Andalucía (Plan Andaluz de Investigación AGR-0137). We appreciate the technical assistance of “Servicio Central de Apoyo a la Investigación (SCAI), Córdoba University.” We thank M. C. Camacho for assistance in the English translation of the manuscript. This manuscript has been prepared in the Uniform Requirements format.
