Abstract
The aim of this study was to report on the lesions occurring in the intestine during experimental classical swine fever (CSF) and to clarify the nature of infected cells and the distribution of viral antigen. Thirty-two pigs were inoculated with the virulent CSF virus (CSFV) isolate Alfort 187 and slaughtered from 2 to 15 postinoculation days; four animals of similar background served as a control group. Immunohistochemistry, electron microscopy, and the transferase-mediated deoxyuridine triphosphate nick-end labeling method were used to detect viral antigens and apoptosis. The results showed progressive lymphoid depletion and mucosal necrosis. The lymphoid depletion could have been caused by apoptosis of lymphocytes but could not be directly attributed to the effect of CSFV on these cells. Vascular changes, pathogenic bacteria, and viral infection of epithelial cells were ruled out as causes of necrotic lesions. However, large virally infected monocytes-macrophages with ultrastructural changes indicative of activation were observed in the intestine. This suggests that monocytes-macrophages play an important role in the pathogenesis of intestinal lesions. An understanding of the function of these cells will require additional study.
Introduction
Bovine viral diarrhea (BVD) and classical swine fever (CSF), both diseases caused by RNA viruses of the pestivirus genus (Flaviviridae family), 35 are characterized by severe clinical and histopathologic changes in the intestine. A number of studies have addressed the pathogenesis of these lesions and the immune response within the digestive mucosa in BVD; 12–14 , 30 however, similar studies have not been carried out for CSF.
CSF takes various clinical forms, depending on the virus strain involved, the environment, and the individual host response. 31 Regardless of the route of entry of the virus into the organism, major pathologic changes take place in the digestive tract; chief among these are watery, greyish-yellow diarrhea, petechial hemorrhages and ecchymosis in the intestine, and catarrhal or necrotic enteritis with focal ulceration in the cecum and the colon. 34 The virus also has a pathogenic effect on gut-associated lymphoid tissue (GALT). 22 , 29 Virus infection takes place initially in monocytes-macrophages and lymphocytes in lymphoid tissue. However, little is known about the sequence of GALT lesions during the course of the disease, the tissue reaction to viral infection, or the mechanism of the considerable lymphoid depletion that characterizes the disease. The following hypotheses have been put forward to account for disease processes in CSF: 1) direct action of the virus on epithelial and endothelial cells, 5 , 15 2) microthrombosis leading to bleeding and ischemic necrosis, 9 , 31 and 3) secondary infection due to immunosuppression. 4 , 27
The aim of this study was to identify lesions in GALT, mainly in interfollicular areas and lymphoid follicles, to ascertain the nature of infected cells and to determine the distribution of viral antigen in the intestine of pigs experimentally infected with CSF virus (CSFV).
Materials and Methods
Animals, virus, and experimental design
Thirty-six 4-month-old Large White × Landrace pigs of both sexes were used. Their live weight at the start of the study was approximately 30 kg. All animals were serologically negative for CSF, African swine fever, porcine reproductive respiratory syndrome, and Aujeszky's disease. All animals were housed in the Centro de Investigación en Sanidad Animal, Valdeolmos, Madrid, Spain. Each of the 32 animals received an intramuscular inoculation of 105 tissue culture infective dose (50%) of the virulent CSFV isolate Alfort 187. 36 Four control pigs received only phosphate-buffered saline (PBS), pH 7.2. After virus inoculation, clinical signs and rectal temperature were monitored daily. This experiment was carried out in accordance with the Code of Practice for Housing and Care of Animals Used in Scientific Procedures, approved by the European Economic Community Union in 1986 (86/609/EEC).
Processing of specimens for light and transmission electron microscopy
Infected pigs were sedated with azaperone (Stresnil®, Janssen Animal Health, Beerse, Belgium) and anaesthetized and killed with thiopental-sodium (Thiovet®, Vet Limited, Leylands, Lancashire, UK) in batches of four at 2, 3, 5, 7, 9, 11, 14, and 15 postinfection days (pid). The four control pigs were killed at the end of the study. Samples were taken from the ileum at 30 and 15 cm (IL30 and IL15) from the ileocecal valve (IV), the IV, and the proximal colon (PC). Samples were fixed in 10% buffered formalin (pH 7.2) and Bouin's for structural and immunohistochemical study. Other samples of these intestinal areas were fixed in 2.5 % glutaraldehyde in 0.1 M phosphate buffer (pH 7.4) for ultrastructural study.
Samples were dehydrated through a graded series of alcohol to xylol and embedded in paraffin wax by routine techniques for light microscopy. Wax-embedded sections (4 μm) were cut and stained with haematoxylin and eosin (HE) or toluidine blue (1% aqueous solution). Histopathologic findings were graded as absent (−), slight (+), moderate (++), and severe (+++).
For transmission electron microscopy, samples were postfixed in 2% osmium tetroxide, dehydrated in acetone, and embedded in Epon 812® (Fluka Chemie AG, Buchs, Switzerland). Epon-812–embedded sections (1 μm) were stained with toluidine blue (1% aqueous solution). Sections (50 nm) of intestinal areas for ultrastructural examination were counterstained with uranyl acetate and lead citrate and viewed using a Philips CM-10 transmission electron microscope.
Apoptosis detection and immunohistochemistry
For detection of DNA fragmentation, formalin-fixed paraffin-embedded sections of intestine were stained by terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling (TUNEL) method (in situ cell death detection, POD® kit, Boehringer Mannheim, Indianapolis, IN, USA), according to the manufacturer's instructions.
To demonstrate CSFV glycoprotein E2 (gp55), the monoclonal antibody WH303 (Veterinary Laboratories Agency, Addlestone, UK) was used on Bouin's fixed samples. 24 , 25 Tissue sections were dewaxed and rehydrated. Endogenous peroxidase activity was abolished by incubation of the sections with 0.3% hydrogen peroxide in methanol for 30 minutes at room temperature. After three 10-minute rinses in PBS, all tissue sections were incubated with 10% normal goat serum for 30 minutes at room temperature. Primary antibody was diluted 1:100 in PBS containing 10% normal goat serum. After incubation with primary antibody overnight at 4 C, the slides were given three 5-minute rinses with PBS, and biotinylated goat anti-mouse immunoglobulin (Dako A/S, Glostrup, Denmark) was applied for 30 minutes at room temperature as the secondary antibody. After two 5-minute rinses with PBS, tissue sections were incubated with avidin–biotin–peroxidase complex (Vector Laboratories, Burlingame, CA, USA) diluted 1:50 in PBS for 1 hour at room temperature. All tissue sections were rinsed with PBS, incubated for 1 minute with the chromogen 3-3′-diaminobenzidine tetrahydrochloride (Sigma-Aldrich Chemie, Steinheim, Germany) diluted to 0.0035% in PBS, rinsed in tap water, and counterstained with Harris' haematoxylin.
PBS and nonimmune serum were used in place of specific primary antibodies as negative controls. Samples from the four uninoculated animals also were used as controls.
Quantitative determination of apoptotic and virally infected cells
TUNEL-positive cells were counted in different anatomic areas of IL30, IL15, IV, and PC. For each area, 10 fields of 0.20 mm2 were evaluated. Results were given as number of cells per square millimeter: −, none; +, 1–2; ++, 2–5; +++, 5–10; ++++, 10–25.
Virally infected cells were identified by immunostaining for gp55 viral antigen. Cell counts were performed on sections of IL30, IL15, IV, and PC from all infected and uninoculated animals. In each section, cells were counted in 50 consecutive areas of 0.20 mm2 that were chosen randomly. Cell density was expressed as the number of cells per square millimeter. Cellular identification was based on morphologic features, location, and size of cells.
Bacteriologic study
IL30, IL15, IV, and PC samples were collected for bacteriologic detection of Salmonella spp using specific culture media (xylose–lysine–deoxycholate and McConkey).
Results
Clinical findings
Control animals remained healthy. The clinical signs observed in infected animals were characteristic of CSF. Infected animals showed nonspecific clinical signs of pyrexia (40.5–41.5 C), anorexia, and constipation from 2 pid. Erythema of the skin and ocular discharge associated with conjunctivitis were present from 4 to 5 pid. From 6 pid onward, deterioration was rapid; pigs developed body temperatures of > 42 C and showed markedly decreased activity. From 8 pid, the typical staggering gait associated with CSF and cyanotic discoloration of the skin of the abdomen, snout, ears, and legs also were observed. From 9 pid onward, all inoculated animals had semiliquid yellowish-grey diarrhea and tenesmus when rectal temperatures were taken.
Bacteriologic detection of Salmonella spp was negative in the intestine of all the animals studied.
Gross lesions
Gross lesions were characteristic of CSF. From 2 pid, hyperemia and petechiae were seen in the submandibular lymph glands and the tonsils. Some animals displayed petechiae in the kidney and bladder from 5 pid. From 7 pid onward, splenic infarcts characteristic of CSF were observed. Reduction in size of the thymus and bleeding in the mesenteric lymph glands were seen from 9 pid. From 5 pid, there was evidence of congestion, diarrhea, and melena at the level of the IL. The first petechiae were observed in the IL, IV, cecum, and colon at 9 pid, whereas 2 days later small necrotic lesions were apparent in the distal portion of the IL and in the IV.
Histopathologic study
Histopathologic findings are summarized in Table 1.
Histopathological findings in different internal areas of pigs inoculated with CSFV.∗

∗ UI = uninoculated animals, (-) = absence, (++) = moderate, (+) = slight, (+++) = severe.
From 3 pid, lymphocyte depletion, pyknosis, and karyorrhexis were observed in the interfollicular areas and the lymphoid follicles of Peyer's patches at IL30 and IL15. In addition, a cellular infiltrate of large mononuclear cells, morphologically resembling macrophages, became more prominent over time. Some of these cells were large, with abundant cytoplasm containing phagocytosed cell debris. Depletion was severe in the IL and IV by 7 pid (Fig. 1), with hypocellular lymphoid follicles and few cells in the interfollicular areas. In the colon these alterations were not observed until 11 pid.
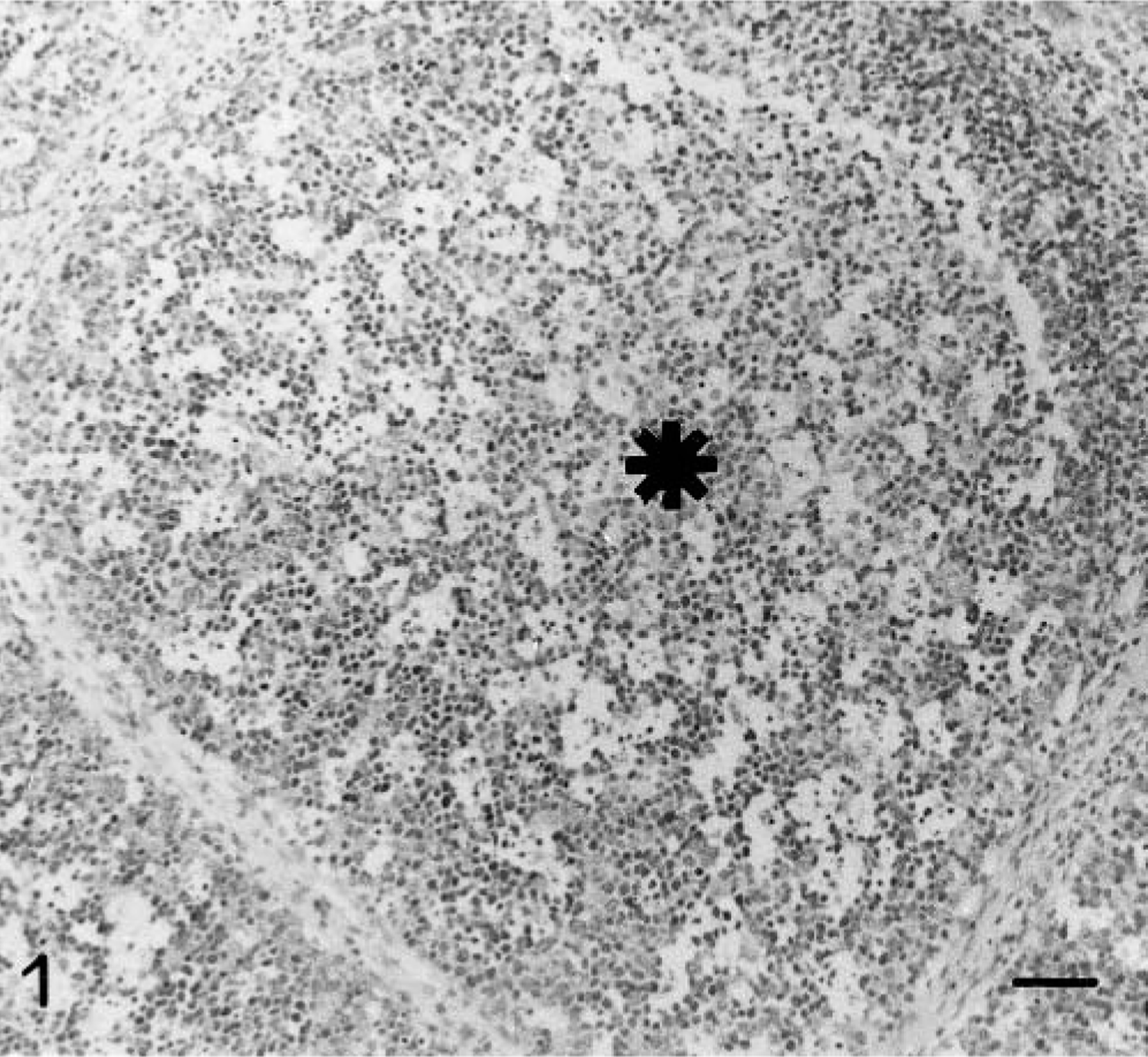
IL 30; 7 pid; lymphoid follicle (∗) of a Peyer's patch in a pig infected with CSFV showing severe lymphocyte depletion. Note the presence of pyknosis and karyorrhexis next to large foamy mononuclear cells. HE. Bar = 60 μm.
Vascular alterations observed in the lamina propria included mild hyperemia and numerous circulating cells in all parts of the intestine from 5 pid onward. A mononuclear infiltrate was visible by 7 pid, mainly in the IV. Edema and microhemorrhage were apparent by 9 pid, although there was no evidence of thrombosis. All these findings were most severe in the IL.
After 3 pid there was an increase in intraepithelial lymphocytes in the crypts of Lieberkühn. At 7 pid there was considerable crypt dilation, with flattening of the epithelium, and cell debris and leukocytes in the lumina; these findings did not vary until 11 pid and were particularly striking in the IL30 and the IL15. Furthermore, pyknosis and karyorhexis of enterocytes, indicative of cell death, were observed occasionally in IL and IV epithelium.
From 11 pid, the IL15 and the IV had small ulcers surrounded by mononuclear infiltrate and covered by a mesh of fibrin and cell debris. By the end of the experiment these ulcers extended into the submucosa.
Distribution of apoptotic cells
The distribution of TUNEL-positive cells is summarized in Table 2. These cells, mainly lymphocytes, increased in number from 2 pid in the interfollicular areas and the lymphoid follicles of Peyer's patches at IL30 (Fig. 2) and IL15 and from 5 pid in the IV. The number of TUNEL-positive cells peaked between 3 and 9 pid in the interfollicular areas (Fig. 3) and between 7 and 11 pid in the lymphoid follicles. The numbers subsequently declined at the end of the experiment. Positive cells were observed sporadically in occasional colon follicles after 9 pid. Lymphoid structures both in the mucosa and in the lamina propria had TUNEL-positive cell debris resulting from cell fragmentation; debris was sometimes in the form of apoptotic bodies undergoing macrophage phagocytosis. From 7 pid, similar numbers of intraepithelial lymphocytes and TUNEL-positive enterocytes were observed in Lieberkühn crypt epithelium in CSF-infected and control animals.
Semiquantitative distribution of TUNEL-positive cells in histological structures of intestinal areas of pigs inoculated with CSFV.

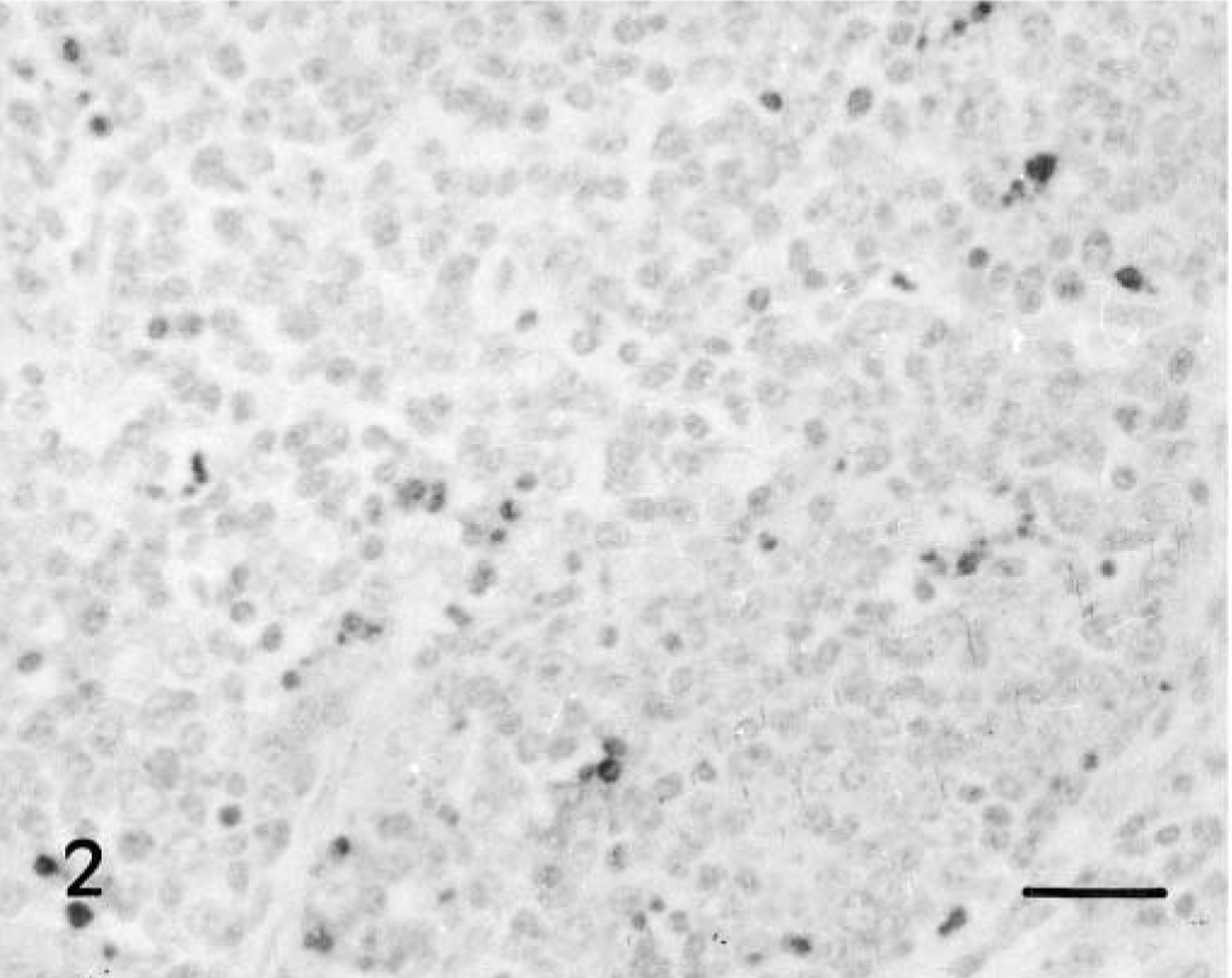
IL 30; 3 pid. Presence of many apoptotic mononuclear cells, mainly lymphocytes, inside the lymphoid structures. TUNEL. Bar = 40 μm.
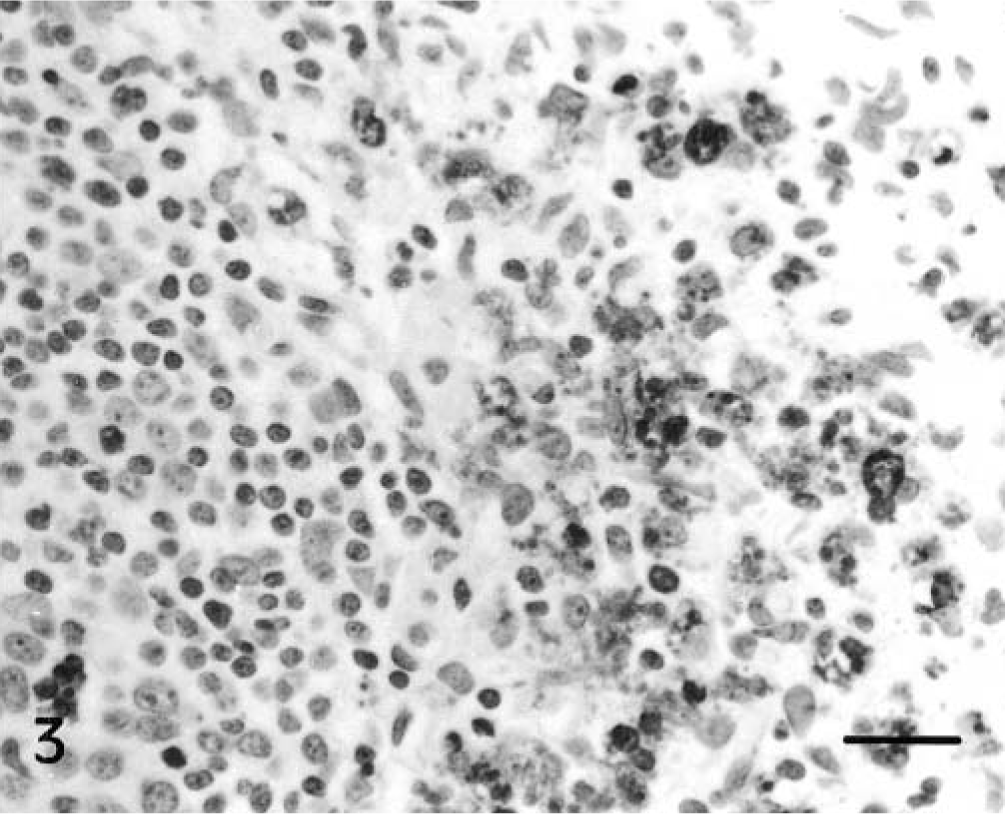
IL 30; 5 pid. Marked apoptotic mononuclear cells and apoptotic bodies phagocytosed by macrophages in interfollicular areas. TUNEL. Bar = 40 μm.
Distribution of viral antigen
No gp55-immunopositive cells were observed in uninoculated controls. In infected animals, gp55-labeled cells included monocytes, macrophages, lymphocytes, epithelial cells, fibroblasts, and endothelial cells. Specific CSFV gp55 reaction product in these cells was evenly distributed in the form of dark brown microgranules or as diffuse stains in the cytoplasm (Fig. 4)
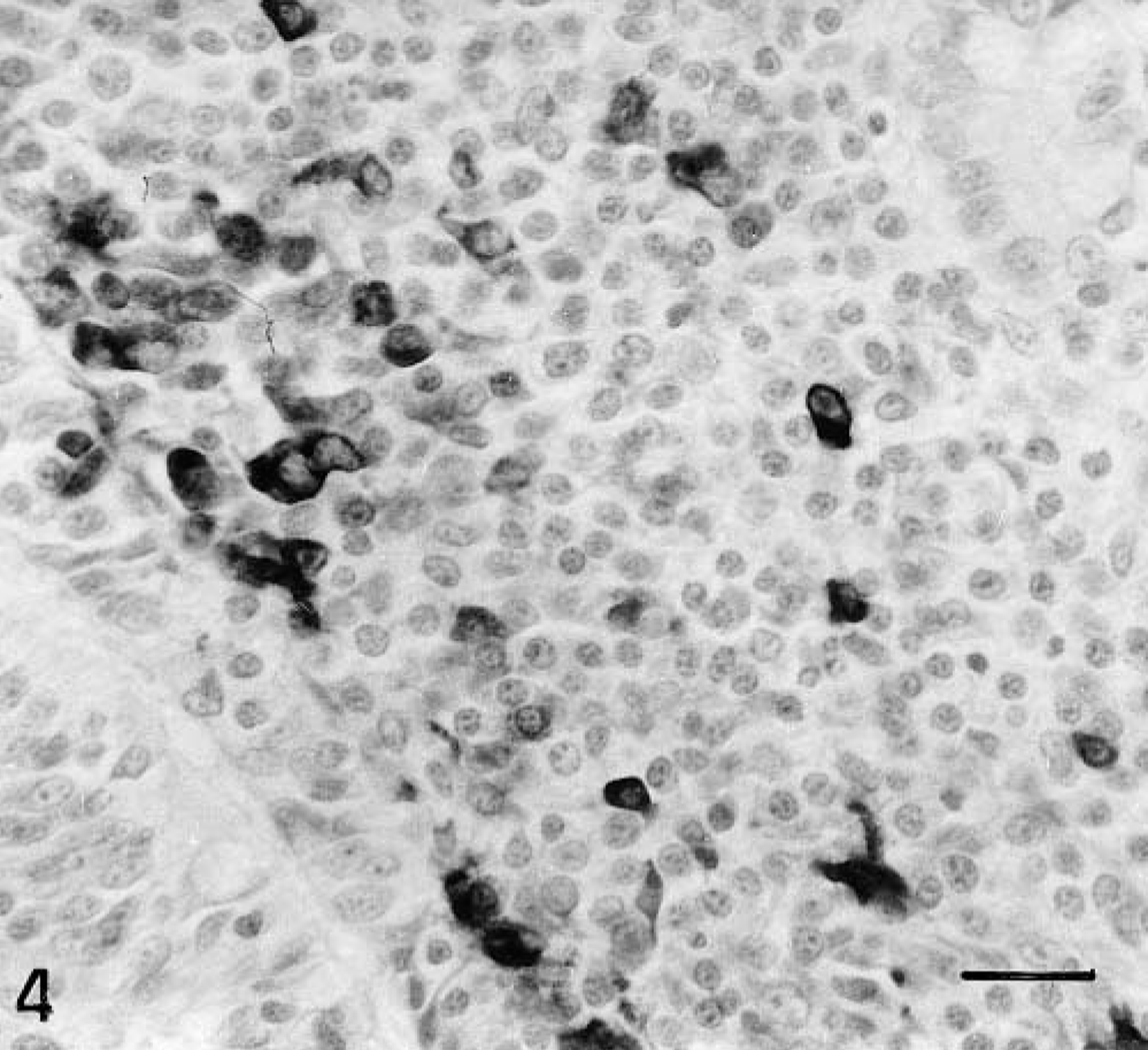
IL 30; 7 pid. Presence of infected mononuclear cells in lamina propria. Note the absence of viral antigen in epithelial cells. ABC method. Bar = 30 μm.
In all areas of the intestine examined, the first cells to be infected were monocytes-macrophages in the interfollicular areas, followed by infection of the follicular lymphocytes. The number of infected follicular lymphocytes increased as the disease progressed, but the most striking increase in infected cells was seen for macrophages. Infection of interstitial capillary endothelial cells and fibroblasts was detected occasionally in the lamina propria from 7 pid onward. Viral antigen was detected in intestinal epithelium, both in crypt epithelial cells and in a small number of intraepithelial lymphocytes.
The distribution (Fig. 5) of virus-infected cells in the various segments of the intestine was focal; areas wholly free of viral antigen were observed to be adjacent to highly infected areas. Viral infection was detected in the IL and the IV from 3 pid but was not apparent in the colon until 7 pid. Infection was later detected in the interfollicular areas, lymphoid follicles, lamina propria, and intestinal epithelium, where it was not related to necrotic areas. Furthermore, viral antigen was detected in the mononuclear infiltrate surrounding necrotic epithelium. In the final stage of the study, the number of cells positive for viral antigen declined in all areas of the intestine, although some viral antigens continued to be evident.
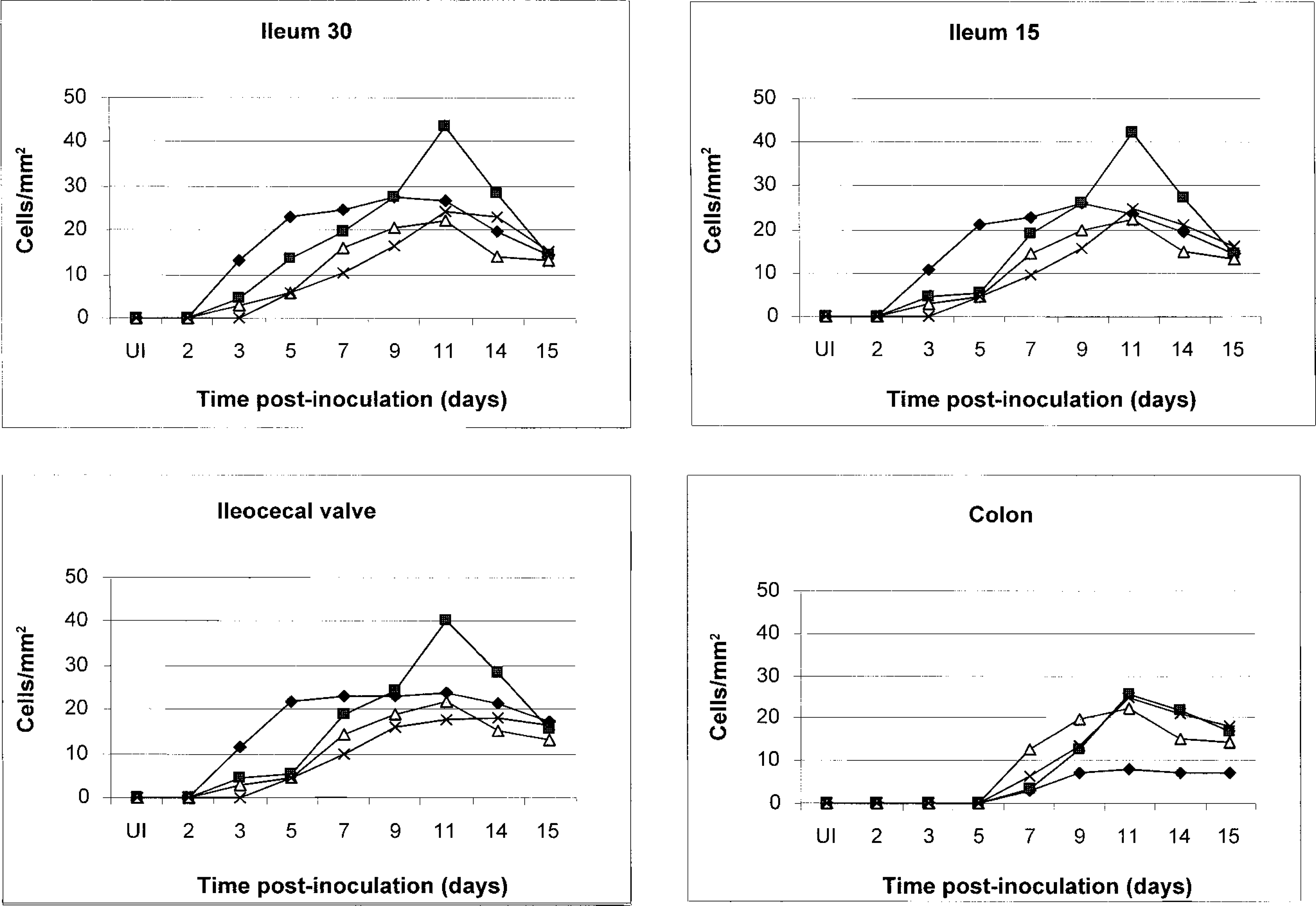
Changes in mean of numbers of gp55-immunopositive cells in interfollicular areas (♦), lymphoid follices (▪), lamina propria (▴), and epithelium (∗) of IL 30, IL 15, IV, and colon in uninoculated (UI) pigs (n = 4) and pigs inoculated with CSFV (n = 4 pigs per time-point).
Ultrastructural study
Lymphocyte apoptosis was observed in lymphoid structures of all intestinal areas, both histologically and by using the TUNEL technique, mainly in the lymphoid follicles and T-cell areas. Lymphoid apoptosis was characterized by condensation and margination of chromatin (Fig. 6) and by fragmentation of lymphocyte nuclei and cytoplasm. Membrane-bound fragments formed apoptotic bodies. A similar process was observed in macrophages and neutrophils. Apoptosis occurred in some epithelial cells in both control and CSF-infected pigs.
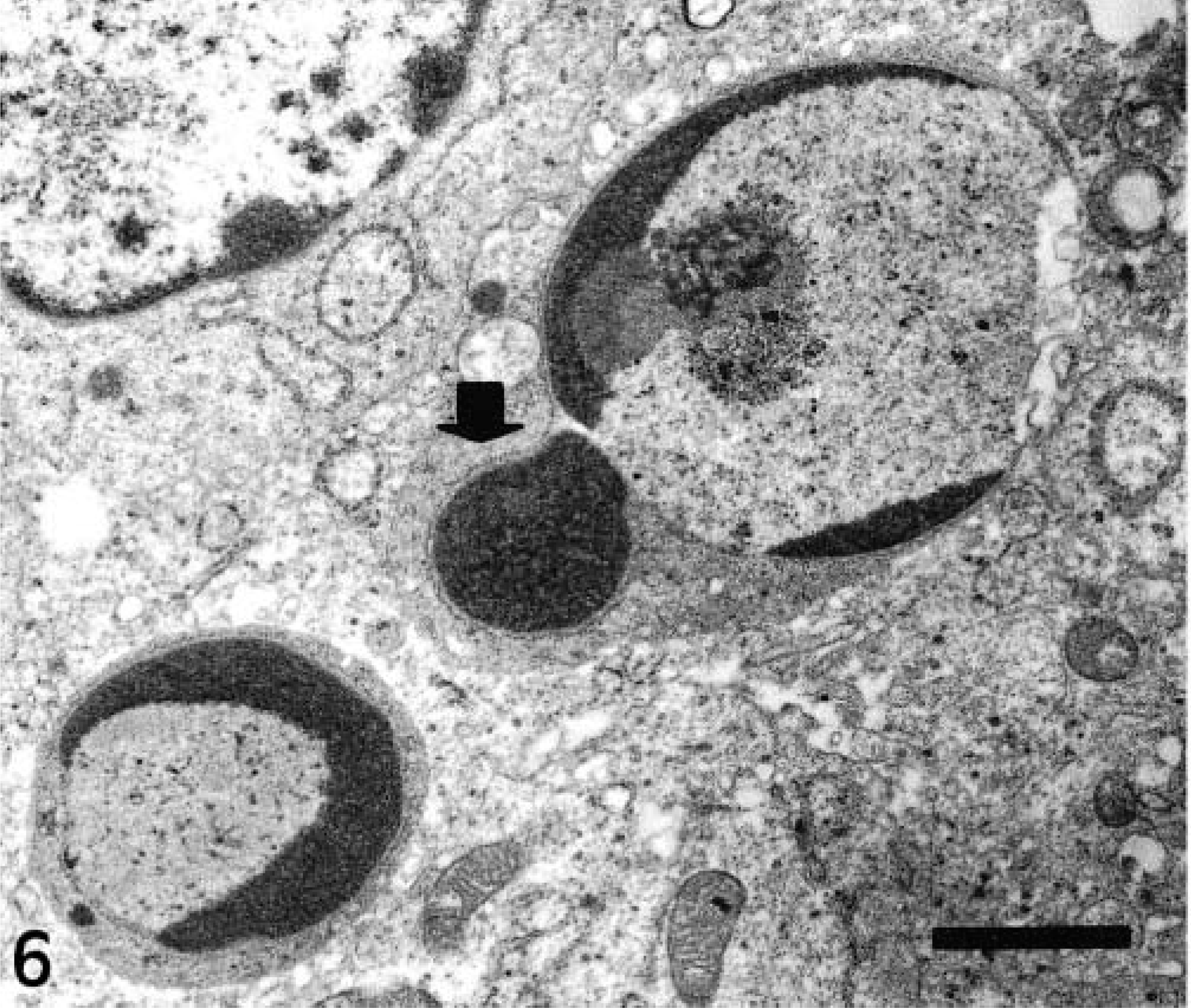
Lymphoid follicle of IL 15; 3 pid. Chromatin condensation and margination in lymphocytes. Note the initial phase of fragmentation of a nucleus (arrow). TEM. Bar = 1.5 μm.
Subcellular structures related to viral infection were membrane-bound inclusions containing granular and moderately electron-dense material with more electron-dense regions. Viruslike particles, 50–55 nm in diameter, resembling CSFV were observed within these inclusions. These structures were not observed in samples from uninoculated control animals. The main host cells for virus were monocytes-macrophages; a few lymphocytes, epithelial cells, fibroblasts, and endothelial cells were infected.
In areas where lymphocyte apoptosis was taking place, macrophages underwent a number of changes consistent with phagocytic and secretory activation from 2 pid. Phagocytic activation was characterized by the presence of phaocytozed cell debris, mostly apoptotic bodies. Some of these macrophages showed signs of viral infection and an increase in cell size and lysosome numbers. Proliferation and dilation of rough endoplasmic reticulum cisternae and Golgi complexes (Fig. 7) were observed in these cells. These subcellular changes suggested secretory activity.
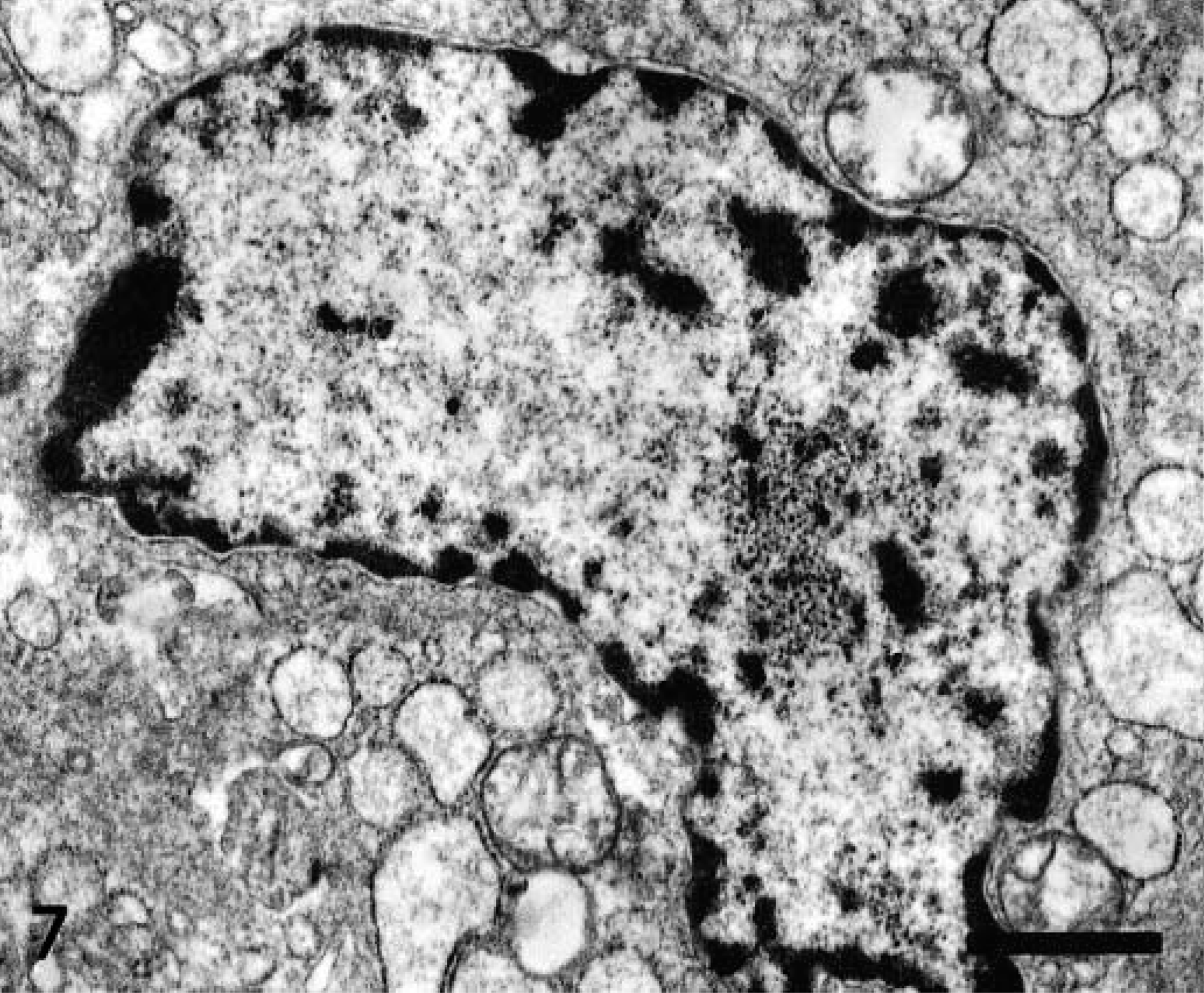
Interfollicular area of IL 15; 3 pid. Subcellular changes in macrophage indicative of secretory activation including dilated rough endoplasmic reticulum cisternae containing moderate electron-dense material. TEM. Bar = 1.5 μm.
Examination of capillaries revealed no significant change in endothelial cells or capillary lumina. There was no evidence of microthrombi or fibrin deposition, although enlarged, degranulated platelets were observed. Only a small number of endothelial cells displayed subcellular changes indicating infection.
Discussion
Lesions observed in the intestines of pigs experimentally inoculated with the virulent Alfort CSFV strain could be directly related to the presence of viral antigen in the structures examined. Changes were observed progressively in the interfollicular areas, lymphoid follicles (marked depletion), lamina propia (vascular changes), and epithelium (necrotic lesions). Lesions appeared after 3 pid in the IL and the IV and after 7 pid in the PC.
In experimental infection with bovine viral diarrhea virus, which belongs to the same genus as CSFV, the number of cells immunopositive for viral antigen has been reported to decline during the course of the disease; 14 a similar decline has been found in the tonsils of pigs inoculated with CSFV (P. J. Sánchez-Cordón, unpublished). Our results showed less decrease in the number of cells that were immunopositive for viral antigen over time. This difference might have been due to the timing of virus spread or differences in the local immune response in our study.
GALT structures were depleted because of massive lymphocyte apoptosis, particularly in B-cell areas. 19 , 26 This apoptosis could not be explained by the direct action of CSFV on lymphocytes 1 , 20 and confirmed the important role of apoptosis caused by indirect mechanisms in the pathogenesis of CSF. 25 , 26 The onset and development of apoptosis coincided with viral infection of macrophages in both interfollicular areas and lymphoid follicles in the various intestinal areas examined. Macrophages, the main CSFV target cells, 8 , 28 also displayed signs of phagocytic activation and secretory changes 2 , 8 , 25 from 3 pid.
The hypothesis that indirect mechanisms that are apparently unrelated to the direct action of the virus may induce apoptosis 25 , 26 , 28 is supported by the results obtained. Our evidence suggests that apoptosis is associated with the quantitative and qualitative changes 8 observed in macrophages. Some authors 8 , 25 , 26 argue that the increase in the macrophage population in the vicinity of lymphoid areas and the release of chemical mediators by these cells (tumor necrosis factor–α and interleukin-1α) 6 , 10 , 25 are jointly responsible for the lymphoid depletion characteristic of CSF. Further research on the behavior of the macrophage population and the expression of various chemical mediators in the intestine is required to identify the pathogenic mechanisms responsible for the depletion of intestinal lymphoid structures in CSF.
Occasionally, small necrotic lesions were observed in the intestinal epithelium at the level of IL15 and the IV from 11 pid. These characteristic necrotic lesions tend to be both more widespread and more severe in subacute or chronic CSF. 27 , 33 A number of hypotheses have been put forward to account for necrotic lesions in the intestinal epithelium in CSF. These include 1) secondary bacterial infections, 2) vascular changes, and 3) viral infection of epithelial cells.
A bacterial disease such as swine salmonellosis 21 was ruled out because bacterial isolation tests proved negative. Also, in swine salmonellosis, necrosis is accompanied by a polymorphonuclear neutrophil infiltrate, 3 , 32 whereas the infiltrate observed in this study consisted of mononuclear cells. 4 , 34 This mononuclear infiltrate has not yet been characterized, although the cell morphology of mononuclear cells was consistent with that of macrophages.
Vascular changes at the intestinal level were localized and were associated with areas displaying more abundant cellular infiltrate. Virus replication caused no apparent damage to endothelial cells; it was observed only occasionally and only after the onset of bleeding. 7 Similarly, there was no evidence of the microthrombi or fibrin deposits reported by other authors 9 , 31 as a cause of bleeding and ischemic necrosis. However, the presence of enlarged, degranulated platelets in the vicinity of mononuclear infiltrate suggested increased permeability and vessel dilation, resulting in the erythrocyte diapedesis and edema observed in this study. 7
We observed that the number of apoptotic epithelial cells was similar in infected and control animals and was thus attributable to biologic renewal of intestinal epithelium. 17
Viral infection was detected occasionally in epithelial cells from 5 pid. However, viral infection was not seen in epithelial cells located near necrotic epithelial foci observed from 11 pid onward. Because there was no correlation in intensity and distribution between viral infection and necrosis, viral infection of epithelial cells was ruled out as a possible cause of necrosis. 18–20 , 37 However, viral infection may be considered a predisposing factor. The main triggering factor for necrosis appeared to be the presence of a mononuclear infiltrate. This infiltrate consisted mainly of macrophages, displaying considerable viral infection and evidence of secretory activation. 2 , 8 , 25 These phenomena might induce necrosis of the epithelium and neighboring structures through the release of chemical mediators. 16 , 23
Footnotes
Acknowledgements
This work was supported financially by grants from DGESIC-MEC (PB98-1033) and Plan Andaluz de Investigación (AGR-0137). P. J. Sánchez-Cordón is the recipient of a fellowship from MCYT (Plan MIT). We appreciate the technical assistance of the Central Service for Support for Research, University of Córdoba, Spain.
