Abstract
A 1.5-year-old female, intact, clinically healthy cat presented for a subcutaneous mass of the ventral abdomen. surgical excision and microscopic examination of the mass were performed. Histologically, this was a discrete, unencapsulated, multilobular, expansile mass, which compressed the surrounding normal mammary tissue. Lobules were composed of tubuloacinar structures formed by atypical round to polygonal cells, which contained foamy to microvacuolated cytoplasm and variably sized, intracytoplasmic, distinct vacuoles causing nuclear peripheralization. Neoplastic cells demonstrated intense and diffuse immunoreactivity for cytokeratin and lacked immunoreactivity for vimentin. The vacuolar contents stained positively with oil RedO and negatively with periodic acid-Schiff and Alcian blue stains. Histomorphologic, histochemical, and immunohistochemial analysis support a diagnosis of lipid-rich mammary carcinoma. This is the first report of a cat with a lipid-rich variant of mammary carcinoma.
Lipid-rich mammary carcinoma, although exceedingly rare, occurs in women and has been reported in the breast of a single male. 3, 6, 8 In the veterinary literature, lipid-rich carcinoma of the mammary gland has been reported in a total of 10 dogs but has not been reported previously in cats. 1, 6 The World Health Organization (WHO) tumor classification system indicates that lipid-rich carcinomas of the canine mammary gland are extremely uncommon; however, it does not identify this tumor type as a variant of feline mammary tumors. 5 This is the first report of a lipid-rich mammary gland carcinoma in a cat.
A 1.5-year-old female, intact, calico, outdoor, farm cat presented for a single mass on the ventral abdomen, which had been present for an unknown period of time. Physical exam revealed a 2.5-cm-diameter (additional dimensions were not provided) subcutaneous mass of the left, fourth mammary gland, which was mottled white to bluish-black, lobular, moderately firm, and nonpainful on palpation. No additional masses were noted, and no lymphadenomegaly was identified. Other than the presence of a mild flea infestation, remaining physical exam findings were unremarkable. Routine blood work was within normal limits, and radiographs were not pursued.
The animal was taken to surgery for mass removal and ovariohysterectomy (OHE). The mass was submitted to the Veterinary Diagnostic Laboratory, University of Illinois, for routine histopathologic examination. Expanding the dermis, compressing surrounding mammary glands and ducts, and elevating the overlying epidermis was an unencapsulated, discrete, multilobulated mass. Lobules were separated by dense bands of fibrovascular connective tissue and composed of closely packed, variably sized (10–40 μm diameter), round to polygonal cells. These cells frequently formed tubuloacinar structures (up to four cell layers thick) of variable luminal diameter. Individual cells contained moderate to abundant, foamy, eosinophilic cytoplasm with variably sized, distinct, round, clear vacuoles, which peripherally compressed the nucleus (Fig. 1). Nuclei demonstrated marked atypia, were round to angular, variably sized, predominantly euchromatic, and basophilic with occasional prominent nucleoli. Mitotic figures were infrequent. A mild mixed inflammatory infiltrate (predominantly lymphoplasmacytic with lesser numbers of neutrophils) was present within the surrounding stroma and overlying superficial dermis. Preliminary differentials included a neoplasm of mammary gland origin or histiocytic/granulomatous inflammation.
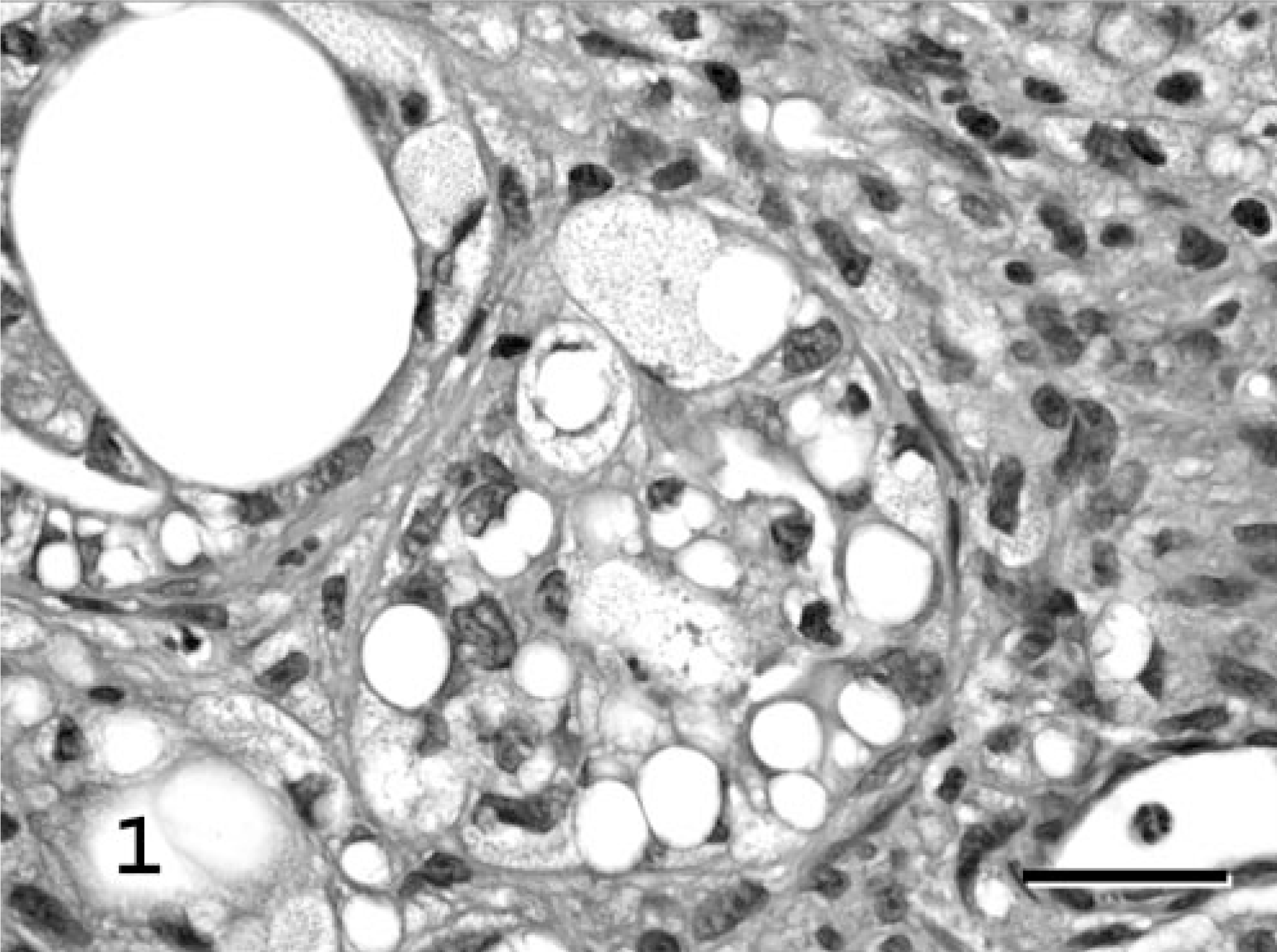
Mammary gland; cat. Lipid-rich carcinoma. Tubuloacinar structures formed by atypical round to polygonal cells with foamy to microvacuolated cytoplasm; variably sized, single to multiple, distinct intracytoplasmic vacuoles; and peripheralized, compressed, basophilic nuclei. HE. Bar = 25 μm.
Histochemical and immunohistochemical stains were used to further characterize the mass lesion and phenotype the atypical cell population. Gomori's methenamine silver (GMS), periodic acid–Shiff (PAS), and Alcian blue stains were performed on 4-μm-thick, formalin-fixed, paraffin-embedded, routinely processed tissue sections. Oil Red-O stain was performed on unprocessed, 4-μm-thick, formalin-fixed, frozen tissue. No organisms or cellular staining were identified by GMS staining. Fragmented basement membranes within the mass lesion were positive for PAS, as were random glandular cells (∼30%) within the compressed, non-neoplastic mammary tissue. The atypical cells of the mass lesion were PAS negative. Similarly, these cells were negative for Alcian blue, whereas random Alcian blue–positive cells were identified in the compressed, nonneoplastic mammary tissue and the superficial dermis. Cytoplasmic vacuoles of the neoplastic cells were negative for both PAS and Alcian blue, but strongly positive for Oil Red-O (Fig. 2, inset). This histochemical profile (negative for PAS and Alcian blue yet positive for Oil Red-O) confirmed the presence of lipid-laden cytoplasmic vacuoles within the neoplastic cells.
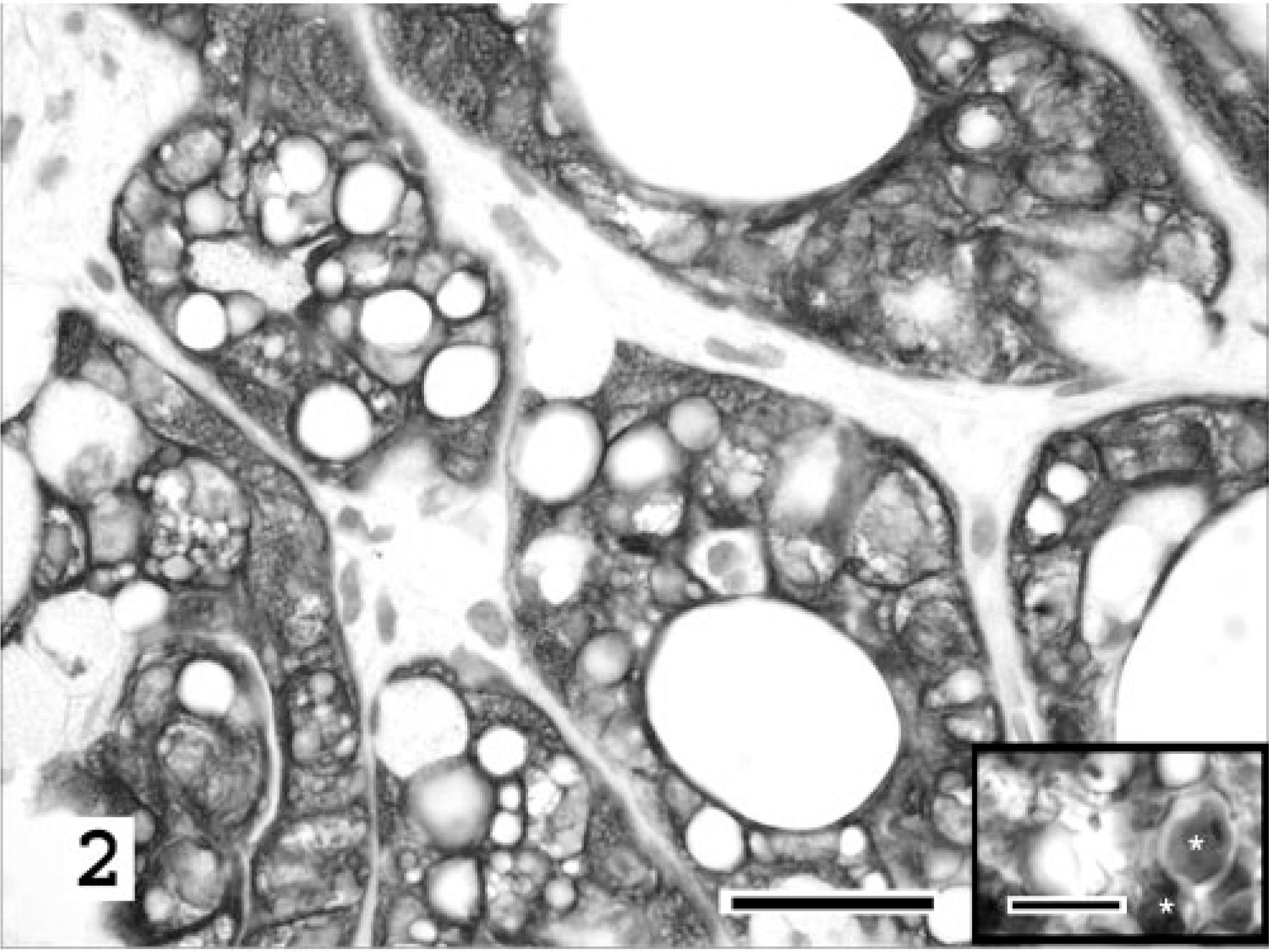
Mammary gland; cat. Lipid-rich carcinoma. Intense and diffuse cytokeratin immunoreactivity of the neoplastic cells forming tubuloacinar structures in contrast to the negative staining of the surrounding stroma. Cytokeratin AE1-AE3, basic immunoperoxidase method with hematoxylin counterstain. Bar = 25 μm. Inset: Oil Red-O–positive staining of intracytoplasmic vacuolar contents (∗) confirms the presence of neutral lipid. Oil Red-O stain. Bar = 10 μm.
Immunohistochemical stains, for cytokeratin (AE1-AE3, BioGenex San Ramon, CA), vimentin (clone V9, Bio-Genex), and lysozyme (Rb polyclonal, DAKO Corporation, Carpinteria, CA), were performed on formalin-fixed, paraffin wax–embedded, 4-μm-thick tissue sections, using steam antigen retrieval in citrate buffer (pH 6.0) and a basic immunoperoxidase method. The tissues were treated with the respective antibody at a prediluted concentration for 30 minutes (15 minutes for vimentin) followed by avidin–biotin–horseradish peroxidase complex with diaminobenzadine substrate. The atypical lipid-laden cells forming tubuloacinar structures had strong and diffuse cytoplasmic cytokeratin staining (Fig. 2). A subset of these cells (<5%) also weakly coexpressed vimentin. The supporting stroma was vimentin positive. The normal, compressed mammary tissue had appropriate staining of epithelial glandular structures with cytokeratin and supporting stroma with vimentin. Lysozyme-positive cells were scattered randomly throughout the mass-associated stroma. These were interpreted as nonneoplastic infiltrating macrophages.
Lipid-rich carcinoma of the mammary gland is defined as a variant of mammary carcinoma, characterized by cells with abundant vacuolated cytoplasm that contain a large amount of neutral lipid. 5 In humans, lipid-rich carcinomas are considered to be a variant of infiltrating ductal carcinomas of the breast, which are extremely rare. 7 Pena et al. described three cases of canine lipid-rich mammary carcinoma as having a “tubular disposition” of the lipid-rich carcinoma cells. 6 The tubuloacinar pattern of the neoplastic cells in this case, combined with the histochemical and immunohistochemical profile, support that lipid-rich mammary carcinomas in cats may also be of ductal origin. In contrast, a recent study by Espinosa De Los Monteros et al. examined seven lipid-rich mammary carcinomas from seven dogs. 1 On the basis of their panel of cytokeratin markers (cytokeratins 5 and 8 [RCK-102 Ab] and cytokeratins 8 and 18 [NCL-5D3 Ab]), it was determined that the majority of neoplastic cells were of glandular epithelial origin.
Six months after complete surgical excision of the mass and OHE, the cat presented in this report is clinically healthy with no signs of local recurrence or metastasis. The presence of a mammary carcinoma in a 1.5-year-old cat is of interest because the mean age of occurrence of mammary carcinoma in the cat is 10–12 years of age. 2 However, the range is reported to be 9 months to 23 years. 2 The diagnosis in this study was based on the marked cellular and nuclear atypia of the neoplastic population and the similarities of this mass, both grossly and histologically, to those reported in humans and dogs. The typical clinical presentation in the dog is a single lobulated mass affecting one mammary gland with potential extension to a second, adjacent gland. Microscopic findings in both humans and dogs are consistent with those described in this study including the abundant cytoplasmic lipid-filled vacuoles, peripheralized nuclei, and rare mitotic figures. 1, 3, 7, 8 Espinosa De Los Monteros et al. reported ∼71% of canine cases had evidence of metastasis. No metastatic lesions have been identified in this case of feline lipid-rich mammary carcinoma to date.
The findings presented in this article suggest that lipid-rich mammary carcinoma should be considered as a variant of mammary carcinoma in the cat.
